Professional Documents
Culture Documents
Poster Miona, Za Gruziju
Uploaded by
sladjad83Original Title
Copyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
Poster Miona, Za Gruziju
Uploaded by
sladjad83Copyright:
Available Formats
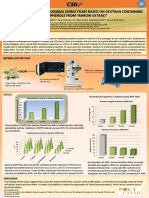
DEXTRANSUCRASE ENTRAPMENT AS AN EFFICIENT ALTERNATIVE FOR INCREASED RECYCLING
EFFICIENCY OF FREE ENZYME WITHIN AGAR-AGAR FILM MATRIX
Miona Miljkovića, Slađana Davidovića, Neda Radovanovićb, Milan Gordićc, Milica Carevića, Aleksandra Nešićc,d, Suzana Dimitrijevića
aUniversity
of Belgrade, Faculty of Technology and Metallurgy, Serbia
bUniversity of Belgrade, Innovation Center of Faculty of Technology and Metallurgy, Serbia
cUniversity of Belgrade, Vinča Institute of Nuclear Sciences, Serbia
dUniversity of Concepción, Technological Development Unit, Coronel, Correo 3, Concepción, Chile
Dextransucrase (DS), the extracellular enzyme is of immense industrial importance, due to ability to produce dextran and oligosaccharides (OS). Worldwide
interest in OS has been increasing, since they have been accorded the prebiotic status. However, the industrial application of DS for OS synthesis is limited, due
to low yield of enzyme production and its low catalytic activity. Hence, there is a great interest for development of new technologies that can provide
improved performance of biocatalyst. Enzyme immobilization technology is considered to be a crucial step for cheaper and more efficient usage of DS.
Entrapment is one of the widely investigated immobilization methods, where enzymes are enclosed or confined within the polymer matrix without altering
their native structure, developing bioreactors for commercial applications. Different matrices such as polyacryl-amide gel, alginate beads and agar–agar have
been used for the entrapment of different enzymes and among them agar–agar is a biocompatible, non-toxic and strong solidifying agent for immobilization of
various enzymes.
Composition • Mechanical properties:
2, 2.5 and 4 wt tensile strength, elongation
Sample Agar-agar DS
% agar – agar (ml) (ml) at break and elastic
glycerol modulus
DS Control 7 (2%*) 0
9:1 6.3 (2%*) 0.7
• Enzymatic activity and
After cooling
After drying reusability
to 40-45oC 4:1 5.6 (2.5%*) 1.4
1:1 3.5 (4%*) 3.5
• HPLC analysis
T = 100 °C *percent of agar-agar mixed with DS
• SEM analysis
OS yield production by immobilized (1:9) and free
Mechanical properties of agar-agar and agar-agar with entrapped DS dextransucrase after 24h is presented in chromatogram
films are presented in Table 1: (Figure 1.):
Thickness, mm Speed, mm/min σ, MPa E, MPa Elongation at break, %
control 0.050 5 23.745 343.423 15.327
1:9 0.045 5 26.637 553.935 12.56
1:4 0.045 5 19.996 439.624 10.35
1:1 0.045 5 † † †
† samples were not uniform so that measurements are not accurate
The lowest tested fraction of enzyme immobilized into polymer matrix
(1:9) improved tensile strength of films in comparison with control film.
Micrographs results at
magnification scale
(1000×) showed
significant difference
between the
morphology of polymer
before and after enzyme
entrapment.
Figure 2. Surface morphological analysis of agar-agar film before (a) and
after (b) dextransucrase entrapment at magnifications (10 0 0×).
Entrapped dextransucrase
showed 60% residual activity
after six cycles. Capability of
entrapped enzyme to catalyze
six reactions also suggests it
potential to be used in various
industrial bioprocesses for
continuous product synthesis. Figure 4.Effect of different enzyme : agar-agar ratio on the
immobilization of dextransucrase from Lc. mesenteroides T3
Figure 3. Reusability of entrapped dextransucrase
Considering the economic feasibility, the entrapped DS indicated imperative recycling efficiency up to six reaction cycles. The results of this study revealed that
an easily available and inexpensive matrix, such as agar-agar, could be successfully employed for DS immobilization and OS production.
