Professional Documents
Culture Documents
Naming CAs Esters and Amides Rules
Uploaded by
Carlton GrantOriginal Title
Copyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
Naming CAs Esters and Amides Rules
Uploaded by
Carlton GrantCopyright:
Available Formats
Naming Carboxylic Acids, Esters and Amides – Chem 30B
Carboxylic Acids
The IUPAC names of carboxylic acids
§ Replace the -e in the alkane name with -oic acid.
CH4 methane HCOOH methanoic acid
CH3—CH3 ethane CH3—COOH ethanoic acid
§ Number substituents from the carboxyl carbon (#1).
§ Extra carbonyl groups present are called –oxo.
§ Other alcohol groups –OH are known as –hydroxyl
The common names of simple carboxylic acids are:
§ Locate substituents using a, b, γ (letters of the greek alphabet) for the carbon atoms adjacent to the carboxyl
carbon.
§ Dicarboxylic acids contain two –COOH groups and they end wiith –dioic acid.
§ Unsaturated acids are named (IUPAC system) with the ending –enoic.
Esters
The name of an ester contains the names of ethanoate (acetate) methyl
§ The alkyl group from the alcohol.
§ The carbon chain from the acid with –ate ending. IUPAC: methyl ethanoate
from alcohol from acid common: methyl acetate
Amides
Amides are named as alkanamides.
§ IUPAC replaces –oic acid ending with –amide.
§ Common names replace -ic acid ending with –amide.
§ An alkyl group bonded to the N atom is named as
N-alkyl in front of the amide name. (Use N,N- if two identical groups are attached.)
Reyes free to copy for educational purposes
You might also like
- The Peptides: Volume II Synthesis, Occurrence, and Action of Biologically Active PolypeptidesFrom EverandThe Peptides: Volume II Synthesis, Occurrence, and Action of Biologically Active PolypeptidesNo ratings yet
- Carboxylic Acids & DerivativesDocument45 pagesCarboxylic Acids & Derivativesayesha sheikhNo ratings yet
- Schaum's Easy Outline of Organic Chemistry, Second EditionFrom EverandSchaum's Easy Outline of Organic Chemistry, Second EditionRating: 3.5 out of 5 stars3.5/5 (2)
- Kakhasan Atom Karbon Dan Gugus FungsinyaDocument34 pagesKakhasan Atom Karbon Dan Gugus FungsinyaAllright ShitNo ratings yet
- Assembly Instructions for Polypeptide Models: Academic Press/Molecular Design Inc. Precision Molecular ModelsFrom EverandAssembly Instructions for Polypeptide Models: Academic Press/Molecular Design Inc. Precision Molecular ModelsNo ratings yet
- Organic Chemistry SummaryDocument8 pagesOrganic Chemistry SummaryZeath ElizaldeNo ratings yet
- CH (CH) CO Butanoate: C H EthylDocument1 pageCH (CH) CO Butanoate: C H EthylmigzpontNo ratings yet
- Practice Makes Perfect in Chemistry: Organic ChemistryFrom EverandPractice Makes Perfect in Chemistry: Organic ChemistryRating: 3 out of 5 stars3/5 (1)
- Chapter 14 Carboxylic Ester Amides PDFDocument22 pagesChapter 14 Carboxylic Ester Amides PDFMADANNo ratings yet
- Practice Makes Perfect in Chemistry: Organic Chemistry with AnswersFrom EverandPractice Makes Perfect in Chemistry: Organic Chemistry with AnswersNo ratings yet
- Notes C16 121Document13 pagesNotes C16 121Amir HussainNo ratings yet
- Practice Makes Perfect in Chemistry: Acids, Bases, and Salts with AnswersFrom EverandPractice Makes Perfect in Chemistry: Acids, Bases, and Salts with AnswersNo ratings yet
