Professional Documents
Culture Documents
Modelling of Diffusion Bonding of Metals
Uploaded by
Marcionilo NeriOriginal Title
Copyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
Modelling of Diffusion Bonding of Metals
Uploaded by
Marcionilo NeriCopyright:
Available Formats
See discussions, stats, and author profiles for this publication at: https://www.researchgate.
net/publication/233591317
Modeling of diffusion bonding of metals
Article in Materials Science and Technology · November 1987
DOI: 10.1179/026708387790221035
CITATIONS READS
50 1,502
2 authors:
Z. Xiao Guo Natassja Ridley
University College London Northeastern State University at Broken Arrow
297 PUBLICATIONS 16,465 CITATIONS 120 PUBLICATIONS 3,707 CITATIONS
SEE PROFILE SEE PROFILE
Some of the authors of this publication are also working on these related projects:
BIOADSORB - Biomass derived microporous carbon adsorbents for CO2 capture and storage View project
Hydrogen storage technologies View project
All content following this page was uploaded by Z. Xiao Guo on 23 February 2015.
The user has requested enhancement of the downloaded file.
Modelling of diffusion bonding has been carried out to quantify the kinetics of the
Modelling of. bonding processes and to predict the time for achieving a sound bond. An
diffusion alternative geometric assumption for the shape of the interfacial cavities to those
considered previously was employed. Three subprocesses of bonding were
bonding of introduced to simplify the modelling. These involved volume and interfacial
diffusion coupled-with creep, rigid collapse, and surface diffusion. The effects of
metals grain size and phase ratio on diffusion bonding have also been considered. The
predictions are compared with existing experimental data for copper and
Ti-6AI-4Valloy and in general show good agreement. MST/588
© 1987 The Institute of Metals. Manuscript received 3 October 1986; infinalform
z.x. Guo 21 April 1987. The authors are in the Department of Metallurgy and Materials
N. Ridley Science, University of Manchester/UM/ST.
R radius of interfacial void
List of symbols Rin initial radius of interfacial void before
deformation
a half of interfacial void width S sign symbol (positive for tensile stress and
A modified creep constant negative for compressive stress)
Ac strain rate power law creep constant t bonding time
Ad area across which flux flows T temperature
A' creep constant from uniaxial creep testing Tm melting temperature
b half of bonded length between two adjacent V cavity volume
voids x, y Cartesian coordinates
bo half of contacted length between two Xi half width of ith slice of void ridge
adjacent voids. after instantaneous .X characteristic length
deformation Yi distance from bondline to ith slice of void
bv magnitude of Burgers vector ridge
B effective diffusion distance Vu chemical potential gradient of atoms
c spacing between cavities along bond
!Xj angle between jth grain boundary and
d half of cavity spacing
bondline
D diffusion coefficient
y surface tension
DGB grain boundary diffusion coefficient
<>GB grain boundary width
DGBO grain boundary diffusion coefficient
<>s surface thickness
(pre-exponential)
Ds surface diffusion coefficient
e strain rate
ee equivalent strain
Dv volume diffusion coefficient .
Fx, Fy atomic fraction of constituents X and Y,
ei strain rate of ith slice of void ridge
respectively, in binary alloy
e half of angle subtended by void surface at
its centre of curvature
FA' FB volume proportions of phases A and B,
p cavity tip radius
respectively, in two-phase alloy
(J stress perpendicular to bondline
G shear modulus
(J e eq ui valen t stress
dG ( 1 ) temperature coefficient of shear modulus (Ji stress acting on ith slice of void ridge
dT G300 (JI, (In average stress normal to bondline in region
G300 shear modulus at 300 K I and II, respectively
h half of interfacial cavity height (J p applied stress
hi height of ith slice of void ridge (J(r) true stress normal to bondline in region I
hin surface roughness (initial height) V(J(r) gradient of true stress normal to bondline
j jth grain boundary in region I
J atom flux (J y yield stress
J GB flux of grain boundary diffusion V(J stress gradient
Jv flux of volume diffusion TO shear stress of parent material
VJ atom flux gradient n atomic volume
k Boltzmann's constant
1 length of arc between two grain Subscripts
boundaries, which corresponds to grain A, B phases in two-phase alloy
diameter C creep
n power law creep exponent D diffusion
N number of grain boundaries around one- i,j ith and jth grain boundary, respectively
quarter of cavity profile o values after instantaneous deformation
QGB activation energy of grain boundary X, Y constituents in binary alloy
diffusion (1) coupled process (from equation (1))
Qv activation energy of volume diffusion (2) rigid collapse (from equation (2))
Qs surface activation energy (3) surface diffusion (from equation (3))
r distance from central point of cavity along I effective diffusion region
x axis in region I II constant stress region
Materials Science and Technology November 1987 Vol. 3 945
946 Guo and Ridley Modelling of diffusion bonding of metals
Introduction c
d
Diffusiori bonding is a solid state process by which two
surfaces are joined under pressure at an elevated
temperature, as a result of diffusion controlled processes. A
minimum macroscopic deformation is involved. Generally,
the clean surfaces to be bonded are not flat. The rate
controlling step in diffusion bonding is the removal of the
interfacial voids due to the surface roughness. A bond is
regarded as being formed when new grains appear in the
zone adjacent to the interface and only a few small pores
remain at the joint line.
Modelling of diffusion bonding has been carried out by
several authors.I-5 In the earlier approaches,I-3 only one
or two mechanisms for diffusion bonding were considered
and some relationships were restricted to specific alloys. A
recent model proposed by Derby and Wallach4,5 has
included all possible bonding mechanisms and the
( b)
predictions have shown reasonable agreement with the
experimental results for copper and iron. Furthermore,
Pilling and co-workers6,7 have considered the effect of
grain size on diffusion bonding which, in addition to the
common mechanisms, is of particular significance in
superplastic materials.
In previous work, the models either assumed a constant
geometry for the interfacial voids during the early stages of
bonding or assumed the voids to be spherical. Neither
assumption is fully adequate in most practical cases.
Surface preparation processes, such as grinding and
machining, usually create semilenticular or semicircular
asperities. When bonding proceeds, the interfacial cavities
will maintain a smooth surface curvature to minimize their
free energy. This is possible because of rapid surface
diffusion. Lenticular cavities can usually be seen in
photographs of the bond interfaces in published
papers.8-10 Therefore, it seems reasonable to assume that
the bonding interface cavities are of lenticular shape.
