Professional Documents
Culture Documents
The Adrenergic Drugs
Uploaded by
Bareera Nudrat0 ratings0% found this document useful (0 votes)
8 views25 pagesOriginal Title
The Adrenergic Drugs (2)
Copyright
© © All Rights Reserved
Available Formats
PPTX, PDF, TXT or read online from Scribd
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
© All Rights Reserved
Available Formats
Download as PPTX, PDF, TXT or read online from Scribd
0 ratings0% found this document useful (0 votes)
8 views25 pagesThe Adrenergic Drugs
Uploaded by
Bareera NudratCopyright:
© All Rights Reserved
Available Formats
Download as PPTX, PDF, TXT or read online from Scribd
You are on page 1of 25
The adrenergic drugs
Affect receptors adrenergic receptor
Sympathomimetics Drugs
Drug that activate adrenergic receptors
Sympatholytic
Drugs that block the activation of
adrenergic receptors
Some sympathomimetics directly activate
adrenergic receptors (direct-acting
agonists)
Some indirectly by enhancing release or
blocking reuptake of norepinephrine
The Adrenergic Neuron
• Release norepinephrine as the primary
neurotransmitter
• Found in the central nervous system (CNS)
• Sympathetic nervous system
• Serve as links b/w ganglia and the effector
organs
• Adrenergic drugs act on adrenergic
receptors located either
• Presynaptically on the neuron
• Postsynaptically on the effector organ
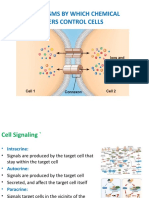
Neurotransmission at adrenergic neurons
• Norepinephrine is the neurotransmitter
• Neurotransmission involves:
• Synthesis
• Storage
• Release
• Receptor binding of norepinephrine
• Removal of the neurotransmitter from
the synaptic gap
Synthesis of norepinephrine:
• Tyrosine is transported by a carrier into
the adrenergic neuron
• Hydroxylated to dihydroxyphenylalanine
(DOPA) by tyrosine hydroxylase
• Rate-limiting step in the formation of
norepinephrine
• DOPA is then decarboxylated to
dopamine
• Aromatic I-amino acid decarboxylase
Storage of norepinephrine in vesicles:
• Dopamine is then transported into
synaptic vesicles
• An amine transporter system
• This carrier system is blocked by
reserpine
• Dopamine is next hydroxylated to form
norepinephrine
• Dopamine β-hydroxylase
Release of norepinephrine:
• An action potential arriving at the
nerve junction
• Triggers an influx of calcium ions into
the cytoplasm of the neuron
• Increase in calcium causes synaptic
vesicles to fuse with the cell
membrane
• Undergo exocytosis to expel their
contents into the synapse
• Guanethidine block this release
Binding to receptors:
• Norepinephrine released diffuses into
the synaptic space
• Binds to postsynaptic receptors on the
effector organ
• Presynaptic receptors on the nerve
ending
• Adrenergic receptors use both the
• Cyclic adenosine monophosphate
(cAMP) second messenger system
• Phosphatidylinositol cycle to transduce
the signal into an effect
Removal of norepinephrine
• Norepinephrine may
• 1) Diffuse out of the synaptic space and
enter the systemic circulation
• 2) be metabolized to inactive metabolites
by catechol-O-methyltransferase
• (COMT) in the synaptic space
• 3) Undergo reuptake back into the neuron
• The reuptake by the neuronal membrane
involves
• Sodium-chloride (Na+/Cl-)-dependent
norepinephrine transporter (NET)
• Can be inhibited by
• Tricyclic antidepressants (TCAs) such as
• Imipramine
• Serotonin–norepinephrine reuptake
inhibitors
• Duloxetine, or by cocaine
• Reuptake of
• norepinephrine into the presynaptic
neuron is the primary mechanism
• for termination of its effects.
