Professional Documents
Culture Documents
Retinal Laser
Uploaded by
Sandra BabuOriginal Description:
Original Title
Copyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
Retinal Laser
Uploaded by
Sandra BabuCopyright:
Available Formats
CLINICAL CASE OPHTHALMOLOGY
RETINAL LASER PHOTOCOAGULATION IN MANAGEMENT OF EALES' DISEASE
Takhchidi KhP, Takhchidi EKh, Kasminina TA, Tebina EP , Mokrunova MV
Pirogov Russian National Medical Research University, Moscow, Russia
Eales’ disease is an idiopathic occlusive inflammatory vasculopathy resulting in peripheral retinal ischemia, neovascularization, recurrent hemophthalmos, and
proliferative tissue formation. It is often complicated by tractional retinal detachment, secondary glaucoma, and central retinal vein occlusion. The management of
patients with Eales’ disease includes mainly glucocorticosteroid therapy, the use of angiogenesis inhibitors, vitreoretinal surgery and laser photocoagulation. The
clinical case reported demonstrates the potential of retinal laser photocoagulation for treatment of the Eales’ disease in the ischemic and proliferative stages. The
results of retinal laser photocoagulation used as monotherapy demonstrate the clinical and functional indices improvement: enhanced visual acuity, stabilized central
retinal sensitivity value, restored clarity to the ocular media, regression of neovascularization and macular edema in the patient’s eye being in the proliferative (3b)
stage, and the process stabilization in the eye being in the ischemic (2a) stage of the disease.
Keywords: Eales' disease, laser photocoagulation, optical coherence tomography, fluorescein angiography
Author contribution: Takhchidi KhP — study concept and design, manuscript editing; Takhchidi EKh — literature analysis; Tebina EP — manuscript writing;
Kasminina TA — laser treatment; Mokrunova MV — data acquisition and processing.
Compliance with ethical standards: the patient submitted informed consent to laser treatment and personal data processing.
Correspondence should be addressed: Ekaterina P. Tebina
Volokolamskoe shosse, 30, str. 2, Moscow, 123182; ekaterinatebina@mail.ru
Received: 20.09.2020 Accepted: 12.10.2020 Published online: 23.10.2020
DOI: 10.24075/brsmu.2020.063
ПРИМЕНЕНИЕ ЛАЗЕРНОЙ КОАГУЛЯЦИИ СЕТЧАТКИ ПРИ БОЛЕЗНИ ИЛЗА
Х. П. Тахчиди, Е. Х. Тахчиди, Т. А. Касмынина, Е. П. Тебина , М. В. Мокрунова
Российский национальный исследовательский медицинский университет имени Н. И. Пирогова, Москва, Россия
Болезнь Илза — идиопатическая окклюзионная воспалительная васкулопатия, которая приводит к периферической ишемии сетчатки,
неоваскуляризации, рецидивирующим гемофтальмам, развитию пролиферативной ткани и нередко осложняется тракционной отслойкой
сетчатки, вторичной глаукомой, окклюзией центральной вены сетчатки. В основном при ведении пациентов с данной патологией используют
глюкокортикостероидную терапию, ингибиторы ангиогенеза, витреоретинальную хирургию и лазерную коагуляцию. Представленный клинический
случай демонстрирует возможность использования лазерной коагуляции сетчатки при лечении болезни Илза на ишемической и пролиферативной
стадиях. Полученные результаты применения лазерной коагуляции сетчатки в качестве монотерапии свидетельствуют об улучшении клинико-
функциональных показателей: увеличении остроты зрения, стабилизации показателя центральной светочувствительности сетчатки, восстановлении
прозрачности оптических сред, регрессе неоваскуляризации и макулярного отека на глазу с пролиферативной стадией (3b), а также стабилизации
процесса на глазу в стадии ишемии (2а).
Ключевые слова: болезнь Илза, лазерная коагуляция, оптическая когерентная томография, флюоресцентная ангиография
Вклад авторов: Х. П. Тахчиди — концепция и дизайн исследования, редактирование текста; Е. Х. Тахчиди — анализ литературных данных; Е. П. Тебина —
написание текста; Т. А. Касмынина — лазерное лечение пациента; М. В. Мокрунова — сбор и обработка материала.
Соблюдение этических стандартов: от пациента получено согласие на лазерное лечение и обработку персональных данных.
Для корреспонденции: Екатерина Павловна Тебина
Волоколамское шоссе, д. 30, корп. 2, г. Москва, 123182; ekaterinatebina@mail.ru
Статья получена: 20.09.2020 Статья принята к печати: 12.10.2020 Опубликована онлайн: 23.10.2020
DOI: 10.24075/vrgmu.2020.063
Eales’ disease is an idiopathic occlusive inflammatory characterized by a number of manifestations of varying intensity:
vasculopathy, which results in peripheral retinal ischemia, inflamed veins, ischemia and retinal neovascularization.
neovascularization, recurrent hemophthalmos and proliferative The listed above manifestations often result in the following
tissue formation [1, 2]. complications: recurrent retinal and vitreous hemorrhages,
The literature indicates that the disorder is most common in traction/rhegmatogenous retinal detachment, rubeosis iridis
healthy young males in their second decade of life, and in 90% and secondary glaucoma [8]. In some cases, central retina is
of patients both eyes are affected [3]. affected, which results in macular edema [9].
The disorder was first described by British ophthalmologist The “gold standard” in the Eales’ disease detection is
Henry Eales in 1880 [4]. Etiopathogenesis of the Eales’ disease fluorescein angiography (FA) allowing one to assess blood flow
is not completely understood. In recent years, as a result of and damage to retinal blood vessels: dye transudation, retinal
immunological, molecular biological and biochemical studies, vascular tortuosity and telangiectasia, vascular shunt, venous
the role of human leukocyte antigen, autoimmune mechanisms, stasis, ischemia, retinal neovascularization [10–12].
