Professional Documents
Culture Documents
Bnbi104 Tutorial1
Uploaded by
Ashria Sonali Prakash0 ratings0% found this document useful (0 votes)
4 views1 pageThis document provides a tutorial sheet for an organic chemistry class, with exercises on drawing line structures, writing condensed structural formulas and IUPAC names, identifying constitutional isomers, and drawing and naming isomers with the molecular formula C7H16. The tutorial covers topics including writing names for alkanes, identifying incorrect IUPAC names, and drawing line structures for various hydrocarbon structures.
Original Description:
Original Title
BNBI104 TUTORIAL1
Copyright
© © All Rights Reserved
Available Formats
PDF, TXT or read online from Scribd
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentThis document provides a tutorial sheet for an organic chemistry class, with exercises on drawing line structures, writing condensed structural formulas and IUPAC names, identifying constitutional isomers, and drawing and naming isomers with the molecular formula C7H16. The tutorial covers topics including writing names for alkanes, identifying incorrect IUPAC names, and drawing line structures for various hydrocarbon structures.
Copyright:
© All Rights Reserved
Available Formats
Download as PDF, TXT or read online from Scribd
0 ratings0% found this document useful (0 votes)
4 views1 pageBnbi104 Tutorial1
Uploaded by
Ashria Sonali PrakashThis document provides a tutorial sheet for an organic chemistry class, with exercises on drawing line structures, writing condensed structural formulas and IUPAC names, identifying constitutional isomers, and drawing and naming isomers with the molecular formula C7H16. The tutorial covers topics including writing names for alkanes, identifying incorrect IUPAC names, and drawing line structures for various hydrocarbon structures.
Copyright:
© All Rights Reserved
Available Formats
Download as PDF, TXT or read online from Scribd
You are on page 1of 1
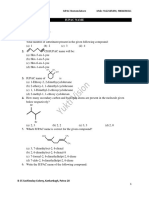
BNBI104: Organic Chemistry; Tutorial Sheet – 1
1. Write a line structure for each of these condensed 4. Write IUPAC names for each of the following
structural formulae. compounds
5. 15.12 Write line structures for these alkanes.
2. Write a condensed structural formula and the
(a) 2,2,4-trimethylhexane
molecular formula for each of the following alkanes.
(h) 2.2-dimethylpropane
(c) 3-ethyl-2,4,5-trimethyloctane
(d) 5-butyl-2.2-dimethylnonane
(e) 4-isopropyloctane
(f) 3,3-dimethylpentane
(g) trans- 1,3-dirnethylcyclopentane
(h) cis-1,2-diethylcyclobutanc
6. Explain why each of the following names is
incorrect IUPAC name, and write the correct IUPAC
3. For each of the following, state whether or not the
name for the intended
two structural formulae shown represent
(a) I ,3-dimethylbutane
constitutional isomers.
(b) 4-methylpentane
(c) 2,2-diethylbutane
(d) 2-ethyl-3-methylpcntane
(e) 2-propylpentane
(f) 2,2-diethylheptane
(g) 2,2-dimethylcyclopropane
(h) 1 -ethyl-5-methy Icyclohexane
7. Draw line structures for the nine constitutional
isomers with the molecular formula C7H16. Also
write their IUPAC names.
