Professional Documents
Culture Documents
CA2575537C Urea
Uploaded by
Ruchita Poilkar0 ratings0% found this document useful (0 votes)
12 views17 pagesUrea Manufacturing process
Original Title
CA2575537C-urea
Copyright
© © All Rights Reserved
Available Formats
PDF or read online from Scribd
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentUrea Manufacturing process
Copyright:
© All Rights Reserved
Available Formats
Download as PDF or read online from Scribd
0 ratings0% found this document useful (0 votes)
12 views17 pagesCA2575537C Urea
Uploaded by
Ruchita PoilkarUrea Manufacturing process
Copyright:
© All Rights Reserved
Available Formats
Download as PDF or read online from Scribd
You are on page 1of 17
Office de la Propriéts Canadian
Intellectuelle Intellectual Property
du Canada Office
Un organisme ‘An agency of
diindustrie Canada Industry Canada
CA 2575537 C 2013/09/10
«nen 2 575 537
(12) BREVET CANADIEN
CANADIAN PATENT
13
(@6) Date de dépét PCT/PCT Filing Date: 2005/07/22
(67) Date publication PCTIPCT Publication Date: 2006/02/16
(4) Date de délivrance/issue Date: 2013/09/10
(@5) Entrée phase nationale/National Entry: 2007/01/29
(6) N° demande PCT/PCT Application No.: EP 2005/008033
(67) N° publication PCT/PCT Publication No.: 2006/015709
(0) Priorite/Priority: 2004/08/10 (EP04018981.3)
(61) Clint int.cl, CO7C 273/10(2006 01),
C018 3/02 (2006.01), CO1C 1/04(2006.01)
(72) Inventeurs/inventors:
ROMITI, DOMENICO, CH;
STICCHI, PAOLO, CH
(73) Propriétaire/Owner:
UREA CASALE S.A., CH
(74) Agent: NORTON ROSE FULBRIGHT CANADA
LPSENCRL, SRL
(64) Titre : PROCESSUS DE PRODUCTION D'UREE A PARTIR D'AMMONIAC ET DE DIOXYDE DE CARBONE
(64) Title: PROCESS FOR UREA PRODUCTION FROM AMMONIA AND CARBON DIOXIDE
(67) Abrége/Abstract:
‘A process for utea production comprises a fist process step in which ammonia (7) and carbon dioxide (6) are obtained, subjecting
natural gas (1) to reforming treatments (12, 14), and a second step of urea (8a) production from such ammonia (7) and from
Cc an ada np:/epie.ge.ca* Otawa/Gatineau KLAOC9 + hap:eipo.geca 6 pI
CA 2575537 C 2013/09/10
«nen 2 575 537
aye
(67) Abrégé{suite)/Abstract(continued):
carbon dioxide, through a formation of a solution comprising urea and ammonium carbamate in a urea synthesis reactor (20) anda
subsequent decomposition of the ammonium carbamate and. urea recovery, the process comprises the steps of: - treating
combustion smokes (5) comprising carbon dioxide with an aqueous solution (9a) comprising a part (7b) of such ammonia (7),
obtaining an aqueous ammonium carbamate solution (8c); - supplying the solution (Sc) thus obtained to the second process step.
cA 02575537 2007-01-29
(12) INTERNATIONAL APPLICATION PUBL
(19) World Intellectual Property Organization
International Bureau
(43) International Publication Date
16 February 2006 (16.02.2006)
Oe
PcT
{ATION TREATY (PCT)
(10) International Publication Number
WO 2006/015709 Al
GL
International Patent Classification
COIC 1/04, CorB 3702
CO7C 273/10,
21) International Application Numbet
PCTAEP2005/008033
(22) International Filing Date: 22 July 2005 (22.07.2005)
25) Filing Languag English
(26) Publication Language: English
(30) Priority Data:
(03018981.3 10 August 2004 (10.08.2008) EP
(TY) Applicant (for all designated States except US): UREA
CASALE S.A. [CHICH], 6, Via Giulio Pocobell,
(CH-6900 Lugano-Besso (CID.
