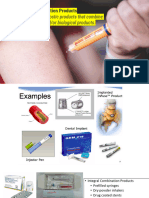
Professional Documents
Culture Documents
Medical Devices Product Classification Guide
Uploaded by
NunoOriginal Title
Copyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
Medical Devices Product Classification Guide
Uploaded by
NunoCopyright:
Available Formats
MEDICAL DEVICES
PRODUCT CLASSIFICATION
GUIDE
Medical Devices Cluster
November 2021
All Rights Reserved, Health Sciences Authority 1
CONTENTS
1. INTRODUCTION
i. Objective
ii. Definitions
iii. Intended Purpose
2. PRODUCTS WITH MEDICAL RELATED PURPOSE
i. Sterilization or Disinfection
ii. Contact Lens Care Products
iii. Products used in Hospital or Laboratories
iv. Ophthalmic Products
v. Solutions or Media for Tissues/Cells
vi. Others
3. GENERAL PURPOSE PRODUCTS
4. ASSISTIVE TECHNOLOGY PRODUCTS
5. PRODUCTS FOR SPORTS/PHYSICAL FITNESS/GENERAL HEALTH
6. PERSONAL PROTECTIVE EQUIPMENTS (PPE)
1) INTRODUCTION
Objective Definitions Intended Purpose
All Rights Reserved, Health Sciences Authority 2
INTRODUCTION
Objective
• Medical device (MD) is defined in the First Schedule of the Health Products Act (HPA)
and products that meet this definition are regulated as MD by HSA. However, for
certain products, it may be challenging to interpret whether or not it would be
classified as a MD, within the scope of the MD definition. This document has been
developed to aid with classification of some of these more challenging products.
• Many a time it is incorrectly assumed that because a product is considered a MD in
some countries, it will also be a medical device in Singapore. This is not the case and
the definition of MD should always be referred to, when determining the classification.
• Products falling outside the scope of the MD regulatory controls may still be
regulated:
- as Health Products under other Product Categories (such as Therapeutic
Product, or Cosmetic Products) under the Health Products Act, or
- under other legislation (e.g. Radiation Protection Act, Consumer Protection (Fair
Trading) Act, etc).
1) INTRODUCTION
Objective Definitions Intended Purpose
All Rights Reserved, Health Sciences Authority 3
INTRODUCTION
MD Definition (First Schedule of HPA)
MEDICAL DEVICE means:
a) Any instrument, apparatus, implement, machine, appliance, implant, reagent for in vitro use, software,
material or other similar or related article that is intended by its manufacturer to be used, whether alone or
in combination, for humans for one or more of the specific purposes of –
– Diagnosis, prevention, monitoring, treatment or alleviate of disease;
– Diagnosis, monitoring, treatment or alleviation of, or compensation for, an injury;
– Investigation, replacement, modification or support of the anatomy or of a physiological process, mainly for
medical purposes;
– Supporting or sustaining life;
– Control of contraception;
– Disinfection of medical devices; or
– Providing information by means of in vitro examination of specimens derived from the human body, for medical
or diagnostic purposes,
and which does not achieve its primary intended action in or on the human body by
pharmacological, immunological or metabolic means, but which may be assisted in its intended
function by such means; and
b) The following articles:
– Any implant for the modification or fixation of any body part;
– Any injectable dermal filler or mucous membrane filler
– Any instrument, apparatus, implement, machine or appliance intended to be used for the removal or degradation
of fat by invasive means
1) INTRODUCTION
Objective Definitions Intended Purpose
All Rights Reserved, Health Sciences Authority 4
INTRODUCTION
Intended Purpose
• In order to determine whether a product is considered a medical device, the following
should be considered:
A. The intended purpose of the product taking into account the way the product is
designed and/or presented.
- If the intended purpose fits the MD definition in the First Schedule of Health
Products Act (HPA), then it would be subject to control as a medical device in
Singapore.
B. The primary mode of action by which the intended purpose is achieved.
– For a medical device, the primary mode of action by which the intended purpose
is typically achieved is by physical means (including mechanical action,
replacement of, or support of the anatomy or of a physiological process).
1) INTRODUCTION
Objective Definitions Intended Purpose
All Rights Reserved, Health Sciences Authority 5
PRODUCTS WITH MEDICAL RELATED PURPOSE
Sterilization or disinfection
PRODUCT(S) CLASSIFICATION REMARKS
Hand or skin antiseptic wipes, Not Medical
hand sanitizers Device
Disinfectant or Sterilant for Medical Device
medical device
Sterilization pouch specifically Medical Device Note: sterilization pouch used for the packaging
intended by its product owner to and sterilization of a medical device prior to the
be used together with the supply of the device is part of the materials used in
sterilizer for re-sterilization of manufacturing process, and hence would not be
medical device classified as a Medical Device.
PRODUCTS WITH GENERAL ASSISTIVE PRODUCTS FOR PERSONAL
MEDICAL RELATED PURPOSE TECHNOLOGY SPORTS/PHYSICAL PROTECTIVE
PURPOSE PRODUCTS PRODUCTS FITNESS/GENERAL HEALTH EQUIPMENT (PPE)
3) PRODUCTS USED IN 5) SOLUTIONS OR
1) STERILIZATION OR 2) CONTACT LENS 4) OPTHALMIC MEDIA FOR 6) OTHERS
HOSPITAL/
DISINFECTION CARE PRODUCTS PRODUCTS TISSUES/CELLS
LABORATORY
All Rights Reserved, Health Sciences Authority 6
PRODUCTS WITH MEDICAL RELATED PURPOSE
Contact Lens Care Products
PRODUCT(S) CLASSIFICATION REMARKS
Rinsing solutions for contact lens Not Medical Device
Cleaning solutions, Enzyme Tablet, or Not Medical Device
Liquid for protein removal of contact
lens
Contact lens multipurpose solution Multipurpose solutions with intended purposes
May be Medical
(such as for cleaning, disinfecting, that includes disinfection or storage of contact
Device
rinsing, and/or storing of contact lens) lens will be classified as MD.
