Professional Documents
Culture Documents
Méthode de Nomenclature Chimique
Uploaded by
Fedoua Benamer0 ratings0% found this document useful (0 votes)
17 views1 pagedhklihuy
Original Title
fchbjh
Copyright
© © All Rights Reserved
Available Formats
DOCX, PDF, TXT or read online from Scribd
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this Documentdhklihuy
Copyright:
© All Rights Reserved
Available Formats
Download as DOCX, PDF, TXT or read online from Scribd
0 ratings0% found this document useful (0 votes)
17 views1 pageMéthode de Nomenclature Chimique
Uploaded by
Fedoua Benamerdhklihuy
Copyright:
© All Rights Reserved
Available Formats
Download as DOCX, PDF, TXT or read online from Scribd
You are on page 1of 1
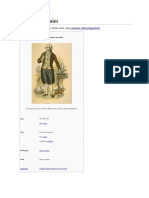
Traité Élémentaire de Chimie (Elementary Treatise of Chemistry) - Antoine Lavoisier
Traité Élémentaire de Chimie, 1789, available in English as Elementary Treatise of
Chemistry
Description: This book was intended as an introduction to new theories in chemistry and
as such, was one of the first Chemistry textbooks.[9]
Importance: Introduction, Influence. Aside from being one of the first chemistry
textbooks, the book was one of the first to state the Law of conservation of mass, define
a chemical element, and contain a list of known elements.[10][3]:154[11]:410
Méthode de Nomenclature Chimique[edit]
Guyton de Morveau, L. B.; Lavoisier, A. L.; Berthollet, C. L.; de Fourcroy, A. F.
Méthode de Nomenclature Chimique, Paris, 1787, available in English as Chymical
Nomenclature.
Some details and a picture available at IUPAC nomenclature#History
Description: This publication laid out a logical system for naming chemical
substances (mainly chemical elements and inorganic compounds).
Importance: Prior to this publication, a multitude of names were often used for the same
substance. This publication led to an international consensus on how to name chemical
substances.
A New System of Chemical Philosophy[edit]
A New System of Chemical Philosophy - John Dalton, 1808–1827
Description: This book explained Dalton's theory of atoms and its applications to
chemistry.
Importance: Topic Creator, Breakthrough, Influence. The book was one of the first to
describe a modern atomic th
Sdxfghnk,lmlkkkkkkkkkkkkkkkkkk
You might also like
- Tellurium in Organic Synthesis: Second, Updated and Enlarged EditionFrom EverandTellurium in Organic Synthesis: Second, Updated and Enlarged EditionNo ratings yet
- Spray TowerDocument3 pagesSpray TowerFedoua BenamerNo ratings yet
- Antoine LavoisierDocument1 pageAntoine LavoisierCreistel KhayNo ratings yet
- Antoine Lavoisier: By: Roberto ÁvilaDocument5 pagesAntoine Lavoisier: By: Roberto ÁvilaROBERTO AVILA ACOSTANo ratings yet
- Antoine-Laurent de Lavoisier: Ferme G N Rale Ancien R GimeDocument3 pagesAntoine-Laurent de Lavoisier: Ferme G N Rale Ancien R GimeValerie Ann FranciscoNo ratings yet
- Module 3 OrgchemDocument7 pagesModule 3 OrgchemJHUNNTY LOZANONo ratings yet
- The Sceptical Chymist: Antoine-Laurent de LavoisierDocument1 pageThe Sceptical Chymist: Antoine-Laurent de LavoisierSeijuro AkashiNo ratings yet
- Antoine LavoisierDocument8 pagesAntoine LavoisierVishal TilokaniNo ratings yet
- Antoine Lavoisier: Presented by Amina.A Physical ScienceDocument8 pagesAntoine Lavoisier: Presented by Amina.A Physical SciencerazeenasalamNo ratings yet
- Antoine-Laurent de Lavoisier (: Lav - Ee - Ay LƏ - Zee-AyDocument1 pageAntoine-Laurent de Lavoisier (: Lav - Ee - Ay LƏ - Zee-AyZenny JabuenNo ratings yet
- The Father of ChemistryDocument2 pagesThe Father of ChemistryJaeho BaeNo ratings yet
- LavoisierDocument1 pageLavoisierEduardo LaloNo ratings yet