- Notes Functional GroupsDocument5 pagesNotes Functional GroupsFrank GaoNo ratings yet
- Organo-Transition Metal Compounds and Related Aspects of Homogeneous Catalysis: Pergamon Texts in Inorganic ChemistryFrom EverandOrgano-Transition Metal Compounds and Related Aspects of Homogeneous Catalysis: Pergamon Texts in Inorganic ChemistryNo ratings yet
- 9.1 Carboxylic Acid and DerivativesDocument34 pages9.1 Carboxylic Acid and DerivativesgoverotaropafadzwaNo ratings yet
- The Important Classes of Organic CompoundsDocument2 pagesThe Important Classes of Organic CompoundsmigzpontNo ratings yet
- Alkanes: Contain Single Bonds CH - CH - CH - CH Saturated Hydrocarbon Substitution ReactionDocument28 pagesAlkanes: Contain Single Bonds CH - CH - CH - CH Saturated Hydrocarbon Substitution ReactionHesham El-RoussassiNo ratings yet
- 1STTTTTTTTTTTDocument15 pages1STTTTTTTTTTTCATALINO, JESSICA MAE W.No ratings yet
- Functional GroupsDocument5 pagesFunctional GroupsGian Austin R. AngelesNo ratings yet
- ChemistryDocument34 pagesChemistrynehaanees100% (1)
- Naming AlcoholsDocument9 pagesNaming AlcoholsMary Margaret "MM" A. Avena0% (1)
- Carboxylic Acid 2Document20 pagesCarboxylic Acid 2brettNo ratings yet
- Aldehydes & Ketones: (Alkanals & Alkanones)Document41 pagesAldehydes & Ketones: (Alkanals & Alkanones)Hunain NadeemNo ratings yet
- Rules in Naming of Carboxylic Acids and Its DerivativesDocument12 pagesRules in Naming of Carboxylic Acids and Its DerivativesJr BalagtasNo ratings yet
- Carbonyl CompoundsDocument11 pagesCarbonyl CompoundsAmon RicoNo ratings yet
- Organic Chem U-2 AlcoholDocument33 pagesOrganic Chem U-2 Alcoholsinte beyuNo ratings yet
- As Noted Earlier: CH (CH) CO ButanoateDocument1 pageAs Noted Earlier: CH (CH) CO ButanoatemigzpontNo ratings yet
- NomenclatureDocument38 pagesNomenclatureMary Darice WongNo ratings yet
- Unit 11 - Alcohols, Phenols and EthersDocument41 pagesUnit 11 - Alcohols, Phenols and EthersPoovaraahan RaghuveeranNo ratings yet
- Hyrocarbons OwnDocument22 pagesHyrocarbons OwnRia PerezNo ratings yet
- Nomenclature of Organic CompoundsDocument6 pagesNomenclature of Organic CompoundsEmhNo ratings yet
- Topic 4.4 Nomenclature and Isomerism in Organic Chemistry New Functional Groups Review of AS Isomerism Optical IsomerismDocument20 pagesTopic 4.4 Nomenclature and Isomerism in Organic Chemistry New Functional Groups Review of AS Isomerism Optical IsomerismRyantyler13No ratings yet
- Functional Groups ContainingDocument8 pagesFunctional Groups ContainingViku GuptaNo ratings yet
- Nomenclature Part 1Document7 pagesNomenclature Part 1gcxnhmzNo ratings yet
- 2021 1.5 - 1.7 Double Bonded Functional GroupsDocument27 pages2021 1.5 - 1.7 Double Bonded Functional GroupsMia PereiraNo ratings yet
- Aldehydes and Ketones-DSVOLDocument107 pagesAldehydes and Ketones-DSVOLMERCY ATUYANo ratings yet
- Classification of Organic CompoundsDocument3 pagesClassification of Organic Compoundskvp0107No ratings yet
- Nomenclature Bansal Iit Jee Organic PDFDocument24 pagesNomenclature Bansal Iit Jee Organic PDFa143deendayal0% (2)
- CH - 8 Aldehydes Ketones and Carboxylic AcidsDocument32 pagesCH - 8 Aldehydes Ketones and Carboxylic Acidspinkykuku12No ratings yet
- Nomenclature of Aldehydes and KetonesDocument12 pagesNomenclature of Aldehydes and KetonesArielle Ciara BajadaNo ratings yet
