Professional Documents
Culture Documents
Atomic Theory
Uploaded by
Cardoza RyanCopyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
Atomic Theory
Uploaded by
Cardoza RyanCopyright:
Available Formats
ATOMIC THEORY
ATOMIC MODELS
What is an atom? What are atoms made of?
Atoms are the basic building blocks of ordinary matter. Atoms can join together to form
molecules, which in turn form most of the objects around you.
Atoms are composed of particles called protons, electrons and neutrons.
Protons carry a positive electrical charge,
electrons carry a negative electrical charge
neutrons carry no electrical charge at all.
The protons and neutrons cluster together in the central part of the atom, called the nucleus, and the
electrons 'orbit' the nucleus.
PROTONS and NEUTRONS are both composed of other particles called quarks and gluons.
Protons contain two 'up' quarks and one 'down' quark
Neutrons contain one 'up' quark and two 'down' quarks. The gluons are responsible for
binding the quarks to one another.
Early Greek Theories
400 B.C. - Democritus thought matter could not be divided indefinitely.
This led to the idea of atoms in a void.
350 B.C - Aristotle modified an earlier theory that matter was made of four elements: earth,
fire, water, air.
Aristotle was wrong. However, his theory persisted for 2000 years.
Daltons billiard ball model
Adding Electrons to the Model
Materials, when rubbed, can develop a charge difference. This electricity is called cathode rays
when passed through an evacuated tube (demos).
These rays have a small mass and are negative.
Thompson noted that these negative subatomic particles were a fundamental part of all atoms.
1) Daltons Billiard ball model (1800-1900)
Atoms are solid and indivisible.
2) Thomson Plum pudding model (1900)
3) Negative electrons in a positive frameworkThe Rutherford model (around 1910)
Atoms are mostly empty space.
Negative electrons orbit a positive nucleus.
Plum Pudding or Raisin bun model of J.J. Thomson
Ernest Rutherford (movie: 10 min.)
Rutherford shot alpha () particles at gold foil.
Most particles passed through. So, atoms are mostly empty.
Some positive -particles deflected or bounced back!
Thus, a nucleus is positive & holds most of an atoms mass.
Ernest Rutherfords Planetary or Nuclear model
Bohrs model
Electrons orbit the nucleus in shells
Electrons can be bumped up to a higher shell if hit by an electron or a photon of light.
There are 2 types of spectra: continuous spectra & line spectra. Its when electrons fall back
down that they release a photon. These jumps down from shell to shell account for the
line spectra seen in gas discharge tubes (through spectroscopes).
Niels Bohr model/s of the atom
Erwin Schrodinger Electron cloud model of the atom
Atomic numbers, Mass numbers
There are 3 types of subatomic particles. We already know about electrons (e) & protons
(p+). Neutrons (n0) were also shown to exist (1930s).
They have: no charge, a mass similar to protons
Elements are often symbolized with their mass number and atomic number
These values are given on the periodic table.
For now, round the mass # to a whole number.
These numbers tell you a lot about atoms.
# of protons = # of electrons = atomic number
# of neutrons = mass number atomic number
Calculate # of e, n0, p+ for Ca, Ar, and Br.
REMEMBER:
For any element:
Number of Protons = Atomic Number
Number of Electrons = Number of Protons = Atomic Number
Number of Neutrons = Mass Number - Atomic Number
The Atoms Family - Atomic Math Challenge
Atomic number
Symbol
Name
Atomic mass
protons electrons
Atomic number equals the number of ____________ or ________________.
Atomic mass equals the number of ______________ + _______________.
neutrons
You might also like
- The Yellow House: A Memoir (2019 National Book Award Winner)From EverandThe Yellow House: A Memoir (2019 National Book Award Winner)Rating: 4 out of 5 stars4/5 (98)
- Hidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceFrom EverandHidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceRating: 4 out of 5 stars4/5 (895)
- The Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeFrom EverandThe Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeRating: 4 out of 5 stars4/5 (5794)
- The Little Book of Hygge: Danish Secrets to Happy LivingFrom EverandThe Little Book of Hygge: Danish Secrets to Happy LivingRating: 3.5 out of 5 stars3.5/5 (400)
- Devil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaFrom EverandDevil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaRating: 4.5 out of 5 stars4.5/5 (266)
- Shoe Dog: A Memoir by the Creator of NikeFrom EverandShoe Dog: A Memoir by the Creator of NikeRating: 4.5 out of 5 stars4.5/5 (537)
- Elon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureFrom EverandElon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureRating: 4.5 out of 5 stars4.5/5 (474)
- Never Split the Difference: Negotiating As If Your Life Depended On ItFrom EverandNever Split the Difference: Negotiating As If Your Life Depended On ItRating: 4.5 out of 5 stars4.5/5 (838)
