Professional Documents
Culture Documents
Levet I Race Tam
Uploaded by
David Torres0 ratings0% found this document useful (0 votes)
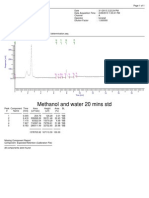
5 views1 pageThis document is a report summarizing the analysis of a sample named Leveti racetam_Di sso_STD. It provides information about the sample name, acquisition method, injection volume, processing channel, and run time. The report details that the sample contained one peak identified as Levetiracetam, which eluted at 5.957 minutes, accounted for 100% of the total area, and met method validation criteria for plate count and tailing.
Original Description:
Original Title
Levet i Race Tam
Copyright
© © All Rights Reserved
Available Formats
PDF, TXT or read online from Scribd
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentThis document is a report summarizing the analysis of a sample named Leveti racetam_Di sso_STD. It provides information about the sample name, acquisition method, injection volume, processing channel, and run time. The report details that the sample contained one peak identified as Levetiracetam, which eluted at 5.957 minutes, accounted for 100% of the total area, and met method validation criteria for plate count and tailing.
Copyright:
© All Rights Reserved
Available Formats
Download as PDF, TXT or read online from Scribd
0 ratings0% found this document useful (0 votes)
5 views1 pageLevet I Race Tam
Uploaded by
David TorresThis document is a report summarizing the analysis of a sample named Leveti racetam_Di sso_STD. It provides information about the sample name, acquisition method, injection volume, processing channel, and run time. The report details that the sample contained one peak identified as Levetiracetam, which eluted at 5.957 minutes, accounted for 100% of the total area, and met method validation criteria for plate count and tailing.
Copyright:
© All Rights Reserved
Available Formats
Download as PDF, TXT or read online from Scribd
You are on page 1of 1
Disso_Assay_SST
Reported by User: General (General )
Proj ect Name: Master_JUN_2014\PARC_ARD_002
17-07-2014
Date Pri nted: 18:51:20 Asi a/Cal cutta
Page: 1 of 1
S A M P L E I N F O R M A T I O N
Leveti racetam_Di sso_STD General Sampl e Name:
Acqui red By:
Leveti racetam_28062014
Sampl e Set Name: 3 Vi al :
Acq. Method Set: Leveti racetam_Mth 1
Inj ecti on #:
Leveti racetam_proc
Processi ng Method:
5.00 ul Inj ecti on Vol ume:
W2489 ChA 205nm
Proc. Chnl . Descr.: Run Ti me: 10.0 Mi nutes
Sampl eName: Leveti racetam_Di sso_STD; Vi al : 3; Inj ecti on: 1; Channel : W2489 ChA; Date
Acqui red: 28-06-2014 12:09:13 IST
5
.
9
5
7
1
Name Injection
Area
(V*sec)
Retention Time
(min)
% Area USP Plate Count USP Tailing Int Type
Levetiracetam 1 3154220 5.957 100.00 9032 1.54 BB
Component Summary Table
You might also like
- Thermal Power Plant: Pre-Operational ActivitiesFrom EverandThermal Power Plant: Pre-Operational ActivitiesRating: 4 out of 5 stars4/5 (4)
- Control Strategy for Time-Delay Systems: Part II: Engineering ApplicationsFrom EverandControl Strategy for Time-Delay Systems: Part II: Engineering ApplicationsMohammad-Hassan KhoobanNo ratings yet
- Peak Summarv: Yial:2 Vial:4Document2 pagesPeak Summarv: Yial:2 Vial:4Wahyuni S FitriNo ratings yet
- General Procedure For AUTDocument19 pagesGeneral Procedure For AUTalinor_tn100% (2)
