Professional Documents
Culture Documents
AHS Prelim 2014 P1
Uploaded by
19Y1H GAO CHENZHANGCopyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
AHS Prelim 2014 P1
Uploaded by
19Y1H GAO CHENZHANGCopyright:
Available Formats
ANGLICAN HIGH SCHOOL
SECONDARY FOUR
PRELIMINARY EXAMINATION 2014
CANDIDATE
NAME
CLASS INDEX NUMBER
CHEMISTRY 5073/01
Paper 1 Multiple Choice 6 August 2014
1 hour
Additional Material: Multiple Choice Answer Sheet
READ THESE INSTRUCTIONS FIRST
Write in soft pencil.
Do not use staples, paper clips, highlighters, glue or correction fluid.
Write you name, class and index number on the Answer Sheet in the spaces provided.
There are forty questions on this paper. Answer all questions. For each question there
are four possible answers A, B, C and D.
Choose the one you consider correct and record your choice in soft pencil on the
separate Answer Sheet.
Read the instructions on the Answer Sheet very carefully.
Each correct answer will score one mark. A mark will not be deducted for a wrong
answer.
Any rough working should be done in this paper.
A copy of the Periodic Table is printed on page 10.
This document consists of 10 printed pages.
2
1 A series of experiments was carried out in order to determine the effect of
concentration on the rate of reaction between hydrochloric acid and aqueous sodium
hydroxide. In each of these experiments, 25.0cm3 of sodium hydroxide was reacted
with different concentrations of hydrochloric acid.
The list of apparatus provided is as follows:
burette conical flask measuring cylinder pipette
stopwatch thermometer
Which four pieces of apparatus would be needed to carry out the experiment?
A burette, conical flask, measuring cylinder, thermometer
B burette, measuring cylinder, stopwatch, thermometer
C burette, pipette, stopwatch, thermometer
D conical flask, measuring cylinder, pipette, stopwatch
2 The diagram shows the chromatogram of four different substances using the same
solvent. Fructose has an Rf value of 0.25. Which sugar is fructose?
Solvent
front
A B C D Base line
3 Astatine boils at 337oC.
Which other property of astatine proves that it is pure?
A It is an inert gas.
B It is neither acidic nor basic in nature.
C It melts at a fixed temperature.
D It reacts with hydrogen to produce ammonia.
4 Which mixture of gases would not change the colour of moist blue litmus paper?
A carbon dioxide and oxygen C hydrogen and ammonia
B chlorine and nitrogen D helium and hydrogen chloride
5 Which reagent could be used to distinguish between aluminium nitrate and zinc
nitrate?
A aqueous ammonia
B aqueous sodium hydroxide
C aqueous sodium hydroxide with powdered aluminium
D litmus solution
Anglican High School 5073/01/SEC4MYE/14
3
28
6 Mg is an isotope of magnesium which is radioactive. Which of these shows
correctly the number of protons, electrons and neutron an atom of 28Mg contain?
protons electrons neutrons
A 12 12 16
B 12 16 16
C 14 12 14
D 14 14 12
7 Propane has the structure shown.
H H H
H–C–C–C–H
H H H
How many electrons in a molecule of propane, C3H8 are not involved in bonding?
A 0 C 6
B 3 D 8
8 Which two statements about ionic compounds are correct?
I They can conduct electricity in the aqueous and molten states.
II They can dissolve in ethanol but not in water.
III Their melting and boiling point is a result of the electrostatic forces of
attraction present.
IV Their melting and boiling point is a result of the intermolecular forces of
attraction present.
A I and II C II and III
B I and III D II and IV
9 Hydrogen chloride dissolves in water to produce a solution. Which ion(s) is/are
responsible for the acidic property of the solution?
