Professional Documents
Culture Documents
Q1 FY20 Press Presentation Highlights Revenue Growth
Uploaded by
Parvathi M LOriginal Description:
Original Title
Copyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
Q1 FY20 Press Presentation Highlights Revenue Growth
Uploaded by
Parvathi M LCopyright:
Available Formats
Press Presentation – Q1 FY20
1 | July 29, 2019 | Q1 FY20 Press Meet
Safe Harbor Statement
This presentation contains forward-looking statements and information that involve risks, uncertainties and assumptions. Forward -looking statements are all statements
that concern plans, objectives, goals, strategies, future events or performance and underlying assumptions and other statemen ts that are other than statements of
historical fact, including, but not limited to, those that are identified by the use of words such as “anticipates”, “believes”, “estimates”, “expects”, “intends”, “plans”,
“predicts”, “projects” and similar expressions. Risks and uncertainties that could affect us include, without limitation:
General economic and business conditions in India and other key global markets in which we operate;
The ability to successfully implement our strategy, our research and development efforts, growth & expansion plans and techno logical changes;
Changes in the value of the Rupee and other currency changes;
Changes in the Indian and international interest rates;
Allocations of funds by the Governments in our key global markets;
Changes in laws and regulations that apply to our customers, suppliers, and the pharmaceutical industry;
Increasing competition in and the conditions of our customers, suppliers and the pharmaceutical industry; and
Changes in political conditions in India and in our key global markets.
Should one or more of such risks and uncertainties materialize, or should any underlying assumption prove incorrect, actual o utcomes may vary materially from those
indicated in the applicable forward-looking statements.
For more detailed information on the risks and uncertainties associated with the Company’s business activities, please see th e company’s annual report filed in Form 20-F
with the US SEC for the fiscal year ended March 31, 2019 and quarterly financial statements filed in Form 6-K with the US SEC for the quarters ended June 30, 2018,
Sep 30, 2018, December 31, 2018 and our other filings with US SEC. Any forward-looking statement or information contained in this presentation speaks only as of the
date of the statement. We are not required to update any such statement or information to either reflect events or circumstan ces that occur after the date the statement
or information is made or to account for unanticipated events.
2 | July 29, 2019 | Q1 FY20 Press Meet
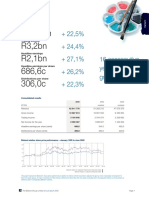
₹ Cr
Financial highlights
Q1 FY20 QoQ Gr% YoY Gr%
Revenues 3,844 -4% 3%
EBITDA* 1,134 29% 41%
PBT* 850 45% 70%
PAT* 663 53% 45%
* Includes Rs. 346 cr received from Celgene towards settlement of any claim the Company or its affiliates may have had for damages under section 8 of the Canadian Patented
Medicines (Notice of Compliance) Regulations in regard to the Company’s ANDS for a generic version of REVLIMID brand capsules , (Lenalidomide) pending before Health Canada
3 | July 29, 2019 | Q1 FY20 Press Meet
₹ Cr
P&L Metrics
Gross margin SG&A Expenses R&D Expenses
2,105 1,238 416
