Professional Documents
Culture Documents
Construction Proposal Form517604220170126 PDF
Uploaded by
mayasOriginal Title
Copyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
Construction Proposal Form517604220170126 PDF
Uploaded by
mayasCopyright:
Available Formats
SAN FRANCISCO BAY AREA SCIENCE FAIR SCIENTIFIC REVIEW COMMITTEE
PROJECT PROPOSAL FORM
This document must be completed by any student, grade 7 through 12, whose project involves
(even if only observing) human subjects, invertebrate or non-human vertebrate animals,
This form must
recombinant DNA, tissues, pathogenic agents, or controlled substances.
be completed, filed, and approved before starting your project and
no later than February 1.
When completed, mail this form to:
Greta Dahlke: 944 Reliez Station Rd., Lafayette, CA 94549
Student's Name: _____________________________________________ Grade: _______
Name of School: __________________________________________________________
Address of School: _________________________________________________________
Home phone #: ______________________ School phone #: ______________________
Where will you conduct your experimentation?
at school:_____ at home:_____ in the field:_____
at a research institution; e.g. university lab, medical center, industrial setting :_____
(If you plan to perform your work at a research institution, you must complete Section 6.)
7th, 8th, and 9th GRADERS
Check here _____ if you are working with humans and complete sections 1, 7, and 8.
Check here _____ if you are working with non-human animals, bacteria, or tissues and
complete sections 1, 7, and 8. Read Section 2 before completing Section 1.
10th, 11th, and 12th GRADERS
Check here _____ if your project involves humans and complete sections 1, 7, and 8.
Check here _____ if your project involves non-human vertebrates and complete sections 1, 4,
7, and 8. Read Section 2 before completing Section 1.
Check here _____ if your project involves bacteria or invertebrates and complete sections 1,
7, and 8. Read Section 2 before completing Section 1.
Check here _____ if your project involves recombinant DNA, pathogenic agents, or controlled
substances and complete sections 1, 4, 7, and 8. Read Section 3 before
completing Section 1.
Check here _____ if your project involves human or animal tissue and complete sections 1, 4, 5,
7, and 8. Read Section 5 before completing Section 1.
page 1 CONTINUE . . .
SECTION 1: To be completed by all students filing this proposal form.
a. What is the question and purpose of your study?
b. Describe your experimental procedure with as much detail as possible. Use and attach
additional page(s) if necessary. If your study involves a survey of humans, please attach a copy
of the survey questions and a copy of the informed consent/waiver form.
SECTION 2: To be completed by all students whose projects involve non-human vertebrate or
invertebrate animals. In your description for Section 1 question b, please include responses to the
following:
a. Are there any alternatives to the use of live animals in your project? If not, why did you find such
alternatives unacceptable for your study? Explain.
b. Describe in detail how the animals will be used. Identify (where appropriate) the species, strain, sex,
age, weight, source , and number of animals proposed for use. Remember to use the MINIMUM number of
animals you deem necessary for your study.
c. Explain the potential impact or contribution this research may have on the broad fields of biology or
medicine.
d. Provide detailed information on the animals' housing and environment. Also, provide information on
the veterinary medical/nursing care in the case of illness or emergency ; give the name of the veterinarian
or veterinary facility.
e. Explain what will happen to the animal(s) after the project is finished. If euthanasia will be performed
by a qualified scientist (students are not permitted to perform euthanasia), describe the method in detail.
page 2 CONTINUE . . .
SECTION 3: To be completed by all students whose projects involve recombinant DNA,
pathogenic agents, or controlled substances. In your description for Section 1 question b, please
include responses to the following:
a. What is the recombinant DNA, pathogen(s), or controlled substance(s) to be used?
b. What is the source of your experimental material?
SECTION 4: To be completed for all students whose projects involve non-human
vertebrate animals, recombinant DNA, tissues, pathogenic agents, or controlled substances by a
qualified scientist (someone who possesses an earned doctoral degree in science or medicine or
someone with a master's degree with equivalent experience and/or expertise).
