Professional Documents
Culture Documents
Class-XII Holiday Assignment
Uploaded by
alta alpOriginal Title
Copyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
Class-XII Holiday Assignment
Uploaded by
alta alpCopyright:
Available Formats
SAI International School
Holiday Assignment
Class-XII
Date of Submission: 26.06.2023
Subject : English
The following are the topics for project as a part of Internal Assessment
There are two topics, you have to do the project on any one topic adhering to the details
given along with the topics.
The project should be handwritten. No typed or printed material will be accepted.
You have to submit the project in a stick file.
Word Limit – 1000
1. The plight of slum dwellers is inescapable poverty in perpetuity
(Prepare a survey report on the basis of evidences from your visit to a nearby slum)
Cover page
Acknowledgement
Index/Table of contents
Slum dwellers’ life- An insight
My visit- the inexplicable story
Childhood- A lost spring and the hollow promises
Where the road ends- An exit possible?
OR
2. Charley of THE THIRD LEVEL is a universal phenomenon of urban chaos, insecurity at large
and the hunt for escape from reality
(Interview several youths to explore the unseen)
Cover page
Acknowledgement
Index/Table of contents
21st century life- The sordid reality
The psychic struggle, the insecurity and the vie for assurance
By-products- Stress, fear and behavioural collapse
Intoxication trap - Futile attempts for escape from the grinding reality
Return to serenity
Class-XII Holiday Assignment-2023 1
Subject : Physics
Chapter-1 (Electric Charges & Fields)
1. Why do the electrostatic field lines not form closed loop?
2. Two charges of magnitudes –20 and + O are located at points (a, 0) and (4a, 0), respectively.
What is the electric flux due to these charges through a sphere of radius 3a with its centre at
the origin?
3. Draw a plot showing the variation of electric field (E) with distance r due to a point charge Q.
4. A proton is placed in a uniform electric field directed along the position X-axis. In which
direction will it tend to move?
5. In which orientation, a dipole placed in a uniform electric field is in (i) stable (ii) unstable
equilibrium?
6. An electric dipole of length 4 cm when placed with its axis making an angle of 60° with a
uniform electric field, experiences a torque of 4√3. Calculate the potential energy of the
dipole if it has charge ± 8 nC.
7. Two point charges q and – 2 q are kept d distance apart, find the location of the point relative
to charge to q at which potential due to this system is zero.
8. A charge q is placed at the centre of a cube of side L. What is the electric flux passingthrough
each face of the cube?
9. If the radius of the Gaussian surface enclosing a charge is halved, how does the electric flux
through the Gaussian surface change?
10. Given a uniform electric held E =2 x 10 i N/C, find the flux of this held through a square of side
20 cm, whose plane is parallel to the YZ-plane. What would be the flux through the same
square if the plane makes an angle of 30° with the X-axis?
11. An electric dipole is placed in a uniform electric field E with its dipole moment p parallel to
the field. Find
(i) The work done in turning the dipole till its dipole moment points in the direction
opposite to E. the orientation of the dipole for which the torque acting on it becomes
maximum.
12. Figure shows two large metal plates and P tightly held against each other
and placed between two equal and unlike point charges perpendicular to
the line joining them.
(i)What will happen to the plates when they are released?
(ii)Draw the pattern of the electric field lines for the system.
13. Two point charges 4Q and Q are separated by 1 m in air. At what point on the line joining of
charges, is the electric field intensity zero?
14. Sketch the pattern of electric field lines due to
(i) a conducting sphere having negative charge on it.
(ii) an electric dipole.
15. Using Gauss’ law, deduce the expression for the electric field due to a uniformly charged
spherical conducting shell of radius R at a point
(i)outside the shell
(ii)inside the shell
Plot a graph showing variation of electric field as a function of r > R and r < R.{r being the
distance from the centre of the shell)
Class-XII Holiday Assignment-2023 2
CHAPTER-2 (ELECCTRIC POTENTIAL & CAPACITANCE)
16. What is the electric potential due to an electric dipole at an equatorial point?
17. What is the work done in moving a test charge q through a distance of 1 cm along the
equatorial axis of an electric dipole?
18. Why electrostatic potential is constant throughout the volume of the conductor and has the
same value as on its surface?