You might also like
- 13 DPP 2 New Batch A-N +ans For StudentsDocument32 pages13 DPP 2 New Batch A-N +ans For StudentskljNo ratings yet
- 11DPP02AIUPACOFALKANEDocument4 pages11DPP02AIUPACOFALKANERIP- PIRNo ratings yet
- 11 DPP 02a Iupac of Alkane Emerge 1657625376769Document4 pages11 DPP 02a Iupac of Alkane Emerge 1657625376769Nothing 2No ratings yet
- Goc PDFDocument56 pagesGoc PDFManik SinghalNo ratings yet
- Arjuna JEE 2.0 (2024) : IUPAC NomenclatureDocument3 pagesArjuna JEE 2.0 (2024) : IUPAC NomenclatureDEV SHARMANo ratings yet
- Classfication and Nomeclature of Organic ComoundsDocument4 pagesClassfication and Nomeclature of Organic ComoundsEunbyeolNo ratings yet
- 655dae70717b6700190a1295 - ## - IUPAC Nomenclature DPP-02 (Of Lecture 03)Document3 pages655dae70717b6700190a1295 - ## - IUPAC Nomenclature DPP-02 (Of Lecture 03)anubhab256224No ratings yet
- 12 U Orgo - 1 - Hydrocarbon Nomenclature WorksheetDocument4 pages12 U Orgo - 1 - Hydrocarbon Nomenclature WorksheetValerie Duran-ArzagaNo ratings yet
- 12 U Orgo - 1 - Hydrocarbon Nomenclature WorksheetDocument4 pages12 U Orgo - 1 - Hydrocarbon Nomenclature WorksheetRehnuma BasherNo ratings yet
- Nomenclature - DPP 02 (Of Lec 04) - (Arjuna JEE 2023)Document2 pagesNomenclature - DPP 02 (Of Lec 04) - (Arjuna JEE 2023)jeemainsmaterial97No ratings yet
- 655eed7353c28900188cd636 - ## - IUPAC Nomenclature DPP-03 (Of Lecture 04)Document3 pages655eed7353c28900188cd636 - ## - IUPAC Nomenclature DPP-03 (Of Lecture 04)anubhab256224No ratings yet
- Nomenclature 1Document3 pagesNomenclature 1vmcuber1100% (1)
- Tutorial Problems, 2013/2014: Che 153 Organic Chemistry For EngineersDocument21 pagesTutorial Problems, 2013/2014: Che 153 Organic Chemistry For EngineersIng Rashid BawahNo ratings yet
- I UpacDocument19 pagesI UpacJay DodiyaNo ratings yet
- Organic Chemistry QuizDocument7 pagesOrganic Chemistry QuizCarlo Jay BasulNo ratings yet
- Answer: CH H: IUPAC Name: C-Atom Classification: 1Document3 pagesAnswer: CH H: IUPAC Name: C-Atom Classification: 1Shaira Dawn PlancoNo ratings yet
- Module 5 OrgchemDocument9 pagesModule 5 OrgchemJHUNNTY LOZANONo ratings yet
- Alkene Structure and PreparationDocument2 pagesAlkene Structure and PreparationCarlo Jay BasulNo ratings yet
- Nomenclature - DPP 03 - Arjuna NEET 2024Document3 pagesNomenclature - DPP 03 - Arjuna NEET 2024mdjakaullah847226No ratings yet
- Review NotesDocument7 pagesReview NotesKatie MoreeNo ratings yet
- Chem 141 Problem-Set 6 Friday 27th September 2013Document3 pagesChem 141 Problem-Set 6 Friday 27th September 2013Riley BenoitNo ratings yet
- 2023 - 03 - 25 - Oc Mka 10 Hours Maha One ShotDocument197 pages2023 - 03 - 25 - Oc Mka 10 Hours Maha One ShotgvygtvcNo ratings yet
- Esr Nomenclature Test OasisDocument4 pagesEsr Nomenclature Test OasisAnamika Barnwal100% (1)
- Basic Organic Chemistry+Anions Cations+Moles: Prof. Rakesh Rathi'S Chemistry Tutorials 15-08-21Document4 pagesBasic Organic Chemistry+Anions Cations+Moles: Prof. Rakesh Rathi'S Chemistry Tutorials 15-08-21Prakash KapadiaNo ratings yet
- IUPAC NomanclatureDocument21 pagesIUPAC NomanclatureDARK KILLERNo ratings yet
- Exercise - Nomenclature of Organic Compounds FinalDocument10 pagesExercise - Nomenclature of Organic Compounds FinalVina YuNo ratings yet
- Nomenclature DiagramsDocument6 pagesNomenclature DiagramsFran ZeneNo ratings yet