" I certify that I have reviewed and approved the research proposal prior to the start of the study, that if the
student or supervising teacher is not trained in the necessary procedures I will ensure his/her training, and
that I will provide advice and supervision during the research. I hereby accept responsibility for the
condition and humane disposition of all living animals used in the project, both during and after its
completion. I am a qualified scientist with a working knowledge of the techniques to be used by the
student in this research plan."
Name of qualified scientist: __________________________________________________
Address: ________________________________________________________________
Title or degree: __________________________ Phone number: _____________________
Signature: ____________________________________________Date: _______________
SECTION 5: To be completed by all students whose projects involve human or animal tissues.
In your description for Section 1 question b, please include responses to the following:
a. What is the tissue and/or organ to be used?
b. What is the source of your material?
To be completed by the provider of your tissue when obtained from a non-commercial source:
"Human blood and blood products will be tested and documented free of AIDS and hepatitis B and C
antibodies and antigens. Human teeth will be certified free of blood and blood products."
"I certify that the above listed materials will be provided by me and that the student will not be involved in
the direct acquisition of the samples provided or purchased."
Provider's Name: ___________________________________ Title: _______________
Institution: _____________________________________________________________
Address: _________________________________________ Phone #: ______________
Provider's signature: ___________________________________ Date: _____________
page 3 CONTINUE . . .
SECTION 6: To be completed by all students whose experimentation is carried out in a
research facility. In your description for Section 1 question b, please include responses to the
following:
a. Where do you plan to do your experimentation?
b. How did you get the idea for your project?
c. Will you work on the project as part of a team or a group?
d. How independently will you work on the project?
"I certify that I have reviewed and approved the research proposal prior to the start of the study, that if the
student is not trained in the necessary procedures I will ensure his/her training, and that I will provide
advice and supervision during the research."
Name of supervising scientist: ________________________________________________
Signature: _________________________________________ Date: ________________
SECTION 7: To be completed by the sponsoring teacher of the student filing this form.
"I have read all the rules and regulations of the SFBASF regarding projects involving humans, animals,
recombinant DNA, tissues, pathogenic agents, and controlled substances and I have discussed these with
the student I am sponsoring. The student has read and understands the SFBASF SRC Guidelines,
Requirements, and Rules for student science projects and I will be responsible for this student's compliance
with the guidelines. I have collected and kept the signed informed consent/waiver forms for this student's
project (if it involves a survey of humans)."
Name of sponsoring teacher: ________________________________________________
Signature: __________________________________________ Date: _______________
SECTION 8: To be completed by all students filing this proposal form.
_____ YES _____ NO Did you read the Guidelines regarding the use of humans, animals,
recombinant DNA, tissue, pathogenic agents, or controlled substances
in a science fair project?
_____ YES _____ NO Did your sponsoring teacher go over the Guidelines regarding your science
fair project with you?
_____ YES _____ NO If your project involved a survey of human subjects, did you give your signed
informed consent/waiver forms to your sponsoring teacher?
Student's Signature: _____________________________________ Date: _____________
PARENT/GUARDIAN APPROVAL
"I have read and understand the risks and possible dangers in the sponsoring teacher-approved research
plan. I consent to my child participating in this research project."