19. For any charge configuration, equipotential surface through a point is a normal to the electric
field. Justify.
20. The figure shows the field lines of a positive charge. Is the work
done by the field is moving a small positive charge from Q to P
positive or negative?
21. A dipole with its charge -q and +q located at the points (0, – b,0) and (0,+ 5,0) is present in a
uniform electric field E. The equipotential surfaces of this field are planes parallel to the Y Z-
plane.
(i)What is the direction of the electric field E?
(ii)How much torque would the dipole experience in this field?
22. Derive an expression for the electric potential at any point along the axial line of an electric
dipole.
23. A wire AB is carrying a steady current of 12 A and is lying on the table. Another wire CD
carrying 5 A is held directly above AB at a height of 1mm. Find the mass per unit length of the
wire CD, so that it remains suspended at its position when left free. Give the direction of the
current flowing in CD with respect to that in [Take the value of g =10 ms-2]
24. Define the dielectric constant of a medium. What is its unit?
25. A metal plate is introduced between the plates of a charged parallel plate capacitor. What is
its effect on the capacitance of the capacitor?
26. The following graph shows the variation of charge Q with voltage V for two capacitors K and L
In which capacitor is more electrostatic energy stored?
27. Net capacitance of three identical capacitors is 1μF. What will be their net capacitance when
connected in parallel? Find the ratio of their energy stored in two configurations?
28. A parallel plate capacitor is charged to a potential difference V by a DC source. The capacitor
is then disconnected from the source. If the distance between the plates is doubled, state
with reason, how the following will change?
(i)Electric field between the plates
(ii)Capacitance
(iii)Energy stored in the capacitor
Class-XII Holiday Assignment-2023 3
29. Find the ratio of the potential differences that must be applied across the parallel and the
series combination of two identical capacitors so that the energy stored in the two cases
becomes the same.
30. A parallel plate capacitor is charged by a battery. After sometime, the battery is disconnected
and a dielectric slab of dielectric constant K is inserted between the plates. How would
(i)the electric field between the plates
(ii)the energy stored in the capacitor be affected? Justify your answer
Subject: Chemistry
Solutions
1. Define the following terms.
(i) Azeotropes (ii) Van’t Hoff factor
(iii) Osmotic pressure (iv) Isotonic solutions
2. State Henry’s law.
3. State Raoult’s law for solution containing volatile liquids.
4. Give reason for the following.
(i) Aquatic species are comfortable in cold water than warm water.
(ii) Vapour pressure of a solution of glucose in water is lower than that of pure water.
(iii) Kb is less than Kf for a pure solvent.
5. Why osmotic pressure method is used for determining molar masses of macromolecules
than other colligative properties?
6. Why is osmotic pressure of 1 M KCl higher than 1 M urea solution?
7. Calculate the boiling point of solution when 2 g of Na2SO4 was dissolved in 50 g of water,
assuming Na2SO4 undergoes complete ionization.
8. (i)What is the value of Van’t of factor if Na2SO4 undergoes 80 % dissociation?
(ii)Why mixture of acetone and chloroform shows -ve deviation?
9. When 2.56 g of Sulphur was dissolved in 100 g of CS2, the freezing point lowered by 0.383
K. Calculate the formula of Sulphur.Given Kf for CS2 is 3.83 K.kg.mol-1
10. 2 g of benzoic acid (C6H5COOH) dissolved in 25 g of benzene shows a depression in
freezing point equal to 1.62 K. Kf for benzene is 4.9 K.kg.mol-1. What is the percentage of
association of acid if it forms dimer in solution?
11. Two liquids X and Y form an ideal solution. The vapour pressure of the solution containing
3 moles of X and 1 mole of Y is 550 mm of Hg. But when 4 moles of X and 1 mole of Y are
mixed, the vapour pressure of the solution becomes 560 mm of Hg. What will be the
vapour pressure of pure X and Y at this temperature?
Class-XII Holiday Assignment-2023 4
12. On dissolving 19.5 g of CH2FCOOH in 500 g of water, a depression of 1oC in freezing point
of water is observed. Calculate the van’t Hoff factor and dissociation constant of
CH2FCOOH. Given Kf for water is 1.86 K.kg.mol-1.
