Professional Documents
Culture Documents
Haematology Haematology Haematology Haematology
Uploaded by
sakshamOriginal Title
Copyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
Haematology Haematology Haematology Haematology
Uploaded by
sakshamCopyright:
Available Formats
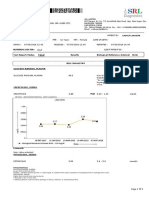
Client Processed By
Wellness Collection Centre (Bareilly) Pathkind Diagnostic Pvt. Ltd.
Shop No.2, E2 Model Town Near Dussera Mela Ground, A-6. Ekta Nagar, Opp. Care Hospital, Stadium Road, Bareilly , U.P. -243122
Bareilly, Uttar Pradesh - 243001
Name : Ms. ABHA DILAWARI Billing Date : 30/09/2021 11:48:39
Age : 20 Yrs Sample Collected on : 30/09/2021 11:51:12
Sex : Female Sample Received on : 30/09/2021 12:37:11
P. ID No. : P1600143806 Report Released on : 30/09/2021 13:45:52
Accession No : 1600211095281 Barcode No. : 14082670
Referring Doctor : SELF
Referred By : Ref no. :
Report Status - Final
Test Name Result Biological Ref. Interval Unit
HAEMATOLOGY
Complete Blood Count (CBC)
Haemoglobin (Hb) 14.2 12 - 15 gm/dL
Sample: Whole Blood EDTA
Method: Photometric measurement
Total WBC Count / TLC 2.1 L 4 - 10 thou/µL
Sample: Whole Blood EDTA
Method: Impedance
RBC Count 4.7 3.8 - 4.8 million/µL
Sample: Whole Blood EDTA
Method: Impedance
PCV / Hematocrit 42.5 36 - 46 %
Sample: Whole Blood EDTA
Method: Impedance
MCV 90.3 83 - 101 fL
Sample: Whole Blood EDTA
Method: Calculated
MCH 30.2 27 - 32 pg
Sample: Whole Blood EDTA
Method: Calculated
MCHC 33.5 31.5 - 34.5 g/dL
Sample: Whole Blood EDTA
Method: Calculated
RDW (Red Cell Distribution Width) 12.5 11.9 - 15.5 %
Sample: Whole Blood EDTA
Method: Calculated
DLC (Differential Leucocyte Count)
Method: Flowcytometry/Microscopy
Neutrophils 56 40 - 80 %
Sample: Whole Blood EDTA
Method: VCS Technology & Microscopy
1600211095281 Ms. ABHA DILAWARI
Page No: 1 of 5
Client Processed By
Wellness Collection Centre (Bareilly) Pathkind Diagnostic Pvt. Ltd.
Shop No.2, E2 Model Town Near Dussera Mela Ground, A-6. Ekta Nagar, Opp. Care Hospital, Stadium Road, Bareilly , U.P. -243122
Bareilly, Uttar Pradesh - 243001
Name : Ms. ABHA DILAWARI Billing Date : 30/09/2021 11:48:39
Age : 20 Yrs Sample Collected on : 30/09/2021 11:51:12
Sex : Female Sample Received on : 30/09/2021 12:37:11
P. ID No. : P1600143806 Report Released on : 30/09/2021 13:45:52
Accession No : 1600211095281 Barcode No. : 14082670
Referring Doctor : SELF
Referred By : Ref no. :
Report Status - Final
Test Name Result Biological Ref. Interval Unit
Lymphocytes 38 20 - 40 %
Sample: Whole Blood EDTA
Method: VCS Technology & Microscopy
Eosinophils 02 1-6 %
Sample: Whole Blood EDTA
Method: VCS Technology & Microscopy
Monocytes 04 2 - 10 %
Sample: Whole Blood EDTA
Method: VCS Technology & Microscopy
Basophils 00 0-2 %
Sample: Whole Blood EDTA
Method: VCS Technology & Microscopy
Absolute Neutrophil Count 1176 L 2000 - 7000 /µL
Sample: Whole Blood EDTA
Absolute Lymphocyte Count 798 L 1000 - 3000 /µL
Sample: Whole Blood EDTA
Absolute Eosinophil Count 42 20 - 500 /µL
Sample: Whole Blood EDTA
Absolute Monocyte Count 84 L 200 - 1000 /µL
Sample: Whole Blood EDTA
Absolute Basophil Count 00 L 20 - 100 /µL
Sample: Whole Blood EDTA
DLC Performed By Automated
Sample: Whole Blood EDTA
Platelet Count 144 L 150 - 410 thou/µL
Sample: Whole Blood EDTA
Method: Impedance
MPV (Mean Platelet Volume) 9.0 6.8 - 10.9 fL
Sample: Whole Blood EDTA
Method: Calculated
1600211095281 Ms. ABHA DILAWARI
Page No: 2 of 5
Client Processed By
Wellness Collection Centre (Bareilly) Pathkind Diagnostic Pvt. Ltd.