Parent/Guardian signature: __________________________________________________
page 4 End of Document
You might also like
- ENVIRONMENT AND HUMANITY: Passbooks Study GuideFrom EverandENVIRONMENT AND HUMANITY: Passbooks Study GuideNo ratings yet
- Checklist For Adult Sponsor (1) : This Completed Form Is Required For ALL ProjectsDocument14 pagesChecklist For Adult Sponsor (1) : This Completed Form Is Required For ALL ProjectsPercen7No ratings yet
- Junior Science Symposium 2018Document2 pagesJunior Science Symposium 2018wes haungNo ratings yet
- CEDREC-ConFormPair-1 (1)Document3 pagesCEDREC-ConFormPair-1 (1)Nor-Haniah B. SulogNo ratings yet
- Methods:: Method 1 - ExperimentDocument9 pagesMethods:: Method 1 - ExperimentFatemaNo ratings yet
- ISEF FormsDocument14 pagesISEF FormsRoma CamilleNo ratings yet
- IACUCFORM+C Research Revised10.24.15Document13 pagesIACUCFORM+C Research Revised10.24.15Ethan Lance CuNo ratings yet
- Interactive Forms Intel ISEF 2015 PDFDocument14 pagesInteractive Forms Intel ISEF 2015 PDFsampdnimNo ratings yet
- Hltinf001 Student Workbook (Word) (c3) .v1.1Document61 pagesHltinf001 Student Workbook (Word) (c3) .v1.1Saima Rashid67% (6)
- Vertebrate Animal Form (5A) : Student's Name(s) Title of ProjectDocument2 pagesVertebrate Animal Form (5A) : Student's Name(s) Title of ProjectEmery FayNo ratings yet
- Form 5Document2 pagesForm 5Biblioteca Brígida ÁlvarezNo ratings yet
- Genetic Disorders ProjectDocument2 pagesGenetic Disorders ProjectbarkhaNo ratings yet
- A Level Biology Course ContentDocument3 pagesA Level Biology Course Contentapi-102850034No ratings yet
- Research Plan (To Accompany Form 1A)Document2 pagesResearch Plan (To Accompany Form 1A)api-484216705No ratings yet
- Integrated Basic Education Department - Senior High School LevelDocument10 pagesIntegrated Basic Education Department - Senior High School LevelJohn SanchezNo ratings yet
- Research II: Special Science Program Quarter 1 - Module 3: International Rules and Research FormsDocument17 pagesResearch II: Special Science Program Quarter 1 - Module 3: International Rules and Research FormsVandon BorelaNo ratings yet
- Checklist For Adult Sponsor (1) : This Completed Form Is Required For ALL ProjectsDocument14 pagesChecklist For Adult Sponsor (1) : This Completed Form Is Required For ALL ProjectsRoberto Delgado WebbNo ratings yet
- Unit 1 Test PsychologyDocument7 pagesUnit 1 Test Psychologycoach_rutNo ratings yet
- Career Civil Service Exam Final Revision-2Document14 pagesCareer Civil Service Exam Final Revision-2Leih Anne Quezon AnonuevoNo ratings yet
- ETHICS FORMH0+ (Clearance+Checklist)Document3 pagesETHICS FORMH0+ (Clearance+Checklist)Laurence Niña X. OrtizNo ratings yet
- 1A Student ChecklistDocument2 pages1A Student ChecklistRosselle De La CruzNo ratings yet
- 2020 MELC Enhanced Subjects for Regional Science High SchoolDocument8 pages2020 MELC Enhanced Subjects for Regional Science High SchoolCholline Zolina100% (3)
- Consent Form SEODocument2 pagesConsent Form SEOPeter Angelo GeleraNo ratings yet
- Pawpaw March 2014Document18 pagesPawpaw March 2014sansan2404No ratings yet
- Evaluation of Research PlanDocument7 pagesEvaluation of Research PlanJenelyn SibulboroNo ratings yet
- Requirements Level 2 OPDDocument5 pagesRequirements Level 2 OPDJAN CAMILLE LENONNo ratings yet
- Eapp Week 7Document5 pagesEapp Week 7Ruben AgacioNo ratings yet
- Sop03 Anx4 Ver1Document13 pagesSop03 Anx4 Ver1techiemedicNo ratings yet