Electrochemistry
1. Define the following.
(i) Limiting molar conductivity (ii) Fuel cell
(iii) Primary cell (iv) Molar conductivity
2. State the following.
(i) Kohlrausch’s law, (ii) Faraday’s 1st law.
3. What is the effect of temperature on ionic conductance?
4. Give reason for the following.
(i) Mercury cell has a constant cell potential throughout its life.
(ii) Conductivity decreases with decrease in concentration.
5. Write expression to determine limiting molar conductivity of
(i) CH3COOH (ii) NH4OH (iii) H2O
6. Calculate the time to deposit 1.27 g of copper when a current of 2 A was passed through
the solution of CuSO4.
7. Write the name of the cell which is used in hearing aids. Write the reactions taking place
at the anode and the cathode of this cell.
8. Give reason for the following.
(i) Alkaline medium inhibits the rusting of iron.
(ii) Iron does not rust even if the zinc coating is broken in a galvanized iron pipe.
9. Conductivity of 0.001028 molL-1 acetic acid is 4.95 x 10-5 S cm-1.Calculate its dissociation
constant if limiting molar conductivity of acetic acid is 390.5 S cm2 mol-1.
10. A voltaic cell is set up at 250C with the following half cells.
Al/Al3+ (0.001 M) and Ni/Ni2+ (0.50 M). Calculate the cell potential.
E0Ni2+/Ni = - 0.25 V, E0Al3+/Al = - 1.66 V, log (8 x 10-6) = - 5.097.
11. The electrical resistance of a column of 0.05 molL-1 NaOH solution of diameter 1 cm and
length 50 cm is 5.55 x 103 ohm. Calculate its resistivity, conductivity, and molar
conductivity.
12. Resistance of a conductivity cell filled with 0.1 M KCl solution is 100 ohm. If the resistance
of the same cell when filled with 0.02 M KCl solution is 520 ohm, calculate the
conductivity and molar conductivity of 0.02 M KCl solution. The conductivity of 0.1 M KCl
solution is 1.29 S m-1.
Class-XII Holiday Assignment-2023 5
Subject: Biology
A – Multiple Choice Questions (MCQs)
1. _______ is a lytic enzyme released by the sperm.
(i) Hyaluronidase (ii) Trypsin (iii) Helicase (iv)None of the above
2. How many autosomes does a human primary spermatocyte have?
(i) 34 (ii) 44 (iii) 54 (iv) 33
3. Where does the ovum receive the sperm?
(i) Animal pole (ii) Vegetal pole (iii) Zona pellucid (iv)None of the above
4. ___________is an organelle that helps the sperm to penetrate the ovum
(i) Acrosome (ii) Zona pellucid (iii) Megalis (iv)None of the above
5. Umbilical cord contains _________
(i) Pluripotent stem cells (ii) Cord blood stem cells
(iii) Blood stem cells (iv) None of the above
6. How does human sperm locomote?
(i) Flagella (ii) Cilia (iii) Neutrophils (iv)None of the above
7. Cryptorchidism is a condition where _________
(i) One of both testes are not developed
(ii) One or both testes fail to descend into the scrotum
(iii) One or both testes are not formed
(iv) None of the above
8. The hormone that is released from the testes is ______
(i) Progesterone (ii) Vasopressin (iii) Testosterone (iv)None of the above
B. Very Short Answer Questions (1 mark each)
1. Failure of testes to descend into scrotal sacs leads to sterility. Why?
2. Both vaccine and colostrum produce immunity. Name type of immunity produced by
these.
3. How many sperms will be produced from 10 primary spermatocytes and how many eggs
will be produced from 10 primary oocytes?
4. The spermatogonial cell has 46 chromosomes in human male. Give the number of
chromosomes in –
(i) Primary spermatocyte (ii) Spermatid
5. In ovary which structure transforms as corpus luteum and name the hormone secreted by
corpus luteum?
6. “Each and every coitus does not results in fertilisation and pregnancy”. Justify the
statement.
Class-XII Holiday Assignment-2023 6
C. Short Answer Questions-II (2 marks each)
1. Give the function of
(i) Corpus luteum (ii) Endometrium
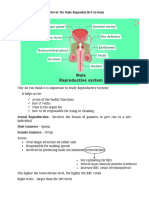
2. In the given figure, give the name and functions of parts labelled A and B.
3. Given below is an incomplete flow chart showing influence of hormone on gametogenesis
in male, observe the flow chart carefully and fill in the blank A, B, C and D.
