Professional Documents
Culture Documents
Stars Academy Lahore: Stars Entry Test System - 2019
Uploaded by
Memoona GullOriginal Title
Copyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
Stars Academy Lahore: Stars Entry Test System - 2019
Uploaded by
Memoona GullCopyright:
Available Formats
STARS ACADEMY LAHORE
Head office: 30 Kashmir Block Allama Iqbal Town Lahore 042-37800293, 0333-1409100, www.starscollege.edu.pk
_____________________ _________________________
Roll No. of Candidate Name of Candidate
STARS ENTRY TEST SYSTEM – 2019
Assignment Code: C5 Session MDCAT Time Allowed: 40 mints
0C
1 The rate constant activation energy and Arrhenius parameter of a reaction at 25 are 3.0 x 10-4s-1,
104.4kJ/mol and 6.0 x
1014s-1respectively. The value of the rate constant at T →∞ is
A) 2.0 x 1018 xs-1 B) 6.0 x 1014s-1 C) 3.6 x 1030s-1 D) ∞
2 The rate constant for the reaction 2N2O5 → 4NO2 +O2 is 3.0 x 10 s-1 . If the rate is 2.4 x 10-5 molL-1,s-1 , the concentration in
-5
mol L-1 is
A) 0.8 B) 0.04 C) 1.2 D) 1.4
3 When A and B are mixed to give C and D , the reaction quotient Q at the initial stages of the reaction
A) Increase with time B) Decrease with time C) Is independent of time D) Is Zero
4 The rate law for the reaction RCl + NaOH → ROH + NaCl is given by rate = k[RCl]. The rate of reaction will be
A) Doubled on doubling the B) Halved on reducing the C) Increased on D) In affected on increasing
concentration of NaOH concentration of alkyl increasing the the temperature of
halide to one half temperature of reaction.
reaction
5 The rate constant of a first order reaction is dependent on
A) Time B) Temperature C) Concentration of D) Concentration of
reaction products
6 The half life period of a first order reaction is 10min . If the initial amount is 0.08mol L -1 and the concentration at some
instant is 0.01mol L-1 , then t =
A) 10min B) 30min C) 20min D) 40min
7 If the t1/2for a reaction is 4min , the time after the reaction is 99.9% completed is
A) 16min B) 8min C) 32mins D) 40min
8 For exothermic reaction, the product are at a lower energy level than the reactants and the decrease in potential energy
appear as _________________ of the product
A) Decrease in kinetic energy B) Increase in kinetic energy C) Decrease in thermal D) Increase in thermal
energy energy
9 If two initial concentration and times for reaction are given then which method is used to find out order of reaction
A) Method of hit and trial B) Method of large excess C) Differential method D) Half life method
basis
10 The half life period of a zero order reaction is
A) Proportional to initial B) Independent of C) Proportional to square D) Inversely proportional to
concentration concentration of of concentration initial concentration
reactants
11 Identify the correct statement
A) The rate of chemical B) The hydrolysis of ethyl C) For a first order D) For a second order
change is directly acetate in acidic medium reaction the rate of reaction the rate of
proportional to is a Secord order reaction reaction doubles as doubles as the
concentration the concentration of concentration of the
the reactant (s) reaction(s) doubles
doubles
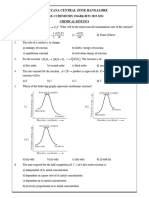
12 A plot of log k Vs 1/T of a reaction give a straight line with
A) A non zero intercept and B) A zero intercept and C) A non zero intercept D) A zero intercept and
negative slope negative slope and positive slope positive slope
13 The diazonium salt decomposes as C6H5N2+Cl-→ C6H5Cl + N2. At O0C , the evolution of N2 becomes two times faster when the
initial concentrating of the salt is doubled , Therefore it is
A) A first order reaction B) A second order reaction C) A zero order reaction D) Independent of the initial
concentration of the salt
14 The formation of nitrogen oxide is endothermic then enthalpy change of reaction is ……………….. KJ/mol
A) 41.6 B) 278.85 C) 285.58 d) 290.87
15 In the thermodynamics, a process is called reversible when
A) The surrounding and B) There is no boundary C) The surroundings are D) The system changes into
system change into each between system and always in equilibrium the surroundings
other surroundings with the system spontaneous
16 Which of the following statement is false
A) Work is a state function B) Temperature is a state C) Change in the state is D) Work appears at the
function completely defined boundary of the system
when the initial and
final states are
specified
17 Which of the following is an exothermic reaction
A) Conversion of graphite to B) Dehydrogenation of C) Decomposition of D) Combustion of methane