2,073 1,211 1,207
1,986 366 361
55.7% 52.4% 51.7% 32.5% 30.8% 31.4% 11.2% 9.1% 9.4%
Q1 Q4 Q1 Q1 Q4 Q1 Q1 Q4 Q1
FY19 FY19 FY20 FY19 FY19 FY20 FY19 FY19 FY20
4 | July 29, 2019 | Q1 FY20 Press Meet
North America
Revenues
Revenue growth driven by contribution from new products and
Q1 YoY Gr 3%
₹ 1,632cr volume traction; partly offset by price erosion in some key
FY20 QoQ Gr 9%
molecules
5 new products launched during the quarter - Daptomycin,
Testosterone gel, Tobramycin, Vitamin K & OTC calcium
carbonate
Re-launched Isotretinoin
U.S. Generics filing update:
1 ANDA filed with the US FDA in this quarter
107 pending approval (104 ANDAs + 3 NDAs)
58 Para IV filings and we believe 34 have first to file status
5 | July 29, 2019 | Q1 FY20 Press Meet
India
Revenues
Growth driven by new products and growth in base business
Q1 YoY Gr 15% We launched 8 brands during the quarter including Cetzine, Ubil &
₹ 696cr
FY20 QoQ Gr 7% Trisertain
As per IQVIA, Dr. Reddy’s growth better than IPM on both MQT &
MAT basis
IQVIA IMS growth rates
June 2019 MQT MAT
IPM 10.4% 10.4%
Dr. Reddy’s 13.0% 12.2%
6 | July 29, 2019 | Q1 FY20 Press Meet
Emerging Markets
Revenues
₹ Cr
Region Q1’20 YoY Gr QoQ Gr
Q1 YoY Gr 10% Russia 396 5% 10%
₹ 730cr
FY20 QoQ Gr 4% CISR 121 2% 2%
RoW 213 27% (4)%
EM 730 10% 4%
Russia: Growth largely driven by new launches and better realizations in
some of the key molecules
CISR: Growth driven by new products launched in this quarter
RoW: YoY growth aided by new products & volume traction in base business
7 | July 29, 2019 | Q1 FY20 Press Meet
Other Businesses ₹ Cr
Europe PSAI
YoY Gr QoQ Gr YoY Gr QoQ Gr
19% 26% (16)% (33)%
677
240
202 191 541
454
Q1 Q4 Q1 Q1 Q4 Q1
FY19 FY19 FY20 FY19 FY19 FY20
Growth aided by new products and volume Decline in the sales volume of certain products
traction on account of improvement in supplies
8 | July 29, 2019 | Q1 FY20 Press Meet
Capex, R&D & free cash flows
R&D expenses (` Cr) Capex (` Cr)
1,783 1,955 1,826 1,202 1,228
1,561 925
696
106
361
FY16 FY17 FY18 FY19 Q1 FY16 FY17 FY18 FY19 Q1
FY20 FY20
Free cash flow (` Cr) Net Debt / Equity
2,827 0.26
2,165 0.24
794 605 850 0.13
0.09
0.04
FY16 FY17 FY18 FY19 Q1
FY20 Mar-18 Sep-18 Dec-18 Mar-19 Jun-19
FCF as above is before acquisition related pay-outs
9 | July 29, 2019 | Q1 FY20 Press Meet
Key Priorities
Achieve market
Resolution of Build healthy
leading growth
CTO 6 Warning pipeline of
across
Letter products
businesses
Continue with Drive
the productivity innovation to
improvements deliver value
10 | July 29, 2019 | Q1 FY20 Press Meet
11 | July 29, 2019 | Q1 FY20 Press Meet
You might also like
- De Lima vs. Guerrero (843 SCRA 1, 20 Oct. 2017)Document101 pagesDe Lima vs. Guerrero (843 SCRA 1, 20 Oct. 2017)Eugene DayanNo ratings yet
- Acebedo Optical Vs CADocument1 pageAcebedo Optical Vs CAJane EnriquezNo ratings yet
- Simpsons - String PDFDocument6 pagesSimpsons - String PDFPaola RussoNo ratings yet
- Agronomy Crop Production (CH)Document45 pagesAgronomy Crop Production (CH)Parvathi M LNo ratings yet
- MCQ NegoDocument20 pagesMCQ NegoMark Hiro NakagawaNo ratings yet
- Plata v. YatcoDocument2 pagesPlata v. YatcoShalom MangalindanNo ratings yet
- Compensation Strategy of Bharti AirtelDocument17 pagesCompensation Strategy of Bharti AirtelParvathi M LNo ratings yet
- (CPR) Collantes v. MabutiDocument5 pages(CPR) Collantes v. MabutiChristian Edward Coronado50% (2)
