Professional Documents
Culture Documents
Uncertainty of Electron
Uploaded by
হাসিন মুসাইয়্যাব আহমাদ পুণ্য0 ratings0% found this document useful (0 votes)
7 views1 pageOriginal Title
Uncertainty of electron.docx
Copyright
© © All Rights Reserved
Available Formats
DOCX, PDF, TXT or read online from Scribd
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
© All Rights Reserved
Available Formats
Download as DOCX, PDF, TXT or read online from Scribd
0 ratings0% found this document useful (0 votes)
7 views1 pageUncertainty of Electron
Uploaded by
হাসিন মুসাইয়্যাব আহমাদ পুণ্যCopyright:
© All Rights Reserved
Available Formats
Download as DOCX, PDF, TXT or read online from Scribd
You are on page 1of 1
Uncertainty of electron : Assume that we want to measure the
position of the electron. the position can be measured by
shorter wavelength. because shorter wavelength means high
frequency.as electron is very small so we need much energy to
see it. Much energy is made by shorter wavelength..But think,if
a high energy photon is conflicted with the electron,it’s velocity
should be changed. so momentum is going to be uncertain.
now assume we want to measure the momentum.so we have
to receive that energy’s photon which can’t change the
momentum of electron significantly.so low energy means large
wavelength and low energy is not applicable to see the photon
in a position.so position is uncertain . .. . .
You might also like
- The Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeFrom EverandThe Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeRating: 4 out of 5 stars4/5 (5794)
- The Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreFrom EverandThe Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreRating: 4 out of 5 stars4/5 (1090)
- Never Split the Difference: Negotiating As If Your Life Depended On ItFrom EverandNever Split the Difference: Negotiating As If Your Life Depended On ItRating: 4.5 out of 5 stars4.5/5 (838)
- Hidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceFrom EverandHidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceRating: 4 out of 5 stars4/5 (895)
- Grit: The Power of Passion and PerseveranceFrom EverandGrit: The Power of Passion and PerseveranceRating: 4 out of 5 stars4/5 (588)
- Shoe Dog: A Memoir by the Creator of NikeFrom EverandShoe Dog: A Memoir by the Creator of NikeRating: 4.5 out of 5 stars4.5/5 (537)
- The Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersFrom EverandThe Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersRating: 4.5 out of 5 stars4.5/5 (344)
- Elon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureFrom EverandElon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureRating: 4.5 out of 5 stars4.5/5 (474)
- Her Body and Other Parties: StoriesFrom EverandHer Body and Other Parties: StoriesRating: 4 out of 5 stars4/5 (821)
- The Sympathizer: A Novel (Pulitzer Prize for Fiction)From EverandThe Sympathizer: A Novel (Pulitzer Prize for Fiction)Rating: 4.5 out of 5 stars4.5/5 (120)
- The Emperor of All Maladies: A Biography of CancerFrom EverandThe Emperor of All Maladies: A Biography of CancerRating: 4.5 out of 5 stars4.5/5 (271)
- The Little Book of Hygge: Danish Secrets to Happy LivingFrom EverandThe Little Book of Hygge: Danish Secrets to Happy LivingRating: 3.5 out of 5 stars3.5/5 (399)
- The World Is Flat 3.0: A Brief History of the Twenty-first CenturyFrom EverandThe World Is Flat 3.0: A Brief History of the Twenty-first CenturyRating: 3.5 out of 5 stars3.5/5 (2259)
- The Yellow House: A Memoir (2019 National Book Award Winner)From EverandThe Yellow House: A Memoir (2019 National Book Award Winner)Rating: 4 out of 5 stars4/5 (98)
- Devil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaFrom EverandDevil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaRating: 4.5 out of 5 stars4.5/5 (266)
- A Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryFrom EverandA Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryRating: 3.5 out of 5 stars3.5/5 (231)
- Team of Rivals: The Political Genius of Abraham LincolnFrom EverandTeam of Rivals: The Political Genius of Abraham LincolnRating: 4.5 out of 5 stars4.5/5 (234)
- On Fire: The (Burning) Case for a Green New DealFrom EverandOn Fire: The (Burning) Case for a Green New DealRating: 4 out of 5 stars4/5 (73)