diamond ethane to ethylene water
18 The standard molar enthalpy of formation of CO2 is equal to
A) Zero B) The standard molar C) The standard molar D) The sum of the standard
enthalpy of combustion entropy of combustion molar enthalpies of CO
of carbon(graphite) of C(gaseous) and CO2
19 Which of the following equations correctly represents the standard heat of formation (∆ H0f) of methane?
A) C(diamond) + 2H2(g) = B) C(graphite) + 2H2(g) = C) C(graphite) + 2H2(g) = D) C(graphite) + 4H =
CH4(g) CH4(l) CH4(g) CH4(g)
20 The surface which separate system and surroundings is called …………………
A) Boundary B) Area C) Linc D) All of these
21 For an endothermic reaction where∆𝑯 represents the enthalpy of the reaction in kJ/mol, the maximum value for the energy
of activation will be
A) Less than ∆𝑯 B) More than ∆𝑯 C) Equal to ∆𝑯 D) Zero
22 The sum all kinds of energy of system is called internal energy. These energies are
A) Bouncing energy B) Elastic potential energy C) All kinds of K.E D) All of these
23 Which of the following reaction is an endothermic reaction?
A) 2H2 + O2 → 2H2O B) 2NaOH +H2SO4 → C) C2H5OH + 3O2→ D) N2 +O2→ 2NO
Na2SO4+2H2O 2CO2 + 3H2O
24 The enthalpy of combustion of cyclohexone, cyclohexanol and H2 is -3920, -3800 and -241 kJ/mol respectively. The heat of
hydrogenation (in kJ/mol) of cyclohexone is
A) -121 B) +121 C) -242 D) +242
25 The products of combustion of an aliphatic thiol (RHS) at 298k are
A) CO2 B) CO2 ,H2Og and SO2g C) CO2g , H2Og and D) CO2(g) and SO2(l)
SO2(g)
26 If pressure on the one mole gas is 2Nm-2 produced 3dm3 volume change and enthalpy of system is 9 J/mole the internal
energy of system is ……………………
A) 12 J/mole B) 6 J/mole C) 3 J/mole d) 9 J/mole
27 In what proportion 1M NaOH and 0.5M H2SO4 are mixed so as to release maximum amount of energy and to form 100ml
solution
A) 33 and 67 B) 67 and 33 C) 40 and 60 D) 50 and 50
28 The enthalpy of combustion of glucose is -2840kJ/mol. The enthalpy of formation of 60 g of glucose is
A) 2840kJ B) 946.6kJ C) -946.6KJ D) 1420kJ
29 The heat of reaction at constant pressure is given by
A) HR - HP B) EP-ER C) ER-EP D) HP-HR
30 The enthalpy change which occurs when the certain number of moles of reactants as indicated by balanced chemical
equation, reacts to form product of standard temperature and pressure is called ………………
A) Enthalpy of reaction B) Enthalpy of atomization C) Enthalpy of formation D) Enthalpy of combustion
TESTS ARE THE BEST FOR YOUR TEST
You might also like
- Senior - 2020 - Class - 12 - Chemistry - Objective Questions - Chemical KineticsDocument5 pagesSenior - 2020 - Class - 12 - Chemistry - Objective Questions - Chemical Kineticsblaise.denzil.rodriguesNo ratings yet
- Concordia Colleges BWP: 1 Year Chemistry Chapter#11Document2 pagesConcordia Colleges BWP: 1 Year Chemistry Chapter#11Hafiz ZainNo ratings yet
- Kinetics Ans Key Master FileDocument10 pagesKinetics Ans Key Master FileJOANA RHEA SAGPAEYNo ratings yet
- Chemical Kinetics FinalDocument7 pagesChemical Kinetics Finalaxiliya6No ratings yet
- (Cbse Board) - Inc-Sr-Cbse-Superchaina-Chemistry Key Q.P 30-03-2024Document8 pages(Cbse Board) - Inc-Sr-Cbse-Superchaina-Chemistry Key Q.P 30-03-2024akshayapraseedaNo ratings yet
