Professional Documents
Culture Documents
C1-3 Revision Mind Map
Uploaded by
cuambyahooCopyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
C1-3 Revision Mind Map
Uploaded by
cuambyahooCopyright:
Available Formats
Metals are found in the Earths crust as ores metal
Transition metals
compounds inside a rock. Metals are then extracted
They are found in the middle of the periodic table between group 2 and group 3 elements. Their
from their ores either by electrolysis or reduction.
strength makes them useful as construction material. The most common transition elements are:
The way we extract metals depend on its place in the
iron, copper, zinc, gold, silver, vanadium, mercury.
reactivity series.
They combine and make alloys mixtures of different metals.
Titanium is a silvery-white metal, strong and very resistant to corrosion.
Extracting copper from its ore chalcocite (copper
(I) sulfide).
There are two ways: smelting and phytominig
reaction with sulfuric acid to produce copper
C1 - 3 Metals and their uses
sulfate solution before extraction by electrolysis.
Copper is another transition metal with many uses:
pipes, electrical wires, cooking pans and pots, as
an alloy with gold.
DIFFERENT ALLOYS: bronze, brass, smart alloys
All metals above carbon (C) are extracted by
electrolysis. Metals below carbon are extracted by
Extracting iron from its ore haematite (iron (III) oxide)
reduction with carbon.
Iron, a transition metal is extracted by reduction with carbon monoxide from its ore haematite
Metals like copper, iron and zinc are found naturally as
(Fe2O3) in the blast furnace:
oxides in their ores.
C + O2
CO2 + C
Extracting aluminium from its ore bauxite (aluminium oxide)
Aluminium is extracted by electrolysis from bauxite because it is
high in the reactivity series and we cannot use carbon to displace
it.
Aluminium has a low density for a metal and can be alloyed with a
wide range of other metals.
Fe2O3 + 3CO
CO2
Raw materials: COKE
2CO
LIME STONE
Fe + 3CO2
HAEMATITE
DIFFERENT STEELS:
Carbon steels are made by alloying iron with small quantities of carbon (from 0.03% to 1.5%).
They are used for the bodies of cars, knives, ships, structural steel for buildings and machinery.
Low-alloy steels (more expensive) contain 1-5% of other metals such as: vanadium, chromium,
nickel, titanium and tungsten.
High-alloy steels contain much higher percentage of other metals (12-25%).
Stainless steels are chromium-nickel steels used for making cooking utensils and cutlery.
You might also like
- Metals. Ferrous Metals. Iron, Steel and Cast IronDocument15 pagesMetals. Ferrous Metals. Iron, Steel and Cast Irontechnologyaljanadic0910No ratings yet
- IGCSE Chemistry - Extraction and Uses of MetalsDocument21 pagesIGCSE Chemistry - Extraction and Uses of MetalsChemistryKlipz90% (10)
- Building Technology 1: Research WorkDocument54 pagesBuilding Technology 1: Research WorkRhobie Shayne BenogsodanNo ratings yet
- Reviewer Crime Scene Investigation Board Exam Criminology Examination of SocialDocument48 pagesReviewer Crime Scene Investigation Board Exam Criminology Examination of SocialJona Addatu96% (24)
- Immigration To Australia-StepbyStep Guide (Subclass 189&190) V3.0Document5 pagesImmigration To Australia-StepbyStep Guide (Subclass 189&190) V3.0cuambyahooNo ratings yet
- Extraction of IronDocument4 pagesExtraction of IronNadita100% (1)
- Metals NewDocument36 pagesMetals NewAbenet GetachewNo ratings yet
- MetalsDocument16 pagesMetalsmhataNo ratings yet
- Extraction of Metals6Document36 pagesExtraction of Metals6manish932No ratings yet
- Extraction of Metals6Document36 pagesExtraction of Metals6Jojo Hany100% (1)
- Types of Metals and Their Uses (With Pictures)Document10 pagesTypes of Metals and Their Uses (With Pictures)Iman SadeghiNo ratings yet
- Metal: More ReactiveDocument7 pagesMetal: More ReactiveJoseph LimNo ratings yet