- BP-HZN - BLYOO349O94: Exhibit No. - Worldwide Court IncDocument5 pagesBP-HZN - BLYOO349O94: Exhibit No. - Worldwide Court IncOSDocs2012No ratings yet
- UO LabDocument1 pageUO LabTyrus TaiNo ratings yet
- VWD1 A, Wavelength 233 NM (AUG2017T2FH0005.D)Document1 pageVWD1 A, Wavelength 233 NM (AUG2017T2FH0005.D)Nataly Grey Montoya GutierrezNo ratings yet
- Manzate WP 066-17-18000Document1 pageManzate WP 066-17-18000Nataly Montoya GutierrezNo ratings yet
- Project HPLC Report: Reported by User: System Page: 1 of 1 Project Name: Imipramine - Diazepam - AshokDocument10 pagesProject HPLC Report: Reported by User: System Page: 1 of 1 Project Name: Imipramine - Diazepam - AshokAshok KumarNo ratings yet
- 2014 08 26-LWBP-1kapasitorDocument93 pages2014 08 26-LWBP-1kapasitorAdnanValentinovNo ratings yet
- H2O - Methanol DistillationDocument1 pageH2O - Methanol DistillationTyrus TaiNo ratings yet
- Instrument Method: PARACETAMOL 2022: UntitledDocument3 pagesInstrument Method: PARACETAMOL 2022: UntitledRubén RuelasNo ratings yet
- Job Information: Engineer Checked ApprovedDocument3 pagesJob Information: Engineer Checked ApprovedRantharu AttanayakeNo ratings yet
- 2014 08 26-LWBP-6kapasitorDocument93 pages2014 08 26-LWBP-6kapasitorAdnanValentinovNo ratings yet
- 2014 08 26-LWBP-5kapasitorDocument93 pages2014 08 26-LWBP-5kapasitorAdnanValentinovNo ratings yet
- F6 Group8Document1 pageF6 Group8Tyrus TaiNo ratings yet
- 48mm - Neg ControlDocument1 page48mm - Neg ControlShaheen GulamaniNo ratings yet
- 144 Report For XRD OnDocument11 pages144 Report For XRD OngobinathdpiNo ratings yet
- TP TPD Pfs TPC QP 12Document29 pagesTP TPD Pfs TPC QP 12erbvp22No ratings yet
- Transformer DifferentialDocument39 pagesTransformer DifferentialcskathirsNo ratings yet
- 6mm - Neg ControlDocument1 page6mm - Neg ControlShaheen GulamaniNo ratings yet
- Report Generated by Test Manager: Title: CMP Author: Doqualkit Date: 28-May-2015 14:23:39Document11 pagesReport Generated by Test Manager: Title: CMP Author: Doqualkit Date: 28-May-2015 14:23:39Janos KovacsNo ratings yet
- Kel 74 - Sampel 3Document1 pageKel 74 - Sampel 3Zainal JufriNo ratings yet
- Alp DRG Rindang 12 Nov 2014.exp 11/13/14-03:12:23 1/14Document14 pagesAlp DRG Rindang 12 Nov 2014.exp 11/13/14-03:12:23 1/14Dadan RamadhanNo ratings yet
- Report Generated by Test Manager: Title: CMP Author: Doqualkit Date: 28-May-2015 14:23:39Document11 pagesReport Generated by Test Manager: Title: CMP Author: Doqualkit Date: 28-May-2015 14:23:39Janos KovacsNo ratings yet
- Report Generated by Test Manager: Title: CMP Author: Doqualkit Date: 28-May-2015 14:23:39Document11 pagesReport Generated by Test Manager: Title: CMP Author: Doqualkit Date: 28-May-2015 14:23:39Janos KovacsNo ratings yet
- GC ValidationDocument8 pagesGC ValidationShashikant DrShashikant BagadeNo ratings yet
- H2O - Methanol DistillationDocument1 pageH2O - Methanol DistillationTyrus TaiNo ratings yet
- Final CTRL-2 ReportDocument26 pagesFinal CTRL-2 ReportKongWeiHernNo ratings yet
- Transformer DifferentialDocument39 pagesTransformer Differentialerbvp22No ratings yet
- 48 MM - 5PPM 1Document1 page48 MM - 5PPM 1Shaheen GulamaniNo ratings yet
- General Information: Zte - PTP - Synce - Test - CSRDocument4 pagesGeneral Information: Zte - PTP - Synce - Test - CSRAsela BamunuarachchiNo ratings yet