Mycobacterium tuberculosis and free radicals in the disease In 2007, the new Eales’ disease classification was proposed
pathogenesis has been shown [5, 6]. The natural course of the based on the ophtalmoscopy and FA data [13].
disease is quite variable, with alternating periods of remission Stage 1
and exacerbation [7, 3]. (1a) Periphlebitis of small caliber vessels
According to the literary sources, the disorder usually (1b) Periphlebitis of large caliber vessels with superficial
presents with involvement of peripheral retina and is retinal hemorrhages
90 BULLETIN OF RSMU | 5, 2020 | VESTNIKRGMU.RU
КЛИНИЧЕСКИЙ СЛУЧАЙ ОФТАЛЬМОЛОГИЯ
А B
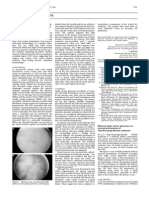
Fig. 1. FA, right eye. А. Optic disc with sharp margins, vascular calibers not altered, A/V ratio 2/3, normal macular area. B. Dye extravasation and aneurisms are
visualized in the area of altered blood vessels in the peripheral fundus (yellow arrow)
Stage 2 regression of retinal neovascularization and vitreoretinal traction
(2а): Capillary nonperfusion after laser photocoagulation is detected in 80–90% of patients
(2b): Neovascularization of the disc and/or retina [16, 23–25]. Moreover, bilateral photocoagulation in the
Stage 3 ischemic areas of asymptomatic eyes is an effective measure
(3а): Fibrovascular proliferation for prevention of the Eales’ disease complications [16, 26]. In
(3b): Hemophthalmos this case study we attempted to assess the effectiveness and
Stage 4 safety of retinal laser photocoagulation for management of
(4а): Traction and/or rhegmatogenous retinal detachment Eales’ disease in the ischemic (2a) and proliferative (3b) stages.
(4b): Rubeosis iridis, neovascular glaucoma, complicated
cataract, optic atrophy Clinical case
The current diagnosis procedures together with wide range
of treatment options significantly improve the Eales’ disease Patient А., aged 20, complaining of floaters and decreased
prognosis and outcome [3, 7]. The management tactics visual acuity in his left eye contacted the Scientific Research
depends on the stage of the disease [14–20] and includes Center for Ophthalmology of Pirogov Russian National Medical
the following: taking glucocorticosteroids (systemic and/or Research University. It was also known from his case history
periocular) in the stage of inflammation [7]; intravitreal injection that the described manifestations emerged spontaneously
of angiogenesis inhibitor [16]; vitreoretinal surgery (in patients and persisted for six months. The patient seeked medical
with recurrent hemophthalmos, vitreoretinal traction and/or assistance from ophthalmologist at his place of residence,
retinal detachment) [21]. and was diagnosed with uveitis of unknown etiology in both
Laser photocoagulation is one of the first-line therapeutic eyes. He received conservative management, which included
options for Eales’ disease in the ischemic and proliferative administration of glucocorticosteroids. No response to
stages [3, 15, 16, 19–22]. According to the foreign authors, treatment was observed.
А B
Fig. 2. А. IR reflectance image of fundus, right eye. Optic disc with sharp margins, normal macular area. B. OCT scan of the right eye. Cross-section image with normal
optic disc and macular area
ВЕСТНИК РГМУ | 5, 2020 | VESTNIKRGMU.RU 91
CLINICAL CASE OPHTHALMOLOGY
А B C
Fig. 3. FA, left eye. А. Optic disc with blurry margins; altered vascular caliber, petaloid (flower-like) pattern of hyperfluorescence is visible through hemophtalmos in
central retina (yellow arrow). B. Periphlebitis with dye extravasation across the visible periphery (blue arrows). C. Protruding proliferative lesion with blurry margins
visualized as a dense hyperfluorescent focus in the clock hour sector 5 (red arrow); hypofluorescent areas corresponding to hemorrhagic foci
The patient underwent complex assessment by the following detected in the paravasal area, the artery to vein (A/V) ratio
methods: visometry aimed at determining the best corrected was 2/3. Aneurysmal dilations and ischemic lesions were
visual acuity (BCVA), ophthalmoscopy using the MaxField observed in peripheral retina. The venous phase FA detected
78D lens (Ocular Inc.; USA), fluorescein angiography (FA) contrast media extravazation and ischemic lesions in the area
and spectral domain optical coherence tomography (SDOCT) of altered vasculature involving the full circumference of the
imaging using the Spectralis HRA+OCT, OCT2 platform with peripheral retina (Fig. 1). SDOCT revealed normal macular
a scanning speed of 85,000 Hz (Heidelberg Engineering; profile, structured retinal layers, and retinal thickness of
Germany), computer perimetry using the Humphrey Field 310 µm (Fig. 2).
Analyzer II (Carl Zeiss Meditec Inc.; USA) and the 30-2 SITA- Vizualization during ophthalmoscopic examination OS
Standard test, computer microperimetry and assessment of was hampered by partial hemophthalmos. The optic disc was
central light sensitivity using the МAIA system (CenterVue Inc.; hyperemic, blurry, and protruded into the vitreous chamber.
Italy). The patient had normal chest x-ray and negative Mantoux There was a fibrous cord over the optic disc extending to the
test. Serology testing revealed no changes. The patient did not inferior outer quadrant of the retina, the macular area was
associate the onset of the disease with anything, and he had blurry. The veins were dilated and tortuous. In peripheral retina,
no hereditary taint. Laser photocoagulation was performed there was a protruding lesion with blurry margins, retinal and
using the VISULAS Trion laser workstation working in the 532 preretinal hemorrhages in the clock hour sector 5. The venous
nm mode (Carl Zeiss; Germany). phase FA detected the hyperfluorescent optic disc. The petaloid
At initial examination, the BCVA in the right eye (OD) was (flower-like) pattern of hyperfluorescence (macular edema) was
1.0, and in the left eye (OS) it was 0.1. Biomicroscopy OU observed in the central retina. Hyperfluorescent focus with
revealed no pathological changes in the anterior segment of hypofluorescent areas was detected on the periphery in the
both eyes. lower sector (Fig. 3). SDOCT revealed the retinal thickness
Ophthalmoscopy OD revealed pale-pink optic disc with increase up to 600 µm in the macular area, and cystic cavities
sharp margins, normal macular reflex. Pathological reflex was in the outer and inner nuclear layers (Fig. 4).
А B C
Fig. 4. А. IR reflectance image of fundus, left eye: partial hemophthalmos, fibrous cord over the optic disc extending to the inferior outer quadrant of the retina (yellow
arrow). B. IR reflectance image of fundus, left eye: retinal veins dilated and tortuous (blue arrows). C. Macula OCT scan, left eye: cystoid edema 600 µm
92 BULLETIN OF RSMU | 5, 2020 | VESTNIKRGMU.RU
КЛИНИЧЕСКИЙ СЛУЧАЙ ОФТАЛЬМОЛОГИЯ
Fig. 5. Macula OCT scan, right eye: normal macular profile, structured retinal layers; no signs of clinical worsening have been revealed within the one month follow-up period
Taking into account the complaints, medical history and parameters were used for central retina in OS: the power was
complex ophthalmological assessment data, the patient 50–100 mW, the exposure time was 0.05–0.1 s, the spot size
was diagnosed with Eales’disease, stage 2a (ischemic was 100 µm, and the distance between the spots was 150 µm.
