Professional Documents
Culture Documents
Biomedical Science in Brief
Uploaded by
Asa HatlahOriginal Title
Copyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
Biomedical Science in Brief
Uploaded by
Asa HatlahCopyright:
Available Formats
British Journal of Biomedical Science
ISSN: 0967-4845 (Print) (Online) Journal homepage: http://www.tandfonline.com/loi/tbbs20
Biomedical Science in Brief
M. Lotfy, G. Badra, W. Burham, F.Q. Alenzi, J.F. Bermejo-Martin, D. Bernardo,
Marta Dominguez-Gil, Ana Alonso, M.C. Garcia-Arevalo, M. Pino, Ortiz
De Lejarazu, J.M. Eiros, J. Ardura, A.J. Leön, J.A. Garrote, S. Resino, A.
Blanco-Quirös, A. Muñoz-Fernández, E. Arranz, V.K. Gopinath, M. Musa,
A.R. Samsudin, W. Sosroseno, M. Shigematsu, Y. Harada, T. Sekizuka, O.
Murayama, S. Takamiya, B.C. Millar, J.E. Moore, M. Matsuda, S. Elshibly, J. Xu,
B.C. Millar, C. Armstrong & J.E. Moore
To cite this article: M. Lotfy, G. Badra, W. Burham, F.Q. Alenzi, J.F. Bermejo-Martin, D. Bernardo,
Marta Dominguez-Gil, Ana Alonso, M.C. Garcia-Arevalo, M. Pino, Ortiz De Lejarazu, J.M. Eiros,
J. Ardura, A.J. Leön, J.A. Garrote, S. Resino, A. Blanco-Quirös, A. Muñoz-Fernández, E. Arranz,
V.K. Gopinath, M. Musa, A.R. Samsudin, W. Sosroseno, M. Shigematsu, Y. Harada, T. Sekizuka,
O. Murayama, S. Takamiya, B.C. Millar, J.E. Moore, M. Matsuda, S. Elshibly, J. Xu, B.C. Millar,
C. Armstrong & J.E. Moore (2006) Biomedical Science in Brief, British Journal of Biomedical
Science, 63:4, 171-184, DOI: 10.1080/09674845.2006.11732742
To link to this article: http://dx.doi.org/10.1080/09674845.2006.11732742
Published online: 23 May 2016.
Submit your article to this journal
View related articles
Full Terms & Conditions of access and use can be found at
http://www.tandfonline.com/action/journalInformation?journalCode=tbbs20
Download by: [University of Exeter] Date: 31 May 2016, At: 14:11
BIOMEDICAL SCIENCE IN BRIEF 171
Combined use of honey, bee propolis and
myrrh in healing a deep, infected wound in
a patient with diabetes mellitus
M. LOTFY*, G. BADRA†, W. BURHAM‡ and F. Q. ALENZI§
*
Biomedical Research Unit, Molecular and Cellular Biology Department, Genetic
Engineering and Biotechnology Research Institute, Minufiya University, Sadat City;
†
Department of Hepatology and Internal Medicine, National Liver Institute,
Minufiya University, Minufiya; ‡Department of Surgery, Faculty of Medicine,
Mansoura University, Mansoura, Egypt; §Department of Medical Laboratories,
College of Applied Medical Sciences, King Faisal University, Dammam, Saudi Arabia
Diabetes mellitus is a group of diseases characterised by high
levels of blood glucose resulting from defects in insulin
production, insulin action, or both. Diabetes can be
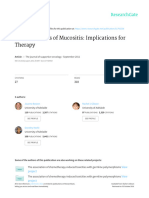
associated with serious complications including diabetic foot Fig. 1. The foot wound showing the healing process during treatment
disease. Diabetic foot disease is estimated to affect with the MPH paste.
Downloaded by [University of Exeter] at 14:11 31 May 2016
15% of people with diabetes.1
Wound healing is a process that involves inflammation, Two days later, the patient’s foot was re-examined and
proliferation/regeneration and finally remodeling. The debridement of the wound was performed to remove dead
normal orderly pattern is disrupted in chronic non-healing skin and necrotic tissue inside the opened cavity.
wounds, which are characterised by decreased levels of At all times during treatment, blood sugar level was
growth factors and increased protease activity. Wound controlled (in the range 150–170 mg/dL) using insulin. The
healing is affected by serum albumin, tissue oxygenation, patient was kept on metronidazole (1500 mg/day) and
infection, hyperglycaemia, cytokines and proteases.2 combined amoxicillin with clavulanic acid (1500 mg/day) for
A marker of non-healing wounds may be the prolonged 10 days. Thereafter, ciprofloxacin (1500 mg/day) was used
presence of extracellular matrix molecules in the dermis.3 instead of the combined amoxicillin with clavulanic acid.
Other markers and potential mediators include increased Pentoxifylline, vitacid calcium (vitamin C and calcium
levels of transforming growth factor (TGF)-β3,4 proteolytic carbonate) and vitazinc (vitamins A and E plus zinc) were
factors such as matrix metalloproteinases,5 and the absence added to the treatment regimen to aid vascularity and
of IGF-I.6 healing. From the beginning of treatment until the deep
Wound care includes a variety of approaches to enhance wound healed, the patient was maintained on an oral dose
healing, with treatment of infection, vascular reconstruction, of bee propolis (400 mg/day). Erythrocyte sedimentation rate
achieving adequate glycaemic control, removal of pressure, (a good indicator of treatment efficacy) was 125 mm prior to
and ongoing wound debridement being important aspects treatment and dropped to 65 mm after two weeks and
of this care.2 25 mm after four weeks, where it stabilised.
A deep wound with tissue loss in the right foot of a The most significant results were obtained during the use
65-year-old male patient with diabetes mellitus was treated of the MPH paste (800 mg bee propolis, 50 g myrrh, mixed
by a standard protocol that included strict control of blood together in honey). The paste was prepared every three days
sugar level. In addition, an antibiotic regimen was included and stored in a refrigerator. Wound cleaning was performed
to combat anaerobic and aerobic infection. Also, a paste daily using standard methods in addition to the MPH paste
consisting of myrrh, bee propolis and honey (MPH) was to fill the wound cavity. The effectiveness of the paste in
applied to the wound. Following treatment, the wound keeping the wound clean was indicated by a complete
settled and healed well (Fig. 1). absence of pus and cellular exudate. After four weeks the
The patient presented to the out-patient clinic of Minufiya wound had healed well and the patient returned to work.
University Hospital (MUH) with possible osteomyelitis. Poor wound healing in people with diabetes is well
A foot wound showed severe oedema and the patient was recognised.7 However, there is little information about many
unable to bear weight on the foot. On examination, the aspects of foot care in people with diabetes, including
patient had a large abscess beneath the skin of the foot. wound healing.8 The American Diabetes Association 9
An incision (3 cm) was made, pus was drained and the suggests a range of predisposing factors to explain poor
wound was cleaned. The patient returned home with healing of wounds in people with diabetes, including
antibiotic (gatifloxacin; 400 mg twice a day for five days) and abnormal cellular and/or inflammatory pathways,
an anti-inflammatory agent. peripheral neuropathy and vascular disease and/or tissue
X-ray examination showed no bony abnormality and hypoxia. Abnormal cellular function, particularly in
inflammation was prominent around the smallest toe. fibroblasts and neutrophils, has been found in people with
diabetes. In vitro, hyperglycaemia may be toxic to these
Correspondence to: Dr Mahmoud Lotfy cellular elements, while in vivo it may result in a greater
Molecular and Cellular Biology Department, Genetic Engineering and Biotechnology susceptibility to infection.
Research Institute, Minufiya University, Sadat City, PO 79, Minufiya, Egypt Modest differences in the function of neutrophils,
Email: mlotfy2000@yahoo.com macrophages and fibroblasts associated with hyperglycaemia
BRITISH JOURNAL OF BIOMEDICAL SCIENCE 2006 63 (4)
172 BIOMEDICAL SCIENCE IN BRIEF
have been postulated, but these have not been Honey is also an effective wound deodorant, an effect
demonstrated conclusively in vivo. Advanced glycosylation attributed to the presence of glucose, which is metabolised
end products accumulate in diabetes as a result of by bacteria in preference to proteinaceous necrotic tissue,
hyperglycaemia, leading to the non-enzymatic glycosylation resulting in the production of lactic acid and not the
of collagen.10 This process results in the production of malodorous compounds generated by protein degradation.33
abnormal collagen, which is highly inflexible and prone to In addition, the observed benefits of honey in infected
breakdown, particularly over pressure areas.11 wounds may be attributed to the high glucose content and
In this report, a paste is described that keeps a wound low pH, both of which stimulate macrophages.34
clean, which is especially important in cases that involve In the present case study, application of MPH resulted in a
tissue loss. The MPH paste contains safe and effective clean and odour-free wound, which healed well. However,
components that have prominent antimicrobial activity. the results of this single case need to be confirmation in
Myrrh is an oleogum resin obtained from the stem of the a study of a larger number of patients. In the meantime,
plant Commiphora molmol. It is a safe, natural flavouring use of the MPH paste would appear to reduce the cost of
substance approved by the US Food and Drug deep wound treatment and improve the outcome in the
Administration.12,13 In experimental studies on Swiss albino patients affected.
mice, myrrh from C. molmol exhibited no mutagenicity and
proved to be a potent cytotoxic drug against Ehrlich solid
tumour cells. The antitumour potential of C. molmol was References
comparable with that of the standard cytotoxic drug
cyclophosphamide. 14 Studies with animal and human 1 Mancini L, Ruotolo V. The diabetic foot: epidemiology. Rays
Downloaded by [University of Exeter] at 14:11 31 May 2016
models demonstrate antischistosomal and other 1997; 22: 511–23.
antiparasitic activity for myrrh and have found it to be safe 2 Bloomgarden ZT. Diabetes complications. Diabetes Care 2004;
and effective.15,16 27: 1506–14.
Myrrh has considerable antimicrobial activity and is 3 Loots MA, Lamme EN, Zeegelaar J, Mekkes JR, Bos JD,
used in a variety of diseases.17 It has antibacterial and Middelkoop E. Differences in cellular infiltrate and extracellular
antifungal activity against standard pathogenic strains of matrix of chronic diabetic and venous ulcers versus acute
Escherichia coli, Staphylococcus aureus, Pseudomonas aeruginosa wounds. J Invest Dermatol 1998; 111: 850–7.
and Candida albicans.18 In addition, it has an antidiabetic 4 Jude EB, Blakytny R, Bulmer J, Boulton AJ, Ferguson MW.
effect, especially in non-insulin-dependent diabetes Transforming growth factor-beta 1, 2, 3 and receptor type I and
mellitus (NIDDM). 19 Moreover, Myrrh has found II in diabetic foot ulcers. Diabet Med 2002; 19: 440–7.
pharmacological application in the reduction of cholesterol 5 Lobmann R, Ambrosch A, Schultz G, Waldmann K, Schiweck S,
and triglycerides.20 Lehnert H. Expression of matrix-metalloproteinases and their
Propolis is a resinous substance collected from trees by the inhibitors in the wounds of diabetic and non-diabetic patients.
bee Apis mellifera, which uses it as a building and insulating Diabetologia 2002; 45: 1011–6.
material in the hive. It is known that propolis has 6 Blakytny R, Jude EB, Martin Gibson J, Boulton AJ, Ferguson MW.
antimicrobial, antioxidative, anti-ulcer and antitumour, Lack of insulin-like growth factor 1 (IGF1) in the basal
anti-inflammatory, hypotensive and immune stimulatory keratinocyte layer of diabetic skin and diabetic foot ulcers.
activities.21 J Pathol 2000; 190: 589–94.
Antimicrobial activity has been observed against 7 Bouter KP, Storm AJ, de Groot RRM et al. The diabetic foot in
S. aureus,22,23 Streptococcus pyogenes,24 Gram-positive and Dutch hospitals: epidemiological features and clinical outcome.
Gram-negative bacterial species and Candida species,25,26 Eur J Med 1993; 2: 215–8.
S. mutans,27 anaerobic bacteria in the human oral cavity,28 8 Pecoraro RE, Ahroni JH, Bpyko EJ et al. Chronology and
salmonellas,29 and other microorganisms including extremities of tissue repair in diabetic lower-extremity ulcers.
mycobacteria.30 Antibacterial activity of propolis against Diabetes 1991; 40: 1305–13.
