Professional Documents
Culture Documents
Bakteri Eukariotik Dan Prokariotik PDF
Bakteri Eukariotik Dan Prokariotik PDF
Uploaded by
Bagus CatshopOriginal Title
Copyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
Bakteri Eukariotik Dan Prokariotik PDF
Bakteri Eukariotik Dan Prokariotik PDF
Uploaded by
Bagus CatshopCopyright:
Available Formats
J. CM Sci.
44, 335-352 (1980)
Printed in Great Britain Company of Biologists Limited 1080
335
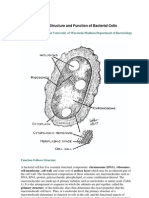
THE ULTRASTRUCTURE OF
PROKARYOTIC-EUKARYOTIC
CELL JUNCTIONS
SIDNEYL.TAMM
Laboratory of Molecular Biology and Department of Zoology, University of Wisconsin,
Madison, Wisconsin 53706, U.S.A.*
SUMMARY
Freeze-fracture and thin-section electron microscopy were used to describe the sites of
attachment of 2 kinds of ectosymbiotic bacteria to a devescovinid flagellate from termites. In
each case, surface specializations in both partners occur at the juncrional complexes. Rod
bacteria lie in pockets of the eukaryotic membrane which are coated by dense material and
contain high densities of intramembrane particles. A double row of closely spaced particles
circumscribes the edges of the pockets on the P face. The surface of the rod bacteria which is
exposed to the external medium bears a thick glycocalyx and flagella. Fusiform bacteria are
attached along ridges of the protozoon surface. Dense material underlies the ridges, and particles
are aggregated on both P and E faces of the ridge membrane. The outer layer of the fusiform
bacteria is grooved to match the host ridge. The bacterialdevescovinid junctions are considered
to serve mainly as attachment sites, and the membrane specializations at the sites of contact are
discussed in this respect. Control of junction assembly by the cortical bacteria is suggested by
the parallel patterns of juncrional replication and bacterial reproduction.
INTRODUCTION
Associations of eukaryotic cells in multicellular organisms are characterized by
specialized regions of contact between the membranes of adjacent cells. These intercellular junctions are of different types, and serve to integrate and coordinate the
various kinds of cellular interactions (Gilula, 1974; McNutt, 1977; Staehelin,
I974)Associations also occur between prokaryotic and eukaryotic cells. These relationships are extremely diverse, but are generally called symbiotic (Ball, 1969; Henry,
1966; Kirby, 1941a; Starr, 1975). This report deals with ectosymbiotic associations,
in which the prokaryotes live permanently attached to the surface of the eukaryotic
cell.
The junctions formed between ectosymbiotic prokaryotes and eukaryotic cells are
not well understood. Conventional electron-microscopic descriptions of prokaryoticeukaryotic cell junctions have revealed various kinds of structural specializations at the
site of contact between prokaryote and eukaryote (Bloodgood & Fitzharris, 1976;
Cleveland & Grimstone, 1964; Hollande & Carruette-Valentin, 1970, 1971, 1972;
Present address: Boston University Marine Program, Marine Biological Laboratory, Woods
Hole, Massachusetts 02543, U.S.A.
336
S. L. Tamm
Hollande & Valentin, 1969; Lavette, 1969; Smith & Arnott, 1974; Smith, Buhse &
Stamler, 1975; Tamm & Tamm, 1974, 1976; Wagner & Barrnett, 1974). Freezefracture replication has greatly clarified the nature of intercellular junctions, but
until recently this method has not been applied to the study of prokaryotic-eukaryotic
cell junctions.
The hindgut of the wood-eating roach, Cryptocercus, and lower termites is a good
place to investigate prokaryotic-eukaryotic cell junctions. This environment harbours
numerous kinds of associations between bacteria andflagellateprotozoa. In particular,
these symbioses reach their highest level of development in devescovinid flagellates
and related polymastigotes (Ball, 1969; Cleveland & Grimstone, 1964; Kirby, 1941a,
b, 1942a, b, 1945, 1949)- The best-known example is the regular attachment of
spirochaetes and rod bacteria to specialized surface structures of Mixotricha paradoxa,
a flagellate from Mastotermes darwiniensis; the coordinated undulations of the spirochaetes propel the protozoon (Cleveland & Grimstone, 1964).
We previously described the association of 2 kinds of ectosymbiotic bacteria with the
surface of a remarkable devescovinid flagellate from a Florida termite (Bloodgood &
Tamm, 1977; Tamm, 1976, 1978 a, b, c, 1979; Tamm & Tamm, 1974, 1976). The
anterior part of this protozoon continually rotates in the same direction relative to the
rest of the cell. Rotational movements are caused by a rod-like axostyle complex
running through the cell body (Tamm, 1978 a). We were interested in the adherent
bacteria because they provided useful markers for following movements of the cell's
plasma membrane. Recently, we discovered that these bacteria are also useful to the
protozoon: one type is flagellated, and provides locomotion for the host (Tamm,
1978c, manuscript in preparation).
In this paper we present a correlated thin-section and freeze-fracture description of
the junctional complexes formed between the 2 kinds of bacteria and the devescovinid.
The results, reported previously in preliminary form (Bloodgood & Tamm, 1977;
Tamm, 19786, 1979), illustrate several new features of the ultrastructure of prokaryotic-eukaryotic cell junctions.
MATERIALS AND METHODS
The organism studied here is the same (unnamed) devescovinid flagellate from Cryptotermes
cavifrons used in previous work (Tamm, 1976, igj&a-c, 1979; Tamm & Tamm, 1974). The
methods for thin-section electron microscopy and freeze-fracture of this protozoon have been
reported previously (Tamm & Tamm, 1974, 1976; Tamm, 1979).
