Professional Documents
Culture Documents
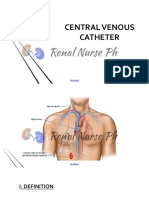
Central Venous Catheter (CVC) Product: RX Only
Central Venous Catheter (CVC) Product: RX Only
Uploaded by
ambitiousamit1Original Title
Copyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
Central Venous Catheter (CVC) Product: RX Only
Central Venous Catheter (CVC) Product: RX Only
Uploaded by
ambitiousamit1Copyright:
Available Formats
Central Venous Catheter (CVC) Product
Rx only.
Indications: 10. Air embolism can occur if air is allowed to enter a central
venous access device or vein. Do not leave open needles or
The Arrow CVC is intended to provide short-term (< 30 days) central venous access for
uncapped, unclamped catheters in central venous puncture
treatment of diseases or conditions requiring central venous access, including, but not
site. Use only securely tightened Luer-Lock connections
limited to the following: with any central venous access device to guard against
• Lack of usable peripheral IV sites inadvertent disconnection.
• Central venous pressure monitoring 11. Clinicians should be aware that slide clamps may be
• Total parenteral nutrition (TPN)
inadvertently removed.
• Infusions of fluids, medications, or chemotherapy
• Frequent blood sampling or receiving blood transfusions/blood products 12. Clinicians must be aware of complications associated with
central venous catheters including, but not limited to:
See additional labeling for product specific indications.
• cardiac tamponade • thrombosis
Contraindications: secondary to vessel, • inadvertent arterial
None known. See additional labeling for product specific contraindications. atrial, or ventricular
puncture
perforation
General Warnings and Cautions • pleural (i.e., • nerve injury
Warnings: pneumothorax) and • hematoma
mediastinal injuries • hemorrhage
1. Sterile, Single use: Do not reuse, reprocess or resterilize.
Reuse of device creates a potential risk of serious injury and/
• air embolism
• fibrin sheath formation
• catheter embolism
or infection which may lead to death.
• catheter occlusion
• exit site infection
2. Read all package insert warnings, precautions and • thoracic duct laceration • vessel erosion
instructions prior to use. Failure to do so may result in severe • bacteremia • catheter tip malposition
patient injury or death.
• septicemia • dysrhythmias
3. Do not place catheter into or allow it to remain in the right
atrium or right ventricle. X-ray exam or other method in Cautions:
compliance with institutional policies and procedures must 1. Do not alter the catheter, guidewire or any other kit/set
show catheter tip located in lower 1/3 of the Superior Vena component during insertion, use or removal.
Cava (SVC), in accordance with institutional guidelines. 2. Procedure must be performed by trained personnel well
versed in anatomical landmarks, safe technique and
4. Clinicians must be aware of potential entrapment of potential complications.
the guidewire by any implanted device in circulatory
3. Use standard precautions and follow established
system. It is recommended that if patient has a circulatory
institutional policies and procedures.
system implant, catheter procedure be done under direct
visualization to reduce risk of guidewire entrapment. 4. Some disinfectants used at catheter insertion site contain
solvents which can weaken the catheter material. Alcohol,
5. Do not use excessive force when introducing guidewire or acetone, and polyethylene glycol can weaken the structure
tissue dilator as this can lead to vessel perforation, bleeding, of polyurethane materials. These agents may also weaken
or component damage. the adhesive bond between catheter stabilization device
and skin.
6. Passage of guidewire into the right heart can cause
dysrhythmias, right bundle branch block, and a perforation • Do not use acetone on catheter surface.
of vessel, atrial or ventricular wall. • Do not use alcohol to soak catheter surface or allow
alcohol to dwell in a catheter lumen to restore catheter
7. Do not apply excessive force in placing or removing catheter
patency or as an infection prevention measure.
or guidewire. Excessive force can cause component damage
or breakage. If damage is suspected or withdrawal cannot be
• Do not use polyethylene glycol containing ointments at
insertion site.
easily accomplished, radiographic visualization should be
obtained and further consultation requested.