Staphylococcus aureus is higher when extracts are prepared in 9 American Diabetes Association. Consensus development
60–80% ethanol.22 In vitro synergy between propolis and conference on diabetic foot wound care. Diabetes Care 1991; 22:
antimicrobial drugs has been investigated, 31,32 and 1354–60.
preparations combining propolis with antibiotic and 10 McInnes A. Guide to the assessment and management
antifungal agents are of potential medical interest.26 of diabetic foot wounds. The Diabetic Foot 200; 4 (Suppl 1):
Honey is an ancient remedy that has regained popularity SI–II.
as an alternative treatment for antibiotic-resistant bacteria. 11 lkes RS, Wolfe JHN. The diabetic foot. BMJ 1991; 303:
Both honey and sugar pastes are considered useful as topical 1053–5.
antimicrobial agents, mainly because of their high 12 Hall BL, Oser BL. Recent progress in the consideration of
osmolarity and the ability to minimise water availability to flavoring ingredients under the food additive amendment. 3.
bacteria.33 Although the dilution of honey by wound fluid is GRAF substances. Food Technol 1965; 19: 151–97.
likely to reduce the efficacy of its osmotic effect, the slow and 13 Ford RA, Api AM, Letizia CS. Monographs on fragrance to raw
sustained production of hydrogen peroxide by some types materials. Food Chem Toxicol 1992; 30 (Suppl): 91S–92S.
of honey (e.g., manuka honey) is capable of maintaining 14 Al Harbi MM, Qureshi S, Ahmed MM, Rafatulla S, Shah AH.
an antimicrobial effect at a concentration approximately Effect of Commiphora molmol (oleogum resin) on the cytological
1000-fold higher than that used commonly in antiseptic and biochemical changes induced by cyclophosphamide in
solutions (i.e., 3%).33 Also, certain components of manuka mice. Am J Chin Med 1994; 22: 77–82.
honey (e.g., flavonoids and aromatic acids) demonstrate 15 Botros S, Sayed H, El-Dusoki H et al. Efficacy of Mirazid in
antimicrobial properties.33 comparison with praziquantel in Egyptian Schistosoma mansoni-
BRITISH JOURNAL OF BIOMEDICAL SCIENCE 2006 63 (4)
BIOMEDICAL SCIENCE IN BRIEF 173
infected school children and households. Am J Trop Med Hyg
2005; 72 (2): 119–23.
Interleukin (IL)-1β, IL-6 and IL-8 in nasal
16 Sheir Z, Nasr AA, Massoud A et al. A safe, effective, herbal secretions: a common role for innate
antischistosomal therapy derived from myrrh. Am J Trop Med
Hyg 2001; 65: 700–4. immunity in viral bronchial infection in
17 El Ashry ES, Rashed N, Salama OM, Saleh A. Components,
therapeutic value and uses of myrrh. Pharmazie 2003; 58 (3):
infants?
163–8.
18 Dolara P, Corte B, Ghelardini C et al. A. Local anaesthetic, J. F. BERMEJO-MARTIN*, D. BERNARDO*, MARTA
antibacterial and antifungal properties of sesquiterpenes from DOMINGUEZ-GIL†, ANA ALONSO‡, M. C. GARCIA-AREVALO*,
myrrh. Planta Med 2000; 66 (4): 356–8. M. PINO‡, R. ORTIZ DE LEJARAZU†, J. Mª EIROS†, J. ARDURA‡,
19 Al-Awadi F, Fatania H, Shamte U. The effect of a plant mixture A. J. LEÓN*, J.A. GARROTE*, S. RESINO§, A. BLANCO-
extract on liver gluconeogenesis in streptozotocin induced QUIRÓS*, Mª. A. MUÑOZ-FERNÁNDEZ§ and E. ARRANZ*
diabetic rats. Diabetes Res 1991; 18 (4):163–8.
20 Michie CA, Cooper E. Frankincense and myrrh as remedies in *
Mucosal Immunity Laboratory, Pediatrics Department. Institute of Molecular
children. J R Soc Med. 1991; 84: 602–5. Biology and Genetics; †Virology Laboratory, Hospital Clínico Universitario;
21 Lotfy M. Biological activity of bee propolis in health and disease. ‡
Pediatrics Deparment. Hospital Clínico Universitario, Valladolid; and
Asian Pac J Cancer Prev 2006; 7: 22–31. §
Immunomolecular Biology Laboratory. HGU Gregorio Marañón, Madrid, Spain
22 Fernandes Júnior A, Balestrin ECC, Cunha MLRS. Anti-
Staphylococcus aureus activity of bee propolis extracts prepared
Downloaded by [University of Exeter] at 14:11 31 May 2016
with different ethanol concentrations. Rev Ciênc Farm 2003; 24: There is growing evidence to support a role for the immune
147–52. response in lower respiratory tract infection (LRTI) due to
23 Fernandes Júnior A, Leomil L, Fernandes AAH, Sforcin JM. viruses in children.1,2 Respiratory syncytial virus (RSV) is the
The antibacterial activity of propolis produced by Apis mellifera most frequent aetiological agent of LRTI, but other
L. and Brazilian stingless bees. J Venom Anim Toxins 2001; respiratory viruses (human metapneumovirus [hMPV],
7: 173–82. parainfluenza virus types 1, 2 and 3, influenza virus B,
24 Bosio K, Avanzini C, D’avolio A, Ozimo O, Savoia D. In vitro adenovirus types 1, 2 and 5, and mycoplasmas) can produce
activity of propolis against Streptococcus pyogenes. Lett Appl symptoms indistinguishable from those elicited by RSV.
Microbiol 2000; 31: 174–7. Currently, however, there is controversy over the ability of
25 Drago I, Mombelli B, De Vecchi E, Fassina MC, Tocalli L, the different viral agents to induce pro-inflammatory
Gismondo MR. In vitro antimicrobial activity of propolis dry cytokines. Differences in the locally secreted cytokine profile
extract. J Chemotherapy 2002; 12: 390–5. in nasal washes between RSV and metapneumovirus
26 Stepanovic S, Antic N, Dakic I, Svabic-Vlahovic M. In vitro infections have been described, as have similarities between
antimicrobial activity of propolis and synergism between RSV and influenza.3 Additionally, immune function in
propolis and antimicrobial drugs. Microbiol Res 2003; 158: neonates differs from that in adults.4
353–7. At birth, the immune system is not fully mature. Neonatal
27 Koo H, Rosalen PL, Cury JA, Park YK, Bowen WH. Effects of antigen-presenting cells tend to be deficient in interleukin
compounds found in propolis on Streptococcus mutans growth (IL)-12. Furthermore, T cells secreting interferon (IFN)-γ
and on glucosiltransferase activity. Antimicrob Agents Chemother (Th1 cells) are more likely to undergo apoptosis after antigen
2002; 46: 1302–9. exposure. This might explain in part the Th2-skewed
28 Santos FA, Bastos EMA, Uzeda B, Carvalho MAR, Farias ESA, immunity in newborns. Neonatal immune cells appear
Braga FC. Antibacterial activity of Brazilian propolis and unable to provide strong responses because neonatal
fractions against oral anaerobic bacteria. J Ethnopharmacol 2002; antigen-presenting cells fail to up-regulate major
80: 1–7. histocompatibility complex (MHC) class II and co-
29 Orsi RO, Sforcin JM, Rall VLM, Funari SRC, Barbosa L, stimulatory molecules. Antibody levels and classes are also
Fernandes Júnior A. Susceptibility profile of Salmonella against different in early life to those found in adulthood.
the antibacterial activity of propolis produced in two regions of Neonatal antibody is characterised by increasing levels of
Brazil. J Venom Anim Toxins 2005; 11: 109–16. maternal immunoglobulin during the last trimester of
30 Banskota AH, Tezuka Y, Kadota S. Recent progress in pregnancy, which is replaced during the first year of life
pharmacological research of propolis. Phytother Res 2001; 15: progressively by neonatal IgM, then IgG and finally IgA
561–71. production. These special features of the neonatal immune
31 Scheller S, Dworniczak S, Waldemar KK, Rajca M, Tomczik A, system mean that young children are much more
Shani J. Synergism between ethanolic extract of propolis (EEP) susceptible to infectious disease; however, with ageing,
and anti-tuberculosis drugs on growth of mycobacteria. Z mortality due to infection decreases rapidly such that by the
Naturforsch C 1999; 54: 549–53. age of 10 years it is reduced 10- to 100-fold.4
32 Fernandes Júnior A, Balestrin EC, Betoni JEC, Orsi RO, Cunha Greater understanding of the immune response in LRTI is
MLRS, Montelli AC. Propolis: anti-Staphylococcus aureus activity important for better understanding of the physiopathology
and synergism with antimicrobial drugs. Mem Inst Oswaldo Cruz, of the condition, which may contribute to the development
Rio de Janeiro 2005; 100: 563–6.
33 Molan PC. The role of honey in the management of wounds. Correspondence to: J. F. Bermejo
J Wound Care 1999; 8: 415–8. Mucosal Immunity Laboratory, Pediatrics and Immunology Department,
34 Cooper RA, Molan PC. Honey in wound care. J Wound Care 1999; Facultad de Medicina, Valladolid, Spain
8: 340. Email: bermejo@ped.uva.es
BRITISH JOURNAL OF BIOMEDICAL SCIENCE 2006 63 (4)
174 BIOMEDICAL SCIENCE IN BRIEF
Table 1. Comparison of cytokine levels. Cytokine values are expressed as median and interquartile range.
Group 1 (A) Group 2 (B) Controls for Controls for P(a) P(b) P(c)
(n=8) (n=14) Group 1 (C) Group 2 (D)
(n=6) (n=11)
IL-6 502 (1033) 135 (296) 19 (33) 28 (34) 0.008* 0.001* 0.110
IL-8 8749 (8765) 3565 (3895) 540 (737) 913 (1479) 0.001* 0.001* 0.029*
IL-10 215 (457) 105 (206) 20 (114) 34 (64) 0.228 0.051 0.330
IL-2 33 (137) 16 (109) 12 (32) 12 (48) 0.414 0.687 0.714
IFNγ 36 (71) 14 (55) 3 (12) 14 (46) 0.345 0.536 0.868
IL-12p70 119 (283) 87 (114) 42 (108) 36 (200) 0.108 0.202 0.365
TNF-α 257 (295) 100 (266) 78 (198) 27 (154) 0.282 0.222 0.330
IL-1β 851 (5036) 704 (937) 104 (83) 124 (295) 0.013 *
0.001 *
0.664
IL-5 33 (63) 35 (75) 14 (36) 10 (17) 0.345 0.120 0.714
IL-4 18 (137) 40 (94) 22 (46) 18 (26) 0.573 0.095 0.664
P : Difference between groups (significance). Table 2. Characteristics of patients in Groups 1 and 2.
P(a): Difference between (A) and (C). Values are expressed as median and interquartile range
Downloaded by [University of Exeter] at 14:11 31 May 2016
P(b): Difference between (B) and (D). (except Gender and Prematurity).
P(c): Difference between (A) and (B).
*
P<0.05.
Group 1 Group 2 P value
of better treatment strategies. This is particular important in (n=8) (n=14)
severe cases of LRTI in young children. Age 1.5 (2.2) 7 (5.25) 0.005*
This study examines the cytokine profile in nasal washes (months)
from children under the age of one year, diagnosed with Gender 3/5 8/6 0.440
severe LRTI that showed bronchial involvement and clinical (male/female)
findings of viral respiratory disease. A control group Severity 4 (2.1) 3.25 (3.75) 0.330
showing no respiratory disease is used for comparison. (M-WCAS score)
The patient group comprised children up to the age of 12 Days 6.5 (3.5) 4 (3.5) 0.188
months who needed hospitalisation in the paediatric in hospital
department of the Hospital Clínico Universitario, Valladolid, Weight 4510 (2655) 7200 (2877) 0.008*
Spain, from February to May 2005, all of whom had signs of (grams)
viral bronchial illness (tachypnoea, prolonged expiratory
C reactive protein 4.2 (8.9) 8.95 (37.85) 0.297
time, wheezing, rales, chest retractions, dyspnoea of sudden (mg/dL)
appearance, fever).5 Those children who showed acute and
Prematurity 6/2 9/5 0.604
severe presentation were recruited for the study (n=22).
(term/premature)
The decision to hospitalise was made independently by the
attending physicians on the basis of clinical findings alone P : Difference between groups (significance). *
P<0.05.
