Professional Documents
Culture Documents
Para Lab Part 1
Uploaded by
Vanessa May Blancio0 ratings0% found this document useful (0 votes)
16 views2 pagespaera
Original Title
para lab part 1
Copyright
© © All Rights Reserved
Available Formats
DOCX, PDF, TXT or read online from Scribd
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this Documentpaera
Copyright:
© All Rights Reserved
Available Formats
Download as DOCX, PDF, TXT or read online from Scribd
0 ratings0% found this document useful (0 votes)
16 views2 pagesPara Lab Part 1
Uploaded by
Vanessa May Blanciopaera
Copyright:
© All Rights Reserved
Available Formats
Download as DOCX, PDF, TXT or read online from Scribd
You are on page 1of 2
INTRODUCTION TO PARA LAB Common terms used for consistency
General symptoms of parasitic infection Watery Loose
Fever Pain Mucoid Mushy
Chills Diarrhea Hard Soft-formed
Fatigue Loss of vitality
Patient’s History- very important in diagnosing parasitic CHEMICAL EXAMINATION
infection 1. Ph – LITMUS PAPER
Specimens used to examine parasites - Clues for metabolic conditions
Stool Urine - Influenced by patient’s diet
Sputum Aspirations - Reported as alkaline or acidic
Blood 2. Occult Blood
- Black with tarry consistency- large amount of
Ova and parasite Fecal examination occult blood
Malarial parasite Blood examination - Indicative of lesion in the intestinal tract due to
Immunologic test Parasitic infection
Ectoparasite (OUTSIDE) Cancerous tumor
- Easily seen and identified Bleeding ulcers
Endoparasite (INSIDE) - Occult blood test is primarily done to screen for
- Intestinal and atrial parasite COLO-RECTAL CANCER
- Blood and tissue parasite - Positive test: BLUE-COLOR
“Examining intestinal parasite, used stool specimen” - Positive and negative control must be run
SPECIMEN COLLECTION - Occult blood detects presence of small amount
- Clean, dry, wide-mouth, leak-proof container, of blood by detecting HB’S PSEUDOPEROXIDASE
tight fitting lid ACTIVITY
- Patient’s names, time, and date of collection - PEROXIDASE from vegetables may cause FALSE
- Process within 2 HOURS POSITIVE when large amount in diet
3 PHASES OF ROUTINE STOOL EXAM 3 types of chemical methods
Macroscopic- color and consistency Benzidine method- most sensitive and
Chemical- pH and occult blood cheapest
Microscopic O- toluidine method
MICROSCOPIC (PHYSICAL EXAMINATION) Gum Guiac method- most specific and most
1. Color expensive
- Presence of parasite and metabolic disorders “Antibodies against human hemoglobin and erythrocytes to
- Presence of blood should be noted detect blood from specimen”
Bright red Bleeding hemorrhoids
Bloody mucus Amoebic ulceration in large MACROSCOPIC EXAMINATION METHODS
(loose/liquid specimen) intestine I. Direct fecal smear (Saline mount/ Wet mount
Bloody tinged Trophic amoebae - Fresh or preserved fecal specimen
(Patches of mucus) - Helminth ova, larvae, protozoan cysts and
Occult blood Gastrointestinal disorder trophozoites
Dark colored blood Bleeding in high up - 2 mg to stool mixed with drop of NSS and
gastrointestinal tract
covered with coverslip
Bright colored blood Bleeding at lower
II. Iodine Wet mount
gastrointestinal tract
- Drop of iodine solution to saline mount or new
“O & P examination- routinely performed for ova and parasite
mount using iodine alone