areas detected by FA), OD, and stage 3b (proliferation and After a month of treatment BCVA OD was 1.0, and BCVA
hemophthalmos), OS. OS had increased to 0.7. Ophthalmoscopy OD revealed pale-
We agreed to perform laser photocoagulation. The extent of pink optic disc with sharp margins. The retinal A/V ratio was
coagulation was determined by the amount of damage to retina. 2/3. The macular anatomy was intact. Pigmented coagula
In OD (stage 2a) the extensive laser photocoagulation of the were observed across the peripheral retina, aneurysms and
peripheral retina was performed (single session), in OS (stage ischemic lesions were sealed (Fig. 5). Examination OS revealed
3b) the panretinal photocoagulation (four sessions at monthly declined hemophtalmos; the optic disc was pale-pink with
intervals) was carried out. The following energy parameters sharp margins; the fibrous cord became smaller. The A/V
were used in OD: the power was 100 mW, the exposure time ratio was 2/3. Partial regression of macular edema and lightly
was 0.1 s, the spot size was 200 µm, the distance between the pigmented coagula were detected in the central retina (except
spots was 300 µm, and the total number of burns was 500. the avascular zone). Pigmented coagula were revealed in the
The following energy parameters were used in OS: the power peripheral retina, the protruding lesion was sealed (Fig. 6).
was 100–120 mW, the exposure time was 0.1 s, the spot size After two years BCVA OU was 1.0. Biomicroscopy OU
was 200 µm, the distance between the spots was 300 µm, revealed no pathological changes in the anterior segment, the
and the total number of burns was 3,000. The following energy ocular media were clear.
А B
Fig. 6. А. IR reflectance image of fundus, left eye: declined hemophtalmos detected during a one month follow-up visit. B. Macula OCT scan, left eye: macular edema
reduced from 600 to 500 µm
ВЕСТНИК РГМУ | 5, 2020 | VESTNIKRGMU.RU 93
CLINICAL CASE OPHTHALMOLOGY
Fig. 7. Macula OCT scan, right eye: normal macular profile, structured retinal layers; no signs of clinical worsening have been revealed during the 2-year
follow-up period
SDOCT detected no signs of clinical worsening in OD (Fig. 7). was reported by many foreign [14, 27] and national authors [28,
Ophthalmoscopy revealed pigmented coagula with sharp 29]. However, the energy parameters of laser treatment play
margins in the peripheral retina, no new lesions were observed. a vital part in the prognosis of the disease. The use of “rigid”
The central retinal sensitivity was 25.7 dB. retinal laser photocoagulation may result in complications,
Ophthalmoscopy OS revealed pale-pink optic disc with such as exudative retinal detachment, iatrogenic choroidal
sharp margins, the fibrous cord over the optic disc became neovascularization, cystoid macular edema, epiretinal fibrosis,
smaller. The retinal A/V ratio was 2/3. There were lightly visual field deficits, deterioration in color vision and decline
pigmented coagula in the macular area (except the avascular in contrast sensitivity [30–32]. According to literary sources,
zone). SDOCT detected restored macular profile, and regression the most commonly used spot size is 400–500 µm, and the
of edema (Fig. 8). The fibrotic neovascularization focus with proposed pulse duration is 0.15–0.2 s with an interpulse
sharp margins was observed in the peripheral retina (Fig. 9). interval of 0.15–0.3 s [3]. Similar energy parameters have been
Pigmented coagula were detected in the paravasal area and used in other studies: spot size of 400 µm, pulse duration of
across the peripheral retina. The central retinal sensitivity value 0.15 s, and the individually adopted average power value of
was 25.4 dB. 160–200 mW [28, 29].
Computer perimetry OU revealed no visual field deficits. In this study, in contrast to the international experience,
we performed laser photocoagulation using the smaller spot
Discussion size (200 µm), lower power (100–120 mW) and exposure
time of 0.1 s in order to improve the clinical and functional
Successful implementation of laser photocoagulation as treatment results in patient with Eales’ disease. The retinal laser
monotherapy for patients not responsive to glucocorticosteroids photocoagulation safety in patient with stage 2a (ischemic) and
А B
Fig. 8. А. Macula OCT scan, left eye: regression of cystoid macular edema, restored anatomic structure of retinal layers. B. Optic disc OCT, left eye: persisting fibrous
cord over the optic disc (yellow arrow)
94 BULLETIN OF RSMU | 5, 2020 | VESTNIKRGMU.RU
КЛИНИЧЕСКИЙ СЛУЧАЙ ОФТАЛЬМОЛОГИЯ
3b (proliferative) Eales’ disease and preserved retinal function
were confirmed by advanced diagnosis methods: computer
perimetry (no visual field deficits), computer microperimetry
(preserved central retinal sensitivity), and optical coherence
tomography imaging (regression of cystoid macular edema).
The proposed energy parameters made it possible to
obtain good clinical and functional results within the long-term
postoperative period, including the improved BCVA, stabilized
central retinal sensitivity value, restored clarity to the ocular
media, regression of neovascularization and macular edema in
the eye being in the proliferative (3b) stage, and the process
stabilization in the eye being in the ischemic (2a) stage of the
disease.
CONCLUSION
The results obtained suggest that the use of retinal laser
photocoagulation as monotherapy for patients with Eales’
disease in the ischemic (2a) and proliferative (3b) stages
contributes to the improvement of clinical and functional
Fig. 9. IR — reflectance image of fundus, left eye: fibrotic neovascularization
treatment results, and the late fate of laser treatment shows focus with sharp margins visualized in the clock hour sector 5 of peripheral retina
long-term remission. (yellow arrow)
References
1. Therese KL, Deepa P, Therese J, Bagyalakshmi R, Biswas J, XXVI Int Cong Ophthalmol – Singapore, 1990.
Madhavan HN. Asso-ciation of mycobacteria with Eales’ disease. 16. Ishaq M, Niazi MK. Usefulness of laser photocoagulation in
Indian J Med Res. 2007; 126: 56–62. managing asymptomatic eyes of Eales disease. J Ayub Med Coll
2. Yannuzzi LA. The Retinal Atlas. Retinal Vascular Diseases, Eales’ Abbottabad. 2002; 14 (4): 22–25.
Disease. Elsevier. 2010: 433–8. 17. Ishaq M, Feroze AH, Shahid M, Baig MA, Ameen SS, Feroze SH,
3. Das T, Pathengay A, Hussain N, Biswas J. Eales’ disease: Chishti RA. Intravitreal steroids may facilitate treatment of Eales'
diagnosis and management. Eye. 2010; 24 (3): 472–82. disease (idiopathic retinal vasculitis): an interventional case series.