(e.g., respiratory distress requiring oxygen therapy, poor
feeding with signs of dehydration and/or apnoea). Evaluation microfugue (Eppendorf), cytokine levels were determined
of clinical severity was performed at admission following the in the supernatants using the FlowCytomix multiplex
modified Wood`s Clinical Asthma Score (M-WCAS).5 human Th1/Th2 10plex kit (Bender) and five-colour flow
Those children under one year old with no respiratory or cytometry (Cytomics, Beckman Coulter).
inflammatory pathology admitted during the observation Differences in infant characteristics and levels of cytokines
period were included in the study as age-matched controls. were analysed for significance by a non-parametric test
In all cases, parent or other legal permission was requested (Mann-Whitney). Children with clinical signs of viral
prior to sample extraction and this was recorded in the bronchial infection who were RSV-positive (Group 1, n=8,
patient’s history. age median [IQR]: 1.5 [2.2] months) were compared with an
Nasopharyngeal aspirate samples were obtained within 24 age-matched control group (n=6, age median [IQR]: 2 [1]
hours of admission to hospital by gently flushing the infants’ months). The differences in ages between the RSV-positive
nostrils with 4 mL sterile saline solution. Secretions were group and the control group were not statistically significant
divided into aliquots, snap frozen immediately in dry ice by the Mann-Whitney test (P=0.852).
and then stored at –70˚C until tested. The group of children with clinical signs of viral bronchial
Viral presence was diagnosed in infected infants and infection but negative for the screened viruses (Group 2,
excluded in controls on nasopharyngeal aspirates by direct n=14, age median [IQR]: 7 [5.2] months) was also compared
immunofluorescence staining (Imagen, Dako, Denmark) for with an age-matched control group (n=11, age median
RSV, adenovirus, parainfluenza types 1, 2 and 3, and [IQR]: 4 [4] months). Once again, the differences in ages
influenza A and B after viral culture on MDCK, LLCMK2, between Group 2 and the control group were not statistically
A549 and Hep2 cells. significant by the Mann-Whitney test (P=0.536). The χ2 test
After centrifugation of nasal samples at 5000 rpm in a was used to compare the effects of prematurity and gender.
BRITISH JOURNAL OF BIOMEDICAL SCIENCE 2006 63 (4)
BIOMEDICAL SCIENCE IN BRIEF 175
Respiratory syncytial virus was isolated in eight children, Finally, IL-1β and IL6, together with the chemokine IL-8,
while 14 children were negative for all the viruses screened. have an important role in mediating viral clearance;
A common pattern in the profile of nasal cytokine secretion however, they may also mediate immune-mediated
in young children suffering from severe LRTI was apparent. pathogenesis, leading to exacerbation of the inflammatory
When the children in Group 1 were compared with its age- response in the bronchial tree. Some authors have proposed
matched control group, IL-1β, IL-8 and IL-6 levels were immunomodulatory approaches to treat the inflammatory
higher in the former (P=0.008, P=0.001 and P=0.013, component in LRTI caused by RSV.8,9
respectively; Table 1). When similar comparison was made On the basis of the results presented here, this approach
between Group 2 and its age-matched control group, IL-1β, deserves further study, not only in RSV infection but also in
IL-8 and IL-6 levels were also higher in the former (P=0.001, those patients with non-identified viral agents who show
Table 1). In addition, when Groups 1 and 2 were compared, clinical criteria of viral LRTI (at least in children less than a
IL-8 levels were significantly higher in Group 1 patients year old).
(P=0.029, Table 1). As shown in Table 2, differences in the M- In conclusion, the high levels of IL-1β, IL-6 and IL-8 in
WCAS score for severity between Groups 1 and 2 were not nasal aspirates reveal the important role played by innate
statistically significant; thus, differences between cytokine immunity in bronchial viral disease in young children. This
levels in these groups could not be attributed to differences is independent of the causative viral agent
in clinical severity.
IL-1β, IL-8, and IL-6 are produced during the very early This study received financial support from the Nutriben Prize of the
stages of infection.6 IL-1β stimulates almost all local and Asociación Española de Pediatría and from Fondo de Investigaciones
systemic inflammatory responses. IL-6, which can be Sanitarias (project PI050358). Dr Bermejo-Martin was supported
Downloaded by [University of Exeter] at 14:11 31 May 2016
induced by IL-1β, is pyrogenic, induces the liberation of by FIS, CD05/00153. David Bernardo received a grant from the
acute-phase reactants by the liver, and, in turn, switches off Ministerio de Educación y Ciencia (programa FPU). Alberto León
pro-inflammatory cytokine production. While IL-1β received a grant from Junta de Castilla y León (O.C. 14/11/03-O.R.
mediates the initial adhesive reaction of neutrophils to the 26/01/0). J. A. Garrote and S. Resino received a grant from Fondo de
endothelium, IL-8 appears to be essential for the directed Investigaciones Sanitarias. E. Arranz was supported by the
migration of leucocytes into infected tissue. Thus, it is not Programa Ramón y Cajal. The authors acknowledge the cooperation
unexpected that these cytokines play an important role of the nursing team in the Infants’ Section of Hospital Clínico
independently of the causative viral agent in children under Universitario de Valladolid, who kindly performed the nasal washes.
the age of one year (with an immature immune response Also, thanks are due to Mrs. Ana Sanz for her assistance.
and underdeveloped specific responses).
Laham et al. previously reported lower pro-inflammatory
cytokine production in human metapneumovirus infection References
than in RSV and influenza infections.3 On the basis of these
differences, and given that these viruses lead to identical 1 McNamara P, Flanagan BF, Selby AM, Hart CA, Smyth RL. Pro-
clinical manifestations, they concluded that innate and anti-inflammatory responses in respiratory syncytial virus
inflammation is not critical for the induction of respiratory bronchiolitis. Eur Respir J 2004; 23:106–12.
symptoms in viral respiratory diseases. 2 Openshaw PJ. Antiviral immune responses and lung
However, Laham’s study compared only infected patients, inflammation after respiratory syncytial virus infection. Proc Am
and it is important to include non-respiratory pathology Thorac Soc 2005; 2 (2): 121–5.
control groups in any study of cytokine secretion profiles. In 3. Laham FR, Israele V, Casellas JM et al. Differential production of
the present study, despite the finding of higher levels of IL- inflammatory cytokines in primary infection with human
8 in Group 1, the inflammation-related mediators (IL-1β, IL8, metapneumovirus and with other common respiratory viruses
IL6) were clearly elevated in Groups 1 and 2, compared with of infancy. J Infect Dis 2004; 189: 47–56.
their respective age-matched controls. Both groups showed 4 Openshaw PJ, Yamaguchi Y, Tregoning JS. Childhood infections,
clinical signs of viral infection, despite the fact that those in the developing immune system, and the origins of asthma.
Group 2 were negative for the viruses screened. However, J Allergy Clin Immunol 2004; 114: 1275–7.
the virus culture methods employed are less sensitive than 5 Martinon-Torres F, Rodriguez-Nunez A, Martinon-Sanchez JM.
are polymerase chain reaction (PCR)-based methods. Heliox therapy in infants with acute bronchiolitis. Pediatrics
The small number of patients studied here, together with 2002; 109: 68–73.
the number of parameters measured, means that larger 6 Van Reeth K, Van Gucht S, Pensaert M. In vivo studies on
studies are required to limit the possible role of type 1 and cytokine involvement during acute viral respiratory disease of
type 2 statistical errors, and such studies are currently swine: troublesome but rewarding. Vet Immunol Immunopathol
underway. Nevertheless, the immature immunological 2002; 87 (3–4): 161–8.
status of the studied children supports a possible role for 7 Tal G, Mandelberg A, Dalal I et al. Association between common
these innate factors of immunity in RSV-mediated disease. Toll-like receptor 4 mutations and severe respiratory syncytial
Comparison of cytokine profiles between patients with mild virus disease. J Infect Dis 2004; 189: 2057–63.
and severe disease is also needed. 8 Sheeran P, Jafri H, Carubelli C et al. Elevated cytokine
Why some children present with severe disease while concentrations in the nasopharyngeal and tracheal secretions of
others do not remains unexplained. However, genetics children with respiratory syncytial virus disease. Pediatr Infect
seems to explain, at least in part, the different clinical Dis J 1999; 18 (2): 115–22.
pictures (i.e., the presence of mutations in the Toll-like 9 Rutigliano JA, Graham BS. Prolonged production of TNF-alpha
receptor family).7 This particular aspect deserves further exacerbates illness during respiratory syncytial virus infection. J
study. Immunol 2004; 173: 3408–17.
BRITISH JOURNAL OF BIOMEDICAL SCIENCE 2006 63 (4)
176 BIOMEDICAL SCIENCE IN BRIEF
Role of interleukin-1β and tumour necrosis
factor-α on hydroxyapatite-induced
phagocytosis by murine macrophages
(RAW264.7 cells)
IL-1b (pg/mL
V. K. GOPINATH*, M MUSA†, A. R. SAMSUDIN*
and W. SOSROSENO*†
*
School of Dental Sciences and †Department of Immunology,
School of Medical Sciences, Universiti Sains Malaysia, 16150 Kota Bharu, Malaysia
Hydroxyapatite (HA) is used in bone reconstruction in
dental and orthopaedic surgery. Characteristic features of
HA such as bioactivity, osteoconduction, osteoinduction and Minutes
the fact that it is similar in composition to bone mineral are Fig. 1. Effect of HA and latex bead particles on the release of IL-1β
factors that favour its use in regeneration of hard tissues.1 by RAW264.7 cells at various incubation times. Untreated cell
However, when HA is implanted in tissue, monocytes culture supernatants were used as negative controls. The bars
Downloaded by [University of Exeter] at 14:11 31 May 2016
/macrophages are attracted to the implant site.2 Analysis of represent standard deviation.
tissue from animal models3 and humans demonstrate the
phagocytosis of HA particles.4 an enzyme-linked immunosorbent assay (ELISA) kit (Bender
The most common causes of failure in implants is aseptic MedSystems, Vienna, Austria) according to the
loosening, which is mostly triggered by wear particles that manufacturer ’s instructions. Unstimulated cell culture
activate macrophages to produce cytokines such as supernatants were used as a control. All materials used in
interleukin (IL)-1 and IL-6, thereby initiating bone the cell culture process were obtained from Sigma.
resorption around the implant.5 It has been shown that both RAW264.7 cells were stimulated with HA or latex beads in
IL-1β and tumour necrosis factor-α (TNFα) are identified in the presence of various concentrations of anti-murine IL-1β
the periprosthetic tissues of patients who undergo revision or TNFα antibody (Sigma). Phagocytic index (PI) was
surgery for total joint replacement,6 indicating that these assessed at 60 min. The number of phagocytosed cells per
cytokines play a crucial role in inflammation and osteolysis. 100 cells was assessed by light microscopy (Leica, Germany).
Indeed, implant materials such as titanium and alumina The PI was calculated by counting the number of engulfed
ceramic particles stimulate the production of pro- particles per macrophage, as described previously.11 All
inflammatory cytokines by murine or human macrophages experiments were carried out in triplicate.
in vitro.7,8 Data analysis included paired t-test at different time
Recently, this group showed that murine macrophage points and one–way analysis of variance followed by
phagocytic activity induced by HA is under the regulation of Fisher’s least squared difference in the experiments where
inducible nitric oxide synthase (iNOS).9 Therefore, this study neutralising IL-1β and TNFα antibodies were used with
aims to evaluate the effect of HA-induced phagocytosis on activated cells (SPSS, Chicago, IL).
IL-1β and TNFα production and to demonstrate whether or IL-1β and TNFα were detected at 30 and 60 min in
not murine macrophages (RAW264.7 cells) use these unstimulated RAW264.7 cell cultures (Figs. 1 and 2).
cytokines in an autocrine fashion. Following activation of the cells, IL-1β concentration was
Hydroxyapatite powder (3.5–8 µm particle size) was higher in cultures treated with latex beads than with HA
suspended in sterile saline.10 Latex beads (3 µm; Sigma, (P<0.05; Fig. 1). Murine macrophages activated with HA and
St. Louis) were used as a control. The HA particles were latex bead particles produced significantly higher levels
stained with crystal violet prior to use in the phagocytic of IL-1β when compared to unstimulated controls at 15,
assay. 30 and 60 min (P<0.05). Levels of IL-1β were not detected at
Hydroxyapatite-induced phagocytosis by RAW264.7 cells 7 min following activation with HA or latex beads.
was assessed as previously described.9 Briefly, RAW 264.7 Examining the trend within groups, IL-1β production
cells, obtained from ATCC, were cultured in Dulbecco’s increased at 7–15 and 15–30 min (P<0.05) but did not
modified Eagle’s medium (supplemented with 1% increase significantly at 30–60 min following activation with
penicillin/streptomycin) and 10% heat-inactivated fetal calf HA or latex beads.
serum in a humidified atmosphere of 5% CO2. Five million TNFα levels in the cultures treated with HA were higher
cells were incubated with 1 x 105 HA particles or latex beads than in those treated with latex beads (P<0.05; Fig. 2).
