Professional Documents
Culture Documents
Seafood Hazards Guide Appendix 3 Decision Tree 06082021
Seafood Hazards Guide Appendix 3 Decision Tree 06082021
Uploaded by
addo ugOriginal Title
Copyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
Seafood Hazards Guide Appendix 3 Decision Tree 06082021
Seafood Hazards Guide Appendix 3 Decision Tree 06082021
Uploaded by
addo ugCopyright:
Available Formats
APPENDIX 3: CRITICAL CONTROL POINT DECISION TREE
This guidance represents the Food and Drug Administration’s (FDA’s) current thinking on this topic. It does not
create or confer any rights for or on any person and does not operate to bind FDA or the public. You can use an alternative
approach if the approach satisfies the requirements of the applicable statutes and regulations. If you want to discuss
an alternative approach, contact the FDA staff responsible for implementing this guidance. If you cannot identify the
appropriate FDA staff, call the telephone number listed on the title page of this guidance.
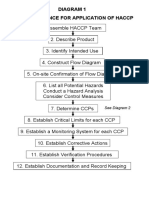
This appendix contains a decision tree that may be used to
assist you with the identification of critical control points
(CCPs). You should not rely exclusively on the decision
tree, because error may result.
The following decision tree is derived from one that
was developed by the National Advisory Committee on
Microbiological Criteria for Foods.
Appendix 3: Critical Control Point Decision
Tree A3 - 1 (June 2021)
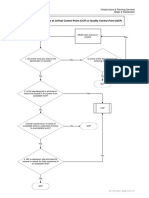
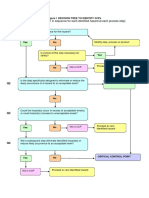
FIGURE A-2: CCP DECISION TREE
Q1: Does this step involve a hazard of sufficient risk and severity to warrant its control?
YES NO NOT A CCP
Q2: Does control measure for the hazard exist at this step?
Modify this step,
YES NO
process or product
Is control at this
step necessary for YES
Safety?
NO NOT A CCP STOP
Q3. Is control at this step necessary to prevent, eliminate or reduce the risk of the hazard to consumers?
YES NO NOT A CCP STOP
CCP
Appendix 3: Critical Control Point Decision
Tree A3 - 2 (June 2021)
BIBLIOGRAPHY
We have placed the following references on display in the Division of Dockets Management, Food and Drug Admin-
istration, 5630 Fishers Lane, rm. 1061, Rockville, MD 20852. You may see them at that location between 9 a.m. and 4
p.m., Monday through Friday. As of [Insert date], FDA had verified the Web site address for the references it makes
available as hyperlinks from the Internet copy of this guidance, but FDA is not responsible for any subsequent changes
to Non-FDA Web site references after [Insert date].
• National Advisory Committee on Microbiological Criteria for Foods. 1992. Hazard Analysis and Critical Control Point
System. Intl. J. Food Microbiol. 16:1-23.
Appendix 3: Critical Control Point Decision
Tree A3 - 3 (June 2021)
NOTES:
Appendix 3: Critical Control Point Decision
Tree A3 - 4 (June 2021)
You might also like
- QMS 140 Root Cause Analysis Investigation Procedure SampleDocument3 pagesQMS 140 Root Cause Analysis Investigation Procedure Samplex engineeringNo ratings yet
- Duck, Duck, Goose by Hank Shaw - RecipesDocument12 pagesDuck, Duck, Goose by Hank Shaw - RecipesThe Recipe Club50% (2)
- AnimalsDocument18 pagesAnimalsMaged MansourNo ratings yet
- Procedue For CapaDocument14 pagesProcedue For Capamariam adebisiNo ratings yet
- ISO 22000 Transition Training Powerpoint FINAL 02-20-19Document105 pagesISO 22000 Transition Training Powerpoint FINAL 02-20-19Anonymous XfIF3Hd86% (7)