Hydrating, Lubricating, or Rewetting Medical Device Intended for use to hydrate or lubricate, and to
solutions (to be instil in the eyes while reduce friction between the lens and eyes.
wearing contact lens)
PRODUCTS WITH GENERAL ASSISTIVE PRODUCTS FOR PERSONAL
MEDICAL RELATED PURPOSE TECHNOLOGY SPORTS/PHYSICAL PROTECTIVE
PURPOSE PRODUCTS PRODUCTS FITNESS/GENERAL HEALTH EQUIPMENT (PPE)
3) PRODUCTS USED IN 5) SOLUTIONS OR
1) STERILIZATION OR 2) CONTACT LENS 4) OPTHALMIC MEDIA FOR 6) OTHERS
HOSPITAL/
DISINFECTION CARE PRODUCTS PRODUCTS TISSUES/CELLS
LABORATORY
All Rights Reserved, Health Sciences Authority 7
PRODUCTS WITH MEDICAL RELATED PURPOSE
Products Used in Hospital/Laboratory
PRODUCT(S) CLASSIFICATION REMARKS
Laboratory equipment for: Medical Device Examples:
- In-vitro diagnostic (IVD) use - Incubators, centrifuges, or analysers for IVD
or use
- In-vitro fertilisation (IVF) - IVF pipettes, transfer catheters or needles.
Blood bags (empty) and blood Medical Device
bags (containing
anticoagulant/preservatives)
PRODUCTS WITH GENERAL ASSISTIVE PRODUCTS FOR PERSONAL
MEDICAL RELATED PURPOSE TECHNOLOGY SPORTS/PHYSICAL PROTECTIVE
PURPOSE PRODUCTS PRODUCTS FITNESS/GENERAL HEALTH EQUIPMENT (PPE)
3) PRODUCTS USED IN 5) SOLUTIONS OR
1) STERILIZATION OR 2) CONTACT LENS 4) OPTHALMIC MEDIA FOR 6) OTHERS
HOSPITAL/
DISINFECTION CARE PRODUCTS PRODUCTS TISSUES/CELLS
LABORATORY
All Rights Reserved, Health Sciences Authority 8
PRODUCTS WITH MEDICAL RELATED PURPOSE
Products Used in Hospital/Laboratory
PRODUCT(S) CLASSIFICATION REMARKS
Blood refrigerator, or freezer Medical Device Intended to preserve blood and blood products
by storing them at cold or freezing temperature
for eventual infusion/ transfusion.
Vaccine/Pharmaceutical/ Not Medical Device Intended to store vaccines and other medical
Medical Refrigerator products at a stable temperature (prevent
(not intended for freezing or temperature fluctuations) to ensure an ideal cold
storing blood bags) temperature storage for sensitive items due to
quality issues and not degrade.
PRODUCTS WITH GENERAL ASSISTIVE PRODUCTS FOR PERSONAL
MEDICAL RELATED PURPOSE TECHNOLOGY SPORTS/PHYSICAL PROTECTIVE
PURPOSE PRODUCTS PRODUCTS FITNESS/GENERAL HEALTH EQUIPMENT (PPE)
3) PRODUCTS USED IN 5) SOLUTIONS OR
1) STERILIZATION OR 2) CONTACT LENS 4) OPTHALMIC MEDIA FOR 6) OTHERS
HOSPITAL/
DISINFECTION CARE PRODUCTS PRODUCTS TISSUES/CELLS
LABORATORY
All Rights Reserved, Health Sciences Authority 9
PRODUCTS WITH MEDICAL RELATED PURPOSE
Ophthalmic Products
PRODUCT(S) CLASSIFICATION REMARKS
Eye drops Medical Device Examples:
(primary mode of action is - Lubricating or moisturizing eye
physical: lubricating or physical drops for relief of dry, irritated eyes
washing) - Eye drops for relief of irritated eyes
via physical washing of eye surface
Eye bath or Eye wash Medical Device An example is the saline eye wash
(primary mode of action is intended for irrigation of the eyes,
physical: mechanical irrigation) during emergency.
PRODUCTS WITH GENERAL ASSISTIVE PRODUCTS FOR PERSONAL
MEDICAL RELATED PURPOSE TECHNOLOGY SPORTS/PHYSICAL PROTECTIVE
PURPOSE PRODUCTS PRODUCTS FITNESS/GENERAL HEALTH EQUIPMENT (PPE)
3) PRODUCTS USED IN 5) SOLUTIONS OR
1) STERILIZATION OR 2) CONTACT LENS 4) OPTHALMIC MEDIA FOR 6) OTHERS
HOSPITAL/
DISINFECTION CARE PRODUCTS PRODUCTS TISSUES/CELLS
LABORATORY
All Rights Reserved, Health Sciences Authority 10
PRODUCTS WITH MEDICAL RELATED PURPOSE
Ophthalmic Products
PRODUCT(S) CLASSIFICATION REMARKS
Fluorescein Ocular strips Medical Device For evaluating the fit of contact lenses
(works via staining of tear film).
Not Medical Device Intended for use to enhance visualisation of
anatomy for diagnostic purposes.
Fluorescein eye drop Not Medical Device Intended for use as diagnostic agent for eye
conditions and/or fitting of contact lens.
PRODUCTS WITH GENERAL ASSISTIVE PRODUCTS FOR PERSONAL
MEDICAL RELATED PURPOSE TECHNOLOGY SPORTS/PHYSICAL PROTECTIVE
PURPOSE PRODUCTS PRODUCTS FITNESS/GENERAL HEALTH EQUIPMENT (PPE)
3) PRODUCTS USED IN 5) SOLUTIONS OR
1) STERILIZATION OR 2) CONTACT LENS 4) OPTHALMIC MEDIA FOR 6) OTHERS
HOSPITAL/
DISINFECTION CARE PRODUCTS PRODUCTS TISSUES/CELLS
LABORATORY
All Rights Reserved, Health Sciences Authority 11
PRODUCTS WITH MEDICAL RELATED PURPOSE
Ophthalmic Products
PRODUCT(S) CLASSIFICATION REMARKS
Contact lens with Medical Device
corrective function
(powered contact lens)
Non-corrective contact Not Medical Device Decorative lens with no corrective function, used to change the
lens (zero powered colour or appearance of the eyes.
contact lens) Refer to Regulatory Guidelines For Devices for Modification of
Appearance or Anatomy [1]
Non-corrective contact Medical Device Some non-corrective contact lens may have a medical
lens labelled with purpose, to treat a number of congenital or traumatic
medical claims conditions. An example is UV blocking contact lenses which
claims to protect against transmission of harmful UV radiation
to the cornea and into the eye, to alleviate photophobia in
albinism.