4. Eales H. Retinal haemorrhages associated with epistaxis and Eye (Lond). 2007; 21: 1403–5.
constipation. Brim Med. 1880; 9: 262. 18. Kumar A, Sinha S. Rapid regression of disc and retinal
5. Madhavan HN, Therese KL, Doraiswamy K. Further investigations neovascularization in a case of Eales disease after intravitreal
on the association of Mycobacterium tuberculosis with Eales' bevacizumab. Can J Ophthalmol. 2007; 42 (2): 335–6.
disease. Indian J Ophthalmol. 2002; 50: 35–39. 19. Magargal LE, Walsh AW, Magargal HO, et al. Treatment of Eales’
6. Patwardhan SD, Azad E, Shah BM, Sharma Y. Role of intravitreal disease with scat-ter laser photocoagulation. Ann Ophthalmol.
bevacizumab in Eales disease with dense vitreous haemorrhage: 1989; 21: 300–2.
a prospective randomized control study. Retina. 2011; 31 (5): 20. Talat L, Lightman S, Tomkins-netzer O. Ischemic retinal vasculitis
866–70. and its manage-ment. J Ophthalmol. 2014; 2014: 197675.
7. Biswas J, K R R, Pal B, Gondhale HP, Kharel Sitaula R. Long- 21. Li J, Liu SM, Dong WT, Li F, Zhou CH, Xu XD, Zhong J. Outcomes of
Term Outcomes of a Large Cohort of Patients with Eales' Disease. transconjunctival sutureless 27-gauge vitrectomy for vitreoretinal
Ocul Immunol Inflamm. 2018; 26 (6): 870–6. diseases. Int J Ophthalmol. 2018; 11 (3): 408–15.
8. Sen A, Paine SK, Chowdhury IH, Mukherjee A, Choudhury S, 22. El-Asrar AM, Al-Kharashi SA. Full panretinal photocoagulation
Mandal LK, Bhattacharya B. Assessment of gelatinase and and early vitrectomy improve prognosis of retinal vasculitis
tumor necrosis factor-alpha level in the vitreous and serum of associated with tuberculoprotein hypersensitivity (Eales' disease).
patients with Eales disease; role of inflammation-mediated Br J Ophthalmol. 2002; 86 (11): 1248–51.
angio-genesis in the pathogenesis of Eales disease. Retina. 23. Dehghan MH, Ahmadieh H, Soheilian M, Azarmina M, Mashayekhi A,
2011; 31 (7): 1412–20. Naghibozakerin J. Therapeutic effects of laser photocoagulation
9. Saxena S, Kumar D. Macular involvement in Eales disease. Ann and/or vitrectomy in Eales' disease. Eur J Ophthalmol. 2005; 15
Ophthalmol. 2000; 32 (2): 98–100. (3): 379–83.
10. Gupta V, Al-Dhibi HA, Arevalo JF. Retinal imaging in uveitis. Saudi 24. Gopal L, Abraham C. Efficacy of photocoagulation in Eales’
J Ophthalmol. 2014; 28: 95–103. disease. Trans Asia-Pacific Acad. 1985; 10: 689.
11. Kumar V, Chandra P, Kumar A. Ultra-wide field angiography in the 25. Nada M, Qanoongo S, Singh SV, Khurana AK. A rare presentation
management of Eales disease. Indian J Ophthalmol. 2016; 64: of exudative macroaneurysms in unilateral Eales' disease. Nepal J
504–7. Ophthalmol. 2017; 9 (18): 95–98.
12. Mwndoza KA, Lauer A. Eales Disease. American Academy of 26. Murphy RP, Gieser SC, Fine SL, et al. Retinal and vitreous findings
Ophthalmology. 2015. Available from: http://eyewiki.org/Eales_ in Eales disease. Invest Ophthalmol Vis Sci. 1986; 27: 121.
Disease. 27. Nicolcescu A, Mocanu C, Dinu L, Olaru A, Ionete M, Stefanescu
13. Saxena S, Kumar D. New classification system-based visual DA. Unilateral Eales' disease a case report. Rom J Ophthalmol.
outcome in Eales’disease. Indian J Ophthalmol. 2007; 55 (4): 267–9. 2017; 61 (2): 144–9.
14. Biswas J, Ravi RK, Naryanasamy A, Kulandai LT, Madhavan HN. 28. Pedanova EK, Burjakov DA. Jeffektivnost' lazerkoaguljacii pri
Eales’ disease — current concepts in diagnosis and management. bolezni Ilza v svete sovremennyh predstavlenij ob jetiopatogeneze
J Ophthalmic Inflamm Infect. 2013; 3: 11. zabolevanija. Sovremennye tehnologii lechenija vitreoretinal'noj
15. Das TP, Namperumalsamy P. Photocoagulation in Eales’ disease. patologii — 2013. 2013: 136. Available from: https://eyepress.ru/
Results of pro-spective randomised clinical study. Presented in article.aspx?12646. Russian.
ВЕСТНИК РГМУ | 5, 2020 | VESTNIKRGMU.RU 95
CLINICAL CASE OPHTHALMOLOGY
29. Tolstuhina EA, Magaramov DA, Volodin PL, Timohov VL, Fomin AV. 31. Moutray T, Evans JR, Lois N, Armstrong DJ, Peto T, Azuara-
Lazer-inducirovannyj regress perifericheskoj retinal'noj Blanco A. Different lasers and techniques for proliferative
neovaskuljarizacii pri bolezni Ilza (klinicheskij sluchaj). Sovremennye diabetic retinopathy. Cochrane Database Syst Rev. 2018; 3 (3):
tehnologii v oftal'mologii. 2019; 1 (26): 381–3. Russian. CD012314.
30. Mainster MA. Decreasing retinal photocoagulation damage: 32. Reddy SV, Husain D. Panretinal Photocoagulation: A Review of
principles and techniques. Semin Ophthalmol. 1999; 14 (4): 200–9. Complications. Semin Ophthalmol. 2018; 33 (1): 83–88.
Литература
1. Therese KL, Deepa P, Therese J, Bagyalakshmi R, Biswas J, Eye (Lond). 2007; 21: 1403–5.
Madhavan HN. Asso-ciation of mycobacteria with Eales’ disease. 18. Kumar A, Sinha S. Rapid regression of disc and retinal
Indian J Med Res. 2007; 126: 56–62. neovascularization in a case of Eales disease after intravitreal
2. Yannuzzi LA. The Retinal Atlas. Retinal Vascular Diseases, Eales’ bevacizumab. Can J Ophthalmol. 2007; 42 (2): 335–6.
Disease. Elsevier. 2010: 433–8. 19. Magargal LE, Walsh AW, Magargal HO, et al. Treatment of Eales’
3. Das T, Pathengay A, Hussain N, Biswas J. Eales’ disease: disease with scat-ter laser photocoagulation. Ann Ophthalmol.
diagnosis and management. Eye. 2010; 24 (3): 472–82. 1989; 21: 300–2.