- HACCP - Identification of CCPS, CPS, PRPS, OPRPs...Document5 pagesHACCP - Identification of CCPS, CPS, PRPS, OPRPs...cher28100% (1)
- PRO 8.2 Validation Procedure TemplatDocument6 pagesPRO 8.2 Validation Procedure TemplatSuresh SubbuNo ratings yet
- Coca Cola Michigan State CCP PDFDocument1 pageCoca Cola Michigan State CCP PDFOzlem MepNo ratings yet
- Corrective Action and Preventive Actions and Its Importance in Quality Management System: A ReviewDocument6 pagesCorrective Action and Preventive Actions and Its Importance in Quality Management System: A ReviewsachinNo ratings yet
- Determining Control MeasuresDocument17 pagesDetermining Control MeasuresGaganpreet KaurNo ratings yet
- Kitchen Essential HistoryDocument35 pagesKitchen Essential Historyeliseo peraltaNo ratings yet
- HMPE3 Culinary Nutrition SyllabusDocument8 pagesHMPE3 Culinary Nutrition SyllabusJaja Dondon67% (3)
- ABCQ Cqe - PDocument13 pagesABCQ Cqe - PAntonio Melgaço da Silva0% (2)
- Food Safety Management System: Asc Consultants (Pty) LTDDocument6 pagesFood Safety Management System: Asc Consultants (Pty) LTDMohammed Imran100% (2)
- Risk-Management-Sandle EsterilesDocument31 pagesRisk-Management-Sandle EsterilesLeyvert De JesusNo ratings yet
- ISO 22000 Audit Checklist ReportDocument37 pagesISO 22000 Audit Checklist ReportFlavones Extract100% (6)
- Jacobi Tds Colorsorb g7 A4 Eng A0214Document2 pagesJacobi Tds Colorsorb g7 A4 Eng A0214Alfonso GarcíaNo ratings yet
- Capa SopDocument14 pagesCapa SopWANDERA ROBERTNo ratings yet
- Moving From HACCP To HARPC - The Practical Steps To ComplianceDocument33 pagesMoving From HACCP To HARPC - The Practical Steps To ComplianceElvira100% (1)
- Haccp ReportDocument52 pagesHaccp Reportmaria dulceNo ratings yet
- Common Remedy For Cough and ColdDocument6 pagesCommon Remedy For Cough and Coldnavneet1107No ratings yet
- Case 14 - Maple Leaf Consumer Foods - Fixing Hot DogsDocument3 pagesCase 14 - Maple Leaf Consumer Foods - Fixing Hot DogsRoseAnnGatuzNicolas100% (1)
- Decision Tree Q1-Q5 Ver 2018Document2 pagesDecision Tree Q1-Q5 Ver 2018Sartika Mutiarasani100% (1)
- Auditing Beyond Compliance: Using the Portable Universal Quality Lean Audit ModelFrom EverandAuditing Beyond Compliance: Using the Portable Universal Quality Lean Audit ModelNo ratings yet
- Codex Decision TreeDocument4 pagesCodex Decision TreeEddy HanifaNo ratings yet
- Haccp 130119031201 Phpapp01Document30 pagesHaccp 130119031201 Phpapp01Maricel SanchezNo ratings yet
- Annex4 - Control Measure Decision TreeDocument1 pageAnnex4 - Control Measure Decision TreeTừ TuấnNo ratings yet
- H4 3a HazardAnalysis Soup ProductsDocument23 pagesH4 3a HazardAnalysis Soup ProductsAelindahNo ratings yet
- COCA COLA MICHIGAN STATE CCP-OPRP TreeDocument1 pageCOCA COLA MICHIGAN STATE CCP-OPRP TreeMuh Nur Afif LuthfiNo ratings yet
- Diagram 1 Logic Sequence For Application of HaccpDocument5 pagesDiagram 1 Logic Sequence For Application of HaccpSlamet ArNo ratings yet
- CRD03e Rev1Document5 pagesCRD03e Rev1mais.zaqdah.2002No ratings yet
- 6) FAAS 608 - Week 9 10Document83 pages6) FAAS 608 - Week 9 10Online NinaNo ratings yet
- ' (V.1) محضر سحب منتج نهائي' .1) محضر سحب منتج نهائيDocument4 pages' (V.1) محضر سحب منتج نهائي' .1) محضر سحب منتج نهائيmohamed reyadNo ratings yet
- Determination of Critical Control Point (CCP) or Quality Control Point (QCP)Document1 pageDetermination of Critical Control Point (CCP) or Quality Control Point (QCP)evil geniusNo ratings yet