RESULTS
General features of the bacterial-devescovinid associations
Rod-shaped and fusiform-like bacteria are found in intimate association with the
surface of the protozoon (Figs. 1, 2). The rod bacteria are ~2~3 /tm long, ~o-6/im
in diameter, and are flagellated on one side (Fig. 3B; Tamm, 1978c). The fusiform
bacteria are longer (~ 5-6/tin) and more slender (~o-2/tm in diameter), and do not
Bacterial-protozoon cell junctions
337
Fig. i. Pattern of ectosymbiotic rod bacteria (rb) on the surface of the protozoon. The
adherent fusiform bacteria are not visible at this magnification. The anterior end of the
cell is continually turned in a clockwise direction by the rod-like axostyle complex
running through the cell body. The bacteria-free zone of membrane between the head
and the body (sz) undergoes continual shear. Zeiss Nomarski optics, x noo.
have flagella. As in other devescovinid flagellates (Kirby, 19416, 1942a, b, 1945,
1949), the bacteria are attached in a characteristic and specific pattern to the surface of
the host. The rod bacteria are arranged end to end in parallel rows which follow a
helical path on the devescovinid's body, but run more transversely on the anterior end,
or head (Fig. 1). The fusiform bacteria live only on the body surface, and lie end toend in single rows or several parallel rows between the rows of rod bacteria (Fig. 2).
The cell envelopes of the bacteria have the multilayered structure typical of Gramnegative bacteria (Sleytr, 1978). The surface layers include a plasma membrane, a
dense peptidoglycan layer (not clearly visible in the fusiform bacteria), and an outer
membrane (Figs. 3-5). Extending from the outer membrane is a fuzzy filamentous
5. L. Tatnm
rb
Fig. 2. Reconstruction of a small area of the body surface to show the arrangement of
the ectosymbiotic bacteria, and the intramembrane particle arrays associated with the
attachment sites. Rod bacteria (rb) lie in deep pockets (p) of the protozoon plasma
membrane. Fusiform bacteria (fb) are attached along ridges (r) of the host surface, and
are grooved along the site of contact. The P face of the eukaryotic membrane (exposed
at right) exhibits aggregations of intramembrane particles (dots) along the ridges and in
the pockets. In addition, a double row of particles circumscribes the opening of the
pocket ('seam', s).
Fig. 3A, B. Adjacent thin sections cut transversely through the rows of rod bacteria
(rb) and fusiform bacteria (fb) on the surface of the protozoon. The pockets of the
devescovinid membrane (dpm) surrounding the rod bacteria (rb) are coated by an electron-dense layer (c) on the cytoplasmic side. Less-dense material (arrows) underlies the
ridges of the devescovinid membrane to which the fusiform bacteria (fb) attach.
Favourable transverse sections through the edge (e, in A) of the pocket membrane show
no opacities corresponding to the particle seam in freeze-fracture replicas (cf. Figs.
Bacterial-protozoan cell junctions
3A
fb
7, 8). The bacterial cell envelopes consist of a plasma membrane (bpm), a dense peptidoglycan layer (d) (not visible in the fusiform bacteria), and an outer membrane
(bom) and glycocalyx (g). The rod bacteria bear a thick glycocalyx andflagella(/) only
on the exposed part of their surface at the pocket opening. Note the groove in the
glycocalyx and outer membrane of the fusiform bacteria where they contact the surface
of the host, x iooooo.
S. L. Tamm
34
bom
Fig. 4. Diagram of the junctional complexes with rod bacteria (rb) and fusiform bacteria (fb) as seen in transverse thin sections (cf. Figs. 3 and 5). The flagella on the exposed surface of the rod bacteria are omitted, dpm, devescovinid pocket membrane;
c, membrane coat; bpm, bacterial plasma membrane; d, dense peptidoglycan layer;
bom, bacterial outer membrane; g, glycocalyx.
layer which is termed a glycocalyx here, although its polysaccharide nature has not
been demonstrated. In this report, only junctional specializations of the bacterial
outer membrane and glycocalyx are described.
As will become apparent, the 2 types of bacteria form 2 structurally distinct kinds of
junctions with the protozoon.
Rod bacterial junctions
The rod bacteria lie in deep invaginations of the protozoon's plasma membrane
(Figs. 3-5). These pockets do not completely enclose the bacteria, but leave part of
their surface exposed to the external medium.
A layer of dense fuzzy material is closely applied to the cytoplasmic side of the
pocket membrane (Fig. 3). This dense coat ends abruptly at the edges of the pockets
where the plasma membrane everts over the cell surface.
Freeze-fracture images reveal specialized distributions of intramembrane particles
in the pocket membrane of the devescovinid (Figs. 69). The most striking specialization is a double row of closely spaced particles that circumscribes the edges of the
pockets on the P face, giving the appearance of a distinct' seam' at the pocket opening.
Bacterial-protozoon cell junctions
dpm
Fig. 5. Section through a fragment of plasma membrane from a glycerinated devescovlnid, showing persistence of bacterial attachment. The characteristic structure of the
junctional complexes is preserved - and even clarified - after disruption and extraction
of the cell./Z>, fusiform bacterium; rb, rod bacterium; dpm, devescovinid pocket membrane; c, membrane coat; bpm, bacterial plasma membrane; d, dense peptidoglycan
layer; bom, bacterial outer membrane; g, glycocalyx. x 68000.
This particle array is not evident on the E face of the pocket membrane, nor are
complementary depressions found there (Fig. 9). In favourable thin sections through
the edges of the pockets, no densities in the lipid bilayer corresponding to the particle
seam are observed (Fig. 3 A).
In addition, the particle density on both faces is approximately 2-fold greater in the
pocket membrane than in surrounding regions of the cell membrane (Figs. 8, 9). For
comparison, it should be noted that the particle distribution on the P face of nonjunctional regions of the devescovinid membrane - i.e. between the sites of bacterial
attachment - is more or less random, with a density of ~ 800 particles/^m2 (Tamm,
1979). The size and shape of the intramembrane particles (approximately 7-11 nm
in diameter) appear similar in both junctional and non-junctional regions of the protozoon membrane.
The rod bacteria also exhibit surface specializations at the junctions. The part of
their surface which faces the external medium bears a prominent glycocalyx, as well
as flagella (Figs. 3-5; Tamm, 1978c). The filamentous nature of the glycocalyx is
particularly evident after glycerination (Fig. 5). In contrast, the glycocalyx is reduced
or absent, andflagellaare missing, on the bacterial surface that is enclosed by the host
pocket. Consequently, only a narrow, somewhat irregular space separates the outer
membrane of the bacterium from the pocket membrane of the devescovinid.
Division stages of the rod bacteria can be found almost anywhere on the surface of
S. L. Tamm
fb
Fig. 6. P face of the body membrane. Particles are aggregated along the ridges (r) to
which the fusiform bacteria (Jb) adhere. Several rod bacteria (rb) are removed from
the membrane pockets (p) by the fracturing process. Only i row of particles in the seam
(j) around the edges of the pockets is visible here. In the centre, i member of a dividing
rod bacterium is retained; the other daughter has been lost, revealing particle arrays
typical of the junctional complex in the replicating pocket of the host, x 43 000.