in culture medium (1 mL) in a sterile tube for 7, 15, 30 and 60 Examining the trend within groups, TNFα production
min. At each time point, culture supernatants were showed significant increases at 7–15 ,7–30 and 7–60 min
harvested and IL-1β and TNFα levels were determined by (P<0.05); however, the HA-treated group also showed
significance at 15–60 min (P<0.05) but did not increase
Correspondence to: Dr. V. K. Gopinath significantly at 15–30 and 30–60 min. In the latex bead-
School of Dental Sciences, Universiti Sains Malaysia, treated group, TNFα production did not increase
16150 Kota Bharu, Malaysia significantly at 15–30, 15–60 or 30–60 min.
Email: gopinath@kb.usm.my Pretreatment of cells with anti-murine IL-1β antibody
BRITISH JOURNAL OF BIOMEDICAL SCIENCE 2006 63 (4)
BIOMEDICAL SCIENCE IN BRIEF 177
TNF-α (pg/mL)
PI
Minutes Anti-IL-1β (µg/mL)
Fig. 2. Effect of HA and latex bead particles on the release of TNFα Fig. 3. RAW264.7 cells stimulated with HA or latex beads in the
by RAW264.7 cells at various incubation times. Untreated cell presence of anti-murine IL-1β. The PI was assessed at 60 min by
culture supernatants were used as negative controls. The bars calculating the number of ingested particles per macrophage. The
Downloaded by [University of Exeter] at 14:11 31 May 2016
represent standard deviation. bars represent standard deviation.
resulted in lower phagocytic activity induced by HA and dependent manner. These results are not unexpected as
latex beads (P<0.05; Fig. 3). Similarly, HA- and latex bead- macrophages during phagocytosis are known to produce
induced phagocytosis by RAW264.7 cells was significantly cytokines.15 Similarly, murine macrophages (ANA-1 cells)
reduced when the cells were coated with anti-murine TNFα and human monocytes (THP-1 cells) synthesise TNFα and
(P<0.05; Fig. 4). IL-1β when the cells come into contact with implant
The present study shows that murine macrophages materials such as titanium and HA.7,8,16,17 This suggests that
upon ingestion of HA and latex beads secreted IL-1β and implant materials activate macrophages to produce pro-
TNFα. Latex beads used as a control in the present study are inflammatory cytokines.
know to activate macrophages to produce cytokines as While TNFα produced by HA-stimulated RAW264.7 cells
observed in previous in vitro studies where latex beads was detected after 7-min incubation, IL-1β production by the
activated J774.2 cells to produce TNF-α12 and human same cell cultures was observed later at 15 min, suggesting
monocyte to secrete IL-1β and TNFα.13 Unstimulated that TNFα may be produced earlier than IL-1β during
RAW 264.7 cells that produced IL-1β and TNFα at 30 and HA-induced macrophage phagocytosis; however, the exact
60 minutes were used as a negative control in the present mechanism to explain these findings remains unclear.
study. This indicated spontaneous activation of the Previous reports have shown that TNFα messenger RNA
macrophages during isolation and culture; this is in (mRNA) expression in human macrophages responding to
correlation with previous studies using RAW 264.7 cells.14 titanium is detected earlier than is IL-1β gene expression.18
The present study shows that HA and latex beads TNF-α is also known to induce production of IL-1β.19
stimulate RAW264.7 cells to produce TNFα in a time- Therefore, it can be assumed that HA stimulates RAW264.7
cells to produce TNFα initially, which then in turn initiates
production of IL-1β; however, this remains to be confirmed.
It should also be noted that IL-1β production by latex
bead-activated cells was higher than that by HA-stimulated
cells. However, HA-activated macrophages secreted
significantly higher amounts of TNFα than did latex bead-
activated cells. This might be due to the physical structure of
the particle, as study has shown that fewer titanium particles
PI
than polyethylene particles are required to stimulate peak
production of TNFα and IL-1β.20
The present study also demonstrated that decreased
phagocytosis of HA and latex beads was observed when the
cells were incubated with HA or latex beads in the presence
of anti-murine TNFα or murine IL-1β antibody. This
suggests that TNFα and IL-1β produced by HA-stimulated
RAW264.7 cells may be required to induce phagocytosis.
Macrophages are the main source of both cytokines,
Anti-TNF-α (ng/mL)
which are required for cell activation and function.19,21
Fig. 4. RAW264.7 cells stimulated with HA or latex bead in Defective phagocytosis, as seen in TNFα- and IL-1β-deficient
the presence of anti-murine TNFα. The PI was assessed at mice,22,23 highlights the importance of the role of TNFα and
60 min by calculating the number of ingested particles per IL-1β in murine macrophage phagocytosis induced by HA.
macrophage. Extrapolation of the results from the present study to
BRITISH JOURNAL OF BIOMEDICAL SCIENCE 2006 63 (4)
178 BIOMEDICAL SCIENCE IN BRIEF
humans remains speculative; however, they confirm Role of nitric oxide in hydroxyapatite-induced phagocytosis by
previous reports that show HA-stimulated human murine macrophage cell line (RAW264.7). Arch Oral Biol 2006;
macrophages produce TNFα and IL-1β,16,17 which suggests 51: 339–44.
that HA-stimulated RAW264.7 cells are a good model of HA- 10 Nordsletten L, Hogasen AK, Konttinen YT, Santavirta S,
induced human macrophage responses. Furthermore, when Aspenberg P, Aasen AO. Human monocytes stimulation by
used as an implant in humans, HA activates macrophages,24 particles of hydroxyapatite, silicon carbide and diamond:
and analysis of periprosthetic tissues from patients with in vitro studies of new prosthesis coatings. Biomaterials 1996; 17:
implants has identified cytokines such as TNFα and IL-1β.6 1521–7.
Thus, it seems plausible that macrophages are activated 11 Barid I, Nawawi S, Sosroseno W. Effects of parachlorophenol
immediately after implantation with HA and then and camphorated parachlorophenol on the phagocytic activities
subsequently produce TNFα and IL-1β. This may enhance of a murine macrophage cell line (RAW264.7). J Endod 2002; 28:
macrophage infiltration and function and induce pro- 806–10.
inflammatory responses to surrounding implanted 12 Olivier CR, Hindie J-L, Duval M, Bomila-Koradjim G, Nagel
materials, as is seen in other pathophysiological disease MD. Uptake of polystyrene bead bearing functional groups by
processes.25 macrophages and fibroblasts. Colloids Surf B Biointerfaces 2004;
In summary, the results of the present study show that HA 33: 23–31.
stimulates murine macrophages (RAW264.7 cells) to produce 13 Harada Y, Wang JT, Doppalapudi VA et al. Differential effects of
IL-1β and TNFα; however, addition of neutralising different forms of hydroxyapatite and hydroxyapatite/
antibodies to IL-1β or TNFα result in reduction of particle- tricalcium phosphate particulates on human monocyte/
induced phagocytosis. This suggests that HA-induced macrophages in vitro. J Biomed Mater Res 1996; 31: 19–26.
Downloaded by [University of Exeter] at 14:11 31 May 2016
phagocytic activity by murine macrophages is dependent on 14 Huang SL, Cheng WL, Lee CT, Huang HC, Chan CC.
the presence of IL-1β and TNFα. Contribution of endotoxin in macrophage cytokine response to
ambient particles in vitro. J Toxicol Environ Health 2002; 65:
This research was supported by an e-IRPA category EA grant (No. 1261–72.
06-02-05-00029 EAR) from the Malaysian Government. The 15 Underhill DM, Ozinsky A. Phagocytosis of microbes: complexity
hydroxyapatite was kindly prepared by staff of the School of in action. Annu Rev Immunol 2002; 20: 825–52.
Material and Mineral Resources Engineering, Universiti Sains 16 Laquerriere P, Grandjean-Laquerriere A, Guenounou M,
Malaysia, Malaysia. Laurent-Maquin D, Frayssinet P, Nardin M. Correlation between
sintering temperature of hydroxyapatite particles and the
production of inflammatory cytokines by human monocytes.
References Colloids Surf B Biointerfaces 2003; 30: 207–13.
17 Grandjean-Laquerriere A, Laquerriere P, Guenounou M,
1 LeGeros RZ. Properties of osteoconductive biomaterials: Laurent-Maquin D, Phillips TM. Importance of the surface area
calcium phosphates. Clin Orthop Relat Res 2002; 395: 81–98. ratio on cytokine production by human monocytes in vitro
2 Heymann D, Pradal G, Benahmed M. Cellular mechanisms of induced by various hydroxyapatite particles. Biomaterials 2005;
calcium phosphate ceramic degradation. Histol Histopathol 1999; 26: 2361–9.
14: 871–7. 18 Garrigues GE, Cho DR, Rubash HE, Goldring SR, Herndon JH,
3 Rahbek O, Kold S, Bendix K, Overgaard S, Soballe K. No effect Shanbhag AS. Gene expression clustering using self-organizing
of hydroxyapatite particles in phagocytosable sizes on implant maps: analysis of the macrophage response to particulate
fixation: an experimental study in dogs. J Biomed Mater Res A biomaterials. Biomaterials 2005; 26: 2933–45.
2005; 73: 150–7. 19 Aggarwal BB, Samanta A, Feldman M. TNF-alpha. In:
4 Bloebaum RD, Beeks D, Dorr LD, Savory CG, DuPont JA, Oppenheim JJ, Feldman M, eds. Cytokines Reference.
Hofmann AA. Complications with hydroxyapatite particulate Amsterdam/New York: Elsevier/Academic Press, 2000: 413–34.
separation in total hip arthroplasty. Clin Orthop Relat Res 1994; 20 Rader CP, Sterner T, Jakob F, Schutze N, Eulert J. Cytokine
298: 19–26. response of human macrophage-like cells after contact with
5 Jiranek WA, Machado M, Jasty M et al. Production of cytokines polyethylene and pure titanium particles. J Arthroplasty 1999; 14:
around loosened cemented acetabular components. Analysis 840–8.
with immunohistochemical techniques and in situ 21 Dinarello CA. Interleukin-1. Cytokine Growth Factor Rev 1997; 8:
hybridization. J Bone Joint Surg Am 1993; 75: 863–79. 253–65.
6 Catelas I, Campbell PA, Dorey F, Frausto A, Mills BG, Amstutz 22 Fantuzzi G, Sacco S, Ghezzi P, Dinarello CA. Physiological and
HC. Semi-quantitative analysis of cytokines in MM THR tissues cytokine responses in IL-1 beta-deficient mice after zymosan-
and their relationship to metal particles. Biomaterials 2003; 24: induced inflammation. Am J Physiol 1997; 273: 400–6.
4785–97. 23 Marino MW, Dunn A, Grail D et al. Characterization of tumor
7 Soloviev A, Schwarz EM, Kuprash DV et al. The role of p105 necrosis factor-deficient mice. Proc Natl Acad Sci USA 1997; 94:
protein in NFkappaB activation in ANA-1 murine macrophages 8093–8.
following stimulation with titanium particles. J Orthop Res 2002; 24 Chatelet JC, Setiey L. Long-term bone behavior in total primary
20: 714–22. hip arthroplasty with a fully hydroxyapatite-coated femoral
8 Yagil-Kelmer E, Kazmier P, Rahaman MN, Bal BS, Tessman RK, stem: a continuous series of 120 cases with twelve years follow-
Estes DM. Comparison of the response of primary human blood up (in French). Rev Chir Orthop Reparatrice Appar Mot 2004; 90:
monocytes and the U937 human monocytic cell line to two 628–35.
different sizes of alumina ceramic particles. J Orthop Res 2004; 22: 25 Esch T, Stefano G. Proinflammation: a common denominator or
832–8. initiator of different pathophysiological disease processes. Med
9 Gopinath VK, Musa M, Samsudin AR, Lalitha P, Sosroseno W. Sci Monit 2002; 8: 1–9.
BRITISH JOURNAL OF BIOMEDICAL SCIENCE 2006 63 (4)
BIOMEDICAL SCIENCE IN BRIEF 179
Genetic heterogeneity of the cytolethal Table 1. Isolates of C. lari analysed in the present study.
