Professional Documents
Culture Documents
Colligative Property Are Properties That Depends On The Number of Solute Particles Present and Not On The Identity of The Solute Particles
Uploaded by
Yssa SayconOriginal Description:
Original Title
Copyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
Colligative Property Are Properties That Depends On The Number of Solute Particles Present and Not On The Identity of The Solute Particles
Uploaded by
Yssa SayconCopyright:
Available Formats
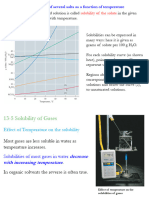
Key Points
Colligative Property are properties that depends on the number of solute particles present and
not on the identity of the solute particles.
Vapor pressure is a colligative property, so the vapor pressure of solutions is directly proportional to the
amount of solute present in a solution.
When a solute is present in a solvent, the vapor pressure is lowered because fewer solvent molecules
are present at the top of the solution.
Raoult’s law details the calculations for acquiring the vapor pressure of an ideal solution.
Key Terms
partial pressure: The pressure that one component of a mixture of gases contributes to the total
pressure.
ideal solution: A solution with thermodynamic properties analogous to those of a mixture of ideal gases.
electrolyte: A substance that, when in solution or when molten, ionizes and conducts electricity.
Electrolyte Solutions
A simple example of an electrolyte solution is sodium chloride in water. In the presence of water, solid
sodium chloride dissociates as it is dissolved, forming an electrolyte solution:
NaCl(s)→Na+(aq)+Cl−(aq)
Vapor Pressure
Vapor pressure is the pressure exerted by a vapor in
equilibrium with its condensed phase, either liquid
or solid, at a particular temperature. Basically, it is a
measure of how much the solvent molecules tend to
escape from a liquid or solid phase into the
atmosphere. Vapor pressure of a liquid is a
colligative property.
Vapor pressure: The picture shows the transition, as
a result of the vapor pressure, from particles in liquid
phase to gas phase and vice-versa.
To better visualize the effect of solute on the vapor
pressure of a solution, consider a pure solvent. This
pure solvent has a certain vapor pressure associated with it. Subjected to temperatures below the
solvent’s boiling point, the molecules going into the gaseous phase are mostly situated on the top layer
of the solution. Now consider a solution composed of both solvent and solute. Some solute molecules
will occupy space near the surface of the liquid, decreasing the number of solvent molecules that can be
there. Therefore, fewer molecules are changing from the liquid phase into the gas phase, and the vapor
pressure of the solvent decreases.
In an electrolyte solution, the number of dissolved particles is larger because the solute breaks apart
into ions. The greater the number of ions, the larger the impact on colligative properties will be.
What are the properties of electrolytes?
A substance that dissociates into ions in solution acquires the capacity to conduct electricity. Sodium,
potassium, chloride, calcium, magnesium, and phosphate are examples of electrolytes
You might also like
- MEIL 122 1 20 1st SEMESTERDocument129 pagesMEIL 122 1 20 1st SEMESTERMhikaella Salangsang90% (10)
- Curry Leaves Extraction MTech ThesisDocument82 pagesCurry Leaves Extraction MTech ThesisMoolam Rao50% (2)
- Modem Aspects of Emulsion ScienceDocument627 pagesModem Aspects of Emulsion ScienceJerzy Sek100% (1)