Bacterial-protozoon cell junctions
Fig. 7A, B. P face of membrane pockets (/>), from which rod bacteria have been removed
during fracturing. A double row of closely spaced particles forms a seam (j) around the
edges of the pockets, A, x 40000; B, X 110 000.
343
S. L. Tamrn
344
Fig. 8. P face of 2 adjacent membrane pockets {p). The particle seams (J) bordering 1
edge of each pocket are visible. The particle density in the pocket membrane is
~ 2 x that in non-junctional regions (centre strip between seams), x 96000.
most devescovinids (Figs. 6, 9). Since the protozoa are not dividing, the bacteria
evidently reproduce in situ throughout the cell cycle of the host. The various junctional
specializations of the devescovinid membrane are reproduced in parallel with the
division of the bacteria (Figs. 6, 9).
Fusiform bacterial junctions
The less-conspicuous fusiform bacteria are attached along their length to ridge-like
elevations of the host surface (Fig. 2). The membrane ridges are underlain by electrondense material which is less .noticeable than the coating of the membrane pockets
Bacterial-protozoon cell junctions
345
Fig. 9. E face of the body membrane. Particles are aggregated under the ridges (r) to
which fusiform bacteria (fb) attach. Note the absence of depressions complementary to
the particle seam at the edges of the membrane pockets (p) (arrow). The elongated
pocket at the lower right has a constriction (c), indicating replication of the junctional
complex in parallel with the division of the enclosed rod bacterium, as shown in Fig. 6.
x 47000.
SSJ
C E L 44
346
S. L. Tamm
surrounding the rod bacteria (Figs. 3, 4). Freeze-fracture replicas reveal aggregations
of 7-11 nm diameter particles along both P and E faces of these ridges (Figs. 6, 9).
Opposite to the surface ridge of the protozoon, the outer membrane and glycocalyx
of the fusiform bacterium are indented to form a matching groove (Figs. 3-5). This
complementary configuration resembles a tongue-and-groove joint, and is probably an
Table 1. Surface specializations of protozoon and bacteria at
junctional complexes
Devescovinid .
Membrane
pocket
Rod bacteria
Glycocalyx and
flagella absent
Dense coat
Particle
aggregation
Particle
' seam'
Devescovinid
Membrane ridge
, Fusiform bacteria
Groove in outer
membrane and
glycocalyx
Dense underlying
material
Particle
aggregation
adaptation for attachment (see Discussion). The fusiform bacteria are completely
covered by a glycocalyx; this layer does not appear modified at the junction but lies
in close contact with the surface ridge of the host. As a result, the grooved outer
membrane of the bacteria is separated from the ridged membrane of the protozoon by
a uniform distance that corresponds approximately to the thickness of the intervening
glycocalyx.
Dividing fusiform bacteria have not been observed in freeze-fracture replica9, but
their greater length compared to the rod bacteria would decrease the chances of finding
fission stages by this method.
Table I summarizes the thin-section and freeze-fracture findings on the various
surface specializations exhibited by the 2 types of bacteria and the devescovinid at the
junctions.
Bacterial attachment to membrane fragments
Devescovinids were disrupted by glycerination or detergent treatment, and the
resulting pieces of cell membrane examined by electron microscopy. Both rod and
fusiform bacteria remained attached in their characteristic pattern to the membrane
fragments (Fig. 5).
The structural specializations at the sites of contact between prokaryotes and protozoon therefore provide firm mechanical anchorage for the bacteria.
Bacterial-protozoon cell junctions
347
DISCUSSION
In comparison to intercellular junctions, the nature of the junctions formed between
prokaryotic and eukaryotic cells is not well understood. We therefore used a correlated thin-section and freeze-fracture approach to describe the attachment sites of 2
kinds of bacteria to the surface of a devescovinid flagellate from termites. The 2
prokaryotes were found to form structurally distinctive junctions with the eukaryote,
and in each case the surface elements of both partners were modified at the sites of
contact (Table 1).
The most obvious structural contribution of the protozoon to the junctions is its
elaborate cortical sculpturing: deep invaginations surround each rod bacterium,
while long ridges underlie the sites of attachment of the fusiform bacteria. The significance of the surface topography for attachment of the prokaryotes is discussed
below.
Complex surface adaptations for accommodating bacteria are also found in Mixotricha paradoxa, a related flagellate from Mastotermes darwiniensis. Spirochaetes are
attached to small depressions on the posterior side of regularly arranged projections
(brackets) of the protozoon's surface, while rod bacteria lie in concavities on the
opposite side of the brackets (Cleveland & Grimstone, 1964). Somewhat similar
attachment sites for spirochaetes occur on the surface of Pyrsonympha, but in addition
a striated rootlet - looking amazingly like a ciliary rootlet - extends from the point of
attachment of the bacterium into the cytoplasm of the protozoon (Smith & Arnott,
1974; Smith et al. 1975).
Shallow pockets or cup-like depressions of the eukaryotic cell surface have been
reported where bacteria attach to other insect gut flagellates (Ball, 1969; Cleveland &
Grimstone, 1964; Hollande & Valentin, 1969; Kirby, 1941a), as well as to intestinal
epithelial cells (Chase & Erlandsen, 1978; Neutra, 1979,1980; Snellen& Savage, 1978;
Wagner & Barrnett, 1974). In addition, ridge-like elevations of the host surface have
been described at the sites of contact with adherent bacteria in other termite flagellates
(Hollande & Carruette-Valentin, 1970, 1971; Lavette, 1969; Tamm&Tamm, 1976).
A common feature shared by most of these junctions and the bacterial-devescovinid
junctions described here is the presence of dense, cytoplasmic material underlying the
junctional membrane of the host. Membrane coats or dense plaques are also found at
desmosomes and synaptic contacts in higher animals (Ellisman, Rash, Staehelin &
Porter, 1976; Gilula, 1974; Heuser & Reese, 1977; McNutt, 1977; Staehelin, 1974).