- Press Presentation q2 Fy 20Document11 pagesPress Presentation q2 Fy 20hau vuvanNo ratings yet
- Press Presentation q1 Fy21Document13 pagesPress Presentation q1 Fy21Pharma researchNo ratings yet
- Q3 FY21 Financial Results Highlight Revenue GrowthDocument13 pagesQ3 FY21 Financial Results Highlight Revenue GrowthRajesh KNo ratings yet
- V-Guard Q1 FY2019 Earnings Presentation Highlights Revenue Growth of 19Document22 pagesV-Guard Q1 FY2019 Earnings Presentation Highlights Revenue Growth of 19Faizan AnsariNo ratings yet
- FY21 Q2 Financial Results: Rs 4,897 Cr Revenue, Rs 762 Cr PATDocument13 pagesFY21 Q2 Financial Results: Rs 4,897 Cr Revenue, Rs 762 Cr PATPharma researchNo ratings yet
- Arihant May PDFDocument4 pagesArihant May PDFBook MonkNo ratings yet
- V-Guard Q1 FY20 Earnings Presentation Highlights Strong GrowthDocument21 pagesV-Guard Q1 FY20 Earnings Presentation Highlights Strong GrowthSelva ThanarajNo ratings yet
- Piramal EnterprisesDocument54 pagesPiramal EnterprisesswarnaNo ratings yet
- V Guard Industries Q4 Results PresentationDocument23 pagesV Guard Industries Q4 Results PresentationIlyasNo ratings yet
- ITC Limited: Ratings Reaffirmed: Summary of Rating ActionDocument6 pagesITC Limited: Ratings Reaffirmed: Summary of Rating ActionSatish RajNo ratings yet
- BKT InvestorPresentation March2020Document34 pagesBKT InvestorPresentation March2020Rina JageNo ratings yet
- BSE Limited The National Stock Exchange of India LimitedDocument31 pagesBSE Limited The National Stock Exchange of India LimitedNic KnightNo ratings yet
- WPP 2020 First Quarter Trading Statement PresentationDocument33 pagesWPP 2020 First Quarter Trading Statement PresentationJuan Carlos Garcia SanchezNo ratings yet
- Reliance Capital (RELCAP) : Businesses Growing, Return Ratios ImprovingDocument15 pagesReliance Capital (RELCAP) : Businesses Growing, Return Ratios Improvingratan203No ratings yet
- Britannia Industries Q1FY18 ResultsDocument8 pagesBritannia Industries Q1FY18 ResultssandyinsNo ratings yet
- Cyient LTD.: Q1FY20: Weak Quarter, Lack VisibilityDocument7 pagesCyient LTD.: Q1FY20: Weak Quarter, Lack VisibilityADNo ratings yet
- MDA YE 2019 EN - Vfinal - RevisedDocument19 pagesMDA YE 2019 EN - Vfinal - Revisedredevils86No ratings yet
- FY10 Earnings & Strategy Update Gy P To Shareholders: August 6, 2010 August 6, 2010Document31 pagesFY10 Earnings & Strategy Update Gy P To Shareholders: August 6, 2010 August 6, 2010Shankar KosuruNo ratings yet
- DABUR INDIA LIMITED INVESTOR COMMUNICATION Q1 FY20 HIGHLIGHTSDocument23 pagesDABUR INDIA LIMITED INVESTOR COMMUNICATION Q1 FY20 HIGHLIGHTSarti guptaNo ratings yet
- Sun Pharma 4QFY19 Results UpdateDocument10 pagesSun Pharma 4QFY19 Results UpdateHarshul BansalNo ratings yet
- October 19, 2022Document39 pagesOctober 19, 2022Indraneel MahantiNo ratings yet
- Pidilite CompanyDocument6 pagesPidilite Companykhalsa.taranjitNo ratings yet
- Aarti Industries Q1 FY22 Results Present Record PerformanceDocument24 pagesAarti Industries Q1 FY22 Results Present Record PerformancerohitnagrajNo ratings yet
- April 2022Document43 pagesApril 2022Indraneel MahantiNo ratings yet
- Dabur India LTD: CMP: Rs 492 Rating: HOLD Target Price: Rs 515Document4 pagesDabur India LTD: CMP: Rs 492 Rating: HOLD Target Price: Rs 515Book MonkNo ratings yet
- ITC Results ReviewDocument13 pagesITC Results ReviewSivas SubramaniyanNo ratings yet