From previous work,4, 5 it is known that the mechanisms
for diffusion bonding are those of diffusion and creep. a initial
void morphology; b after instantaneous deformation;
c coupled diffusion and creep process; d rigid collapse process;
Since vapour phase transport in the cavity is not believed e combined effect of processes c and d; fvoid morphology at a late
to be very important,5 it is not included in the present stage of bOl)ding when (d V /dt)o» (d V /dt)c; g void morphology at
model. Thus, the mechanisms considered are: late stage of bonding when (d V /dt)o« (d V /dt)c
Schematic of void shrinkage processes
1. Bonded interface diffusion, volume diffusion, and
grain boundary diffusion.
2. Plastic deformation by creep, and, in the early stages, COUPLED DIFFUSION AND CREEP
by instantaneous yielding. This process decreases cavity volume only by the mass
3. Cavity surface· diffusion. transferred to the cavity from the boundary and the bulk,
through boundary diffusion, volume diffusion, and creep.
For simplicity, the following assumptions are proposed: This is assumed to result only in a decrease of cavity width
2a; cavity height does not change (Fig. Ie). Therefore, the
(i) the two surfaces to be bonded are brought together volume change rate from the coupled process is
in such a manner as to give ridge-to-ridge contact as (dV/dt)(I) = (dV/dt)D(I) + (dV/dt)ql) (1)
shown in Fig. la; bonds are obtained immediately
in the contact regions where (d V /dt)D(l) is the rate of decrease of volume due to
(ii) during bonding, interfacial voids are of lenticular mass transferred into the cavity through boundary and
shape as shown in Fig. Ib volume diffusion; (d V/dt)ql) is the rate due to the mass
(iii) the ridges between the cavities are in a state of plane transferred by matrix creep.
strain; the strain in the direction of the cavity length
is zero RIGID COLLAPSE
(iv) cavities are uniformly distributed along the bond This process decreases the cavity volume as a result of the
with a spacing e and have unit length along the z cavity surfaces coming together and so causing changes of
axis cavity width 2a and height 2h. The curvature of the cavity
(v) there is no pressure in the cavities during bonding profile remains constant, so that the radius R is unchanged
(vi) since the cavities are very long compared with their (Fig. Id). Thus, the rate of variation of cavity volume is
width, end effects can be ignored
(dV/dt)(2) = (dV/dt)D(2) + (dV/dt)q2) (2)
To quantify the bonding process under the present where (dV/dt)D(2) and (dV/dt)q2) are the rates of cavity
geometric assumptions and simplify the modelling, the shrinkage ascribed to the approaching cavity surfaces,
cavity shrinkage is assumed to result from three caused by mass transference away from the boundary and
subprocesses, shown in Fig. 1, which are described below. by matrix creep, respectively.
Materials Science and Technology November 1987 Vol. 3
Guo and Ridley Modelling of diffusion bonding of metals 947
In fact, h2/a is the tip radius of an elliptic void with half
I :
I
I J I axes a and h. This assumption is appropriate in that rapid
:Region I ,Region II : surface diffusion will maintain a smooth variation of
I I I
, I I surface curvature along the void profile and an elliptic void
I I I
'. B •I : is likely to exist in the early stage of bonding. This
: I I
consideration has been adopted for simplicity and is
o expected to give rise to only a very small error.
a b •
I
I
I
I
d
-, I
I
Power law creep
I
2 Stress distribution along diffusion bond During bonding, the creep of void ridges also contributes
to the shrinkage of voids. If power law creep is assumed,
the strain rate is given by
SURFACE DIFFUSION
. . . (10)
This process changes the void geometry but not the volume
and can be represented as The volume variation due to bonding surfaces coming
(dV/dt)(3)=0 .... (3) together by creep (Fig. 3) is derived in Appendix 2 as
(~~t2) = 4aSA ( ci)"
Diffusion around grain boundary
When a pressure is uniformly applied perpendicular to a
and the variation
x J: {d-[R2-(Y:R-W]'/2
due to mass transfer by creep is
r dy (11)
grain boundary, the stress in the boundary will redistribute
around boundary cavities. In the region adjacent to the
= 4dSAlup/GI"
cavity, termed region I, the compressive stress increases (dV)
dt C(1)
r-
with increasing distance r from the origin (Fig. 2). I
However, in region II, midway between the cavities,
stresses are constant, as shown in Fig. 2.
A stress gradient results in a chemical potential gradient
x J: {d_[R2_(Y:R_h)2]1/2 dy (12)
Equations (11) and (12) can be solved using numerical
which causes diffusion. Therefore, diffusion along the
integration.
boundary takes place only in region I, termed the effective
diffusion region. In region II, power law creep occurs.
Chen and Argon 11 have introduced a characteristic
length X for the effective diffusion region, which is
Geometric relations
X = [DGBbGBQUpJ1/3 (4)
AkT(up/G)n
For the coupled process, the cavity height is constant.
The effective diffusion distance B corresponding to region I Hence, from Figs. 1 and 4
can be defined as
(da/dt)(1) = (h/a)(dR/dt)(l) (13)
B = b when X> b, and B = X when X < b (5)
(dh/dt)(1) =0 (14)
When X> b, region II does not exist.
As shown in Appendix 1, the average normal stress
(d8/dt)(1) = (h/a-a/R)(dR/dt)(1) (15)
in region I is given by
2dBup+ [3(B-2a)(d-a-B)
(~~t 2
= {[~ (a +h )sin-
2 1
(}:hh2) J }(~~t
-4a
x (1/B+ l/p)-2B(d-a-B)/p]y . . (16a)
(6)
2B2 +3(B-2a)(d-a-B) or
and the rates of the volume variation, due to the diffusive
mass transfer and the cavity surfaces coming together are,
respectively,
(~~
t (~~)j =
2
{[~(a2 +h )sin -I C h
22: h2)J-4a}
(16b)
(~~)D(I)= B~T (DGB"GB+2Dvp) (JI- i-~) (7)
For the rigid collapse
constant. Therefore
process, the void radius R is
and (da/dt)(2) = [(a2 - h2)/(2ah)]( dh/dt)(2) (17)
(dV)
dt D(2)
= 6aDGBbGB
B2kT
(ul
- ~
B
_!) P
(8)
(dh/dt)(2) = (d V/dt)(2/(4a)
(dv/dt)(2) = 4a(dh/dt)(2)
(18)
(19)
Equations (7) and (8) can be readily solved if the radius of
the void tip can be determined.