4. Give reason for the following :
(i) The first half of the menstrual cycle is called follicular phase as well as proliferative
phase.
(ii) The second half of the menstrual cycle is called luteal phase as well as secretory
phase.
5. What is meant by L.H. Surge? Write the role of L.H.
6. Explain significance of the condition in which the testes remain suspended in scrotum
outside the abdomen.
D. Short Answer Questions-I (3 marks each)
1. Mention the name and role of hormones which are involved in regulation of gamete
formation in human male.
2. Three of the steps of neuro endocrine mechanism in respect of parturition are
mentioned below.
Class-XII Holiday Assignment-2023 7
Write the missing steps in proper sequence.
(a) Signals originate from fully developed foetus and placenta.
(b) ______________________________.
(c) ______________________________.
(d) Oxytocin causes strong uterine contraction
(e) Uterine contraction stimulates further secretion of oxytocin.
(f) ______________________________.
3. The events of the menstrual cycle are represented below. Answer the following
questions.
(i) State the levels of FSH, LH and Progesterone simply by mentioning high or low
around 13th and 14th day and 21st to 23rd day.
(ii) In which of the above mentioned phases does egg travel to fallopian tube?
(iii) Why there is no mensuration after fertilisation?
4. (a) Read the graph given below. Correlate the ovarian events that take place in the human
female according to the level of the pituitary hormone during the following day.
(i) 10th – 14th days
(ii) 14th –15th days
(iii)16th – 23th days
(iv) 25th – 29th days (If the ovum is not fertilised)
(b) What are the uterine events that follow beyond 29th day if the ovum is not
fertilised?
Class-XII Holiday Assignment-2023 8
5. T.S. of mammalian testis revealing seminiferous tubules show different types of cell.
(i) Name the two types of cells of germinal epithelium.
(ii) Name of cells scattered in connective tissue and lying between seminiferous tubules.
(iii) Differentiate between them on the basis of their functions.
E. Long Answer Questions (5 marks each)
1.
Study the figure given :
(i) Pick out and name the cells that undergo spermiogenesis.
(ii) Name ‘A’ and ‘C’ cells.
(iii) Give ploidy of ‘B’ and ‘E’
(iv) What are the cells marked as ‘F’? Mention their function.
(v) Mention the type of cell division in A and B.
Class-XII Holiday Assignment-2023 9
Subject: Mathematics
Answer the following:
Topic
SECTION- A
1 Let us define a relation R in set of real number R as aRb if a ≥ 𝑏 . Then the relation R is :
( a ) an equivalence relation ( b) reflexive, transitive but not symmetric
( c ) symmetric, transitive but not reflexive ( d ) None of these
2 Total number of possible matrices of order 3x3 with each entry 2 or 0 is
(a ) 9 ( b ) 27 ( c ) 81 ( d ) 512
33𝜋
3 −1
The value of sin ( 𝑐𝑜𝑠 ) ) is :
5
3𝜋 −7𝜋 𝜋 −𝜋
(a) 5 ( b) ( c ) 10 (d ) 10
5
4 The function f ( x ) =[ x ] where [ x ] denotes the greatest integer function of x, is
continuous at
(a)4 (b)0 ( c ) 1.5 ( d ) None of these
5 −1 7
The value of cot (cos ( 25 ) ) is …………… .
𝜋
6 The value of cot ( 4 − 2𝑐𝑜𝑡 −1 3 ) is ………….. .
7 If A and B are symmetric matrices , then AB – BA is a ……….. matrix.
8 If set A contains 5 elements and the set B contains 6 elements, then the number of one
one and onto function from A to B is ………………… .
9 If A & B are matrices of order 3 and | A | = 5, | B | = 3, then find the value of | 3AB |
SECTION- B
−1 −1
10 If 3tan 𝑥 + cot 𝑥 = 𝜋, then find the value of x.
11 𝑎 0 0
If A = 0 𝑎 0 , find the value of |adjA|.
0 0 𝑎
12 1 2 𝑥
If [ 2x 3 ] = 0, find the value of x.