PRODUCTS WITH GENERAL ASSISTIVE PRODUCTS FOR PERSONAL
MEDICAL RELATED PURPOSE TECHNOLOGY SPORTS/PHYSICAL PROTECTIVE
PURPOSE PRODUCTS PRODUCTS FITNESS/GENERAL HEALTH EQUIPMENT (PPE)
3) PRODUCTS USED IN 5) SOLUTIONS OR
1) STERILIZATION OR 2) CONTACT LENS 4) OPTHALMIC MEDIA FOR 6) OTHERS
HOSPITAL/
DISINFECTION CARE PRODUCTS PRODUCTS TISSUES/CELLS
LABORATORY
All Rights Reserved, Health Sciences Authority 12
PRODUCTS WITH MEDICAL RELATED PURPOSE
Solutions/Media for Tissues/Cells
PRODUCT(S) CLASSIFICATION REMARKS
Cell culture media for cell or tissue Not Medical Considered as processing reagents.
therapy Device
Organ preservation solution Not Medical Comparable to processing reagents. An example
Device is solution for preserving of corneal material prior
transplant.
PRODUCTS WITH GENERAL ASSISTIVE PRODUCTS FOR PERSONAL
MEDICAL RELATED PURPOSE TECHNOLOGY SPORTS/PHYSICAL PROTECTIVE
PURPOSE PRODUCTS PRODUCTS FITNESS/GENERAL HEALTH EQUIPMENT (PPE)
3) PRODUCTS USED IN 5) SOLUTIONS OR
1) STERILIZATION OR 2) CONTACT LENS 4) OPTHALMIC MEDIA FOR 6) OTHERS
HOSPITAL/
DISINFECTION CARE PRODUCTS PRODUCTS TISSUES/CELLS
LABORATORY
All Rights Reserved, Health Sciences Authority 13
PRODUCTS WITH MEDICAL RELATED PURPOSE
Solutions/Media for Tissues/Cells
PRODUCT(S) CLASSIFICATION REMARKS
In vitro fertilisation (IVF) media for: Medical Device Replacement or modification of a physiological
• Sperm preparation process.
• Follicle Aspiration
• Rinsing
• Fertilization
• Embryo Culture
• Embryo Transfer
In vitro diagnostic (IVD) culture Medical Device Labelled for IVD use. For definition of IVD
media product, please refer to Guidance on Risk
Classification of IVD Medical Devices (GN-14) [2].
PRODUCTS WITH GENERAL ASSISTIVE PRODUCTS FOR PERSONAL
MEDICAL RELATED PURPOSE TECHNOLOGY SPORTS/PHYSICAL PROTECTIVE
PURPOSE PRODUCTS PRODUCTS FITNESS/GENERAL HEALTH EQUIPMENT (PPE)
3) PRODUCTS USED IN 5) SOLUTIONS OR
1) STERILIZATION OR 2) CONTACT LENS 4) OPTHALMIC MEDIA FOR 6) OTHERS
HOSPITAL/
DISINFECTION CARE PRODUCTS PRODUCTS TISSUES/CELLS
LABORATORY
All Rights Reserved, Health Sciences Authority 14
PRODUCTS WITH MEDICAL RELATED PURPOSE
Others
PRODUCT(S) CLASSIFICATION REMARKS
Cold laser therapy (for pain management Medical device
and/or to accelerate healing)
Liposuction Machine Medical Device Intended to remove fats from the body using
suction (by invasive means).
Refer to Regulatory Guidelines For Devices
for Modification of Appearance or Anatomy
[1]
Sitz bath Medical Device Generally intended for pain relief.
Medical gases such as oxygen for Not Medical Device
breathing and anaesthetic gases
(supplied in cylinders)
PRODUCTS WITH GENERAL ASSISTIVE PRODUCTS FOR PERSONAL
MEDICAL RELATED PURPOSE TECHNOLOGY SPORTS/PHYSICAL PROTECTIVE
PURPOSE PRODUCTS PRODUCTS FITNESS/GENERAL HEALTH EQUIPMENT (PPE)
3) PRODUCTS USED IN 5) SOLUTIONS OR
1) STERILIZATION OR 2) CONTACT LENS 4) OPTHALMIC MEDIA FOR 6) OTHERS
HOSPITAL/
DISINFECTION CARE PRODUCTS PRODUCTS TISSUES/CELLS
LABORATORY
All Rights Reserved, Health Sciences Authority 15
PRODUCTS WITH MEDICAL RELATED PURPOSE
Others
PRODUCT(S) CLASSIFICATION REMARKS
Acupuncture Needles, Electro- Medical Device
acupuncture machines
Dental aligners, Dental retainers for Medical Device
use after orthodontic treatment
In-vivo diagnostic agents such as x- Not Medical
ray contrast media and gases for Device
lung function test
PRODUCTS WITH GENERAL ASSISTIVE PRODUCTS FOR PERSONAL
MEDICAL RELATED PURPOSE TECHNOLOGY SPORTS/PHYSICAL PROTECTIVE
PURPOSE PRODUCTS PRODUCTS FITNESS/GENERAL HEALTH EQUIPMENT (PPE)
3) PRODUCTS USED IN 5) SOLUTIONS OR
1) STERILIZATION OR 2) CONTACT LENS 4) OPTHALMIC MEDIA FOR 6) OTHERS
HOSPITAL/
DISINFECTION CARE PRODUCTS PRODUCTS TISSUES/CELLS
LABORATORY
All Rights Reserved, Health Sciences Authority 16
PRODUCTS WITH MEDICAL RELATED PURPOSE
Others
PRODUCT(S) CLASSIFICATION REMARKS
Hot or cold packs, Heat pads, Medical Device Generally intended for pain relief.
Hot or cold water bottles
Not Medical Labelled with specific intended purpose to
Device exclude these products from being a medical
device. An example is heat pads labelled for
keeping warmth in cold weather.
PRODUCTS WITH GENERAL ASSISTIVE PRODUCTS FOR PERSONAL
MEDICAL RELATED PURPOSE TECHNOLOGY SPORTS/PHYSICAL PROTECTIVE
PURPOSE PRODUCTS PRODUCTS FITNESS/GENERAL HEALTH EQUIPMENT (PPE)
3) PRODUCTS USED IN 5) SOLUTIONS OR
1) STERILIZATION OR 2) CONTACT LENS 4) OPTHALMIC MEDIA FOR 6) OTHERS
HOSPITAL/
DISINFECTION CARE PRODUCTS PRODUCTS TISSUES/CELLS
LABORATORY
All Rights Reserved, Health Sciences Authority 17
PRODUCTS WITH MEDICAL RELATED PURPOSE
Others
PRODUCT(S) CLASSIFICATION REMARKS
Peritoneal dialysis solution Not Medical Device
Hemofiltration solution Not Medical Device Note: It is a replacement/substitution solution
used in renal replacement therapy, where the
exchange of solutes is by convection
(hemofiltration).
Haemodialysis solution Medical Device Note: Exchange of solutes is by diffusion, across
a semi-permeable membrane.