There are many factors influencing the void tip radius,
such as surface tension, surface diffusivity, or grain Diffusion bonding around surface areas
boundary diffusion/surface diffusion ratio,12 void size, and
void geometry. For simplicity, an assumption is made that
The difference of curvature between void tip and void
the radius is a function of void width and height, given by
surface results in a chemical potential gradient described by
p = h2/a ..... . . . . . . (9) the Gibbs-Thomson relationship. From Ref. 4, the volume
Materials Science and Technology November 1987 Vol. 3
948 Guo and Ridley Modelling of diffusion bonding of metals
Yi
o
I
3 Creep of void ridge I
f
I
.'
I
,
I
I
flux is given by I
I
(
dV)
dt (3)
= 8A DQy
d
kT P
(1- ~)(~_~)
R p b
. (20) 4 Schematic of interfacial void intersected by group
I
I
of straight grain boundaries
It should be noted that diffusion around the surface area
occurs not only through a thin layer of material (surface
diffusion) but also through the volume adjacent to the
surface (volume diffusion). If the v'olume diffusion is determined approximately by using slipline theory for a
assumed to pass through a layer with the thickness of the trapezoidal shape. Therefore, the contact width of the void
void neck, then the above equation becomes ridge after instantaneous deformation can be estimated
dV)
( -dt (3)
8Qy ( 1--
=-
kT
p)'
R
(Ds~s+Dvp) '(1 1)
---
P b
(21)
as10
b = )3d I O"p I
This material is assumed to be taken uniformly from the
o 2+n-2tg-1(hin/d) O"y (30)
void surface. Thus, it can be estimated that Thus, after contact, the void width and height are,
(dh/dt)(3) = (d V/dt)(3/(48R) . (22) respectively,
Since the diffusion only alters the shape of the void, the a = d-b (31)
volume is constant. Hence10 and
da) [1-(a2/h2)]{R8-a- [ah/2(R2 _a2)1/2]} (dh) h = hin[hrn + (d -b)2 + bd -(bhrn/d)]
. . . . . (32)
( dt (3)
2 2 2 2
= {(h -a )/[2(R -a )1/2]}+2a(R8-a)/h dt (3) d2 +hrn
. . . . . (23)
As "can be seen from equation (23), when a = h, surface
diffusion' does not occur. Effect of grain size
The equations derived so far, to achieve the rates of
variation of void geometry, can be summarized by:
For superplastic materials, the grains are usually very
(dV/dt)(l) = (dV/dt)D(1) + (dV/dt)q1) (24) small compared with the cavity width at the bonding
interface. Thus, the profile of the cavity may be intersected
(dV/dt)(2) = (dV/dt)D(2)+ (dV/dt)q2! (25)
by several grain boundaries, through which atoms diffuse
dV/dt = (dV/dt)(l)+ (dV/dt)(2) (26) into the cavity and contribute to the cavity shrinkage.
.., This effect can be estimated by simplifying the grain
dh/dt = (dh/dt\l) + (dh/dt)(2) + (dh/dt\3) (27)
boundaries as a set of straight lines or planes, distributed
da/dt = (da/dt)(l) + (da/dt)(2) + (dh/dt)(3) (28) around the cavity profile, directed towards the cavity
centre, and intersecting the profile with a spacing I apart.
At a later stage of bonding, the value of void width may
Here, I is the diameter of the grain, as shown in Fig. 4.
approach that of vO,id height. Then, the above equations
Thus the number of such boundaries N around a quarter
can be simplified to
of the profile is given by
da/dt = dh/dt = (d V/dt)/(2nR) . . (29)
N = INT(8R/l) . (33)
As can be seen, these relationships are functions of the
where R is the radius of the profile and 8 the angle of one-
variables a and h, and can be solved by using numerical
quarter of the profile.
methods. It should be noted that in all the equations
For one such boundary j (j = 1, 2, ... , N), since' a stress
concerned, tensile stresses are assumed positive and
gradient exists in region Ij adjacent to the cavity, a flux of
compressive stresses negative.
atoms diffuses through the boundary into the cavity.
Surface diffusion redistributes the mass along the profile of
the cavity to maintain a smooth surface. This contribution
should be merged into the coupled process, (d V/dt)D(1)'
Instantaneous plastic deformation
However, the rigid collapse of the boundary is part of the
matrix creep and has already been considered. The volume
It has been assumed that the bonding surfaces are brought diffusion adjacent to those boundaries is neglected because,
int() contact in a ridge-to-ridge and valley-to-valley for the material with a small grain size, boundary diffusion
manner, as shown in Fig. 1a. When pressure is applied, the seems much more important.
void ridge will deform instantaneously to such an extent Similarly, the same types of relationship as those for
that the contact stress is no more than the deforming diffusion through a bondline can be obtained by sub-
resistance of the metal. The deforming resistance can be stituting aj, bj, Bj, dj, O"pj for a, b, B, d, O"p, as shown in
Materials Science and Technology November 1987 Vol. 3
Guo and Ridley Modelling of diffusion bonding of metals 949
Fig. 4, i.e. and Y. It is obvious that this treatment is not suitable for
interstitial solutions.
X.-
_ [DGBbGBQcrPi]1/3
---- As for two-phase alloys, the law of mixtures has been
(34)
J AkT(crpj/Gt used for a microduplex titanium alloy in Ref. 6, in the
Bj = bj when Xj> bj, and Bj = Xj when Xj < bj (35) absence of an alternative more rigorous approach, and
has led to predictions in reasonable agreement with
1dBcrpj+ [3(Bj-2a)(dj-aj-B) experiment.
x (l/Bj+ 1/p)-2Bj(dj-aj-B)/pjJy Hence, if FA and FB are the volume proportions of phase
1j
(36)
cr = 2BJ+3(Bj-2a)(dj-aj-B) A and phase B in the alloy, it is assumed that
FADA + FBDB
(dV) Y Y)
12DGBbGB ( D = . (45)
. . . (37)
dt D(1)j = BjkT crIj- Bj - P; The relationship can only be regarded as approximate
since it is assumed that matter is transported by diffusion
where to an extent related to the volume fractions of the phases
a2 + h2 present. For boundary diffusion it assumes that two types
pj~R=~ · (38) of boundary, a/a and fl/fl, rather than a/a, a/fl, and fl/fl
boundaries, are involved in the diffusion process. In
The sum of the contributions of all the grain boundaries is practice few data exist for diffusion along boundaries
expressed as between dissimilar phases. However, it is unlikely that the
(dV)
dt D(l)
= (dV)
dt D(1)0
+ f (dV)dt D(1)j
j= 1
· (39)
approach will lead to significant error unless the slowest
diffusion process is rate controlling and is appreciably less
rapid than other diffusional processes.
where, on the right hand side, the first term is due to the
coupled process without considering the effect of grain size,
and the second term is the summation of the contributions
of all other grain boundaries.