- The Unwinding: An Inner History of the New AmericaFrom EverandThe Unwinding: An Inner History of the New AmericaRating: 4 out of 5 stars4/5 (45)
- Brac Bank ACCountDocument1 pageBrac Bank ACCountহাসিন মুসাইয়্যাব আহমাদ পুণ্যNo ratings yet
- Annexure 1Document3 pagesAnnexure 1হাসিন মুসাইয়্যাব আহমাদ পুণ্যNo ratings yet
- E-GP Registration and Tendering Process enDocument3 pagesE-GP Registration and Tendering Process enহাসিন মুসাইয়্যাব আহমাদ পুণ্যNo ratings yet
- Circular 16172Document2 pagesCircular 16172হাসিন মুসাইয়্যাব আহমাদ পুণ্যNo ratings yet
- 2019 04 11 17 21 44 PPA 2006, PPR 2008 With All Amendments Up To January, 2019 PDFDocument274 pages2019 04 11 17 21 44 PPA 2006, PPR 2008 With All Amendments Up To January, 2019 PDFMd Shamim HasanNo ratings yet
- Annexure 1Document4 pagesAnnexure 1হাসিন মুসাইয়্যাব আহমাদ পুণ্যNo ratings yet
- Ar (t"Lc'4: Qr3reDocument3 pagesAr (t"Lc'4: Qr3reহাসিন মুসাইয়্যাব আহমাদ পুণ্যNo ratings yet
- Constructs Ex-OR Gate Using Basic Gates and Verify Its/ Constructs Ex-NOR Gate Using Basic Gates and Verify Its.Document4 pagesConstructs Ex-OR Gate Using Basic Gates and Verify Its/ Constructs Ex-NOR Gate Using Basic Gates and Verify Its.হাসিন মুসাইয়্যাব আহমাদ পুণ্যNo ratings yet
- Tft0'lb (Ql-Cn. - : FrffiDocument1 pageTft0'lb (Ql-Cn. - : Frffiহাসিন মুসাইয়্যাব আহমাদ পুণ্যNo ratings yet
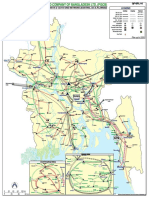
- Transmission Network Map MainDocument1 pageTransmission Network Map Mainহাসিন মুসাইয়্যাব আহমাদ পুণ্যNo ratings yet
- Construction of All Basic Gates Using Universal (NAND/NOR) Gates and Verify Its.Document4 pagesConstruction of All Basic Gates Using Universal (NAND/NOR) Gates and Verify Its.হাসিন মুসাইয়্যাব আহমাদ পুণ্যNo ratings yet
- Verify The Truth Table For Basic Gates and Others Logic Gates.Document2 pagesVerify The Truth Table For Basic Gates and Others Logic Gates.হাসিন মুসাইয়্যাব আহমাদ পুণ্যNo ratings yet
- Construction of All Basic Gates Using Universal (NAND/NOR) Gates and Verify Its.Document4 pagesConstruction of All Basic Gates Using Universal (NAND/NOR) Gates and Verify Its.হাসিন মুসাইয়্যাব আহমাদ পুণ্যNo ratings yet
- Chemistry Class9 Part1Document5 pagesChemistry Class9 Part1হাসিন মুসাইয়্যাব আহমাদ পুণ্যNo ratings yet
- Higher K Value or Lower PK Valu Indicates Strong AcidDocument3 pagesHigher K Value or Lower PK Valu Indicates Strong Acidহাসিন মুসাইয়্যাব আহমাদ পুণ্যNo ratings yet
- Generic Wind by PSSEDocument31 pagesGeneric Wind by PSSEহাসিন মুসাইয়্যাব আহমাদ পুণ্যNo ratings yet
- Equation Editor The Word 2007/2010 Equation Editor Shortcut CommandsDocument11 pagesEquation Editor The Word 2007/2010 Equation Editor Shortcut CommandspanchopanzaNo ratings yet
- Load Flow Study in BuetDocument4 pagesLoad Flow Study in Buetহাসিন মুসাইয়্যাব আহমাদ পুণ্যNo ratings yet
- Differentiation Test: Marks:100 Time:1.30 HoursDocument2 pagesDifferentiation Test: Marks:100 Time:1.30 Hoursহাসিন মুসাইয়্যাব আহমাদ পুণ্যNo ratings yet
- Word Collection by MEDocument2 pagesWord Collection by MEহাসিন মুসাইয়্যাব আহমাদ পুণ্যNo ratings yet
- Equation Editor The Word 2007/2010 Equation Editor Shortcut CommandsDocument11 pagesEquation Editor The Word 2007/2010 Equation Editor Shortcut CommandspanchopanzaNo ratings yet
- Experiment 1 PDFDocument5 pagesExperiment 1 PDFJarin TasnimNo ratings yet
- Some Information About PointerDocument1 pageSome Information About Pointerহাসিন মুসাইয়্যাব আহমাদ পুণ্যNo ratings yet
- PSP Lab 1Document5 pagesPSP Lab 1হাসিন মুসাইয়্যাব আহমাদ পুণ্য0% (1)
- Prepared by Hasin Mussayab Ahmed, Lecturer, Dept of EEE, UU: Power Plant Engineering Lecture On Nuclear Power PlantDocument26 pagesPrepared by Hasin Mussayab Ahmed, Lecturer, Dept of EEE, UU: Power Plant Engineering Lecture On Nuclear Power Plantহাসিন মুসাইয়্যাব আহমাদ পুণ্যNo ratings yet
- AP Electron Config U ExceptionsDocument8 pagesAP Electron Config U Exceptionsহাসিন মুসাইয়্যাব আহমাদ পুণ্যNo ratings yet
- Editable - Application For Learner's Driving LicenseDocument2 pagesEditable - Application For Learner's Driving LicenseFoisul AlamNo ratings yet
- Fault AnalysisDocument5 pagesFault Analysisহাসিন মুসাইয়্যাব আহমাদ পুণ্য100% (1)
- Short Circuit Study For A Test NetworkDocument32 pagesShort Circuit Study For A Test Networkহাসিন মুসাইয়্যাব আহমাদ পুণ্যNo ratings yet