- 1A Student Checklist FebcenDocument2 pages1A Student Checklist FebcenKimmmiNo ratings yet
- Hltinf001 Student Workbook (Word) (c3) .v1.1Document71 pagesHltinf001 Student Workbook (Word) (c3) .v1.1navjot kaur50% (2)
- 1011 Chestnut St. Alameda, CA 94501 T: (510) 523-1526 WDocument10 pages1011 Chestnut St. Alameda, CA 94501 T: (510) 523-1526 WalexrherreraNo ratings yet
- Qualified Scientist Form (2) : Student's Name(s) Title of ProjectDocument1 pageQualified Scientist Form (2) : Student's Name(s) Title of ProjectBiblioteca Brígida ÁlvarezNo ratings yet
- MODULE4-Designs A Research Used in Daily LifeDocument14 pagesMODULE4-Designs A Research Used in Daily LifeMira Rochenie CuranNo ratings yet
- Research Ethics Review Committee (Who Erc)Document4 pagesResearch Ethics Review Committee (Who Erc)Evan Permana PutraNo ratings yet
- Research Paper On Service AnimalsDocument4 pagesResearch Paper On Service Animalsibhuglbkf100% (1)
- An Autonomous Healthcare Provider for Remote CommunitiesDocument8 pagesAn Autonomous Healthcare Provider for Remote CommunitiescessNo ratings yet
- Adult Sponsor Checklist for Student Research ProjectsDocument6 pagesAdult Sponsor Checklist for Student Research ProjectsErijah YapNo ratings yet
- Medical Report For Ecology Action Intern Applicants: (Page 1 of 2)Document2 pagesMedical Report For Ecology Action Intern Applicants: (Page 1 of 2)Daniel SantiagoNo ratings yet
- Monthly Test PR2Document2 pagesMonthly Test PR2Jonathan PacificoNo ratings yet
- All 2023 Isef FormsDocument15 pagesAll 2023 Isef FormsIrish alexis AycochoNo ratings yet
- Application Procedure AssessmentDocument12 pagesApplication Procedure AssessmentYamuna MadamNo ratings yet
- WEEK-4 - LectureDocument2 pagesWEEK-4 - LectureBritney TamuraNo ratings yet
- 1A Student Checklist Research Plan InstructionsDocument2 pages1A Student Checklist Research Plan InstructionsEmmanuel ValenzuelaNo ratings yet
- 1A Student Checklist ISEF FORMDocument2 pages1A Student Checklist ISEF FORMKirzten Avril R. AlvarezNo ratings yet
- Student Checklist for Amygdalin ProjectDocument2 pagesStudent Checklist for Amygdalin ProjectJulyanna AteNo ratings yet
- All Ethical FormsDocument27 pagesAll Ethical Formsramandeep kaurNo ratings yet
- FullcsDocument37 pagesFullcsapi-344457268No ratings yet
- Experimental Research Design Thesis PDFDocument8 pagesExperimental Research Design Thesis PDFninavazquezhartford100% (2)
- Hltwhs002 Student Workbook (Word) (c3) .v1.1Document45 pagesHltwhs002 Student Workbook (Word) (c3) .v1.1yghimire100No ratings yet
- Checklist For Adult Sponsor (1) : This Completed Form Is Required For ALL ProjectsDocument14 pagesChecklist For Adult Sponsor (1) : This Completed Form Is Required For ALL Projectswhipped assNo ratings yet
- M2 FinalDocument61 pagesM2 FinalJeranz ColansiNo ratings yet
- Fetal Pig Research PaperDocument8 pagesFetal Pig Research Paperiyarmacnd100% (1)
- Practical Research-2: Quarter 2 - Module 17Document12 pagesPractical Research-2: Quarter 2 - Module 17Jerald M. MacapagalNo ratings yet
- Qualified Scientist FormDocument1 pageQualified Scientist FormlepoyNo ratings yet
- Las 3is Week 7 Sns OcenarDocument13 pagesLas 3is Week 7 Sns OcenarJun Elbert JaboliNo ratings yet
- Biology3 7a Dec12Document7 pagesBiology3 7a Dec12api-292477453No ratings yet
- Research PlanDocument2 pagesResearch Planapi-484250469No ratings yet
- PR1-ACTIVITY SHEET-WEEK1Document4 pagesPR1-ACTIVITY SHEET-WEEK1Geriza AshleyNo ratings yet
