Professional Documents
Culture Documents
GHANSHYAM PATEL - Graphical Abstract
Uploaded by
nitinOriginal Title
Copyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
GHANSHYAM PATEL - Graphical Abstract
Uploaded by
nitinCopyright:
Available Formats
QbD Assisted Fabrication of Optimized Mouth Dissolving Film Formulation of
Ivabradine Hydrochloride
Ghanshyam Patel, Upendra Bhadoriya, Nitin Dubey
IPS Academy College of Pharmacy, Indore (M.P.), India
ABSTRACT
Objective: The present study was aimed to design and optimize Ivabradine Hydrochloride
loaded mouth dissolving film (MDF) formulation for enhanced bioavailability and effective
oral delivery.
Material: Design of experiment (DoE) based formulation and optimization was done by
three-factor, three-level Box–Behnken design approach. The excipients comprise polymer,
plasticizer, and surfactant as an independent variables and thickness, folding endurance, and
% drug release as response variables. The suggested optimized formulation of Ivabradine HCl
loaded MDF with highest desirability was prepared by solvent casting method and
characterized for all desired critical quality attributes (CQAs) and stability studies.
Results: The optimized MDF formulation exhibited experimentally observed responses close
to the predicted values, thickness (99.2 μm), folding endurance (209 folds), and % drug
release (95.90%). The solid-state characterization studies by SEM, DSC, FT-IR, and XRD
revealed excellent film characteristics.
Conclusion: The developed Ivabradine HCl loaded MDF formulation possessed all
recommended CQAs in optimal range with enhanced bioavailability and remained physically
stable.
Keywords: Optimization, Bioavailability, Mouth dissolving film, Design of Experiments,
Ivabradine HCl.
GRAPHICAL ABSTRACT
Selection of
Variables
Independent Response
Polymer (mg) Thickness (μm)
Plasticizer (mg) Folding endurance (folds)
Surfactant (mg) In-vitro Drug releases (%)
Formulation Optimization Box–Behnken design representing
using QbD Approach independent variables at three levels
(–1, 0, +1)
Response Surface Analysis of MDF Formulations
Fabrication of MDF Formulations
SEM
Optimized MDF Formulation
DSC
FTIR
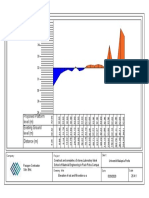
% Drug
Prediction of Optimized MDF Formulation releases
100
90
% Drug release
80
70
MQA3016
60
50
40
30
20
0
XRD
2 4 6
Time (minutes)
8 10
You might also like
- CBL Interpretation Chart PDFDocument1 pageCBL Interpretation Chart PDFReza Alfiano100% (1)
- Dasha PhalDocument105 pagesDasha Phalnitin100% (1)
- Govindarajulu Computer ArchitectureDocument4 pagesGovindarajulu Computer ArchitectureLori0% (2)
- Physics of Microwave OvenDocument8 pagesPhysics of Microwave Ovenabdel100% (1)
- Impact of Micro/Nanofibrillated Cellulose Preparation On The Reinforcement Properties of Paper and Composites FilmsDocument20 pagesImpact of Micro/Nanofibrillated Cellulose Preparation On The Reinforcement Properties of Paper and Composites FilmsGita WulandariNo ratings yet
- 07 - Hofmann - Hofalign Forged Steel Girth Gears Brochure - A4Document2 pages07 - Hofmann - Hofalign Forged Steel Girth Gears Brochure - A4Jorge VillalobosNo ratings yet
- Designed Appurtenance Loading: DLG IngenieriaDocument1 pageDesigned Appurtenance Loading: DLG IngenieriaCarlos Andres RodriguezNo ratings yet
- 2024.03.14 YOKOCHO - Sample Roof No 2Document1 page2024.03.14 YOKOCHO - Sample Roof No 2christopheryusNo ratings yet
- B.Tech IVSem IBM 2020-2024Document1 pageB.Tech IVSem IBM 2020-2024lala solankiNo ratings yet