2)
5)
Inventors; and
Inventors/Applicants
Domenico [CH
Lugano (CHD.
Tesserete, CH-6900 Massagno
(for US only): ROMITI,
1], 9, Via Gaggini da Bissone, CH-6900
STICCHI, Paolo [CHICH]; 19, Via
)
(74) Agent: ZARDI, Marco; M. Zardi & CO. S.A., Via Pda
6, CH-6900 Lugano (CH).
(BL) Designated States (unless otherwise indicated, for every
kind of national protection available}: AE, AG, AL, AM,
AT, AU, AZ, BA, BB, BG, BR, BW, BY, BZ, CA, CH, CN,
€O, CR, CU, CZ, DE, DK, DM, DZ, Et
GR, GD, GE, GH, GM, HR, HU, ID, IL, IN, IS, 1, KE,
KG, KM, KP, KR, KZ, LC, LK, LR, LS, LT, LU, LV, MA,
MK, MN, MW, MX, M
‘OM, PG, PH, PL, PT, RO, RU, S
SM, SY, TJ, TM, TN, TR, TT, TZ, UA, UG, US, UZ, VC,
VN, YU, ZA, 2M, ZW.
(ss)
Designated States (unless otherwise indicated, for every
kind of regional protection available): ARIPO (BW, GH,
RO, SE, SI, SK, TR), OAPI (BE, BI, CF,
GN, GQ, GW, ML, MR, NE, SN, TD, TG),
i, C1LCM,
Publishes
with international search report
For two-letter codes and other abbreviations, refer tothe "Guid
‘ance Notes on Codes and Abbreviations” appearing at the begin-
ning of each regular issue of the PCT Gazette.
WO 2006/015709 At lll
64) 7
PROCI
(57) Abstrac
POR UREA PRODUCTION FROM AMMONIA AND CARBON DIOXIDE,
|\ process for urea production comprises a first process step in which ammonia (7) and earbon dioxide (6) are ob-
tained, subjecting natural gas (1) (0 reforming treatments (I2, 14), and a second step of urea (8a) procuction from such ammonia (7)
and from carbon dioxide, through a formation of solution comprising urea and ammonium carbamate in aurea syathesis reactor
(20) and a subsequent decomposition of the ammonium carbamate and. urea recovery, the process comprises the steps of
ing combustion smokes (5) comprising carbon dioxide with an aqueous solution (9a) comprising a part (7b) of such ammonia (7),
obtaining an aqueous ammonium carbamate solution (9e) ; - supplying the solution (9e) thus obtained to the Second process step.
10
15
20
25
cA 02575537 2007-01-29
WO 2006/015709 PCT/EP2005/008033
Title: “Process for urea production from ammonia and carbon
dioxide”
DESCRIPTION
Field of application
The present invention refers, in its most general aspect,
to a process for urea production from ammonia and carbon
dioxide.
In particular this invention refers to a process for urea
production of the type in which the ammonia and the carbon
dioxide necessary for the urea synthesis are obtained in
the same ambit, in the same processing line, in the same
industrial area or in the same plant that produces said
urea.
More specifically, the present invention concerns a process
of the type considered above, comprising a first step in
which ammonia and carbon dioxide are produced by subjecting
natural gas to a steam reforming treatment, and a second
step of urea production from said ammonia and carbon
dioxide, through respective steps of formation of a
solution of urea and ammonium carbamate in a urea synthesis
reactor and of subsequent decomposition of the ammonium
carbamate and urea recovery.
Prior Art
Processes for urea production from ammonia and carbon
dioxide are well known, and it is known that when the urea
thus obtained is intended for fertilizer production, there
is still a widespread requirement to use natural gas as
primary material of the entire production process and, in
CONFIRMATION COPY
10
15
20
30
cA 02575537 2012-05-22
particular, for the production of ammonia synthesis gases
(hydrogen and nitrogen) .