Shop No.2, E2 Model Town Near Dussera Mela Ground, A-6. Ekta Nagar, Opp. Care Hospital, Stadium Road, Bareilly , U.P. -243122
Bareilly, Uttar Pradesh - 243001
Name : Ms. ABHA DILAWARI Billing Date : 30/09/2021 11:48:39
Age : 20 Yrs Sample Collected on : 30/09/2021 11:51:12
Sex : Female Sample Received on : 30/09/2021 12:37:11
P. ID No. : P1600143806 Report Released on : 30/09/2021 13:45:52
Accession No : 1600211095281 Barcode No. : 14082670, 14082669
Referring Doctor : SELF
Referred By : Ref no. :
Report Status - Final
Test Name Result Biological Ref. Interval Unit
SEROLOGY
WIDAL
Sample: Serum
Method: aggluttination method
Salmonella Typhi 'O' < 1:80 < 1:80
Salmonella Typhi 'H' < 1:80 < 1:80
Salmonella Paratyphi 'AH' < 1:80 < 1:80
Salmonella Paratyphi 'BH' < 1:80 < 1:80
Result : Negative
Complete Blood Count (CBC)
Clinical Significance :
CBC comprises of estimation of the cellular componenets of blood including RBCs, WBCs and Platelets. Mean corpuscular volume (MCV) is a
measure of the size of the average RBC, MCH is a measure of the hemoglobin cointent of the average RBC and MCHC is the hemoglobin
concentration per RBC. The red cell distribution width (RDW) is a measure of the degree of variation in RBC size (anisocytosis) and is helpful in
distinguishing between some anemias. CBC examination is used as a screening tool to confirm a hematologic disorder, to establish or rule out a
diagnosis, to detect an unsuspected hematologic disorder, or to monitor effects of radiation or chemotherapy. Abnormal results may be due to a
primary disorder of the cell-producing organs or an underlying disease. Results should be interpreted in conjunction with the patient's clinical picture
and appropriate additional testing performed.
WIDAL
While the definitive diagnosis oftyphoid fever depends on the isolation of S typhi from blood, stools, urine orother body fluids, the role of the Widal test
had been to increase the index ofsuspicion for the presence of typhoid fever by demonstrating a positive agglutinationduring the acute and convalescent
period of infection with evidence of a four-fold rise of antibody titre.In many developing countries, including India, the Widal test appears to be the only
laboratory means employed in the diagnosis of typhoidfever among suspected patients. As the test suffers from serious cross-reactivitywith other
infectious agents, it may produce false-positive results, leading to anover-diagnosis of typhoid fever. The Widal test reaction involves the use of bacterial
1600211095281 Ms. ABHA DILAWARI
Page No: 3 of 5
Client Processed By
Wellness Collection Centre (Bareilly) Pathkind Diagnostic Pvt. Ltd.