- EXTRACTION OF METALS Form 3 .2Document9 pagesEXTRACTION OF METALS Form 3 .2itsshaunboteNo ratings yet
- Metals and PolymersDocument37 pagesMetals and PolymerscharlotteconsigoNo ratings yet
- METALSDocument44 pagesMETALSDivine CollargaNo ratings yet
- MetalDocument14 pagesMetalsusanNo ratings yet
- Tcbe 1202-3 MetalsDocument15 pagesTcbe 1202-3 MetalsshafikNo ratings yet
- PPT-10C 6Document18 pagesPPT-10C 6samairabahriNo ratings yet
- Cloze MetalsDocument3 pagesCloze MetalsDeepa ShreenathNo ratings yet
- Class 8 Metals and Non - MetalsDocument53 pagesClass 8 Metals and Non - Metalsmanjulata.mohanty85No ratings yet
- Ballad May Alyssa G. MetalsDocument18 pagesBallad May Alyssa G. MetalsMay Alyssa BalladNo ratings yet
- DownloadedDocument12 pagesDownloadedAniket shuklaNo ratings yet
- Sat PalDocument4 pagesSat PalAmit Rashmi MishraNo ratings yet
- MetalsDocument10 pagesMetalsfergardeNo ratings yet
- Metal and NonmetalDocument26 pagesMetal and NonmetalSudhanshu Sekhar PandaNo ratings yet
- Metals: Properties of Metals Extraction of Metals Uses of MetalsDocument29 pagesMetals: Properties of Metals Extraction of Metals Uses of MetalsdhawandNo ratings yet
- UNIT 2 PPT 2Document21 pagesUNIT 2 PPT 2neha yarrapothuNo ratings yet
- Chemistry - Topic 4Document8 pagesChemistry - Topic 4Maity SkandaNo ratings yet
- Extraction of AluminiumDocument15 pagesExtraction of AluminiumdahliazamaniNo ratings yet
- Chem ch10Document5 pagesChem ch10Wendy SitNo ratings yet
- Metals HandoutDocument17 pagesMetals HandoutEyoatem TeferiNo ratings yet
- Assignment No. 6: Jhommer V. Angcon JR Bsmar-E 1F E-MAT 111Document4 pagesAssignment No. 6: Jhommer V. Angcon JR Bsmar-E 1F E-MAT 111Jeoy AngconNo ratings yet
- Metals and Non Metals NotesDocument3 pagesMetals and Non Metals NotesVUDATHU SHASHIK MEHERNo ratings yet
- Uses of MetalsDocument6 pagesUses of Metalsdan964No ratings yet
- Transition MetalDocument3 pagesTransition MetalZain KhanNo ratings yet
- Extractive Metallurgy of IronDocument11 pagesExtractive Metallurgy of Ironnot meanieNo ratings yet
- Chapter - 8 MetalDocument12 pagesChapter - 8 Metalamit_idea1No ratings yet
- Chapter 13, 14 - Metals PDFDocument9 pagesChapter 13, 14 - Metals PDFAarush SharmaNo ratings yet
- ChemistrysummaryDocument7 pagesChemistrysummaryapi-253698991No ratings yet
- Different Types of MetalsDocument9 pagesDifferent Types of MetalsEllie Henderson Year 08No ratings yet
- Fe 2O3 Fe 3O4: Extracting Iron From Iron Ore Using A Blast FurnaceDocument5 pagesFe 2O3 Fe 3O4: Extracting Iron From Iron Ore Using A Blast FurnaceTej SHahNo ratings yet
- Extraction of MetalsDocument9 pagesExtraction of MetalsNeha FaisalNo ratings yet
- Mbeya University of Science and TechnologyDocument36 pagesMbeya University of Science and TechnologyJacob kapingaNo ratings yet
- Leson3 FerrousandNon-FerrousMetalsDocument35 pagesLeson3 FerrousandNon-FerrousMetalsKaushik SenguptaNo ratings yet
- Notes, MetalsDocument7 pagesNotes, MetalsindaiNo ratings yet
- Gen Chem - Finals ReviewerDocument7 pagesGen Chem - Finals ReviewerMariel Dela Cruz TeanilaNo ratings yet
- Gen Chem - Finals ReviewerDocument9 pagesGen Chem - Finals ReviewerMariel Dela Cruz TeanilaNo ratings yet
- CEM Unit 4-1Document52 pagesCEM Unit 4-1kunalsharmaa2929No ratings yet
- Metals Non MetalsDocument49 pagesMetals Non MetalsthinkiitNo ratings yet
- R20 Advance Materials NotesDocument116 pagesR20 Advance Materials NotesPraveen 007No ratings yet
- Pure Metals: 3.2 Bonding: The Structure of MatterDocument5 pagesPure Metals: 3.2 Bonding: The Structure of MatterVickneswary MuniyanNo ratings yet
- Uses of Different Metals Through HistoryDocument18 pagesUses of Different Metals Through HistorygabriellamauriceNo ratings yet
- 10 Unit 3 PrintDocument10 pages10 Unit 3 PrintClaresse NavarroNo ratings yet
- Metals and Non MetalsDocument10 pagesMetals and Non MetalsKaran MahajanNo ratings yet
- Extraction of MetalsDocument5 pagesExtraction of MetalsALVINNo ratings yet
