Professional Documents
Culture Documents
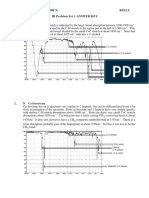
IR Values Table 1
IR Values Table 1
Uploaded by
bbtbadalCopyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
IR Values Table 1
IR Values Table 1
Uploaded by
bbtbadalCopyright:
Available Formats
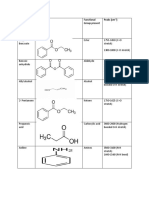
Table 1: Principal IR Absorptions for Certain Functional Groups
Functional Group Names
& Example compounds
Absorption Ranges(cm-1)
[Look for a single absorption in these regions, unless stated otherwise.]
Type of Vibration
causing IR absorption
Alkanes:
H H C H H
Methane
3000-2800
(Note: The absorptions can be seen as several distinct peaks in this region.)
H-C-H Asymmetric & Symmetric Stretch H-C-H Bend C=C-H Asymmetric Stretch C-C=C Symmetric Stretch
C H Stretch
1500-1440 3100-3000
Alkenes:
H H3C C H H
1-Propene
1675-1600 3300-3200
Alkynes:
HC C CH3
Propyne
2200-2100 3100-3000
C Stretch
Aromatic Rings:
H H H C C C C H C C H
Benzene
1600-1580 1500-1450 3600-3100
C=C-H Asymmetric Stretch C-C=C Symmetric Stretch C-C=C Asymmetric Stretch Hydrogen-bonded O-H Stretch
(This peak usually appears much broader than the other IR absorptions.
Phenols & Alcohols:
H H H C C C C C C OH H H H C H
Methanol (Alcohol)
OH
(Note: Phenols MUST have Aromatic Ring Absorptions too.)
H Phenol
Carboxylic Acids:
O H C
Formic Acid
3400-2400
(This peak always covers the entire region with a VERY BROAD peak.)
Hydrogen-bonded O-H Stretch
[Note: This peak can obscure other peaks in this region.]
OH
1730-1650
Ketones:
O H3C C
Acetone
C=O Stretch
1750-1625
C=O Stretch
CH3
Aldehydes:
O H3C C
Ethanal
1750-1625 2850-2800
H
C=O Stretch C-H Stretch off C=O C-H Stretch off C=O
2750-2700
Table 1: Principal IR Absorptions for Certain Functional Groups
Functional Group Names
& Example compounds
Absorption Ranges(cm-1)
[Look for a single absorption in these regions, unless stated otherwise.]
Type of Vibration
causing IR absorption
Esters:
O H C O CH3
Methyl Formate
1755-1650 (1300-1000) (1300-1000) 3500-3100 (TWO PEAKS!) 1640-1560 3500-3100 (ONE PEAK!) 1550-1450
H
C=O Stretch (C-O Stretch) (C-O Stretch) N-H Stretch N-H Bend N-H Stretch N-H Bend
Ethers:
O
Diethyl Ether (aka-Ethyl Ether)
AminesPrimary:
N H H
Ethylamine
AminesSecondary:
N H CH3
N-Methylethylamine
Nitriles:
H H C C N
Methanenitrile
2300-2200
C N Stretch
Nitro Groups:
O H3C N
+
1600-1500
Nitromethane
N=O Stretch
1400-1300
(Note: Both peaks are <200 cm-1 apart.)