Acknowledgements
The authors gratefully acknowledge the financial support of Ministry of Science, Education and Technological Development of the Republic of Serbia
under the project TR 31035.
*mmiljkovic@tmf.ac.bg.rs
You might also like
- Product Technical Sheet - Saflex Structural 092618 0Document5 pagesProduct Technical Sheet - Saflex Structural 092618 0J RogerNo ratings yet
- Mechanical Properties, Accuracy, and Cytotoxicity of UV-polymerized 3D Printing Resins Composed of BisEMA, UDMA, and TEGDMA PDFDocument6 pagesMechanical Properties, Accuracy, and Cytotoxicity of UV-polymerized 3D Printing Resins Composed of BisEMA, UDMA, and TEGDMA PDF林元敏No ratings yet
- Thinking Kids’™ Math Analogies, Grade 5From EverandThinking Kids’™ Math Analogies, Grade 5Rating: 5 out of 5 stars5/5 (1)
- Collaboration Processes: Looking Inside the Black BoxDocument13 pagesCollaboration Processes: Looking Inside the Black Boxเข้าใจว่า น่าจะชื่อตุ้นNo ratings yet
- Encapsulated Sample Rheometer (Premier ESR)Document10 pagesEncapsulated Sample Rheometer (Premier ESR)Amie SedlockNo ratings yet
- Methodology To Predict Mechanical Properties of PA-12 Lattice StructuresDocument13 pagesMethodology To Predict Mechanical Properties of PA-12 Lattice Structuresnicoleta.popa2093No ratings yet
- IMECE2006-15221: Strain Measurement of Microsamples Using Laser InterferometryDocument5 pagesIMECE2006-15221: Strain Measurement of Microsamples Using Laser InterferometryKanagarajanNo ratings yet
- JA M672 385 FichaTecnicaDocument2 pagesJA M672 385 FichaTecnicaEverardo Bracamontes DíazNo ratings yet
- Lap Shear Paper PDFDocument7 pagesLap Shear Paper PDFmaheshNo ratings yet
- Comparison of Material Properties and Biofilm FormDocument5 pagesComparison of Material Properties and Biofilm FormadistiharahapNo ratings yet
- Discrete Element Modelling of Vibrating ScreensDocument15 pagesDiscrete Element Modelling of Vibrating ScreensJuliana AguiarNo ratings yet
- Technical Data SheetDocument2 pagesTechnical Data Sheetrenaat.vanwayenbergeNo ratings yet
- ASTM D6272 Tech NoteDocument2 pagesASTM D6272 Tech NoteJuniantoro SuryantoNo ratings yet
- Optimization of Wedm Parameters Using Taguchi Method and Fuzzy Logic Technique For Al-6351Document10 pagesOptimization of Wedm Parameters Using Taguchi Method and Fuzzy Logic Technique For Al-6351TJPRC PublicationsNo ratings yet
- 8th Gen Processor Family S Platform Datasheet Vol 1Document8 pages8th Gen Processor Family S Platform Datasheet Vol 1Namelezz ShadowwNo ratings yet
- Prusament Tough Resin Technical DatasheetDocument3 pagesPrusament Tough Resin Technical DatasheetRobert AnderssonNo ratings yet
- RMSFaultseal Allows Geologists to Analyze Fault Sealing EffectsDocument2 pagesRMSFaultseal Allows Geologists to Analyze Fault Sealing EffectsbelahmeursNo ratings yet
- PRS Geotech Data Spec Neoloy Category A v8.2 PDFDocument2 pagesPRS Geotech Data Spec Neoloy Category A v8.2 PDFbehcetayNo ratings yet
- Tensile and Morphological Properties of PMMA Composite Reinforced by Pistachio Shell Powder Used in Denture ApplicationsDocument9 pagesTensile and Morphological Properties of PMMA Composite Reinforced by Pistachio Shell Powder Used in Denture ApplicationsJawad K. OleiwiNo ratings yet