distending toxin B (cdtB) gene locus among
isolates of Campylobacter lari Isolate Source Country Accession
number
UN C. lari JCM2530T Seagull Japan AB 266778
M. SHIGEMATSU*, Y. HARADA*, T. SEKIZUKA*, O. MURAYAMA*, UN C. lari 28 Mussel N. Ireland AB 266779
S. TAKAMIYA†, B. C. MILLAR‡, J. E. MOORE‡ and M. MATSUDA*
*
UN C. lari 48 Mussel N. Ireland AB 266780
Laboratory of Molecular Biology, Graduate School of Environmental Health
Sciences, Azabu University, Fuchinobe 1-17-71, Sagamihara 229-8501; †Department UN C. lari 84C-1 Human N. Ireland AB 266781
of Molecular and Cellular Parasitology, Juntendo University School of Medicine, UN C. lari 84C-2 Human N. Ireland AB 266782
Bunkyoku Hongo 2-1-1, Tokyo 113-8421, Japan; and ‡Department of Bacteriology, UN C. lari 99 Sea water N. Ireland AB 266783
Northern Ireland Public Health Laboratory, Belfast City Hospital, Belfast BT9 7AD, UN C. lari 170 Seagull Japan AB 266784
Northern Ireland, UK
UN C. lari 264 Mussel N. Ireland AB 266785
UN C. lari 274 Mussel N. Ireland AB 266786
Thermophilic Campylobacter species, primarily C. jejuni and C. UN C. lari 288 Black-tailed gull Japan AB 266787
coli, are curved Gram-negative bacteria that are the UN C. lari 293 Seagull Japan AB 266788
recognised cause of acute bacterial diarrhoea around the UN C. lari 296 Human Canada AB 266789
world.1,2 C. lari is a relatively recently discovered thermophilic UN C. lari 298 Human Canada AB 266790
Downloaded by [University of Exeter] at 14:11 31 May 2016
Campylobacter species first isolated from mammalian and UN C. lari 299 Human USA AB 266791
avian species, particularly seagulls of the genus Larus.1,3 C. lari
UN C. lari 381 Mussel N. Ireland AB 266792
has also been shown to be a cause of clinical infection.4–7
An atypical group of isolates of urease-positive UN C. lari 2316A3 NA NA AB 266793
thermophilic campylobacters (UPTC) was first isolated from UPTC CF89-12 River water Japan AB 266794
the natural environment in England in 1985.8 Thereafter, UPTC CF89-14 River water Japan AB 266795
these organisms were described as a biovar or variant of UPTC NCTC12892 River water England AB 266796
C. lari.9,10 Subsequent reports described four human isolates UPTC NCTC12893 River water England AB 266797
in France.9,11 Some additional isolates of UPTC have also been
UPTC A1 Seagull N. Ireland AB 266798
reported in Ireland,12–14 in The Netherlands15 and in Japan.16,17
The possible association of UPTC with human disease UPTC A3 Seagull N. Ireland AB 266799
remains unclear. Two representative taxa, namely urease- UPTC 89049 Human France AB 266800
negative (UN) C. lari and UPTC, occur within the species of UPTC 92251 Human France AB 266801
C. lari.18 NA: not available.
Although several Campylobacter species’ cytotoxins have
been identified,19,20 only the cytolethal distending toxin
(CDT) has been characterised in detail.21,22 The cdt genes of A schematic representation of the cdtB gene and its
C. jejuni have been cloned and characterised by Pickett et al.21 genetic loci for C. lari RM2100 (GenBank Accession No.
However, in relation to the cdt genes, no reports have yet AAFK00000000)23 including the locations of a primer pair for
appeared for C. lari. the cdtB (JCB common up and JCB common down [Asakura
Therefore, the aim of the present study is to clone, primer])24 employed in the present study for PCR
sequence and analyse the cdtB gene of C. lari isolates and amplification is shown in Figure 1. This primer pair was
compare the sequences obtained with those of other designed to generate a product of approximately 700 bp
thermophilic campylobacters. (equivalent to a 90% segment of the cdtB structural gene of
Twenty-four isolates of C. lari (UN C. lari [n=16] and UPTC C. lari RM2100, AAFK00000000) of the cdtB gene with
[n=8]) were used in the present study (Table 1), together C. jejuni, C. coli and C. fetus isolates.24 The polymerase chain
with three reference strains (JCM2530T, NCTC12892 and reaction (PCR) was performed in 50-µL reaction volumes, for
NCTC12893). The test organisms were isolated from 30 cycles at 94˚C for 30 sec, 55˚C for 30 sec, 72˚C for 45 sec,
different sources in several countries. The organisms were followed by a final extension of 72˚C for 5 min.
cultured on blood agar containing defibrinated horse blood Amplified PCR products were separated by 1.0% (w/v)
(Nippon Bio-Test, Tokyo, Japan) and supplemented with agarose gel electrophoresis in 0.5x TBE at 100 V and detected
campylobacter-selective medium (Nissui, Tokyo, Japan), by ethidium bromide staining. PCR products amplified by
under microaerophilic conditions at 37˚C for two days. the constructed primer pair for the partial cdtB gene were
Template DNA was prepared by boiling in water at 95˚C purified using a QIA quick PCR purification kit (Qiagen, CA,
for five minutes. The PCR mixture contained 10 mmol/L USA) and inserted in the pGEM-T vector using the pGEM-T
Tris-HCl (pH 8.3), 50 mmol/L KCl, 1.5 mmol/L MgCl2, Easy Vector System (Promega, Tokyo, Japan).
400 µmol each dNTP, 1 µmol each primer, and 1 unit of Thermus Sequencing of the cloned cdtB gene fragment was
aquaticus (Taq) DNA polymerase (Takara Bio, Shiga, Japan). performed (Hitachi DNA autosequencers SQ-5500L and SQ-
5500EL) after a dideoxy nucleotide sequencing reaction,
Correspondence to: Dr. Motoo Matsuda using a Thermo Sequenase premixed cycle sequencing kit
Laboratory of Molecular Biology, School of Environmental Health Sciences, (Amersham Pharmacia Biotech, Tokyo, Japan). Sequence
Azabu University, Fuchinobe 1-17-71, Sagamihara 229-8501, Japan analysis of the PCR amplicons was carried out using the
Email: matsuda@azabu-u.ac.jp GENETYX-MAC (version 9) computer software.
BRITISH JOURNAL OF BIOMEDICAL SCIENCE 2006 63 (4)
180 BIOMEDICAL SCIENCE IN BRIEF
nucleotide and deduced amino acid sequences with the
24 C. lari isolates examined. An extremely high sequence
variability of the cdtB was also demonstrated between the
24 C. lari isolates and the other two thermophilic
campylobacters (C. jejuni and C. coli).
In relation to the cdt gene in campylobacters, Martinez et al.
found the cdt gene variant, a putative shortened cdtB gene
fragment in two of the 100 C. jejuni isolates derived from
several sources (humans and animals) and countries, by a
multiplex PCR procedure. Although, they observed several
point mutations throughout the remaining cdtA, -B and -C
sequences, it has become very clear that cdt genes from
C. jejuni are highly homologous.26
In the present study, the partial cdtB gene (approximately
Fig. 1. Schematic representation of the cdtB genes of C. lari, 720 bp), similar in length in 24 isolates of C. lari, was
including A) the locations of a primer pair for the amplification of the examined using a constructed PCR primer in silico (Asakura
cdtB gene fragment of C. lari and B) nucleotide sequences of the primer). Thus, essentially all the C. lari isolates employed in
primer pair.24 the present study had the cdtB gene. However, high genetic
heterogeneity of 74.7–100% nucleotide sequence similarity
Nucleotide sequences of approximately 720 bp of the of the partial cdtB gene fragment was identified among all
Downloaded by [University of Exeter] at 14:11 31 May 2016
partial cdtB gene fragments from 24 C. lari isolates were 24 isolates of C. lari by cloning and sequencing procedures.
compared to each other and to accessible sequence data of This is the first demonstration of the genetic heterogeneity
other thermophilic campylobacters (C. lari RM2100, of cdt genes among C. lari isolates by cloning and sequencing
AAFK00000000; C. jejuni RM1221, CP000025; C. coli RM2228, procedures.
AAFL00000000), employing CLUSTAL W software (1.7 A dendrogram showing phylogenetic relationships was
program),25 which was incorporated in DDBJ. A constructed using UPGMA, based on the nucleotide
phylogenetic tree was constructed by the unweighted pair sequence information of the partial cdtB gene fragments
group method, using arithmetic means analysis (UPGMA) amplified from 24 C. lari isolates (Fig. 2) and demonstrated
available on the GENETYX-MAC program (version 9.0). hypervariability among the cdtB genes of 24 C. lari isolates.
In the present study, the primer pair (JCB common up and This implies that the C. lari organisms examined are
down, Asakura primer) 24 amplified PCR products of genetically variable, at least based on the cdtB gene
approximately 720 bp in length with all 24 isolates of C. lari information obtained, and separate from the other two
(UN C. lari [n=16] and UPTC [n=8]) (data not shown). For thermophilic campylobacters, C. jejuni and C. coli.
PCR cloning of the partial cdtB fragments, PCR products Previously, this group demonstrated genetic
were purified and inserted in the pGEM-T vector using the hypervariability of 31 isolates of UPTC, as determined by
TA cloning procedure. The nucleotide and deduced amino multilocus enzyme electrophoresis typing (MLEE).14 The
acid sequence data of the partial and possible open reading present results for the cdtB gene from eight UPTC isolates is
frame (ORF) of the cdtB gene fragments cloned and consistent with the results of the genetic hypervariability
sequenced from 24 isolates of C. lari are accessible in the obtained by MLEE.
DDBJ/EMBL/GenBank nucleotide sequence database (Table 1).
The nucleotide sequences of the partial cdtB gene UPTC A1
fragments (approximately 720 bp) of 16 UN C. lari isolates UPTC A3
UPTC 92251
showed 81–100% similarity to each other. Those of eight UN C. lari 381
UPTC 89049
UPTC isolates showed 74.7–99.6% similarity to each other. In UN C. lari 274
addition, those of the cdtB gene fragments showed UN C. lari 84C-2
UN C. lari 288
74.7–100 % similarity among 16 UN C. lari isolates and eight UN C. lari 299
UN C. lari 84C-1
UPTC isolates. Moreover, the nucleotide sequences of the UN C. lari 48
cdtB gene fragments of 25 C. lari isolates, including C. lari UN C. lari 2316A3
UN C. lari RM2100
RM2100, showed 63.9–88.0% sequence similarity to those of UN C. lari 264
UN C. lari 296
C. jejuni RM1221 and C. coli RM2228. UN C. lari 28
When the deduced amino acid sequences of the partial UN C. lari JCM2530T
UN C. lari 298
and possible ORFs of the cdtB fragments of 24 C. lari isolates UN C. lari 170
were determined, the partial and possible ORFs were UN C. lari 293
UPTC CF89-12
estimated to consist of 241–242 amino acids. The deduced UPTC CF89-14
UN C. lari 99
amino acid sequence alignments were also examined for UPTC NCTC12892
partial and possible ORFs of the cdtB fragments of 24 C. lari UPTC NCTC12893
C. coli RM2228
isolates, as well as those of C. lari RM2100, C. jejuni RM1221 C. jejuni RM1221
and C. coli RM2228. The partial and possible ORFs of the cdtB 0.5 0.1 0.1824
gene fragments from 25 C. lari isolates, including C. lari
RM2100, showed 78.2–99.6 % amino acid sequence similarity Fig. 2. Phylogenetic tree based on sequence similarity data of the
to each other, and 66.6–78.5% amino acid sequence similarity cdtB gene fragments from 24 C. lari isolates examined and
to those of the ORFs of C. jejuni RM1221 and C. coli RM2228. thermophilic isolates of C. lari RM2100, C. jejuni RM1221 and
Thus, cdtB had a high sequence heterogeneity in both C. coli RM2228. Values in the figure represent evolutionary distances.
BRITISH JOURNAL OF BIOMEDICAL SCIENCE 2006 63 (4)
BIOMEDICAL SCIENCE IN BRIEF 181
References Prevalence of cytolethal distending toxin production in
Campylobacter jejuni and relatedness of Campylobacter spp. cdtB
1 Benjamin J, Leaper S, Owen R J, Skirrow MB. Description of genes. Infect Immun 1996; 64: 2070–8.
Campylobacter laridis, a new species comprising the nalidixic 22 Pickett CL, Whitehouse CA. The cytolethal distending toxin
acid-resistant thermophilic Campylobacter (NARTC) group. Curr family. Trends Microbiol 1999; 7: 292–7.
Microbiol 1983; 8: 231–8. 23 Fouts DE, Mongodin EF, Mandrell RE et al. Major structural
2 Blaser MJ, Taylor DN, Feldman RA. Epidemiology of differences and novel potential virulence mechanisms from the
Campylobacter jejuni infections. Epidemiol Rev 1983; 5: 157–76. genomes of multiple Campylobacter species. PLoS Biol. 2005; 3:
3 Skirrow MB, Benjamin J. ‘1001’ Campylobacters: cultural e15. 0072–85. http://biology.plosjournals.org/
characteristics of intestinal campylobacters from man and 24 Asakura M, Yoshida E, Taguchi M, Kobayashi K, Yamazaki S.
animals. J Hyg (Camb) 1980; 85: 427–42. Poster presentation at the 77th annual meeting of the Japanese
4 Nachamkin I, Stowell C, Skalina D, Jones AM, Hoop RM, Society for Bacteriology, Abstract (in Japanese). Jpn J Bacteriol
Smibert RM. Campylobacter laridis causing bacteremia in an 2004; 59: 283.
immunosuppressed patient. Ann Intern Med 1984; 101: 55–7. 25 Thompson JD, Higgins DG, Gibson TJ. CLUSTAL W: improving
5 Simor A E, Wilcox L. Enteritis associated with Campylobacter the sensitivity of progressive multiple sequence alignment
laridis. J Clin Microbiol 1987; 25: 10–2. through sequence weighting, position-specific gap penalties
6 Martinot M, Jaulhac B, Moog R et al. Campylobacter lari and weight matrix choice. Nucleic Acids Res 1994; 212: 4673–80.
bacteremia. Clin Microbiol Infect 2001; 7: 96–7. 26 Martinez I, Mateo E, Churruca E, Girbau C, Alonso R,
7 Werno AM, Klena JD, Shaw GM, Murdoch DR. Fatal case of Fernandez-Astorga A. Detection of cdtA, cdtB and cdtC genes in
Campylobacter lari prosthetic joint infection and bacteremia in an Campylobacter jejuni by multiplex PCR. Int J Med Microbiol 2006;
Downloaded by [University of Exeter] at 14:11 31 May 2016
immunocompetent patient. J Clin Microbiol 2002; 40: 1053–5. 296: 45–8.