It is also known that in the aforementioned processes, the
production of synthesis gases is generally carried out by
first subjecting the predetermined natural gas to
subsequent steam reforming treatments and then separating
hydrogen and nitrogen from the carbon dioxide that is
obtained with them through the aforementioned treatments.
This carbon dioxide itself is used with the ammonia for the
urea product ior
However, it is well known that, by using natural gas as
primary material, the ammonia and the carbon dioxide
produced are not in the stoichiometric quantities required
by the subsequent urea synthesis, but generally there is
excess ammonia with respect to C02. Consequently, to
produce urea using all the ammonia produced, it is
necessary to have an additional amount of carbon dioxide.
For such a purpose, the prior art teaches to carry out a
recovery of pure carbon dioxide actually within the ambit
of the same process, in particular from smokes resulting
om the burning of natural gas (methane), burnt to provide
the necessary heat for the steam reforming treatment of the
process natural gas for producing synthesis gases, and
discharged by the reforming treatment itself, in which it
is contained in significant quantities. In particular, said
recovery is carried out by treating (washing) the same
combustion smokes with ethanolamines, for example
monoethanolamine in aqueous solution (MEA).
Whilst advantageous from various points of view, however,
this technique of supplying additional carbon dioxide is
10
15
20
25
30
cA 02575537 2007-01-29
WO 2006/015709 PCT/EP2005/008033
complex to be carried out, laborious and not cost-
effective. Indeed, it necessarily involves a whole series
of chemical reactions variously interconnected and
interrelated, which are difficult to carry out and control,
which require a plant and relative apparatuses that must be
e. to recover pure carbon
designed for just that purpose,
dioxide from the combustion smokes discharged by the
reforming step of the natural gas, as well as for the
elimination of the discharge products (including oxidized
MEA), in considerable quantity, resulting from such a
technique.
Summary of the invention
The technical problem underlying the present invention is
that of devising and providing a process for urea
production from natural gas, having characteristics such as
to allow the aforementioned drawbacks with reference to the
prior art to be overcome.
Such a technical problem is solved, according to the
present invention, by a process for urea production
comprising a first process step in which ammonia and carbon
dioxide are obtained, subjecting natural gas to reforming
treatments, and a second step of urea production from said
ammonia and from carbon dioxide, through a formation of a
solution comprising urea and ammonium carbamate in a urea
synthesis reactor and a subsequent decomposition of the
ammonium carbamate and urea recovery, characterized in that
it comprises the steps of:
- treating combustion smokes comprising carbon dioxide with
an aqueous solution comprising a part of said ammonia,
obtaining an aqueous ammonium carbamate solution;
10
15
20
25
A 02575537 2012-05-22
- supplying the solution thus obtained to said second
process step.
More particularly, there is provided an improved process
for urea production from ammonia and carbon dioxide
comprising a first process step in which ammonia and carbon
dioxide are obtained, which comprises subjecting natural
gas to two reforming treatments, and a second step
comprising urea production from said ammonia and from
carbon dioxide, through formation of a solution comprising
urea and ammonium carbamate in a urea synthesis reactor and
a subsequent decomposition of said ammonium carbamate and
to produce urea; the improvement comprising treating
combustion smokes comprising carbon dioxide with an aqueous
solution comprising a part of said ammonia, such that said
part of said ammonia is reacted with carbon dioxide
recovered from said combustion smokes obtaining an aqueous
ammonia carbamate solution; and supplying the aqueous
ammonium carbamate solution thus obtained to the second
process step for urea production.
The characteristics and the advantages of the process
according to the invention shall become clearer from the
following description of an embodiment thereof, made with
reference to the attached drawings, given for indicating
and not limiting purposes.
Brief description of the drawings
In such drawings:
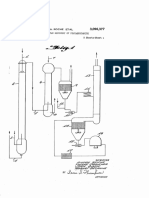
- figure 1 shows the block diagram of a process for urea
production from ammonia, according to the present
invention;
10
15
A 02575537 2012-05-22
- figure 2 schematically shows units operating in the
process of figure 1, according to the present invention.