- 1 Chapter 1.1 Engineering Materilas (Metals,... )Document39 pages1 Chapter 1.1 Engineering Materilas (Metals,... )KidusNo ratings yet
- Metal 11Document5 pagesMetal 11Bereket BlessingNo ratings yet
- Unit One Cshemistry GCSE Revision Subsection 3-Metals and Their UsesDocument4 pagesUnit One Cshemistry GCSE Revision Subsection 3-Metals and Their Usesmws_97No ratings yet
- Copper and Its Alloys: IntroductionDocument5 pagesCopper and Its Alloys: IntroductionAhmed AlbayatiNo ratings yet
- Mechanical EngineeringDocument8 pagesMechanical EngineeringkekeNo ratings yet
- InTech-Copper and Copper Alloys Casting Classification and Characteristic MicrostructuresDocument29 pagesInTech-Copper and Copper Alloys Casting Classification and Characteristic MicrostructuresCarlos Ortega JonesNo ratings yet
- Faculty of Business - Report WritingDocument16 pagesFaculty of Business - Report WritingcuambyahooNo ratings yet
- Balance Sheet Format PDFDocument1 pageBalance Sheet Format PDFpraveenyarandoleNo ratings yet
- Advt - No - 1-2018Document5 pagesAdvt - No - 1-2018aminaNo ratings yet
- Faculty of Business - Report WritingDocument16 pagesFaculty of Business - Report WritingcuambyahooNo ratings yet
- Peterdruckers Whatmakesaneffectiveleaderpps 150823134802 Lva1 App6891Document20 pagesPeterdruckers Whatmakesaneffectiveleaderpps 150823134802 Lva1 App6891cuambyahooNo ratings yet
- PPSC Advt 60-2017 - 48cmx8colDocument1 pagePPSC Advt 60-2017 - 48cmx8colcuambyahooNo ratings yet
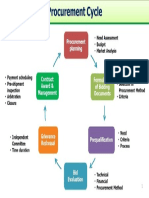
- Procurement Cycle PDFDocument1 pageProcurement Cycle PDFcuambyahoo100% (1)
- Quetta Attack PDFDocument1 pageQuetta Attack PDFcuambyahooNo ratings yet
- Medialeer Engels HsDocument1 pageMedialeer Engels HscuambyahooNo ratings yet
- Advt - No - 1-2018Document5 pagesAdvt - No - 1-2018aminaNo ratings yet
- Pharmacy Act 67Document11 pagesPharmacy Act 67Aqeel AhmedNo ratings yet
- Schmal - Cdncrimtdy - 2e - ch03 (Compatibility Mode) PDFDocument2 pagesSchmal - Cdncrimtdy - 2e - ch03 (Compatibility Mode) PDFcuambyahooNo ratings yet
- Pharmacy Act 67Document11 pagesPharmacy Act 67Aqeel AhmedNo ratings yet
- HomeostasisDocument36 pagesHomeostasisUwais AhmedNo ratings yet
- Richard Nixon - Resignation Address PDFDocument4 pagesRichard Nixon - Resignation Address PDFcuambyahooNo ratings yet
- Aleppo and World ConscienceDocument28 pagesAleppo and World ConsciencecuambyahooNo ratings yet
- Workshop IntroDocument41 pagesWorkshop Introjdpatel28No ratings yet
- Democracy Accountability and RepresentationDocument25 pagesDemocracy Accountability and RepresentationcuambyahooNo ratings yet
- Schmal Cdncrimtdy 2e Ch11 (Compatibility Mode)Document2 pagesSchmal Cdncrimtdy 2e Ch11 (Compatibility Mode)cuambyahooNo ratings yet
- Quetta Attack PDFDocument1 pageQuetta Attack PDFcuambyahooNo ratings yet
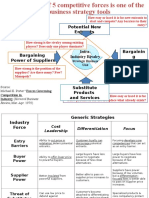
- Potential New Entrants: Strategic Business UnitDocument6 pagesPotential New Entrants: Strategic Business UnitcuambyahooNo ratings yet
- Enthalpy S&G 06Document13 pagesEnthalpy S&G 06OnSolomonNo ratings yet
- Impact of Demographic Changes On Inflation in Pakistan: A A J, F F F MDocument1 pageImpact of Demographic Changes On Inflation in Pakistan: A A J, F F F McuambyahooNo ratings yet
- 17 LectureDocument61 pages17 LecturecuambyahooNo ratings yet
- Area Population Density and Urban Rural Proportion, PakistanDocument1 pageArea Population Density and Urban Rural Proportion, PakistanZeibJahangirNo ratings yet
- Minority Report 2016Document70 pagesMinority Report 2016cuambyahooNo ratings yet
- A Kashmir Statement by Ashraf JhangirDocument5 pagesA Kashmir Statement by Ashraf JhangircuambyahooNo ratings yet
- Un-Employment Rates: Administrative 1981 Unit Both Sexes Male Female Census 1998-CensusDocument1 pageUn-Employment Rates: Administrative 1981 Unit Both Sexes Male Female Census 1998-CensuscuambyahooNo ratings yet