- Act#09 Ocampo THC003Document2 pagesAct#09 Ocampo THC003jumarri ilardeNo ratings yet
- Principle 2 Identifying CCPsDocument10 pagesPrinciple 2 Identifying CCPsNel WenceslaoNo ratings yet
- Ehaccp - CCP Decision TreeDocument1 pageEhaccp - CCP Decision TreeEvinder AnejaNo ratings yet
- T09-MOTIVA Sample Continual Improvement ProcedureDocument9 pagesT09-MOTIVA Sample Continual Improvement ProcedurefarheenNo ratings yet
- Risk Analysis For The Inland Waterways Hygiene Code Food and FeedDocument16 pagesRisk Analysis For The Inland Waterways Hygiene Code Food and FeedEEder118No ratings yet
- Design_of_CCP_Monitoring_ProgramsDocument13 pagesDesign_of_CCP_Monitoring_Programssyed wovaizhNo ratings yet
- Policy and Standard Operating Procedure OTO 203 - Quality Management and Corrective Action Plans Attachment B - CAPA WorksheetDocument3 pagesPolicy and Standard Operating Procedure OTO 203 - Quality Management and Corrective Action Plans Attachment B - CAPA WorksheetRezi ZazadzeNo ratings yet
- Deviation Management - Why Have We Not Solved The Problem?: David W. HusmanDocument9 pagesDeviation Management - Why Have We Not Solved The Problem?: David W. HusmanVENKATNo ratings yet
- Principle2 CCPDocument24 pagesPrinciple2 CCPNitesh PanchalNo ratings yet
- Python Cheat SheetDocument16 pagesPython Cheat Sheetphamxtien3741No ratings yet
- 2 Determine Critical Control Points (CCPS)Document5 pages2 Determine Critical Control Points (CCPS)Andre MarsNo ratings yet
- The Basics of Healthcare Failure Mode and Effect AnalysisDocument89 pagesThe Basics of Healthcare Failure Mode and Effect AnalysisAnita Sriwaty PardedeNo ratings yet
- Dairy Grade A Voluntary HACCP - NCIMS HACCP Hazard Analysis WorksheetDocument2 pagesDairy Grade A Voluntary HACCP - NCIMS HACCP Hazard Analysis WorksheetSanjana NekkantiNo ratings yet
- 1 Reliability Growth Analysis OverviewDocument10 pages1 Reliability Growth Analysis OverviewFakhrul HidayatNo ratings yet
- Haccp d3 3 TMDocument10 pagesHaccp d3 3 TM2C50Serly SeptiyantiD3 GIZINo ratings yet
- Cap ADocument2 pagesCap AibikgibikNo ratings yet
- Determination of Preventive and Control Measures in Food SafetyDocument30 pagesDetermination of Preventive and Control Measures in Food SafetyMd Kamruzzaman MonirNo ratings yet
- Continued Process VerificationDocument19 pagesContinued Process VerificationMinh LeNo ratings yet
- Risk Management in Sterile EnvironmentsDocument30 pagesRisk Management in Sterile Environmentsdhanu_lagwankarNo ratings yet
- FMEA Managemen RisikoDocument10 pagesFMEA Managemen RisikonurNo ratings yet
- Answer Each Question in Sequence For Each Identified Hazard at Each Process StepDocument1 pageAnswer Each Question in Sequence For Each Identified Hazard at Each Process StepLaura Castellanos SuárezNo ratings yet
- HFMEAIntro PDFDocument10 pagesHFMEAIntro PDFSyamsul ArifinNo ratings yet
- CCP Determination Tree, 008Document1 pageCCP Determination Tree, 008sajid waqasNo ratings yet
- 10.2 Incident, Nonconformity and Corrective Action: Endix A - Comparison Table - ISO45001 Vs OHSAS18001Document1 page10.2 Incident, Nonconformity and Corrective Action: Endix A - Comparison Table - ISO45001 Vs OHSAS18001Anabela GibraltarNo ratings yet
- CAPA Overview Training 1686363145Document35 pagesCAPA Overview Training 1686363145Nurul FikriNo ratings yet
- Be SMARTDocument5 pagesBe SMARTYareni RosasNo ratings yet
- Food Safety Handbook - Part 2Document203 pagesFood Safety Handbook - Part 2Yasser MostafaNo ratings yet