The values of ai' dj, bj, and crpj can be determined as10 Effect of stress state
aj = [R2+(R:-h)2_2R(R-h) cos (8-8)J1/2
From the analyses in the modelling,10 it is obvious that
= {h2+2R(R-h)[1-cos(8-8)J}1/2 '. (40) atomic diffusion depends only on the normal stress
_ d _ daj gradient, which provides the driving force for diffusion.
d (41a) Therefore, it seems that stress state alone does not directly
j - cos aj - Rsin(8-8)
influence the atomic diffusion in certain directions, if the
bj = dj-aj (41b) stress gradient is the same. However, it should be noted
that, for biaxial and triaxial stress states, the main flux of
6pj = 0·56p (1 + cos ~- ~ sin I1j cos I1j) · (42) atoms will be in the direction of the resultant gradient,
which is not necessarily the direction of the bondline. In
where this case, the atomic flux along the bond line will be
influenced by other components of the stress gradient.
8j = (l/R)j }
Moreover, stress state influences the degree of plastic
cos aj = (R/a) sin (8-8) · (43)
deformation and the creep behaviour. In a fixed condition,
aj = cos-1 [(R/a)sin(8-8) deformation may become easier, the fewer the compressive
Now, equations (34)-(37) can be solved by using equations stresses. In the case of diffusion bonding, humplike surface
(40)-(43). asperities may cause faster bonding than long ridges,
because the former result only in a uniaxial compressive
stress state when contacted together, whereas the latter
lead to a biaxial stress state (plane strain).
Similarly, the creep strain rate under uniaxial com-
Diffusion bonding of alloys pressive stress is higher than that under a biaxial or triaxial
compressive stress state. Therefore, for power law creep in
different stress states the constant A should be different.
As indicated in Ref. 13, it is obvious that the above In the equation
modelling for diffusion around a grain boundary is suitable
mainly for pure metals but may be used for some low- 8 = A'(cr/Gt . . . . (46)
alloyed materials. As far as most alloys are concerned, the constants A' and n are usually obtained from uniaxial
modifications need to be made. tensile creep tests. For creep in the biaxial stress state,
Darken 14 has proposed a phenomenological theory of which is the case in the present model, the concept of
diffusion in binary alloys based on the assumption that equivalent stress cre and equivalent strain Ce is
each constituent diffuses independently, relative to a fixed introduced.16 Hence
reference frame, and the atomic volume is essentially
constant. This theory has been proved to be identical to cre = (J3/2)cr (47a)
the mechanism of diffusion via vacant sites, if the vacant
sites are in local thermal equilibrium.15 This mechanism is
Ce = (2/J3)c (47b)
consistent with that of diffusion around a boundary in Therefore,
diffusion bonding. Therefore, the theory can be used in the
8 = (J3/2t+ A'lcr/Gln
1
present paper, in the absence of measured diffusion data.
Accordingly, if Fx and Fy are the atomic fractions of or
constituents X and Y in a binary solid solution alloy, the 8 = Alcr/Gln (48a)
diffusion coefficient is 15
A = (J3/2)n+ 1 A' (48b)
D = FxDy+FyDX ••••• • (44)
where A is the modified constant for the power law creep of
where Dx and Dy are the diffusivities of the components X long ridges in diffusion bonding.
Materials Science and Technology November 1987 Vol. 3
950 Guo and Ridley Modelling of diffusion bonding of metals
1.0 10 -
0 O.7MPo.
0 1.4M Pa \ • > 0.95 "to
0.8 \ o <0.95 to
0
l.LJ
0
• 2.1 M Pa \
\
_._.- Experim!ntal
Z
a \
(D
0.6
\ \
z
a
t=
« 0.4 -
lJ
0
Q..
o o 0-"
o. \
o 000.. \
\
\
0.::: L \ \
u- .•.
1.4MPa lLJ 0.\ \
« 0:::
. \
w
0:: 0.2 ~
«
til o O. ~.. \
V)
llJ
0::: \ \
n. \ \
o e\ \
\ \\
\ \
5 Comparison of present model (full lines) and that of \
Pilling et al.6 (dashed lines) with experimental
results7 (points) for Ti-6AI-4V; temperature 877°C;
grain size 5 Jim, surface roughness height 30 Jim,
wavelength 60 Jim
I
105
TIME,s
Comparison of predictions with 6 Comparison of predictions of present model (full
experimental data line) with predictions (dashed line) and experi-
mental results (points and chain dotted line) of
Pilling et al.6 for Ti-6AI-4V; temperature 927°C,
Few systematic experimental studies appear to have been grain size 5 Jim, surface roughness height 30 Jim, and
made of the factors which influence diffusion bonding in wavelength 60 Jim
metals and alloys. The most detailed data are those
available for Ti-6AI-4V alloy and for copper, and it is
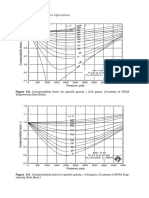
these data which have been compared with the predictions The area fraction bonded at 877°C for three pressures
of the· current model. The material parameters used in the (0'7, 1-4, and 2·1 MPa) for material of grain size 5 Jlm with
model are summarized in Table 1 for the titanium alloys constant surface roughness is shown in Fig. 5. The
and in Table 2 for copper. predictions of the present model are compared with those
of the diffusion bonding model of Pilling et al.6 and with
DIFFUSION BONDING OF Ti-6AI-4V ALLOY the limited experimental data of Pilling and Ridley.7 The
The experimental data on the diffusion bonding of present model shows an improved agreement with
titanium alloys available in the literature are surprisingly experiment for the times required to produce a sound bond
limited in view of the increasing commercial significance of (i.e. high area fraction bonded) compared with the earlier
the process as part of the superplastic forming-diffusion model.