A Cl- C OH-
B H+ D H+ and Cl-
10 Which oxide reacts in the same manner as aluminium oxide, Al2O3?
A Li2O C MgO
B FeO D PbO
11 Which reactants could be used safely to prepare potassium chloride?
A aqueous potassium carbonate and hydrochloric acid
B aqueous potassium nitrate and aqueous sodium chloride
C potassium and hydrochloric acid
D potassium and aqueous sodium chloride
Anglican High School 5073/01/SEC4MYE/14
4
12 10cm3 of a hydrocarbon was used to react with excess oxygen to produce 70cm3 of
gaseous products. Which equation best represents the reaction?
A C3H8 (g) + 5O2 (g) 3CO2 (g) + 4H2O (g)
B C7H16 (l) + 11O2 (g) 7CO2 (g) + 8H2O (g)
C 2C2H5OH (l) + 6O2 (g) 4CO2 (g) + 6H2O (g)
D 2C3H7OH (l) + 9O2 (g) 6CO2 (g) + 8H2O (g)
13 25.0cm3 of 0.2mol/dm3 of an aqueous carbonate reacts completely with 50.0cm3 of
0.1mol/dm3 of an acid. What is the charge of the anion of the acid?
A 1+ C 1-
B 2+ D 2-
14 Which fertiliser provides the most nitrogen per mole?
A NaNO3 C (NH4)2SO4
B NH4NO3 D (NH4)3PO4
15 In an experiment, a quantity of electricity plated 0.120 mole of copper metal onto an
object from a solution of copper (II) ions. The same quantity of electricity plated
0.080 mole of gold onto an object. What is the formula of the gold ions?
A Au+ C Au3+
B Au2+ D Au4+
16 Element A is found in the same group as lithium. What can be deduced about this
element?
A It is a strong oxidising agent.
B It has one valence electron.
C It reacts with oxygen to form a covalent compound.
D Its oxide dissolves in water to form an acidic solution.
17 Across a period, in a periodic table, there is a trend from metallic to non-metallic
elements. This is caused by
A an increase in the number of electrons.
B an increase in the number of electron shells.
C an increase in the number of protons.
D an increase in the number of valency electrons.
Anglican High School 5073/01/SEC4MYE/14
5
18 The rate of reaction between a given mass of metal and an excess of hydrochloric
acid is studied by collecting the hydrogen gas in a graduated syringe. The results are
as shown in the table below.
Time taken / s Volume of
hydrogen gas / cm3
0 0
20 20
40 34
60 38
80 40
100 40
How much time is required for half the metal to react?
A 20s C 80s
B 60s D 100s
19 Which statement about the use of nickel in the hydrogenation of alkenes is correct?
A It increases the amount of kinetic energy the particles possess.
B It increases the number of reactants per unit volume.
C It increase the surface area to volume ratio of the reactants.
D It lowers the activation energy required.
20 Which compound, when added to aqueous iron(II) sulfate, takes part in a redox
reaction?
A acidified barium chloride
B acidified potassium manganate (VII)
C sodium hydroxide
D sulfur dioxide
21 Which is not an oxidation and/or reduction reaction?
A CuO + H2 Cu + H2O
B 2H2 + O2 2H2O
C Mg + 2HCl MgCl2 + H2
D NaOH + HNO3 NaNO3 + H2O
22 Which property of zinc is not typical of a transition metal?
A It has high density.
B It has high melting point.
C It is used to catalyse chemical reactions.
D Its compounds are mostly colourless.
Anglican High School 5073/01/SEC4MYE/14
6
23 Which carbonate does not produce an acidic gas upon heating?
A copper (II) carbonate C potassium carbonate
B iron (II) carbonate D zinc carbonate
24 Which two gases are produced mainly as waste gases in the blast furnace?
A carbon dioxide and carbon monoxide
B carbon dioxide and nitrogen
C carbon monoxide and oxygen
D carbon monoxide and nitrogen
25 Ethene, C2H4, undergoes atomisation to form carbon and hydrogen atoms according
to the equation below.
C2H4 2C +4H
Using the data give, calculate the total energy required for the reaction.