• Take care when infusing drugs with a high concentration
of alcohol.
8. Using catheters not indicated for high pressure injection • Allow insertion site to dry completely prior to applying
for such applications can result in inter-lumen crossover or dressing.
rupture with potential for injury. 5. Ensure catheter patency prior to use. Do not use syringes
9. Do not secure, staple and/or suture directly to outside smaller than 10 mL (a fluid filled 1 mL syringe can exceed 300
diameter of catheter body or extension lines to reduce risk of psi) to reduce risk of intraluminal leakage or catheter rupture.
cutting or damaging the catheter or impeding catheter flow. 6. Minimize catheter manipulation throughout procedure to
Secure only at indicated stabilization locations. maintain proper catheter tip position.
S15703111A1.indd 1 4/24/2015 10:19:47 AM
Kits/Sets may not contain all accessory components Arrow® Raulerson Syringe (where provided):
detailed in these instructions for use. Become familiar with Arrow Raulerson Syringe is used in conjunction with Arrow Advancer for guidewire insertion.
instructions for individual component(s) before beginning 9. Insert introducer needle or catheter/needle with attached syringe or Arrow Raulerson
the procedure. Syringe (where provided) into vein and aspirate.
Warning: Do not leave open needles or uncapped, unclamped catheters in
A Suggested Procedure: Use sterile technique. central venous puncture site. Air embolus can occur with these practices.
Prep Puncture Site: Caution: Do not reinsert needle into introducer catheter (where provided) to
reduce risk of catheter embolus.
1. Position patient as appropriate for insertion site.
• Subclavian or Jugular approach: Place patient in slight Trendelenburg position as Verify Venous Access:
tolerated to reduce risk of air embolism and enhance venous filling. Utilize one of the following techniques to verify venous access because of the potential for
• Femoral approach: Place patient in supine position. inadvertent arterial placement:
2. Prepare clean skin with an appropriate antiseptic agent. • Central Venous Waveform:
• Insert fluid primed blunt tip pressure transduction probe into rear of plunger
3. Drape puncture site.
and through valves of Arrow Raulerson Syringe and observe for central venous
4. Administer local anesthetic per institutional policies and procedures. pressure waveform.
5. Dispose of needle. ◊ Remove transduction probe if using Arrow Raulerson Syringe.
• Pulsatile Flow (if hemodynamic monitoring equipment is not available):
SharpsAway II™ Locking Disposal Cup (where provided): • Use transduction probe to open syringe valving system of Arrow Raulerson Syringe
The SharpsAway II Locking Disposal Cup is used for disposal of needles (15 Ga. - 30 Ga.). and observe for pulsatile flow.
• Disconnect syringe from needle and observe for pulsatile flow.
• Using one-handed technique, firmly push needles into disposal cup holes (refer to
Warning: Pulsatile flow is usually an indicator of inadvertent arterial puncture.
Figure 1).
Caution: Do not rely on blood aspirate color to indicate venous access.
Insert Guidewire:
Guidewire:
Kits/Sets are available with a variety of guidewires. Guidewires are provided in different
diameters, lengths and tip configurations for specific insertion techniques. Become familiar
with the guidewire(s) to be used with the specific technique before beginning the actual
insertion procedure.
Arrow Advancer (where provided):
Figure 1 Arrow Advancer is used to straighten “J” Tip of guidewire for introduction of the guidewire
• Once placed into disposal cup, needles will be automatically secured in place so that into Arrow Raulerson Syringe or a needle.
they cannot be reused. • Using thumb, retract “J” (refer to Figure 2).
Caution: Do not attempt to remove needles that have been placed into
SharpsAway II Locking Disposal Cup. These needles are secured in place. Damage
may occur to needles if they are forced out of disposal cup.
• Where provided, a foam SharpsAway® system may be utilized by pushing needles into
foam after use.