- Specific Heat Lab ReportDocument29 pagesSpecific Heat Lab ReportWeebly LoLNo ratings yet
- 1981 Book FoodProcessEngineering PDFDocument441 pages1981 Book FoodProcessEngineering PDFSabrina Fernandes100% (1)
- Chem LeeDocument3 pagesChem LeeLee Alvin MagyayaNo ratings yet
- Colligative Properties of Nonelectrolyte Solutions - Boundless ChemistryDocument15 pagesColligative Properties of Nonelectrolyte Solutions - Boundless Chemistryprashant_cool_4_uNo ratings yet
- 1 Lecture Note Chem 2 ColligativeDocument2 pages1 Lecture Note Chem 2 ColligativeMarjorie BrondoNo ratings yet
- Raoult's LawDocument8 pagesRaoult's LawHARSH DHOLAKIYANo ratings yet
- Liquid Solutions PDFDocument50 pagesLiquid Solutions PDFAniruddha KawadeNo ratings yet
- Boiling Point ElevationDocument2 pagesBoiling Point ElevationHannah VisitacionNo ratings yet
- Colligative Properties of SolutionsDocument8 pagesColligative Properties of SolutionspieNo ratings yet
- Hukum RoultDocument25 pagesHukum RoultZaqiya zahwa alifaNo ratings yet
- Solutions G Colligative PropDocument14 pagesSolutions G Colligative PropHusnul YaqinNo ratings yet
- Chemistry Q2Document3 pagesChemistry Q2Kristel Anne Magaru-Macauggal-LugoNo ratings yet
- Biochemistry Lab Hand OutsDocument12 pagesBiochemistry Lab Hand Outsrica sebabillonesNo ratings yet
- Colligative Properties of SolutionsDocument5 pagesColligative Properties of SolutionsrizzmuizzNo ratings yet
- Chemistry Picture Vocabulary - SolutionsDocument28 pagesChemistry Picture Vocabulary - Solutionsapi-254514513No ratings yet
- Raoult's Law123Document25 pagesRaoult's Law123Abdur RehmanNo ratings yet
- HANDOUT Separation PhasesDocument14 pagesHANDOUT Separation PhasesDrumz StaffNo ratings yet
- Solution of Non-ElectrolytesDocument133 pagesSolution of Non-Electrolytesneha_dand1591No ratings yet
- Physiochemical and Colligative Properties of SolutionsDocument10 pagesPhysiochemical and Colligative Properties of SolutionsAhmad ButtNo ratings yet
- SPT 120 Non ElectrolytesDocument36 pagesSPT 120 Non Electrolytestafadzwachikuni21septemberNo ratings yet
- SolutionsDocument4 pagesSolutionsOvijit KarmokarNo ratings yet
- GC2Document12 pagesGC2venuskcalzadoNo ratings yet
- Colligative Properties of SolutionsDocument7 pagesColligative Properties of SolutionsYSAIDEN123No ratings yet
- Types of Solutions & Expressing Concentration of SolutionsDocument13 pagesTypes of Solutions & Expressing Concentration of SolutionsTr Mazhar PunjabiNo ratings yet
- Theory of SolutionDocument9 pagesTheory of SolutionrajuNo ratings yet
- Colligative Properties - WikipediaDocument22 pagesColligative Properties - WikipediaManojkumarNo ratings yet
- Colligative Properties of SolutionsDocument2 pagesColligative Properties of SolutionsJeremy BunquinNo ratings yet
- Chemistry: TopicDocument10 pagesChemistry: TopicMuzammil ShabirNo ratings yet
- Gen Chem Rev Pre Fi (2nd Sem)Document6 pagesGen Chem Rev Pre Fi (2nd Sem)Caeyla NhinaNo ratings yet
- New Microsoft Word DocumentDocument18 pagesNew Microsoft Word DocumentinatuxedoNo ratings yet
- GC2 PrelimDocument158 pagesGC2 Prelimkmarissee100% (1)
- ColligativeDocument24 pagesColligativeHarry WinstonNo ratings yet
- 4 Colligativeproperties 180825211533Document88 pages4 Colligativeproperties 180825211533Rmon ianNo ratings yet
- Liquid SolutionDocument64 pagesLiquid Solutiontapas kunduNo ratings yet
- Special CaseDocument1 pageSpecial CaseHarshit SrivastavaNo ratings yet