Bond Bond energy / kJ/mol
C-C 345
C=C 835
C-H 415
H-H 432
A 2005kJ C 2495 kJ
B 2073kJ D 2563kJ
26 On combustion, which fuel never produces pollutants?
A diesel C methane
B hydrogen D petrol
Anglican High School 5073/01/SEC4MYE/14
7
27 The reaction between propene molecules and hydrogen gas releases heat to the
surrounding. The energy profile diagram for the reaction between propene and
hydrogen is shown below:
Energy
reactants
products
Extent of reaction
When would a reaction occur between a propene molecule and a hydrogen
molecule?
A Each time they collide.
B Only when they collide with energy exactly equal to Y.
C Only when they collide with energy equal to or greater than Y.
D Only when they collide with energy equal to or greater than X.
28 In the electrolysis of concentrated sodium chloride solution, 0.12dm3 of hydrogen is
produced at the negative electrode. What is the volume of gas produced at the
positive electrode?
A 0.06dm3 C 0.18dm3
B 0.12dm3 D 0.24dm3
29 The diagram below shows a simple cell.
Copper
electrode
Zinc
electrode
Sulfuric
acid
Which statement about the cell is true?
A Copper is the negative electrode.
B Electrons move from the copper to the zinc electrode.
C The voltage registered will be less than that of a simple cell with magnesium and
copper as electrodes.
D The zinc electrode gains electrons to produce zinc ions.
Anglican High School 5073/01/SEC4MYE/14
8
30 Which gas contributes to a rise in sea levels?
A methane C ozone
B oxides of nitrogen D sulfur dioxide
31 Why are catalytic converters fitted to cars?
A To decrease the amount of carbon dioxide emitted.
B To decrease the amount of oxides of nitrogen emitted.
C To decrease the amount of sulfur dioxide emitted.
D To improve fuel consumption.
32 A sample of air is slowly passed through aqueous sodium hydroxide and then over
heated copper. Which gases are removed by this process?
A carbon dioxide and oxygen
B carbon dioxide and water vapour
C nitrogen and oxygen
D nitrogen and water vapour
33 In the Haber process, nitrogen and hydrogen react to form ammonia. What is the
source of hydrogen?
A air C ethanol
B crude oil D sulfuric acid
34 In which of the following can kerosene be used as the energy source?
A air conditioning units C heavy lorries
B aircraft D power stations
35 Which statement is not true about members in a homologous series?
A They have the same chemical properties.
B They have the same functional group.
C They have the same general formula.
D They have the same physical properties.
Anglican High School 5073/01/SEC4MYE/14
9
36 The structures of 3 compounds are as shown below.
H O H H H O H O
C C H–C–C–O–C–H H–C–O–C–H
H–O H H H H
I II III
Which compounds are isomers?
A I and II C II and III
B I and III D I, II and III
37 A hydrocarbon Q, on heating with a catalyst, forms a solid compound R. Both Q and
R have the same empirical formula. What type of reaction is this?
A addition polymerisation C hydrogenation
B condensation polymerisation D substitution
38 Ethanol is a .....(1)..... which is made from .....(2)..... and .....(3)..... in the laboratory. It
can also be produced by .....(4)..... .
(1) (2) (3) (4)
A fuel ethanoic acid potassium manganate (VII) addition polymerisation
B fuel ethene steam fermentation
C plastic ethanoic acid potassium manganate (VII) addition polymerisation
D plastic ethene steam fermentation
39 Which reagent will react with propanoic acid to produce a 'sweet smelling'
compound that can be used as food flavouring?
A aqueous potassium hydroxide C ethanol
B concentrated sulfuric acid D sodium
40 Glycine is the smallest of the 20 amino acids commonly found in proteins and has
the structure as shown below.