−3 0 8
SECTION- C
13 x 1, if x is odd
Show that f:N N , given by f(x) is both one-one and onto.
x 1, if x is even
14 0 tan( / 2)
Let A and I be the identity matrix of order 2. Show
tan( / 2) 0
cos sin
that I A (I A)
sin cos
Class-XII Holiday Assignment-2023 10
Subject: Physical Education
PRACTICAL-1 FITNESS TEST ADMINISTRATION (SAI KHELO INDIA TEST)
i) Plate tapping test
ii) 50 meters Dash
iii) 600 meters walk/Run
iv) Sit and Reach Test
v) Partial Curl-up
vi) Push-ups for Boys and Modify Push-ups for Girls
(You must Paste the color Picture of each test)
PRACTICAL -2 ASANAS
Write about any two ASANA for each Lifestyle disease
(They must write the procedure for Asanas, Benefits, and Contraindications)
Lifestyle diseases are:-
i) Obesity ii) Diabetic iii) Asthma iv) Back pain v) hypertension.
Subject: Computer Science
1. Write a program to count the number of lines in text file TEXT.txt.
2. WAP containing a function that appends the content of one file at the end of another file.
Read both file names from the user and pass the file names to the function.
3. Write a program to create a copy of an existing text file by capitalizing first letter of every
sentence.
4. Write a program containing a user defined function that displays the number of lines
ending with 'H' in the file para.txt.
5. A text file named MESSAGE.TXT contains some text. Another text file named SMS.TXT
needs to be created such that it would store only the first 150 characters from the file
MESSAGE.TXT.
Write a user-defined function LongToShort() in Python that would perform the above task
of creating SMS.TXT from the already existing file MESSAGE.TXT.
6. A text file named CONTENTS.TXT contains some text. Write a user-defined function
LongWords() in Python which displays all such words of the file whose length is more than
9 alphabets.
For example: if the file CONTENTS.TXT contains:
"Conditional statements of Python programming language are if only"
Then the function LongWords() should display the output as : Conditional
statements
programming
Class-XII Holiday Assignment-2023 11
7. Write a method/function ISTOUPCOUNT() in python to read contents from a text file
WRITER.TXT, to count and display the occurrence of the word ‘‘IS’’ or ‘‘TO’’ or ‘‘UP’’.
For example : If the content of the file is
IT IS UP TO US TO TAKE CARE OF OUR SURROUNDING. IT IS NOT POSSIBLE ONLY FOR THE
GOVERNMENT TO TAKE RESPONSIBILITY
The method/function should display
Count of IS TO and UP is 6
8. Write a method/function AEDISP() in python to read lines from a text file WRITER.TXT,
and display those lines, which are starting either starting with A or starting with E.
For example : If the content of the file is
A CLEAN ENVIRONMENT IS NECESSARY FOR OUR GOOD HEALTH. WE SHOULD TAKE CARE
OF OUR ENVIRONMENT. EDUCATIONAL INSTITUTIONS SHOULD TAKE THE LEAD.
The method should display
A CLEAN ENVIRONMENT IS NECESSARY FOR OUR GOOD HEALTH.
EDUCATIONAL INSTITUTIONS SHOULD TAKE THE LEAD.
Subject: Economics
PROJECT WORK: - Make the hard copy of project file.
TOPIC: - As finalized after discussion in the class and consultation with the teacher.
Finalize the topic and objectives.
Complete the entire project.
Note:- reference given in the group.