Shop No.2, E2 Model Town Near Dussera Mela Ground, A-6. Ekta Nagar, Opp. Care Hospital, Stadium Road, Bareilly , U.P. -243122
Bareilly, Uttar Pradesh - 243001
Name : Ms. ABHA DILAWARI Billing Date : 30/09/2021 11:48:39
Age : 20 Yrs Sample Collected on : 30/09/2021 11:51:12
Sex : Female Sample Received on : 30/09/2021 12:37:11
P. ID No. : P1600143806 Report Released on : 30/09/2021 13:45:52
Accession No : 1600211095281 Barcode No. : 14082670, 14082669
Referring Doctor : SELF
Referred By : Ref no. :
Report Status - Final
Test Name Result Biological Ref. Interval Unit
suspensions of S typhi andS paratyphi‘A’ and ‘B’, treated to retain only the ‘O’ and ‘H’ antigens. Theseantigens are employed to detectcorresponding
antibodies in the serum of apatient suspected of having typhoid fever. The IgM somatic O antibody appearsfirst and represents the initial serologic
response in acute typhoid fever, while theIgG flagella H antibody usually develops more slowly but persists for longer.
In an individual with no prior exposureto S typhi infection (either lack of active infection or absence of passive immunisation),a higher than 1:80 or 1:160
titre on an initial single test, usually correlatesfairly well with exposure to typhoid fever. However, even these single highvaluetitres in an endemic area
where repeated exposures to S typhi may haveoccurred, do not have any clinical relevance in the absence of a positive isolate ofthe causative organism.
Researchers from different parts of India have reported that in normally health blood donors, the baseline titre for antibodies to “O” and “H” antigens of
Salmonella enterica serotype typhi was 1:40 and hence, based on the above results, it could be recommended to use a cutoff level of ≥1:80 for a single
antibody test titre. Similarly, baseline titre for antibody to H antigen of Salmonella enterica serotype paratyphiA and paratyphiB was 1:80 and the cutoff
level was ≥1:160 for a single antibody test titre.
** End of Report**
Dr. Somya Agarwal
MBBS. MD (Pathology)
Lab Head –Bareilly
1600211095281 Ms. ABHA DILAWARI
Page No: 4 of 5
NATIONAL REFERENCE LAB
PATHKIND DIAGNOSTICS PVT. LTD.
Plot No. 55 - 56, Udyog Vihar, Phase 4, Gurugram - 122015
E-Mail: care@pathkindlabs.com | Website: www.pathkindlabs.com
Customer Care: 782-784-4444
In our quest to keep you hale, hearty & healthy, we recommend these following
preventive packages
Healthkind - Preventive health check-up packages
Package Healthkind Complete Healthkind Advance Healthkind Platinum
Tests 77 Tests 83 Tests 85 Tests
Price 6589* Now at 3199 7879* Now at 3999 9480* Now at 4999
Immusure - Immunity Check Package
A comprehensive package that is designed to help you track your immunity level against variety
of threats including virus, bacteria & other pathogens.