Caution: Do not re-use needles after they have been placed into the foam
SharpsAway system. Particulate matter may adhere to needle tip.
Figure 2
• Place tip of Arrow Advancer – with “J” retracted – into the hole in rear of Arrow
Prepare Catheter: Raulerson Syringe plunger or introducer needle (refer to Figure 3).
6. Flush each lumen with sterile normal saline for injection to establish patency and
prime lumen(s).
7. Clamp or attach Luer-Lock connector(s) to extension line(s) to contain saline within
lumen(s).
8. Leave distal extension line uncapped for guidewire passage.
Warning: Do not cut catheter to alter length.
Gain Initial Venous Access:
Figure 3
Echogenic Needle (where provided):
10. Advance guidewire into Arrow Raulerson Syringe approximately 10 cm until it passes
An echogenic needle is used to allow access to the vascular system for the introduction of a through syringe valves or into introducer needle.
guidewire to facilitate catheter placement. The needle tip is enhanced for approximately 1 cm • Advancement of guidewire through Arrow Raulerson Syringe may require a gentle
for clinician to identify exact needle tip location when puncturing the vessel under ultrasound. rotating motion.
11. Raise thumb and pull Arrow Advancer approximately 4 - 8 cm away from Arrow
Protected Needle/Safety Needle (where provided): Raulerson Syringe or introducer needle. Lower thumb onto Arrow Advancer and while
A protected needle/safety needle should be used in accordance with manufacturer’s maintaining a firm grip on guidewire, push assembly into syringe barrel to further
instructions for use. advance guidewire. Continue until guidewire reaches desired depth.
2
S15703111A1.indd 2 4/24/2015 10:19:48 AM
12. Use centimeter markings (where provided) on guidewire as a reference to assist in • If resistance is encountered, withdraw catheter relative to guidewire about 2-3 cm
determining how much guidewire has been inserted. and attempt to remove guidewire.
NOTE: When guidewire is used in conjunction with Arrow Raulerson Syringe (fully • If resistance is again encountered, remove guidewire and catheter simultaneously.
aspirated) and a 2-1/2” (6.35 cm) introducer needle, the following positioning Warning: Do not apply undue force on guidewire to reduce risk of possible
references can be made:
breakage.
• 20 cm mark (two bands) entering back of plunger = guidewire tip at end
of needle 21. Always verify entire guidewire is intact upon removal.
• 32 cm mark (three bands) entering back of plunger = guidewire tip
approximately 10 cm beyond end of needle Complete Catheter Insertion:
Caution: Maintain firm grip on guidewire at all times. Keep sufficient guidewire 22. Check lumen patency by attaching a syringe to each extension line and aspirate until
length exposed for handling purposes. A non-controlled guidewire can lead to free flow of venous blood is observed.
wire embolus. 23. Flush lumen(s) to completely clear blood from catheter.
Warning: Do not aspirate Arrow Raulerson Syringe while guidewire is in place; 24. Connect all extension line(s) to appropriate Luer-Lock connector(s) as required.
air may enter syringe through rear valve. Unused port(s) may be “locked” through Luer-Lock connector(s) using standard
Caution: Do not reinfuse blood to reduce risk of blood leakage from rear (cap) institutional policies and procedures.
of syringe. • Slide clamp(s) are provided on extension lines to occlude flow through each lumen
Warning: Do not withdraw guidewire against needle bevel to reduce risk of during line and Luer-Lock connector changes.
possible severing or damaging of guidewire. Warning: Open slide clamp prior to infusion through lumen to reduce risk of
13. Remove introducer needle and Arrow Raulerson Syringe (or catheter) while holding damage to extension line from excessive pressure.
guidewire in place.
14. Use centimeter markings on guidewire to adjust indwelling length according to Secure Catheter:
desired depth of indwelling catheter placement. 25. Use a catheter stabilization device, catheter clamp and fastener, staples or sutures
15. If necessary, enlarge cutaneous puncture site with cutting edge of scalpel, positioned (where provided).
away from guidewire. • Use triangular juncture hub with side wings as primary suture site.