- O Chapter 1Document15 pagesO Chapter 1Anupa GhoseNo ratings yet
- Chemical Bonding and StructureDocument42 pagesChemical Bonding and Structurecoga194No ratings yet
- Development of the Periodic Table and Contributions of Key ScientistsDocument12 pagesDevelopment of the Periodic Table and Contributions of Key Scientistsalfie abadNo ratings yet
- Introduction to Chemistry History and DefinitionsDocument7 pagesIntroduction to Chemistry History and DefinitionsDiana AnchetaNo ratings yet
- Anti OneDocument7 pagesAnti OneJade ClarkNo ratings yet
- Father of ChemistryDocument15 pagesFather of ChemistryApril Angela MonterosoNo ratings yet
- Chapter 1 General View of Chemistry and MatterDocument17 pagesChapter 1 General View of Chemistry and MatterJoshua Kulot100% (1)
- ModernchemistryDocument7 pagesModernchemistryprinceuduma2021No ratings yet
- Antoine LavoisierDocument3 pagesAntoine LavoisierMark allenNo ratings yet
- IUPAC Nomenclature: Blue Book Red Book Green Book Gold BookDocument9 pagesIUPAC Nomenclature: Blue Book Red Book Green Book Gold BookRakesh RanjanNo ratings yet
- Introduction to ChemistryDocument5 pagesIntroduction to ChemistryHina RabbaniNo ratings yet
- Chemical Bonding and StructureDocument45 pagesChemical Bonding and Structurehymerchmidt100% (1)
- Antoine Lavoisier ChemDocument5 pagesAntoine Lavoisier ChemshristiNo ratings yet
- 146-148 7atom-ADocument3 pages146-148 7atom-AToh YangNo ratings yet
- Organic Chemistry DefinitionsDocument1 pageOrganic Chemistry DefinitionsLocked Up ColossalNo ratings yet
- Chemistry: Chemistry - The Study of Interactions of Chemical Substances WithDocument14 pagesChemistry: Chemistry - The Study of Interactions of Chemical Substances WithmahmoodurrahmankaziNo ratings yet
- NomenclatureDocument140 pagesNomenclatureAnonymous G296iP5No ratings yet
- Adge CompiledDocument18 pagesAdge CompiledNovelyn LumboyNo ratings yet
- Development of Atomic TheoryDocument13 pagesDevelopment of Atomic TheoryAbegail GabineNo ratings yet
- Early History of the Atom and Dalton's ContributionsDocument3 pagesEarly History of the Atom and Dalton's ContributionsMarivic Tanque SedayonNo ratings yet
- REVIEWERDocument10 pagesREVIEWERNathasha Marie BanalNo ratings yet
- Corpuscles To Chemical Atomic TheoryDocument17 pagesCorpuscles To Chemical Atomic TheorySta Lucia National High SchoolNo ratings yet
- The Origin of The Mole ConceptDocument1 pageThe Origin of The Mole ConceptJorge Alfonso Patiño FlorezNo ratings yet
- Organic Chemistry Describes The StructuresDocument3 pagesOrganic Chemistry Describes The StructuresRevanthNo ratings yet
- Module 1Document16 pagesModule 1Joan Dadivas AmancioNo ratings yet
- Untitled Extract Pages - PDGDFDocument1 pageUntitled Extract Pages - PDGDFofajs84No ratings yet
- Antoine LavoisierDocument9 pagesAntoine LavoisierLeo HuangNo ratings yet
- Branches of ChemistryDocument9 pagesBranches of ChemistryYhan Brotamonte Boneo100% (1)
- Introduction To ChemistryDocument17 pagesIntroduction To ChemistryLeovihilda BalalangNo ratings yet
- Organic Chemistry MasterDocument128 pagesOrganic Chemistry MasterLeigh DensingNo ratings yet
- History of Organic Chemistry PDFDocument14 pagesHistory of Organic Chemistry PDFIRISH REEM LINAOTANo ratings yet
- Antoine LavoisierDocument7 pagesAntoine Lavoisierapi-286661738No ratings yet
- 2000 235 Octobre p39 Bues PDFDocument4 pages2000 235 Octobre p39 Bues PDFسمير دبيليNo ratings yet
- NatSci 301 Instructional Learning Module 1Document7 pagesNatSci 301 Instructional Learning Module 1Grace VillanuevaNo ratings yet
- Chem 110Document110 pagesChem 110Raymond LO OtucopiNo ratings yet
- Historical ReviewDocument2 pagesHistorical ReviewSidny BatulanNo ratings yet