Potential fates of recaptured norepinephrine:
• Once norepinephrine re-enters the
adrenergic neuron
• It may be taken up into synaptic vesicles
via the amine transporter system
• Sequestered for release by another action
potential
• Norepinephrine can be oxidized by
monoamine oxidase (MAO)
• Present in neuronal mitochondria
Direct-acting agents
• Albuterol β2
• Clonidine α2
• Dobutamine β1
• Dopamine doses, α1 doses, β1
• Epinephrine α1, α2, β1- and β2
• Formoterol
• Isoproterenol β1- and β2
• Norepinephrine
• Phenylephrine α1
• Salmeterol
• Terbutaline β2
• Fenoldopam
• Indirect-acting agents
• Amphetamine
• Cocaine
• Direct and indirect acting (mixed action)
agents
• Ephedrine
• Pseudoephedrine
B2 receptor
• This receptor is directly associated with one of its
ultimate effectors, the class C L-type calcium
channel Ca
• Receptor-channel complex is coupled to the Gs G
protein, which activates adenylyl cyclase
• Catalysing the formation of cyclic (cAMP)
• Which then activates protein kinase A
• Counterbalancing phosphatase
• Protein kinase A then goes on to phosphorylate
(and thus inactivate) myosin light-chain kinase,
which causes smooth muscle relaxation
Direct-acting adrenergic agonists
• Bind to adrenergic receptors on effector organs
• As a group, these agents are widely used
clinically
• Epinephrine
• Epinephrine is a catecholamines
• Commonly used in therapy
• Norepinephrine is methylated to yield
epinephrine in the adrenal medulla
• Adrenal medulla releases about 80%
epinephrine and 20% norepinephrine
• Epinephrine interacts with both α and β
receptors
Actions
• Cardiovascular:
• Strengthens the contractility of the
myocardium
• Increases its rate of contraction increase
cardiac output
• Increase oxygen demands on the
myocardium
• Activates β1 receptors on the kidney
release renin
• Renal blood flow is decreased
• Cumulative effect is an increase in systolic
blood pressure
• Respiratory:
• Epinephrine causes powerful
bronchodilation acting directly β2 action
• Inhibits the release of allergy mediators
such as histamines from mast cells
• Hyperglycemia:
• Epinephrine has a significant hyperglycemic
effect
• Increase glycogenolysis in the liver (β2
effect)
• Increased release of glucagon (β2 effect)
• Decreased release of insulin (α2 effect)
• Lipolysis:
• Epinephrine initiates lipolysis
• Increased levels of cAMP stimulate a
hormone-sensitive lipase
Therapeutic uses:
• Bronchospasm:
• Primary drug used in the emergency
treatment of bronchoconstriction
• Use in the treatment of acute asthma
• Anaphylactic shock
• Epinephrine is life saving in this setting
• Within a few minutes after s/c inj
respiratory function greatly improves
• Selective β2 agonist are favored in the
chronic treatment of asthma
• Anaphylactic shock:
• Drug of choice for the treatment of type-I
hypersensitivity reactions (including
anaphylaxis)
• Cardiac arrest:
• Epinephrine may be used to restore cardiac
rhythm in patients with cardiac arrest
• Anesthetics:
• Local anesthetic solutions may contain low
concentrations (for example, 1:100,000
parts) of epinephrine
• Greatly increases the duration of local
anesthesia
• Very weak solutions of epinephrine can
also be applied topically to vasoconstrict
mucous membranes and control oozing of
capillary blood.
Pharmacokinetics:
• Epinephrine has a rapid onset but a brief
duration of action (due to rapid degradation).