4. Eales H. Retinal haemorrhages associated with epistaxis and 20. Talat L, Lightman S, Tomkins-netzer O. Ischemic retinal vasculitis
constipation. Brim Med. 1880; 9: 262. and its manage-ment. J Ophthalmol. 2014; 2014: 197675.
5. Madhavan HN, Therese KL, Doraiswamy K. Further investigations 21. Li J, Liu SM, Dong WT, Li F, Zhou CH, Xu XD, Zhong J. Outcomes of
on the association of Mycobacterium tuberculosis with Eales' transconjunctival sutureless 27-gauge vitrectomy for vitreoretinal
disease. Indian J Ophthalmol. 2002; 50: 35–39. diseases. Int J Ophthalmol. 2018; 11 (3): 408–15.
6. Patwardhan SD, Azad E, Shah BM, Sharma Y. Role of intravitreal 22. El-Asrar AM, Al-Kharashi SA. Full panretinal photocoagulation
bevacizumab in Eales disease with dense vitreous haemorrhage: and early vitrectomy improve prognosis of retinal vasculitis
a prospective randomized control study. Retina. 2011; 31 (5): associated with tuberculoprotein hypersensitivity (Eales' disease).
866–70. Br J Ophthalmol. 2002; 86 (11): 1248–51.
7. Biswas J, K R R, Pal B, Gondhale HP, Kharel Sitaula R. Long- 23. Dehghan MH, Ahmadieh H, Soheilian M, Azarmina M, Mashayekhi A,
Term Outcomes of a Large Cohort of Patients with Eales' Disease. Naghibozakerin J. Therapeutic effects of laser photocoagulation
Ocul Immunol Inflamm. 2018; 26 (6): 870–6. and/or vitrectomy in Eales' disease. Eur J Ophthalmol. 2005; 15
8. Sen A, Paine SK, Chowdhury IH, Mukherjee A, Choudhury S, (3): 379–83.
Mandal LK, Bhattacharya B. Assessment of gelatinase and tumor 24. Gopal L, Abraham C. Efficacy of photocoagulation in Eales’
necrosis factor-alpha level in the vitreous and serum of patients disease. Trans Asia-Pacific Acad. 1985; 10: 689.
with Eales disease; role of inflammation-mediated angio-genesis 25. Nada M, Qanoongo S, Singh SV, Khurana AK. A rare presentation
in the pathogenesis of Eales disease. Retina. 2011; 31 (7): 1412–20. of exudative macroaneurysms in unilateral Eales' disease. Nepal J
9. Saxena S, Kumar D. Macular involvement in Eales disease. Ann Ophthalmol. 2017; 9 (18): 95–98.
Ophthalmol. 2000; 32 (2): 98–100. 26. Murphy RP, Gieser SC, Fine SL, et al. Retinal and vitreous findings
10. Gupta V, Al-Dhibi HA, Arevalo JF. Retinal imaging in uveitis. Saudi in Eales disease. Invest Ophthalmol Vis Sci. 1986; 27: 121.
J Ophthalmol. 2014; 28: 95–103. 27. Nicolcescu A, Mocanu C, Dinu L, Olaru A, Ionete M, Stefanescu
11. Kumar V, Chandra P, Kumar A. Ultra-wide field angiography in the DA. Unilateral Eales' disease a case report. Rom J Ophthalmol.
management of Eales disease. Indian J Ophthalmol. 2016; 64: 2017; 61 (2): 144–9.
504–7. 28. Педанова Е. К., Буряков Д. А. Эффективность лазеркоагуляции
12. Mwndoza KA, Lauer A. Eales Disease. American Academy of при болезни Илза в свете современных представлений об
Ophthalmology. 2015. Available from: http://eyewiki.org/Eales_ этиопатогенезе заболевания. Современные технологии
Disease. лечения витреоретинальной патологии — 2013. 2013: 136.
13. Saxena S, Kumar D. New classification system-based visual Доступно по ссылке: https://eyepress.ru/article.aspx?12646.
outcome in Eales’disease. Indian J Ophthalmol. 2007; 55 (4): 267–9. 29. Толстухина Е. А., Магарамов Д. А., Володин П. Л., Тимохов В. Л.,
14. Biswas J, Ravi RK, Naryanasamy A, Kulandai LT, Madhavan HN. Фомин А. В. Лазер-индуцированный регресс периферической
Eales’ disease — current concepts in diagnosis and management. ретинальной неоваскуляризации при болезни Илза (клинический
J Ophthalmic Inflamm Infect. 2013; 3: 11. случай). Современные технологии в офтальмологии. 2019; 1
15. Das TP, Namperumalsamy P. Photocoagulation in Eales’ disease. (26): 381–3.
Results of pro-spective randomised clinical study. Presented in 30. Mainster MA. Decreasing retinal photocoagulation damage:
XXVI Int Cong Ophthalmol – Singapore, 1990. principles and techniques. Semin Ophthalmol. 1999; 14 (4): 200–9.
16. Ishaq M, Niazi MK. Usefulness of laser photocoagulation in 31. Moutray T, Evans JR, Lois N, Armstrong DJ, Peto T, Azuara-
managing asymptomatic eyes of Eales disease. J Ayub Med Coll Blanco A. Different lasers and techniques for proliferative
Abbottabad. 2002; 14 (4): 22–25. diabetic retinopathy. Cochrane Database Syst Rev. 2018; 3 (3):
17. Ishaq M, Feroze AH, Shahid M, Baig MA, Ameen SS, Feroze SH, CD012314.
Chishti RA. Intravitreal steroids may facilitate treatment of Eales' 32. Reddy SV, Husain D. Panretinal Photocoagulation: A Review of
disease (idiopathic retinal vasculitis): an interventional case series. Complications. Semin Ophthalmol. 2018; 33 (1): 83–88.