Immusure 31 Tests 4410*
- CBC 3250
- Iron Studies (Iron, UIBC, TIBC & % Saturation)
- Vitamin-D,
- Immunoglobulin IgE Total
- Immunoglobulin Profile (IgA, IgG, IgM)
from the promoters of
You might also like
- PathkindDocument5 pagesPathkindgovt.job4692No ratings yet
- FRM Patient View All ReportDocument6 pagesFRM Patient View All ReportShailesh KumarNo ratings yet
- Department of Hematology Test Name Result Unit Bio. Ref. IntervalDocument3 pagesDepartment of Hematology Test Name Result Unit Bio. Ref. Intervalaf dNo ratings yet
- ReportDocument19 pagesReportpparmar871No ratings yet
- AstapathiDocument3 pagesAstapathiumamahes637No ratings yet
- Department of Haematology: Test Name Result Unit Bio. Ref. Range MethodDocument2 pagesDepartment of Haematology: Test Name Result Unit Bio. Ref. Range Methodgsm2008No ratings yet
- ReportDocument6 pagesReportvenkateshgoudvenkateshgouNo ratings yet
- Labreportnew - 2023-02-17T141822.094Document8 pagesLabreportnew - 2023-02-17T141822.094pavanimatteguntaNo ratings yet
- D-37/1, TTC MIDC, Turbhe, Navi Mumbai-400 703: Name Ref. by Test Asked::: Patientid: Home CollectionDocument2 pagesD-37/1, TTC MIDC, Turbhe, Navi Mumbai-400 703: Name Ref. by Test Asked::: Patientid: Home Collectionrohit singhNo ratings yet
- Report 1Document2 pagesReport 1Kanchan KarmakarNo ratings yet
- LabTest 18jul2023Document5 pagesLabTest 18jul2023Kedarnath KundaNo ratings yet
- KUNALYADAVreportDocument2 pagesKUNALYADAVreportSourabhNo ratings yet
- Hepatitis "B" Surface Antigen (Hbsag) Parameter Results MethodDocument4 pagesHepatitis "B" Surface Antigen (Hbsag) Parameter Results MethodSachin AgarwalNo ratings yet
- Lab ReportDocument4 pagesLab ReportdipikaNo ratings yet
- Kgxrdtqpylps1aixlnec2p3y PDFDocument1 pageKgxrdtqpylps1aixlnec2p3y PDFNaveen PathakNo ratings yet
- Report PDFDocument6 pagesReport PDFAayushiNo ratings yet
- LabReport 2Document2 pagesLabReport 2Tathagata ChaudhuriNo ratings yet
- ReportDocument2 pagesReportSupriya DewanNo ratings yet
- Report 220719 195623Document11 pagesReport 220719 195623Abdullah AlviNo ratings yet
- Diagnostic Report: FinalDocument2 pagesDiagnostic Report: FinalJohn AbhishekNo ratings yet
- Date 30/nov/2021 02:37AM 29/nov/21 07:51PM Unit Bio Ref IntervalDocument2 pagesDate 30/nov/2021 02:37AM 29/nov/21 07:51PM Unit Bio Ref IntervalSaurabh PuriNo ratings yet
- RSLT An21100870 PDFDocument3 pagesRSLT An21100870 PDFMuhammed Ameen MoulaviNo ratings yet
- Remotesensing 12 00857 v2Document1 pageRemotesensing 12 00857 v2Rajarshi BhattacharyaNo ratings yet
- Date 15/feb/2024 12:05PM 31/jul/23 11:10AM Unit Bio Ref IntervalDocument4 pagesDate 15/feb/2024 12:05PM 31/jul/23 11:10AM Unit Bio Ref IntervalrajorajisunnyNo ratings yet
- DN RainaDocument17 pagesDN RainaAjit Raina100% (1)
- Fever Panel BasicDocument7 pagesFever Panel BasicMadNo ratings yet
- L0075RE000419YDocument2 pagesL0075RE000419YAyana NandyNo ratings yet
- Department of Genetics: Covid-19 RT PCRDocument1 pageDepartment of Genetics: Covid-19 RT PCRMuhammed HadhiNo ratings yet
- Falaan ReportDocument4 pagesFalaan ReportAzeem ChauhanNo ratings yet