H
H–N–C–C–O–H
H H O
Which shows the correct type of polymerisation it undergoes and the type of linkage
produced?
polymerization linkage
A addition amide
B addition ester
C condensation amide
D condensation ester
Anglican High School 5073/01/SEC4MYE/14
10
END OF PAPER
Anglican High School 5073/01/SEC4MYE/14
You might also like
- 2020 Sec 4 Pure Chemistry SA2 Ngee Ann SecondaryDocument35 pages2020 Sec 4 Pure Chemistry SA2 Ngee Ann SecondaryDORA SIN YU KWOKNo ratings yet
- Nanocatalytic Materials For Energy-Related Small-Molecules Conversions: Active Site Design, Identification and Structure Performance Relationship DiscoveryDocument11 pagesNanocatalytic Materials For Energy-Related Small-Molecules Conversions: Active Site Design, Identification and Structure Performance Relationship DiscoveryJohnNo ratings yet
- A Level Chemistry PDFDocument71 pagesA Level Chemistry PDFPaida Heart50% (8)
- 2019 Sec 4 Pure Chemistry SA2 Fuhua SecondaryDocument37 pages2019 Sec 4 Pure Chemistry SA2 Fuhua SecondaryTimothy CNo ratings yet
- SULIT 4541/1 Chemistry Paper 1 Mei 2007Document22 pagesSULIT 4541/1 Chemistry Paper 1 Mei 2007AMINNo ratings yet
- 2019 Sec 4 Pure Chemistry SA2 Geylang MethodistDocument32 pages2019 Sec 4 Pure Chemistry SA2 Geylang MethodistErOn TaNNo ratings yet
- 5070 w11 QP 12Document12 pages5070 w11 QP 12mstudy123456No ratings yet
- University of Cambridge International Examinations General Certificate of Education Ordinary LevelDocument12 pagesUniversity of Cambridge International Examinations General Certificate of Education Ordinary LevelStuart KanyesigyeNo ratings yet
- 2018 Sec 4 Science Chemistry SA1 Pasir Ris Crest SecondaryDocument34 pages2018 Sec 4 Science Chemistry SA1 Pasir Ris Crest SecondaryAnqi DaiNo ratings yet
- HanksDocument20 pagesHanksRia MandasariNo ratings yet
- Grade 10 O Level Chemistry - Mock Test 1 (7-04-2021)Document29 pagesGrade 10 O Level Chemistry - Mock Test 1 (7-04-2021)Roselyn TrixieNo ratings yet
- 2018 Sec 4 Pure Chemistry SA2 Geylang Methodist SchoolDocument42 pages2018 Sec 4 Pure Chemistry SA2 Geylang Methodist SchoolginkaineoNo ratings yet
- Admiralty Secondary Prelim 2021 ChemistryDocument50 pagesAdmiralty Secondary Prelim 2021 ChemistryAlexisNo ratings yet
- 5070 s10 QP 12 PDFDocument16 pages5070 s10 QP 12 PDFNeural Spark Physics CieNo ratings yet
- 5070 s16 QP 12 PDFDocument16 pages5070 s16 QP 12 PDFNeen Naaz100% (1)
- Chem G10-Quiz1 19-5-2020Document13 pagesChem G10-Quiz1 19-5-20201211200107No ratings yet
- Cambridge International Examinations Cambridge Ordinary LevelDocument16 pagesCambridge International Examinations Cambridge Ordinary LevelRaheel Ahmed KhanNo ratings yet
- Chemistry PDFDocument38 pagesChemistry PDFAddict- ionNo ratings yet
- 4024q1 Specimen PaperdocxDocument12 pages4024q1 Specimen PaperdocxLeses MayNo ratings yet
- O-Levels Chemistry ExemplarDocument40 pagesO-Levels Chemistry Exemplartendaisithole02No ratings yet
- Cambridge O Level: Chemistry 5070/11Document16 pagesCambridge O Level: Chemistry 5070/11Jack DoeNo ratings yet