96 BULLETIN OF RSMU | 5, 2020 | VESTNIKRGMU.RU
You might also like
- Management of Macular HemorrhageFrom EverandManagement of Macular HemorrhageLars-Olof HattenbachNo ratings yet
- JournalDocument15 pagesJournalagungNo ratings yet
- Complications in UveitisFrom EverandComplications in UveitisFrancesco PichiNo ratings yet
- E255406 FullDocument3 pagesE255406 Fullroweis AdelNo ratings yet
- Cystoid Macular EdemaDocument5 pagesCystoid Macular EdemaAgnes Triana BasjaNo ratings yet
- Vascular Abnormalities in Uveitis: SciencedirectDocument15 pagesVascular Abnormalities in Uveitis: Sciencedirect2456708893No ratings yet
- Ischemic Eye Syndrome in Aortic Arch DiseaseDocument3 pagesIschemic Eye Syndrome in Aortic Arch DiseaseCentral Asian StudiesNo ratings yet
- April 2017 Ophthalmic PearlsDocument2 pagesApril 2017 Ophthalmic PearlsNur Asri AinunNo ratings yet
- Central Retinal Vein Occlusion Revealing Anti Phospholipid Antibody Syndrome Case ReportDocument4 pagesCentral Retinal Vein Occlusion Revealing Anti Phospholipid Antibody Syndrome Case ReportInternational Journal of Innovative Science and Research TechnologyNo ratings yet
- Diagnostic and Therapeutic Challenges: Edited by H. Richard McdonaldDocument4 pagesDiagnostic and Therapeutic Challenges: Edited by H. Richard McdonaldAndhi Riawan Eko WiikramatunggadewaNo ratings yet
- 0410RT Feature BennettDocument4 pages0410RT Feature BennettRaissaNo ratings yet
- Observations On Time Course Changes of The CherryDocument16 pagesObservations On Time Course Changes of The CherryAndi Tiara S. AdamNo ratings yet
- Magnetic Resonance Tractographyas A Method of Choice For Neuroimaging in Ocular Ischemic Syndrome Against The Background of HypertensionDocument4 pagesMagnetic Resonance Tractographyas A Method of Choice For Neuroimaging in Ocular Ischemic Syndrome Against The Background of HypertensionCentral Asian StudiesNo ratings yet
- Ni Hms 403652 Patología Oculares Relacionadas A HASDocument15 pagesNi Hms 403652 Patología Oculares Relacionadas A HASLuis Esteban HoyoNo ratings yet
- Ischaemic Optic Neuropathy Induced Sudden BlindnesDocument2 pagesIschaemic Optic Neuropathy Induced Sudden Blindneszazaza94No ratings yet
- Ijo 69 525Document10 pagesIjo 69 525Eduardo Espindola ElizaldeNo ratings yet
- Neovascular GlaucomaDocument4 pagesNeovascular GlaucomaMahmoud ElsaidNo ratings yet
- Hypopyon in Acute Lymphoblastic Leukemia A Case ReportDocument2 pagesHypopyon in Acute Lymphoblastic Leukemia A Case ReportInternational Journal of Innovative Science and Research TechnologyNo ratings yet
- Retrospective Analysis of Sterile Corneal Infiltrates in Patients With Keratoconus After CrossLinking Procedure 2022Document10 pagesRetrospective Analysis of Sterile Corneal Infiltrates in Patients With Keratoconus After CrossLinking Procedure 2022fn_millardNo ratings yet
- NCP Rheum 0268Document9 pagesNCP Rheum 0268drheriNo ratings yet
- 2022 10 50 Images KimDocument2 pages2022 10 50 Images KimJuan David Pineda MontoyaNo ratings yet
- 1141 FullDocument9 pages1141 FullemikoNo ratings yet
- Corneal Neovascularization: Updates On Pathophysiology, Investigations & ManagementDocument8 pagesCorneal Neovascularization: Updates On Pathophysiology, Investigations & ManagementAsniar RNo ratings yet
- Acute Retinal Necrosis A Potentially Visual Devastating AffectionDocument3 pagesAcute Retinal Necrosis A Potentially Visual Devastating AffectionNAFILAH NAFILAHNo ratings yet
- Uveitis Associated With Multiple Sclerosis: Complications and Visual PrognosisDocument4 pagesUveitis Associated With Multiple Sclerosis: Complications and Visual PrognosisnovywardanaNo ratings yet
- Clinical and Epidemiological Characterization of KeratoconusDocument12 pagesClinical and Epidemiological Characterization of KeratoconusAndrés MontoyaNo ratings yet
- Tacrolimus Optic NeuropathyDocument7 pagesTacrolimus Optic NeuropathyVlady BordaNo ratings yet
- Ocular Ischemic Syndrome Survey OphthalmologyDocument33 pagesOcular Ischemic Syndrome Survey OphthalmologyamaiacNo ratings yet
- Cytokines Associated With Hemorrhage in Proliferative Diabetic RetinopathyDocument9 pagesCytokines Associated With Hemorrhage in Proliferative Diabetic RetinopathyQabil GilbranNo ratings yet
- Optic NeuritisDocument12 pagesOptic NeuritisScerbatiuc CristinaNo ratings yet
- Is Episcleritis Associated To Glaucoma?Document3 pagesIs Episcleritis Associated To Glaucoma?Alvin JulianNo ratings yet
- Jurnal Mata 1 PDFDocument4 pagesJurnal Mata 1 PDFyayan latuconsinaNo ratings yet
- Fundus Autofluorescence and Optical Coherence TomoDocument4 pagesFundus Autofluorescence and Optical Coherence TomoAndi Tiara S. AdamNo ratings yet
- Retinoblastoma PDFDocument5 pagesRetinoblastoma PDFRiska PermatasariNo ratings yet
- Ischemic Optic NeuropathyDocument7 pagesIschemic Optic NeuropathyJhoni Akbar DalimuntheNo ratings yet
- Acc or Inti 2007Document6 pagesAcc or Inti 2007iwanNo ratings yet
- Retinal Vasculitis DDDocument17 pagesRetinal Vasculitis DDPopuri SandhyaNo ratings yet
- Indirect Carotid-Cavernous Fistula Mimicking Scleritis: MagesDocument1 pageIndirect Carotid-Cavernous Fistula Mimicking Scleritis: MagesClarithq LengguNo ratings yet
- Retninal Vein OcclusionDocument10 pagesRetninal Vein OcclusionuhibNo ratings yet
- Results of Staged Combined Laser Treatment of Neovascular GlaucomaDocument4 pagesResults of Staged Combined Laser Treatment of Neovascular GlaucomaCentral Asian StudiesNo ratings yet
- Probable Vogt-Koyanagi-Harada Disease: A Case ReportDocument5 pagesProbable Vogt-Koyanagi-Harada Disease: A Case ReportIJAR JOURNALNo ratings yet