• Preferred route is intramuscular (anterior thigh)
due to rapid absorption
• In emergency intravenously (IV) for the most
rapid onset of action
• Given subcutaneously by endotracheal tube &
by inhalation
• It is rapidly metabolized by MAO and COMT
• Metabolites metanephrine and vanillylmandelic
acid are excreted in urine
Indirect-acting adrenergic agonists
• Indirect-acting adrenergic agonists cause the
• Release
• Inhibit the reuptake
• Inhibit the degradation of epinephrine or
norepinephrine
• Potentiate the effects of epinephrine or
norepinephrine
• Do not directly affect postsynaptic receptors
Amphetamine
• Marked central stimulatory action
• Often mistaken by drug abusers as its
only action
• Can also increase blood pressure
significantly by α1 agonist action on the
vasculature
• As well as β1-stimulatory effects on the
heart
• Increase in nonvascular release of
catecholamines
• Dopamine and norepinephrine from nerve
terminals
• Amphetamine is an indirect-acting
adrenergic drug
• Use as CNS stimulants of the CNS
Adverse effects:
• Adverse effects CNS include
• Anxiety, fear, tension, headache, and
tremor
• Trigger cardiac arrhythmias, particularly if
the patient is receiving digoxin
• Also induce pulmonary edema
• Enhanced cardiovascular actions in
patients with hyperthyroidism
You might also like
- Adrenergic AgonistsDocument74 pagesAdrenergic AgonistsBolanle Ojokoh100% (1)
- Adrenergic Drugs: Receptors, Agonists & AntagonistsDocument13 pagesAdrenergic Drugs: Receptors, Agonists & AntagonistsBareera NudratNo ratings yet
- Adrenergic and NonadrenergicDocument49 pagesAdrenergic and Nonadrenergicsweta sumanNo ratings yet
- Adrenergic Agonists and AntagonistsDocument41 pagesAdrenergic Agonists and Antagonistsclement simukokoNo ratings yet
- Adrenergic AgonistsDocument40 pagesAdrenergic AgonistsBenedict Brashi100% (1)
- Adrenergic and Nonadrenergic - 2Document44 pagesAdrenergic and Nonadrenergic - 2sweta sumanNo ratings yet
- Adrenergic Agonist and AntagonistDocument35 pagesAdrenergic Agonist and AntagonistFirjat AfiataNo ratings yet
- GFDGFDGFDGDocument11 pagesGFDGFDGFDGلو ترىNo ratings yet
- Autonomic Nervous System DrugsDocument111 pagesAutonomic Nervous System DrugsCres Padua QuinzonNo ratings yet
- Adrenergic Agonists: Classification and UsesDocument44 pagesAdrenergic Agonists: Classification and UsesAneeza AhmadNo ratings yet
- Adrenergic Agonist 22222Document64 pagesAdrenergic Agonist 22222Sifat DewanNo ratings yet
- Antiseizure AgentsDocument62 pagesAntiseizure Agentsakoeljames8543No ratings yet
- AnalgesicsDocument19 pagesAnalgesicsRogelio Junior RiveraNo ratings yet
- Adrenergic DrugsDocument50 pagesAdrenergic DrugscoleenNo ratings yet
- Ans Drugs Pharmacology.Document81 pagesAns Drugs Pharmacology.SAMUEL - NGUNJIRINo ratings yet
- Adrenergic Drug MNsDocument37 pagesAdrenergic Drug MNsTakale BuloNo ratings yet
- General AnestDocument35 pagesGeneral AnestMauz AshrafNo ratings yet
- General Anesth 1Document16 pagesGeneral Anesth 1api-3699361No ratings yet
- OpioidsDocument57 pagesOpioidsSteven Okta ChandraNo ratings yet
- SympathomimeticDocument71 pagesSympathomimeticahmad tariqNo ratings yet