- North Vista 2015 Prelim Paper 1Document20 pagesNorth Vista 2015 Prelim Paper 1GM MonsterEtaNo ratings yet
- Chem-CGS P1Document17 pagesChem-CGS P1dimpledblissNo ratings yet
- Cambridge O Level: Chemistry 5070/12Document16 pagesCambridge O Level: Chemistry 5070/12pinkNo ratings yet
- 2019 Sec 4 Science Chemistry SA2 Singapore Sports SchoolDocument30 pages2019 Sec 4 Science Chemistry SA2 Singapore Sports School19Y1H GAO CHENZHANGNo ratings yet
- 2019 Sec 4 Science Chemistry SA2 Singapore Sports SchoolDocument30 pages2019 Sec 4 Science Chemistry SA2 Singapore Sports School19Y1H GAO CHENZHANGNo ratings yet
- University of Cambridge International Examinations General Certificate of Education Ordinary Level Chemistry Paper 1 Multiple Choice May/June 2005 1 HourDocument20 pagesUniversity of Cambridge International Examinations General Certificate of Education Ordinary Level Chemistry Paper 1 Multiple Choice May/June 2005 1 Hourkaran79No ratings yet
- Cambridge IGCSE: Chemistry 0620/13Document16 pagesCambridge IGCSE: Chemistry 0620/13Aasil AtifNo ratings yet
- Chemistry June 2004 - Paper 1Document16 pagesChemistry June 2004 - Paper 1theyaasir0% (1)
- 5070 s10 QP 12Document16 pages5070 s10 QP 12Ruby ChongNo ratings yet
- 5070 w05 QP 1Document16 pages5070 w05 QP 1mstudy123456No ratings yet
- Cambridge International Examinations Cambridge International General Certificate of Secondary EducationDocument16 pagesCambridge International Examinations Cambridge International General Certificate of Secondary EducationAhmed MasoudNo ratings yet
- Ejc H1 Chem P1Document11 pagesEjc H1 Chem P1Lim EnningNo ratings yet
- 2020 Yearly Exam PaperDocument22 pages2020 Yearly Exam PaperYu-Tang LinNo ratings yet
- ChemistryDocument14 pagesChemistryAdil ArifNo ratings yet
- Hci H2 Chem P1Document16 pagesHci H2 Chem P1clarissa yeoNo ratings yet
- upload-WCH05 01 Que 201506151446484865Document28 pagesupload-WCH05 01 Que 201506151446484865Sadaf ChowdhuryNo ratings yet
- 5070 w14 QP 12Document16 pages5070 w14 QP 12hakimonlyNo ratings yet
- Cambridge International Examinations Cambridge International General Certificate of Secondary EducationDocument16 pagesCambridge International Examinations Cambridge International General Certificate of Secondary EducationDenver DemisNo ratings yet
- 0620 w12 QP 11Document16 pages0620 w12 QP 11n0tsewNo ratings yet
- University of Cambridge International Examinations General Certificate of Education Ordinary LevelDocument18 pagesUniversity of Cambridge International Examinations General Certificate of Education Ordinary LevelG M Ali KawsarNo ratings yet
- Cambridge IGCSE: Chemistry 0620/23Document16 pagesCambridge IGCSE: Chemistry 0620/23SasukeNo ratings yet
- 5070 s15 QP 12Document13 pages5070 s15 QP 12Saima SohailNo ratings yet
- 0620 s15 QP 13Document16 pages0620 s15 QP 13karan79No ratings yet
- CHEMISTRYDocument41 pagesCHEMISTRYLindsayyNo ratings yet
- 2020 Yearly Exam SolutionsDocument19 pages2020 Yearly Exam SolutionsYu-Tang LinNo ratings yet
- 2021 JC2 Prelim H1 Chemistry Paper 1 QPDocument12 pages2021 JC2 Prelim H1 Chemistry Paper 1 QPShengxin PanNo ratings yet