Warning: Do not cut guidewire to alter length. • Use catheter clamp and fastener as a secondary suture site as necessary.
Warning: Do not cut guidewire with scalpel. Caution: Minimize catheter manipulation throughout procedure to maintain
• Position cutting edge of scalpel away from guidewire. proper catheter tip position.
• Engage safety and/or locking feature of scalpel (where provided) when not in
Catheter Stabilization Device (where provided):
use to reduce the risk of sharps injury.
A catheter stabilization device should be used in accordance with manufacturer’s
16. Use tissue dilator to enlarge tissue tract to the vein as required. Follow the angle of the
instructions for use.
guidewire slowly through the skin.
Warning: Do not leave tissue dilator in place as an indwelling catheter. Leaving Catheter Clamp and Fastener (where provided):
tissue dilator in place puts patient at risk for possible vessel wall perforation. A catheter clamp and fastener are used to secure catheter when an additional securement
site other than the catheter hub is required for catheter stabilization.
Advance Catheter: • After guidewire has been removed and necessary lines have been connected or locked,
17. Thread tip of catheter over guidewire. Sufficient guidewire length must remain spread wings of rubber clamp and position on catheter making sure catheter is not
exposed at hub end of catheter to maintain a firm grip on guidewire. moist, as required, to maintain proper tip location.
18. Grasping near skin, advance catheter into vein with slight twisting motion. • Snap rigid fastener onto catheter clamp.
Warning: Do not attach catheter clamp and fastener (where provided) until • Secure catheter clamp and fastener as a unit to patient by using either catheter
guidewire is removed. stabilization device, stapling or suturing. Both catheter clamp and fastener need to
be secured to reduce risk of catheter migration (refer to Figure 5).
19. Using centimeter marks on catheter as positioning reference points, advance catheter
to final indwelling position.
NOTE: Centimeter marking symbology is referenced from catheter tip.
• numerical: 5, 15, 25, etc.
• bands: each band denotes a 10 cm interval, with one band indicating 10 cm, two
bands indicating 20 cm, etc.
• dots: each dot denotes a 1 cm interval
20. Hold catheter at desired depth and remove guidewire.
Figure 5
Caution: If resistance is encountered when attempting to remove guidewire
after catheter placement, guidewire may be kinked around tip of catheter 26. Ensure insertion site is dry before applying dressing per manufacturer’s instructions.
within vessel (refer to Figure 4). 27. Assess catheter tip placement in compliance with institutional policies and procedures.
28. If catheter tip is malpositioned, assess and replace or reposition according to
institutional policies and procedures.
Care and Maintenance:
Dressing:
Figure 4 Dress according to institutional policies, procedures, and practice guidelines. Change
• In this circumstance, pulling back on guidewire may result in undue force being immediately if the integrity becomes compromised e.g. dressing becomes damp, soiled,
applied resulting in guidewire breakage. loosened or no longer occlusive.
3
S15703111A1.indd 3 4/24/2015 10:19:48 AM
Catheter Patency: 34. Apply direct pressure to site until hemostasis is achieved followed by an ointment-
Maintain catheter patency according to institutional policies, procedures and practice based occlusive dressing.
guidelines. All personnel who care for patients with central venous catheters must be
knowledgeable about effective management to prolong catheter’s dwell time and Warning: Residual catheter track remains an air entry point until site is
prevent injury. epithelialized. Occlusive dressing should remain in place for at least 24 hours or
until site appears epithelialized.
Catheter Removal Instructions:
29. Position patient as clinically indicated to reduce risk of potential air embolus. 35. Document catheter removal procedure including confirmation that entire catheter
30. Remove dressing. length and tip has been removed per institutional policies and procedures.
31. Release catheter and remove from catheter securement device(s).
32. Ask patient to take a breath and hold it if removing internal jugular or subclavian
catheter.