- Chapter 2 Atoms Molecules IonsDocument55 pagesChapter 2 Atoms Molecules IonsVuthy CheyNo ratings yet
- Chemistry As Science: The Sceptical ChymistDocument2 pagesChemistry As Science: The Sceptical Chymistterran2020No ratings yet
- Atoms and MoleculesDocument1 pageAtoms and Moleculessaisha khannaNo ratings yet
- Chemistry As Scienceaazzqqww: The Sceptical ChymistDocument2 pagesChemistry As Scienceaazzqqww: The Sceptical Chymistterran2020No ratings yet
- Chemical Reactions Module: Figure 2: Aristotle Believed That Everything in The World Was ComposedDocument9 pagesChemical Reactions Module: Figure 2: Aristotle Believed That Everything in The World Was ComposedNabeela TunisNo ratings yet
- Chemistry As Scienceaazzqq: The Sceptical ChymistDocument2 pagesChemistry As Scienceaazzqq: The Sceptical Chymistterran2020No ratings yet
- 1 - Introduction - To - Chemistry 1Document19 pages1 - Introduction - To - Chemistry 1Hosnie AlfonsoNo ratings yet
- Lesson 5 Chemical Nomenclature Part 1Document37 pagesLesson 5 Chemical Nomenclature Part 1mywaykissNo ratings yet
- Chemical Nomenclature - WikipediaDocument11 pagesChemical Nomenclature - WikipediaJamella Jael Jumao-asNo ratings yet
- CHAPTER II Atoms and The Atomic TheoryDocument19 pagesCHAPTER II Atoms and The Atomic TheoryEunice Rzn NmttcNo ratings yet
- Organic ChemistryDocument44 pagesOrganic ChemistryJopie ArandaNo ratings yet
- A Guide For ChemistsDocument188 pagesA Guide For Chemistsneculaitarabuta100% (2)
- Understanding T-WPS OfficeDocument6 pagesUnderstanding T-WPS OfficeJoebelle GiananNo ratings yet
- Cyclar PDFDocument3 pagesCyclar PDFFedoua BenamerNo ratings yet
- Les Phénomènes D'adsorption, D'échange Ou de Rétention À L'interface Solide-Solution Aqueuse.1. Connaissance Des Propriétés Structurales, Texturales Et Superficielles Des SolidesDocument11 pagesLes Phénomènes D'adsorption, D'échange Ou de Rétention À L'interface Solide-Solution Aqueuse.1. Connaissance Des Propriétés Structurales, Texturales Et Superficielles Des SolidesFedoua BenamerNo ratings yet
- 1 Step Oh + HCL CL + H O: AaaaayyyyyyyyyyyyyyyyyyyyzzzzzzzzzzzzzzzzzzeeeeeeeeDocument1 page1 Step Oh + HCL CL + H O: AaaaayyyyyyyyyyyyyyyyyyyyzzzzzzzzzzzzzzzzzzeeeeeeeeFedoua BenamerNo ratings yet
- KphyifyfhvDocument1 pageKphyifyfhvFedoua BenamerNo ratings yet
- KJHGFHDocument1 pageKJHGFHFedoua BenamerNo ratings yet
- KphyifyfhvDocument1 pageKphyifyfhvFedoua BenamerNo ratings yet
- Gas Scrubbing - General: Synonyms, Abbreviations And/or Process NamesDocument6 pagesGas Scrubbing - General: Synonyms, Abbreviations And/or Process NamesFedoua BenamerNo ratings yet
- PouyghjhggfgbnDocument1 pagePouyghjhggfgbnFedoua BenamerNo ratings yet
- JbgfffffffhcyfDocument1 pageJbgfffffffhcyfFedoua BenamerNo ratings yet
- B, N, CNNBJKZ N, BBJFLK, NJKZHFJKNC, C, NffffffffjjjjfkljiDocument1 pageB, N, CNNBJKZ N, BBJFLK, NJKZHFJKNC, C, NffffffffjjjjfkljiFedoua BenamerNo ratings yet
- Surf ChemDocument1 pageSurf ChemFedoua BenamerNo ratings yet
- 4 Phrases: Come Is A Verb, Letter Is A Noun and Great Is An AdjectiveDocument1 page4 Phrases: Come Is A Verb, Letter Is A Noun and Great Is An AdjectiveFedoua BenamerNo ratings yet
- XN, ZDocument1 pageXN, ZFedoua BenamerNo ratings yet
- The Grammatical Units and Elements of the English LanguageDocument1 pageThe Grammatical Units and Elements of the English LanguageFedoua BenamerNo ratings yet
- TtyyuuzzDocument2 pagesTtyyuuzzFedoua BenamerNo ratings yet
- ChimioDocument1 pageChimioFedoua BenamerNo ratings yet
- FD FDVDocument1 pageFD FDVFedoua BenamerNo ratings yet
- SurfchemDocument1 pageSurfchemFedoua BenamerNo ratings yet
- 1Document1 page1Fedoua BenamerNo ratings yet