PRODUCTS WITH GENERAL ASSISTIVE PRODUCTS FOR PERSONAL
MEDICAL RELATED PURPOSE TECHNOLOGY SPORTS/PHYSICAL PROTECTIVE
PURPOSE PRODUCTS PRODUCTS FITNESS/GENERAL HEALTH EQUIPMENT (PPE)
3) PRODUCTS USED IN 5) SOLUTIONS OR
1) STERILIZATION OR 2) CONTACT LENS 4) OPTHALMIC MEDIA FOR 6) OTHERS
HOSPITAL/
DISINFECTION CARE PRODUCTS PRODUCTS TISSUES/CELLS
LABORATORY
All Rights Reserved, Health Sciences Authority 18
PRODUCTS WITH MEDICAL RELATED PURPOSE
Others
PRODUCT(S) CLASSIFICATION REMARKS
Pressure or gas regulators for Medical Device
medical gases (to control flow
rate)
Female intimate hygiene Not Medical Device An example is douche or gel intended to restore
products natural pH in vagina, for symptomatic relief of
(primary mode of action is via vaginal irritation, or infections.
restoration of vaginal pH)
PRODUCTS WITH GENERAL ASSISTIVE PRODUCTS FOR PERSONAL
MEDICAL RELATED PURPOSE TECHNOLOGY SPORTS/PHYSICAL PROTECTIVE
PURPOSE PRODUCTS PRODUCTS FITNESS/GENERAL HEALTH EQUIPMENT (PPE)
3) PRODUCTS USED IN 5) SOLUTIONS OR
1) STERILIZATION OR 2) CONTACT LENS 4) OPTHALMIC MEDIA FOR 6) OTHERS
HOSPITAL/
DISINFECTION CARE PRODUCTS PRODUCTS TISSUES/CELLS
LABORATORY
All Rights Reserved, Health Sciences Authority 19
PRODUCTS WITH MEDICAL RELATED PURPOSE
Others
PRODUCT(S) CLASSIFICATION REMARKS
Lubricants Medical Device Lubricants with a medical purpose.
Examples:
- Lubricant intended for use in medical procedures with medical
devices such as endoscopes
- Lubricant intended for use with condoms (medical device) to facilitate
the function of condom
- Vaginal lubricant intended for a medical purpose such as for
treatment of vaginal dryness
Not Medical Device Personal lubricants that is intended for a non-medical purpose (e.g.
sexual comfort/ for pleasure).
Note: Personal lubricants which claims compatibility to condom (e.g.
can be used with condom) are not medical device as it is only to
indicate the fact that it can be safely used in combination with (latex)
condom. Such personal lubricants compatible for use with condoms (ie.
latex) are not medical devices.
PRODUCTS WITH GENERAL ASSISTIVE PRODUCTS FOR PERSONAL
MEDICAL RELATED PURPOSE TECHNOLOGY SPORTS/PHYSICAL PROTECTIVE
PURPOSE PRODUCTS PRODUCTS FITNESS/GENERAL HEALTH EQUIPMENT (PPE)
3) PRODUCTS USED IN 5) SOLUTIONS OR
1) STERILIZATION OR 2) CONTACT LENS 4) OPTHALMIC MEDIA FOR 6) OTHERS
HOSPITAL/
DISINFECTION CARE PRODUCTS PRODUCTS TISSUES/CELLS
LABORATORY
All Rights Reserved, Health Sciences Authority 20
GENERAL PURPOSE PRODUCTS
Products that serves multiple purposes may occasionally be used within a medical setting. These
are not considered medical devices, unless the product is intended for a specific medical purpose.
PRODUCT(S) CLASSIFICATION REMARKS
Air Decontamination or Not Medical Device Products intended to remove allergens (dust, pollen,
Purification Units etc) and/or to remove bacteria from the air in a
specific area/room. They may be used in hospitals
for air decontamination, aiming to reduce infection
risks in intensive care units and operating theatres.
Not for disinfection of medical devices.
Multipurpose general Not Medical Device These products are intended for disinfecting various
disinfectant surfaces such as floors, walls and sanitary facilities.
Not for disinfection of medical devices.
Multipurpose computer Not Medical Device These products are not medical devices unless they
and printer are specifically intended* to be used in
combination with a medical device as an accessory.
*Specifically intended refers to a clear indication of such use in the information accompanying the medical device.
PRODUCTS WITH GENERAL ASSISTIVE PRODUCTS FOR PERSONAL
MEDICAL RELATED PURPOSE TECHNOLOGY SPORTS/PHYSICAL PROTECTIVE
PURPOSE PRODUCTS PRODUCTS FITNESS/GENERAL HEALTH EQUIPMENT (PPE)
All Rights Reserved, Health Sciences Authority 21
GENERAL PURPOSE PRODUCTS
PRODUCT(S) CLASSIFICATION REMARKS
Toothbrush,
Toothpaste, Primarily intended for basic oral hygiene and/or
Not Medical Device
Mouthwash, cleaning.
Dental floss
Denture cleansing tablets Not Medical Device
Dental disclosing
Not Medical Device Generally used to disclose dental plaques on teeth.
solution/tablets
Tooth whitening or Tooth whitening instruments: refer to Regulatory
bleaching products Not Medical Device Guideline for Devices for Modification of
(Instruments and gel) Appearance or Anatomy [1]
PRODUCTS WITH GENERAL ASSISTIVE PRODUCTS FOR PERSONAL
MEDICAL RELATED PURPOSE TECHNOLOGY SPORTS/PHYSICAL PROTECTIVE
PURPOSE PRODUCTS PRODUCTS FITNESS/GENERAL HEALTH EQUIPMENT (PPE)
All Rights Reserved, Health Sciences Authority 22
GENERAL PURPOSE PRODUCTS
PRODUCT(S) CLASSIFICATION REMARKS
Female hygiene products Not Medical Device
such as: Tampons,
Menstrual Cup,
Pads/sanitary towel
Diapers (for baby and/ or Not Medical Device Generally used to contain or absorb urine and waste.
adults) and
Incontinence pads
Commodes and Not Medical Device Generally used to contain body waste.
Bedpans (Body waste
receptacles)
Gallipots, Not Medical Device Generally intended for a variety of purposes such as
Kidney dishes containing fluids, mixing solutions, carrying or holding
instruments prior to or during a procedure, and
collecting body waste or other matters.
PRODUCTS WITH GENERAL ASSISTIVE PRODUCTS FOR PERSONAL
MEDICAL RELATED PURPOSE TECHNOLOGY SPORTS/PHYSICAL PROTECTIVE
PURPOSE PRODUCTS PRODUCTS FITNESS/GENERAL HEALTH EQUIPMENT (PPE)
All Rights Reserved, Health Sciences Authority 23
GENERAL PURPOSE PRODUCTS
PRODUCT(S) CLASSIFICATION REMARKS
Breast pumps Not Medical Device Generally, breast pumps are intended for milk
extraction.