- Adrenergic DrugsDocument20 pagesAdrenergic DrugsEdwardRoshanNo ratings yet
- Pharma Nca1 Notes MidtermDocument8 pagesPharma Nca1 Notes MidtermAICEL A. ABILNo ratings yet
- 5 Cholinergic AgonistDocument53 pages5 Cholinergic AgonistLoai Mohammed IssaNo ratings yet
- Chapter - 015 Adrenergic Agonists and AntagonistsDocument52 pagesChapter - 015 Adrenergic Agonists and AntagonistsMaureen CuartoNo ratings yet
- Pharmacology of Autonomic Nervous SystemDocument29 pagesPharmacology of Autonomic Nervous SystemTahir AzizNo ratings yet
- Adrenoceptor-activating and other sympathomimetic drugs: classification, mechanism of action, uses, and adverse effectsDocument43 pagesAdrenoceptor-activating and other sympathomimetic drugs: classification, mechanism of action, uses, and adverse effectsRupal RaghuwanshiNo ratings yet
- Cholinergic Drugs for Smoking Cessation & Alzheimer's DiseaseDocument7 pagesCholinergic Drugs for Smoking Cessation & Alzheimer's DiseaseAaron Lee100% (1)
- Central Nervous System Agents: Carmencita R. Pacis PHD, ManDocument52 pagesCentral Nervous System Agents: Carmencita R. Pacis PHD, ManTWINNY JOSSANNE CHEERISHANE MINIONNo ratings yet
- Cns Trans & Drug DetoxificationDocument57 pagesCns Trans & Drug DetoxificationAlvin LaurenceNo ratings yet
- Inotropes and VasopressorsDocument56 pagesInotropes and VasopressorsMuhammad Ali Yousuf100% (6)
- Toxicology: by Group 4 2018/2019 Tan Geok Eng Reena DewiDocument59 pagesToxicology: by Group 4 2018/2019 Tan Geok Eng Reena DewiTan Geok EngNo ratings yet
- General Anesthesia ExplainedDocument26 pagesGeneral Anesthesia ExplainedAlphahin 17No ratings yet
- Chapter 4: PsychopharmacologyDocument37 pagesChapter 4: PsychopharmacologyGummyCola100% (2)
- Adrenrgic AgonistsDocument35 pagesAdrenrgic Agonistssamar yousif mohamedNo ratings yet
- Nursing Sympathomimetics and SympatholyticsDocument87 pagesNursing Sympathomimetics and SympatholyticsFrancis PeterosNo ratings yet
- Neurotransmitters 2023Document41 pagesNeurotransmitters 2023Nuhu SibaNo ratings yet
- Sympathetic Agonist and Antagonist 23Document12 pagesSympathetic Agonist and Antagonist 23PNo ratings yet
- Anti Depressant Drug LatestDocument65 pagesAnti Depressant Drug LatestAnonymous zOMkw9100% (1)
- AnalgesicsDocument36 pagesAnalgesicsTakale BuloNo ratings yet
- Book ReadingDocument37 pagesBook Reading東廣謀No ratings yet
- FAR 381 - Opioids - 2018 - Lecture UploadDocument36 pagesFAR 381 - Opioids - 2018 - Lecture UploadHuzaifa KhanNo ratings yet
- Sympathomimetic Drugs: Dr. Shazana RanaDocument78 pagesSympathomimetic Drugs: Dr. Shazana Ranaمحمد رافع تنویر100% (1)
- Adrenergic Drugs 20.07.018 PDFDocument59 pagesAdrenergic Drugs 20.07.018 PDFHitesh karnNo ratings yet
- 4 Adrenergic and Anti-Adrenergic DrugsDocument51 pages4 Adrenergic and Anti-Adrenergic DrugsLoai Mohammed IssaNo ratings yet
- Adrenergic Drugs 20.06.018 PDFDocument55 pagesAdrenergic Drugs 20.06.018 PDFHitesh karnNo ratings yet
- Anesthetic Agents Ga/La: Rabindra AdhikaryDocument40 pagesAnesthetic Agents Ga/La: Rabindra AdhikaryAnaliza Kitongan LantayanNo ratings yet
- NarcoticsDocument6 pagesNarcoticsanaeshkl100% (1)