33. Remove catheter by slowly pulling it parallel to skin. If resistance is met while For reference literature concerning patient assessment, clinician education, insertion
removing catheter STOP
technique, and potential complications associated with this procedure, consult
Caution: Catheter should not be forcibly removed, doing so may result
standard textbooks, medical literature, and Arrow International, Inc. website:
in catheter breakage and embolization. Follow institutional policies and
procedures for difficult to remove catheter. www.teleflex.com
Symbol Glossary
Do not use Not made
Consult Do not Do not Sterilized by Keep away Keep
Caution if package with natural
instructions for use reuse resterilize ethylene oxide from sunlight dry
is damaged rubber latex
Catalogue Lot
Use by Manufacturer
number number
Arrow International, Inc.
Subsidiary of Teleflex Incorporated
2400 Bernville Road | Reading, PA 19605 USA
1-800-523-8446 | 1-610-378-0131
S-15703-111A, Rev. 1 (4/15)
S15703111A1.indd 4 4/24/2015 10:19:49 AM
You might also like
- Dissociative Experiences ScaleDocument3 pagesDissociative Experiences ScaleLucretia DespinoiuNo ratings yet
- How To Cure Almost Any CancerDocument49 pagesHow To Cure Almost Any CancerNikhil VelpulaNo ratings yet
- Gene Therapy HistoryDocument18 pagesGene Therapy HistorySharon Samuel50% (2)
- Advances in Tunneled CVCDsDocument12 pagesAdvances in Tunneled CVCDsroykelumendekNo ratings yet
- Port-A-Cath: Ayah Omairi Mrs. Boughdana JaberDocument13 pagesPort-A-Cath: Ayah Omairi Mrs. Boughdana Jaberayah omairi100% (1)
- Gordon's Functional Assessment HistoryDocument5 pagesGordon's Functional Assessment HistoryJachel Kathleen Laguio100% (1)
- The Perfect Neutropenic Diet Cookbook; The Complete Nutrition Guide To Reinstating Overall Health For General Wellness With Delectable And Nourishing RecipesFrom EverandThe Perfect Neutropenic Diet Cookbook; The Complete Nutrition Guide To Reinstating Overall Health For General Wellness With Delectable And Nourishing RecipesNo ratings yet
- COLLECTION OF BLOOD SPECIMENS MANUAL 2018-2020 - For Lab Guide PDFDocument27 pagesCOLLECTION OF BLOOD SPECIMENS MANUAL 2018-2020 - For Lab Guide PDFGonzalez ArturoNo ratings yet
- CanulationDocument21 pagesCanulationJason Liando100% (1)
- Cranial Ultrasound: DR Muhammad Jazib ShahidDocument43 pagesCranial Ultrasound: DR Muhammad Jazib ShahidJazib ShahidNo ratings yet
- Discharge Summary, Tabiano DeliaDocument3 pagesDischarge Summary, Tabiano DeliaGerald AndersonNo ratings yet
- Central Venous Catheters: Iv Terapy &Document71 pagesCentral Venous Catheters: Iv Terapy &Florence Liem0% (1)
- Central Venous Access Devices (Cvads)Document40 pagesCentral Venous Access Devices (Cvads)DocRNNo ratings yet
- HAI bUNDLESDocument54 pagesHAI bUNDLESNurhayati100% (1)
- Merit Microcatheter InstructionDocument2 pagesMerit Microcatheter InstructionnlongsisvnNo ratings yet
- 18 Sapphire II PRO IFU G-10-0252 Rev 04 EnglishDocument1 page18 Sapphire II PRO IFU G-10-0252 Rev 04 EnglishmanagervavilovaNo ratings yet
- CVC PDFDocument32 pagesCVC PDFEmerson Ocampo AlindoganNo ratings yet