/occult blood exam”
- Increases the visibility of protozoan cysts
2. Consistency
- Example: weak iodine solution
Indication of the types of organism
Lugol’s
Formed Semi-formed
Mushy Liquid D’ Antoni’s
Dobell and O’ conner
Liquid and soft stool Trophozoites III. Kato thick smear
Fully formed stool Cysts - 50-60 mg of fresh stool and covered with a CUT
Liquid/formed stool Helminths eggs CELLOPHANE soaked in a mixture of MALACHITE
GREEN GLYCERINE
- Alternative: colored cellophane strip - 40% HCl to dissolve albuminous materials
- Thin-shelled ova- difficult to find due to clearing - Ether to dissolve neutral fats
agent (GLYCERINE) - Protozoan cysts may be destroyed using this
- Most sensitive for detection of thick-shelled ova technique
- Cannot be used for liquid stool and preserved Quantitative and Semi-quantitative methods
samples X. Stoll’s egg counting methods
IV. Kato-Katz smear - Used for formed and liquid stools
- Uses a measured amount of stool for - Samples are dilutes using 0.1 N NaOH
quantitation - Exact amount is measured using STOLL PIPETTE
- Samples passed through SIEVE to remove large and read microscope
particles - Counts are multiplied by a factor depending on
- All eggs are counted and multiplied by a factor the amount of stool sample used and the
depending on the amount of stool used consistency
CONCENTRATION METHODS XI. Kato-Katz
- Increased the possibility of detecting parasites - Only for formed stool samples
when only few are present in feces
Floating methods
- Uses liquid of a higher specific gravity
- Top layer is skimmed from the surface and
examined microscopically
- Performed with METICULOUS ATTENTION
- Ova and cysts are selectively floated out from
the sample using a reagent with a high specific
gravity
- Not useful for detecting trophozoites
- Uses large amount of stool, increases the
specificity
V. Zinc Sulfate Flotation
- 33% zinc sulfate with S.G of 1.18-1.20
- Provide cleaner concentration
- Good for recovery of protozoan cysts and most
eggs
VI. Sheather’s sugar flotation
- Boiled sucrose solution preserved with phenol
Sedimentation methods
- Present in large amount of feces about 2 gm of
sediments
Straining the stool Remove large particles
Formalizing To kill and preserve the
organisms
Adding a solvent such as To dissolve fats
ether or ethyl acetate
- Uses a reagent with LOW SPECIFIC GRAVITY
- The parasite sinks to the bottom
- Not useful for detecting trophozoites
VII. Formalin ether concentration technique
(FECT/Ritchie’s method)
- 10% formalin used as preservatives
- Ether to dissolve neutral fats and sediment the
parasite
- Ether is explosive and flammable so it may
replaced with ether acetate
VIII. KOH Method for Cyclospora
- 10% KOH and saline
IX. Acid ether concentration technique (AECT)
You might also like
- College of Medical Laboratory Science Our Lady of Fatima University-VelenzuelaDocument33 pagesCollege of Medical Laboratory Science Our Lady of Fatima University-VelenzuelaClaire GonoNo ratings yet
- Introduction To Parasitology Laboratory - A - SRGDocument3 pagesIntroduction To Parasitology Laboratory - A - SRGGAIL BATHANNo ratings yet
- CM Stool Examination (Ocfemia, Eliazel Galorio - BSLABSCI-PLTCIDocument30 pagesCM Stool Examination (Ocfemia, Eliazel Galorio - BSLABSCI-PLTCIeliazel ocfemiaNo ratings yet