- 410W MBB Bifacial Mono PERC Module Generates Higher Output PowerDocument2 pages410W MBB Bifacial Mono PERC Module Generates Higher Output PowerAntonio CanalesNo ratings yet
- Ali Goger IngeniaDocument16 pagesAli Goger IngeniaMiguel Angel Perez EsparzaNo ratings yet
- Particle Size Analysis in Dry Powder Cell Culture Media ProductionDocument5 pagesParticle Size Analysis in Dry Powder Cell Culture Media ProductionSAFC-GlobalNo ratings yet
- Sbrs-CapsDocument1 pageSbrs-CapsVICTOR FQNo ratings yet
- Specific Provisions For Rigid Geo-CellDocument1 pageSpecific Provisions For Rigid Geo-Cellvenkateswara rao PothinaNo ratings yet
- Article 2Document1 pageArticle 2bhagat103No ratings yet
- Plastic Deformation in Tensile Testing: Renewable Energy Equipment DesignDocument17 pagesPlastic Deformation in Tensile Testing: Renewable Energy Equipment DesignNoman AhmedNo ratings yet
- LajakLaku Tablet Production Analysis For April 2023Document3 pagesLajakLaku Tablet Production Analysis For April 2023Aiman ZanNo ratings yet
- Channel Prime Alliance 5R2 Polypropylene Data SheetDocument1 pageChannel Prime Alliance 5R2 Polypropylene Data SheetdianaNo ratings yet
- PRS Geotech Data Spec Neoloy Category D v8.2Document2 pagesPRS Geotech Data Spec Neoloy Category D v8.2Thameem SharaafNo ratings yet
- Calibration of Angle of ReposeDocument2 pagesCalibration of Angle of ReposesmendozaNo ratings yet
- Raise3D Premium ABS Technical Data Sheet: Physical PropertiesDocument3 pagesRaise3D Premium ABS Technical Data Sheet: Physical PropertiesdeepNo ratings yet
- Facebaker Baking Character Facial Rigs With Machine LearningDocument2 pagesFacebaker Baking Character Facial Rigs With Machine LearningPackNo ratings yet
- Mejora Continua en Molinos SAGDocument24 pagesMejora Continua en Molinos SAGRelining MineralsNo ratings yet
- Prediction of Compressive Strength of Self-Compacting Concrete Using Intelligent Computational ModelingDocument18 pagesPrediction of Compressive Strength of Self-Compacting Concrete Using Intelligent Computational ModelingAfaq AhmedNo ratings yet
- Dense Medium SeparationDocument37 pagesDense Medium SeparationFelipe Goes Ribeiro BrideNo ratings yet
- Face Reconstruction - Generic ModelDocument4 pagesFace Reconstruction - Generic ModelBeulah ChristudasNo ratings yet
- Effect of Silica & Prepolymer Nanoparticles on Mechanical Properties of Denture Base AcrylicDocument8 pagesEffect of Silica & Prepolymer Nanoparticles on Mechanical Properties of Denture Base AcrylicindahNo ratings yet
- Realize The Potential: Apparent Hardness vs. Thickness in Over-Mold ApplicationsDocument2 pagesRealize The Potential: Apparent Hardness vs. Thickness in Over-Mold Applicationsisraelevy1No ratings yet
- Comparison GBX11 TriAxDocument1 pageComparison GBX11 TriAxjordiNo ratings yet
- University of British Columbia: Mpowell@eng - Uct.ac - ZaDocument7 pagesUniversity of British Columbia: Mpowell@eng - Uct.ac - Zaluis martinezNo ratings yet
- Scale Up of HSM GranulatorsDocument15 pagesScale Up of HSM Granulatorshicham1963No ratings yet