- Groshong PICC and Catheters: Instructions For UseDocument20 pagesGroshong PICC and Catheters: Instructions For UseLucas FerreiraNo ratings yet
- CLABSI NewDocument41 pagesCLABSI NewyasmeenNo ratings yet
- 2009 Venous AccessDocument3 pages2009 Venous AccessDaniela MéndezNo ratings yet
- Central Venous Catheter: Insertion GuideDocument22 pagesCentral Venous Catheter: Insertion GuidePhil St Flour NPMT Central CoastNo ratings yet
- Guidelines 1.cannulation of Fistula and Grafts and Central Venous Catheters and Port Catheter SystemsDocument5 pagesGuidelines 1.cannulation of Fistula and Grafts and Central Venous Catheters and Port Catheter SystemsGrafe ChuaNo ratings yet
- Option X9585712Document2 pagesOption X9585712kohatian27796105No ratings yet
- Loss of A Guidewire: SciencedirectDocument3 pagesLoss of A Guidewire: SciencedirectSA SHNo ratings yet
- Recognizing, Preventing, and Troubleshooting Central-Line ComplicationsDocument7 pagesRecognizing, Preventing, and Troubleshooting Central-Line ComplicationsFauziaNo ratings yet
- CVC RemovalDocument5 pagesCVC RemovalRiza ZaharaNo ratings yet
- Central Venous Catheter Removal: Procedures and Rationale: LinicalDocument9 pagesCentral Venous Catheter Removal: Procedures and Rationale: LinicalVendiNo ratings yet
- Nursing Proses Diabetes ESRDDocument9 pagesNursing Proses Diabetes ESRDJamilah SudinNo ratings yet
- Central Venous Cannulation: Learning ObjectivesDocument2 pagesCentral Venous Cannulation: Learning ObjectivesRicardoNo ratings yet
- PMLS Lesson 5Document3 pagesPMLS Lesson 5Void MelromarcNo ratings yet
- 11 Caring For Patients Post PCI Emil 2019Document3 pages11 Caring For Patients Post PCI Emil 2019ameliajamirusNo ratings yet
- Guiding Catheter 2Document3 pagesGuiding Catheter 2Sara MartínezNo ratings yet
- Central Venous Catheters in Dialysis The Good TheDocument8 pagesCentral Venous Catheters in Dialysis The Good TheLaurențiu-Claudiu TAFLAN-TUDORACHENo ratings yet
- 012 Complications Associated With IVTherapyDocument5 pages012 Complications Associated With IVTherapyERIKA MAE SIROYNo ratings yet
- IV TherapyDocument9 pagesIV TherapyJackson Pukya Gabino PabloNo ratings yet
- Check List For Peripheral Venous Cannulation and Blood TransfusionDocument3 pagesCheck List For Peripheral Venous Cannulation and Blood Transfusionmaria hmNo ratings yet
- Catheter Ablation of Intramural Outflow Tract Premature Ventricular Complexes - A Multicentre StudyDocument9 pagesCatheter Ablation of Intramural Outflow Tract Premature Ventricular Complexes - A Multicentre Studyahmed mostafaNo ratings yet
- High Venous Pressure: 110.8 Venous Catheters: Problems and Measures in Nursing CareDocument8 pagesHigh Venous Pressure: 110.8 Venous Catheters: Problems and Measures in Nursing CareSherlaz EfhalNo ratings yet
- Activity 5 Blood Specimen Collection by Venipuncture: Safety PrecautionsDocument5 pagesActivity 5 Blood Specimen Collection by Venipuncture: Safety PrecautionsBadit GamutNo ratings yet
- IFU1062 Celosia Covered Stent Rev. BDocument11 pagesIFU1062 Celosia Covered Stent Rev. BAnnie NguyễnNo ratings yet