Class-XII Holiday Assignment-2023 12
You might also like
- MCQ Gynecological CancerDocument6 pagesMCQ Gynecological Cancerlimaihwa1990100% (7)
- Electricity and Magnetism: Problems in Undergraduate PhysicsFrom EverandElectricity and Magnetism: Problems in Undergraduate PhysicsRating: 5 out of 5 stars5/5 (1)
- Amol Potential Class 12Document9 pagesAmol Potential Class 12kaizenpro01No ratings yet
- Electrostatics Xii HDHW Vsa &VS 2023Document6 pagesElectrostatics Xii HDHW Vsa &VS 2023mishra.sanskar12345No ratings yet
- CBSE Class 12 Physics Electric Charges and FieldsDocument6 pagesCBSE Class 12 Physics Electric Charges and Fieldsshikha sharmaNo ratings yet
- Apeejay School, Saket First Terminal Examination - 2016: Class Xii (Physics) General InstructionsDocument4 pagesApeejay School, Saket First Terminal Examination - 2016: Class Xii (Physics) General InstructionsabhilashagoelNo ratings yet
- 12 Science HHWDocument26 pages12 Science HHWOmkar VikalNo ratings yet
- 6185-Revision Test 1 Class XII Physics Dec 16Document5 pages6185-Revision Test 1 Class XII Physics Dec 16Rehan AhmadNo ratings yet
- CBSE Class 12 Physics Electric Charges and FieldsDocument6 pagesCBSE Class 12 Physics Electric Charges and FieldsSayantan YadavNo ratings yet
- Holiday Homework For 12 Science-2022-23: EnglishDocument23 pagesHoliday Homework For 12 Science-2022-23: EnglishSamadrita MondalNo ratings yet
- Holiday HomeworkDocument26 pagesHoliday HomeworkSOUMYADEEP DENo ratings yet
- Capacitance Test-1Document4 pagesCapacitance Test-1vinayakNo ratings yet
- Electrostatics: Physics Worksheet Class: 12Document4 pagesElectrostatics: Physics Worksheet Class: 1211F10 RUCHITA MAARANNo ratings yet
- SA - ElectrostaticsDocument10 pagesSA - Electrostaticsdheeraj831No ratings yet
- Class-12 Holiday Homework (2023-24)Document17 pagesClass-12 Holiday Homework (2023-24)EmmReelsNo ratings yet
- CBSE Class 12 Physics Electric Charges and Fields PDFDocument6 pagesCBSE Class 12 Physics Electric Charges and Fields PDFRanjith M100% (1)
- ElectrostaticsDocument4 pagesElectrostaticsSharan RajNo ratings yet
- ElectrostaticsDocument1 pageElectrostaticsSayan SenguptaNo ratings yet
- Assignment ELECTROSTATICSDocument3 pagesAssignment ELECTROSTATICSabhinavNo ratings yet
- ELECTRIC CHARGES AND FIELDS Bank of Board QuestionsDocument11 pagesELECTRIC CHARGES AND FIELDS Bank of Board QuestionsNishy GeorgeNo ratings yet
- Chapter Wise TheoriticalQuestionsDocument10 pagesChapter Wise TheoriticalQuestionsUdaibir PradhanNo ratings yet
- Physics XII Chapter Wise Question BankDocument87 pagesPhysics XII Chapter Wise Question BankAdiba khanNo ratings yet
- Holidays Homework 2017 PhysicsDocument25 pagesHolidays Homework 2017 PhysicsGunveer SinghNo ratings yet
- Class 12 Revision Sheet HW PDFDocument5 pagesClass 12 Revision Sheet HW PDFAditya ChauhanNo ratings yet
- CH 2 Electric Potential and CapacitanceDocument7 pagesCH 2 Electric Potential and CapacitanceShaurya SinghNo ratings yet
- Assignment ElectrostaticsDocument2 pagesAssignment ElectrostaticsSubhanjan Mukherjee - pikuNo ratings yet
- Assignment 3 Potential & Capacitance XIIDocument4 pagesAssignment 3 Potential & Capacitance XIIRAUNAK DEYNo ratings yet
- Answer The Following QuestionsDocument5 pagesAnswer The Following Questionsneelesh sNo ratings yet
- PB 2 Physics XII 2020-21Document5 pagesPB 2 Physics XII 2020-21Atul DubeyNo ratings yet
- Electro Statics Worksheet CbseDocument19 pagesElectro Statics Worksheet CbseAmanNo ratings yet