- Grit: The Power of Passion and PerseveranceFrom EverandGrit: The Power of Passion and PerseveranceRating: 4 out of 5 stars4/5 (588)
- A Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryFrom EverandA Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryRating: 3.5 out of 5 stars3.5/5 (231)
- The Emperor of All Maladies: A Biography of CancerFrom EverandThe Emperor of All Maladies: A Biography of CancerRating: 4.5 out of 5 stars4.5/5 (271)
- The World Is Flat 3.0: A Brief History of the Twenty-first CenturyFrom EverandThe World Is Flat 3.0: A Brief History of the Twenty-first CenturyRating: 3.5 out of 5 stars3.5/5 (2259)
- On Fire: The (Burning) Case for a Green New DealFrom EverandOn Fire: The (Burning) Case for a Green New DealRating: 4 out of 5 stars4/5 (73)
- The Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersFrom EverandThe Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersRating: 4.5 out of 5 stars4.5/5 (344)
- Team of Rivals: The Political Genius of Abraham LincolnFrom EverandTeam of Rivals: The Political Genius of Abraham LincolnRating: 4.5 out of 5 stars4.5/5 (234)
- The Unwinding: An Inner History of the New AmericaFrom EverandThe Unwinding: An Inner History of the New AmericaRating: 4 out of 5 stars4/5 (45)
- The Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreFrom EverandThe Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreRating: 4 out of 5 stars4/5 (1090)
- The Sympathizer: A Novel (Pulitzer Prize for Fiction)From EverandThe Sympathizer: A Novel (Pulitzer Prize for Fiction)Rating: 4.5 out of 5 stars4.5/5 (121)
- Her Body and Other Parties: StoriesFrom EverandHer Body and Other Parties: StoriesRating: 4 out of 5 stars4/5 (821)
- Particle Molecule AtomDocument3 pagesParticle Molecule AtomGermaeGonzalesNo ratings yet
- Naval Reactor Handbook Vol 1Document1,562 pagesNaval Reactor Handbook Vol 1Peter AngeloNo ratings yet
- GM Counting System Experiments PDFDocument58 pagesGM Counting System Experiments PDFbalakrishnanit100% (1)
- Chapter Four (Nuclear Radiation)Document10 pagesChapter Four (Nuclear Radiation)White HeartNo ratings yet
- Atomic ModelsDocument25 pagesAtomic ModelsAubry DurangparangNo ratings yet
- BibliografiaDocument2 pagesBibliografiaVeriato BalõesNo ratings yet
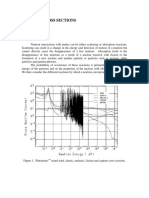
- Ragheb-Neutron Cross Sections-2011 PDFDocument26 pagesRagheb-Neutron Cross Sections-2011 PDFsujayan2005No ratings yet
- C H A P T E R 2 5 - Electric Potential: ProblemsDocument2 pagesC H A P T E R 2 5 - Electric Potential: ProblemsPAULINA ARIASNo ratings yet
- FCAT Questions: Atoms, Elements, & Molecules Module Grades 9-12 Teacher VersionDocument4 pagesFCAT Questions: Atoms, Elements, & Molecules Module Grades 9-12 Teacher VersionMae Uayan Prisco-TagaraoNo ratings yet
- Ge BWR4Document119 pagesGe BWR4rs_sechiNo ratings yet
- Annotated Bib MidtermDocument2 pagesAnnotated Bib Midtermapi-494398715No ratings yet
- Chapter 4 Test The Periodic Table of ElementsDocument6 pagesChapter 4 Test The Periodic Table of ElementsNeeraj MeenaNo ratings yet
- Strange Heart of Neutron StarDocument3 pagesStrange Heart of Neutron StarMani VannanNo ratings yet
- Radioactive Waste ManagementDocument20 pagesRadioactive Waste ManagementArka DattaNo ratings yet
- Counts Per Minute: Count RatesDocument3 pagesCounts Per Minute: Count RatesLee Kwon ZeeNo ratings yet
- THE BIGGER PICTURE. Beyond The Illusion of Matter. Johan OldenkampDocument43 pagesTHE BIGGER PICTURE. Beyond The Illusion of Matter. Johan OldenkampStephen J. RobertsNo ratings yet
- Yr 9 Physics Revision 3Document4 pagesYr 9 Physics Revision 3KitanNo ratings yet
- 32 ConcepTests ClickersDocument53 pages32 ConcepTests ClickersJasdeep SinghNo ratings yet
- 08 - Chapter 2 PDFDocument44 pages08 - Chapter 2 PDFthegubesNo ratings yet
- Insag 7Document1 pageInsag 7Vittorio BeltottiNo ratings yet
- Nuclear (AS Physics) Compiled By: Sir SumairDocument4 pagesNuclear (AS Physics) Compiled By: Sir SumairBerry101No ratings yet
- Nuclear Power Plant Engineering - BUETDocument79 pagesNuclear Power Plant Engineering - BUET0410058100% (6)
- Timeline: (Hide) 1 2 3 4 o 4.1 o 4.2 o 4.3 o 4.4 o 4.5 5 6 7 8 9 10Document7 pagesTimeline: (Hide) 1 2 3 4 o 4.1 o 4.2 o 4.3 o 4.4 o 4.5 5 6 7 8 9 10mimiyuhNo ratings yet
- Selected Reference Material United StateDocument1,103 pagesSelected Reference Material United StatehaibhNo ratings yet
- G 8 Third Revision......Document67 pagesG 8 Third Revision......Christina AdelNo ratings yet
- Power Plant EquipmentDocument54 pagesPower Plant EquipmentJoy PalitNo ratings yet
- Analytic Expressions For Alpha Decay Half-Lives and Potential Barriers-RoyerDocument13 pagesAnalytic Expressions For Alpha Decay Half-Lives and Potential Barriers-RoyerJhoan PerezNo ratings yet
- Decay: What Is Meant by A Radioactive Substance? (1mk)Document12 pagesDecay: What Is Meant by A Radioactive Substance? (1mk)geofry lutondoNo ratings yet
- Iaea Position PaperDocument2 pagesIaea Position Paperapi-278012231No ratings yet
- B61 Nuclear BombDocument2 pagesB61 Nuclear Bombbabibu12345No ratings yet