- Nomenclature of Organic CompoundsDocument36 pagesNomenclature of Organic CompoundsRx Nadeem ChhipaNo ratings yet
- NSSCAS Chemistry Theme 4 Topic 4.1 - TsumebDocument91 pagesNSSCAS Chemistry Theme 4 Topic 4.1 - Tsumebsikereteromanus9No ratings yet
- AVCL 9A Properties of Carboxylic Acids and EstersDocument8 pagesAVCL 9A Properties of Carboxylic Acids and EstersGiane MadrigalNo ratings yet
- Lecture-1: Introduction To Organic ChemistryDocument53 pagesLecture-1: Introduction To Organic ChemistryNatha ShaNo ratings yet
- New CHY3201 Chapter 6 Nucleophilic Acyl Substitution ReactionDocument33 pagesNew CHY3201 Chapter 6 Nucleophilic Acyl Substitution Reaction222418No ratings yet
- AldehydesDocument19 pagesAldehydesSeverina MamauagNo ratings yet
- IUPAC NomenclatureDocument36 pagesIUPAC NomenclatureRx Nadeem ChhipaNo ratings yet
- Alkanes 2Document30 pagesAlkanes 2Jaymarie ZabateNo ratings yet
- Nomenclature - Carbon BondingDocument28 pagesNomenclature - Carbon BondingRobertMurray100% (1)
- Chapter 21-25 PDFDocument134 pagesChapter 21-25 PDFHimanshu RanjanNo ratings yet
- Introduction To Organic ChemistryDocument31 pagesIntroduction To Organic Chemistryauni ramizahNo ratings yet
- Week 3 Hydrocarbon Alkanes Alkenes AlkynesDocument67 pagesWeek 3 Hydrocarbon Alkanes Alkenes AlkynesNikol BaltazarNo ratings yet
- Alcohols - Nomenclature and ClassificationDocument10 pagesAlcohols - Nomenclature and ClassificationDionisio BrinosaNo ratings yet
- Alkohol, Eter, Aldehid, KetonDocument21 pagesAlkohol, Eter, Aldehid, KetonFaesal AmrullahNo ratings yet
- Alkane 1Document17 pagesAlkane 1Aneesa JalilNo ratings yet
- Organic ChemistryDocument33 pagesOrganic ChemistryTr Mazhar PunjabiNo ratings yet
- Class12 Chemistry2 Unit12 NCERT TextBook EnglishEditionDocument32 pagesClass12 Chemistry2 Unit12 NCERT TextBook EnglishEditionSidharth ThukralNo ratings yet
- In Context: Subject Area: Organic Chemistry Level: 14-16 Years (Higher) Topic: Alcohols Source: RSC - Li/3NtocpmDocument3 pagesIn Context: Subject Area: Organic Chemistry Level: 14-16 Years (Higher) Topic: Alcohols Source: RSC - Li/3NtocpmCarlton GrantNo ratings yet
- Balanced Unbalanced WorksheetDocument7 pagesBalanced Unbalanced Worksheetlizetteburks100% (1)
- In Context: Subject Area: Organic Chemistry Level: 14-16 Years (Higher) Topic: Alcohols Source: RSC - Li/3NtocpmDocument4 pagesIn Context: Subject Area: Organic Chemistry Level: 14-16 Years (Higher) Topic: Alcohols Source: RSC - Li/3NtocpmCarlton GrantNo ratings yet
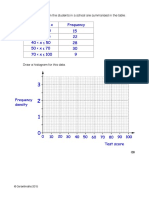
- Histograms PDF 002 003Document2 pagesHistograms PDF 002 003Carlton GrantNo ratings yet
- Knowledge Check: Subject Area: Organic Chemistry Level: 14-16 Years (Foundation) Topic: Alcohols Source: RSC - Li/3NtocpmDocument3 pagesKnowledge Check: Subject Area: Organic Chemistry Level: 14-16 Years (Foundation) Topic: Alcohols Source: RSC - Li/3NtocpmCarlton GrantNo ratings yet
- Hooks Law 2Document21 pagesHooks Law 2Carlton GrantNo ratings yet
- Naming Aldehydes and Ketones RulesDocument1 pageNaming Aldehydes and Ketones RulesCarlton GrantNo ratings yet
- 6-10.2-4 Hydrocarbon ReactionsDocument13 pages6-10.2-4 Hydrocarbon ReactionsSpandan GhoshalNo ratings yet