- Survey-62-P770 Keratoconus Review - QueratoconoDocument14 pagesSurvey-62-P770 Keratoconus Review - QueratoconoMaría Antonela NicolóNo ratings yet
- Masquerade SyndromesDocument10 pagesMasquerade Syndromestony_chrisNo ratings yet
- Connective Tissue Diseases: Focus On Microcirculatory Bed: O.I. Zarudna, I.K. Venher, A.V. DovbushDocument8 pagesConnective Tissue Diseases: Focus On Microcirculatory Bed: O.I. Zarudna, I.K. Venher, A.V. DovbushdjdNo ratings yet
- Tahmasbi Arashlow2016Document4 pagesTahmasbi Arashlow2016Jonathan PanchiNo ratings yet
- How Does Hipertension Affects Your Eye - Anure 2011Document13 pagesHow Does Hipertension Affects Your Eye - Anure 2011Germano Freire Bezerra FilhoNo ratings yet
- Bilateral Radiation Retinopathy 17 Years Following.62Document3 pagesBilateral Radiation Retinopathy 17 Years Following.62ShivcharanLalChandravanshiNo ratings yet
- Hematom 2Document2 pagesHematom 2omidazadmehr1375No ratings yet
- Central Serous RetinopathyDocument7 pagesCentral Serous RetinopathyshintasissyNo ratings yet
- Multiple Sclerosis With Ophthalmologic Onset - Case ReportDocument5 pagesMultiple Sclerosis With Ophthalmologic Onset - Case ReportVannyNo ratings yet
- V36N4p293 PDFDocument5 pagesV36N4p293 PDFsiscaNo ratings yet
- Age-Related Macular DegenerationDocument5 pagesAge-Related Macular DegenerationHawan SMNo ratings yet
- Issue Apr2012 Pdffiles CasereportDocument5 pagesIssue Apr2012 Pdffiles CasereportyunjaealwaysNo ratings yet
- Review: Ocular and Orbital Involvement in LeukemiaDocument22 pagesReview: Ocular and Orbital Involvement in LeukemiadechastraNo ratings yet
- Vascular: Changes The Chronic Anterior UveitisDocument5 pagesVascular: Changes The Chronic Anterior Uveitisandrema123No ratings yet
- Imprimir 2Document3 pagesImprimir 2Gino VitteriNo ratings yet
- 10.1007@s11940 019 0579 9Document16 pages10.1007@s11940 019 0579 9AlexNo ratings yet
- Uveitis 2Document9 pagesUveitis 2srihandayaniakbarNo ratings yet
- Enfermedad Renal y Desordenes HematologicasDocument11 pagesEnfermedad Renal y Desordenes HematologicasIris GzlzNo ratings yet
- ManagementDocument3 pagesManagementElison J PanggaloNo ratings yet
- Common Retinal Diseases: FloatersDocument7 pagesCommon Retinal Diseases: FloatersDan-Dan Irika CentinoNo ratings yet
- Education For Physically Challenged Children Set 1Document6 pagesEducation For Physically Challenged Children Set 1Ahmed KhanNo ratings yet
- Ophthalmology Investigation and Examination Techniques PDFDocument237 pagesOphthalmology Investigation and Examination Techniques PDFJavier Andrés Pinochet SantoroNo ratings yet
- Children's Activity BookDocument48 pagesChildren's Activity Bookojossaludables100% (3)
- Optical Express Brochure PDFDocument16 pagesOptical Express Brochure PDFmari_ana_b1320No ratings yet
- A Review of Netra ShariraDocument6 pagesA Review of Netra ShariraEditor IJTSRDNo ratings yet
- Diabetic RetinopathyDocument95 pagesDiabetic Retinopathyhassan qureshiNo ratings yet
- Structure and Function of EyesDocument11 pagesStructure and Function of EyesSamar Ameen SialNo ratings yet
- Iol Power Calculation Warren HillDocument4 pagesIol Power Calculation Warren HillMariana Luzardo bravoNo ratings yet
- Visual Acuity Test: Equipment NeededDocument3 pagesVisual Acuity Test: Equipment Neededmelannie napisaNo ratings yet
- Diagnostic Considerations in Uveitis: AAO Reading Section 9Document43 pagesDiagnostic Considerations in Uveitis: AAO Reading Section 9evaNo ratings yet
- Abnormal Retinal CorrespondenceDocument6 pagesAbnormal Retinal CorrespondenceJhonathan MoránNo ratings yet
- Question Papers of BHUDocument13 pagesQuestion Papers of BHUgyogi1989No ratings yet
- Acute Angle-Closure GlaucomaDocument1 pageAcute Angle-Closure GlaucomaMr. LNo ratings yet
- Eye & Ear Embryology ObjectivesDocument3 pagesEye & Ear Embryology ObjectivesDr. DNo ratings yet
- Ophthalmology: Cataract Surgery in Eyes With Pseudoexfoliation (PEX) SyndromeDocument5 pagesOphthalmology: Cataract Surgery in Eyes With Pseudoexfoliation (PEX) SyndromeCahya AlfalizaNo ratings yet
- Daftar Pustaka ReferatDocument2 pagesDaftar Pustaka ReferatWidyastutiNo ratings yet
- Neuro OphthalmologyDocument61 pagesNeuro OphthalmologySurat TanprawateNo ratings yet
- Assignment 2022 B213298B OPTC201Document4 pagesAssignment 2022 B213298B OPTC201Kudzai RusereNo ratings yet
- RAAB Di IndonesiaDocument42 pagesRAAB Di IndonesiaRaissa100% (2)
- NCM 116: Care of Clients With Problems in Nutrition and Gastrointestinal, Metabolism and Endocrine,, Acute and ChronicDocument18 pagesNCM 116: Care of Clients With Problems in Nutrition and Gastrointestinal, Metabolism and Endocrine,, Acute and ChronicDiego DumauaNo ratings yet
- Heal Your Eye Problems With Herbs, Minerals and Vitamins PDFDocument144 pagesHeal Your Eye Problems With Herbs, Minerals and Vitamins PDFangelobuffalo100% (6)
- Ch14 Jarvis Test BankDocument13 pagesCh14 Jarvis Test BankRaghavanJayaraman100% (12)
- Vitreous HemorrhageDocument7 pagesVitreous HemorrhageindahNo ratings yet
- 43-Articles Content-164-1-10-20190131 PDFDocument7 pages43-Articles Content-164-1-10-20190131 PDFInyraiputramarthanaNo ratings yet
- Glaucoma NCLEX Questions QuizDocument22 pagesGlaucoma NCLEX Questions QuizGabrielle G. MaldoNo ratings yet
- Eye Eye: Visit For More The Ultimate Online Montessori Resource !Document21 pagesEye Eye: Visit For More The Ultimate Online Montessori Resource !uncorreopararegistroNo ratings yet
- Visual AcuityDocument21 pagesVisual AcuityLynCapiliNo ratings yet
- Letters To The EditorDocument8 pagesLetters To The EditorGheavita Chandra DewiNo ratings yet