N=O Bend N-H Stretch (similar to amines) C=O Stretch N-H Bend
Amides:
O H3C C
Methanamide
3500-3100 1670-1600 1640-1550
NH2
You might also like
- Biografia de Hugo SchiffDocument26 pagesBiografia de Hugo SchiffMartin LopezNo ratings yet
- Colour Reactions of ProteinsDocument7 pagesColour Reactions of ProteinsTARIQNo ratings yet
- Mono Methyl at IonDocument15 pagesMono Methyl at Ionlocolocolocoxoxo100% (1)
- Infra Red Infra Red: Atmanto Heru WDocument15 pagesInfra Red Infra Red: Atmanto Heru WSetya Dhana Santika AjiNo ratings yet
- Aplikasi Infra Merah Dalam Bidang Farmasi: Muhammad Taupik Laboratorium Kimia Analisis Farmasi UNGDocument35 pagesAplikasi Infra Merah Dalam Bidang Farmasi: Muhammad Taupik Laboratorium Kimia Analisis Farmasi UNGVsten MramisNo ratings yet
- IR Spectroscopy Problem Set 4 Answer Key PDFDocument6 pagesIR Spectroscopy Problem Set 4 Answer Key PDFHuy Đặng AnhNo ratings yet
- Aplikasi Infra Merah Dalam Bidang Farmasi: Abdul Rohman Laboratorium Kimia Analisis Fakultas Farmasi UGMDocument38 pagesAplikasi Infra Merah Dalam Bidang Farmasi: Abdul Rohman Laboratorium Kimia Analisis Fakultas Farmasi UGMKhumairah MohtarNo ratings yet
- 5 IR Analisis KualitatifDocument38 pages5 IR Analisis KualitatifPAHTMA PURNAMA SARI DEWINo ratings yet
- IR Analisis KualitatifDocument38 pagesIR Analisis KualitatifRetno SulistyaningrumNo ratings yet
- Aplikasi Spek IR Untuk FarmasiDocument38 pagesAplikasi Spek IR Untuk FarmasiHifdzurRashifRija'iNo ratings yet
- IR Exercise: 1-Butanol AcetophenoneDocument6 pagesIR Exercise: 1-Butanol AcetophenoneDedi saputraNo ratings yet
- Nurul Aisyah BT Yusaffendy - 2020628694Document8 pagesNurul Aisyah BT Yusaffendy - 2020628694ainaa' najwaNo ratings yet
- Spec Ir NMR Spectra Tables PDFDocument15 pagesSpec Ir NMR Spectra Tables PDFYuppie RajNo ratings yet
- Ir 3000-4000Document34 pagesIr 3000-4000heijustmeNo ratings yet
- If You Already Know The Frequency Range Use This IR Spectrum Chart. If You Already Know The Frequency Use The AboveDocument11 pagesIf You Already Know The Frequency Range Use This IR Spectrum Chart. If You Already Know The Frequency Use The AboveAdza LuailikNo ratings yet
- Basic Concepts and Hydrocarbons PDFDocument6 pagesBasic Concepts and Hydrocarbons PDFDr.CharinNo ratings yet
- 13C and 1H NMR (RMN 1H y 13C)Document1 page13C and 1H NMR (RMN 1H y 13C)veromendoNo ratings yet
- 6 Organic Chemistry IDocument27 pages6 Organic Chemistry IAwil AhmedNo ratings yet
- 6 - Organic ChemistryDocument27 pages6 - Organic ChemistryAlvaro CatalaNo ratings yet
- Organic Chemistry I EdexcelDocument28 pagesOrganic Chemistry I EdexcelAinara Román MacíasNo ratings yet
- IR Spectroyscopy: Pending Region Finger Print 600-1100 Stretching Region 1100-4000Document1 pageIR Spectroyscopy: Pending Region Finger Print 600-1100 Stretching Region 1100-4000QantLeRabyNo ratings yet
- Organic Chemistry I EdexcelDocument28 pagesOrganic Chemistry I Edexcelvyb8qpq57yNo ratings yet
- List GoodDocument2 pagesList GoodSumonNo ratings yet
- Infrared Spectra: It Is Important To Remember That The Absence of AnDocument10 pagesInfrared Spectra: It Is Important To Remember That The Absence of AnSaad NadeemNo ratings yet
- Edexcel Chemistry Data Sheet AlevelDocument6 pagesEdexcel Chemistry Data Sheet AlevelManoj oliNo ratings yet
- 3.1 Revision Guide Introduction Organic AqaDocument9 pages3.1 Revision Guide Introduction Organic AqaIman KhanNo ratings yet
- 3.1 Revision Guide Introduction Organic AqaDocument9 pages3.1 Revision Guide Introduction Organic Aqashafiqur rahmanNo ratings yet
- Hydrocarbons Final Revision WorksheetDocument21 pagesHydrocarbons Final Revision WorksheetMoonesh MKNo ratings yet