- Chemical Kinetics 20-21Document5 pagesChemical Kinetics 20-21Mahek KatharotiyaNo ratings yet
- 1032 1st Exam 1040325 A PDFDocument8 pages1032 1st Exam 1040325 A PDFTrung TranNo ratings yet
- CHEMISTRY 12th SubejctiveDocument3 pagesCHEMISTRY 12th SubejctiveVivek SanwalNo ratings yet
- DPP 02Document1 pageDPP 02prathmfedNo ratings yet
- Cbse+2 Chemistry 1mark Bits 2023-2024Document41 pagesCbse+2 Chemistry 1mark Bits 2023-2024lama lamaNo ratings yet
- GZB - Xii - WS-8 - Chemistry - Chemical Kinetics - OctoberDocument3 pagesGZB - Xii - WS-8 - Chemistry - Chemical Kinetics - OctoberSaman PanwarNo ratings yet
- Practice Sheet, CH # 9, C-I, Chemical KineticsDocument5 pagesPractice Sheet, CH # 9, C-I, Chemical Kineticsfreeacount468No ratings yet
- Chem. Assig.Document8 pagesChem. Assig.aryan asliaNo ratings yet
- Practice Chapter 12 and 13 Chemistry TestDocument9 pagesPractice Chapter 12 and 13 Chemistry Testluis arauzNo ratings yet
- CH-4 Kinetics MaterialDocument18 pagesCH-4 Kinetics MaterialBishal MishraNo ratings yet
- Semester-6 3360503 CRE MCQ KRD PDFDocument9 pagesSemester-6 3360503 CRE MCQ KRD PDFDhruv RanaNo ratings yet
- Chemical Kinetics TestDocument5 pagesChemical Kinetics Testrajneesh kumarNo ratings yet
- Class-12 Chemistry ElectroDocument4 pagesClass-12 Chemistry ElectroHemant ChaudharyNo ratings yet
- Chapter 1 Solutions Multiple Choice QuestionsDocument29 pagesChapter 1 Solutions Multiple Choice Questionsjkc collegeNo ratings yet
- 16PCH1103Document22 pages16PCH1103MoneeshsabapathiNo ratings yet
- CHEMICAL KINETICS FinalDocument6 pagesCHEMICAL KINETICS FinalBOTHRA CLASSESNo ratings yet
- Chemistry (Maninagar-Target) Section-I (Only One Option Correct)Document4 pagesChemistry (Maninagar-Target) Section-I (Only One Option Correct)Rajeev GangwarNo ratings yet
- End Term ALLDocument31 pagesEnd Term ALLJulie Anne CristalesNo ratings yet
- Chemical Kinetics Chap 8Document2 pagesChemical Kinetics Chap 8Eliza BethNo ratings yet
- Chemical Kinetics Class 12: Rates, Orders, and EnergyDocument2 pagesChemical Kinetics Class 12: Rates, Orders, and EnergyShubham KumarNo ratings yet
- CRE - Diagnostic Exam (USA)Document2 pagesCRE - Diagnostic Exam (USA)Kuo SarongNo ratings yet
- Chemical Kinetics Worksheet Online PDFDocument7 pagesChemical Kinetics Worksheet Online PDFRISHIKESH SHIRSATHNo ratings yet
- Chemical Kinetic - Dec2016 PDFDocument137 pagesChemical Kinetic - Dec2016 PDFFaisal AzamNo ratings yet
- Mid_term_general_chem_II_fall_2001Document6 pagesMid_term_general_chem_II_fall_2001dr.ibrahimsalemvpNo ratings yet
- MCD4390 Week 10 Tutorial QuestionsDocument5 pagesMCD4390 Week 10 Tutorial QuestionsGabbar100% (1)
- Adobe Scan 16 Nov 2022Document16 pagesAdobe Scan 16 Nov 2022Shaik mohammed NizamuddinNo ratings yet
- h2 2 Midterm2 ch2Document6 pagesh2 2 Midterm2 ch2api-263846648No ratings yet
- Class XII Chemical KineticsDocument6 pagesClass XII Chemical KineticsvartikasinghNo ratings yet
- Shalini Memorial School: SC O LDocument2 pagesShalini Memorial School: SC O LPŕį Ņćę ĂãťîfNo ratings yet
- Chemical KineticsDocument4 pagesChemical KineticsShubhankar SinhaNo ratings yet
- Chemical Kinetics JEE MAINS 2022 - 966591 - 2022 - 08 - 19 - 16 - 17Document8 pagesChemical Kinetics JEE MAINS 2022 - 966591 - 2022 - 08 - 19 - 16 - 17AbhinavNo ratings yet