- Date 28/mar/2023 10:09AM Unit Bio Ref Interval: Laboratory Investigation ReportDocument12 pagesDate 28/mar/2023 10:09AM Unit Bio Ref Interval: Laboratory Investigation ReportChauhanNo ratings yet
- GANESANDocument8 pagesGANESANBuvanesh BalajiNo ratings yet
- S81 - Ram Ratan Lab-CcDocument1 pageS81 - Ram Ratan Lab-CcCHANDAN KUMARNo ratings yet
- Date 28/mar/2023 09:29AM Unit Bio Ref Interval: Laboratory Investigation ReportDocument10 pagesDate 28/mar/2023 09:29AM Unit Bio Ref Interval: Laboratory Investigation ReportbhailogofficiaNo ratings yet
- Department of Haematology Test Name Result Unit Bio. Ref. Range MethodDocument6 pagesDepartment of Haematology Test Name Result Unit Bio. Ref. Range MethodjudgerajaNo ratings yet
- 23rglmgb2punhj3tbk4mpfhzDocument1 page23rglmgb2punhj3tbk4mpfhzHarsh AryaNo ratings yet
- Department of Clinical Biochemistry: Total Cholesterol, SerumDocument1 pageDepartment of Clinical Biochemistry: Total Cholesterol, SerumSabarinath RK PillaiNo ratings yet
- Report Geeti PaulDocument22 pagesReport Geeti PaulSAIKAT PAULNo ratings yet
- Savitri Singh ThyrocareDocument20 pagesSavitri Singh ThyrocareAbhishek SinghNo ratings yet
- Thyrocare Processed atDocument3 pagesThyrocare Processed atKarim SirNo ratings yet
- Complete Blood Count: Name DR Chetanbhai MojidaraDocument2 pagesComplete Blood Count: Name DR Chetanbhai Mojidarachetan mojidraNo ratings yet
- Date 02/sep/2023 10:08AM Unit Bio Ref Interval: Laboratory Investigation ReportDocument12 pagesDate 02/sep/2023 10:08AM Unit Bio Ref Interval: Laboratory Investigation ReportlbhsinhaNo ratings yet
- Arogyam1 3Document12 pagesArogyam1 3Sashikanta NayakNo ratings yet
- Report 400d0061Document18 pagesReport 400d0061Santosh BhandarkarNo ratings yet
- Thirumalai N:::: Patient Age / Sex 34 Y / Male BranchDocument6 pagesThirumalai N:::: Patient Age / Sex 34 Y / Male Branchthirumalai narayananNo ratings yet
- Ak 19105144Document2 pagesAk 19105144raghavremNo ratings yet
- Department of Clinical Biochemistry: Ppbs 215 MG/DLDocument1 pageDepartment of Clinical Biochemistry: Ppbs 215 MG/DLMunniNo ratings yet
- Date 03/aug/2023 07:32AM Unit Bio Ref Interval: Laboratory Investigation ReportDocument12 pagesDate 03/aug/2023 07:32AM Unit Bio Ref Interval: Laboratory Investigation ReportRishabh GuptaNo ratings yet
- Afridi Shaikh-23Document5 pagesAfridi Shaikh-23Altamash AnsariNo ratings yet
- Department of Haematology: Platelet CountDocument2 pagesDepartment of Haematology: Platelet Countbinto johnNo ratings yet
- Department of Haematology: Platelet CountDocument2 pagesDepartment of Haematology: Platelet Countbinto johnNo ratings yet
- Blood Tests ReportDocument7 pagesBlood Tests ReportLalith GoudNo ratings yet
- Tam1726834776305969 - RLS 4Document16 pagesTam1726834776305969 - RLS 4Rahul GNo ratings yet
- Self C Nagarajan (38Y/M) C Nagarajan Flat 3 MB Flats No 27 Bazaar Street Nesapakkam West KK Nagar Chennai 600078 Opp Ravi Travels Parking ADocument12 pagesSelf C Nagarajan (38Y/M) C Nagarajan Flat 3 MB Flats No 27 Bazaar Street Nesapakkam West KK Nagar Chennai 600078 Opp Ravi Travels Parking ANagarajan ChandrasekaranNo ratings yet