8 Bolton FJ, Holt AV, Hutchinson DN. Urease-positive
thermophilic campylobacters. Lancet 1985; i: 1217–8.
9 Mégraud F, Chevrier D, Desplaces N, Sedallian A, Guesdon JL.
Urease-positive thermophilic Campylobacter (Campylobacter
Molecular diagnosis of native mitral valve
laridis variant) isolated from an appendix and from human endocarditis due to Corynebacterium
feces. J Clin Microbiol 1988; 26: 1050–1.
10 Owen RJ, Costas M, Sloss L, Bolton FJ. Numerical analysis of
striatum
electrophoretic protein patterns of Campylobacter laridis and
allied thermophilic campylobacters from the natural S. ELSHIBLY*, J. XU†, B. C. MILLAR†, C. ARMSTRONG*
environment. J Appl Bacteriol 1988; 65: 69–78. and J. E. MOORE†
11 Bezian MC, Ribou G, Barberis-Giletti C, Megraud F. Isolation of *
Department of Microbiology, Craigavon Area Hospital, Craigavon, Co. Armagh;
a urease-positive thermophilic variant of Campylobacter lari from and †Northern Ireland Public Health Laboratory, Department of Bacteriology,
a patient with urinary tract infection. Eur J Clin Microbiol Infect Belfast City Hospital, Lisburn Road, Belfast, Northern Ireland
Dis 1990; 9: 895–7.
12 Wilson IG, Moore JE. Presence of Salmonella spp. and
Campylobacter spp. in shellfish. Epidemiol Infect 1996; 116: 147–53. Corynebacterium striatum is a relatively rare causal agent of
13 Kaneko A, Matsuda M, Miyajima M, Moore JE, Murphy PG. infective endocarditis (IE). Its association with IE is
Urease-positive thermophilic strains of Campylobacter isolated complicated by its relatively slow growth, its role as a
from seagulls (Larus spp.). Lett Appl Microbiol 1999; 29: 7–9. possible contaminant in such cases, and the phenotypic
14 Matsuda M, Kaneko A, Stanley T et al. Characterization of difficulty in laboratory identification.
urease-positive thermophilic Campylobacter subspecies by The present study reports a case of culture-positive
multilocus enzyme electrophoresis typing. Appl Environ endocarditis due to C. striatum in a 77-year-old woman who
Microbiol 2003; 69: 3308–10. showed no risk factors for endocarditis. To date, there have
15 Endtz HP, Vliegenthart JS et al. Genotypic diversity of been 12 reports of endocarditis due to C. striatum, which are
Campylobacter lari isolated from mussels and oysters in The discussed and summarised.
Netherlands. Int J Food Microbiol 1997; 34: 79–88. Currently, use of molecular microbiological methods has
16 Matsuda M, Kaneko A, Fukuyama M et al. First finding of been limited, but such techniques have been adopted in
urease-positive thermophilic strains of Campylobacter in river several clinical microbiology laboratories. Molecular
water in the Far East, namely, in Japan, and their phenotypic methods of identification using the polymerase chain
and genotypic characterization. J Appl Bacteriol 1996; 81: 608–12. reaction (PCR) and sequencing of 16S ribosomal DNA
17 Matsuda M, Shibuya T, Itoh Y et al. First isolation of urease- (rDNA) from the causal agent isolated by blood culture may
positive thermophilic Campylobacter (UPTC) from crows (Coruvs be very useful in the identification of causal agents in
levaillantii) in Japan. Int J Hyg Environ Health 2002; 205: 321–4. culture-positive endocarditis, which prove difficult to
18 Matsuda M, Moore JE. Urease-positive thermophilic identify using a conventional approach.
Campylobacter species. Appl Environ Microbiol 2004; 70: 4415–8. The 77-year-old patient presented to hospital with a
19 Johnson WM, Lior H. A new heat-labile cytolethal distending chronic three-month history of weight loss, fatigue and
toxin (CLDT) produced by Campylobacter spp. Microb Pathog arthralgia. A history was taken in accordance with the
1988; 4: 115–26.
20 Schulze F, Hanel I, Borrmann E. Formation of cytotoxins by Correspondence to: Dr. B. Cherie Millar
enteric Campylobacter in humans and animals. Zentralbl Bakteriol Northern Ireland Public Health Laboratory,
1998; 288: 225–36. Belfast City Hospital, Belfast BT9 7AD, Northern Ireland, UK
21 Pickett CL, Pesci EC, Cottle DL, Russell G, Erdem AN, Zeytin H. Email: bcmillar@niphl.dnet.co.uk
BRITISH JOURNAL OF BIOMEDICAL SCIENCE 2006 63 (4)
182 BIOMEDICAL SCIENCE IN BRIEF
questionnaire criteria for endocarditis, as described a final extension at 72˚C for 10 min. During each run,
previously by Paturel et al.1 The patient had no risk factors molecular-grade water was included randomly as several
for IE nor any underlying disease. She was febrile, anaemic negative controls and DNA templates from Staphylococcus
and demonstrated septic embolisation. On auscultation, aureus were included as a positive control, as appropriate.
she had a new cardiac murmur, which was shown to Following amplification, samples (15 µL) were removed
originate from leakage from her native mitral valve. In from each reaction mixture and examined by electrophoresis
addition, the presence of a vegetation on this valve was (80V, 45 min) in gels composed of 2% (w/v) agarose (Gibco,
demonstrated. UK) in TAE buffer (40 mmol/L Tris, 20 mmol/L acetic acid,
On presentation, she had an elevated C-reactive protein 1 mmol/L EDTA [pH 8.3]), and then stained with ethidium
(CRP) of 244.7 mg/L. This rose to 255.5 mg/L and then bromide (5 µg/100 mL).
decreased to 46.4 mg/L nine days after commencement of Gels were visualised under ultraviolet (UV) illumination
intravenous antibiotics. Three sets of blood cultures were using a gel image analysis system (UVP Products, England)
taken, all of which grew a Gram-positive rod provisionally and all images were archived as digital graphic (.bmp) files.
identified by conventional techniques as a Corynebacterium Amplicons chosen for automated sequencing were purified
sp. Antibiotic treatment was continued for a further seven using a QIAquick PCR purification kit (Qiagen, UK) and
weeks and the patient was discharged without any signs or eluted in Tris–HCl (10 mmol/L [pH 8.5]) prior to sequencing,
symptoms of IE. in order to remove dNTPs, polymerases, salts and primers.
Phenotypic identification of the Gram-positive organism, The amplicon was sequenced on the ALF II Express
isolated from three sets of blood cultures (isolate identifier: automated sequencer using the primer PSL which was
CAHE68720) was performed using the API Corynebacterium labelled with Cy-5 fluorescent dye and used in conjunction
Downloaded by [University of Exeter] at 14:11 31 May 2016
system (bioMérieux, Las Halles, France). It gave the profile with the Sequenase fluorescence-labelled primer cycle
3140345 and an identification of Brevibacterium sp. (47%). sequencing kit (Thermo, Amersham, UK).
A repeat test gave the API profile 3100305, yielding The resulting sequence obtained (570 bp) was compared
Corynebacterium group G (55.6%). The organism was with those stored in the GenBank data system using FASTA
sensitive to vancomycin, teicoplanin, gentamicin and alignment software (www.ebi.ac.uk), and deposited in
rifampicin by standard in vitro antibiotic disc diffusion GenBank (accession number DQ018338). On BLAST analysis
susceptibility assay. in combination with previously reported criteria used for
Given the relatively poor phenotypic identification interpretation of partial 16S rRNA gene sequences,5 the
obtained, the isolate was forwarded for molecular sequence gave a 100% identification for C. striatum
identification through PCR amplification and direct (GenBank accession number AY008302) followed by
sequencing of a large but partial region of the 16S rRNA C. xerosis X81906 (99% identity) and an uncultured
gene, corresponding to the base position of approximately Rhodococcus sp. (98% identity).
811–1374 of Escherichia coli ATCC 25922 16S rRNA (GenBank At present, there are approximately 73 recognised species
accession number: X80724). All DNA isolation procedures in the genus Corynebacterium, including C. striatum. This
were carried out in accordance with the DNA contamination species was first described by Chester in 1901 as Bacterium
management guidelines of Millar et al.2 and in a Class II striatum, and then by Eberson in 1918. 6 Several
biological safety cabinet (MicroFlow, England). This was Corynebacterium spp. have been described as causal agents of
situated in a room separated from that used to set up nucleic IE.7 Over the past 15 years, however, there have only been 10
acid amplification reaction mixes, and also from the ‘post- cases of C. striatum endocarditis. In a review of the 12 cases
PCR’ room, in order to minimise contamination and the in the literature, seven patients were male. The median age
possibility of false-positive results. was 62.6 years for females (age range: 46–72 years) and 59
Bacterial DNA was extracted from the isolate using the years for males (age range: 24–76 years), as shown in Table 1.
Roche high-purity PCR template preparation kit (Roche, Native valves have been most frequently infected (three
England) in accordance with the manufacturer ’s mitral, three aortic, one pulmonary and one tricuspid);
instructions. Extracted DNA was transferred to a clean tube however, there have been three cases of prosthetic valve
and stored at –80˚C prior to PCR. For each batch of endocarditis (two aortic, one mitral). In total to date, two
extractions, a negative extraction control containing all deaths have been attributed to C. striatum endocarditis, with
reagents other than the isolate was performed. Reaction aortic valve involvement (Table 18–17).
mixes (50 µL) were set up as follows: 10 mmol/L Tris–HCl The current case highlights the important role of
(pH 8.3), 50 mmol/L KCl, 2.5 mmol/L MgCl2 200µmol/L (each) molecular methods in the correct identification of the causal
dATP, dCTP, dGTP and dTTP, 1.25 units Thermus aquaticus agent, namely C. striatum. Identification of the genus
(Taq) DNA polymerase (Amplitaq, Perkin Elmer), 0.2 µmol/L Corynebacterium and differentiation of species in the genus is
appropriate ‘broad range’ primers (PSL3 [forward] 5’ -AGG usually based on differential biochemical tests.18 However, in
ATT AGA TAC CCT GGT AGT CCA-3’ and P13P4 [reverse] most diagnostic laboratories, the API system is employed for
5’ - AGG CCC GGG AAC GTA TTC AC -3’) and 4 µL DNA the rapid and convenient speciation of isolates in this genus.
template. Previously, Funke et al.,19 although concluding that the API
Prior to PCR cycling, sealed tubes containing DNA Coryne system is a useful tool for identifying the diverse
template and all PCR reagents were introduced to the group of coryneform bacteria encountered in the routine
thermal cycler at 96˚C to avoid non-specific annealing during clinical laboratory, noted that additional tests were required
the initial ramp stage. The reaction mixtures were subjected when using version 2.0 of the kit instead of version 1.0, in
to the following thermal cycling parameters in a Perkin Elmer order to identify strains completely. In the case reported
Cetus 2400 thermocycler: 96˚C for 3 min followed by 40 here, the API Coryne system was not able to identify the
cycles at 96˚C for 1 min, 55˚C for 1 min, 72˚C for 1 min, and isolate correctly, even to the genus level, yielding the close
BRITISH JOURNAL OF BIOMEDICAL SCIENCE 2006 63 (4)
BIOMEDICAL SCIENCE IN BRIEF 183
Table 1. A review of the literature describing cases of Corynebacterium striatum endocarditis previously reported.