- FABIG TM79 ChallengesinUsingModelledBlastLoadsinStructuralResponseAnalysis ASariDocument26 pagesFABIG TM79 ChallengesinUsingModelledBlastLoadsinStructuralResponseAnalysis ASariAbabNo ratings yet
- CSC2541: Differentiable Inference and Generative ModelsDocument46 pagesCSC2541: Differentiable Inference and Generative ModelslordpersonNo ratings yet
- SALD-2300: Laser Diffraction Particle Size AnalyzerDocument20 pagesSALD-2300: Laser Diffraction Particle Size AnalyzerAwais RiazNo ratings yet
- Flotation After A Direct Contact of Flotation Reagents With Carbonate Particles Part 1. Model InvestigationsDocument12 pagesFlotation After A Direct Contact of Flotation Reagents With Carbonate Particles Part 1. Model Investigationskhairiltfort14No ratings yet
- Layout Plan BroncoDocument3 pagesLayout Plan BroncoAchmad NofandiNo ratings yet
- HCD 3,5/50-II: General Lens DataDocument2 pagesHCD 3,5/50-II: General Lens DataZbigniew LipinskiNo ratings yet
- Construction and Building Materials: Ali A. Mohammed, Allan C. Manalo, Ginghis B. Maranan, Yan Zhuge, P.V. VijayDocument11 pagesConstruction and Building Materials: Ali A. Mohammed, Allan C. Manalo, Ginghis B. Maranan, Yan Zhuge, P.V. VijayAliNo ratings yet
- Noise & The City ProjectDocument10 pagesNoise & The City ProjectMike Eugene CollinsNo ratings yet
- HCD24 EN v8Document2 pagesHCD24 EN v8dirrleonardNo ratings yet
- Titan Le LimestoneDocument32 pagesTitan Le LimestonelehilarinoNo ratings yet
- Arikawa 2007Document7 pagesArikawa 2007Mariana Carolayne Almeida ResendeNo ratings yet
- Transparent Conductive Oxides: Solems Solems Solems Solems Solems Solems Solems SolemsDocument2 pagesTransparent Conductive Oxides: Solems Solems Solems Solems Solems Solems Solems SolemsErvin CrespoNo ratings yet
- ExperimentsDocument1 pageExperimentsOsamaZaidNo ratings yet
- General Lens Data: DatasheetDocument2 pagesGeneral Lens Data: DatasheetZbigniew LipinskiNo ratings yet
- La Guia MetAs 10 06 Densidad AguaDocument19 pagesLa Guia MetAs 10 06 Densidad AguaMayreneDavilaNo ratings yet
- Electrospinning Nanofibers of PANI/PMMA BlendsDocument6 pagesElectrospinning Nanofibers of PANI/PMMA BlendsDanay ManzoNo ratings yet
- D6P00301G0Document4 pagesD6P00301G0Hieu NguyenNo ratings yet
- Shri Vaishnav Vidyapeeth VishwavidyalayaDocument1 pageShri Vaishnav Vidyapeeth VishwavidyalayaRajat SaxenaNo ratings yet
- Tapak Analisis Item f4f5Document1 pageTapak Analisis Item f4f5Ticher InaNo ratings yet
- TRA Copertina SpateDocument1 pageTRA Copertina SpatePan AmNo ratings yet
- 6 Soil - CompactionDocument58 pages6 Soil - CompactionLiv CBNo ratings yet
- Lingaraj College (Autonomous) Belgaum: Karnataka - IndiaDocument3 pagesLingaraj College (Autonomous) Belgaum: Karnataka - IndiaSnehal KocheriNo ratings yet
- Dynamic Traffic Assignment and ApplicationsDocument38 pagesDynamic Traffic Assignment and ApplicationsaldojudinNo ratings yet
- Development of A Computational Model For Optimal SDocument7 pagesDevelopment of A Computational Model For Optimal Sbavin rajNo ratings yet
- 2017.10.23 Denton Presentation v1.2Document38 pages2017.10.23 Denton Presentation v1.2LaryNo ratings yet
- Captrate Combo Filters: Introducing The ImprovedDocument2 pagesCaptrate Combo Filters: Introducing The ImprovedYvanNo ratings yet
- Ultrasonic Sensor UB6000-F42-E6-V15: DimensionsDocument6 pagesUltrasonic Sensor UB6000-F42-E6-V15: DimensionsSpring SuperbNo ratings yet
- Examples of Lien and Bar GraphsDocument17 pagesExamples of Lien and Bar GraphsTemi FatokunNo ratings yet