- Date 01/dec/2021 04:33AM 30/nov/21 05:17PM Unit Bio Ref IntervalDocument2 pagesDate 01/dec/2021 04:33AM 30/nov/21 05:17PM Unit Bio Ref IntervalSaurabh PuriNo ratings yet
- Doctor: EW192357-EW/CA1 68/M 25/05/2019 03:21 PM: Pareeth K M Sample Collected atDocument1 pageDoctor: EW192357-EW/CA1 68/M 25/05/2019 03:21 PM: Pareeth K M Sample Collected atAnwar P.sNo ratings yet
- Department of Haematology Test Name Result Unit Bio. Ref. Range MethodDocument5 pagesDepartment of Haematology Test Name Result Unit Bio. Ref. Range MethodSunil KhandekarNo ratings yet
- Po4009507960 540Document15 pagesPo4009507960 540PRADEEP KUMARNo ratings yet
- Department of Clinical Biochemistry: Creatinine, SerumDocument3 pagesDepartment of Clinical Biochemistry: Creatinine, SerumrishikundanNo ratings yet
- Altum Report of Dr. SnehaDocument3 pagesAltum Report of Dr. SnehaSham AmshNo ratings yet
- MrsSANGEETASAHU 45Y FemaleDocument6 pagesMrsSANGEETASAHU 45Y FemalesayyedatfatmaNo ratings yet
- Tulshan-Dhage2019 Chapter SurveyOnVirtualAssistantGoogleDocument13 pagesTulshan-Dhage2019 Chapter SurveyOnVirtualAssistantGoogleRushiNo ratings yet
- Cyber Security Unit 2 NotesDocument32 pagesCyber Security Unit 2 NotessakshamNo ratings yet
- ActiugiDocument20 pagesActiugisakshamNo ratings yet
- Geographical PlotsDocument4 pagesGeographical PlotssakshamNo ratings yet
- Engineering & Managerial Economics EHU 501Document3 pagesEngineering & Managerial Economics EHU 501sakshamNo ratings yet
- What Is Knowledge RepresentationDocument10 pagesWhat Is Knowledge RepresentationsakshamNo ratings yet
- ActiugiDocument20 pagesActiugisakshamNo ratings yet
- ActiugiDocument20 pagesActiugisakshamNo ratings yet
- MLT NotesDocument17 pagesMLT NotessakshamNo ratings yet
- Notice/Circular: All Students of B. Tech. (2019), B. Pharm. (2019), MBA (2020) & MCA (2020)Document1 pageNotice/Circular: All Students of B. Tech. (2019), B. Pharm. (2019), MBA (2020) & MCA (2020)sakshamNo ratings yet
- MLT 1 Unit NotesDocument16 pagesMLT 1 Unit NotessakshamNo ratings yet
- CDDocument254 pagesCDsakshamNo ratings yet
- The Oriental Insurance Company LimitedDocument3 pagesThe Oriental Insurance Company LimitedsakshamNo ratings yet
- Notice: Centre of Excellence Under Advanced AI, ML and Data Science LabDocument1 pageNotice: Centre of Excellence Under Advanced AI, ML and Data Science LabsakshamNo ratings yet
- Event List FinalDocument1 pageEvent List FinalsakshamNo ratings yet
- Project Synopsis Format GuidelinesDocument2 pagesProject Synopsis Format GuidelinessakshamNo ratings yet
- B.Tech 2nd Year CSAI - CSML - CSDS - CSIOT - 2021-22 0709Document14 pagesB.Tech 2nd Year CSAI - CSML - CSDS - CSIOT - 2021-22 0709sakshamNo ratings yet
- Student College PortalDocument11 pagesStudent College PortalsakshamNo ratings yet
- Student College PortalDocument11 pagesStudent College PortalsakshamNo ratings yet
- Final Rulebook HunkaarDocument4 pagesFinal Rulebook HunkaarsakshamNo ratings yet
- Root BiomodificationDocument88 pagesRoot Biomodificationperiodontics07No ratings yet
- Dnb-General Surgery: Competency Based Training ProgrammeDocument49 pagesDnb-General Surgery: Competency Based Training ProgrammeLakshmi Mounica Grandhi100% (1)