- Pharmacology Local AnestheticsDocument60 pagesPharmacology Local Anestheticsalianaeem839No ratings yet
- Adrenal GlandDocument49 pagesAdrenal GlandRaphael AnajeNo ratings yet
- Farmakologi ICUDocument54 pagesFarmakologi ICULin Phoponk100% (2)
- General Anesthetic AgentsDocument151 pagesGeneral Anesthetic AgentsArwa QishtaNo ratings yet
- Ans For PCL 301Document26 pagesAns For PCL 301olowoyoanjolaNo ratings yet
- L9 Sympathomimetics Lytics REVISED 2017 PDFDocument49 pagesL9 Sympathomimetics Lytics REVISED 2017 PDFVea AngelesNo ratings yet
- Pharmacology of Vasopressors and InotropesDocument56 pagesPharmacology of Vasopressors and InotropesBalemlay HailuNo ratings yet
- Fentanyl & CongenersDocument30 pagesFentanyl & Congenersnacimay21No ratings yet
- Drugs Acting On The Nervous SystemDocument123 pagesDrugs Acting On The Nervous SystemIretiola AdeleruNo ratings yet
- Drugs Acting On The Nervous SystemDocument123 pagesDrugs Acting On The Nervous SystemIretiola Adeleru100% (1)
- Vasoactive Drugs (PICUCOURSE)Document52 pagesVasoactive Drugs (PICUCOURSE)surasuarezlopezNo ratings yet
- TerminologyDocument14 pagesTerminologyBareera NudratNo ratings yet
- Volume of DistributionDocument9 pagesVolume of DistributionBareera NudratNo ratings yet
- Plasma Half LifeDocument7 pagesPlasma Half LifeBareera NudratNo ratings yet
- ReceptorsDocument31 pagesReceptorsBareera NudratNo ratings yet
- Facotors Affecting Drug AbsorptionDocument5 pagesFacotors Affecting Drug AbsorptionBareera NudratNo ratings yet
- IntroductionDocument13 pagesIntroductionBareera NudratNo ratings yet
- Top Drug Target Proteins: GPCRs, Enzymes, Ion ChannelsDocument6 pagesTop Drug Target Proteins: GPCRs, Enzymes, Ion ChannelsBareera NudratNo ratings yet
- Drug Distribution & MetabolismDocument23 pagesDrug Distribution & MetabolismBareera NudratNo ratings yet
- Factors affecting the rate of diffusionDocument24 pagesFactors affecting the rate of diffusionBareera NudratNo ratings yet
- Drug Absorption Across Plasma MemDocument8 pagesDrug Absorption Across Plasma MemBareera NudratNo ratings yet
- PoliomyelitisDocument46 pagesPoliomyelitisBareera Nudrat100% (1)
- Drug Metabolism and Elimination PathwaysDocument14 pagesDrug Metabolism and Elimination PathwaysBareera NudratNo ratings yet
- Drug ActionDocument20 pagesDrug ActionBareera NudratNo ratings yet
- Bio A ViabilityDocument8 pagesBio A ViabilityBareera NudratNo ratings yet
- Everything You Need to Know About Constipation Causes and TreatmentsDocument30 pagesEverything You Need to Know About Constipation Causes and TreatmentsBareera NudratNo ratings yet
- Crisis and Critique On Preparedness, Authoritarianism and The Regulatory StateDocument11 pagesCrisis and Critique On Preparedness, Authoritarianism and The Regulatory StateNikos KarfakisNo ratings yet
- Iso PDFDocument14 pagesIso PDFZoran BubinskiNo ratings yet
- SELF-EFFICACY THEORY SOURCESDocument14 pagesSELF-EFFICACY THEORY SOURCESagungNo ratings yet
- PepsiCo Prj-Recruitment SelectionDocument70 pagesPepsiCo Prj-Recruitment Selectionnarsimhamusunuri33% (3)
- Risk and Associated Factors of Depression and AnxiDocument27 pagesRisk and Associated Factors of Depression and AnxiDian Oktaria SafitriNo ratings yet