- HCL Tech q1 2019 Investor Release PDFDocument23 pagesHCL Tech q1 2019 Investor Release PDFmanishaNo ratings yet
- NomuraSecuritiesCoLtd QuickNote-FortisHealthcare (FORHIN) (Buy) - 2QFY23Stronghospitalbusiness Nov 14 2022Document8 pagesNomuraSecuritiesCoLtd QuickNote-FortisHealthcare (FORHIN) (Buy) - 2QFY23Stronghospitalbusiness Nov 14 2022Gaurav AhirkarNo ratings yet
- Redginton Q4 FY22Document26 pagesRedginton Q4 FY22nitin2khNo ratings yet
- BCSL Investor Meet - 29.05.2020 - Final For BSE Upload - 0 PDFDocument22 pagesBCSL Investor Meet - 29.05.2020 - Final For BSE Upload - 0 PDFPruthviraj DeshmukhNo ratings yet
- Galaxy Surfactants Limited: Niranjan Arun KetkarDocument41 pagesGalaxy Surfactants Limited: Niranjan Arun KetkarparthchillNo ratings yet
- Albermarle November 2022 Investor Presentation v04Document39 pagesAlbermarle November 2022 Investor Presentation v04Guillaume De SouzaNo ratings yet
- Results-Presentation 2019Document32 pagesResults-Presentation 2019Shahin AlamNo ratings yet
- V-Guard-Industries - Q4 FY21-Results-PresentationDocument17 pagesV-Guard-Industries - Q4 FY21-Results-PresentationanooppattazhyNo ratings yet
- Iiww 161210Document4 pagesIiww 161210rinku23patilNo ratings yet
- V Guard Industries Q3 FY22 Results PresentationDocument17 pagesV Guard Industries Q3 FY22 Results PresentationRATHINo ratings yet
- LPPF 4q18 - NomuraDocument6 pagesLPPF 4q18 - Nomurakrisyanto krisyantoNo ratings yet
- RelianceJan2024Document56 pagesRelianceJan2024jaikumar608jainNo ratings yet
- Press ReleaseDocument4 pagesPress ReleaseNaveen AnchanNo ratings yet
- Third Quarter 2020 Earnings Results Presentation: October 14, 2020Document16 pagesThird Quarter 2020 Earnings Results Presentation: October 14, 2020Zerohedge100% (1)
- Dabur India's strong domestic volume growth lifts revenues 18Document9 pagesDabur India's strong domestic volume growth lifts revenues 18Raghavendra Pratap SinghNo ratings yet
- Generic Engineering PreesentationDocument50 pagesGeneric Engineering Preesentationshailesh1978kNo ratings yet
- JM Fy2019Document35 pagesJM Fy2019Krishna ParikhNo ratings yet
- Parag Milk Foods Limited: Investor Presentation August 2019Document46 pagesParag Milk Foods Limited: Investor Presentation August 2019Raju GummaNo ratings yet
- KPITTECH 03082020160338 KPITInvestorUpdateSEuploadDocument28 pagesKPITTECH 03082020160338 KPITInvestorUpdateSEuploadSreenivasulu Reddy SanamNo ratings yet
- Axiata Group Berhad: 4Q 2019 ResultsDocument39 pagesAxiata Group Berhad: 4Q 2019 ResultsFarhadNo ratings yet
- ITC Limited: Resilient Quarter Amidst Weak Industry DemandDocument5 pagesITC Limited: Resilient Quarter Amidst Weak Industry DemandGeebeeNo ratings yet
- Britannia Industries - Q1FY20 Result Update - Centrum 10082019Document12 pagesBritannia Industries - Q1FY20 Result Update - Centrum 10082019darshanmaldeNo ratings yet
- GSK Q3 2020 Results SummaryDocument65 pagesGSK Q3 2020 Results SummarymarceloNo ratings yet
- Syngene 3Document44 pagesSyngene 3Puneet KapoorNo ratings yet
- Third Quarter Results FY 2019: Investor ReleaseDocument15 pagesThird Quarter Results FY 2019: Investor Releasevikalp123123No ratings yet
- Presentation To Investors: First Nine Months 2020 ResultsDocument27 pagesPresentation To Investors: First Nine Months 2020 ResultsMiguel García MirandaNo ratings yet