- Adobe Scan 13-Dec-2023Document6 pagesAdobe Scan 13-Dec-2023sourabh singhNo ratings yet
- NomenclatureDocument3 pagesNomenclaturecrystalgal9462No ratings yet
- Hydrocarbon 1.2 SolutionsDocument2 pagesHydrocarbon 1.2 SolutionsbaldobenrayNo ratings yet
- Post TestDocument3 pagesPost TestJaydione MakiawayNo ratings yet
- Ola ChemDocument10 pagesOla ChemChan ChanNo ratings yet
- Chem Test 1Document5 pagesChem Test 1dishaali110No ratings yet
- Xi STD (Class Test) Organic Chemistry + Moles + IupacDocument4 pagesXi STD (Class Test) Organic Chemistry + Moles + IupacPrakash KapadiaNo ratings yet
- He Following Are The List of Aliphatic Compounds, According To The Number of Carbon Atoms They Contain. Complete The Table BelowDocument17 pagesHe Following Are The List of Aliphatic Compounds, According To The Number of Carbon Atoms They Contain. Complete The Table BelowJohn Rafael DiazNo ratings yet
- Organic Chemistry QuizDocument7 pagesOrganic Chemistry QuizCarlo Jay BasulNo ratings yet
- Oc01 Alkanes Worksheet AnswersDocument3 pagesOc01 Alkanes Worksheet Answerswade aryanNo ratings yet
- NomenDocument4 pagesNomensushil kumarNo ratings yet
- Tutorial 5Document4 pagesTutorial 5Eqieyn JerrNo ratings yet
- DPPONIUPACSUPERSIXER4Document5 pagesDPPONIUPACSUPERSIXER4Kartik YadavNo ratings yet
- Iupac WorksheetDocument8 pagesIupac WorksheetSujan BansodNo ratings yet
- DPPS - 1 GocDocument4 pagesDPPS - 1 GocRajeev GangwarNo ratings yet
- WS SL NamingPracticeDocument1 pageWS SL NamingPracticeGüşta İrem SakızNo ratings yet
- Document 1Document9 pagesDocument 1Nishi tomarNo ratings yet
- Chapter 4 QuestionsDocument2 pagesChapter 4 Questionsdaniday1977100% (1)
- Worksheet 8. Aromatic HCDocument2 pagesWorksheet 8. Aromatic HCHafza MacabatoNo ratings yet
- Nomenclature - Prob SetDocument7 pagesNomenclature - Prob SetGenny MarantanNo ratings yet
- Nomenclature DPPDocument4 pagesNomenclature DPPRitesh BogatiNo ratings yet
- JR & ER - Eng. PC PDFDocument33 pagesJR & ER - Eng. PC PDFmarmaduke32No ratings yet
- Ejercicios de AlacanosDocument16 pagesEjercicios de AlacanosRicardo Alcántara ReyesNo ratings yet
- Css Chemistry2 2023Document3 pagesCss Chemistry2 2023Shoukat hussain PirzadaNo ratings yet
- LdaDocument23 pagesLdaelveNo ratings yet
- IupacDocument8 pagesIupacAmit YadavNo ratings yet
- General Organic Chemistry DPP 02 DPP 02 General Organic ChemistryDocument2 pagesGeneral Organic Chemistry DPP 02 DPP 02 General Organic ChemistryAwnikant AjayNo ratings yet
- Work Sheet IsomersDocument14 pagesWork Sheet IsomerslynlwsNo ratings yet
- IUPAC - Practice Sheet - IUPAC Nomenclature - ManzilDocument9 pagesIUPAC - Practice Sheet - IUPAC Nomenclature - ManzilShio100% (1)
- Work Sheet Alkanes Sbi 09Document6 pagesWork Sheet Alkanes Sbi 09neneng rohayatiNo ratings yet
- Nomenclature Test 1Document7 pagesNomenclature Test 1Soham SagaonkarNo ratings yet
- Differentiation of Chiral Compounds Using NMR SpectroscopyFrom EverandDifferentiation of Chiral Compounds Using NMR SpectroscopyNo ratings yet
- CH Test 4 PDFDocument151 pagesCH Test 4 PDFcvo123100% (1)
- D2374 PDFDocument3 pagesD2374 PDFChandraditya Iman FirmanshaNo ratings yet
- Ngo Et Al. - 2018 - Prediction of Degree of Impregnation in Thermoplastic Unidirectional Carbon Fiber Prepreg by Multi-Scale ComputationDocument12 pagesNgo Et Al. - 2018 - Prediction of Degree of Impregnation in Thermoplastic Unidirectional Carbon Fiber Prepreg by Multi-Scale ComputationNgô Ích SơnNo ratings yet