- Output 2Document8 pagesOutput 2Fitria MaharaniNo ratings yet
- Uk-C: Marshall Mix Design Method (Astm D 1559 or JKR Method)Document5 pagesUk-C: Marshall Mix Design Method (Astm D 1559 or JKR Method)AiniHazwaniNo ratings yet
- Enhancement of Surface Integrity Using Spark Assisted Abrasive Flow Machining ProcessDocument8 pagesEnhancement of Surface Integrity Using Spark Assisted Abrasive Flow Machining ProcessTJPRC PublicationsNo ratings yet
- 10.18400-tekderg.683568-953930Document20 pages10.18400-tekderg.683568-953930sekarchand33No ratings yet
- Radiography Image Quality Control User ManualDocument16 pagesRadiography Image Quality Control User Manualsamel abreNo ratings yet
- MONOPERC 370-390 BIFACIALJAM72D09 BP Global - EN - 20190703A PDFDocument2 pagesMONOPERC 370-390 BIFACIALJAM72D09 BP Global - EN - 20190703A PDFAmaro BorgesNo ratings yet
- PRS Geotech Data Spec Neoloy Category B V8.1aDocument2 pagesPRS Geotech Data Spec Neoloy Category B V8.1aJonas MachadoNo ratings yet
- Our Visit Today: Multiple Stress Creep Recovery (MSCR) Task ForceDocument5 pagesOur Visit Today: Multiple Stress Creep Recovery (MSCR) Task ForceAndresNo ratings yet
- 320 MsDocument8 pages320 MsJian ZhangNo ratings yet
- Characterization and Indentificatio of Plastic PDFDocument24 pagesCharacterization and Indentificatio of Plastic PDFWatthana KlairutsameeNo ratings yet
- MeCaTeC MeCaCorr 700 Surface Polymer Ceramic CoatingDocument1 pageMeCaTeC MeCaCorr 700 Surface Polymer Ceramic CoatingjaviermvsNo ratings yet
- Construction and Building Materials: Lyes Bal, François Buyle-BodinDocument7 pagesConstruction and Building Materials: Lyes Bal, François Buyle-Bodinit does not exist for the [mpossibleNo ratings yet
- MeCaTeC MeCaCorr 700Document1 pageMeCaTeC MeCaCorr 700Camilo Andrés Vargas JiménezNo ratings yet
- Particle Design via Spray Drying TechniquesDocument27 pagesParticle Design via Spray Drying Techniquesfatemeh afariNo ratings yet
- Optimization of The Process Parameter in Drilling of GFRP Using Hss DrillDocument6 pagesOptimization of The Process Parameter in Drilling of GFRP Using Hss DrillTJPRC PublicationsNo ratings yet
- Study of MEMS Pressure Sensor For TPMS UDocument3 pagesStudy of MEMS Pressure Sensor For TPMS UBasescu Matei-AlexandruNo ratings yet
- A Robust Method For Ship and Wake DetectionDocument56 pagesA Robust Method For Ship and Wake DetectionSaravanaNo ratings yet
- Modelling Dense Media SeparationDocument18 pagesModelling Dense Media SeparationbaberoniNo ratings yet
- Zhermack 2019 Catalogue - Dental Lab Solutions for Stone Models, Masks, Duplication and MoreDocument111 pagesZhermack 2019 Catalogue - Dental Lab Solutions for Stone Models, Masks, Duplication and MoreZurazis LabNo ratings yet
- High-Performance Gradient Elution: The Practical Application of the Linear-Solvent-Strength ModelFrom EverandHigh-Performance Gradient Elution: The Practical Application of the Linear-Solvent-Strength ModelNo ratings yet
- Facile Synthesis of Silver Chloride Nanocubes and Their DerivativesDocument5 pagesFacile Synthesis of Silver Chloride Nanocubes and Their Derivativessladjad83No ratings yet