- Lesson 7 Colligative Properties of Nonelectrolytes and ElectrolyteDocument27 pagesLesson 7 Colligative Properties of Nonelectrolytes and ElectrolyteLyndy PantaoNo ratings yet
- CHM 151 Lecture Note - 2023-2024Document15 pagesCHM 151 Lecture Note - 2023-2024jimisolaoluwayinkaNo ratings yet
- Lec. 4, AeronauticalDocument8 pagesLec. 4, AeronauticalRifat UddinNo ratings yet
- 1.9 Colligative Properties of SolutionsDocument9 pages1.9 Colligative Properties of Solutionsfikerdereje697No ratings yet
- Higher Vapor Pressure Lower Vapor PressureDocument10 pagesHigher Vapor Pressure Lower Vapor PressureCatalina PerryNo ratings yet
- Ideal and Real SolutionsDocument57 pagesIdeal and Real SolutionsAhammed Hussain MadhaniNo ratings yet
- Colligative Properties: Vapour Pressure Boiling Point Freezing Point Osmotic PressureDocument27 pagesColligative Properties: Vapour Pressure Boiling Point Freezing Point Osmotic PressureRSNo ratings yet
- Colligative PropertiesDocument13 pagesColligative PropertiesretorbarjosephinemercolitaNo ratings yet
- Colligative Properties of Solution (3rd Week)Document41 pagesColligative Properties of Solution (3rd Week)SomeThingNo ratings yet
- ComsDocument8 pagesComsJoseph EstellosoNo ratings yet
- Colligative PropertiesDocument8 pagesColligative PropertiesValerie Ann Francisco100% (1)
- Colligative Properties of Solutions: Abdallah AskarDocument9 pagesColligative Properties of Solutions: Abdallah AskarAbdallah AskarNo ratings yet
- Colligative Properties of SolutionsDocument6 pagesColligative Properties of SolutionsUmbrellaNo ratings yet
- Chapter 13 Lecture-2Document10 pagesChapter 13 Lecture-2Çetin UzişNo ratings yet
- SH Hetler in PakDocument14 pagesSH Hetler in Pakpatiser mNo ratings yet
- Type of SolutionsDocument8 pagesType of SolutionsbharathNo ratings yet
- Arindam Das Bs-15 253 B.SC Chemistry Honors Supervised By:-Dr. S MuniDocument25 pagesArindam Das Bs-15 253 B.SC Chemistry Honors Supervised By:-Dr. S MuniArindam DasNo ratings yet
- CHM 203 by AdasDocument7 pagesCHM 203 by Adaslilianmatt81No ratings yet
- Liquid State FosDocument32 pagesLiquid State FosSrirama LakshmiNo ratings yet
- Asc0304 Chemistry I: Phase EquilibriumDocument27 pagesAsc0304 Chemistry I: Phase EquilibriumhadassahhadidNo ratings yet
- Class Discussion: Colligative Properties: SolutionDocument2 pagesClass Discussion: Colligative Properties: SolutionARRIANE CYREL CAMACHONo ratings yet
- Colligative Properties of SolutionsDocument66 pagesColligative Properties of Solutionspalitpa moreNo ratings yet
- UntitledDocument72 pagesUntitledRemelyn Marie RegaladoNo ratings yet
- Liquid Solution (13th)Document19 pagesLiquid Solution (13th)Raju SinghNo ratings yet
- Colligative PropertiesDocument6 pagesColligative PropertiesMILLENNIUM CREATIVE STUDIONo ratings yet
- Working Guide to Vapor-Liquid Phase Equilibria CalculationsFrom EverandWorking Guide to Vapor-Liquid Phase Equilibria CalculationsRating: 5 out of 5 stars5/5 (1)
- Phyc 121Document38 pagesPhyc 121Yssa SayconNo ratings yet
- RSCH 121Document2 pagesRSCH 121Yssa SayconNo ratings yet
- RSCH 1Document72 pagesRSCH 1Yssa SayconNo ratings yet
- Physical Education and Health 3 Nature of The Different DancesDocument84 pagesPhysical Education and Health 3 Nature of The Different DancesYssa SayconNo ratings yet
- Meil 1Document59 pagesMeil 1Yssa SayconNo ratings yet
- Outlines of A New System of Thermodynamic Chemistry G. N. Lewis 1907Document36 pagesOutlines of A New System of Thermodynamic Chemistry G. N. Lewis 1907Victor VazquezNo ratings yet