- Xi Chem 13.01.24Document2 pagesXi Chem 13.01.24faraazahmed70058No ratings yet
- MCH 05 2020 April Final Exam Option OneDocument20 pagesMCH 05 2020 April Final Exam Option Onepaul kapambweNo ratings yet
- JEE - Chemistry - Chemical KineticsDocument27 pagesJEE - Chemistry - Chemical Kineticsdaiwikchilukuri321No ratings yet
- XII - Revision Sheet - 2 - ChemistryDocument3 pagesXII - Revision Sheet - 2 - ChemistryVipin VNo ratings yet
- Chemical Kinetics Class 12Document137 pagesChemical Kinetics Class 12Liyutsa ZirangeNo ratings yet
- 4 - Chemical Kinetics & RadioactivityDocument19 pages4 - Chemical Kinetics & RadioactivityNimeshNo ratings yet
- PLTL Ch. 16 AssignmentDocument6 pagesPLTL Ch. 16 AssignmentJules BrunoNo ratings yet
- 2012EC 2 Semester 3 Quarter Chemistry Worksheet For Grade 11. I. Choose The Best Answer From The Given AlternativesDocument5 pages2012EC 2 Semester 3 Quarter Chemistry Worksheet For Grade 11. I. Choose The Best Answer From The Given AlternativesPatrix ParkerNo ratings yet
- QuestionsDocument22 pagesQuestionsTriciaMichi HoNo ratings yet
- @bohring - Bot CHEMICAL KINETICS ASSIGNMENT @HeyitsyashXDDocument77 pages@bohring - Bot CHEMICAL KINETICS ASSIGNMENT @HeyitsyashXDxkryxxzNo ratings yet
- Chemical Sciences: Paper IIDocument12 pagesChemical Sciences: Paper IIRavikanthNo ratings yet
- Kinetics II (Multiple Choice) QPDocument10 pagesKinetics II (Multiple Choice) QPMZWAANo ratings yet
- Rate of Reaction: Key Concepts ExplainedDocument73 pagesRate of Reaction: Key Concepts ExplainedLiyutsa ZirangeNo ratings yet
- CH 12 Prac Test Kinetics1Document15 pagesCH 12 Prac Test Kinetics1NolemNo ratings yet
- Test ReviewDocument3 pagesTest ReviewgraceNo ratings yet
- CHNG 3004 - 2019-2020 AssignmentsDocument26 pagesCHNG 3004 - 2019-2020 AssignmentsXheikhKaleem100% (1)
- 3 - QP - Chemical KineticsDocument5 pages3 - QP - Chemical Kineticsssheeladevi84No ratings yet
- Chem Class Xi-2022Document7 pagesChem Class Xi-2022Gourav SwainNo ratings yet
- Chemistry: Crash Course For JEE Main 2020Document17 pagesChemistry: Crash Course For JEE Main 2020QSQFNo ratings yet
- 1.chemical KineticsDocument24 pages1.chemical KineticsVinod AgrawalNo ratings yet
- (Section A) - Answer The Following For 1 Mark.Document6 pages(Section A) - Answer The Following For 1 Mark.vivek daveNo ratings yet
- Kinetics Lec-1 NEET ChalisaDocument35 pagesKinetics Lec-1 NEET Chalisaashustarguy005No ratings yet
- Chemical Kinetics PYQDocument9 pagesChemical Kinetics PYQAmresh MohantyNo ratings yet
- Physics: © The PreceptorDocument10 pagesPhysics: © The PreceptorAsad RehanNo ratings yet
- 2 BhattiAcademy - Com Chemistry 3. Azeem Acadmy (Subjective)Document10 pages2 BhattiAcademy - Com Chemistry 3. Azeem Acadmy (Subjective)Memoona GullNo ratings yet
- Physics:: AnswersDocument3 pagesPhysics:: AnswersMuhammad Zaheer KhalilNo ratings yet
- Physics: © The PreceptorDocument12 pagesPhysics: © The PreceptorMemoona GullNo ratings yet
- © The Preceptor: PhysicsDocument12 pages© The Preceptor: PhysicsAitazaz AhsanNo ratings yet
- 10 BhattiAcademy - Com Chemistry 7. Unique (Obj)Document7 pages10 BhattiAcademy - Com Chemistry 7. Unique (Obj)Memoona GullNo ratings yet
- Physics:: AnswersDocument3 pagesPhysics:: AnswersMuhammad Zaheer KhalilNo ratings yet