- White Paper CPV Lets Foster QualityDocument7 pagesWhite Paper CPV Lets Foster QualitySrinivasaRaoNo ratings yet
- Corrective Action Project WorkDocument6 pagesCorrective Action Project WorkRizwan JavaidNo ratings yet
- Process Verification VsDocument6 pagesProcess Verification Vschemist_tmaNo ratings yet
- Continued Process Verification (CPV) Signal Responses in Biopharma - Pharmaceutical EngineeringDocument22 pagesContinued Process Verification (CPV) Signal Responses in Biopharma - Pharmaceutical EngineeringJohn PerezNo ratings yet
- The Vaccine IndustryDocument8 pagesThe Vaccine IndustryAlejandro MotoliníaNo ratings yet
- Published by BS Central: 515 2nd Ave. S - Glasgow, MT 59230 406-228-4558 - Fax: 406-228-4578Document6 pagesPublished by BS Central: 515 2nd Ave. S - Glasgow, MT 59230 406-228-4558 - Fax: 406-228-4578BS Central, Inc. "The Buzz"No ratings yet
- Objective of The RestaurantDocument5 pagesObjective of The RestaurantRaman JoshiNo ratings yet
- HC3 2.1 SITXFSA001 002 GDDocument65 pagesHC3 2.1 SITXFSA001 002 GDMmc MixNo ratings yet
- Jungle SurvivalDocument82 pagesJungle SurvivalArja ArentNo ratings yet
- 2 - Carbohydrate MetabolismDocument30 pages2 - Carbohydrate MetabolismcayyoanisNo ratings yet
- Ve Ingredients List Week 3Document1 pageVe Ingredients List Week 3migil66256No ratings yet
- Dietary Assessment Tools - Group 6Document20 pagesDietary Assessment Tools - Group 6Muhammad JavaidNo ratings yet
- L.P. Drinks MenuDocument9 pagesL.P. Drinks MenuMatthewKangNo ratings yet
- Properties of Matter - Grade 5 PPT W2Document45 pagesProperties of Matter - Grade 5 PPT W2carleen kaye lozanoNo ratings yet
- SQLDocument6 pagesSQLalokkumar012345100% (1)
- Design & Fabrication of A Motorized Maize Shelling Machine: Conference PaperDocument9 pagesDesign & Fabrication of A Motorized Maize Shelling Machine: Conference PaperGodwin UmoffiaNo ratings yet
- LSI Boston Accommodation OptionsDocument4 pagesLSI Boston Accommodation OptionsOpen Hearts Int'l EducationNo ratings yet
- Ultimate Chocolate Cupcakes With Ultimate Chocolate Cream Cheese FrostingDocument3 pagesUltimate Chocolate Cupcakes With Ultimate Chocolate Cream Cheese Frostingpstojanovska1670No ratings yet
- Of 39-Invitation For Seminar.Document3 pagesOf 39-Invitation For Seminar.gadirajuNo ratings yet
- Exploring Impact of Meal Planning and Preparation On The Eating Habits and Nutrition of 11-He B StudentsDocument43 pagesExploring Impact of Meal Planning and Preparation On The Eating Habits and Nutrition of 11-He B StudentsPELINA, Carlos Jade L.No ratings yet
- Dragon Boat Festival Conversation Topics Dialogs 136266Document12 pagesDragon Boat Festival Conversation Topics Dialogs 136266Sonja NedeljkovicNo ratings yet
- Semiotic Analysis of BillboardsDocument14 pagesSemiotic Analysis of Billboardsapi-375702257100% (1)
- Hyperbolic DietDocument9 pagesHyperbolic DietJuan Jacobo Giraldo RodriguezNo ratings yet
- Critical Reading - SaltDocument2 pagesCritical Reading - SaltAKSHATNo ratings yet
- Timun MasDocument7 pagesTimun MasarutemiaNo ratings yet
- Dairy Development in KarnatakaDocument31 pagesDairy Development in KarnatakaShivamShekharNo ratings yet
- Cooking With Herbs and Spices: "Variety Is The Spice of Life"Document16 pagesCooking With Herbs and Spices: "Variety Is The Spice of Life"Lila KarapostoliNo ratings yet
- First Voyage Around The WorldDocument6 pagesFirst Voyage Around The WorldMicko Alcala IINo ratings yet