Sunscreens Not Medical Device
Insect repellent, Mosquito Not Medical Device
repellent or patches
Lice comb Not Medical Device For removal of lice and lice eggs from human hair
Medicine spoons, Medicine Not Medical Device
droppers,
Dispensers or Storage
containers for pills, capsules
and tablets
PRODUCTS WITH GENERAL ASSISTIVE PRODUCTS FOR PERSONAL
MEDICAL RELATED PURPOSE TECHNOLOGY SPORTS/PHYSICAL PROTECTIVE
PURPOSE PRODUCTS PRODUCTS FITNESS/GENERAL HEALTH EQUIPMENT (PPE)
All Rights Reserved, Health Sciences Authority 24
GENERAL PURPOSE PRODUCTS
PRODUCT(S) CLASSIFICATION REMARKS
Laser equipment Not Medical Device Intended to remove fats from the body (by non-invasive
(for non-invasive lipolysis) means). Refer to Regulatory Guidelines For Devices for
Modification of Appearance or Anatomy [1]
Needles for tattooing Not Medical Device Refer to Regulatory Guidelines For Devices for
Modification of Appearance or Anatomy [1]
Microneedles or dermal Not Medical Device Refer to Regulatory Guidelines For Devices for
rollers (to create micro Modification of Appearance or Anatomy [1]
punctures on the skin) for
skin rejuvenation or for
scars
Brushes, or Sponges for Not Medical Device An example is brush or sponge for use by healthcare
washing and/or cleaning of professionals prior surgery (on intact skin not relating to
nails and/or hands wound care), for cleaning of nails and/or hands.
PRODUCTS WITH GENERAL ASSISTIVE PRODUCTS FOR PERSONAL
MEDICAL RELATED PURPOSE TECHNOLOGY SPORTS/PHYSICAL PROTECTIVE
PURPOSE PRODUCTS PRODUCTS FITNESS/GENERAL HEALTH EQUIPMENT (PPE)
All Rights Reserved, Health Sciences Authority 25
GENERAL PURPOSE PRODUCTS
PRODUCT(S) CLASSIFICATION REMARKS
Pipelines/ manifolds in Not Medical Device
hospital (for anaesthetics
and other medical gases)
Ceiling pendants, fixed Not Medical Device Generally, infrastructures are not medical devices.
installations, treatment However, if the fixtures include examination lights
rooms in hospitals (also or gas flow control function (of medical gases) etc.,
including salt rooms or please contact the Medical Devices Cluster with
oxygen hyperbaric rooms product information.
outside of hospital setting)
General Laboratory Not Medical Device General lab equipment not for In-vitro diagnostic
Equipment use are not regulated as Medical Devices. For
definition of In-vitro diagnostic (IVD) devices,
please refer to Guidance on Risk Classification of
IVD Medical Devices (GN-14) [2].
PRODUCTS WITH GENERAL ASSISTIVE PRODUCTS FOR PERSONAL
MEDICAL RELATED PURPOSE TECHNOLOGY SPORTS/PHYSICAL PROTECTIVE
PURPOSE PRODUCTS PRODUCTS FITNESS/GENERAL HEALTH EQUIPMENT (PPE)
All Rights Reserved, Health Sciences Authority 26
ASSISTIVE TECHNOLOGY PRODUCTS
Assistive technology products are generally intended for alleviation of, or compensation for disability. They
are designed to help individual with disability to overcome barriers they face in carrying out their daily
living activities. Depending on the primary intended purpose, these products may be considered either
medical devices or general consumer products.
The main determining factor will be whether there is a:
(a) direct link between the corrective function of the product and the individual with disability and,
(b) that there is a stated medical purpose
MEDICAL DEVICES NOT MEDICAL DEVICES
Wheelchair Alarm system in toilets/homes
Walking stick Toilet equipment for disabled
Hearing Aid Grab rails at doorways or stairs
Prosthetic Devices Portable ramps
PRODUCTS WITH GENERAL ASSISTIVE PRODUCTS FOR PERSONAL
MEDICAL RELATED PURPOSE TECHNOLOGY SPORTS/PHYSICAL PROTECTIVE
PURPOSE PRODUCTS PRODUCTS FITNESS/GENERAL HEALTH EQUIPMENT (PPE)
All Rights Reserved, Health Sciences Authority 27
PRODUCTS FOR SPORTS/PHYSICAL FITNESS
/GENERAL HEALTH
In general, products intended for sport or leisure purposes are not considered as medical
devices. Similarly, products intended for physical fitness, maintenance of general state of health
(ie. weight management, recreational use, sleep management, detoxification, improving and
enhancing flow of (qi) are not considered medical devices.
PRODUCT(S) CLASSIFICATION REMARKS
Gym equipment or Watch with a May or May not be Refer to Regulatory Guidelines for
function to measure heart rate a Medical Device Telehealth Products [3] for
determination of product classification.
Foot patch intended to maintain Not Medical Device
good health, promote good
sleep, to soothe or relax muscle
Bandages or support for sprains Medical Device
or injuries
PRODUCTS WITH GENERAL ASSISTIVE PRODUCTS FOR PERSONAL
MEDICAL RELATED PURPOSE TECHNOLOGY SPORTS/PHYSICAL PROTECTIVE
PURPOSE PRODUCTS PRODUCTS FITNESS/GENERAL HEALTH EQUIPMENT (PPE)
All Rights Reserved, Health Sciences Authority 28
PERSONAL PROTECTIVE EQUIPMENT (PPE)
• PPE are products that are generally worn to protect users from workplace hazards.
• The primary determining factor will be whether the product is for use in a healthcare setting,
or for a medical purpose.
PRODUCT(S) CLASSIFICATION REMARKS
Gloves or gowns for non- Not Medical Device Gloves or gowns intended for other purposes
medical purposes (outside of healthcare settings) such as food
industry would not be considered as medical device.
Medical/ Examination/ Medical Device Gloves or gowns intended to be worn by healthcare
Surgical gloves, personnel during medical/surgical procedures for
Surgical/Isolation gowns the protection of:
• healthcare professional
• patient who may contract illnesses or medical
conditions from being exposed to potentially
infectious bacteria
PRODUCTS WITH GENERAL ASSISTIVE PRODUCTS FOR PERSONAL
MEDICAL RELATED PURPOSE TECHNOLOGY SPORTS/PHYSICAL PROTECTIVE
PURPOSE PRODUCTS PRODUCTS FITNESS/GENERAL HEALTH EQUIPMENT (PPE)
All Rights Reserved, Health Sciences Authority 29
PERSONAL PROTECTIVE EQUIPMENT (PPE)
PRODUCT(S) CLASSIFICATION REMARKS
Facemask/N95 respirator for Not Medical Device Facemask/ N95 respirator labelled solely for
non-medical purposes occupational uses would not be a medical
device.