bonding fabrication route, used particularly for the The combinations of pressures and times required to
manufacture of aerospace components. produce a sound bond at 927°C in Ti-6AI-4V alloy of
1.0
(0) (b) (c)
o
~o.'il
z
o
m
~ 0.6
~
«
•
•
----: --:::: J
600 700 800 900 1000
--- 500 600 700 800 900 600 700 eoo 900 1000
T EM PERATURE, °c . TEMPERATURE ,oC TEMPERATURE ,~C
a 3/60; b 30/250; c 70/500
7 Comparison of predictions of present model (full lines) and that of Derby and Wallach8 (dashed lines) with
experimental data18 (points) for pure copper at various values of surface roughness (height, Jim/wavelength,
Jim); pressure 4·9 MPa, time 4 min
Materials Science and Technology November 1987 Vol. 3
Guo and Ridley Modelling of diffusion bonding of metals 951
to Table 1 Material parameters for a-Ti and P-Ti (After
Ref. 17) and Ti-6AI-4V (After Ref. 6)
(0)
0 Parameter rx-Ti fJ-Ti
l.J.J
a 0.8
z a-Ti and P-Ti
0 Atomic volume, m3 1·76 x 10-29 1·81 x 10-29
co Burgers vector, m 2·95 x 10-10 2·86 x 10-10
« 0.6
LJJ
Shear modulus at 300 K,
MN m-2 4·36x104 2·05x103
0:: Temperature coefficient of
« shear modulus, K-l 6·2x10-4 2'6x10-4
--I Surface energy, J m-2 1·0 1·0
«
z: 0.4
Volume diffusion coefficient
(pre-exponential), m2 S-1 8·6 x 10-10 1·9 X 10-7
0 Volume diffusion activation
J- energy, kJ mol-1 150 153
u Grain boundary diffusion
« 0.2 coefficient (pre-exponential),
a::: m2s-1 6 x 10-7 9 x 10-8
u..
Grain boundary width, m 5·9 x 10-10 5·72 x 10-10
Grain boundary diffusion
activation energy, kJ mol-I 97 153
0 Ti-6AI-4V (stress dependence of strain rate)
1.0 Strain rate e=Ac<a/G)n
(b) Strain rate power law creep constant
Ac = A<5DGBO exp (- QGB/ RT) (G/KT)
D
w Exponent n = 1 ·43
Creep constant A' = 1·2 x 10-9
z 0_8
0
Yield stress ay= 9·4 x 108 x [1 -4 x 10-4 X (T -300)]
a
co
« 0.6
w
0::
« ) experimental data and that it shows an improvement over
the predictions of the Derby- Wallach model for the
roughest surface finish (Fig. 7c).
The effect of bonding pressure on the fractional area
/:/
--I
« 0.4 bonded at 800 e
for 4 min for two values of surface
0
z roughness is shown in Figs. 8a and 8b. Agreement between
.-« 0.2
a
u
the predictions of the present model and experimental data
is excellent, again with improved agreement for the
roughest surface finish compared with that of the Derby-
0::
u.. // Wallach model (Fig. 8b).
0-----·- -- The effect of time on the fractional area bonded at 550
for a bonding pressure of 35 MPa and constant surface
roughness is shown in Fig. 9. It can be seen that the
0
e
0.1 1.0 10 experimental results available show considerable scatter at
the relatively low bonding temperature involved, but the
BONDING PRESSURE,MPa predictions of the present model are in satisfactory
a 3/60; b 70/500 agreement with the data from Ref. 8.
8 Comparison of predictions of present model (full
lines) and that of Derby and Wallach8 (dashed lines)
with experimental data18 (points) for pure copper at
two values of surface roughness (height, Jlm/ Conclusions
wavelength, Jim); temperature-SOOo-C,time 4 min
A model for diffusion bonding has been established, which
grain size 5 ~m and constant surface roughness are shown is based on realistic geometric assumptions and which
in Fig. 6. The predictions of the present model are
compared with the experimental data of Pilling et ai.,6 for
which a sound bond is defined as one having a lap shear Table 2 Material parameters for copper (After Ref. 8)
strength which is greater than 95% of that of the parent
metal. The current model predicts essentially the correct Parameter Value
relationship between bonding pressure and time, but tends
to overestimate the times required to produce a sound Atomic volume, m 3
1·18 x 10-29
bond. However, the model shows an improvement over the Burgers vector, m 2·56 x 10-10
Melting temperature, K 1356
times predicted from the model of Pilling et al.6 The Shear modulus at 300 K, M N m-2 4·29 x 104
origins of the discrepancies are uncertain but could reflect Temperature coefficient of shear modulus, K-1 -3·97 X 10-4
the reliability of the available diffusion data. Surface energy, J m-2 1·75
Volume diffusion coefficient (pre-exponential), m2 S-1 6·2x10-5
Volume diffusion activation energy, kJ mol-1 207
DIFFUSION BONDING OF COPPER Product of grain boundary diffusion coefficient (pre-
The experimental data used are from the work of Ohashi exponential) and grain boundary width, m S-1
3
5·12x10-15
and Hashimoto,18 or are those measured by Derby and Grain boundary diffusion activation energy, kJ mol-1 105
Product of surface diffusion coefficient (pre-
Wallach.8 In Figs. 7a-c, the experimental and predicted exponential) and surface thickness, m3 S-1 . 6·0 X 10-10
effect of temperature on the fractional area bonded is Surface activation energy, kJ mol-1 205
shown for three values of surface roughness at a bonding Creep constant A' 7·42 x 105
Power law creep exponent n 4·8
pressure of 4·9 MPa and bonding time of 4 min. It can be
Normalized yield stress ay/ G 5 x 10-3
seen that the present model is in good agreement with the
Materials Science and Technology November 1987 Vol. 3
952 Guo and Ridley Modelling of diffusion bonding of metals
1.0 17. H. J. FROST and M. F. ASHBY: 'Deformation-mechanism maps',
D 44; 1982, Oxford, Pergamon Press. .
~ 0.8 18. O. OHASHI and T. HASHIMOTO: J. Jpn Weld. Soc., 1976,45,76.
o
CD
~ 0.6
0::
«
....J
« ./
...- .-----
z
o
i=
LJ
• Appendix 1
«
0::
LL.