- In Context: Subject Area: Organic Chemistry Level: 14-16 Years (Foundation) Topic: Alcohols Source: RSC - Li/3NtocpmDocument5 pagesIn Context: Subject Area: Organic Chemistry Level: 14-16 Years (Foundation) Topic: Alcohols Source: RSC - Li/3NtocpmCarlton GrantNo ratings yet
- Petroleum and Alkanes Fractional Distillation Cracking CombustionDocument10 pagesPetroleum and Alkanes Fractional Distillation Cracking CombustionCarlton GrantNo ratings yet
- AMMONIADocument3 pagesAMMONIACarlton GrantNo ratings yet
- Solubility ChartDocument1 pageSolubility ChartVida Faith GalvezNo ratings yet
- ChemSchool Naming AlcoholsDocument1 pageChemSchool Naming AlcoholsCarlton GrantNo ratings yet
- Fractional DistillationDocument1 pageFractional DistillationCarlton GrantNo ratings yet
- How To Calculate Resistors in Series and ParallelDocument4 pagesHow To Calculate Resistors in Series and ParallelGeoff HampsonNo ratings yet
- Gravimetric AnalysisDocument4 pagesGravimetric AnalysisCarlton Grant100% (1)
- Topic 17 NotesDocument40 pagesTopic 17 NotessnigdhaNo ratings yet
- ResistanceDocument1 pageResistanceCarlton GrantNo ratings yet
- How To Calculate Resistors in ParallelDocument3 pagesHow To Calculate Resistors in ParallelCarlton GrantNo ratings yet
- Mass SpectrometryDocument4 pagesMass SpectrometryCarlton GrantNo ratings yet
- Faraday's Second Law of ElectrolysisDocument2 pagesFaraday's Second Law of ElectrolysisCarlton GrantNo ratings yet
- Esters: The Reaction Between A Carboxylic Acid and An Alcohol Produces An Ester Molecule and A Molecule of WaterDocument5 pagesEsters: The Reaction Between A Carboxylic Acid and An Alcohol Produces An Ester Molecule and A Molecule of WaterCarlton GrantNo ratings yet
- How To Calculate Resistors in SeriesDocument2 pagesHow To Calculate Resistors in SeriesCarlton GrantNo ratings yet
- Kidney Stone FormationDocument1 pageKidney Stone FormationCarlton GrantNo ratings yet
- نام گذاری اسیدها و بازهاDocument2 pagesنام گذاری اسیدها و بازهاapi-3706290100% (1)
- Probability: How Likely Something Is To HappenDocument2 pagesProbability: How Likely Something Is To HappenCarlton GrantNo ratings yet
- Trig Ratios: Another Angle Is Often Labeled, and The Three Sides Are Then CalledDocument1 pageTrig Ratios: Another Angle Is Often Labeled, and The Three Sides Are Then CalledCarlton GrantNo ratings yet
- Set TheoryDocument1 pageSet TheoryCarlton GrantNo ratings yet
- Pythagoras TheoremDocument2 pagesPythagoras TheoremCarlton GrantNo ratings yet
- Organic Chemistry for Schools: Advanced Level and Senior High SchoolFrom EverandOrganic Chemistry for Schools: Advanced Level and Senior High SchoolNo ratings yet
- Periodic Tales: A Cultural History of the Elements, from Arsenic to ZincFrom EverandPeriodic Tales: A Cultural History of the Elements, from Arsenic to ZincRating: 3.5 out of 5 stars3.5/5 (137)
- The Disappearing Spoon: And Other True Tales of Madness, Love, and the History of the World from the Periodic Table of the ElementsFrom EverandThe Disappearing Spoon: And Other True Tales of Madness, Love, and the History of the World from the Periodic Table of the ElementsRating: 4 out of 5 stars4/5 (146)
- Is That a Fact?: Frauds, Quacks, and the Real Science of Everyday LifeFrom EverandIs That a Fact?: Frauds, Quacks, and the Real Science of Everyday LifeRating: 5 out of 5 stars5/5 (4)