Detailed description of a preferred embodiment
With reference to figure 1, a process according to the
present invention, for urea production, essentially
comprises two process steps, variously interconnected, as
shall become clear in the rest of the description. A first
step in which, from natural gas, ammonia is produced and at
the same time substantially pure carbon dioxide is
obtained; and a second step in which urea is produced,
using the ammonia and the carbon dioxide, generated in said
previous first step.
In the first step, carried out in an ammonia plant globally
indicated with 1, a natural gas 2, for example methane,
after possible desulphurisation in a desulphurisation unit
10, is subjected, in a totally conventional way, to
10
20
25
cA 02575537 2012-05-22
successive catalytic reforming treatments, firstly with
steam 3 in a primary reformer 12 and then with air 4 in a
secondary reformer 14.
The heat necessary for the primary reforming treatment
of the process natural gas is obtained by burning a further
flow of natural gas (methane), not represented, thus
generating combustion smokes 5 comprising carbon dioxide;
whereas a gaseous flow essentially comprising hydrogen,
nitrogen and carbon dioxide is discharged from the
secondary reformer 14. Hydrogen and nitrogen, freed from
the carbon dioxide 6 through a conventional decarbonation
treatment in a decarbonation unit 16, for example with MEA,
ave supplied to a reactor 18, for synthesizing the ammonia
7.
The ammonia 7 and the carbon dioxide 6 at the outlet from
the first process step schematically described above, are
supplied to the second process step, for the urea
production.
It should be remembered that, as known, the ammonia and the
carbon dioxide obtained from natural gas in the
aforementioned way, are not in the quantitative proportions
stoichiometrically provided for the urea production, but
the ammonia is in excess with respect to the carbon
dioxide.
For this reason, in the second process step, carried out in
a urea plant globally indicated with 8, only a part 7a of
the ammo: and all of the carbon dioxide produced in the
first process step, are made to react in a per se known
way, in an appropriate synthesis reactor 20, obtaining a
solution comprising urea, ammonium carbamate and free
ammonia. The ammonium carbamate present in said solution
10
15
20
30
cA 02575537 2012-05-22
decomposed and the resulting Carbon dioxide and ammonia
vapours, together with the free ammonia, are condensed in a
decomposition and recovery unit 22 and are recycled to the
synthesis reactor 20. The process solution is then
subjected to concentration in a concentration unit 24 in
which an aqueous solution comprising residual ammonium
carbamate is separated from a concentrated urea solution.
The concentrated urea solution is then sent, for example,
to a granulation stage 26 where solid urea (granules) 8a is
obtained, which is then packaged. The aqueous solution
om the aforementioned urea concentration
resulting
treatment in the unit 24 is, on the other hand, sent to a
water treatment unit 34, in which the residual ammonium
carbamate still present in the solution is separated and
recycled to the synthesis reactor 20 through the
decomposition and recovery unit 22, whereas the water thus
purified is discharged from the urea plant 8 through the
line 36
In order to use, for the urea. production, all of the
ammonia 7 produced in the aforementioned first process
step, in other words also the ammonia 7b stoichiometrically
in excess with respect to the carbon dioxide, in accordance
with the present invention, preferably said excess ammonia
7b is advantageously used to "recover" the carbon dioxide
present in combustion smokes.
Such combustion smokes can be smokes generated for
obtaining heat through combustion of natural gas in said
first and/or second process step of the process itself, or
they can also be generated outside of the industrial area
in which the process of the invention is carried out.
10
15
20
25
cA 02575537 2007-01-29
WO 2006/015709 PCT/EP2005/008033
In the example of figure 1, the combustion smokes 5
generated by the primary reforming treatment of the natural
gas are advantageously used. Alternatively, for example,
the combustion smokes of boilers present in the plant could
be used.
Advantageously and according to a preferred embodiment of
the aforementioned “recovery”, carried out in a carbon
dioxide recovery section globally indicated with 9 and
shown in greater detail in figure 2, the combustion smokes
5, purified in a DeNOx purifier 28 (per se known and
therefore not described in detail) from the nitrogen oxides
possibly contained in them and cooled in a heat exchanger
30, are treated in a washing tower 32 with an aqueous
solution 9a of said excess ammonia.