- January 19, 2022: Priyadarshi NI MahapatraDocument43 pagesJanuary 19, 2022: Priyadarshi NI MahapatraPuneet KapoorNo ratings yet
- 2010 First Quarter Business Review: (Unaudited)Document19 pages2010 First Quarter Business Review: (Unaudited)qtipxNo ratings yet
- S E Intimation Earnings Presentation 31.03.2023Document25 pagesS E Intimation Earnings Presentation 31.03.2023dinesh suresh KadamNo ratings yet
- Ankur JainDocument34 pagesAnkur Jainorganic.machliNo ratings yet
- Firstsource Solutions Limited: Q1 FY2019 Earnings UpdateDocument10 pagesFirstsource Solutions Limited: Q1 FY2019 Earnings UpdateCA EwapNo ratings yet
- Cipla Q3FY23 Press ReleaseDocument4 pagesCipla Q3FY23 Press ReleaseMantu HugarNo ratings yet
- DCB Bank Q4 FY 2019 Investor PresentationDocument35 pagesDCB Bank Q4 FY 2019 Investor PresentationrohanNo ratings yet
- A Comparative Analysis of Tax Administration in Asia and the Pacific: Fifth EditionFrom EverandA Comparative Analysis of Tax Administration in Asia and the Pacific: Fifth EditionNo ratings yet
- Easy Ways To Remember All The Current Affairs (CH)Document1 pageEasy Ways To Remember All The Current Affairs (CH)Parvathi M LNo ratings yet
- Defence and SecurityDocument9 pagesDefence and SecurityParvathi M LNo ratings yet
- Case: Eureka Forbes LTD.: Managing The Sales EffortDocument8 pagesCase: Eureka Forbes LTD.: Managing The Sales EffortParvathi M LNo ratings yet
- Yearly Current Affairs PDF (CH)Document2 pagesYearly Current Affairs PDF (CH)Parvathi M LNo ratings yet
- Current: AffairsDocument55 pagesCurrent: AffairsAnagh TiwariNo ratings yet
- Yearly Current Affairs PDF (CH)Document2 pagesYearly Current Affairs PDF (CH)Parvathi M LNo ratings yet
- Assignment 1: Risk Management Banking Financial IndustryDocument2 pagesAssignment 1: Risk Management Banking Financial IndustryParvathi M LNo ratings yet
- CMDocument1 pageCMParvathi M LNo ratings yet
- LearningDocument1 pageLearningParvathi M LNo ratings yet
- Analyze Financial Ratios & DuPont AnalysisDocument2 pagesAnalyze Financial Ratios & DuPont AnalysisParvathi M LNo ratings yet
- LearningsDocument3 pagesLearningsParvathi M LNo ratings yet
- Chairman Joseph C. Wilson, His Successor KearnsDocument2 pagesChairman Joseph C. Wilson, His Successor KearnsParvathi M LNo ratings yet
- SalaryDocument3 pagesSalaryParvathi M LNo ratings yet
- Jefferies Global Healthcare Conference 2016: Alok Sonig EVP & Head North America GenericsDocument32 pagesJefferies Global Healthcare Conference 2016: Alok Sonig EVP & Head North America GenericsDevendra JainNo ratings yet
- Reliance Jio Industry, Market and SWOT AnalysisDocument30 pagesReliance Jio Industry, Market and SWOT AnalysisParvathi M LNo ratings yet
- Stock Comp 2021Document13 pagesStock Comp 2021Parvathi M L0% (1)
- LearningsDocument1 pageLearningsParvathi M LNo ratings yet
- Project Assets Class Calculation 20105Document34 pagesProject Assets Class Calculation 20105Parvathi M LNo ratings yet
- Answer CasesDocument9 pagesAnswer CasesSafayetChowdhury67% (3)
- Q2Document6 pagesQ2Parvathi M LNo ratings yet
- A Comparative Analysis of Returns of Various Fi Nancial Asset Classes in South Africa: A Triumph of Bonds?Document21 pagesA Comparative Analysis of Returns of Various Fi Nancial Asset Classes in South Africa: A Triumph of Bonds?Parvathi M LNo ratings yet
- Products and ServicesDocument1 pageProducts and ServicesParvathi M LNo ratings yet