- Monkeys, Myths, and Molecules: Separating Fact from Fiction, and the Science of Everyday LifeFrom EverandMonkeys, Myths, and Molecules: Separating Fact from Fiction, and the Science of Everyday LifeRating: 4 out of 5 stars4/5 (1)
- The Regenerative Grower's Guide to Garden Amendments: Using Locally Sourced Materials to Make Mineral and Biological Extracts and FermentsFrom EverandThe Regenerative Grower's Guide to Garden Amendments: Using Locally Sourced Materials to Make Mineral and Biological Extracts and FermentsRating: 5 out of 5 stars5/5 (3)
- The Elements We Live By: How Iron Helps Us Breathe, Potassium Lets Us See, and Other Surprising Superpowers of the Periodic TableFrom EverandThe Elements We Live By: How Iron Helps Us Breathe, Potassium Lets Us See, and Other Surprising Superpowers of the Periodic TableRating: 3.5 out of 5 stars3.5/5 (22)
- The Periodic Table: A Very Short IntroductionFrom EverandThe Periodic Table: A Very Short IntroductionRating: 4.5 out of 5 stars4.5/5 (3)
- The Nature of Drugs Vol. 1: History, Pharmacology, and Social ImpactFrom EverandThe Nature of Drugs Vol. 1: History, Pharmacology, and Social ImpactRating: 5 out of 5 stars5/5 (5)
- Chemistry for Breakfast: The Amazing Science of Everyday LifeFrom EverandChemistry for Breakfast: The Amazing Science of Everyday LifeRating: 4.5 out of 5 stars4.5/5 (90)
- Handbook of Formulating Dermal Applications: A Definitive Practical GuideFrom EverandHandbook of Formulating Dermal Applications: A Definitive Practical GuideNo ratings yet
- A Perfect Red: Empire, Espionage, and the Quest for the Color of DesireFrom EverandA Perfect Red: Empire, Espionage, and the Quest for the Color of DesireRating: 4 out of 5 stars4/5 (129)
- Water-Based Paint Formulations, Vol. 3From EverandWater-Based Paint Formulations, Vol. 3Rating: 4.5 out of 5 stars4.5/5 (6)
- Essential Oil Chemistry Formulating Essential Oil Blends that Heal - Aldehyde - Ketone - Lactone: Healing with Essential OilFrom EverandEssential Oil Chemistry Formulating Essential Oil Blends that Heal - Aldehyde - Ketone - Lactone: Healing with Essential OilRating: 5 out of 5 stars5/5 (1)
- Bioplastics: A Home Inventors HandbookFrom EverandBioplastics: A Home Inventors HandbookRating: 4 out of 5 stars4/5 (2)
- Chemistry: a QuickStudy Laminated Reference GuideFrom EverandChemistry: a QuickStudy Laminated Reference GuideRating: 5 out of 5 stars5/5 (1)
- Formulating, Packaging, and Marketing of Natural Cosmetic ProductsFrom EverandFormulating, Packaging, and Marketing of Natural Cosmetic ProductsNo ratings yet
- Guidelines for Integrating Process Safety into Engineering ProjectsFrom EverandGuidelines for Integrating Process Safety into Engineering ProjectsNo ratings yet
- The Periodic Table of Elements - Alkali Metals, Alkaline Earth Metals and Transition Metals | Children's Chemistry BookFrom EverandThe Periodic Table of Elements - Alkali Metals, Alkaline Earth Metals and Transition Metals | Children's Chemistry BookNo ratings yet
- The Periodic Table of Elements - Post-Transition Metals, Metalloids and Nonmetals | Children's Chemistry BookFrom EverandThe Periodic Table of Elements - Post-Transition Metals, Metalloids and Nonmetals | Children's Chemistry BookNo ratings yet