- NSCLC Feasibility QuestionnaireDocument4 pagesNSCLC Feasibility Questionnairezaq rewNo ratings yet
- Boysen Paints Calculator-1579263160926Document2 pagesBoysen Paints Calculator-1579263160926mayasNo ratings yet
- Boysen Paints Calculator-1579261731230Document2 pagesBoysen Paints Calculator-1579261731230mayasNo ratings yet
- Boysen Paints Calculator-1579261175845Document2 pagesBoysen Paints Calculator-1579261175845mayasNo ratings yet
- FBDDocument7 pagesFBDmayasNo ratings yet
- Boysen Paints Calculator-1579261166769Document2 pagesBoysen Paints Calculator-1579261166769mayasNo ratings yet
- Boysen Paints Calculator-1579260493111Document2 pagesBoysen Paints Calculator-1579260493111mayasNo ratings yet
- Boysen Paints Calculator-1579259585908 PDFDocument2 pagesBoysen Paints Calculator-1579259585908 PDFmayasNo ratings yet
- Boysen Paints Calculator-1579260064743Document2 pagesBoysen Paints Calculator-1579260064743mayasNo ratings yet
- Boysen Paints Calculator-1579260493111 PDFDocument2 pagesBoysen Paints Calculator-1579260493111 PDFmayasNo ratings yet
- A 2 PDFDocument1 pageA 2 PDFmayasNo ratings yet
- Boysen Paints Calculator-1579260058147Document2 pagesBoysen Paints Calculator-1579260058147mayasNo ratings yet
- Boysen Paints Calculator-1579260506426Document2 pagesBoysen Paints Calculator-1579260506426mayasNo ratings yet
- Almira Slab 2 DetailsDocument1 pageAlmira Slab 2 DetailsbobosNo ratings yet
- Boysen Paints Calculator-1579259925650Document2 pagesBoysen Paints Calculator-1579259925650mayasNo ratings yet
- Boysen Paints Calculator-1579259585908 PDFDocument2 pagesBoysen Paints Calculator-1579259585908 PDFmayasNo ratings yet
- Boysen Paints Calculator-1579259585908Document2 pagesBoysen Paints Calculator-1579259585908mayasNo ratings yet
- Boysen Paints Calculator-1579259578224Document2 pagesBoysen Paints Calculator-1579259578224mayasNo ratings yet
- Detail Two PDFDocument1 pageDetail Two PDFmayasNo ratings yet
- CE Roof Beam Framing Final - MiasDocument1 pageCE Roof Beam Framing Final - MiasmayasNo ratings yet
- CE Slab Detail Final 1 - Mias PDFDocument1 pageCE Slab Detail Final 1 - Mias PDFmayasNo ratings yet
- Measurement of Earthquakes - MarquezDocument36 pagesMeasurement of Earthquakes - MarquezmayasNo ratings yet
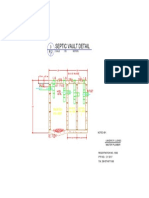
- Plsanitary Moni'sDocument1 pagePlsanitary Moni'smayasNo ratings yet
- CE Beam Detail Final 2 - MiasDocument1 pageCE Beam Detail Final 2 - MiasmayasNo ratings yet
- Img 20180325 0005 PDFDocument1 pageImg 20180325 0005 PDFmayasNo ratings yet
- Eelighting Moni'sDocument1 pageEelighting Moni'smayasNo ratings yet
- Eepower Moni'sDocument1 pageEepower Moni'smayasNo ratings yet
- Img 20180325 0004Document1 pageImg 20180325 0004bobosNo ratings yet
- Arch Moni'sDocument1 pageArch Moni'smayasNo ratings yet
- Plwatersupply Moni's PDFDocument1 pagePlwatersupply Moni's PDFmayasNo ratings yet
- Ordinary Annuity - MarquezDocument10 pagesOrdinary Annuity - MarquezmayasNo ratings yet
- Record Retention ChartDocument2 pagesRecord Retention ChartVatsalPatelNo ratings yet
- Dade BehringDocument85 pagesDade Behringmichaelburke47No ratings yet
- The Breast - Part 2 Dr. Roberto B. Acuña: Hormonal Non-HormonalDocument14 pagesThe Breast - Part 2 Dr. Roberto B. Acuña: Hormonal Non-HormonalMaf BNo ratings yet
- Nursing Care Plan for Diverticulitis PatientDocument3 pagesNursing Care Plan for Diverticulitis PatientLeny GallardoNo ratings yet