Patient Predisposing Echocardiographic Valve Antibiotic therapy Surgery Survival Ref
(age, sex) condition for IE findings
72, F RF in childhood. Mitral TOE: inconclusive due to NK Vancomycin/rifampicin None Yes (8)
valvotomy and prosthetic what was believed to be
mitral valve replacement calcified vegetations from
30 y later. Culture-negative previous episode of
IE, 18 m previously culture-negative IE
61, F RF in childhood Partially calcified vegetation Mitral Vancomycin/gentamicin None Yes (8)
on mitral valve
50, M* Previous mycotic aneurysm TOE: 15 mm vegetation on Aortic Antibiotic therapy unsuccessful. Aortic Yes (9)
for which he underwent aortic valve visible before i. Penicillin/gentamicin valve
surgical treatment. and after antibiotic therapy ii. Vancomycin/netilmicin replaced
iii Vancomycin/gentamicin/
doxycycline
62, F Diastolic murmur after TOE: No evidence of Bioprosthetic Vancomycin None Yes (10)
surgery for a bioprosthetic vegetation or abscess aortic
aortic valve replacement formation valve
a few years previously
Downloaded by [University of Exeter] at 14:11 31 May 2016
72, F Aortic valve replacement TTE indicated a 15 mm Periprosthetic i.Vancomycin/gentamicin, None No (11)
with a metallic prosthesis vegetation on the mitral aortic ii benzyl penicillin
valve; however, this was valve
not confirmed at
post mortem
68, M Mitral valve regurgitation TTE: moderate left Mitral i.Vancomycin, None Yes (12)
(2 y) ventricular dysfunction, ii. Penicillin
mitral valve regurgitation.
TOE: mitral regurgitation
and vegetation on atrial
aspect of anterior
mitral valve leaflet
73, M Pacemaker 6 y previously. TTE: normal Tricuspid i. Vancomycin Removal of Yes (13)
Batteries replaced and TOE: vegetation on old ii. Oral co-trimoxazole/ old electrode
electrode cut, although electrode wire which rifampin wire via
distal part left in place. remained after previous iii. Vancomycin due to jugular vein
Draining sinus tract removal penicillin resistance
TOE after surgical removal
of wire. Vegetation visible
on tricuspid valve
24, M Ventriculoatrial shunt TTE/TOE: 10 mm vegetation Pulmonary i. Amoxicillin/netilmicin Bed sore treated Yes (14)
on pulmonary valve close ii. Amoxicillin (source of infection)
to distal extremity of iii Amoxicillin, Endovascular
ventriculoatrial shunt catheter; netilmicin/teicoplanin process attempted
TTE after 7 w therapy: iv. Oral amoxicillin to remove catheter
vegetation unchanged; in pulmonary
TOE 20 m later: no vegetations artery (failed)
54, M None TTE: moderate aortic Aortic i. Ampicillin/vancomycin Aortic valve Yes (7)
insufficiency following detection ii. Ampicillin + gentamicin replacement
of new murmur. iii. Vancomycin alone
TTE/TOE: ruptured aortic (due to rash)
tricuspid valve cusp with severe
aortic insufficiency
76, M† No history of Echo: aortic/tricuspid Aortic Parenteral ampicillin/ Died prior to No (15)
heart disease insufficiency gentamicin planned emergency
surgery to replace
aortic valve
46, F Unknown Unknown Unknown Initial treatment failure None Yes (16)
with linezolid.
Switched to daptomycin/
rifampicin
68, M Aortic and mitral TTE: 10 mm vegetation on Prosthetic Vancomycin due to antibiotic None Yes (17)
prosthetic valves prosthetic mitral valve mitral valve pan-resistance
RF: rheumatic fever; IE: infective endocarditis; TTE: transthoracic echocardiography; TOE: transoesphageal echocardiography; NK: not known.
*
Polymicrobial, multiresistant infective endocarditis due to Staphylococcus epidermidis and Corynebacterium striatum.
†
Causative organism most closely resembled Corynebacterium striatum.
BRITISH JOURNAL OF BIOMEDICAL SCIENCE 2006 63 (4)
184 BIOMEDICAL SCIENCE IN BRIEF
phylogenetic neighbour Brevibacterium sp. (47%) on first 3 Campbell PW 3rd, Phillips JA 3rd, Heidecker GJ, Krishnamani
testing and eventually Corynebacterium group G (55.6%) on MR, Zahorchak R, Stull TL. Detection of Pseudomonas
subsequent testing. (Burkholderia) cepacia using PCR. Pediatr Pulmonol 1995; 20: 44–9.
Where conventional blood culture yields a viable culture, 4 Widjojoatmodjo MN, Fluit AC, Verhoef J. Rapid identification of
it is important to be able to identify it reliably to aid patient bacteria by PCR-single-strand conformation polymorphism.
management, in particular antimicrobial susceptibility and J Clin Microbiol 1994; 32: 3002–7.
the prescribing of an appropriate regimen of anti-infective 5 Goldenberger D, Kunzli A, Vogt P, Zbinden R, Altwegg M.
chemotherapy, and for epidemiological reporting of Molecular diagnosis of bacterial endocarditis by broad-range
aetiological agents of IE. Molecular methods may be PCR amplification and direct sequencing. J Clin Microbiol 1997;
introduced as part of a polyphasic approach to aid the 35: 2733–9.
identification of a culture-positive case, but are not required 6 Eberson F. A bacteriologic study of the diphtheroid organisms
in the primary diagnostic setting. with special reference to Hodgkin’s disease. J Infect Dis 1918;
Conventional phenotypic characterisation of cultures can 23: 1–42.
be problematic (e.g., speciation of enterococci and coagulase- 7 Rufael DW, Cohn SE. Native valve endocarditis due to
negative staphylococci) and molecular identification Corynebacterium striatum: case report and review. Clin Infect Dis
primarily by sequence analysis allows these organisms to be 1994; 19: 1054–61.
speciated in such cases. Additionally, occasional 8 Stoddart B, Sandoe JA, Denton M. Corynebacterium striatum
employment of these molecular methods may help to endocarditis masquerading as connective tissue disorders.
quality assure the correct phenotypic identification of Rheumatology (Oxford) 2005; 44: 557–8.
cultures from patients with IE. 9 Kocazeybek B, Ozder A, Kucukoglu S, Kucukates E, Yuksel H,
Downloaded by [University of Exeter] at 14:11 31 May 2016
Although primary diagnostic laboratories may be able to Olga R. Report of a case with polymicrobial endocarditis related
detect and isolate a significant organism from the blood to multiresistant strains. Chemotherapy 2002; 48: 316–9.
cultures of patients with IE, these laboratories may have 10 Houghton T, Kaye GC, Meigh RE. An unusual case of infective
problems with the correct and reliable identification of such endocarditis. Postgrad Med J 2002; 78: 290–1.
isolates. This may be due to the fact that the cultures are 11 de Arriba JJ, Blanch JJ, Mateos F, Martinez-Alfaro E, Solera J.
unusual in that they are not observed frequently in the Corynebacterium striatum first reported case of prosthetic valve
clinical laboratory, either in the context of IE or other endocarditis. J Infect 2002; 44: 193.
infections (e.g., Weissella confusa20) or where the organism is 12 Juurlink DN, Borczyk A, Simor AE. Native valve endocarditis
new (e.g., Streptococcus nov. sp.21). due to Corynebacterium striatum. Eur J Clin Microbiol Infect Dis
Where a new organism is identified by 16S rRNA 1996; 15: 963–5.
sequence-based methods, further work is required to fully 13 Melero-Bascones M, Munoz P, Rodriguez-Creixems M, Bouza E.
characterise and describe the new taxon, whether a new Corynebacterium striatum: an undescribed agent of pacemaker-
species or indeed a new genus. Generally, this necessitates related endocarditis. Clin Infect Dis 1996; 22: 576–7.
the forwarding of such isolates to a specialist taxonomy 14 Tattevin P, Cremieux AC, Muller-Serieys C, Carbon C. Native
laboratory, as even those primary diagnostic laboratories valve endocarditis due to Corynebacterium striatum: first
with specialist molecular ability are often not suited for an reported case of medical treatment alone. Clin Infect Dis 1996;
in-depth analysis of the taxonomic positioning of such 23: 1330–1.
isolates. Limitations of routine identification systems 15 Markowitz SM, Coudron PE. Native valve endocarditis caused
(e.g., API, Vitek, etc.) in primary diagnostic laboratories by an organism resembling Corynebacterium striatum. J Clin
create problems in identifying difficult-to-identify Microbiol 1990; 28: 8–10.
organisms reliably, and employment of 16S rRNA gene 16 Shah M, Murillo JL. Successful treatment of Corynebacterium
sequencing has proved reliable in such cases.22 striatum endocarditis with daptomycin plus rifampin. Ann
In conclusion, this report demonstrates the value of Pharmacother 2005; 39: 1741–4.
molecular methods in the correct identification of 17 Mashavi M, Soifer E, Harpaz D, Beigel Y. First report of
phenotypically difficult-to-identify causal agents of IE. prosthetic mitral valve endocarditis due to Corynebacterium
Furthermore, it represents the first instance in which PCR striatum: successful medical treatment. Case report and
and direct sequencing has been employed to aid the literature review. J Infect 2006; 52: e139–41.
identification of C. striatum endocarditis. Diagnostic clinical 18 Anon. Aerobic, non-spore forming Gram-positive bacilli
laboratories should be familiar with the existence of such (Ch 34). In: Baron EJ, Peterson LR, Finegold SM, eds. Bailey &
techniques and have a mechanism in place to permit the Scott’s Diagnostic Microbiology (9th edn). St. Louis: Mosby, 1994:
referral of such organisms to a specialist/reference laboratory 457–73.
with experience in these molecular techniques. 5 19 Funke G, Renaud FN, Freney J, Riegel P. Multicenter evaluation
of the updated and extended API (RAPID) Coryne database 2.0.
J Clin Microbiol 1997; 35: 3122–6.
References 20 Flaherty JD. Levett PN, Dewhirst FE, Troe TE, Warren JR,
Johnson S. Fatal case of endocarditis due to Weissella confusa.
1 Paturel L, Casalta JP, Habib G, Nezri M, Raoult D. Actinobacillus J Clin Microbiol 2003; 41: 2237–9.
actinomycetemcomitans endocarditis. Clin Microbiol Infect 2004; 21 Woo PC, Tam DM, Leung KW et al. Streptococcus sinensis sp. nov.,
10: 98–118. a novel species isolated from a patient with infective
2 Millar BC. Xu J, Moore JE. Risk assessment models and endocarditis. J Clin Microbiol 2002; 40: 805–10.
contamination management: implications for broad-range 22 Millar B. Moore J, Mallon P et al. Molecular diagnosis of infective
ribosomal DNA PCR as a diagnostic tool in medical bacteriology. endocarditis – a new Duke’s criterion. Scand J Infect Dis 2001;
J Clin Microbiol 2002; 40: 1575–80. 33: 673–80.