Diffusion around grain boundary
o
o 20 40 60 B0 100 120
BONDING Tl ME, min From Ref. 13, it is known that the atom flux J from the
grain boundary to the void neck surface is given by
9 Comparison of predictions of present model (full
line) with predictions (dashed lines) and experi- -D
mental data (points) of Derby and WaliachB for J = nkT Vu · (49)
copper; temperature 550°C, pressure 35 M Pa,
surface roughness height 3 pm, wavelength 40 Jim where Vu is the chemical potential gradient of atoms. By
assuming a constant divergence of the flux along the
boundary and an identical chemical potential gradient for
grain boundary diffusion and volume diffusion, Johnson13
takes into account most of the possible mechanisms for
showed that
diffusion bonding. The effective diffusion distance and the
effects of grain size, phase proportions, and stress state are Vu = nV(J(r) · (50)
also considered quantitatively.
Therefore,
With the aid of the model, the time to form a sound
bond and the fractional area bonded after a certain time D
can be predicted. The changes in interfacial void volume VJ = - kT V2(J(r) = const · (51)
and void shape can be obtained as a function of bonding
time or fractional area bonded. The predictions of the
or
model have been compared with the experimental results V2(J(r) = const . (52)
available for Ti-6AI-4V and copper and show good From previous assumptions, equation (52) can be solved
agreement. using the following conditions (Fig. 2):
(i) (J~Y/p, atr=a
(ii) d(J/dr = 0, at r = a+B
Acknowledgments
(iii) r+ B
adr = a,8-y
The authors are grateful to Dr J. Pilling for helpful (iv) (J = (In at r > a+B
discussions. One of the authors (ZXG) wishes to thank the (v) (Jpd = (JIB+(Jn(d-a-B)
Government of the People's Republic of China and the
Thus,
British Council for financial support.
Va = ;2 [a,8-Y (1 + ;)] · (53)
2dB(Jp + [3(B - 2a)(d - a - B)
References x (l/B+ 1/p)-2B(d-a-B)/pJy
2B2 +3(B-2a)(d-a-B)
1. c. H. HAMILTON: in Proc. 2nd Int. Conf. on 'Titanium science
and technology', (ed. R. I. Jaffee and H. M. Burte), Vol. 1, (equation (6) above).
625; 1973, New York, Plenum Press. The atom fluxes through a grain boundary and through
2. G. GARMONG, N. E. PATON, and A. S. ARGON: Metall. Trans., the volume adjacent to the boundary are, respectively,
((J _ !B _!)
1975, 6A, 1269.
3. A. A. L. WHITE and D. J. ALLEN: in Proc. Conf. on 'Joining of 1 =_ 3DGB £5GB (54)
metals', Vol. 2, 96; 1981, London, Institution of Metallurgists. GB BkT I p
4. B. DERBY and E. R. WALLACH: Met. Sci., 1982, 16, 49.
5. B. DERBY and E. R. WALLACH: Met. Sci., 1984, 18, 427.
6. J. PILLING, D. W. LIVESEY, J. B. HAWKYARD, and N. RIDLEY: Met.
Sci., 1984, 18, 117.
J v ~~;(a,-i-~)
=- . (55)
7. J. PILLING and N. RIDLEY: in Proc. 5th Int. Conf. on 'Titanium For volume diffusion, it is assumed that the limiting area
science and technology' (ed. G. Lutjering et al.), Vol. 2, through which the flux travels is equal to the neck radius.
733-739; 1985, Oberursel, FRG, Deutsche Gesellschaft fur Therefore,
Metallkunde.
(dV)
8. B. DERBY and E. R. WALLACH: J. Mater. Sci., 1984, 19, 3140.
= -2JGB-4Jv
9. B. DERBY and E. R. WALLACH: J. Mater. Sci., 1984, 19,3149.
10. z. x. GUO: MSc thesis, University of Manchester, 1985. dt 0(1)
II. I. W. CHEN and A. S. ARGON: Acta Metall., 1981, 29, 1759. 6
=--(DGB£5GB+2Dvp) y
((JI---- y)
12. L. MARTINEZ and w. D. NIX: Scr. Metall., 1981, 15,757. BkT B p
13. D. L. JOHNSON: J. Appl. Phys., 1969,40, 192.
14. L. s. DARKEN: Met. Technol., 1948,15, (1), AIMME TP2311.
15. J. BARDEEN: Phys. Rev., 1949, 76, 1403. (dV) = JGB 2a = 6aDGB£5GB ((J _! _!)
dt 0(2) B B2 k T I B p
16. W. JOHNSON and P. B. MELLOR: 'Plasticity for mechanical
engineers', 57; 1962, London, Van Nostrand Reinhold. (equations (7) and (8) above).
Materials Science and Technology November 1987 Vol. 3
Guo and Ridley Modelling of diffusion bonding of metals 953
From Fig. 3, it can be shown that
Appendix 2 x = d_[R2_(y+R_h)2]1/2 (59)
Combining equations (58) and (59) we have
Power law creep (~~)c= I~I"J: SA {d-[R2_(Y~R-h)2r/2
. . . . . .
r
.
dy
. (60)
As shown in Fig. 3, the void ridge is divided into N slices; This corresponds to the rate of collapse of bond surfaces
N can be any integer that satisfies the accuracy required. due to creep. Thus,
The stress, acting on the ith slice with the thickness of h/ N
(i = 1,2, ... , N), is
(Ji = (d/xi)(Jp (56)
(~~)c(2) = 4a (~~ \ . . . . . . . . . . (61)
If a constant volume is assumed, the mass transferred into
so the strain rate of the slice is
the void due to the creep of the slice i is
Sz' = SAI(JJGln . . (57)
d~ = dhi X Xi = (h/N)SAI(JJGln dt Xi
From the definition of true strain and strain rate
Thus,
N
~i = (~~) = dei = Sidt = SAlo)GI"dt d Vc = 4 lim I d~
N-+(fJ i= 1
dhi = (h/N)SAI(JJGln dt Therefore,
N N h
dh = lim L dhi = SA lim L -lo)Gln dt
(ddtV) = 4dSAIa p/Gln
N-+(fJ i=l N-+(fJ i=l N C(l)
Therefore,
X f
h
0
{
d_[R2_(y+R_h)2]1/2
d }n-l dy
dh = SA fh I(JJGln dh . (58)
dt 0 (equation (12) above).