In the example of figure 1, the aqueous solution 9a for
washing the combustion smokes is obtained by mixing the
excess ammonia 7b coming from the ammonia plant 1 with an
aqueous ammonium carbamate solution 9b coming from the
concentration unit 24 of the urea plant 8 associated with
it. Such mixing can take place outside of the washing tower
32 as indicated in figure 1, or even inside the tower 32
itself.
In other words, the combustion smokes 5 are washed with
such an ammonia solution 9a, in which a large amount of the
carbon dioxide comprised in said smokes is absorbed and
reacts to form a more concentrated ammonium carbamate
aqueous solution 9c.
The smokes 9d, impoverished of carbon dioxide, are then
xeleased into the atmosphere in a per se conventional way.
10
15
20
25
30
cA 02575537 2007-01-29
WO 2006/015709 PCT/EP2005/008033
As an alternative to the example of figure 1, it is also
possible to provide the use of an ammonia solution 9a
obtained using a part of ammonia 7 in a smaller amount with
respect to said excess 7b, or, in a greater amount than it,
for example through the use of an additional amount of
ammonia coming from the outside of the ammonia plant 1.
In accordance with another characteristic of the present
invention, the ammonium carbamate solution 9c in which the
carbon dioxide of the combustion smokes 5 has been
absorbed, is supplied to the second process step.
More specifically, the ammonium carbamate solution 9c is
advantageously supplied to the urea synthesis reactor 20,
preferably following passage through the waters treatment
unit 34 and the decomposition and recovery unit 22 together
with the ammonium carbamate separated in said waters
treatment unit 34. In other words, preferably, the aqueous
carbamate solution 9c is combined with the recycled aqueous
carbamate solution of the urea plant 8, which follows a
path that conventionally passes from the waters treatment
unit 34 and from the decomposition and recovery unit 22.
Alternatively, according to a further embodiment of the
invention, not represented, the aqueous ammonium carbamate
solution 9c is supplied to another part of the urea plant,
for example to an additional decomposition unit of the
ammonium carbamate (not represented) in which the vapours
comprising carbon dioxide and ammonia are then supplied to
the synthesis reactor following suitable condensation.
The advantages achieved by the process for urea production
fundamentally consist of the fact that all of the ammonia
produced in the first step of said process is used for urea
10
15
20
cA 02575537 2012-05-22
production in the second step thereof and the fact that the
“recovery” of carbon dioxide from combustion smokes, for
example from the smokes discharged from the primary
reformer, is easy and cost-effective to carry out, not
requiring the complicated operations provided by the prior
art.
Moreover, the aforementioned process can be carried out
through’ plants designed ex novo, ensuring the possibility
of selecting, without any restriction, the amount of
ammonia to be converted into urea, or through existing
plants appropriately equipped with a simple new coz
“recovery” section.
Another advantage of the invention is linked to the fact
that in the carbon dioxide recovery section 9 an exothermal
carbamate formation reaction develops: the heat freed in
such a reaction can thus be used, for example to produce
steam.
A 02575537 2012-05-22
CLAIMS
1. An improved process for urea production from ammonia
and carbon dioxide comprising a first process step in which
ammonia and carbon dioxide are obtained, which comprises
subjecting natural gas to two reforming treatments, and a
second step comprising urea production from said ammonia and
from carbon dioxide, through formation of a solution
comprising urea and ammonium carbamate in a urea synthesis
reactor and a subsequent decomposition of said ammonium
carbamate and to produce urea; the improvement comprising
treating combustion smokes comprising carbon dioxide
with an aqueous solution comprising a part of said ammonia,
such that said part of said ammonia is reacted with carbon
dioxide recovered from said combustion smokes obtaining an
aqueous ammonia carbamate solution; and
supplying the aqueous ammonium carbamate solution thus
obtained to the second process step for urea production.