- Separation of Pollutants From RestaurantDocument12 pagesSeparation of Pollutants From RestaurantFahmi IzzuddinNo ratings yet
- Dailo - BSTRM1-1D (#3)Document2 pagesDailo - BSTRM1-1D (#3)Laica Mae R DailoNo ratings yet
- Imds Recommendation 001 Annex IDocument33 pagesImds Recommendation 001 Annex IAnjali DeshpandeNo ratings yet
- TOTAL PROTEIN Biuret Method: BiolaboDocument2 pagesTOTAL PROTEIN Biuret Method: BiolaboVenura VishwajithNo ratings yet
- EnvChmAnswers PDFDocument109 pagesEnvChmAnswers PDFTsamara Luthfia50% (6)
- 3m High Modulus Low Conductivity Polyurethane Adhesive Sealant 58015 50681 Technical Datasheet en Eu - 0820Document5 pages3m High Modulus Low Conductivity Polyurethane Adhesive Sealant 58015 50681 Technical Datasheet en Eu - 0820victorNo ratings yet
- Applied - Physics - Notes 1Document248 pagesApplied - Physics - Notes 1Ashutosh SinghNo ratings yet
- Mitsubishi Safeline DatasheetDocument34 pagesMitsubishi Safeline Datasheetgodama42No ratings yet
- Moisture Management FinishDocument4 pagesMoisture Management FinishDr. Sanket ValiaNo ratings yet
- Natural DyesDocument23 pagesNatural DyesSenelisile MoyoNo ratings yet
- Evaluating Structural Buildup at Rest of Self-Consolidating Concrete Using Workability TestsDocument10 pagesEvaluating Structural Buildup at Rest of Self-Consolidating Concrete Using Workability TestsHuaman Chavez Jesus AngelNo ratings yet
- FM 306: Size Reduction and Sieving: ObjectiveDocument6 pagesFM 306: Size Reduction and Sieving: ObjectiveErcille Mae Oraiz PacamoNo ratings yet
- Chloride Attack of Reinforced ConcreteDocument8 pagesChloride Attack of Reinforced ConcreteBraydon GoyetteNo ratings yet
- Water Flooding MCQDocument42 pagesWater Flooding MCQpriya dharshiniNo ratings yet
- Archimedes: 287 B.C. - 212 B.CDocument32 pagesArchimedes: 287 B.C. - 212 B.CVladislav CoteleaNo ratings yet
- Carbohydrates NotesDocument9 pagesCarbohydrates NotesAshley Saron100% (1)
- Class 10 Test Chapter-Acids, Bases, and SaltsDocument1 pageClass 10 Test Chapter-Acids, Bases, and Saltsharsheen kaurNo ratings yet
- Formulation and Evaluation of Solid Self Micro Emulsifying Drug Delivery System Using Aerosil 200 As Solid CarrierDocument6 pagesFormulation and Evaluation of Solid Self Micro Emulsifying Drug Delivery System Using Aerosil 200 As Solid CarriermorganvilleNo ratings yet
- Transformer DesignDocument33 pagesTransformer DesignHenock TadesseNo ratings yet
- Chm1 Gases 1002 FDDocument14 pagesChm1 Gases 1002 FDaerkn skjksNo ratings yet
- شرح وتدريبات ساينس للصف الرابع الابتدائي لغات - ملزمتيDocument24 pagesشرح وتدريبات ساينس للصف الرابع الابتدائي لغات - ملزمتيDina WaleedNo ratings yet
- Theory of StructureDocument15 pagesTheory of StructureClaire BarriosNo ratings yet
- Makalah Bahasa Inggris ToniDocument14 pagesMakalah Bahasa Inggris ToniLuthfi LukmannudinNo ratings yet
- Dr. A B Rajib Hazarika PHD FRAS AESDocument7 pagesDr. A B Rajib Hazarika PHD FRAS AESDrabrajib HazarikaNo ratings yet
- Antioxidant Activity of Eugenol: A Structure-Activity Relationship StudyDocument12 pagesAntioxidant Activity of Eugenol: A Structure-Activity Relationship Studyseema yadavNo ratings yet
- Animation Diagrama de Fases Kfs PL H2ODocument19 pagesAnimation Diagrama de Fases Kfs PL H2OFer PereyraNo ratings yet
- Prob Thermo chp2Document7 pagesProb Thermo chp2Muhammad FaizanNo ratings yet