- Lab Cromatografia GaseosaDocument1 pageLab Cromatografia GaseosaCarolina RoblesNo ratings yet
- RF Test Report 1851136 PDFDocument30 pagesRF Test Report 1851136 PDFlucas barriosNo ratings yet
- Basics of HAZOPDocument26 pagesBasics of HAZOPNg JoshuaNo ratings yet
- FIT741177Document8 pagesFIT741177Carlos AndrésNo ratings yet
- H2O - Methanol DistillationDocument1 pageH2O - Methanol DistillationTyrus TaiNo ratings yet
- PV Elite ResultDocument18 pagesPV Elite ResultVeny MartianiNo ratings yet
- Algeria 1 GTFT Gas Plant 19-02-2012: Load Flow AnalysisDocument23 pagesAlgeria 1 GTFT Gas Plant 19-02-2012: Load Flow Analysisnksuthar5399No ratings yet
- 2mM - 25PPM 1Document1 page2mM - 25PPM 1Shaheen GulamaniNo ratings yet
- XTRACT Analysis Report - : Section DetailsDocument1 pageXTRACT Analysis Report - : Section DetailsMadalina CirstianNo ratings yet
- System IQ ReportDocument3 pagesSystem IQ ReportRiad ManamanniNo ratings yet
- GC Fid SopDocument6 pagesGC Fid SopHamidReza ShahbaaziNo ratings yet
- UT DocumentationDocument3 pagesUT DocumentationMatthew BallardNo ratings yet
- Report Generated by Test ManagerDocument36 pagesReport Generated by Test ManagerJanos KovacsNo ratings yet
- Daunorubicin Working Standard Validation: Biolyse PharmaDocument1 pageDaunorubicin Working Standard Validation: Biolyse PharmaNishit PatelNo ratings yet
- EMC Test Procedure V2 0Document18 pagesEMC Test Procedure V2 0Gaurav AroraNo ratings yet
- Durham E-Theses: Investigation Into The Use of Zero Angle Ultrasonic Probe Array For Defect Detection and LocationDocument191 pagesDurham E-Theses: Investigation Into The Use of Zero Angle Ultrasonic Probe Array For Defect Detection and LocationRakesh SrivastavaNo ratings yet
- Subject: Returned Damaged Detector To DRTECH For ReparationDocument1 pageSubject: Returned Damaged Detector To DRTECH For ReparationMed MahmoudNo ratings yet
- 2mm - Neg ControlDocument1 page2mm - Neg ControlShaheen GulamaniNo ratings yet
- EMC31143B-NEBS Draft 5 PDFDocument86 pagesEMC31143B-NEBS Draft 5 PDFIsrael VazquezNo ratings yet
- A Sample: Ultrasonic Inspection ProcedureDocument5 pagesA Sample: Ultrasonic Inspection ProcedureMark BrailsfordNo ratings yet
- Us Navy Practical Guide Flame Bending PipeDocument102 pagesUs Navy Practical Guide Flame Bending PipeBrian DohertyNo ratings yet
- Test Report For Lab Iso 17025Document6 pagesTest Report For Lab Iso 17025vines7831No ratings yet
- H2O - Methanol DistillationDocument1 pageH2O - Methanol DistillationTyrus TaiNo ratings yet
- Ulllted States Patent (10) Patent N0.: US 8,019,846 B2Document6 pagesUlllted States Patent (10) Patent N0.: US 8,019,846 B2Miguel Angel Bernabe CruzNo ratings yet
- Wrong Answer Summary: Fleet Management Limited MDB CES 5.3.3 Arnab Datta 02.05.2018 54371 STCW 111 India Engine SupportDocument255 pagesWrong Answer Summary: Fleet Management Limited MDB CES 5.3.3 Arnab Datta 02.05.2018 54371 STCW 111 India Engine SupportRAHUL SINGHNo ratings yet
- GC TCD SopDocument6 pagesGC TCD Soparfan.irinbestindoNo ratings yet
- Fid1 A, (Ekafeits/Minggu-4.D)Document1 pageFid1 A, (Ekafeits/Minggu-4.D)Elisabeth TessaNo ratings yet
- REPORT 2016 06 23 1646 g14Document3 pagesREPORT 2016 06 23 1646 g14Asela BamunuarachchiNo ratings yet