- General Lens Data: DatasheetDocument2 pagesGeneral Lens Data: DatasheetZbigniew LipinskiNo ratings yet
- Risk Assessment PDFDocument54 pagesRisk Assessment PDFTeju AshuNo ratings yet
- General Lens Data: DatasheetDocument2 pagesGeneral Lens Data: DatasheetMarcin PyszynskiNo ratings yet
- General Lens Data: DatasheetDocument2 pagesGeneral Lens Data: DatasheetZbigniew LipinskiNo ratings yet
- Product Information SWEDAC Damping CassettesDocument8 pagesProduct Information SWEDAC Damping CassettesJay JayNo ratings yet
- Calypso TRV-3 InstructionDocument2 pagesCalypso TRV-3 Instructionjimmy.hallberg.jhNo ratings yet
- CC06 P09 Qualitative Uncertainty Reliability of Chemical Identification With High Resolution Mass Spectrometry - Marios KostakisDocument8 pagesCC06 P09 Qualitative Uncertainty Reliability of Chemical Identification With High Resolution Mass Spectrometry - Marios KostakisAmira BaihaniNo ratings yet
- CellTiter 96 AQueous One Solution Cell Proliferation Assay TB245Document13 pagesCellTiter 96 AQueous One Solution Cell Proliferation Assay TB245Teh Chye PhingNo ratings yet
- Modern Particle Characterization Techniques Image AnalysisDocument46 pagesModern Particle Characterization Techniques Image AnalysisSONWALYOGESHNo ratings yet
- Estandar Disolucion LevoDocument5 pagesEstandar Disolucion LevoBrendapaez3No ratings yet
- PRTG Report 400 - Summary Report For All Sensors - Created 2023-09-01 23-05-02 (2023-08-25 00-00 - 2023-09-01 00-00) UTCDocument9 pagesPRTG Report 400 - Summary Report For All Sensors - Created 2023-09-01 23-05-02 (2023-08-25 00-00 - 2023-09-01 00-00) UTCMuhammed KunnappillyNo ratings yet
- Elevation Section A-A-Layout1Document1 pageElevation Section A-A-Layout1lawrenceNo ratings yet
- CBL Interpretation Chart: Through-Pipe EvaluationDocument1 pageCBL Interpretation Chart: Through-Pipe EvaluationFalza WihdanyNo ratings yet
- Company Comparison Economics: Merak ®Document13 pagesCompany Comparison Economics: Merak ®Tripoli ManoNo ratings yet
- Luzenac10M0 - TalcDocument1 pageLuzenac10M0 - Talcsriatul2006No ratings yet
- Luzenac10M0 - Talc PDFDocument1 pageLuzenac10M0 - Talc PDFsriatul2006No ratings yet
- Luzenac10M0 - Talc PDFDocument1 pageLuzenac10M0 - Talc PDFsriatul2006No ratings yet
- Luzenac10M0 - Talc PDFDocument1 pageLuzenac10M0 - Talc PDFsriatul2006No ratings yet
- Loop Formation: Special Cases in Optimization Model-IDocument1 pageLoop Formation: Special Cases in Optimization Model-ISamyuktha KNo ratings yet
- Noctilux M50 TechnicalDataDocument7 pagesNoctilux M50 TechnicalDataAlessio MalinverniNo ratings yet
- Sds PDFDocument1 pageSds PDFrajeeshNo ratings yet
- Blower Conventional Double Blower Double Inlet: ConstructionDocument1 pageBlower Conventional Double Blower Double Inlet: ConstructionrajeeshNo ratings yet
- CT2012 Deployable Structures: - Concepts & ExplorationsDocument40 pagesCT2012 Deployable Structures: - Concepts & ExplorationsJay PatelNo ratings yet
- KW Oxides 2019-V9-Part-6Document3 pagesKW Oxides 2019-V9-Part-6qkzxbik734No ratings yet
- Granite Comparator StandDocument3 pagesGranite Comparator Standรุ่งทิวา ทวีทรัพย์No ratings yet
- Abstract NidhiDocument1 pageAbstract NidhinitinNo ratings yet
- Dhona Balance001Document1 pageDhona Balance001nitinNo ratings yet
- Good Clinical Practices:: ProtocolsDocument12 pagesGood Clinical Practices:: ProtocolsnitinNo ratings yet
- Dhona Balance02Document1 pageDhona Balance02nitin100% (1)
- 1Document11 pages1nitinNo ratings yet
- Bhava VicharDocument3 pagesBhava VicharnitinNo ratings yet