- Poster Sa Konferencije U GruzijiDocument1 pagePoster Sa Konferencije U Gruzijisladjad83No ratings yet
- KefirBeer PDFDocument6 pagesKefirBeer PDFAugusto TorresNo ratings yet
- Postrer Za PTEPDocument1 pagePostrer Za PTEPsladjad83No ratings yet
- Animal NutritionDocument6 pagesAnimal Nutritionsladjad83No ratings yet
- Animal Feed Science and TechnologyDocument10 pagesAnimal Feed Science and Technologysladjad83No ratings yet
- From Waste To New Biobased Edible Coatings: An Ecological Approach To Improve The Safety and Shelf-Life of FoodsDocument1 pageFrom Waste To New Biobased Edible Coatings: An Ecological Approach To Improve The Safety and Shelf-Life of Foodssladjad83No ratings yet
- Kvantitativno Određivanje Beta GlukanaDocument8 pagesKvantitativno Određivanje Beta Glukanasladjad83No ratings yet
- Esej 1Document1 pageEsej 1sladjad83No ratings yet
- Mushroom Polysaccharides ReviewDocument40 pagesMushroom Polysaccharides Reviewsladjad83No ratings yet
- Zrno Vodenog Kefira Kao Izvor Bakterija Mlečno Kiselinskog Vrenja Potentnih Producenata DekstranaDocument10 pagesZrno Vodenog Kefira Kao Izvor Bakterija Mlečno Kiselinskog Vrenja Potentnih Producenata Dekstranasladjad83No ratings yet
- Homework 2Document1 pageHomework 2sladjad83No ratings yet
- Determination of Molecular Weights of Dextrans by Means of Alkaline Copper ReagentsDocument6 pagesDetermination of Molecular Weights of Dextrans by Means of Alkaline Copper Reagentssladjad83No ratings yet
- Response Surface Methodology For Optimisation of Edible Coatings Based On Dextran From Leuconostoc Mesenteroides T3Document7 pagesResponse Surface Methodology For Optimisation of Edible Coatings Based On Dextran From Leuconostoc Mesenteroides T3sladjad83No ratings yet
- AMERSHAM Hydrophobic Interaction ManualDocument104 pagesAMERSHAM Hydrophobic Interaction ManualAnil ReddyNo ratings yet
- Mechanical Properties and Water Vapor TransmissionDocument7 pagesMechanical Properties and Water Vapor Transmissionsladjad83No ratings yet
- Phenol Sulphuric Acid Carbohydrate AssayDocument1 pagePhenol Sulphuric Acid Carbohydrate Assaysladjad83100% (1)
- Phenol Sulphuric Acid Carbohydrate AssayDocument1 pagePhenol Sulphuric Acid Carbohydrate Assaysladjad83100% (1)
- Wuthering Heights Chapter Summary and AnalysisDocument215 pagesWuthering Heights Chapter Summary and Analysissladjad83No ratings yet
- Development and Characterisation of A New Biodegradable Edible Film Made From KefiranDocument7 pagesDevelopment and Characterisation of A New Biodegradable Edible Film Made From Kefiransladjad83No ratings yet
- Cromatografia Exclasion PharmaciaDocument124 pagesCromatografia Exclasion PharmaciaNaybethGuzmánNo ratings yet
- Stabilization of Dextransucrase From Leuconostoc MesenteroidesDocument5 pagesStabilization of Dextransucrase From Leuconostoc Mesenteroidessladjad83No ratings yet
- Antimicrobial and Healing Activity of Kefir and Kefiran ExtractDocument5 pagesAntimicrobial and Healing Activity of Kefir and Kefiran Extractsladjad83No ratings yet
- Recombi Prot HandbookDocument112 pagesRecombi Prot Handbookpacman6666No ratings yet
- Creme Brulee - Printer Friendly PDFDocument1 pageCreme Brulee - Printer Friendly PDFmtlpcguysNo ratings yet