Adhesive junctions, such as desmosomes, also possess a constant intercellular space,
typically ~ 25-3511111 wide, containing dense filamentous material (Gilula, 1974;
McNutt, 1977; Staehelin, 1974). Similar well-defined gaps, also spanned by filaments
or dense material, are found at many prokaryotic-eukaryotic junctions, particularly
those in which only one end of the bacterium is attached to the eukaryote (Cleveland &
Grimstone, 1964; Hollande & Valentin, 1969; Smith & Arnott, 1974; Smith et al.
1975; Wagner & Barrnett, 1974). In several such end-on attachments, a 30-40-nm
space, bridged periodically by filaments specific to the junction complex, separates the
bacterial cell wall from the membrane of the eukaryotic cell (Hollande & Valentin,
23-2
348
S. L. Tamm
1969; Wagner & Barrnett, 1974). In one case (Hollande & Valentin, 1969), this filamentous material exhibits a central dense stratum resembling that found in the interspace of spot desmosomes. Whether the extracellular material originates from the
prokaryote or the eukaryote, or both partners, is not known. However, the marked
resemblance of this feature of prokaryotic-eukaryotic junctions to desmosomes
suggests a similar function.
In associations where the bacteria adhere along their length, the junctions often
exhibit less obvious filamentous connexions between the prokaryote and eukaryote
(Bloodgood & Fitzharris, 1976; Cleveland & Grimstone, 1964; Hollande & CarruetteValentin, 1970, 1971; Lavette, 1969; Tamm & Tamm, 1976). In many of these cases,
as in the fusiform bacterial-devescovinid association described here, the bacterial
glycocalyx rests in direct contact with the junctional membrane of the host. Since no
other structural connexion between prokaryote and eukaryote is evident, adhesion is
evidently mediated by the (bacterial) glycocalyx alone.
The fusiform bacterial-devescovinid junctions described here have an additional
feature which seems uniquely designed to promote the association. The outer layer of
the bacterium is grooved to match the apposing surface ridge of the host. This
tongue-and-groove configuration is probably a structural adaptation for adhesion.
Similar ridge-groove joints have been reported between another devescovinid flagellate
and adherent fusiform bacteria (Tamm & Tamm, 1976).
That the bacterialdevescovinid junctions serve mainly, if not exclusively, as
attachment sites seems self-evident from the nature of the association. Further evidence that the junctions provide strong mechanical anchorage for the prokaryotes
comes from the persistent adhesion of the bacteria to disrupted fragments of the host
membrane. The junctional complexes are therefore considered to perform an adhesive function, analogous to the role played by desmosomes in higher organisms.
Because of the morphological asymmetry of the prokaryotic-eukaryotic junctions,
however, a better analogy may be to hemidesmosomes - i.e. the devescovinid would be
comparable to the basal epithelial cell, and the outer layer of the bacteria to the extracellular lamina.
Freeze-fracture
Freeze-fracture replicas of the devescovinid membrane demonstrated specific
arrays and/or distributions of intramembrane particles at the sites of contact with
each type of bacterium. In this respect the prokaryotic-eukaryotic junctions resemble
intercellular junctions in multicellular organisms.
The morphological similarities between desmosomes and prokaryotic-eukaryotic
cell contacts, based on thin-section electron microscopy, were pointed out above. Our
freeze-fracture images, however, reveal important differences between the bacterialdevescovinid junctions and desmosomes. The fracture faces of desmosomes either
bear few particles (belt desmosomes), or, in the case of spot desmosomes, contain
patches of irregularly shaped particles, often fibrillar in form and apparently unorganized (McNutt, 1977; Staehelin, 1974). In contrast, the particles seen in the
Bacterial-protozoon cell junctions
349
devescovinid membrane at the bacterial attachment sites are dimensionally uniform,
and highly organized in the case of the particle seams.
These differences may be significant, since the fibrillar appearance of particles in
fractured desmosomes is interpreted to represent transmembrane connexions which
have been broken off and plastically deformed during the fracturing process (McNutt,
1977; Staehelin, 1974). Such differences do not necessarily rule out participation of the
particles observed here in cell adhesion, but rather point up the uniqueness of the
prokaryotic-eukaryotic cell contacts.
The double seam of closely spaced particles lining the edges of the membrane pockets may function in a novel way to promote attachment of the rod bacteria. The
circumference of these surface imaginations remains constant, never closing completely over the bacteria, nor opening wider. The particle seam may stabilize this highly
asymmetric configuration of the membrane by acting as a fixed aperture. By so doing,
this particle array would help to retain the bacteria, as well as to ensure that the
flagellated portion of their surface - which provides motility for the protozoon (see
below) - is not covered over.
To date, the freeze-fracture method has been applied to only a few other prokaryotic-eukaryotic junctions. Chase & Erlandsen (1978) and Snellen & Savage
(1978) found changes in intramembrane particle density in rat mucosal cell membranes
at the attachment sites of filamentous bacteria. In contrast, a freeze-fracture study of
the attachment sites of 2 kinds of bacteria to primate intestinal cells revealed no
particle specializations in the host cell membrane, but particle arrays were found on
the outer membranes of both types of bacteria (Neutra, 1979, 1980). In the present
study, we did not determine whether specialized particle arrays also occurred in the
outer membrane of the rod or fusiform bacteria at the junctions with the devescovinid.
An important corollary of this study of the bacterialdevescovinid junctions is that
the intimate and highly specialized nature of the association provides strong justification for using the ectosymbiotic bacteria as visible markers to follow movements
of the devescovinid plasma membrane (Tamm, 1976, 1979; Tamm & Tamm, 1974,
1976).
Relations between junctions and the type of symbiotic association
Bloodgood & Fitzharris (1976) considered it unlikely that junctional structures
apparently specialized for promoting and maintaining prokaryote-eukaryote associations would evolve in either the protozoon or the bacteria if the association conveyed
no selective advantage upon the partner elaborating the specialization. They argued
that a partner which contributed a structural specialization to the junctional complex
must therefore benefit from the association. These authors used this approach to
classify the type of symbiotic relationships between bacteria and insect gut flagellates.
Although examples were found in which one or the other partner provided structural
specializations for attachment, Bloodgood & Fitzharris did not observe cases where
both partners contributed to the formation of the junctional complexes.
Such associations, indicative of true mutualistic symbiosis, have been reported
between adherent spirochaetes and Pyrsonympha (Smith & Arnott, 1974; Smith et al.