Magnetic materials have many
MAGNETISM important uses. This book begins
AND with a development of the basic
ideas of magnetism from first
MAGNETIC principles and goes on to an
MATERIALS elementary treatment of the
theory of different types of
J. ~ Jakubovics materials.
CONTENTS
.Fundamentals of magnetism • Bulk magnetic properties and
.Classification of materials by their measurement
magnetic properties • Magnetic materials for practical
applications
Book 401 210x148mm 148pp ISBN 0 904357 95 3 Paperback Published 1987
Price: UK£15.000ther Countries US$30.00{Institute of Metals members deduct 20%)
Student price: UK£6.00 Other countries US$12.00
Orders with remittance should be sent to: THE INSTITUTE OF METALS, 1 Carlton
House Terrace, London SW1Y 50B Tel. 01-839 4071 Telex 8814813 Fax. 01-839 2289
Orders originating in Canada and the United States should be sent direct to:
THE INSTITUTE OF METALS NORTH AMERICAN PUBLICATIONS CENTER, Old Post Road,
Brookfield, VT 05036, U.S.A. Tel. (802) 276 3162 Telex 759615 Brookfield UD
View publication stats Materials Science and Technology November 1987 Vol. 3
You might also like
- The Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeFrom EverandThe Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeRating: 4 out of 5 stars4/5 (5795)
- The Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreFrom EverandThe Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreRating: 4 out of 5 stars4/5 (1091)
- Never Split the Difference: Negotiating As If Your Life Depended On ItFrom EverandNever Split the Difference: Negotiating As If Your Life Depended On ItRating: 4.5 out of 5 stars4.5/5 (838)
- Hidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceFrom EverandHidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceRating: 4 out of 5 stars4/5 (895)
- Grit: The Power of Passion and PerseveranceFrom EverandGrit: The Power of Passion and PerseveranceRating: 4 out of 5 stars4/5 (588)
- Shoe Dog: A Memoir by the Creator of NikeFrom EverandShoe Dog: A Memoir by the Creator of NikeRating: 4.5 out of 5 stars4.5/5 (537)
- The Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersFrom EverandThe Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersRating: 4.5 out of 5 stars4.5/5 (345)
- Elon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureFrom EverandElon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureRating: 4.5 out of 5 stars4.5/5 (474)
- Her Body and Other Parties: StoriesFrom EverandHer Body and Other Parties: StoriesRating: 4 out of 5 stars4/5 (821)
- The Emperor of All Maladies: A Biography of CancerFrom EverandThe Emperor of All Maladies: A Biography of CancerRating: 4.5 out of 5 stars4.5/5 (271)
- The Sympathizer: A Novel (Pulitzer Prize for Fiction)From EverandThe Sympathizer: A Novel (Pulitzer Prize for Fiction)Rating: 4.5 out of 5 stars4.5/5 (121)
- The Little Book of Hygge: Danish Secrets to Happy LivingFrom EverandThe Little Book of Hygge: Danish Secrets to Happy LivingRating: 3.5 out of 5 stars3.5/5 (400)
- The World Is Flat 3.0: A Brief History of the Twenty-first CenturyFrom EverandThe World Is Flat 3.0: A Brief History of the Twenty-first CenturyRating: 3.5 out of 5 stars3.5/5 (2259)
- The Yellow House: A Memoir (2019 National Book Award Winner)From EverandThe Yellow House: A Memoir (2019 National Book Award Winner)Rating: 4 out of 5 stars4/5 (98)
- Devil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaFrom EverandDevil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaRating: 4.5 out of 5 stars4.5/5 (266)
- A Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryFrom EverandA Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryRating: 3.5 out of 5 stars3.5/5 (231)
- Team of Rivals: The Political Genius of Abraham LincolnFrom EverandTeam of Rivals: The Political Genius of Abraham LincolnRating: 4.5 out of 5 stars4.5/5 (234)
- On Fire: The (Burning) Case for a Green New DealFrom EverandOn Fire: The (Burning) Case for a Green New DealRating: 4 out of 5 stars4/5 (74)
- The Unwinding: An Inner History of the New AmericaFrom EverandThe Unwinding: An Inner History of the New AmericaRating: 4 out of 5 stars4/5 (45)
- Concise Guide To Quantum Computing: Sergei Kurgalin Sergei BorzunovDocument129 pagesConcise Guide To Quantum Computing: Sergei Kurgalin Sergei BorzunovVi Kem0% (1)
- State of The Art of MicromachiningDocument16 pagesState of The Art of MicromachiningMarcionilo NeriNo ratings yet
- Evaluation On Diffusion Bonded Joints of TiAl Alloy To Ti3SiC2Document6 pagesEvaluation On Diffusion Bonded Joints of TiAl Alloy To Ti3SiC2Marcionilo NeriNo ratings yet
- Interfacial Microstructure and Mechanical Property of BrazedDocument8 pagesInterfacial Microstructure and Mechanical Property of BrazedMarcionilo NeriNo ratings yet
- Processing, Microstructure and Mechanical Properties of Vacuum-Brazed Al2O3-Ti6Al4V JointsDocument6 pagesProcessing, Microstructure and Mechanical Properties of Vacuum-Brazed Al2O3-Ti6Al4V JointsMarcionilo NeriNo ratings yet
- Aluminetos de TitânioDocument7 pagesAluminetos de TitânioMarcionilo NeriNo ratings yet
- Guedes-Pinto2012 Article ActiveMetalBrazingOfMachinableDocument7 pagesGuedes-Pinto2012 Article ActiveMetalBrazingOfMachinableMarcionilo NeriNo ratings yet
- Formation of Intermetallics During Brazing of Alumina With Fe, Ni and CR Using Ag-30 Cu-10 SN As Filler MetalDocument7 pagesFormation of Intermetallics During Brazing of Alumina With Fe, Ni and CR Using Ag-30 Cu-10 SN As Filler MetalMarcionilo NeriNo ratings yet