- 9701 s09 QP 1 PDFDocument16 pages9701 s09 QP 1 PDFAinthu IbrahymNo ratings yet
- Cambridge IGCSE: Chemistry 0620/12Document16 pagesCambridge IGCSE: Chemistry 0620/12Mohammed khaled GhazalNo ratings yet
- 5070 w04 QP 1Document16 pages5070 w04 QP 1mstudy123456No ratings yet
- 2 1 The Diagrams Show Four Different Methods of Collecting GasesDocument12 pages2 1 The Diagrams Show Four Different Methods of Collecting GasesMujeeb SiddiqueNo ratings yet
- Cambridge International General Certificate of Secondary EducationDocument16 pagesCambridge International General Certificate of Secondary Educationlila davinci100% (1)
- Cambridge IGCSE: Chemistry 0620/23Document16 pagesCambridge IGCSE: Chemistry 0620/23mostafa barakatNo ratings yet
- Cambridge International General Certificate of Secondary EducationDocument16 pagesCambridge International General Certificate of Secondary EducationKim Yen GohNo ratings yet
- QC - 2019-20 - Mock - S6 - Chem 1ADocument12 pagesQC - 2019-20 - Mock - S6 - Chem 1AOof GucciNo ratings yet
- 5070 s11 QP 11Document16 pages5070 s11 QP 11chuasioklengNo ratings yet
- Cambridge IGCSE: Chemistry 0620/12Document16 pagesCambridge IGCSE: Chemistry 0620/12190377964No ratings yet
- Chapter 9 Part 5Document2 pagesChapter 9 Part 519Y1H GAO CHENZHANGNo ratings yet
- Electricity QuizDocument5 pagesElectricity Quiz19Y1H GAO CHENZHANGNo ratings yet
- Chapter 9 Pa 6: Why Does Evaporation Cause Cooling?Document1 pageChapter 9 Pa 6: Why Does Evaporation Cause Cooling?19Y1H GAO CHENZHANGNo ratings yet
- HBL Physics MCQ 4octDocument43 pagesHBL Physics MCQ 4oct19Y1H GAO CHENZHANGNo ratings yet
- Magnetism and ElectromagnetismDocument2 pagesMagnetism and Electromagnetism19Y1H GAO CHENZHANGNo ratings yet
- Chapter 9 Part 2Document2 pagesChapter 9 Part 219Y1H GAO CHENZHANGNo ratings yet
- Chapter 9 Pa 1: What Is The Internal Energy of A Substance?Document2 pagesChapter 9 Pa 1: What Is The Internal Energy of A Substance?19Y1H GAO CHENZHANGNo ratings yet
- Chapter 17 - Practical ElectricityDocument2 pagesChapter 17 - Practical Electricity19Y1H GAO CHENZHANGNo ratings yet
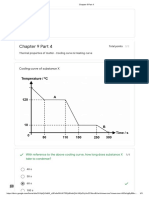
- Chapter 9 Pa 4: Cooling Curve of Substance XDocument3 pagesChapter 9 Pa 4: Cooling Curve of Substance X19Y1H GAO CHENZHANGNo ratings yet
- Green Revolution ImpactDocument4 pagesGreen Revolution ImpactAditya JainNo ratings yet
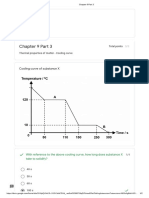
- Chapter 9 Pa 3: Cooling Curve of Substance XDocument2 pagesChapter 9 Pa 3: Cooling Curve of Substance X19Y1H GAO CHENZHANGNo ratings yet
- Chapter 9 Part 2Document2 pagesChapter 9 Part 219Y1H GAO CHENZHANGNo ratings yet
- Chapter 9 Part 5Document2 pagesChapter 9 Part 519Y1H GAO CHENZHANGNo ratings yet
- Chapter 9 Pa 1: What Is The Internal Energy of A Substance?Document2 pagesChapter 9 Pa 1: What Is The Internal Energy of A Substance?19Y1H GAO CHENZHANGNo ratings yet
- Chapter 9 Pa 3: Cooling Curve of Substance XDocument2 pagesChapter 9 Pa 3: Cooling Curve of Substance X19Y1H GAO CHENZHANGNo ratings yet