350
S. L. Tamm
1975), and possibly in several other cases (Hollande & Valentin, 1969; Wagner &
Barrnett, 1974). The junctional complexes described here are additional examples of
participation between surface elements of both the bacteria and the eukaryote
(Table 1), and thus may indicate a mutualistic symbiosis. The presumed advantage
conferred upon the bacteria by this association is not understood. However, recent
work using selective inhibitors of prokaryotic vs. eukaryotic motility has shown that the
flagellated rod bacteria provide locomotion for the devescovinid (Tamm, 1978c,
manuscript in preparation), analogous to the spirochaete motility-symbiosis in Mixotricha (Cleveland & Grimstone, 1964). It is also possible that the prokaryotes provide
other functions for the host, such as cellulose digestion (cf. Bloodgood & Fitzharris,
1976) or nitrogen fixation (Breznak, 1975). The junctions may therefore serve other
purposes besides adhesion, and a role in metabolic or physiological coupling between
the partners cannot be ruled out by ultrastructural comparisons alone.
Control of junction assembly
The positional information that specifies the location and spatial organization of cell
junctions is unknown. The bacterial-devescovinid association provides some insight
into this problem. As noted above, the bacteria reproduce in situ on the surface of the
host. The various junctional structures contributed by the protozoon, including the
specialized arrays of intramembrane particles, are reproduced in parallel to the division
of the bacteria (cf. Figs. 6, 9).
Of the 2 concurrent processes, bacterial reproduction and junctional replication, the
former seems more likely to be the primary causative event. If so, the sites and arrangement of the junctional complexes would be determined by the reproductive pattern of
the attached prokaryotes. The positional information for the assembly of the junctional
membrane specializations of the protozoon may thus be provided by the bacteria
themselves - much like the pattern of cortical inheritance in ciliate Protozoa
(Sonneborn, 1970). This hypothesis could be tested by determining the effects of
inhibiting bacterial growth on the formation of junctional specializations by the
protozoon.
I am grateful to Doctors Birgit Satir, Peter Satir and Caroline Schooley for instruction in
freeze-fracture technique at the Electron Microscope Laboratory, University of California,
Berkeley, California. The Wisconsin Regional Primate Research Center, University of Wisconsin, Madison, generously provided use of a Balzers freeze-etch unit. It is also a pleasure to
thank Dr Gary Borisy and the Laboratory of Molecular Biology, University of Wisconsin, for
electron-microscope facilities and general laboratory support. This work was supported by
National Science Foundation Grant PCM 77-09880.
REFERENCES
BALL, G. H. (1969). Organisms living on and in Protozoa. In Research in Protozoology, vol. 3
(ed. T.-T. Chen), pp. 565-718. New York: Pergamon.
BLOODGOOD, R. A. & FITZHARRIS, T. P. (1976). Specific associations of prokaryotes with
symbiotic flagellate Protozoa from the hindgut of the termite Reticulitermes and the woodeating roach Cryptocercus. Cytobios 17, 103-122.
Bacterial-protozoon cell junctions
351
BLOODGOOD, R. A. & TAMM, S. L. (1977). Membrane specializations involved in the association
of prokaryotes with symbiotic flagellate protozoa. Vth int. Cong. Protozool., New York, abstract 260.
BREZNAK, J. A. (1975). Symbiotic relationships between termites and their intestinal microbiota. In Symbiosis, Symp. Soc. exp. Biol. (ed. D. H. Jennings & D. L. Lee), 29, pp. 559-580.
London: Cambridge University Press.
CHASE, D. G. & ERLANDSEN, S. (1978). The morphology of cell-microbe junctions at the apical
surface of rat ileal enterocytes. J. Cell Biol. 79, 262 a.
CLEVELAND, L. R. & GRIMSTONE, A. V. (1964). The fine structure of the flagellate Mixotricha
paradoxa and its associated micro-organisms. Proc. R. Soc. B 159, 668-686.
ELLISMAN, M. H., RASH, J. E., STAEHELIN, L. A. & PORTER, K. R. (1976). Studies of excitable
membranes. II. A comparison of specializations at neuromuscular junctions and nonjunctional
sarcolemmas of mammalian fast and slow twitch muscle fibers. J. Cell Biol. 68, 752-774.
GILULA, N. B. (1974). Junctions between cells. In Cell Communication (ed. R. P. Cox), pp. 1-29.
New York: Wiley.
HENRY, S. M. (1966). Symbiosis, vol. 1. New York and London: Academic Press.
HEUSER, J. E. & REESE, T. S. (1977). Structure of the synapse. In Handbook of Physiology,
sect. I: The Nervous System, vol. 1: Cellular Biology of Neurons (ed. E. R. Kandel), pp.
261-294. Baltimore: Williams and Wilkins.
HOLLANDE, A. & CARRUETTE-VALENTIN, J. (1970). La ligne'e des Pyrsonymphines et les caracteres infrastructuraux communs aux genres Opisthomitus, Oxymonas, Saccinobacculus,
Pyrsonympha et Streblomastix. C. r. hebd. Sianc. Acad. Sci., Paris 270, 1587-1590.
HOLLANDE, A. & CARRUETTE-VALENTIN, J. (1971). Les atractophores, l'induction du fuseau et la
division cellulaire chez les hypermastigines: 6tude infrastructurale et revision syst6matique
des Trichonymphines et des Spirotrichonymphines. Protistologica 7, 5-100.
HOLLANDE, A. & CARRUETTE-VALENTIN, J. (1972). Le probleme du centrosome et la cryptoleuromitose atractophorienne chez Lophomonas striata. Protistologica 8, 267-278.
HOLLANDE, A. & VALENTIN, J. (1969). Appareil de Golgi, pinocytose, lysosomes, mitochondries,
bacte'ries symbiotiques, atractophores et pleuromitose chez les Hypermastigines du genre
Joenia. Affinite's entre Joeniides et Trichomonadines. Protistologica 5, 39-86.
KIRBY, H. (1941a). Organisms living on and in Protozoa. In Protozoa in Biological Research
(ed. G. N. Cakins & F. M. Summers), pp. 1009-1113. New York: Columbia University Press.
KIRBY, H. (1941 b). Devescovinidflagellatesof termites. I. The genus Devescovina. Univ. Calif.
Pubis Zool. 45, 1-92.
KIRBY, H. (1942 a). Devescovinidflagellatesof termites. II. The genera Caduceia and Macrotrichomonas. Univ. Calif. Publs Zool. 45, 93-166.