- Parasitologyclass 2021Document207 pagesParasitologyclass 2021notsoninjaninjaNo ratings yet
- Seminal Fluid AnalysisDocument11 pagesSeminal Fluid AnalysisKenneth Jake BatiduanNo ratings yet
- UrinalysisDocument16 pagesUrinalysisJestha Parayno De Vera100% (6)
- Non Blood SpecimenDocument4 pagesNon Blood Specimendorothymaebanks970No ratings yet
- Semen Analysis - Aubf (BSMLS Olfu)Document8 pagesSemen Analysis - Aubf (BSMLS Olfu)Mitch IbayNo ratings yet
- Microscopic Exam Body FluidsDocument61 pagesMicroscopic Exam Body FluidsCarl PinedaNo ratings yet
- Lab Exercise 10 Stool Specimen CollectionDocument12 pagesLab Exercise 10 Stool Specimen CollectionArianne Jans Munar100% (1)
- (HANDOUT) Phar 112 Lab - Fecalysis and Fecal Occult Blood TestDocument2 pages(HANDOUT) Phar 112 Lab - Fecalysis and Fecal Occult Blood TestHan SoloNo ratings yet
- Stool Exam - Routine, Conc. Method - Students'Document27 pagesStool Exam - Routine, Conc. Method - Students'AnastasiaNo ratings yet
- EXPERIMENTDocument58 pagesEXPERIMENTYriel GuilaranNo ratings yet
- Maldigestion/malabsorption Syndromes: Fecal AnalysisDocument2 pagesMaldigestion/malabsorption Syndromes: Fecal AnalysisMonica DomingoNo ratings yet
- Topic 9 The Clinical Microscopy SectionDocument4 pagesTopic 9 The Clinical Microscopy SectionHanna EstebanNo ratings yet
- Lesson 6 - Coccidian PDFDocument11 pagesLesson 6 - Coccidian PDFAnya IgnacioNo ratings yet
- Iii. Medical Management A. Diagnostic and Laboratory ProceduresDocument11 pagesIii. Medical Management A. Diagnostic and Laboratory ProceduresNickaela CalalangNo ratings yet
- MLSP Lec UrinalysisDocument7 pagesMLSP Lec UrinalysisALEXANDRA MAE MAIPIDNo ratings yet
- Amoeba: Ms. Helga SyDocument7 pagesAmoeba: Ms. Helga Syanti romantic txtNo ratings yet
- E. Histolytica: Associated DiseasesDocument11 pagesE. Histolytica: Associated DiseasesCorinne MandrezaNo ratings yet
- Routine FecalysisDocument25 pagesRoutine FecalysisE. B. F.No ratings yet
- (PROTOZOANS) Phylum ApicomplexaDocument8 pages(PROTOZOANS) Phylum ApicomplexaEunice AndradeNo ratings yet
- Introduction To UrinalysisDocument8 pagesIntroduction To UrinalysisKyle PicocNo ratings yet
- Clinical Microscopy: Standard Operating Procedures FORDocument8 pagesClinical Microscopy: Standard Operating Procedures FORRochell OcampoNo ratings yet
- Accurate Diagnosis of Parasitic Infections Is Important To Decrease The Prevalence andDocument4 pagesAccurate Diagnosis of Parasitic Infections Is Important To Decrease The Prevalence andManulat VicaiiNo ratings yet
- Semen AnalysisDocument31 pagesSemen AnalysisVinodkhanna Kairamkonda100% (1)
- Clinical Pathology (Body Fluids) 4 SemesterDocument12 pagesClinical Pathology (Body Fluids) 4 SemesterSiddhi KaulNo ratings yet
- Routine Stool ExaminationDocument2 pagesRoutine Stool ExaminationarvenemartinNo ratings yet
- Clinical Pathology Final PresentationDocument16 pagesClinical Pathology Final Presentationmomin.laangNo ratings yet
- GROUP 1 - Sweat and UrineDocument9 pagesGROUP 1 - Sweat and Urinechocoholic potchiNo ratings yet
- CLM IiDocument24 pagesCLM IiKuma BelloNo ratings yet
- Clin Path Lab 6 Urinalysis Part 2Document7 pagesClin Path Lab 6 Urinalysis Part 2api-3743217100% (3)