- Pediatric Blood TransfusionDocument10 pagesPediatric Blood TransfusionELISION OFFICIALNo ratings yet
- 4 EL Husseinys Essentials of Cardiovascular System @eduwaves360Document236 pages4 EL Husseinys Essentials of Cardiovascular System @eduwaves360ahmed_abu_alrobNo ratings yet
- AIP Chap13 Shoulder DystociaDocument13 pagesAIP Chap13 Shoulder DystociaJeremia KurniawanNo ratings yet
- Dementia - StatPearls - NCBI BookshelfDocument7 pagesDementia - StatPearls - NCBI BookshelfSMA N 1 TOROHNo ratings yet
- Marijuana: A Medicine For CancerDocument11 pagesMarijuana: A Medicine For CancerXylvia Hannah Joy CariagaNo ratings yet
- GAD Aos 40 AnosDocument7 pagesGAD Aos 40 AnosrafaelplNo ratings yet
- Brief Intervention: Mhgap-Ig Base Course - Field Test Version 1.00 - May 2012 1Document16 pagesBrief Intervention: Mhgap-Ig Base Course - Field Test Version 1.00 - May 2012 1TEOFILO PALSIMON JR.No ratings yet
- Re EvaluationDocument4 pagesRe EvaluationchandanaNo ratings yet
- Submental Abscess After Deoxycholic Acid Injection: ReferenceDocument1 pageSubmental Abscess After Deoxycholic Acid Injection: Referencechash75No ratings yet
- CMC Exam HandbookDocument18 pagesCMC Exam HandbookNílo StárnNo ratings yet
- Modul Normal Delivery 2016Document8 pagesModul Normal Delivery 2016Alvin FarhanNo ratings yet
- Throat Diseases and SepsisDocument15 pagesThroat Diseases and SepsisOlga GoryachevaNo ratings yet
- Basic Life SupportDocument29 pagesBasic Life SupportRyan-Jay AbolenciaNo ratings yet
- Session 20Document2 pagesSession 20Bernadeth Barrientos ZamoraNo ratings yet
- Unit 3 Part 2 Rheumatoid ArthritisDocument10 pagesUnit 3 Part 2 Rheumatoid ArthritisReman AlingasaNo ratings yet
- Module 4: Mood Disorders: Disorders and Individuals With Bipolar Disorders. The Key Difference BetweenDocument7 pagesModule 4: Mood Disorders: Disorders and Individuals With Bipolar Disorders. The Key Difference BetweenLiz Evermore0% (1)
- Carillo vs. People G.R. No. 86890 FULL TEXTDocument8 pagesCarillo vs. People G.R. No. 86890 FULL TEXTJeng PionNo ratings yet
- Review Material Exam TypeDocument9 pagesReview Material Exam TypeFelimon BugtongNo ratings yet
- Chronic Hepatitis BDocument5 pagesChronic Hepatitis BMurat HessesNo ratings yet
- Psychiatric Assessment and Evaluation: History TakingDocument9 pagesPsychiatric Assessment and Evaluation: History TakingWaheedullah AhmadiNo ratings yet
- HSG PresentationDocument18 pagesHSG Presentationashikin92No ratings yet
- Drug StudyDocument5 pagesDrug Studyanon_168410816No ratings yet
- Κατάσταση Πρωτοτύπων-Αντιγράφων φαρμάκων - Απρίλιος 2011Document121 pagesΚατάσταση Πρωτοτύπων-Αντιγράφων φαρμάκων - Απρίλιος 2011nosfarNo ratings yet
- C - Syiam Amalia Rahmah - Topik 4Document7 pagesC - Syiam Amalia Rahmah - Topik 4Syiam Amalia RahmahNo ratings yet
- Haemostasis: 1. Vascular SpasmDocument5 pagesHaemostasis: 1. Vascular SpasmAnurag YadavNo ratings yet
- Planning For A Health CareerDocument5 pagesPlanning For A Health CareerDanch CatamuraNo ratings yet
- Labor Monitoring RecordDocument4 pagesLabor Monitoring RecordFrancis PeterosNo ratings yet
- TrypanosomiasisDocument39 pagesTrypanosomiasisNani HendrianiNo ratings yet