Medical/ Surgical Facemasks Medical Device Medical/ surgical facemasks and N95
and N95 Respirators respirators intended for use in healthcare
settings.
Mouth guards for non-medical Not Medical Device Mouth guards intended for sports purposes.
purposes
PRODUCTS WITH GENERAL ASSISTIVE PRODUCTS FOR PERSONAL
MEDICAL RELATED PURPOSE TECHNOLOGY SPORTS/PHYSICAL PROTECTIVE
PURPOSE PRODUCTS PRODUCTS FITNESS/GENERAL HEALTH EQUIPMENT (PPE)
All Rights Reserved, Health Sciences Authority 30
References
[1] Regulatory Guideline for Devices for Modification of Appearance or
Anatomy
[2] Guidance on Risk Classification of IVD Medical Devices (GN-14)
[3] Regulatory Guidelines for Telehealth Products
Above References can be accessed online at:
https://www.hsa.gov.sg/medical-devices/guidance-documents
All Rights Reserved, Health Sciences Authority 31
CONTACT INFORMATION
Medical Devices Branch
Medical Devices Cluster
Health Products Regulation Group
Health Sciences Authority
11 Biopolis Way, #11-03 Helios
Singapore 138667
www.hsa.gov.sg
https://crm.hsa.gov.sg/event/feedback
All Rights Reserved, Health Sciences Authority 32
You might also like
- AHCC: The Medical Breakthrough in Natural ImmunotherapyFrom EverandAHCC: The Medical Breakthrough in Natural ImmunotherapyRating: 5 out of 5 stars5/5 (1)
- The SFDA General Rules For Products Claim 2.0Document16 pagesThe SFDA General Rules For Products Claim 2.0Ahmed Khalid ZohairyNo ratings yet
- Combination ProductDocument25 pagesCombination ProductVaibhav Narayan LagadNo ratings yet
- Class Guide SFDADocument60 pagesClass Guide SFDAAhmed AlyNo ratings yet
- Health Week 1Document21 pagesHealth Week 1lana del reyNo ratings yet
- Gourav Barai - Pradipta Mondal - Priyanka Kurmi - Ranajit Nath M.Pharmacy (Pharmaceutics) Regulatory Affair MPT-1064 07 November' 2019Document10 pagesGourav Barai - Pradipta Mondal - Priyanka Kurmi - Ranajit Nath M.Pharmacy (Pharmaceutics) Regulatory Affair MPT-1064 07 November' 2019Pradipta MondalNo ratings yet
- DeltaLAB Catalogue PDFDocument320 pagesDeltaLAB Catalogue PDFViktorija AvicenaNo ratings yet
- Act1 Abdullah Amera PDFDocument6 pagesAct1 Abdullah Amera PDFZyra Erylle Rodriguez CapistranoNo ratings yet
- Regulation For Medical Devices in IndiaDocument5 pagesRegulation For Medical Devices in IndiaPavan kumar NaikNo ratings yet
- Guideline For Classification of in Vitro Diagnostic Medical Devices - EFDADocument14 pagesGuideline For Classification of in Vitro Diagnostic Medical Devices - EFDAa.razakhajiNo ratings yet
- Catalogo Deltalab 2019 enDocument320 pagesCatalogo Deltalab 2019 enClarita Muller LeigueNo ratings yet
- APPENDIX 1 Food Drug Interphase FDI ProductsDocument9 pagesAPPENDIX 1 Food Drug Interphase FDI Productsmissmew91No ratings yet
- APPENDIX 1 Food Drug Interphase FDI ProductsDocument9 pagesAPPENDIX 1 Food Drug Interphase FDI ProductsVCARE 2UNo ratings yet
- S.No Substances Regulated by The FdaDocument6 pagesS.No Substances Regulated by The Fdasaurav choudharyNo ratings yet
- Control of The Practice of Pharmacy.Document4 pagesControl of The Practice of Pharmacy.RANIELLE SIMNo ratings yet
- Who Reuse Single UseDocument13 pagesWho Reuse Single UsenonanoviNo ratings yet
- Veterinary Medicines: Safe Use by Farmers and Other Animal HandlersDocument8 pagesVeterinary Medicines: Safe Use by Farmers and Other Animal HandlersRamesh BeniwalNo ratings yet
- Pemaparan Program Denok Kosasi, DRDocument17 pagesPemaparan Program Denok Kosasi, DRdenok kosasiNo ratings yet
- Medical Product Safety and RegulationDocument77 pagesMedical Product Safety and Regulationreycardo100% (1)
- Concepts in Using Products and Health Services: Grade-Level IndicatorDocument10 pagesConcepts in Using Products and Health Services: Grade-Level Indicatorสุขศึกษาและพละศึกษา อยวNo ratings yet
- Science Dan Regulasi Herbal Medicines: Dr. Eka Fitrianda, M.Farm, AptDocument19 pagesScience Dan Regulasi Herbal Medicines: Dr. Eka Fitrianda, M.Farm, Aptmufida adilaNo ratings yet
- Medical Instruments and Devices at Home: Prepared by Dr.P.VinuprithaDocument33 pagesMedical Instruments and Devices at Home: Prepared by Dr.P.VinuprithaAdityanair RA1711009010128No ratings yet
- GN 17 Guidance On Preparation of A Product Registration Submission For General Medical Devices Using The Asean CSDTDocument32 pagesGN 17 Guidance On Preparation of A Product Registration Submission For General Medical Devices Using The Asean CSDTShankar BNo ratings yet
- Guideline For Registration of Drug-Medical Device and Medical Device-Drug Combinations ProductsDocument33 pagesGuideline For Registration of Drug-Medical Device and Medical Device-Drug Combinations ProductsTZ LABNo ratings yet
- GN-13-R1.1 Guidance On The Risk Classification of General Medical DevicesDocument31 pagesGN-13-R1.1 Guidance On The Risk Classification of General Medical DevicesJoresp EspinozaNo ratings yet
- Are Healthcare Antiseptics Toxic To Staff and Patients?Document5 pagesAre Healthcare Antiseptics Toxic To Staff and Patients?aksinuNo ratings yet
- Inhaler Registration - Drug-MD GuidelineDocument33 pagesInhaler Registration - Drug-MD Guidelinetan_hoe_1No ratings yet