2. The process according to claim 1, wherein said aqueous
ammonium carbamate solution is supplied to the urea
synthesis reactor.
3. The process according to claim 1, wherein said aqueous
ammonium carbamate solution is supplied to the urea
synthesis reactor following passage through a water
treatment unit and a decomposition and recovery unit of said
second step of urea production.
4. The process according to claim 1, wherein said
combustion smokes comprising carbon dioxide are treated for
A 02575537 2012-05-22
removal of any nitrogen oxides contained in them, before
said treatment with the aqueous ammonia solution.
5. The process according to claim 1, wherein said aqueous
ammonia solution is obtained by mixing said part of ammonia
with an aqueous ammonium carbamate solution coming from a
concentration unit of said second step of urea production.
6. The process according to claim 1, wherein said
combustion smokes are generated by said reforming
treatments.
7. The process according to claim 1, wherein said
combustion smokes are produced in said first process step,
or second process step or both process steps of the process
itself.
cA 02575537 2007-01-29
WO 2006/015709 PCT/EP2005/008033
Fig. 2 ae
You might also like
- Grit: The Power of Passion and PerseveranceFrom EverandGrit: The Power of Passion and PerseveranceRating: 4 out of 5 stars4/5 (588)
- Salt Purification - 3Document4 pagesSalt Purification - 3Ruchita PoilkarNo ratings yet
- The Yellow House: A Memoir (2019 National Book Award Winner)From EverandThe Yellow House: A Memoir (2019 National Book Award Winner)Rating: 4 out of 5 stars4/5 (98)
- US3743699Document12 pagesUS3743699Ruchita PoilkarNo ratings yet
- The Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeFrom EverandThe Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeRating: 4 out of 5 stars4/5 (5795)
- US4343954 FormaldehydeDocument4 pagesUS4343954 FormaldehydeRuchita PoilkarNo ratings yet
- Never Split the Difference: Negotiating As If Your Life Depended On ItFrom EverandNever Split the Difference: Negotiating As If Your Life Depended On ItRating: 4.5 out of 5 stars4.5/5 (838)
- US2462413 FormaldehydeDocument5 pagesUS2462413 FormaldehydeRuchita PoilkarNo ratings yet
- Hidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceFrom EverandHidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceRating: 4 out of 5 stars4/5 (895)
- Salt Purification - 2Document8 pagesSalt Purification - 2Ruchita PoilkarNo ratings yet
- The Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersFrom EverandThe Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersRating: 4.5 out of 5 stars4.5/5 (345)
- US3779883 PentaDocument6 pagesUS3779883 PentaRuchita Poilkar100% (1)
- Shoe Dog: A Memoir by the Creator of NikeFrom EverandShoe Dog: A Memoir by the Creator of NikeRating: 4.5 out of 5 stars4.5/5 (537)
- Ei. A. G : 1,202,995. Patented 0ct, 31, 1916Document5 pagesEi. A. G : 1,202,995. Patented 0ct, 31, 1916Ruchita PoilkarNo ratings yet
- US8367867 - UreaDocument12 pagesUS8367867 - UreaRuchita PoilkarNo ratings yet
- The Little Book of Hygge: Danish Secrets to Happy LivingFrom EverandThe Little Book of Hygge: Danish Secrets to Happy LivingRating: 3.5 out of 5 stars3.5/5 (400)
- EP0435008A1 UreaDocument8 pagesEP0435008A1 UreaRuchita PoilkarNo ratings yet
- Elon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureFrom EverandElon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureRating: 4.5 out of 5 stars4.5/5 (474)
- United States PatentDocument10 pagesUnited States PatentRuchita PoilkarNo ratings yet