- 1 Identification of Chemical SystemsDocument5 pages1 Identification of Chemical SystemsCris CorsinoNo ratings yet
- 9 Crystals: Growth, Properties, and ApplicationsDocument150 pages9 Crystals: Growth, Properties, and ApplicationscosmowmarchalNo ratings yet
- Thermodynamics 1 - LN-Topic 2 (Pure Substances)Document8 pagesThermodynamics 1 - LN-Topic 2 (Pure Substances)Mondaya, Jake Armond D.No ratings yet
- Mass Transfer 1 CLB 20804Document54 pagesMass Transfer 1 CLB 20804muhammad fazmiNo ratings yet
- Adv Funct Materials - 2023 - Zhang - Water Adsorption in MOFs Structures and ApplicationsDocument32 pagesAdv Funct Materials - 2023 - Zhang - Water Adsorption in MOFs Structures and ApplicationsnemaderakeshNo ratings yet
- Alternative Extractive Distillation System For CO2-Ethane Azeotrope Separation in Enhanced Oil Recovery Processes - Ebrahimzadeh2016Document9 pagesAlternative Extractive Distillation System For CO2-Ethane Azeotrope Separation in Enhanced Oil Recovery Processes - Ebrahimzadeh2016Hesam AhmadianNo ratings yet
- Aspenhysysoliif2006 RefDocument232 pagesAspenhysysoliif2006 RefKrishanu SahaNo ratings yet
- HPLC TroubleshootingDocument27 pagesHPLC TroubleshootingwingroupsNo ratings yet
- Powder Processed Aluminium AlloysDocument10 pagesPowder Processed Aluminium AlloysFatih BozkurtNo ratings yet
- Chemical Process Design and Optimization: Chapter 3a - Separation Train SynthesisDocument26 pagesChemical Process Design and Optimization: Chapter 3a - Separation Train SynthesisLam DesmondNo ratings yet
- Basic Thermodynamics: Module 01: Fundamental Concepts and Definitions 1.1 DefinitionDocument22 pagesBasic Thermodynamics: Module 01: Fundamental Concepts and Definitions 1.1 DefinitionVinayaka G PNo ratings yet
- Slurry Airlift CFDDocument9 pagesSlurry Airlift CFDlrodriguez_892566No ratings yet
- Mechanical Engineering PapersDocument34 pagesMechanical Engineering PapersshivarajeshNo ratings yet
- 1 s2.0 S0360319916001695 MainDocument7 pages1 s2.0 S0360319916001695 Mainabhishek992198No ratings yet
- Chemistry Made Easy An Illustrated Study Guide For Students To EasilyDocument220 pagesChemistry Made Easy An Illustrated Study Guide For Students To EasilyAnna Vyluschak100% (2)
- PT FLASH CALCULATION (Using Peng Robinson EOS) : Chemical Engineer's GuideDocument88 pagesPT FLASH CALCULATION (Using Peng Robinson EOS) : Chemical Engineer's GuideDaniel Marcelo Velasquez100% (1)
- Engineering Thermodynamics 1 THRM1001: Basic Concepts and DefinitionsDocument50 pagesEngineering Thermodynamics 1 THRM1001: Basic Concepts and DefinitionsTashi BestNo ratings yet
- DDDDDDDocument2 pagesDDDDDDapi-254428474No ratings yet
- Cre Lab ManualDocument190 pagesCre Lab Manualmanishtiwari877No ratings yet
- ChE 124 SyllabusDocument5 pagesChE 124 SyllabussexyrustyNo ratings yet
- Proper Regeneration of Molecular Sieves in TSA Processes-Part 1Document7 pagesProper Regeneration of Molecular Sieves in TSA Processes-Part 1G P100% (1)
- Mathematical Modeling of Melting and Freezing Processes PDFDocument34 pagesMathematical Modeling of Melting and Freezing Processes PDFAjit KambleNo ratings yet
- HW 9 2011 SolutionsDocument15 pagesHW 9 2011 SolutionsParamesh Anugu100% (1)
- Chapter 12 Thermodynamics PDFDocument94 pagesChapter 12 Thermodynamics PDFNitish MehraNo ratings yet
- Basic Concept ThermodynamicsDocument62 pagesBasic Concept Thermodynamicscjdbbt1No ratings yet