- Urinalysis - BALOLOY (Moonchild)Document6 pagesUrinalysis - BALOLOY (Moonchild)Rose Andrea De los SantosNo ratings yet
- Intro To para Lab Part 2Document1 pageIntro To para Lab Part 2Vanessa May BlancioNo ratings yet
- Body FluidsDocument81 pagesBody FluidsAris ResurreccionNo ratings yet
- Stool AnalysisDocument11 pagesStool AnalysisMohsen Haleem100% (1)
- Aubf PDFDocument48 pagesAubf PDFJeaniz dignosNo ratings yet
- Far Eastern University - Nicanor Reyes Medical Foundation: Clinical Diagnosis B Lab Activity - Stool Analysis OrientationDocument3 pagesFar Eastern University - Nicanor Reyes Medical Foundation: Clinical Diagnosis B Lab Activity - Stool Analysis OrientationKaren EstavilloNo ratings yet
- NS1 Case PresDocument8 pagesNS1 Case PresjoanaalpayNo ratings yet
- Clinical MicrosDocument14 pagesClinical MicrosKRISTINE JOY PANGAHINNo ratings yet
- Paps SmearDocument51 pagesPaps SmearCatherine MerillenoNo ratings yet
- Seminal and Other Body Fluids Module PrelimDocument6 pagesSeminal and Other Body Fluids Module PrelimRachelle OzicniNo ratings yet
- Parasitology Lab M2 Routine FecalysisDocument4 pagesParasitology Lab M2 Routine Fecalysiseumhir7No ratings yet
- Forensic Chemistry Module 2Document11 pagesForensic Chemistry Module 2Andrhey BayanNo ratings yet
- Prof. Ethel Marie Mangada, RMT: Microscopic Examination of UrineDocument17 pagesProf. Ethel Marie Mangada, RMT: Microscopic Examination of UrineKelvin CafirmaNo ratings yet
- Salmonella: DR - Vasanthi .R Professor Dept Microbiology ChriDocument52 pagesSalmonella: DR - Vasanthi .R Professor Dept Microbiology ChrividhyashreeNo ratings yet
- AUBF Lec M Microscopic Examination of Urine Sediment Part 1Document11 pagesAUBF Lec M Microscopic Examination of Urine Sediment Part 1Haniah DsNo ratings yet
- Culture Media & Culture MethodsDocument60 pagesCulture Media & Culture MethodsbiochemiNo ratings yet
- Microscopic Examination of UrineDocument13 pagesMicroscopic Examination of UrineMark Raymund Galvez Nava100% (1)
- Stool ExaminationDocument93 pagesStool ExaminationWajid SultanNo ratings yet
- Se Me N Analysis - C Asa: Somanatha Sharma. S II Year Resident Stanley Medical CollegeDocument55 pagesSe Me N Analysis - C Asa: Somanatha Sharma. S II Year Resident Stanley Medical CollegephoenixibexNo ratings yet
- GastrontestinalSystem PDFDocument1 pageGastrontestinalSystem PDFGeorge KanparNo ratings yet
- AmoebaeDocument23 pagesAmoebaeMeenakshisundaram CNo ratings yet
- Physical Examination of Urine: Analysis of Urine and Other Body Fluids Module 4Document19 pagesPhysical Examination of Urine: Analysis of Urine and Other Body Fluids Module 4Francis ValdezNo ratings yet
- General Stool ExaminationDocument6 pagesGeneral Stool Examinationزكريا خالدNo ratings yet
- PMLS Transes W1Document9 pagesPMLS Transes W1J BatumbakalNo ratings yet
- Hematuria, (Blood in Urine) A Simple Guide to The Condition, Related Diseases And Use in Diagnosis of DiseasesFrom EverandHematuria, (Blood in Urine) A Simple Guide to The Condition, Related Diseases And Use in Diagnosis of DiseasesRating: 5 out of 5 stars5/5 (2)
- How To Draw Blood From a Goat: How To Collect and Send Specimens to Test for Pregnancy, Johnes, CAE, CL, and MoreFrom EverandHow To Draw Blood From a Goat: How To Collect and Send Specimens to Test for Pregnancy, Johnes, CAE, CL, and MoreNo ratings yet