- QRP Yu1lm SDR-RX TXDocument299 pagesQRP Yu1lm SDR-RX TXD BNo ratings yet
- CD 0400 CH 4 X 100 ML: For in Vitro Diagnostic Use Only. LinearityDocument1 pageCD 0400 CH 4 X 100 ML: For in Vitro Diagnostic Use Only. LinearityNguyễn ThơiNo ratings yet
- FT Aeroterme GEADocument15 pagesFT Aeroterme GEACrisTimNo ratings yet
- Wda 201Document72 pagesWda 201MoussaNo ratings yet
- Recycle ProgramDocument2 pagesRecycle ProgramKaps BlazeNo ratings yet
- RCS England Trainees Guide To A Quality Improvement Project 2021Document17 pagesRCS England Trainees Guide To A Quality Improvement Project 2021Wee K WeiNo ratings yet
- Understanding of AVO and Its Use in InterpretationDocument35 pagesUnderstanding of AVO and Its Use in Interpretationbrian_schulte_esp803100% (1)
- BrosurDocument68 pagesBrosurKiki Xhui7No ratings yet
- Memorial Garden Policies and Guidelines St. Mark's Episcopal Church, Raleigh, North CarolinaDocument7 pagesMemorial Garden Policies and Guidelines St. Mark's Episcopal Church, Raleigh, North Carolinaestela maria dulaganNo ratings yet
- Generador APD550PEDocument4 pagesGenerador APD550PEFTL1990No ratings yet
- Walet - Further Mathematical Methods PDFDocument79 pagesWalet - Further Mathematical Methods PDFPeter BraamsNo ratings yet
- Encore 700 Diaphragm Pump 1Document12 pagesEncore 700 Diaphragm Pump 1bagurtoNo ratings yet
- Shock Classification and PathophysiologyDocument40 pagesShock Classification and PathophysiologyErick Anca100% (2)
- The Picture of Dorian GrayDocument137 pagesThe Picture of Dorian GrayАнгелаС.No ratings yet
- Kyle 106 - 119 - 131 - 141Document1 pageKyle 106 - 119 - 131 - 141Gerson SouzaNo ratings yet
- Unit 4 Early HumansDocument8 pagesUnit 4 Early HumansSepfira ReztikaNo ratings yet
- Gem EscortingDocument7 pagesGem Escortingsuman sutharNo ratings yet
- Conduction Calorimetric Investigation of The Effect of Retarders On The Hydration of Portland CementDocument15 pagesConduction Calorimetric Investigation of The Effect of Retarders On The Hydration of Portland CementAlfredo Landaverde GarcíaNo ratings yet
- 30-300 MHZ Transmission Line Trainer Tla05Document1 page30-300 MHZ Transmission Line Trainer Tla05Kishor HandoreNo ratings yet
- Mitsubishi Motors: Service ManualDocument29 pagesMitsubishi Motors: Service ManualCristobalNo ratings yet
- Postmodern EthicsDocument1 pagePostmodern Ethicsgeorge elerickNo ratings yet
- Osha Top 25 Physical HazardsDocument100 pagesOsha Top 25 Physical HazardsHasan Shakeel100% (2)
- Atlas MothDocument1 pageAtlas Mothlililala92No ratings yet
- Saej401v002 PDFDocument6 pagesSaej401v002 PDFLuis LujanoNo ratings yet
- GROHE Specification Sheet 19443000-1Document2 pagesGROHE Specification Sheet 19443000-1Fred PrzNo ratings yet
- Wood Plastic and CompositesDocument5 pagesWood Plastic and CompositesBenjie LatrizNo ratings yet
- VSD Power Supply Connections and EarthingDocument4 pagesVSD Power Supply Connections and EarthingHumaid ShaikhNo ratings yet
- Encapsulation and Inheritance in Object-Orlented Programming LanguagesDocument8 pagesEncapsulation and Inheritance in Object-Orlented Programming Languageszsolt kormanyNo ratings yet