- Seminar: Penny A Asbell, Ivo Dualan, Joel Mindel, Dan Brocks, Mehdi Ahmad, Seth EpsteinDocument11 pagesSeminar: Penny A Asbell, Ivo Dualan, Joel Mindel, Dan Brocks, Mehdi Ahmad, Seth EpsteinMagny DcrNo ratings yet
- The Age of Magical Overthinking: Notes on Modern IrrationalityFrom EverandThe Age of Magical Overthinking: Notes on Modern IrrationalityRating: 4 out of 5 stars4/5 (23)
- Summary: Outlive: The Science and Art of Longevity by Peter Attia MD, With Bill Gifford: Key Takeaways, Summary & AnalysisFrom EverandSummary: Outlive: The Science and Art of Longevity by Peter Attia MD, With Bill Gifford: Key Takeaways, Summary & AnalysisRating: 4.5 out of 5 stars4.5/5 (42)
- By the Time You Read This: The Space between Cheslie's Smile and Mental Illness—Her Story in Her Own WordsFrom EverandBy the Time You Read This: The Space between Cheslie's Smile and Mental Illness—Her Story in Her Own WordsNo ratings yet
- Summary: The Psychology of Money: Timeless Lessons on Wealth, Greed, and Happiness by Morgan Housel: Key Takeaways, Summary & Analysis IncludedFrom EverandSummary: The Psychology of Money: Timeless Lessons on Wealth, Greed, and Happiness by Morgan Housel: Key Takeaways, Summary & Analysis IncludedRating: 5 out of 5 stars5/5 (80)
- Raising Mentally Strong Kids: How to Combine the Power of Neuroscience with Love and Logic to Grow Confident, Kind, Responsible, and Resilient Children and Young AdultsFrom EverandRaising Mentally Strong Kids: How to Combine the Power of Neuroscience with Love and Logic to Grow Confident, Kind, Responsible, and Resilient Children and Young AdultsRating: 5 out of 5 stars5/5 (1)
- The Body Keeps the Score by Bessel Van der Kolk, M.D. - Book Summary: Brain, Mind, and Body in the Healing of TraumaFrom EverandThe Body Keeps the Score by Bessel Van der Kolk, M.D. - Book Summary: Brain, Mind, and Body in the Healing of TraumaRating: 4.5 out of 5 stars4.5/5 (266)
- Think This, Not That: 12 Mindshifts to Breakthrough Limiting Beliefs and Become Who You Were Born to BeFrom EverandThink This, Not That: 12 Mindshifts to Breakthrough Limiting Beliefs and Become Who You Were Born to BeNo ratings yet
- The Obesity Code: Unlocking the Secrets of Weight LossFrom EverandThe Obesity Code: Unlocking the Secrets of Weight LossRating: 4 out of 5 stars4/5 (5)
- Why We Die: The New Science of Aging and the Quest for ImmortalityFrom EverandWhy We Die: The New Science of Aging and the Quest for ImmortalityRating: 4 out of 5 stars4/5 (3)
- Sleep Stories for Adults: Overcome Insomnia and Find a Peaceful AwakeningFrom EverandSleep Stories for Adults: Overcome Insomnia and Find a Peaceful AwakeningRating: 4 out of 5 stars4/5 (3)
- Gut: the new and revised Sunday Times bestsellerFrom EverandGut: the new and revised Sunday Times bestsellerRating: 4 out of 5 stars4/5 (392)
- Dark Psychology & Manipulation: Discover How To Analyze People and Master Human Behaviour Using Emotional Influence Techniques, Body Language Secrets, Covert NLP, Speed Reading, and Hypnosis.From EverandDark Psychology & Manipulation: Discover How To Analyze People and Master Human Behaviour Using Emotional Influence Techniques, Body Language Secrets, Covert NLP, Speed Reading, and Hypnosis.Rating: 4.5 out of 5 stars4.5/5 (110)
- The Ritual Effect: From Habit to Ritual, Harness the Surprising Power of Everyday ActionsFrom EverandThe Ritual Effect: From Habit to Ritual, Harness the Surprising Power of Everyday ActionsRating: 3.5 out of 5 stars3.5/5 (3)
- ADHD is Awesome: A Guide to (Mostly) Thriving with ADHDFrom EverandADHD is Awesome: A Guide to (Mostly) Thriving with ADHDRating: 5 out of 5 stars5/5 (1)
- When the Body Says No by Gabor Maté: Key Takeaways, Summary & AnalysisFrom EverandWhen the Body Says No by Gabor Maté: Key Takeaways, Summary & AnalysisRating: 3.5 out of 5 stars3.5/5 (2)
- An Autobiography of Trauma: A Healing JourneyFrom EverandAn Autobiography of Trauma: A Healing JourneyRating: 5 out of 5 stars5/5 (2)
- Raising Good Humans: A Mindful Guide to Breaking the Cycle of Reactive Parenting and Raising Kind, Confident KidsFrom EverandRaising Good Humans: A Mindful Guide to Breaking the Cycle of Reactive Parenting and Raising Kind, Confident KidsRating: 4.5 out of 5 stars4.5/5 (169)
- Mindset by Carol S. Dweck - Book Summary: The New Psychology of SuccessFrom EverandMindset by Carol S. Dweck - Book Summary: The New Psychology of SuccessRating: 4.5 out of 5 stars4.5/5 (328)
- Outlive: The Science and Art of Longevity by Peter Attia: Key Takeaways, Summary & AnalysisFrom EverandOutlive: The Science and Art of Longevity by Peter Attia: Key Takeaways, Summary & AnalysisRating: 4 out of 5 stars4/5 (1)
- Summary: The Myth of Normal: Trauma, Illness, and Healing in a Toxic Culture By Gabor Maté MD & Daniel Maté: Key Takeaways, Summary & AnalysisFrom EverandSummary: The Myth of Normal: Trauma, Illness, and Healing in a Toxic Culture By Gabor Maté MD & Daniel Maté: Key Takeaways, Summary & AnalysisRating: 4 out of 5 stars4/5 (9)
- 12 Rules for Life by Jordan B. Peterson - Book Summary: An Antidote to ChaosFrom Everand12 Rules for Life by Jordan B. Peterson - Book Summary: An Antidote to ChaosRating: 4.5 out of 5 stars4.5/5 (207)
- Cult, A Love Story: Ten Years Inside a Canadian Cult and the Subsequent Long Road of RecoveryFrom EverandCult, A Love Story: Ten Years Inside a Canadian Cult and the Subsequent Long Road of RecoveryRating: 4 out of 5 stars4/5 (44)
- The Courage Habit: How to Accept Your Fears, Release the Past, and Live Your Courageous LifeFrom EverandThe Courage Habit: How to Accept Your Fears, Release the Past, and Live Your Courageous LifeRating: 4.5 out of 5 stars4.5/5 (253)