- Quiz 1 PharmaDocument13 pagesQuiz 1 PharmaVanessa May BlancioNo ratings yet
- Clinical Chemistry 2 2ueDocument18 pagesClinical Chemistry 2 2ueVanessa May BlancioNo ratings yet
- Para CompleteDocument120 pagesPara CompleteVanessa May BlancioNo ratings yet
- Clinical Microscopy ExamsDocument36 pagesClinical Microscopy ExamsVanessa May BlancioNo ratings yet
- AUBF Reinforcement ExamDocument67 pagesAUBF Reinforcement ExamVanessa May BlancioNo ratings yet
- 2nd Ue Pharma - MergedDocument90 pages2nd Ue Pharma - MergedVanessa May BlancioNo ratings yet
- Gantt Chart FinalDocument2 pagesGantt Chart FinalVanessa May BlancioNo ratings yet
- Intro To para Lab Part 2Document1 pageIntro To para Lab Part 2Vanessa May BlancioNo ratings yet
- Introduction To Parasitology 2022Document7 pagesIntroduction To Parasitology 2022Vanessa May BlancioNo ratings yet
- Genetics TestDocument26 pagesGenetics TestBaitz5No ratings yet
- Para Lec FinalsDocument3 pagesPara Lec FinalsVanessa May BlancioNo ratings yet
- Course Offerings - First Sem 2020Document21 pagesCourse Offerings - First Sem 2020Vanessa May BlancioNo ratings yet
- AUP Student Life Care Plan For OFFCAMDocument1 pageAUP Student Life Care Plan For OFFCAMVanessa May BlancioNo ratings yet
- Session 1, Introduction To GlobalizationDocument38 pagesSession 1, Introduction To GlobalizationVanessa May BlancioNo ratings yet
- Session 2 The Global EconomyDocument45 pagesSession 2 The Global EconomyVanessa May BlancioNo ratings yet
- Global Inter-State System (Political Globalization)Document43 pagesGlobal Inter-State System (Political Globalization)Vanessa May BlancioNo ratings yet
- Aspirin WorksheetDocument12 pagesAspirin WorksheetVanessa May BlancioNo ratings yet
- Bwiset PlanDocument2 pagesBwiset PlanVanessa May BlancioNo ratings yet
- Acknowledgement, Dedi, AbsDocument3 pagesAcknowledgement, Dedi, AbsVanessa May BlancioNo ratings yet
- Calibration HOMEWORKDocument1 pageCalibration HOMEWORKVanessa May BlancioNo ratings yet
- Lab7 Panugao, KAC Th4 7Document3 pagesLab7 Panugao, KAC Th4 7Vanessa May BlancioNo ratings yet
- Jack Ma's Story Behind His SuccessDocument2 pagesJack Ma's Story Behind His SuccessVanessa May BlancioNo ratings yet
- Jack Ma's Story Behind His SuccessDocument2 pagesJack Ma's Story Behind His SuccessVanessa May BlancioNo ratings yet
- Maganda Si VanessaDocument4 pagesMaganda Si VanessaVanessa May BlancioNo ratings yet
- Where Did I Come From?Document1 pageWhere Did I Come From?Vanessa May BlancioNo ratings yet
- 16 Digestive SystemDocument5 pages16 Digestive SystemVanessa May BlancioNo ratings yet
- Table of ContentsDocument2 pagesTable of ContentsVanessa May BlancioNo ratings yet
- A Case Study On The Insufficiency of Pharmaceutical Practitioners in Katapatan PharmacyDocument1 pageA Case Study On The Insufficiency of Pharmaceutical Practitioners in Katapatan PharmacyVanessa May BlancioNo ratings yet
- NothingDocument79 pagesNothingVanessa May BlancioNo ratings yet
- Review Problems Chapter 6Document8 pagesReview Problems Chapter 6Yue FeiNo ratings yet
- Trockel Flash Art 1987Document4 pagesTrockel Flash Art 1987cvg_geNo ratings yet
- Roy B. Kantuna, MPRM, CESE: (For Midterm) (Instructor)Document8 pagesRoy B. Kantuna, MPRM, CESE: (For Midterm) (Instructor)Athena Mae DitchonNo ratings yet