- Magadan Kardex SurgpavDocument2 pagesMagadan Kardex Surgpavdon7daneNo ratings yet
- HCD 1 PDFDocument714 pagesHCD 1 PDFDinaNo ratings yet
- Endocrine System Drugs and Their EffectsDocument3 pagesEndocrine System Drugs and Their EffectsJerica Jaz F. VergaraNo ratings yet
- Check your well-being with this situations listDocument2 pagesCheck your well-being with this situations listArlen Mae RayosNo ratings yet
- 713-Article Text-2302-1-10-20180613Document9 pages713-Article Text-2302-1-10-20180613nareNo ratings yet
- BARANGAY TrainingDocument2 pagesBARANGAY TrainingAngel BenateroNo ratings yet
- ICCD Power Point 2015Document16 pagesICCD Power Point 2015OhioOhioNo ratings yet
- Hypnosis For Beginners Written by DRDocument196 pagesHypnosis For Beginners Written by DRbharath_mv7100% (4)
- Handbook (Chemistry Workshop)Document34 pagesHandbook (Chemistry Workshop)DocsNo ratings yet
- Air Alert III PDFDocument8 pagesAir Alert III PDFEric Wijaya100% (1)
- Course Details - CHC32015 Certificate III in Community Services - Mark Oliphant CollegeDocument1 pageCourse Details - CHC32015 Certificate III in Community Services - Mark Oliphant CollegeSaroja ChhetriNo ratings yet
- Assessment and Diagnosis of StrokeDocument34 pagesAssessment and Diagnosis of StrokeMohd Khateeb100% (1)
- Mojwh 08 00237Document9 pagesMojwh 08 00237EndalkNo ratings yet
- Pathology ReportDocument2 pagesPathology ReportdenisdembskeyNo ratings yet
- Transparency Concerns Led To Energy Minister Change: Remembering An Uprising, 25 Years OnDocument88 pagesTransparency Concerns Led To Energy Minister Change: Remembering An Uprising, 25 Years OnThe Myanmar TimesNo ratings yet
- Epistaxis: Dr. Wachuka G. Thuku Tutorial Fellow-ENT RegistrarDocument32 pagesEpistaxis: Dr. Wachuka G. Thuku Tutorial Fellow-ENT RegistrarAlexNo ratings yet
- Jaundice-Review of Clinical Features, Differential Diagnosis and RemediesDocument13 pagesJaundice-Review of Clinical Features, Differential Diagnosis and RemediesDr-Dalya ShakirNo ratings yet
- 2013 State of Oklahoma EMS ProtocolsDocument493 pages2013 State of Oklahoma EMS ProtocolsMarian Ioan-Lucian100% (1)
- PC 4 NursingcareplanDocument2 pagesPC 4 Nursingcareplanapi-316769247No ratings yet
- Quality Certificate CHEPDocument2 pagesQuality Certificate CHEPKumar RajadhyakshNo ratings yet
- Foreign StudiesDocument2 pagesForeign StudiesNebea AdorableNo ratings yet
- Yoga Swing Installation InstructionsDocument10 pagesYoga Swing Installation Instructionskarmasherbs100% (2)
- 2 51 1588072528 6.ijmpsapr20206Document14 pages2 51 1588072528 6.ijmpsapr20206TJPRC PublicationsNo ratings yet
- ACC8278 Annual Report 2021Document186 pagesACC8278 Annual Report 2021mikeNo ratings yet
- Mixed Connective Tissue DiseaseDocument6 pagesMixed Connective Tissue Diseasemasa depanNo ratings yet
- Ati RN 2016 Mental HealthDocument6 pagesAti RN 2016 Mental HealthStan Tan0% (1)
- Dark Matter and the Dinosaurs: The Astounding Interconnectedness of the UniverseFrom EverandDark Matter and the Dinosaurs: The Astounding Interconnectedness of the UniverseRating: 3.5 out of 5 stars3.5/5 (69)
- Roxane Gay & Everand Originals: My Year of Psychedelics: Lessons on Better LivingFrom EverandRoxane Gay & Everand Originals: My Year of Psychedelics: Lessons on Better LivingRating: 5 out of 5 stars5/5 (5)