- Chapter 9 Pa 4: Cooling Curve of Substance XDocument3 pagesChapter 9 Pa 4: Cooling Curve of Substance X19Y1H GAO CHENZHANGNo ratings yet
- Practice Question For MYEDocument2 pagesPractice Question For MYE19Y1H GAO CHENZHANGNo ratings yet
- 2019 Sec 4 Science Chemistry SA2 Singapore Sports SchoolDocument30 pages2019 Sec 4 Science Chemistry SA2 Singapore Sports School19Y1H GAO CHENZHANGNo ratings yet
- A Green Revolution for Africa Through Drought-Resistant MaizeDocument8 pagesA Green Revolution for Africa Through Drought-Resistant Maize19Y1H GAO CHENZHANGNo ratings yet
- 2019 Sec 4 Science Chemistry SA2 Singapore Sports School AnswerDocument7 pages2019 Sec 4 Science Chemistry SA2 Singapore Sports School Answer19Y1H GAO CHENZHANGNo ratings yet
- The Toxic Consequences of The Green Revolution - Indian Agrarian CrisisDocument4 pagesThe Toxic Consequences of The Green Revolution - Indian Agrarian Crisis19Y1H GAO CHENZHANGNo ratings yet
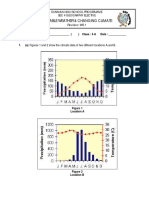
- Revision WS 1 (Weather & Climate) (Qns Only)Document3 pagesRevision WS 1 (Weather & Climate) (Qns Only)19Y1H GAO CHENZHANGNo ratings yet
- 2018 Sec 4 A Math SA2 School of Science and TechnologyDocument58 pages2018 Sec 4 A Math SA2 School of Science and Technology19Y1H GAO CHENZHANGNo ratings yet
- 2018 Sec 4 A Math SA2 School of Science and TechnologyDocument58 pages2018 Sec 4 A Math SA2 School of Science and Technology19Y1H GAO CHENZHANGNo ratings yet
- 2019 Sec 4 Science Chemistry SA2 Singapore Sports School AnswerDocument7 pages2019 Sec 4 Science Chemistry SA2 Singapore Sports School Answer19Y1H GAO CHENZHANGNo ratings yet
- Sec 4 Physics Set B Paper 1: Exam Papers With Worked SolutionsDocument15 pagesSec 4 Physics Set B Paper 1: Exam Papers With Worked Solutions19Y1H GAO CHENZHANGNo ratings yet
- 2019 Sec 4 Science Chemistry SA2 Singapore Sports SchoolDocument30 pages2019 Sec 4 Science Chemistry SA2 Singapore Sports School19Y1H GAO CHENZHANGNo ratings yet
- Microsoft Word - S4PH Set B - 2 - QNDocument20 pagesMicrosoft Word - S4PH Set B - 2 - QN19Y1H GAO CHENZHANGNo ratings yet
- AHS Prelim 2014 P1Document11 pagesAHS Prelim 2014 P119Y1H GAO CHENZHANGNo ratings yet
- MCQs Electromagnetism QuizDocument17 pagesMCQs Electromagnetism Quiz19Y1H GAO CHENZHANGNo ratings yet
- Study of Hydrogen as the First ElementDocument10 pagesStudy of Hydrogen as the First ElementAyush DasNo ratings yet
- IGCSE Cam Chemistry P4 Topical Worksheet 2012-16Document651 pagesIGCSE Cam Chemistry P4 Topical Worksheet 2012-16Piya SabooNo ratings yet
- Photocatalytic Reaction Using CDS and RGO PDFDocument68 pagesPhotocatalytic Reaction Using CDS and RGO PDFNANDITA DOLOI Dept. of Chemical Engg. & Tech., IIT (BHU) VaranasiNo ratings yet
- Aldol Notes PDFDocument8 pagesAldol Notes PDFAna100% (1)
- Psi-Ap-Chemistry-Equilibrium-Multiple-Choice 3Document30 pagesPsi-Ap-Chemistry-Equilibrium-Multiple-Choice 3Tricyver ChienNo ratings yet
- 14 Chemical KineticsDocument6 pages14 Chemical KineticsPriyadharshni RaviNo ratings yet