KIRBY, H. (1942 b). Devescovinidflagellatesof termites. III. The genera Foaina and Parajoenia.
Univ. Calif. Publs Zool. 45, 167-246.
KIRBY, H. (1945). Devescovinid flagellates of termites. IV. The genera Metadevescovina and
Pseudodevescovina. Univ. Calif. Publs Zool. 45, 247-318.
KIRBY, H. (1949). Devescovinid flagellates of termites. V. The genus Hyperdevescovina, the
genus Bullanympha, and undescribed or unrecorded species. Univ. Calif. Publs Zool. 45,
3I9-422-
A. (1969). Sur la veture schizophytique des Flagelles symbiotes de Termites. C. r.
hebd. Sianc. Acad. Sci., Paris 268, 2585-2587.
MCNUTT, N. S. (1977). Freeze-fracture techniques and applications to the structural analysis
of the mammalian plasma membrane. In Dynamic Aspects of Cell Surface Organisation (ed.
G. Poste & G. L. Nicolson), Cell Surface Reviews, vol. 3, pp. 75-126. New York: Elsevier/
North-Holland.
NEUTRA, M. R. (1979). Attachment of bacteria to epithelial cells in primate large intestine:
EM and freeze-fracture studies. Anat. Rec. 193, 636a.
NEUTRA, M. R. (1980). Prokaryotic-eukaryotic cell junctions: attachment of spirochetes and
flagellated bacteria to primate large intestinal cells. J. Ultrastruct. Res. 70, 186203.
SLEYTR, U. B. (1978). Regular arrays of macromolecules on bacterial cell walls: structure,
chemistry, assembly, and function. Int. Rev. Cytol. 53, 1-64.
SMITH, H. E. & ARNOTT, H. J. (1974). Epi- and endobiotic bacteria associated with Pyrsonympha
vertens. Trans. Am. microsc. Soc. 93, 180-194.
LAVETTE,
352
5. L. Tamm
H. E., BUHSE, H. E. JR & STAMLER, S. J. (1975). Possible formation and development of
spirochaete attachment sites found on the surface of symbiotic polymastigoteflagellatesof the
termite Reticulitermesflavipes.BioSystems 7, 374-379.
SNELLEN, J. E. & SAVAGE, D. C. (1978). Freeze-fracture study of the filamentous, segmented
microorganism attached to the murine small bowel. J. Bad. 134, 1099-1107.
SONNEBORN, T. M. (1970). Determination, development, and inheritance of the structure of the
cell cortex. Symp. int. Soc. Cell Biol., vol. 9, pp. 1-13. New York and London: Academic
Press.
STAEHELIN, L. A. (1974). Structure and function of intercellular junctions. Int. Rev. Cytol.
39, 191-283.
STARR, M. P. (1975). A generalized scheme for classifying organismic associations. In Symbiosis,
Symp. Soc. exp. Biol. (ed. D. H. Jennings & D. L. Lee), 29, pp. 1-20. London: Cambridge
University Press.
TAMM, S. L. (1976). Properties of a rotary motor in eukaryotic cells. In Cell Motility (ed.
R. Goldman, T. Pollard & J. Rosenbaum). Cold Spring Harbor Conferences on Cell Proliferation, vol. 3, pp. 949-967. New York: Cold Spring Harbor Laboratory.
TAMM, S. L. (1978 a). Laser microbeam study of a rotary motor in termite flagellates. Evidence
that the axostyle complex generates torque. J. Cell Biol. 78, 76-92.
TAMM, S. L. (19786). Membrane specializations associated with the attachment of prokaryotes
to a eukaryotic cell. J. Cell Biol. 79, 216 a.
TAMM, S. L. (1978c). Prokaryoticflagellapropel a eukaryotic cell. J. Cell Biol. 79, 310a.
TAMM, S. L. (1979). Membrane movements and fluidity during rotational motility of a termite
flagellate. J. Cell Biol. 80, 141-149.
TAMM, S. L. & TAMM, S. (1974). Direct evidence for fluid membranes. Proc. natn. Acad. Sci.
U.S.A. 71, 4580-4593.
TAMM, S. L. & TAMM, S. (1976). Rotary movements and fluid membranes in termite flagellates.
J. Cell Sci. 20, 619-639.
WAGNER, R. C. & BARRNETT, R. J. (1974). The fine structure of prokaryotic-eukaryotic cell
junctions. J. Ultrastruct. Res. 48, 404-413.
SMITH,
(Received 14 November 1979)
You might also like
- Comparative Cell Membranes and Transport Lab ReportDocument10 pagesComparative Cell Membranes and Transport Lab Reportapi-314520592100% (4)
- Kingdoms and Domains: An Illustrated Guide to the Phyla of Life on EarthFrom EverandKingdoms and Domains: An Illustrated Guide to the Phyla of Life on EarthRating: 4.5 out of 5 stars4.5/5 (4)
- Introduction To Pharmacology: Renato I. Dalmacio, RPH Pharmacology 325 College of PharmacyDocument40 pagesIntroduction To Pharmacology: Renato I. Dalmacio, RPH Pharmacology 325 College of PharmacyJon Paulo Bautista100% (3)
- Microbial Physiology LectureDocument88 pagesMicrobial Physiology LectureDaniNo ratings yet
- STreptomyces Plasmid GiantDocument3 pagesSTreptomyces Plasmid GiantA2 ZNo ratings yet
- The Key Role of The Surface Membrane in Why Gastropod Nacre Grows in TowersDocument6 pagesThe Key Role of The Surface Membrane in Why Gastropod Nacre Grows in TowersfronterasdelconNo ratings yet
- Field JCellBiol 2011Document10 pagesField JCellBiol 2011huouinkyoumaNo ratings yet
- (H2) CI2.10 - Genetics of Bacteria (N) (STDT)Document37 pages(H2) CI2.10 - Genetics of Bacteria (N) (STDT)Timothy HandokoNo ratings yet
- A4. Thilander H, Bloom G. Cell Contacts in Oral Epithelia. J Perio Res 1968Document16 pagesA4. Thilander H, Bloom G. Cell Contacts in Oral Epithelia. J Perio Res 1968Daniel CubasNo ratings yet