- Key HW 3 Part II SpecDocument16 pagesKey HW 3 Part II SpecTha KantanaNo ratings yet
- 6: Organic Chemistry I: 6A. Introduction To Organic Chemistry Basic Definitions To KnowDocument27 pages6: Organic Chemistry I: 6A. Introduction To Organic Chemistry Basic Definitions To KnowJam GeejeeNo ratings yet
- Butane - Rotate With C3-C4 Bond.: C H C H C H CH H H H HDocument11 pagesButane - Rotate With C3-C4 Bond.: C H C H C H CH H H H HMadison FullerNo ratings yet
- HW Ir 2020Document5 pagesHW Ir 2020iris glzNo ratings yet
- Sample: Structure Functional Group Present Peaks (CM)Document2 pagesSample: Structure Functional Group Present Peaks (CM)MUHAMMAD USMANNo ratings yet
- Sample: Structure Functional Group Present Peaks (CM)Document2 pagesSample: Structure Functional Group Present Peaks (CM)MUHAMMAD USMANNo ratings yet
- Frequency Table For IR & NMRDocument6 pagesFrequency Table For IR & NMRYogesh PingleNo ratings yet
- Alcohol Phenol Ether (1) 6Document9 pagesAlcohol Phenol Ether (1) 6sdnishacNo ratings yet
- Dia orDocument8 pagesDia orNaman MahawarNo ratings yet
- Alcohol Phenol Ether (1) 4Document9 pagesAlcohol Phenol Ether (1) 4subhashitamurapakaNo ratings yet
- Alcohol and Carboxylic AcidDocument6 pagesAlcohol and Carboxylic AcidPutri Nur SyafieqahNo ratings yet
- Organic Chemistry SummaryDocument10 pagesOrganic Chemistry SummaryKiara LimNo ratings yet
- Raj1 PDFDocument35 pagesRaj1 PDFAvdhoot Gautam100% (1)
- Halogen DerivativeDocument32 pagesHalogen Derivativesachin pantNo ratings yet
- Elimination Reactions: Elimination Reaction: A Reaction in Which A Molecule Loses Atoms or Groups of AtomsDocument8 pagesElimination Reactions: Elimination Reaction: A Reaction in Which A Molecule Loses Atoms or Groups of AtomsMohammed Adil ShareefNo ratings yet
- Cycloalkanes ConformersDocument15 pagesCycloalkanes ConformersSabila IzzatiNo ratings yet
- Functional Class Range (CM) Intensity Assignment Alkanes: AlkenesDocument1 pageFunctional Class Range (CM) Intensity Assignment Alkanes: AlkenesStoica AlexandruNo ratings yet
- IR Absorption Table PDFDocument3 pagesIR Absorption Table PDFDavid QuinteroNo ratings yet
- MEQ L (Introductory Organic Chemistry)Document62 pagesMEQ L (Introductory Organic Chemistry)Furious SKNo ratings yet
- Dodecanoyl Chloride: CH BendDocument15 pagesDodecanoyl Chloride: CH BendFatima AhmedNo ratings yet
- AlkynesDocument3 pagesAlkynesnandankheniNo ratings yet
- L01 - 1 Petroleum Engineering (v1.0)Document35 pagesL01 - 1 Petroleum Engineering (v1.0)Santiago ZapataNo ratings yet
- Organic Chem SyllabusDocument7 pagesOrganic Chem SyllabusHriday VakhariaNo ratings yet
- Combustion and Fuels: - Usu. Hydrocarbons or Hydrogen in Air Are UsedDocument29 pagesCombustion and Fuels: - Usu. Hydrocarbons or Hydrogen in Air Are UsedJefferson CaponeroNo ratings yet
- Alkanes: Quicktime™ and A Tiff (Uncompressed) Decompressor Are Needed To See This PictureDocument30 pagesAlkanes: Quicktime™ and A Tiff (Uncompressed) Decompressor Are Needed To See This Pictureafreenbegum-0502No ratings yet
- Adobe Scan 31 May 2023Document1 pageAdobe Scan 31 May 2023Imuu IsmuuNo ratings yet
- Conceciones Entre Alquenos y AlquinosDocument1 pageConceciones Entre Alquenos y Alquinos(E)Ambar Rubie GarciaNo ratings yet
- 15 Carbonyls Carboxylic Acids and Chirality Iedxcel PDFDocument20 pages15 Carbonyls Carboxylic Acids and Chirality Iedxcel PDFKazi Unysah NabiNo ratings yet
- Chem CH 12 NIE Premium NOtesDocument22 pagesChem CH 12 NIE Premium NOtesansarahmad6931No ratings yet
- Hydrocarbons PDFDocument48 pagesHydrocarbons PDFAniruddha KawadeNo ratings yet