BRITISH JOURNAL OF BIOMEDICAL SCIENCE 2006 63 (4)
You might also like
- Original Article at YashDocument4 pagesOriginal Article at YashVicky DwiNo ratings yet
- Mushrooms and immunityDocument13 pagesMushrooms and immunityMartinaNo ratings yet
- Polysaccharides from natural resources exhibit great potential in the treatment of ulcerative colitis_ A reviewDocument12 pagesPolysaccharides from natural resources exhibit great potential in the treatment of ulcerative colitis_ A reviewMartinaNo ratings yet
- Therapeutical Properties of Apigenin a Review on the Experimental Evidence and Basic MechanismsDocument27 pagesTherapeutical Properties of Apigenin a Review on the Experimental Evidence and Basic MechanismsMario CoelhoNo ratings yet
- 2020 The Potential of Probiotics For Treating Acne Vulgaris - A Review of Literature On Acne and MicrobiotDocument6 pages2020 The Potential of Probiotics For Treating Acne Vulgaris - A Review of Literature On Acne and MicrobiotJeslyn TengkawanNo ratings yet
- Pie Diabetico 2Document6 pagesPie Diabetico 2danielNo ratings yet
- The Physiological Relevance of The Intestinal Microbiota - Contributions To Human HealthDocument6 pagesThe Physiological Relevance of The Intestinal Microbiota - Contributions To Human HealthLuisNo ratings yet
- Frankova 2021Document12 pagesFrankova 2021Anonymous gzXHCYNo ratings yet
- Medicina: Supplement Consumption and Periodontal Health: An Exploratory Survey Using The Bigmouth RepositoryDocument9 pagesMedicina: Supplement Consumption and Periodontal Health: An Exploratory Survey Using The Bigmouth RepositoryDadi SindhuNo ratings yet
- bioengineering-11-00357Document15 pagesbioengineering-11-00357Shahmeer HassanNo ratings yet
- Pubmed Zip - Versions - 1 - Package Entries - Fvets 10 1283679 - Fvets 10 1283679Document14 pagesPubmed Zip - Versions - 1 - Package Entries - Fvets 10 1283679 - Fvets 10 1283679Danielle OliveiraNo ratings yet
- Clinical Evidence On The Potential Beneficial Effects ofDocument12 pagesClinical Evidence On The Potential Beneficial Effects ofDyane VatriciaNo ratings yet
- Hallopeau 10.1001@jamadermatol.2019.3515Document1 pageHallopeau 10.1001@jamadermatol.2019.3515Faten HayderNo ratings yet
- Microbiome and Probiotics in Acne Vulgaris-A Narrative ReviewDocument11 pagesMicrobiome and Probiotics in Acne Vulgaris-A Narrative ReviewVanNo ratings yet
- Fecal Microbiota Transplantation To Prevent and TRDocument5 pagesFecal Microbiota Transplantation To Prevent and TRaimane.makerNo ratings yet
- Characteristics of Microbial Drug Resistance and Its Correlates in Chronic Diabetic Foot Ulcer InfectionsDocument9 pagesCharacteristics of Microbial Drug Resistance and Its Correlates in Chronic Diabetic Foot Ulcer InfectionsPutri Alif PermatasariNo ratings yet
- Alimentos Funcionais e PancreasDocument2 pagesAlimentos Funcionais e PancreasGeovani LopesNo ratings yet
- 1 s2.0 S0753332222011520 MainDocument20 pages1 s2.0 S0753332222011520 MainTejas krishnakanthNo ratings yet
- FULLTEXT01Document124 pagesFULLTEXT01menaxieNo ratings yet
- Nutrients 14 04940 v3Document15 pagesNutrients 14 04940 v3Tamires SantanaNo ratings yet
- Nutrients: Nutrition and Rheumatoid Arthritis in The Omics' EraDocument23 pagesNutrients: Nutrition and Rheumatoid Arthritis in The Omics' EraMikhael FeliksNo ratings yet
- New Hypotheses For The Health ProtectiveDocument70 pagesNew Hypotheses For The Health ProtectiveMiguelArceMonroyNo ratings yet
- .60 Subyek 18 Mohseni Et Al 2017 - A Randomized, Double Placebo, Controlled Trial - The Effects of Probiotic Administration On Wound Healing and Metabolic Status in Patients With Diabetic Foot UlcerDocument25 pages.60 Subyek 18 Mohseni Et Al 2017 - A Randomized, Double Placebo, Controlled Trial - The Effects of Probiotic Administration On Wound Healing and Metabolic Status in Patients With Diabetic Foot Ulcerbangd1f4nNo ratings yet
- Kerry Bone Covid 19 PresentationDocument27 pagesKerry Bone Covid 19 PresentationLukasNo ratings yet
- 2023 Nano Pharmaceuticals ArtigoDocument16 pages2023 Nano Pharmaceuticals ArtigoLuís Rodrigues da SilvaNo ratings yet
- Invitro Studies On The Effect of Adhatoda Vasica Nees. in Adipocyte 3T3 - L1 Cell LinesDocument15 pagesInvitro Studies On The Effect of Adhatoda Vasica Nees. in Adipocyte 3T3 - L1 Cell LinesDR. BALASUBRAMANIAN SATHYAMURTHYNo ratings yet
- Artigo - Dieta e Endometriose - 2024Document26 pagesArtigo - Dieta e Endometriose - 2024nutricionistataianeNo ratings yet
- G1 Alimentos Com Propriedades de Redução Do Risco de CâncerDocument13 pagesG1 Alimentos Com Propriedades de Redução Do Risco de CâncerAna PaulaNo ratings yet
- Nutrients: Mediterranean Diet and Soy Isoflavones For Integrated Management of The Menopausal Metabolic SyndromeDocument3 pagesNutrients: Mediterranean Diet and Soy Isoflavones For Integrated Management of The Menopausal Metabolic SyndromeAndrés ZapataNo ratings yet
- Plants: Effect of Propolis Paste and Mouthwash Formulation On Healing After Teeth Extraction in Periodontal DiseaseDocument12 pagesPlants: Effect of Propolis Paste and Mouthwash Formulation On Healing After Teeth Extraction in Periodontal DiseaseSalsabila Tri YunitaNo ratings yet
- The Bidirectional Relationship Between EndometriosDocument9 pagesThe Bidirectional Relationship Between EndometriossilsilviasilNo ratings yet
- Nutrition and Nutritional Supplementation PDFDocument5 pagesNutrition and Nutritional Supplementation PDFnanda agnessiaNo ratings yet
- endoperioDocument21 pagesendoperioLuis KabreraNo ratings yet
- Muhammad Abba Sheriff SCI/13/BCH/00339 ApplicationsofbiotechnologyandgeneticengineeringDocument5 pagesMuhammad Abba Sheriff SCI/13/BCH/00339 ApplicationsofbiotechnologyandgeneticengineeringKabir ShariffNo ratings yet
- Management of Puberty Associated Gingival Enlargement in An Adolescent Female - A Case Report With A Follow-Up of 1 YearDocument4 pagesManagement of Puberty Associated Gingival Enlargement in An Adolescent Female - A Case Report With A Follow-Up of 1 YearDha Dina SevofrationNo ratings yet
- Insulin Resistance and Periodontal BacteriaDocument27 pagesInsulin Resistance and Periodontal BacteriaramadhancunbertNo ratings yet
- Role of Dietary Edible Mushrooms in the Modulation of Gut MicrobiotaDocument12 pagesRole of Dietary Edible Mushrooms in the Modulation of Gut MicrobiotaMartinaNo ratings yet
- Understanding Insulin in The Age of Precision Medicine and Big Data - Under-Explored Nature of GenomicsDocument38 pagesUnderstanding Insulin in The Age of Precision Medicine and Big Data - Under-Explored Nature of GenomicsGestne AureNo ratings yet
- Aceite de Coco PeriodontalDocument8 pagesAceite de Coco PeriodontalErick PiñerosNo ratings yet
- Serum Vitamin D Levels in Pulmonary Tuberculosis: SJIF Impact Factor 6.647Document7 pagesSerum Vitamin D Levels in Pulmonary Tuberculosis: SJIF Impact Factor 6.647rendyjiwonoNo ratings yet
- Role of Antioxidant Vitamins and Other Micronutrients OnDocument19 pagesRole of Antioxidant Vitamins and Other Micronutrients OnMasna ArahmanNo ratings yet
- Periodontal Health and DiabetesDocument2 pagesPeriodontal Health and Diabetesesra gdNo ratings yet
- Uterine Fibroids and DietDocument15 pagesUterine Fibroids and DietELISABETE PIMENTELNo ratings yet
- Marton, Et Al ('10) - The Role of Sprouts in Human Nutrition - A Review (Marked)Document37 pagesMarton, Et Al ('10) - The Role of Sprouts in Human Nutrition - A Review (Marked)aida-69No ratings yet
- The Role of Intestinal Microbiota in Atopic.5Document5 pagesThe Role of Intestinal Microbiota in Atopic.5Rifqi arinaNo ratings yet
- Diet and Nutrients and ImmuneDocument21 pagesDiet and Nutrients and ImmuneKaty Carreño RojasNo ratings yet
- PsoriasisDocument382 pagesPsoriasisCindy_MayuryNo ratings yet
- Non-Surgical Periodontal Care TrendsDocument5 pagesNon-Surgical Periodontal Care TrendsRavi KumarNo ratings yet
- An Investigation Into The Prevalence and Treatment of Oral MucositisDocument11 pagesAn Investigation Into The Prevalence and Treatment of Oral MucositisHilya Aliva AufiaNo ratings yet
- Role of Biotechnology in Improving Human HealthDocument7 pagesRole of Biotechnology in Improving Human HealthdeepaksagarNo ratings yet
- Module 5 WPS OfficeDocument2 pagesModule 5 WPS OfficeJohn MontejoNo ratings yet
- Research Proposal: Role of Nurse in Delay Wound HealingDocument25 pagesResearch Proposal: Role of Nurse in Delay Wound HealingNaveeda IqbalNo ratings yet
- THE IMPORTANCE OF GUT HEALTH IN CANCER PREVENTION & THERAPYDocument33 pagesTHE IMPORTANCE OF GUT HEALTH IN CANCER PREVENTION & THERAPYNishi RuciNo ratings yet
- Nutrition and ImmunityFrom EverandNutrition and ImmunityMaryam MahmoudiNo ratings yet
- GutMicrobiotaHomeostasisRestoration and BCDocument8 pagesGutMicrobiotaHomeostasisRestoration and BCerdost450No ratings yet
- 3 PDFDocument8 pages3 PDFJethro Floyd QuintoNo ratings yet
- Deraya 2020 IOP Conf. Ser. Earth Environ. Sci. 457 012079Document8 pagesDeraya 2020 IOP Conf. Ser. Earth Environ. Sci. 457 012079Irwina eka derayaNo ratings yet
- Animal_Models_of_Mucositis_Implications_for_TherapDocument9 pagesAnimal_Models_of_Mucositis_Implications_for_TherapÁgnesJanovszkyNo ratings yet
- Russian Federation Country Codes from World Development IndicatorsDocument262 pagesRussian Federation Country Codes from World Development IndicatorsMuhammad Talha TalhaNo ratings yet
- Lentz Criminal ComplaintDocument16 pagesLentz Criminal ComplaintKristen Faith SchneiderNo ratings yet
- Conduct CardsDocument9 pagesConduct Cardsapi-248309459No ratings yet
- Accounting for Business CombinationsDocument52 pagesAccounting for Business CombinationsEliza BethNo ratings yet
- DVD S2300Document106 pagesDVD S2300cristakeNo ratings yet
- Upliftment of Recto AveDocument6 pagesUpliftment of Recto AveFrance CortezanoNo ratings yet
- Data Protection Act (DPA)Document14 pagesData Protection Act (DPA)Crypto SavageNo ratings yet
- Missing Number Series Questions Specially For Sbi Po PrelimsDocument18 pagesMissing Number Series Questions Specially For Sbi Po PrelimsKriti SinghaniaNo ratings yet
- Introduction To Major Crop FieldsDocument32 pagesIntroduction To Major Crop FieldsCHANDANINo ratings yet
- The 2012 FedEx Ketchum Social Business StudyDocument40 pagesThe 2012 FedEx Ketchum Social Business StudyEric PrenenNo ratings yet
- Vodacom Fibre - Application Form CPD - 23 August 2018Document8 pagesVodacom Fibre - Application Form CPD - 23 August 2018Mla Emmanuel ZondoNo ratings yet
- Event Management Study Material Free PDFDocument2 pagesEvent Management Study Material Free PDFKim0% (1)
- Seasonal Work Brochure 05Document2 pagesSeasonal Work Brochure 05R-lau R-pizNo ratings yet
- If You Are Wondering How To Get To Visayas and Mindanao by LandDocument7 pagesIf You Are Wondering How To Get To Visayas and Mindanao by LandHisoka HillingtonNo ratings yet
- Problem Solving 5Document4 pagesProblem Solving 5Raphael Pizarro ArceoNo ratings yet
- Complete HSE document kit for ISO 14001 & ISO 45001 certificationDocument8 pagesComplete HSE document kit for ISO 14001 & ISO 45001 certificationfaroz khanNo ratings yet
- Quiz ME 1Document4 pagesQuiz ME 1Radytia BimantaraNo ratings yet
- NHBC Standards 2011 Bre SD 1 SRCDocument5 pagesNHBC Standards 2011 Bre SD 1 SRCSuresh RaoNo ratings yet
- Central Council For Research in Ayurvedic Sciences: CCRAS Post Doctoral Fellowship Scheme (CCRAS PDF Scheme)Document2 pagesCentral Council For Research in Ayurvedic Sciences: CCRAS Post Doctoral Fellowship Scheme (CCRAS PDF Scheme)TUSHAR DASHNo ratings yet
- Unit 4:: Incident Commander and Command Staff FunctionsDocument16 pagesUnit 4:: Incident Commander and Command Staff FunctionsAntonio Intia IVNo ratings yet
- Global CityDocument3 pagesGlobal Citycr lamigoNo ratings yet
- OriginalDocument4 pagesOriginalJob ValleNo ratings yet
- Written Report Maneco FinalDocument31 pagesWritten Report Maneco FinalKymicah DesiertoNo ratings yet
- PTL Ls Programme HandbookDocument34 pagesPTL Ls Programme Handbooksalak946290No ratings yet
- Building C# Applications: Unit - 2Document25 pagesBuilding C# Applications: Unit - 2mgsumaNo ratings yet
- 12 December 1996Document116 pages12 December 1996Monitoring Times100% (1)
- Agfa CR 10 X User ManualDocument4 pagesAgfa CR 10 X User ManualpietrokoNo ratings yet
- TRAIN Briefing - Revised Personal Income Tax RatesDocument73 pagesTRAIN Briefing - Revised Personal Income Tax RatesElizaFaithEcleoNo ratings yet
- The Sssessential List of Microsoft Outlook Keyboard ShortcutsDocument3 pagesThe Sssessential List of Microsoft Outlook Keyboard Shortcutsabidaliabid1No ratings yet
- Genetic Modified FoodDocument16 pagesGenetic Modified FoodKishoNo ratings yet