- External Jugular Vein Peripheral IntravenousDocument7 pagesExternal Jugular Vein Peripheral Intravenoushector nuñezNo ratings yet
- 2 CytoSorb DeviceInstructionsForUseDocument2 pages2 CytoSorb DeviceInstructionsForUseThomasHeblingerNo ratings yet
- Pmls2 MidtermDocument13 pagesPmls2 MidtermJainee Chen JavillonarNo ratings yet
- Pmls2 MidtermDocument13 pagesPmls2 MidtermJainee Chen JavillonarNo ratings yet
- Peripheral Intravenous Cannulation 2Document4 pagesPeripheral Intravenous Cannulation 2valecharapova11No ratings yet
- Catheter-Associated Urinary Tract Infection (CAUTI) : Prevention Diagnosis and ManagementDocument6 pagesCatheter-Associated Urinary Tract Infection (CAUTI) : Prevention Diagnosis and ManagementTamil SelvaNo ratings yet
- Tubing DevicesDocument13 pagesTubing DevicesAki YashinoNo ratings yet
- Central LinesDocument7 pagesCentral LineslaneeceshariNo ratings yet
- Bts ChestdrainDocument7 pagesBts ChestdrainSUMAN RIMALNo ratings yet
- Ins and Outs of Urinary Catheters: Indwelling Urethral Catheterisation (Short Term and Long Term)Document5 pagesIns and Outs of Urinary Catheters: Indwelling Urethral Catheterisation (Short Term and Long Term)Yudhi AuliaNo ratings yet
- CVP Line CareDocument37 pagesCVP Line CareArvindJoshiNo ratings yet
- Central Line Removal Policy JCUHDocument3 pagesCentral Line Removal Policy JCUHhellodrvigneshwarNo ratings yet
- Permanent Tunnel CatheterDocument52 pagesPermanent Tunnel CatheterJanice MuliadiNo ratings yet
- Managing Complications of Iv TherapyDocument48 pagesManaging Complications of Iv TherapyRandom Guy/GirlNo ratings yet
- 321971029397/virtualeducation/20122/anuncios/37790/CVC Femoral NEJM PDFDocument3 pages321971029397/virtualeducation/20122/anuncios/37790/CVC Femoral NEJM PDFvalentina cadena correaNo ratings yet
- FORMATDocument3 pagesFORMATjanekieannearlosNo ratings yet
- Central Lines AND Arterial LinesDocument41 pagesCentral Lines AND Arterial LinesSalinKaurNo ratings yet
- ACAreerSummer Fall2017Document4 pagesACAreerSummer Fall2017jelenaNo ratings yet
- Chapter 5Document17 pagesChapter 5Angelo Dell'AnnaNo ratings yet
- Practical Implications of Cytotoxic Drug AdministrDocument10 pagesPractical Implications of Cytotoxic Drug AdministrSundas EjazNo ratings yet
- Clinical Review - Full PDFDocument5 pagesClinical Review - Full PDFsitikhumaironiNo ratings yet
- Final 16-17Document5 pagesFinal 16-17Churrizo IslamiNo ratings yet
- Reocor D S User GuideDocument38 pagesReocor D S User GuideTuchilo ViorelNo ratings yet
- Clearway RX IfuDocument46 pagesClearway RX Ifubiomedical_com_brNo ratings yet
- NCM 112 Lesson5Document6 pagesNCM 112 Lesson5Trisha LopezNo ratings yet
- Arrow Central Venous SystemsDocument28 pagesArrow Central Venous Systemsambitiousamit1No ratings yet
- Ijrc 7 4Document10 pagesIjrc 7 4ambitiousamit1No ratings yet
- AnnalsATS 202003-233PSDocument4 pagesAnnalsATS 202003-233PSambitiousamit1No ratings yet
- ISA Public Awareness Flier 2020Document2 pagesISA Public Awareness Flier 2020ambitiousamit1No ratings yet