- Cara Baca SWISSADME-TargetPredDocument38 pagesCara Baca SWISSADME-TargetPredVictor DelacroixNo ratings yet
- Dr. Nirali Patel Pediatric Emergency MedicineDocument31 pagesDr. Nirali Patel Pediatric Emergency Medicinemarsan12No ratings yet
- Sop 105 Manual HandlingDocument3 pagesSop 105 Manual HandlingRobiyyana0% (1)
- Pelvic Organ Prolapse Causes and TreatmentDocument23 pagesPelvic Organ Prolapse Causes and TreatmentAbdullah Matar100% (2)
- Young Mothers' HIV/AIDS Prevention Knowledge, Attitudes, and PracticesDocument11 pagesYoung Mothers' HIV/AIDS Prevention Knowledge, Attitudes, and PracticesSITI MONAUWARAH BT MOKHTARNo ratings yet
- Government SchemesDocument86 pagesGovernment Schemesanon_462060725No ratings yet
- The Deteriorating PatientDocument29 pagesThe Deteriorating PatientvarymacNo ratings yet
- Guardian Angel - Cara MaloneDocument177 pagesGuardian Angel - Cara MalonenoNo ratings yet
- Dr. Bondoc Found Liable for Grave Misconduct in Delivery CaseDocument3 pagesDr. Bondoc Found Liable for Grave Misconduct in Delivery CaseSalie VillafloresNo ratings yet
- Taking A History of DEPRESSIONDocument3 pagesTaking A History of DEPRESSIONPrarthana Thiagarajan100% (13)
- A Textbook of Clinical Pharmacology and TherapeuticsDocument476 pagesA Textbook of Clinical Pharmacology and TherapeuticsAli Hassan100% (1)
- The Workshop Powerpoint For The Therapy of Infidelity CrisesDocument16 pagesThe Workshop Powerpoint For The Therapy of Infidelity Crisesclaudiaghergari100% (1)
- Consanguinity Tulyagotriya Vivaha Impact On Child Health A Review LiteratureDocument4 pagesConsanguinity Tulyagotriya Vivaha Impact On Child Health A Review LiteratureEditor IJTSRDNo ratings yet
- Pre AnestesiDocument64 pagesPre AnestesiKevin YonathanNo ratings yet
- Hearfulness Consciousness and Cognition 2020Document10 pagesHearfulness Consciousness and Cognition 2020Hugo MagalhãesNo ratings yet
- Bacabac Vs NYK-FIL Ship Management, G.R. No. 228550, July 28, 2021Document3 pagesBacabac Vs NYK-FIL Ship Management, G.R. No. 228550, July 28, 2021Gi NoNo ratings yet
- Pediatric Physical Examination An Illustrated Handbook 3rd Edition 2019Document758 pagesPediatric Physical Examination An Illustrated Handbook 3rd Edition 2019Anonymous n0uVxZm0W87% (23)
- Emergency Supply QRG PDFDocument3 pagesEmergency Supply QRG PDFOmar AbdulahNo ratings yet
- Self-Assessment of Nursing Standards of PracticeDocument32 pagesSelf-Assessment of Nursing Standards of Practiceapi-242114301No ratings yet
- Stress ManagementDocument54 pagesStress ManagementjomelofillartosNo ratings yet
- Patton, C. Sanchez-Eppler, B. (Editors) (2000) - Queer DiasporasDocument317 pagesPatton, C. Sanchez-Eppler, B. (Editors) (2000) - Queer Diasporastutkjhf0% (1)
- 30.plDocument54 pages30.plRobert GarciaNo ratings yet
- Surfactantreplacementtherapy 160926164838Document33 pagesSurfactantreplacementtherapy 160926164838revathidadam55555No ratings yet
- Rectification of Met Material by Roll FormingDocument8 pagesRectification of Met Material by Roll Formingolegprikhodko2809No ratings yet
- MDR 040501991289Document1 pageMDR 040501991289REZEL JOHN MAYRENANo ratings yet