- Phy Board LatestDocument52 pagesPhy Board LatestChithambaramNo ratings yet
- Class 12 HHW PDFDocument45 pagesClass 12 HHW PDFHarshit SinghNo ratings yet
- Chapter 2 - Electric Potential and Capacitance Previous Year Question WorksheetDocument7 pagesChapter 2 - Electric Potential and Capacitance Previous Year Question WorksheetLILYNo ratings yet
- FLT - Iii (2016-17)Document4 pagesFLT - Iii (2016-17)ChetanNo ratings yet
- Suggestion XiiDocument2 pagesSuggestion Xiiphysicshari.bwnNo ratings yet
- Em WavesDocument9 pagesEm WavesAyush shuklaNo ratings yet
- Assignment 2022Document2 pagesAssignment 2022BirajNo ratings yet
- Worksheet ElectrostaticsDocument6 pagesWorksheet ElectrostaticsDEEPA PAPNAINo ratings yet
- Summer Holiday Homework Class Xii Subject: Physics: Chapter - 1: Electric Charges and FieldsDocument19 pagesSummer Holiday Homework Class Xii Subject: Physics: Chapter - 1: Electric Charges and FieldsRishiNo ratings yet
- Assignment ElectrostaticsDocument3 pagesAssignment ElectrostaticsDr. Pradeep Kumar SharmaNo ratings yet
- Sample Paper Sample Paper Sample Paper Sample Paper - 2014 2014 2014 2014Document3 pagesSample Paper Sample Paper Sample Paper Sample Paper - 2014 2014 2014 2014manishaNo ratings yet
- Class 12 Physics Chapter 1Document11 pagesClass 12 Physics Chapter 1Garry BroNo ratings yet
- Vidhyasagar International Public School: I. Answer All The FollowingDocument2 pagesVidhyasagar International Public School: I. Answer All The FollowingPhysicsNo ratings yet
- Electric Charges and FieldsDocument15 pagesElectric Charges and Fieldsmili groupNo ratings yet
- Electrostatic Potential and CapascitanceDocument9 pagesElectrostatic Potential and CapascitancemapuclouddigitalworldNo ratings yet
- CBSE Board Class XII Physics - Set 1 Board Paper - 2011 Time: 3 Hours (Total Marks: 70) General Instructions: 1. 2. 3. 4Document6 pagesCBSE Board Class XII Physics - Set 1 Board Paper - 2011 Time: 3 Hours (Total Marks: 70) General Instructions: 1. 2. 3. 4Chandan GuptaNo ratings yet
- XII Q Group 1Document3 pagesXII Q Group 1Sanskriti VermaNo ratings yet
- CL 12holiday HomeworkDocument4 pagesCL 12holiday HomeworkSrijit SahaNo ratings yet
- Class 12Document10 pagesClass 12Vicky mishraNo ratings yet
- Physics 1 WEBDocument18 pagesPhysics 1 WEBSsNo ratings yet
- Kumpulan Soal Fisika 2 PDFDocument3 pagesKumpulan Soal Fisika 2 PDFwaridho IskandarNo ratings yet
- Chapter-1 &2 Ncert Practic EquestionsDocument15 pagesChapter-1 &2 Ncert Practic EquestionsRAHIM MDNo ratings yet
- ELECTROSTATICS AssignmentsDocument5 pagesELECTROSTATICS AssignmentsGlendNo ratings yet
- XII UT 2 PhysicsDocument4 pagesXII UT 2 Physicsharsh JoshiNo ratings yet
- Chapter 1 Ws G12Document2 pagesChapter 1 Ws G12Vinushree SanthoshkumarNo ratings yet
- Class: XII Subject: Physics Assignment 1 Chapter: ElectrostaticsDocument2 pagesClass: XII Subject: Physics Assignment 1 Chapter: ElectrostaticsAryan KhannaNo ratings yet
- Physics Part 1 ProblemsDocument14 pagesPhysics Part 1 Problemsdimdamfly100% (1)
- Class-12 Workbook Physics New 5 MarchDocument175 pagesClass-12 Workbook Physics New 5 Marchshijubs1977No ratings yet
- CBSE Class 12 Physics Sample Paper 2018Document3 pagesCBSE Class 12 Physics Sample Paper 2018Faculty, Shahid Matangini Hazra Government College for WomenNo ratings yet
- Class 12 - HY2 - RevisionDocument6 pagesClass 12 - HY2 - RevisionSam SolomonNo ratings yet
- Geophysical Field Theory and Method, Part B: Electromagnetic Fields IFrom EverandGeophysical Field Theory and Method, Part B: Electromagnetic Fields INo ratings yet