- Juan LunaDocument2 pagesJuan LunaClint Dave OacanNo ratings yet
- ADCD LowDocument8 pagesADCD LowrahulmultivisionNo ratings yet
- Advanced Calculus For Applications - Francis B Hilderand 1962Document657 pagesAdvanced Calculus For Applications - Francis B Hilderand 1962prabu201No ratings yet
- UNIT 5 Standard Costing - Variance AnalysisDocument50 pagesUNIT 5 Standard Costing - Variance AnalysisMohsin SheikhNo ratings yet
- UntitledDocument4 pagesUntitledapi-223522684No ratings yet
- Saes TABLEDocument13 pagesSaes TABLERiyaz BasheerNo ratings yet
- 1250kva DG SetDocument61 pages1250kva DG SetAnagha Deb100% (1)
- Sociology Internal AssessmentDocument21 pagesSociology Internal AssessmentjavoughnNo ratings yet
- ESET TechnologyDocument21 pagesESET TechnologyValentin SalcianuNo ratings yet
- Litotriptor Intracorporeo NeumaticoDocument4 pagesLitotriptor Intracorporeo NeumaticoJuan VasquezNo ratings yet
- Wooden Buildings: exposed to tiếp xúc với dramatic renewal sự làm mới đáng kểDocument6 pagesWooden Buildings: exposed to tiếp xúc với dramatic renewal sự làm mới đáng kểNguyễn Phạm Thảo NguyênNo ratings yet
- Āyāt Al-Aḥkām, Ayat Al-AhkamDocument6 pagesĀyāt Al-Aḥkām, Ayat Al-AhkamRasoul NamaziNo ratings yet
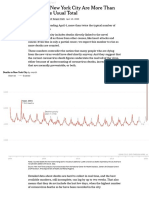
- Deaths in New York City Are More Than Double The Usual TotalDocument3 pagesDeaths in New York City Are More Than Double The Usual TotalRamón RuizNo ratings yet
- Research Propsal v1.2Document3 pagesResearch Propsal v1.2john7904No ratings yet
- English The Smiles and Tears of RasoolullahDocument130 pagesEnglish The Smiles and Tears of RasoolullahwildqafNo ratings yet
- Dassault Systems Academic CalenderDocument5 pagesDassault Systems Academic CalenderSarath KumarNo ratings yet
- BarDocument1 pageBarJoannalyn Libo-onNo ratings yet
- 78EZDM Product Specifications Conector para Cable de 7 OctavosDocument4 pages78EZDM Product Specifications Conector para Cable de 7 OctavosEdwin Santiago QuispeNo ratings yet
- Language - Introduction To The Integrated Language Arts CompetenciesDocument7 pagesLanguage - Introduction To The Integrated Language Arts CompetenciesHari Ng Sablay100% (1)
- Expt Lipid - Influence of Bile in The Action of LipaseDocument3 pagesExpt Lipid - Influence of Bile in The Action of LipaseAngela CaguitlaNo ratings yet
- The Trend Trader Nick Radge On Demand PDFDocument8 pagesThe Trend Trader Nick Radge On Demand PDFDedi Tri LaksonoNo ratings yet
- Group3 The Development of Moral Character of Moral Agent EspantoHipolitoLepitinNamoc 1Document5 pagesGroup3 The Development of Moral Character of Moral Agent EspantoHipolitoLepitinNamoc 1Novelyn DuyoganNo ratings yet
- CBLM FinalDocument33 pagesCBLM Finalopep7775% (8)
- Arid Agriculture University, RawalpindiDocument4 pagesArid Agriculture University, RawalpindiIsHa KhAnNo ratings yet
- Am Jetstream Pre-Int Unit 8 Lesson 2Document2 pagesAm Jetstream Pre-Int Unit 8 Lesson 2Jennyfer Guevara50% (2)
- Draft Technical Notes SGLG 2023 - National OrientationDocument109 pagesDraft Technical Notes SGLG 2023 - National OrientationZane ZyneNo ratings yet
- Case Presentation On Management of Depressive Disorders-1Document40 pagesCase Presentation On Management of Depressive Disorders-1Fatima MuhammadNo ratings yet