- GD Classification Draft1Document31 pagesGD Classification Draft1Suresh AnandNo ratings yet
- Pharmaceuticals and Medical Devices Safety Information 224: No. May 2006Document36 pagesPharmaceuticals and Medical Devices Safety Information 224: No. May 2006osama.khalifaNo ratings yet
- Guideline For MD RegisterationDocument36 pagesGuideline For MD RegisterationIsidoro OlveraNo ratings yet
- Policy On Medical PDT TranportationDocument4 pagesPolicy On Medical PDT Tranportationqariasif1900No ratings yet
- Health 6 Q4 Module 4. Final 1Document15 pagesHealth 6 Q4 Module 4. Final 1minminlubay13No ratings yet
- Unit 3 Industrial Pharmacy 2 7th SemesterDocument27 pagesUnit 3 Industrial Pharmacy 2 7th SemesterDurgha SureshNo ratings yet
- Welcome To Today's FDA/CDRH WebinarDocument29 pagesWelcome To Today's FDA/CDRH WebinarCris GalendezNo ratings yet
- Health Q1-Consumer HealthDocument55 pagesHealth Q1-Consumer HealthCharles Victor ConsultaNo ratings yet
- MDReg General Overview - 70Document70 pagesMDReg General Overview - 70sakiba2003No ratings yet
- Week 2-Health 10Document7 pagesWeek 2-Health 10lana del reyNo ratings yet
- Week 2-Health 10Document7 pagesWeek 2-Health 10lana del reyNo ratings yet
- 1 Presentation 120604 Drug 1600 Griffin (Compatibility Mode)Document28 pages1 Presentation 120604 Drug 1600 Griffin (Compatibility Mode)jmittalNo ratings yet
- FDA FAQs On AO 2015-003Document4 pagesFDA FAQs On AO 2015-003Jacques oltrade100% (2)
- Leitfaden Fuer App Entwickler Def ENDocument53 pagesLeitfaden Fuer App Entwickler Def ENYousra El MeriniNo ratings yet
- Medical Devices 2021Document67 pagesMedical Devices 2021Vighnesh Vikki100% (1)
- (MDS-G10) Ar, en - 1 LabelingDocument20 pages(MDS-G10) Ar, en - 1 LabelingAhmed AlyNo ratings yet
- FDA Presentation Sept. 20 2016Document141 pagesFDA Presentation Sept. 20 2016José GuapachaNo ratings yet
- Pharmaceutical TechnologyDocument32 pagesPharmaceutical TechnologyYonas TarekegnNo ratings yet
- Medical Device Design Using Polymer CompoundsDocument6 pagesMedical Device Design Using Polymer CompoundsAkash DoshiNo ratings yet
- Medical Device RegulationDocument6 pagesMedical Device RegulationAnonymous iqoU1mt100% (2)
- Branded Drugs Are Initially Marketed As New Chemical IntitiesDocument5 pagesBranded Drugs Are Initially Marketed As New Chemical IntitiesErwin CabangalNo ratings yet
- Embedded Sensor Systems in Medical Devices: Requisites and Challenges AheadDocument28 pagesEmbedded Sensor Systems in Medical Devices: Requisites and Challenges AheadAbdullah Bin MasoodNo ratings yet
- Eu MDRDocument10 pagesEu MDRumeshchandra24276No ratings yet
- Chapter 6 Part 4 Health Products (1) - 1-1Document56 pagesChapter 6 Part 4 Health Products (1) - 1-1Benedict Justine MagsinoNo ratings yet
- Regulatory Requirments of Tga & RowDocument29 pagesRegulatory Requirments of Tga & RowGautami UbhraniNo ratings yet
- Decree - 4725 - 2005 Label p-25Document34 pagesDecree - 4725 - 2005 Label p-25Manuela BernalNo ratings yet
- Biomedical Inst. Lec1 PDFDocument29 pagesBiomedical Inst. Lec1 PDFLari EvergardenNo ratings yet
- Covigilance Question BankDocument44 pagesCovigilance Question BankDrx Rinku KunduNo ratings yet
- Challenges and Issues Related To Regulation of Traditional MedicineDocument41 pagesChallenges and Issues Related To Regulation of Traditional MedicineKavish BhajbhujeNo ratings yet
- Regulatory GuidlineDocument88 pagesRegulatory GuidlineM Usman KhanNo ratings yet
- Weg Product-Literature Detox-Footbath V003Document8 pagesWeg Product-Literature Detox-Footbath V003cancerhipertermiaNo ratings yet
- Health 10 LM Month 2Document6 pagesHealth 10 LM Month 2Aubrey Rose Dela Peña - MagluyanNo ratings yet
- HTechDocument6 pagesHTechDelica AloNo ratings yet
- O8449v83 GRW 28024 H229 enGBDocument2 pagesO8449v83 GRW 28024 H229 enGBNunoNo ratings yet
- Lifting CarsDocument1 pageLifting CarsNunoNo ratings yet
- UX360DDocument5 pagesUX360DNunoNo ratings yet
- OnwardHP4 PassengerLithium IonOperatorManualDocument90 pagesOnwardHP4 PassengerLithium IonOperatorManualNuno100% (1)
- UN490DDocument8 pagesUN490DNunoNo ratings yet
- Outrigger Pads - TILBOXDocument62 pagesOutrigger Pads - TILBOXNunoNo ratings yet
- MS1 Med Instrument GuideDocument11 pagesMS1 Med Instrument GuideNunoNo ratings yet
- UX360DDocument5 pagesUX360DNunoNo ratings yet
- Gear Pumps and Motors Cast Iron Body - KAPPA 30 CHS (K30CHS 04 T E)Document124 pagesGear Pumps and Motors Cast Iron Body - KAPPA 30 CHS (K30CHS 04 T E)NunoNo ratings yet
- XCMG Qy50kDocument4 pagesXCMG Qy50kNunoNo ratings yet
- Outrigger Pads - TVHDocument8 pagesOutrigger Pads - TVHNunoNo ratings yet
- Engen Premium EP SeriesDocument2 pagesEngen Premium EP SeriesNunoNo ratings yet
- Mitas Con-Mh 2020-21 en LRDocument100 pagesMitas Con-Mh 2020-21 en LRNunoNo ratings yet
- zeppelin-ZT400 - Crawler-Cranes-SpecDocument4 pageszeppelin-ZT400 - Crawler-Cranes-SpecNunoNo ratings yet
- Luna All Terrain Cranes Spec B8e943Document8 pagesLuna All Terrain Cranes Spec B8e943NunoNo ratings yet