- GILLICK FRASER GUIDELINES 1pageDocument1 pageGILLICK FRASER GUIDELINES 1pageStacyNo ratings yet
- Kode TindakanDocument3 pagesKode Tindakanlobow100% (1)
- Chapter 6 - MCNDocument2 pagesChapter 6 - MCNPrincess Queenie OlarteNo ratings yet
- A Study of Women Perception Towards Personal Hygiene ProductsDocument53 pagesA Study of Women Perception Towards Personal Hygiene ProductsSachin ChoudharyNo ratings yet
- Multiple Choice Questions: (Aipgme 2006) (Aipgme 1996)Document11 pagesMultiple Choice Questions: (Aipgme 2006) (Aipgme 1996)Dr-Sanjay SinghaniaNo ratings yet
- Project b.1696516385336Document9 pagesProject b.169651638533610a.shyamleekaushalNo ratings yet
- C2b - Hydatidiform Mole (H-Mole Pregnancy) ContentDocument4 pagesC2b - Hydatidiform Mole (H-Mole Pregnancy) ContentGLADYS MAY GUDELOS MEJIASNo ratings yet
- Mutual MasturbationDocument31 pagesMutual MasturbationEafael Andrade veraNo ratings yet
- Assessment of InfantDocument6 pagesAssessment of InfantSAGAR ADHAONo ratings yet
- Gonasi - Human Chorionic Gonadotrophin Injection Reprinted Sept 2018Document3 pagesGonasi - Human Chorionic Gonadotrophin Injection Reprinted Sept 2018JonNo ratings yet
- Reviewer ReproDocument4 pagesReviewer Repromelody111mendozaNo ratings yet
- Latihan Soal TERMINOLOGI MEDISDocument10 pagesLatihan Soal TERMINOLOGI MEDISdelia yuliantiNo ratings yet
- Difference Between Human Sperm and Ovum (With Comparison Chart) - Bio DifferencesDocument1 pageDifference Between Human Sperm and Ovum (With Comparison Chart) - Bio DifferencesAhmediqraNo ratings yet
- WM Matahari 22 September 2020Document94 pagesWM Matahari 22 September 2020Riri TambogoNo ratings yet
- Vulvitis in Childhood: Information For Parents and CarersDocument8 pagesVulvitis in Childhood: Information For Parents and CarersSammar ShahbazNo ratings yet
- Mod 7 Report FINALDocument16 pagesMod 7 Report FINALZia Ammarah SaripNo ratings yet
- Research Proposal On Evolution of Surrogacy LawsDocument11 pagesResearch Proposal On Evolution of Surrogacy LawsAbhay GuptaNo ratings yet
- Endometrial Hyperplasia Debate 2020 FINAL PDFDocument24 pagesEndometrial Hyperplasia Debate 2020 FINAL PDFPanitia Symposium UroginekologiNo ratings yet
- Skripsi Diana Novitadewi B - P07124214006Document129 pagesSkripsi Diana Novitadewi B - P07124214006Arsya OfficialNo ratings yet
- Science PTDocument9 pagesScience PTorpillaleah27No ratings yet
- Ectopic PregnancyDocument54 pagesEctopic Pregnancypatriciaatan1497No ratings yet
- 1a ReproductiveAndSexualHealthDocument2 pages1a ReproductiveAndSexualHealthKenjie Palaganas CentenoNo ratings yet
- Toygasms Preview PDFDocument25 pagesToygasms Preview PDFRush VeltranNo ratings yet
- (International Handbooks of Population - 11) John F. May, Jack A. Goldstone - International Handbook of Population Policies-Springer (2022)Document862 pages(International Handbooks of Population - 11) John F. May, Jack A. Goldstone - International Handbook of Population Policies-Springer (2022)Kalsoom100% (1)
- From Egg and Sperm To NewbornDocument63 pagesFrom Egg and Sperm To NewbornCristine Joy GuinoNo ratings yet
- Definition of TermsDocument6 pagesDefinition of TermsCindy AbcideNo ratings yet
- A Collection of Post-Objectivst Essays.Document221 pagesA Collection of Post-Objectivst Essays.Leo_The_Magnificent50% (2)
- Obstetrics NotesDocument114 pagesObstetrics NotesKiara Luna100% (2)
- List of FP Service Providers in PVT - Sector-2016-17Document4 pagesList of FP Service Providers in PVT - Sector-2016-17AartiNo ratings yet