- A Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryFrom EverandA Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryRating: 3.5 out of 5 stars3.5/5 (231)
- Volumetric Determination of NitrateDocument3 pagesVolumetric Determination of NitrateRuchita PoilkarNo ratings yet
- On Fire: The (Burning) Case for a Green New DealFrom EverandOn Fire: The (Burning) Case for a Green New DealRating: 4 out of 5 stars4/5 (74)
- US5741956 PentaerythritolDocument5 pagesUS5741956 PentaerythritolRuchita PoilkarNo ratings yet
- The Emperor of All Maladies: A Biography of CancerFrom EverandThe Emperor of All Maladies: A Biography of CancerRating: 4.5 out of 5 stars4.5/5 (271)
- US2373717Document2 pagesUS2373717Ruchita PoilkarNo ratings yet
- Filed June 18, 1954: Feb. 26, 1957 I, Christoffel. 2,782,918Document4 pagesFiled June 18, 1954: Feb. 26, 1957 I, Christoffel. 2,782,918Ruchita PoilkarNo ratings yet
- Formaldehyde - H2CO - PubChemDocument95 pagesFormaldehyde - H2CO - PubChemRuchita PoilkarNo ratings yet
- Devil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaFrom EverandDevil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaRating: 4.5 out of 5 stars4.5/5 (266)
- Para-Cumylphenol (PCP) : Typical Properties Property TypicalDocument1 pagePara-Cumylphenol (PCP) : Typical Properties Property TypicalRuchita PoilkarNo ratings yet
- The Unwinding: An Inner History of the New AmericaFrom EverandThe Unwinding: An Inner History of the New AmericaRating: 4 out of 5 stars4/5 (45)
- By Swill 39Re-A-2J: July 2, 1963 A. Roche Etal 3,096,377Document5 pagesBy Swill 39Re-A-2J: July 2, 1963 A. Roche Etal 3,096,377Ruchita PoilkarNo ratings yet
- Team of Rivals: The Political Genius of Abraham LincolnFrom EverandTeam of Rivals: The Political Genius of Abraham LincolnRating: 4.5 out of 5 stars4.5/5 (234)
- Modelagem Fuzzy Aplicada Na Detec o Da Vulnerabildade Eros o CosteiraDocument172 pagesModelagem Fuzzy Aplicada Na Detec o Da Vulnerabildade Eros o CosteiraRuchita PoilkarNo ratings yet
- United States Patent Office: Patented Mar. 13, 1951 ... ."Document2 pagesUnited States Patent Office: Patented Mar. 13, 1951 ... ."Ruchita PoilkarNo ratings yet
- Ijrras 7 2 15Document10 pagesIjrras 7 2 15Ruchita PoilkarNo ratings yet
- The World Is Flat 3.0: A Brief History of the Twenty-first CenturyFrom EverandThe World Is Flat 3.0: A Brief History of the Twenty-first CenturyRating: 3.5 out of 5 stars3.5/5 (2259)
- TAM BasicTheory Applications 2019Document310 pagesTAM BasicTheory Applications 2019Ruchita PoilkarNo ratings yet
- The Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreFrom EverandThe Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreRating: 4 out of 5 stars4/5 (1090)
- VPS JtacDocument9 pagesVPS JtacRuchita PoilkarNo ratings yet
- United States Patent (10) Patent No.: US 7,875,163 B2: Gilliam Et Al. (45) Date of Patent: Jan. 25, 2011Document26 pagesUnited States Patent (10) Patent No.: US 7,875,163 B2: Gilliam Et Al. (45) Date of Patent: Jan. 25, 2011Ruchita PoilkarNo ratings yet
- US20110091366A1Document32 pagesUS20110091366A1Ruchita PoilkarNo ratings yet
- March 3, 1936. J. W. Hayes E Al 2,032,699: W. We Arza Wa Wag 562ufzarzDocument4 pagesMarch 3, 1936. J. W. Hayes E Al 2,032,699: W. We Arza Wa Wag 562ufzarzRuchita PoilkarNo ratings yet
- The Sympathizer: A Novel (Pulitzer Prize for Fiction)From EverandThe Sympathizer: A Novel (Pulitzer Prize for Fiction)Rating: 4.5 out of 5 stars4.5/5 (121)
- Her Body and Other Parties: StoriesFrom EverandHer Body and Other Parties: StoriesRating: 4 out of 5 stars4/5 (821)