- Mdcat/Lmdcat (Phase I) Virtual at Kipslms Scheme of Studies (LMS)Document6 pagesMdcat/Lmdcat (Phase I) Virtual at Kipslms Scheme of Studies (LMS)Memoona GullNo ratings yet
- Stars Academy Lahore: Stars Entry Test SystemDocument2 pagesStars Academy Lahore: Stars Entry Test SystemMemoona GullNo ratings yet
- C1 Home AssignmentDocument2 pagesC1 Home AssignmentMemoona GullNo ratings yet
- C1 Home AssignmentDocument2 pagesC1 Home AssignmentMemoona GullNo ratings yet
- P6 Home AssignmentDocument5 pagesP6 Home AssignmentMemoona GullNo ratings yet
- Second Year: English Book-II Part-IDocument22 pagesSecond Year: English Book-II Part-IMuhammad Tayyab IqbalNo ratings yet
- NUMS/NMDCAT Additional Topics ScheduleDocument9 pagesNUMS/NMDCAT Additional Topics ScheduleMemoona GullNo ratings yet
- MDCAT REPEATER SESSIONDocument1 pageMDCAT REPEATER SESSIONMemoona GullNo ratings yet
- P6 Home AssignmentDocument5 pagesP6 Home AssignmentMemoona GullNo ratings yet
- Stars Academy Lahore Entry Test SystemDocument2 pagesStars Academy Lahore Entry Test SystemMemoona GullNo ratings yet
- C1 Home AssignmentDocument2 pagesC1 Home AssignmentMemoona GullNo ratings yet
- B3 Home AssignmentDocument2 pagesB3 Home AssignmentMemoona GullNo ratings yet
- P4 Home AssignmentDocument2 pagesP4 Home AssignmentMemoona GullNo ratings yet
- P6 Home AssignmentDocument5 pagesP6 Home AssignmentMemoona GullNo ratings yet
- Entry Test Scheme of Studies MDCAT 2020: HomeostasisDocument4 pagesEntry Test Scheme of Studies MDCAT 2020: HomeostasisCaptain MesencephalonNo ratings yet
- B2 Home AssignmentDocument2 pagesB2 Home AssignmentMemoona GullNo ratings yet
- Stars Entry Test System 2019Document2 pagesStars Entry Test System 2019Memoona GullNo ratings yet
- Stars Academy Lahore Entry Test SystemDocument2 pagesStars Academy Lahore Entry Test SystemMemoona GullNo ratings yet
- C6 Home AssignmentDocument4 pagesC6 Home AssignmentMemoona GullNo ratings yet
- Step Test #12Document25 pagesStep Test #12Memoona GullNo ratings yet
- C1 Home AssignmentDocument2 pagesC1 Home AssignmentMemoona GullNo ratings yet
- C3 Home AssignmentDocument2 pagesC3 Home AssignmentMemoona GullNo ratings yet
- DPG10 Power Choke TesterDocument2 pagesDPG10 Power Choke TesterJawadi HamdiNo ratings yet
- Fuji Electric RSA30LCC + ROR30LCTservice ManualDocument27 pagesFuji Electric RSA30LCC + ROR30LCTservice ManualMaurizio DumitracheNo ratings yet
- 03 Definition - Classification of Power System Stability IEEE-CIGRE Joint Task Force On Stability Terms and DefinitionsDocument63 pages03 Definition - Classification of Power System Stability IEEE-CIGRE Joint Task Force On Stability Terms and DefinitionsRosa Elvira Montalvo MartínezNo ratings yet
- How To Determine Correct Number of Earthing Electrodes Strips Plates and Pipes Part 1Document6 pagesHow To Determine Correct Number of Earthing Electrodes Strips Plates and Pipes Part 1Francisco MartinezNo ratings yet
- Word Games in MechanicsDocument4 pagesWord Games in MechanicsJeazel MosendoNo ratings yet
- M1000230AC5MDocument1 pageM1000230AC5MPlant Site KMI-KJANo ratings yet
- Enidine Shock 2007Document120 pagesEnidine Shock 2007sveznalicamozesNo ratings yet