- Deep-Sea Bacteria Harboring Bacterial Endosymbionts in A Cytoplasm - 3D Electron Microscopy by Serial Ultrathin Sectioning of Freeze-Substituted SpecimenDocument3 pagesDeep-Sea Bacteria Harboring Bacterial Endosymbionts in A Cytoplasm - 3D Electron Microscopy by Serial Ultrathin Sectioning of Freeze-Substituted SpecimenFernandoNo ratings yet
- The Three-Dimensional Structure of The Cytostome-CytopharynxDocument11 pagesThe Three-Dimensional Structure of The Cytostome-CytopharynxUNIG Aluana SantanaNo ratings yet
- A Comparative Cytological Analysis of Fu20160623 13956 q2w8t5 With Cover PageDocument15 pagesA Comparative Cytological Analysis of Fu20160623 13956 q2w8t5 With Cover PageCristián MaximilianoNo ratings yet
- Bacteria TermitasDocument8 pagesBacteria TermitasRashaverak RomNo ratings yet
- FibroblastDocument6 pagesFibroblastMOISES AREVALONo ratings yet
- Spirogyra (Spiro Coiled and Gyra Curved) .: External Features and Cell StructureDocument112 pagesSpirogyra (Spiro Coiled and Gyra Curved) .: External Features and Cell StructureNinjax 1805No ratings yet
- NIH Public Access: Bacterial Swarming: A Model System For Studying Dynamic Self-AssemblyDocument31 pagesNIH Public Access: Bacterial Swarming: A Model System For Studying Dynamic Self-AssemblyverdosNo ratings yet
- tmp8556 TMPDocument26 pagestmp8556 TMPFrontiersNo ratings yet
- Bab 6 Sruktur Dan Fungsi Sel Bakteria (I)Document9 pagesBab 6 Sruktur Dan Fungsi Sel Bakteria (I)Shakira AmiraNo ratings yet
- Core Idea 2) Genetics and Inheritance: 2.9A) Structure and Organisation of Prokaryotic DnaDocument10 pagesCore Idea 2) Genetics and Inheritance: 2.9A) Structure and Organisation of Prokaryotic DnaReyes ChengNo ratings yet
- PIIS0969212608003857Document11 pagesPIIS0969212608003857Rin ChanNo ratings yet
- Lecture 2 - Bacterial Cell Structure and FunctionDocument90 pagesLecture 2 - Bacterial Cell Structure and FunctionOdurNo ratings yet
- Lampbrush ChromosomesDocument8 pagesLampbrush ChromosomesSurajit BhattacharjeeNo ratings yet
- Lab 9Document3 pagesLab 9alyssa cambaNo ratings yet
- Chloroplast SymbiosisDocument10 pagesChloroplast SymbiosisMaria Quesada BrunaNo ratings yet
- Lom2003 MicrosporidiaFishPhylogenDocument21 pagesLom2003 MicrosporidiaFishPhylogenJorge Manuel Cardenas CallirgosNo ratings yet
- Biyoloji Ödev Yazısı KaynakDocument4 pagesBiyoloji Ödev Yazısı KaynakbirlikteyizNo ratings yet
- Micro CH 2a StructureDocument155 pagesMicro CH 2a StructureBernadette Joyce PascualNo ratings yet
- The Physiology of Prokaryotic CellDocument22 pagesThe Physiology of Prokaryotic CellAnnysaNurmalaSariNo ratings yet
- The Bacterial Cell EnvelopeDocument19 pagesThe Bacterial Cell EnvelopetheiasitNo ratings yet
- Nichols1996 ConjunctivaDocument24 pagesNichols1996 Conjunctivamono1144No ratings yet
- Drosophila Melanogaster: The Inter, Actions of Some Moi Phogenetic GenesDocument16 pagesDrosophila Melanogaster: The Inter, Actions of Some Moi Phogenetic GenesDe Hoyos TorresNo ratings yet
- Nematocitos PolypodiumDocument11 pagesNematocitos PolypodiumKevin RucciNo ratings yet
- Essential Biology 2.2 and 2.3Document7 pagesEssential Biology 2.2 and 2.3MrNewbsNo ratings yet
- Southworth Et Al 2001-Male GametogenesisDocument16 pagesSouthworth Et Al 2001-Male GametogenesisAde MulyanaNo ratings yet
- BIO350 BacStrucDocument65 pagesBIO350 BacStrucPrincewill SeiyefaNo ratings yet
- Bacterial Flegella, Pilli and Cell MembraneDocument12 pagesBacterial Flegella, Pilli and Cell MembraneSai SatishNo ratings yet
- Geobiology - 2020 - LeeDocument10 pagesGeobiology - 2020 - LeeDuy Pham HoangNo ratings yet
- Diliman, Florilyn - Laboratory-Activity-3Document3 pagesDiliman, Florilyn - Laboratory-Activity-3Florilyn Apatan DilimanNo ratings yet
- Fine Structure and Morphogenic Movements in The Gastrula of The Treefrog, Hyla RegillaDocument24 pagesFine Structure and Morphogenic Movements in The Gastrula of The Treefrog, Hyla Regillaimanuel kuneNo ratings yet
- Laporan Genetika Giant ChromosomeDocument21 pagesLaporan Genetika Giant ChromosomeSyaifulBachryNo ratings yet
- Chapter 2. The Plasma Membrane & Cytoplasm: ObjectivesDocument24 pagesChapter 2. The Plasma Membrane & Cytoplasm: ObjectivesRichard BradshawNo ratings yet
- Driks-Las EsporasDocument3 pagesDriks-Las EsporasRenan Saldaña RochaNo ratings yet
- Morphology and Fine StructureDocument67 pagesMorphology and Fine StructureDiksha PadamNo ratings yet
- Molecules 25 00223Document19 pagesMolecules 25 00223Muh. RizalNo ratings yet
- tmp2CC8 TMPDocument8 pagestmp2CC8 TMPFrontiersNo ratings yet
- Lynn Margulis and The Endosymbiont Hypothesis: 50 Years LaterDocument3 pagesLynn Margulis and The Endosymbiont Hypothesis: 50 Years LaterJeisson GarzonNo ratings yet
- Bacteriology and Virology by KaziDocument35 pagesBacteriology and Virology by KaziMohibNo ratings yet
- Palaeontology - 2003 - Rouget - Embryonic Ammonoid Shell Features Intraspecific Variation RevisitedDocument12 pagesPalaeontology - 2003 - Rouget - Embryonic Ammonoid Shell Features Intraspecific Variation RevisitedFossilpremiaNo ratings yet