- Roxane Gay & Everand Originals: My Year of Psychedelics: Lessons on Better LivingFrom EverandRoxane Gay & Everand Originals: My Year of Psychedelics: Lessons on Better LivingRating: 3.5 out of 5 stars3.5/5 (33)
- Alex & Me: How a Scientist and a Parrot Discovered a Hidden World of Animal Intelligence—and Formed a Deep Bond in the ProcessFrom EverandAlex & Me: How a Scientist and a Parrot Discovered a Hidden World of Animal Intelligence—and Formed a Deep Bond in the ProcessNo ratings yet
- Fire Season: Field Notes from a Wilderness LookoutFrom EverandFire Season: Field Notes from a Wilderness LookoutRating: 4 out of 5 stars4/5 (142)
- The Ancestor's Tale: A Pilgrimage to the Dawn of EvolutionFrom EverandThe Ancestor's Tale: A Pilgrimage to the Dawn of EvolutionRating: 4 out of 5 stars4/5 (811)
- When You Find Out the World Is Against You: And Other Funny Memories About Awful MomentsFrom EverandWhen You Find Out the World Is Against You: And Other Funny Memories About Awful MomentsRating: 3.5 out of 5 stars3.5/5 (13)
- Spoiled Rotten America: Outrages of Everyday LifeFrom EverandSpoiled Rotten America: Outrages of Everyday LifeRating: 3 out of 5 stars3/5 (19)
- Make It Stick by Peter C. Brown, Henry L. Roediger III, Mark A. McDaniel - Book Summary: The Science of Successful LearningFrom EverandMake It Stick by Peter C. Brown, Henry L. Roediger III, Mark A. McDaniel - Book Summary: The Science of Successful LearningRating: 4.5 out of 5 stars4.5/5 (55)
- Why Fish Don't Exist: A Story of Loss, Love, and the Hidden Order of LifeFrom EverandWhy Fish Don't Exist: A Story of Loss, Love, and the Hidden Order of LifeRating: 4.5 out of 5 stars4.5/5 (699)
- World of Wonders: In Praise of Fireflies, Whale Sharks, and Other AstonishmentsFrom EverandWorld of Wonders: In Praise of Fireflies, Whale Sharks, and Other AstonishmentsRating: 4 out of 5 stars4/5 (222)
- Wayfinding: The Science and Mystery of How Humans Navigate the WorldFrom EverandWayfinding: The Science and Mystery of How Humans Navigate the WorldRating: 4.5 out of 5 stars4.5/5 (18)
- The Secret Life of Lobsters: How Fishermen and Scientists Are Unraveling the Mysteries of Our Favorite CrustaceanFrom EverandThe Secret Life of Lobsters: How Fishermen and Scientists Are Unraveling the Mysteries of Our Favorite CrustaceanNo ratings yet
- The Other End of the Leash: Why We Do What We Do Around DogsFrom EverandThe Other End of the Leash: Why We Do What We Do Around DogsRating: 5 out of 5 stars5/5 (63)
- The Revolutionary Genius of Plants: A New Understanding of Plant Intelligence and BehaviorFrom EverandThe Revolutionary Genius of Plants: A New Understanding of Plant Intelligence and BehaviorRating: 4.5 out of 5 stars4.5/5 (137)
- The Hidden Life of Trees: What They Feel, How They CommunicateFrom EverandThe Hidden Life of Trees: What They Feel, How They CommunicateRating: 4 out of 5 stars4/5 (1002)
- Come Back, Como: Winning the Heart of a Reluctant DogFrom EverandCome Back, Como: Winning the Heart of a Reluctant DogRating: 3.5 out of 5 stars3.5/5 (10)
- The Mind of Plants: Narratives of Vegetal IntelligenceFrom EverandThe Mind of Plants: Narratives of Vegetal IntelligenceRating: 4.5 out of 5 stars4.5/5 (11)
- The Big, Bad Book of Botany: The World's Most Fascinating FloraFrom EverandThe Big, Bad Book of Botany: The World's Most Fascinating FloraRating: 3 out of 5 stars3/5 (10)
- Gathering Moss: A Natural and Cultural History of MossesFrom EverandGathering Moss: A Natural and Cultural History of MossesRating: 4.5 out of 5 stars4.5/5 (347)