- Lab Report: Investigation of First Order Response Kinetics: I.AbstractDocument5 pagesLab Report: Investigation of First Order Response Kinetics: I.AbstractHoàng Thu HằngNo ratings yet
- Sulfonation Mechanism of Benzene With SO3 in Sulfuric Acid or Oleum PDFDocument37 pagesSulfonation Mechanism of Benzene With SO3 in Sulfuric Acid or Oleum PDFLaely Dian MarlindawatiNo ratings yet
- Safety Data Sheet: 1. Product IdentificationDocument6 pagesSafety Data Sheet: 1. Product Identificationmohamed AdelNo ratings yet
- Reversible Reactions and EquilibriumDocument8 pagesReversible Reactions and Equilibriummahika gaurNo ratings yet
- Biology Unit 4 Part 1 Last Minute Revision ReallyacademicsDocument67 pagesBiology Unit 4 Part 1 Last Minute Revision ReallyacademicsWill AndyNo ratings yet
- Biochemistry Chapter 4Document16 pagesBiochemistry Chapter 4Jayson AguilarNo ratings yet
- Chapter 11-Reactions of Alcohols: 3° Alcohols Are Resistant To OxidationDocument30 pagesChapter 11-Reactions of Alcohols: 3° Alcohols Are Resistant To Oxidation張湧浩No ratings yet
- Chemistry Vol 2 EM PDFDocument320 pagesChemistry Vol 2 EM PDFTharani ThangamaniNo ratings yet
- Rate of Decomposition of H2O2Document41 pagesRate of Decomposition of H2O2Beeta Moulik100% (1)
- Ujian Bab5 Form 3Document2 pagesUjian Bab5 Form 3Azween SabtuNo ratings yet
- A Level Chemistry Paper 2 Exam 28Document3 pagesA Level Chemistry Paper 2 Exam 28Anthony AndyNo ratings yet
- KNC1063 Organic Chemistry: DR Md. Rezaur RahmanDocument19 pagesKNC1063 Organic Chemistry: DR Md. Rezaur RahmanKunashiny RamashNo ratings yet
- Anti Crease Finishing of Cotton Fabrics Based On Crosslinking of Cellulose With Oxidized SucroseDocument12 pagesAnti Crease Finishing of Cotton Fabrics Based On Crosslinking of Cellulose With Oxidized SucroseNattaya VuthiganondNo ratings yet
- Reactions Alkenes & AlkynesDocument62 pagesReactions Alkenes & AlkynesDeniseNo ratings yet
- A Study On Making of Compact Manual Paper Recycling Plant For Domestic PurposeDocument22 pagesA Study On Making of Compact Manual Paper Recycling Plant For Domestic PurposeEinjhel RamosNo ratings yet
- Condensation Reactions - Organic Chemistry Questions and Answers - SanfoundryDocument10 pagesCondensation Reactions - Organic Chemistry Questions and Answers - SanfoundryMary Francia RicoNo ratings yet
- Class 8 Science Notes Ch-1Document9 pagesClass 8 Science Notes Ch-1deepakatoliaNo ratings yet
- Complete Chem (9) 78-82 - 20200428 - 0001Document5 pagesComplete Chem (9) 78-82 - 20200428 - 0001Wisdom PhanganNo ratings yet
- IITAIIMS 2023 SCREENING CUM SCHOLARSHIP TEST Sample Questions PDFDocument207 pagesIITAIIMS 2023 SCREENING CUM SCHOLARSHIP TEST Sample Questions PDFReeshba Reji0% (1)
- Sir Gerobel Narrative Report in ChemistryDocument19 pagesSir Gerobel Narrative Report in Chemistryangelo aquinoNo ratings yet
- NSS Chemistry Part 4 Acids and Bases - MCDocument35 pagesNSS Chemistry Part 4 Acids and Bases - MCFelix YueNo ratings yet
- One-Pot Multistep Reactions Based on ThiolactonesDocument4 pagesOne-Pot Multistep Reactions Based on ThiolactonesUsman ShaukatNo ratings yet