- Task24 Client2 Parvathi ML 21WM60 (B2.)Document11 pagesTask24 Client2 Parvathi ML 21WM60 (B2.)Parvathi M LNo ratings yet
- Dialogues Case 1Document3 pagesDialogues Case 1Parvathi M LNo ratings yet
- Task 5Document11 pagesTask 5Parvathi M LNo ratings yet
- Current Elective New Elective Risk Management in Banks and Financial InstitutionsDocument2 pagesCurrent Elective New Elective Risk Management in Banks and Financial InstitutionsParvathi M LNo ratings yet
- References (CH)Document6 pagesReferences (CH)Parvathi M LNo ratings yet
- qt9gz4f4gj NosplashDocument195 pagesqt9gz4f4gj NosplashNhu QuynhNo ratings yet
- GARVICH, Javier. El Caracter Chicha en La Cultura Peruana ContemporaneaDocument10 pagesGARVICH, Javier. El Caracter Chicha en La Cultura Peruana ContemporaneaDarloxNo ratings yet
- Shurts v. Shurts, Ariz. Ct. App. (2015)Document6 pagesShurts v. Shurts, Ariz. Ct. App. (2015)Scribd Government DocsNo ratings yet
- RW2011-15 Toc 183749110917062932Document6 pagesRW2011-15 Toc 183749110917062932Dwi AgungNo ratings yet
- Ethnicity and Crime A3 Knowledge Organiser FinalDocument3 pagesEthnicity and Crime A3 Knowledge Organiser FinalCharlotte BrennanNo ratings yet
- 2ndlastmeritlist PDFDocument8 pages2ndlastmeritlist PDFMahnoor AsimNo ratings yet
- Down These Mean StreetsDocument4 pagesDown These Mean StreetsAndra Elena AgafiteiNo ratings yet
- Comprehensive car insurance cover noteDocument2 pagesComprehensive car insurance cover noteSupernau Admin KuchingNo ratings yet
- Ventureshield™ Paint Protection Film 7510Cc/Cs-Ld: Technical Data Sheet April 2011Document2 pagesVentureshield™ Paint Protection Film 7510Cc/Cs-Ld: Technical Data Sheet April 2011Димитър ПетровNo ratings yet
- Right to Privacy in IndiaDocument38 pagesRight to Privacy in IndiaMonika100% (1)
- 404 Damodhar Narayan Sawale V Shri Tejrao Bajirao Mhaske 4 May 2023 472234Document13 pages404 Damodhar Narayan Sawale V Shri Tejrao Bajirao Mhaske 4 May 2023 472234Avinash SrivastavaNo ratings yet
- Business Ethics and Social Responsibility Weekly HomeworkDocument2 pagesBusiness Ethics and Social Responsibility Weekly HomeworkAdrian GalanoNo ratings yet
- Policy and Guidelines in Requesting A CCTVDocument2 pagesPolicy and Guidelines in Requesting A CCTVChristopher LaputNo ratings yet
- Modules 13, 14 and 15Document11 pagesModules 13, 14 and 15George EvangelistaNo ratings yet
- United Feature Vs MunsingwearDocument1 pageUnited Feature Vs Munsingwearsally deeNo ratings yet
- Fauquier Supervisors May 14, 2020, AgendaDocument3 pagesFauquier Supervisors May 14, 2020, AgendaFauquier NowNo ratings yet
- Certificate of EarningsDocument1 pageCertificate of EarningsRiasat AliNo ratings yet
- Albert vs. University PublishingDocument10 pagesAlbert vs. University PublishingMadam JudgerNo ratings yet
- Timeline of Philippine HistoryDocument6 pagesTimeline of Philippine HistoryCarlos Baul DavidNo ratings yet
- OHVA Monthly House ReportDocument2 pagesOHVA Monthly House ReportKimberly Marie SousaNo ratings yet
- InvoiceDocument2 pagesInvoiceSelva maniNo ratings yet
- Tony Cliff State Capitalism in Russia (1955-1974)Document160 pagesTony Cliff State Capitalism in Russia (1955-1974)sergevictorNo ratings yet
- Pki (Sran15.1 02)Document129 pagesPki (Sran15.1 02)VVL1959No ratings yet
- TaskRabbit COI 2022Document1 pageTaskRabbit COI 2022daNo ratings yet