- RT100 Operation Manual-ND 21031217Document174 pagesRT100 Operation Manual-ND 21031217NunoNo ratings yet
- Sany SCC1800A 180tonDocument68 pagesSany SCC1800A 180tonNunoNo ratings yet
- Sany - SCC1000ACrawler CraneDocument32 pagesSany - SCC1000ACrawler CraneNunoNo ratings yet
- Hiab 022 T 1Document2 pagesHiab 022 T 1NunoNo ratings yet
- Sany - SCC8300 Crawler CraneDocument52 pagesSany - SCC8300 Crawler CraneNunoNo ratings yet
- Catálogo Equipamento para Barcos e NaviosDocument603 pagesCatálogo Equipamento para Barcos e NaviosNunoNo ratings yet
- Hiab 022 T 1Document2 pagesHiab 022 T 1NunoNo ratings yet
- Luna All Terrain Cranes Spec dc26f4Document2 pagesLuna All Terrain Cranes Spec dc26f4NunoNo ratings yet
- Hiab 035 2Document4 pagesHiab 035 2NunoNo ratings yet
- Hiab 035 2Document4 pagesHiab 035 2NunoNo ratings yet
- KTI20Document888 pagesKTI20NunoNo ratings yet
- Hiab 035 2Document4 pagesHiab 035 2NunoNo ratings yet
- Hiab 022 T 1Document2 pagesHiab 022 T 1NunoNo ratings yet
- Liebherr - LTR - 11200 DiagramaDocument60 pagesLiebherr - LTR - 11200 DiagramaNunoNo ratings yet
- Proportional Directional Valves Dhzo-Te Dkzor-Te: Direct Operated, With Position Transducer, ISO 4401 Size 06 and 10Document8 pagesProportional Directional Valves Dhzo-Te Dkzor-Te: Direct Operated, With Position Transducer, ISO 4401 Size 06 and 10NunoNo ratings yet
- Application Form For Accreditation SPDocument4 pagesApplication Form For Accreditation SPMyk Twentytwenty NBeyondNo ratings yet
- Hardy Et Al-2018-Australian and New Zealand Journal of Public HealthDocument5 pagesHardy Et Al-2018-Australian and New Zealand Journal of Public HealthAnggita RifkyNo ratings yet
- ED Produk KF - KF (Nama Apotek)Document19 pagesED Produk KF - KF (Nama Apotek)Eko FebryandiNo ratings yet
- 1823 - Part - A - DCHB - Kamrup Metropolitan PDFDocument224 pages1823 - Part - A - DCHB - Kamrup Metropolitan PDFSaddam FaruqueNo ratings yet
- A Seminar Report On Pharmacy ServicesDocument20 pagesA Seminar Report On Pharmacy Servicesrimjhim chauhanNo ratings yet
- Haad 3 Questions GiDocument16 pagesHaad 3 Questions GiPatpat De Guzman67% (3)
- CelecoxibDocument2 pagesCelecoxibXtinegoNo ratings yet
- BinderDocument13 pagesBinderisabel carreauNo ratings yet
- Foundations For Clinical Mental Health Counseling - 3rd EditionDocument369 pagesFoundations For Clinical Mental Health Counseling - 3rd EditionJamie Finotti100% (7)
- 9c5a4module 2 Self EsteemDocument14 pages9c5a4module 2 Self EsteemHarshita Jakhodia100% (1)
- Msds Material Safety Data Sheet For #2 Diesel: of TermsDocument7 pagesMsds Material Safety Data Sheet For #2 Diesel: of TermsAndrew PaulNo ratings yet
- Monkeypox: VirusDocument18 pagesMonkeypox: Virusdiela dwi lestariNo ratings yet
- Transport OshDocument260 pagesTransport OshAnlugosiNo ratings yet
- SoW - PPE - Attachment 5Document31 pagesSoW - PPE - Attachment 5أحمد خيرالدين عليNo ratings yet
- Forging An International MentorshipDocument3 pagesForging An International MentorshipAbu SumayyahNo ratings yet
- Schedule of Charges-Holy Family - Sep 08Document64 pagesSchedule of Charges-Holy Family - Sep 08Quirinevv100% (1)
- Segmentos Ica 2022 PDFDocument9 pagesSegmentos Ica 2022 PDFRoberto RodríguezNo ratings yet
- E The Real Facts Supporting Jeanne Calment As The Oldest Ever HumanDocument9 pagesE The Real Facts Supporting Jeanne Calment As The Oldest Ever Humanliz201177No ratings yet
- Literature Review On Voluntary Blood DonationDocument7 pagesLiterature Review On Voluntary Blood Donationakjnbowgf100% (1)
- Week 3 PATIENTS RIGHTS AND RESPONSIBILITIES 19-20Document32 pagesWeek 3 PATIENTS RIGHTS AND RESPONSIBILITIES 19-20maha abdallahNo ratings yet
- REACH - Substances of Very High ConcernDocument3 pagesREACH - Substances of Very High Concerna userNo ratings yet
- BTEC - Assignment - Brief A2 Unit 21 2Document3 pagesBTEC - Assignment - Brief A2 Unit 21 2Dariusz LangeNo ratings yet
- Philippine Health Care Delivery SystemDocument24 pagesPhilippine Health Care Delivery SystemoisloeNo ratings yet
- BJGPbookreview Justiceby SandelDocument2 pagesBJGPbookreview Justiceby SandelEm DeNo ratings yet
- Effects of Smoking and Alcohol On Complete Denture PatientsDocument2 pagesEffects of Smoking and Alcohol On Complete Denture PatientsAshwin ThejaswiNo ratings yet
- Basic Maternity ConceptsDocument14 pagesBasic Maternity ConceptsDivine Grace Arreglo AbingNo ratings yet
- LCSW Additional CourseworkDocument8 pagesLCSW Additional Courseworkiuhvgsvcf100% (2)
- Project PrimaryDocument7 pagesProject PrimaryLisanwork HonseboNo ratings yet
- Cancer-Relatedfatiguein Cancersurvivorship: Chidinma C. Ebede,, Yongchang Jang,, Carmen P. EscalanteDocument13 pagesCancer-Relatedfatiguein Cancersurvivorship: Chidinma C. Ebede,, Yongchang Jang,, Carmen P. EscalanteMahdhun ShiddiqNo ratings yet
- L 65 - Prevention of Fire and Explosion, and Emergency Response On Offshore Installations - Approved Code of Practice and Guidance - HSE - 2010Document56 pagesL 65 - Prevention of Fire and Explosion, and Emergency Response On Offshore Installations - Approved Code of Practice and Guidance - HSE - 2010Barkat UllahNo ratings yet