- Effects of Interfacial Reaction and Atomic Diffusion On The Mechanical Property of Ti3SiC2 Ceramic To Cu Brazing Joints, VacuumDocument7 pagesEffects of Interfacial Reaction and Atomic Diffusion On The Mechanical Property of Ti3SiC2 Ceramic To Cu Brazing Joints, VacuumMarcionilo NeriNo ratings yet
- Growth Kinetics of Reaction Layers Formed During Diffusion Bonding of SiC Ceramic To TiAl AlloyDocument5 pagesGrowth Kinetics of Reaction Layers Formed During Diffusion Bonding of SiC Ceramic To TiAl AlloyMarcionilo NeriNo ratings yet
- Effect of Brazing Temperature On The Shear Strength of InconelDocument8 pagesEffect of Brazing Temperature On The Shear Strength of InconelMarcionilo NeriNo ratings yet
- Data Sheet TI-6AL-4VDocument3 pagesData Sheet TI-6AL-4VMarcionilo NeriNo ratings yet
- Joining of Ti6Al4V To Al2O3 Using NanomultilayersDocument15 pagesJoining of Ti6Al4V To Al2O3 Using NanomultilayersMarcionilo NeriNo ratings yet
- Effect of Thermal Residual Stresses On The Strength For BothDocument13 pagesEffect of Thermal Residual Stresses On The Strength For BothMarcionilo NeriNo ratings yet
- Effect of ZnO Whisker Content On Sinterability and Fracture Behaviour of PZTDocument6 pagesEffect of ZnO Whisker Content On Sinterability and Fracture Behaviour of PZTMarcionilo NeriNo ratings yet
- A Study of The Transient Liquid Phase Bonding Process Applied To A Ag-Cu-Ag Sandwich JointDocument12 pagesA Study of The Transient Liquid Phase Bonding Process Applied To A Ag-Cu-Ag Sandwich JointMarcionilo NeriNo ratings yet
- Diffusion Bonding of Ti-Ni Under The InfluenceDocument10 pagesDiffusion Bonding of Ti-Ni Under The InfluenceMarcionilo NeriNo ratings yet
- Brazing: Selection Brazing Processes and Filler MetalsDocument14 pagesBrazing: Selection Brazing Processes and Filler MetalsMarcionilo NeriNo ratings yet
- Aços Memoria de FormaDocument12 pagesAços Memoria de FormaMarcionilo NeriNo ratings yet
- Data Sheet Al2O3 IIDocument3 pagesData Sheet Al2O3 IIMarcionilo NeriNo ratings yet
- Al O 99.8% Datasheet: Material Properties Properties Test Standard Unit ValueDocument1 pageAl O 99.8% Datasheet: Material Properties Properties Test Standard Unit ValueMarcionilo NeriNo ratings yet
- I 3031Document222 pagesI 3031Daniel Venancio VieiraNo ratings yet
- Manual - Book-Vektor Ablerex - MsiiDocument24 pagesManual - Book-Vektor Ablerex - MsiiIndonesian's LOVENo ratings yet
- TEMA Shell Bundle Entrance and Exit AreasDocument3 pagesTEMA Shell Bundle Entrance and Exit AreasArunkumar MyakalaNo ratings yet
- VD 2Document4 pagesVD 2Hồ BảoNo ratings yet
- DIP Unit 1 MCQDocument12 pagesDIP Unit 1 MCQSanthosh PaNo ratings yet
- ACPI Embedded SATAIII mSATA SSD MSS4Q-L 3K PE Datasheet 20190611Document16 pagesACPI Embedded SATAIII mSATA SSD MSS4Q-L 3K PE Datasheet 20190611Daniel CrespoNo ratings yet
- 14 - Part 3 - Ch. 7 - A Lesson With Czerny - Tempo Rubato PDFDocument21 pages14 - Part 3 - Ch. 7 - A Lesson With Czerny - Tempo Rubato PDFErduandNo ratings yet
- DrilflexDocument4 pagesDrilflexmurdicksNo ratings yet
- EE203 01 Digital and Number SystemsDocument34 pagesEE203 01 Digital and Number SystemsrayNo ratings yet
- Inorganic Qualitative AnalysisDocument9 pagesInorganic Qualitative AnalysisShireen SuhailNo ratings yet
- Icm U1d7 Simplifying Exponential Expressions 1Document2 pagesIcm U1d7 Simplifying Exponential Expressions 1kazamNo ratings yet
- TURCKDocument3 pagesTURCKNegreaDanielaNo ratings yet
- Manual Hameg 3010Document27 pagesManual Hameg 3010Yéred Josué Rugama MontenegroNo ratings yet
- Basic Aerodynamic TheoryDocument24 pagesBasic Aerodynamic TheoryOganga JackNo ratings yet
- QUCM CCM Application ManualDocument30 pagesQUCM CCM Application ManualibanvegaNo ratings yet
- Prospect ATR901.RevB Eng PDFDocument25 pagesProspect ATR901.RevB Eng PDFUntea LiviuNo ratings yet
- Cast Iron Gate Valve Non Rising Stem PN16: Features & BenefitsDocument1 pageCast Iron Gate Valve Non Rising Stem PN16: Features & BenefitsMohamed RaafatNo ratings yet
- Torque - Slip Characteristic of A Three - Phase Induction MachineDocument28 pagesTorque - Slip Characteristic of A Three - Phase Induction MachineAli AltahirNo ratings yet
- Group 4 Winds and CloudsDocument15 pagesGroup 4 Winds and Cloudsghita hasnaNo ratings yet
- Biology Final Exam - Print - QuizizzDocument5 pagesBiology Final Exam - Print - QuizizzrubelliteNo ratings yet
- Lab Exe No. 10 - Design of Simple Bioethanol Production System - FermentationDocument2 pagesLab Exe No. 10 - Design of Simple Bioethanol Production System - FermentationJamil Mansor M. LaguiabNo ratings yet
- Finite Element Analysis Using MATLAB ANSYS 16 HrsDocument2 pagesFinite Element Analysis Using MATLAB ANSYS 16 HrsMOHIT PANTNo ratings yet
- Advances in High-Performance CoolingDocument15 pagesAdvances in High-Performance Coolingygrao100% (1)
- Fourth Floor PlanDocument1 pageFourth Floor Planvenkatalakshmi natarasanNo ratings yet
- Gravedad Especifico PDFDocument1 pageGravedad Especifico PDFCrisca FumeNo ratings yet
- Testing ResumeDocument4 pagesTesting ResumeBaji TulluruNo ratings yet
- General Handout - MathematicsDocument7 pagesGeneral Handout - MathematicsRaven St. LouisNo ratings yet
- CNQ#1 PDFDocument32 pagesCNQ#1 PDFareejaNo ratings yet
- Differential Diagnosis of Finger DropDocument4 pagesDifferential Diagnosis of Finger DropR HariNo ratings yet