- IndianJMedRes1464459-4813122 132211 PDFDocument9 pagesIndianJMedRes1464459-4813122 132211 PDFambitiousamit1No ratings yet
- Alpha & PH StatDocument46 pagesAlpha & PH Statambitiousamit1No ratings yet
- Blood Bank Drawing 20-4-2016Document1 pageBlood Bank Drawing 20-4-2016ambitiousamit1No ratings yet
- TSX ULT Freezers - NorthAmerica - 0719 v2 PDFDocument9 pagesTSX ULT Freezers - NorthAmerica - 0719 v2 PDFambitiousamit1No ratings yet
- Cardiac ArrestDocument21 pagesCardiac Arrestashrf seadNo ratings yet
- Journal: Effects of Intrathecal Fentanyl On Quality of Spinal Anesthesia in Children Undergoing Inguinal Hernia RepairDocument7 pagesJournal: Effects of Intrathecal Fentanyl On Quality of Spinal Anesthesia in Children Undergoing Inguinal Hernia RepairputiridhaNo ratings yet
- The Many Health Benefits of SwimmingDocument1 pageThe Many Health Benefits of SwimmingRobert ReynoldsNo ratings yet
- Data Sarana PrasaranaDocument18 pagesData Sarana PrasaranaFlora Indah FajarwattyNo ratings yet
- Bridging The Care GapDocument50 pagesBridging The Care GapChetna AroraNo ratings yet
- Thesis Topics in PharmacyDocument13 pagesThesis Topics in Pharmacyromeofatima100% (1)
- LIDocument28 pagesLIErven VenturaNo ratings yet
- AneurysmsDocument18 pagesAneurysmsSaad Anwar GujjarNo ratings yet
- Bree Doran - ResumeDocument2 pagesBree Doran - Resumeapi-546694141No ratings yet
- en Colitis TuberculosisDocument7 pagesen Colitis TuberculosisATIKA INDAH SARINo ratings yet
- ReferensiDocument243 pagesReferensiyasminNo ratings yet
- Acuteleukemia: Hayley Rose-Inman,, Damon KuehlDocument18 pagesAcuteleukemia: Hayley Rose-Inman,, Damon KuehlJorge Tovar AvilaNo ratings yet
- Sari Clinical Care Training Learning Sequence 6 Oxygen TherapyDocument22 pagesSari Clinical Care Training Learning Sequence 6 Oxygen TherapyEstefania Cuta HernandezNo ratings yet
- Science 1stQ Lesson 3 - Importance of Your Senses and Medical Specialists For Sense OrgansDocument12 pagesScience 1stQ Lesson 3 - Importance of Your Senses and Medical Specialists For Sense OrgansVeronica RayosNo ratings yet
- Pediatric Case 6: Eva Madison (Complex) : Guided Reflection QuestionsDocument3 pagesPediatric Case 6: Eva Madison (Complex) : Guided Reflection QuestionsKathy ArenasNo ratings yet
- Ethical Issues in NeonateDocument11 pagesEthical Issues in Neonatethilaga880% (1)
- Anti-Infective AgentsDocument7 pagesAnti-Infective AgentsAlyssaGrandeMontimor100% (1)
- Icd 10 Penyakit Terbanyak VaskularDocument8 pagesIcd 10 Penyakit Terbanyak VaskularAlma WijayaNo ratings yet
- Arnold-Chiari Malformations Clinicl Features PDFDocument3 pagesArnold-Chiari Malformations Clinicl Features PDFsridharNo ratings yet
- Relapse & RecoveryDocument22 pagesRelapse & RecoveryRadeeka ShresthaNo ratings yet
- Pelod Vs Sofa Scoring in PediatricDocument6 pagesPelod Vs Sofa Scoring in PediatricAdrian KhomanNo ratings yet
- IPSG May28Document62 pagesIPSG May28S. Pichammal - Quality ManagerNo ratings yet
- 005 DeckingDocument3 pages005 DeckingMarvinNo ratings yet
- Bio Infertility Before Final PDFDocument31 pagesBio Infertility Before Final PDFmanojkumar200624No ratings yet