- Pharmacy Council RECIPT PDFDocument2 pagesPharmacy Council RECIPT PDFnitinNo ratings yet
- MPPC201715959 DigiDocument1 pageMPPC201715959 DiginitinNo ratings yet
- STUDENTS Info LIST 2018-19Document191 pagesSTUDENTS Info LIST 2018-19nitinNo ratings yet
- 14 - Part 3 - Ch. 7 - A Lesson With Czerny - Tempo Rubato PDFDocument21 pages14 - Part 3 - Ch. 7 - A Lesson With Czerny - Tempo Rubato PDFErduandNo ratings yet
- Scope and Sequence of Math CurriculumDocument2 pagesScope and Sequence of Math CurriculumFaisal MunirNo ratings yet
- Finite Element Analysis Using MATLAB ANSYS 16 HrsDocument2 pagesFinite Element Analysis Using MATLAB ANSYS 16 HrsMOHIT PANTNo ratings yet
- Design of Band BrakeDocument23 pagesDesign of Band BrakeRushikesh WandhekarNo ratings yet
- RN Nemo Outdoor 7 6 0 7 30th June 2015Document55 pagesRN Nemo Outdoor 7 6 0 7 30th June 2015yatishjainkotaNo ratings yet
- ASTM e 186Document4 pagesASTM e 186Ashish GuptaNo ratings yet
- Nizami Akturk 1993 PHD ThesisDocument299 pagesNizami Akturk 1993 PHD ThesisGerman ToledoNo ratings yet
- Analog - RF VLSI Design (ECE 503) (Makeup) PDFDocument2 pagesAnalog - RF VLSI Design (ECE 503) (Makeup) PDFRahul PinnamaneniNo ratings yet
- Structural and Process Dimensions of OrgDocument12 pagesStructural and Process Dimensions of OrgrafaelwidiNo ratings yet
- Chains For Power TransmissionDocument11 pagesChains For Power TransmissionQasim JavedNo ratings yet
- Electromagnetic InductionDocument25 pagesElectromagnetic Inductionnivasan aNo ratings yet
- Kavitha Brunswick: SAP HANA, BODS and BI Consultant - ADOBEDocument6 pagesKavitha Brunswick: SAP HANA, BODS and BI Consultant - ADOBEparag3482No ratings yet
- YAW - S Cross ReliefDocument5 pagesYAW - S Cross ReliefRonaldNo ratings yet
- The Key of It AllDocument91 pagesThe Key of It AllSimonida Mona VulićNo ratings yet
- BUILDING SERVICES-I Sanitary Fixtures, Fittings & Wellness-1Document48 pagesBUILDING SERVICES-I Sanitary Fixtures, Fittings & Wellness-1Vidya Hitti100% (2)
- ADC and DAC Glossary - Tutorial - Maxim PDFDocument16 pagesADC and DAC Glossary - Tutorial - Maxim PDFshaan_patilNo ratings yet
- Botta-Dukát 2005 (RaoQ)Document8 pagesBotta-Dukát 2005 (RaoQ)BeatrizNo ratings yet
- 2 Separator 123 KV in Linie-Paralel Fara CLP - B92145MCDC09-B - R00Document2 pages2 Separator 123 KV in Linie-Paralel Fara CLP - B92145MCDC09-B - R00Nicolae Dan OnacaNo ratings yet
- Communicating With The MicroprocessorDocument13 pagesCommunicating With The MicroprocessorRyan John de LaraNo ratings yet
- Brahe 1000starsDocument55 pagesBrahe 1000starsefrain dalmagroNo ratings yet
- Sarojini Naidu Govt Girls P.G. College: Bca Vi Sem. Cce I JavaDocument12 pagesSarojini Naidu Govt Girls P.G. College: Bca Vi Sem. Cce I Javamaria khanNo ratings yet
- Manual de AVERIAS Codigos SC RICOH MP C4500Document84 pagesManual de AVERIAS Codigos SC RICOH MP C4500Erika Velásquez100% (1)
- Using Scoria As Fine Aggregate in Lightweight Mortar and ConcreteDocument14 pagesUsing Scoria As Fine Aggregate in Lightweight Mortar and ConcreteFoolad GharbNo ratings yet
- Jurnal AutorefractometerDocument11 pagesJurnal AutorefractometerNeida Valeria DanunNo ratings yet
- The Physics of Bowling BallsDocument34 pagesThe Physics of Bowling Ballskhisham20005389100% (2)
- Arria 10 FPGA Development Kit User Guide: Subscribe Send FeedbackDocument135 pagesArria 10 FPGA Development Kit User Guide: Subscribe Send FeedbacklolNo ratings yet
- Theory of ProductionDocument24 pagesTheory of ProductionKanika BakshiNo ratings yet