- Connections, Displays and Operating Elements: MTN299200/MTN299201/MTN299202Document2 pagesConnections, Displays and Operating Elements: MTN299200/MTN299201/MTN299202fikrifazNo ratings yet
- Stephen Danso - MensahDocument126 pagesStephen Danso - MensahfikrifazNo ratings yet
- Shimadzu FTIR Academic 12-1Document1 pageShimadzu FTIR Academic 12-1fikrifazNo ratings yet
- TB Solubility SaccharinDocument1 pageTB Solubility SaccharinfikrifazNo ratings yet
- Torpac Rat Gpig PricingDocument1 pageTorpac Rat Gpig PricingfikrifazNo ratings yet
- Additive 369Document3 pagesAdditive 369fikrifazNo ratings yet
- tp189 c6Document7 pagestp189 c6fikrifazNo ratings yet
- A Lead CompoundDocument2 pagesA Lead CompoundfikrifazNo ratings yet
- Case Histories of Amine Plant Equipment Corrosion Problems - Part 2Document4 pagesCase Histories of Amine Plant Equipment Corrosion Problems - Part 2jdgh1986No ratings yet
- Syllabus For Pharmacognosy-III, B. Pharma (3 Yr)Document1 pageSyllabus For Pharmacognosy-III, B. Pharma (3 Yr)Deepak PradhanNo ratings yet
- NCERT Solutions For Class 12 Chemistry Chapter 12 Aldehydes, Ketones and Carboxylic AcidsDocument17 pagesNCERT Solutions For Class 12 Chemistry Chapter 12 Aldehydes, Ketones and Carboxylic Acidssinghrathoreganga814No ratings yet
- Proteins IAL Edexcel Unit 1Document4 pagesProteins IAL Edexcel Unit 1MahmoudNo ratings yet
- Chemistry Reviewer 9Document2 pagesChemistry Reviewer 9Jade RonquilloNo ratings yet
- Synthesis of Amides of Lysergic AcidDocument5 pagesSynthesis of Amides of Lysergic Acid0j1u9nmkv534vw9v100% (1)
- Dietanolamina - TDS - DOWDocument2 pagesDietanolamina - TDS - DOWhenriquefxs2926No ratings yet
- ACS Review 22 AminesDocument10 pagesACS Review 22 AminesJana BazziNo ratings yet
- Vehicle WashDocument12 pagesVehicle Washyilmaz_uuurNo ratings yet
- DM pp81-104Document24 pagesDM pp81-104MLUNGISI MkhwanaziNo ratings yet
- J. Chem. Thermodynamics: Kun Fu, Pan Zhang, Dong FuDocument10 pagesJ. Chem. Thermodynamics: Kun Fu, Pan Zhang, Dong FuTobias GonzalezNo ratings yet
- Aldehydes Ketones and Carboxylic Acids - NCERT SolutionsDocument27 pagesAldehydes Ketones and Carboxylic Acids - NCERT SolutionsVyjayanthiNo ratings yet
- Analysis of Amino Acids by Paper ChromatDocument8 pagesAnalysis of Amino Acids by Paper ChromatasdfNo ratings yet
- 12th Board Sprint-Amines (15.12.2020)Document62 pages12th Board Sprint-Amines (15.12.2020)Harsh ShahNo ratings yet
- 29th Jan Shift - 2 - AakashDocument16 pages29th Jan Shift - 2 - AakashMusic BanglaNo ratings yet
- ISC 12 Chemistry SyllabusDocument11 pagesISC 12 Chemistry SyllabusShivaNo ratings yet
- Progressive GR 9 2nd Q EDocument12 pagesProgressive GR 9 2nd Q ERAMIL BAUTISTANo ratings yet
- Enzymatic Synthesis of Amides 2015Document44 pagesEnzymatic Synthesis of Amides 2015bluedolphin7No ratings yet
- Chemistry Pase-II MDCAT Paper 2017Document25 pagesChemistry Pase-II MDCAT Paper 2017Aarish KhanNo ratings yet
- NCERT Important Name Reactions For RevisionDocument34 pagesNCERT Important Name Reactions For Revisionyimisa2927No ratings yet
- Ioc 9Document3 pagesIoc 9KarthikeyanNo ratings yet
- 0901b803801522b2 PDFDocument36 pages0901b803801522b2 PDFoviangstaNo ratings yet
- Chemistry Chapter 19 4ed Problems ScannedDocument18 pagesChemistry Chapter 19 4ed Problems ScannedeuniawapsNo ratings yet
- Chapter 2.6 Aldehyde & KetoneDocument40 pagesChapter 2.6 Aldehyde & Ketone0JTINGNo ratings yet
- Bảng phổ IRDocument5 pagesBảng phổ IRĐan KhanhNo ratings yet
- Mahmoud Sadeq RDocument8 pagesMahmoud Sadeq RWalid Ebid ElgammalNo ratings yet
- Protein Chemistry Section ExamDocument9 pagesProtein Chemistry Section ExamJoelNo ratings yet