- The Forever Rechargeable VARIABLE Super Capacitor Battery !!!Document11 pagesThe Forever Rechargeable VARIABLE Super Capacitor Battery !!!shadyghanemNo ratings yet
- Time Rates ExplainedDocument6 pagesTime Rates ExplainedAljohn Escalona100% (1)
- Voltage Drop Calculation-TransformerDocument2 pagesVoltage Drop Calculation-TransformerLPKumar290% (1)
- Dem Unit 1 2 3 4 5Document17 pagesDem Unit 1 2 3 4 5Revathy KannanNo ratings yet
- Finalreport SwetasinghDocument19 pagesFinalreport Swetasinghsaurav kumarNo ratings yet
- ( (Premier Batch: Problem Solving DQG ( (HuflvhsheetDocument31 pages( (Premier Batch: Problem Solving DQG ( (HuflvhsheetAYUSH GUPTANo ratings yet
- Lightning Electromagnetic Fields and Their Induced Voltages On Overhead Lines: The Effect of A Non-Flat Lossy GroundDocument4 pagesLightning Electromagnetic Fields and Their Induced Voltages On Overhead Lines: The Effect of A Non-Flat Lossy GroundJavad PaknahadNo ratings yet
- Cart Brake Material Selection and Stress CalculationDocument5 pagesCart Brake Material Selection and Stress CalculationHailemariam WeldegebralNo ratings yet
- Bifacial Dual Glass: Monocrystalline ModuleDocument2 pagesBifacial Dual Glass: Monocrystalline ModuleJose VicenteNo ratings yet
- UP33TOP140KGAAZ01B - Datasheet - Garun 40KL - DatasheetDocument2 pagesUP33TOP140KGAAZ01B - Datasheet - Garun 40KL - DatasheetAnton TonchevNo ratings yet
- 8.2 Aerodynamics 8.2.1: Airflow Around A BodyDocument6 pages8.2 Aerodynamics 8.2.1: Airflow Around A BodyNayem Hashan NayemNo ratings yet
- Data Sheet - Kodaki - 2.5kW6.0kWDocument2 pagesData Sheet - Kodaki - 2.5kW6.0kWmustang460No ratings yet
- Magnetic Force MicrosDocument15 pagesMagnetic Force MicrosArvindGovaNo ratings yet
- S.N. Cat No. Qty. Description Quoted MRP J 1117 (Final Excel Sheet Not Recevied Till Date)Document2,412 pagesS.N. Cat No. Qty. Description Quoted MRP J 1117 (Final Excel Sheet Not Recevied Till Date)Amit SharmaNo ratings yet
- AP Physics Work Energy Power PDFDocument17 pagesAP Physics Work Energy Power PDFAli khan7No ratings yet
- Turbine MaintainanceDocument9 pagesTurbine Maintainanceer_sanjaypatel100% (2)
- Thermal Mass, Insulation and Ventilation in Sustainable Housing - An Investigation Across Climate and OccupancyDocument9 pagesThermal Mass, Insulation and Ventilation in Sustainable Housing - An Investigation Across Climate and OccupancySérgio GnipperNo ratings yet
- Characteristics of Ideal Op-AmpDocument14 pagesCharacteristics of Ideal Op-AmpShubham MannaNo ratings yet
- Abb - 1sda054321r1 PDFDocument3 pagesAbb - 1sda054321r1 PDFInnov8 OilNo ratings yet
- The Effect of Collector Aspect Ratio On The Collector Efficiency of Flat-Plate Solar Air HeatersDocument7 pagesThe Effect of Collector Aspect Ratio On The Collector Efficiency of Flat-Plate Solar Air Heatersaxel_oscNo ratings yet
- Bearing Types and Concept of FrictionDocument29 pagesBearing Types and Concept of Frictionjamesv52_743942786No ratings yet
- Magneto-Hydro Dynamic Generator: Hannes AlfvénDocument34 pagesMagneto-Hydro Dynamic Generator: Hannes AlfvénHuzaifa WasimNo ratings yet
- How Much Ac Ripple in A DC Power Supply Is Too Much?: Editor'S Note: Pdfs of This Article AreDocument2 pagesHow Much Ac Ripple in A DC Power Supply Is Too Much?: Editor'S Note: Pdfs of This Article Areparvinder ranaNo ratings yet