- Meningitidis), or More Than Once To Produce A Chain (Streptococcus Pyogenes), Divides Regularly in Two Planes at RightDocument14 pagesMeningitidis), or More Than Once To Produce A Chain (Streptococcus Pyogenes), Divides Regularly in Two Planes at RightMichael Vincent Pizarro BarbaNo ratings yet
- Reproduction in BacteriaDocument18 pagesReproduction in Bacteriaسيفل إسلامNo ratings yet
- Area Opaca (Figures 1, 2) - The Latter Region Contributes Only To ExtraembryonicDocument13 pagesArea Opaca (Figures 1, 2) - The Latter Region Contributes Only To ExtraembryonicgiuseppegnrNo ratings yet
- Bacteria MorphologyDocument38 pagesBacteria MorphologyFuad Hasan Pranto 1921147049No ratings yet
- Cells: Nuclear Envelope Proteins Modulating The Heterochromatin Formation and Functions in Fission YeastDocument21 pagesCells: Nuclear Envelope Proteins Modulating The Heterochromatin Formation and Functions in Fission YeastLê Khánh ToànNo ratings yet
- Gupta 2011Document12 pagesGupta 2011nyxcNo ratings yet
- Molecular Map of The Desmosomal PlaqueDocument12 pagesMolecular Map of The Desmosomal PlaqueLucas TobingNo ratings yet
- Phage DisplayDocument26 pagesPhage DisplaySurajit BhattacharjeeNo ratings yet
- Biology Marking For Third Term 2023Document13 pagesBiology Marking For Third Term 2023Mutaganda Ami fideleNo ratings yet
- Elife 91194 v1Document30 pagesElife 91194 v1PranavNo ratings yet
- Molecular Phylogenetics and EvolutionDocument14 pagesMolecular Phylogenetics and EvolutionNadin AriaNo ratings yet
- Volland Et Al 2022Document7 pagesVolland Et Al 2022Juan Sebastian Rodriguez MartinezNo ratings yet
- Dependence of Local Cell Density On Concentric RinDocument9 pagesDependence of Local Cell Density On Concentric RinMukhaddam MuhammadNo ratings yet
- FAD1001 TutorialsDocument14 pagesFAD1001 TutorialsKhairul AmeenNo ratings yet
- 2B Carbs and LipidsDocument26 pages2B Carbs and LipidsShawn NagraNo ratings yet
- Ion ChannelsDocument22 pagesIon ChannelsGanesh V GaonkarNo ratings yet
- CorningDocument272 pagesCorningAvinashkumar KaparapuNo ratings yet
- Cell Transport Review Worksheet: Complete The Table by Checking The Correct Column For Each StatementDocument3 pagesCell Transport Review Worksheet: Complete The Table by Checking The Correct Column For Each StatementMa.Janice GarciaNo ratings yet
- Endocrinology Exam KeyDocument11 pagesEndocrinology Exam KeyLouise SalvadorNo ratings yet
- TNPSC Group 2 Model Question Paper 11 General StudiesDocument31 pagesTNPSC Group 2 Model Question Paper 11 General StudiesNARUTO UZUMAKINo ratings yet
- AP Bio Notes FullDocument54 pagesAP Bio Notes FullThatKidNo ratings yet
- PEROXISOMESDocument3 pagesPEROXISOMESFajar Emman AsmatNo ratings yet
- AP Biology SGO ContentDocument5 pagesAP Biology SGO ContentIrina ShutovskyNo ratings yet
- NUR11O1 Integrated Human Anatomy and Physiology Department of Biology Institute of Arts and Sciences Far Eastern UniversityDocument5 pagesNUR11O1 Integrated Human Anatomy and Physiology Department of Biology Institute of Arts and Sciences Far Eastern UniversityYda Maxine PalmaNo ratings yet
- Unit 2 Practice Test PDFDocument5 pagesUnit 2 Practice Test PDFCandace LawNo ratings yet
- Code RCH 2003Document23 pagesCode RCH 2003Rhenso Victor Albites CondoriNo ratings yet
- Department of Education Mababanaba National High SchoolDocument5 pagesDepartment of Education Mababanaba National High SchoolMaricris LamugNo ratings yet
- Introduction To Cell PhysiologyDocument39 pagesIntroduction To Cell Physiologywaqas_xsNo ratings yet
- Post Lab (Exercise 1 Compound Microscope)Document66 pagesPost Lab (Exercise 1 Compound Microscope)Jenilyn FarnacioNo ratings yet
- Botany 2019-2020 Botany Xi Book LetDocument194 pagesBotany 2019-2020 Botany Xi Book LetAmmad U dinNo ratings yet
- Nature and Composition of Plants NotesDocument15 pagesNature and Composition of Plants NotesMc Wilson DecenaNo ratings yet
- Biology Lecture Membrane ProteinsDocument7 pagesBiology Lecture Membrane ProteinslimesoxroxNo ratings yet
- 2021 LIFE SCIENCES GR 10 ATP (Life at Molecular Cellular and Tissue Level)Document4 pages2021 LIFE SCIENCES GR 10 ATP (Life at Molecular Cellular and Tissue Level)Ejay OlebogengNo ratings yet
- WORKSHEET 2.2 Cell Structure and FunctionDocument4 pagesWORKSHEET 2.2 Cell Structure and Functionemaran100% (1)
- Comparative Evaluation of Alternate Emergency FixativesDocument14 pagesComparative Evaluation of Alternate Emergency FixativesIJAR JOURNALNo ratings yet
- 1st Lec On Endo Physiology by DR RoomiDocument24 pages1st Lec On Endo Physiology by DR RoomiMudassar RoomiNo ratings yet
- Exam98 5Document12 pagesExam98 5Harun MohamedNo ratings yet
- Cells Are The Basic Unit of Life. The Cell Theory States ThatDocument4 pagesCells Are The Basic Unit of Life. The Cell Theory States ThatMet XiiNo ratings yet
- Cell OrgenellesDocument10 pagesCell OrgenellesShruti TiwariNo ratings yet
- Video Recap of Cell Transport by Amoeba SistersDocument2 pagesVideo Recap of Cell Transport by Amoeba Sisters5zhrbrtbwdNo ratings yet
- III. Biological Membranes: Brenda Leady Fall 2005Document71 pagesIII. Biological Membranes: Brenda Leady Fall 2005Rif'atul AlfiyahNo ratings yet