Professional Documents
Culture Documents
Solution Manual Chemical Engine PDF
Solution Manual Chemical Engine PDF
Uploaded by
Abid Rahardjo0 ratings0% found this document useful (0 votes)
3K views621 pagesOriginal Title
36045063-Solution-Manual-Chemical-Engine.pdf
Copyright
© © All Rights Reserved
Available Formats
PDF or read online from Scribd
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
© All Rights Reserved
Available Formats
Download as PDF or read online from Scribd
0 ratings0% found this document useful (0 votes)
3K views621 pagesSolution Manual Chemical Engine PDF
Solution Manual Chemical Engine PDF
Uploaded by
Abid RahardjoCopyright:
© All Rights Reserved
Available Formats
Download as PDF or read online from Scribd
You are on page 1of 621
Tnstructor’s Soltutions
Introduction to
CHEMICAL ENGINEERING
THERMODYNAMICS
Sixth Editson
J.M. Smith ¢ H.C. Van Ness ¢ M.M. Abbott
Chapter 1 - Section A - Mathcad Solutions
1.4 The equation that relates deg F to deg C is: t(F
‘equation by setting t(F) = «(C).
‘Guess solution:
Given Find(t) 240
5 a F
1.5. By definiti Peo F = massg
3000bar
P-A
1.6 By definition: Pp = = F = massg
3000atm
1.7 Pabs = p-g:h + Patm
13,535.28
em?
Patm = 101.78kPa
18 p= 13.535-50
om
Pam !=29.86in_Hg Pats == p-8-h+ Patm
4
8 (C) + 32. Solve this
Ans.
Note: Pressures are in
gauge pressure.
A= 12.566mm"
mass = 3844kg = Ans.
A= 0.023 in?
mass='10007 Ibm Ams.
b= $6.38em
Pabs = 176.808kPa Ans,
f= 25.62in
£27179 psia Ans.
Pats
1.10 Assume the following: p := 13.52% gi 98%
om’, ea
400bar his 2
Ps h=302.3m. Ans.
1.11 The force on a spring is described by: F = K x where K is the spring
constant, First calculate K based on the earth measurement then 2ytars
based on spring measurement on Mars.
On Earth:
F = massg mass := 0.40kg,
F c= massg F = 3.924N
On Mars:
0.40em Fears °= Kx EMars = 1-453N
FMars
mass
“Sears: Ans,
BMars =
112 Given: 4 ps—pg and: p= MP
a &
Poenver Denver
Separating variables and integrating: lope {*3) i
P RT
Pree °
After integrating:
‘Taking the exponential of both sides —Me
Ap Benver
and rearranging:
latm
Psea
3
cov atm
R = 82.06 Tes (10+273.19K ZDenver = Imi
mol
Me
ne zpemver = 0.194
Rr
Mey
(tome)
Ppenver >= Psca'e Ppeniee = 0.823atm Ans.
denver = 0:834bar Ams.
1.13. The same proportionality applies as in Pb. 1.11.
2186-5 Almoon ?= 18.76
¢
learth = Sloot Aleath = 113.498
Smoon
M i= Alegrthlbm MS.113,498lbm Ans.
Wimoon = M: moon
B.767IbE = Ams.
O0dolt T
114 cost = =20dollars jg Ltr costeleg = SGollars. 19 ht row
100k day Whe day
4 dollars
costhuty = 18.262 Solas costelgg = 25.567 Solar
yF yr
goo dollars
COSttotal *= COStbulb + COSteIee
o “Ans.
11S Ds 125k mass == 250lbm gis 2.169%
§
Patm i= 30.12in_Hg A= 120787
(2) F = PainrA + mase-g
Ans.
(b) Paps ae Paks + Ans.
A
(© Ale 17 Work = Fal
APE := mass-g-Al
1.16 D:= 047m mass = 150kg
Pot ‘= 101.57kPa Asp
(a) F 2 Paige A + mass-g FS1900%10'N Ans.
F 2
()Pabs == Paps = 110! Ans,
© Al:=0.83m Work := F-Al Work = 15,8481) Ans.
AEp := mass-g-Al D2k7 Ans.
1.18, mass := 1250kg
Eg six 10s Ans.
Wotk =F 10°kJ Ans.
119 Weot = M2S-8-Ah 9 91.9.9
time
Wdot 00W
Wdot
dot = ———>
g-Ah-0.91-0.92
Chapter 2 - Section A - Mathcad Solutions
24 (@) | Myp= 35-kg ze Sem
Work := My-g-Az Work:= 1715} Ans.
(b) AU total = Work AUsoigh = LTISKI Ans.
(©) By Eqs. (2.14) and (2.21): dU + d(PV) = Cp-dT
Since P is constant, this can be written:
Miz0-Cp-d? = Myo-dU + Mypo:P-dV
Take Cp and V constant and integrate: My2o-Cp-(t2~ t1) = AU total
25-kg
O-deg’
AUrotal
~— ty = 20,02degC Ans.
Miz0-Cp
(d) For the restoration process, the change in internal energy is equal but of
opposite sign to that of the initial process. Thus
S1715k) Ans.
Q = AU otal Q
© In all cases the total internal energy change of the universe is zero.
2.2. Similar to Pb, 2.1 with mass of water = 30 kg.
Answers ar @) W=3.43 kt
(b) Internal energy change of
the water = 2.86 kJ
(©) Final temp. = 20.027 deg C
(@) Q=-2.86 kd
5
2.4 The electric power supplied to the motor must equal the work done by the
motor plus the heat generated by the motor.
9.7amp E:= ov Wdotmech = 1-25hp
Weotelect = hE Wetecg = 1.067 10° W
Qdot := Wdotelect ~ Wdotimoch Qdot £134.875W Ans.
2.5 Eq. (2.3): AU'= Q+W
Step 1t02: AUty := -200) Wyo := -60003
Qi2 == AUY—Wi2 Qi $8109 Ans,
Step 3 t0 4:
Ans.
Step 1 to 2 to 3 to 4 to 1: Since AU' isa state function, AU' for a series of
steps that leads back to the initial state must be zero. Therefore, the sum of
the AU' values for all of the steps must sum to zero.
AUtyy = 4700) AUty3 = ~AUty2 - AUEsy- ALLY,
AUg; = 40007 Ans.
Step2to3: AUtp3=-4% 10°F Qo3 = -38003
Ans.
Wo3 = AUQ3- Qos Wha = 5200"
For a series of steps, the total work done is the sum of the work done for each
step.
Wat := W134i - Wi2 — W23 ~ W34.
Step 4 tot: AUty == 47001
Qat = AUtg) - War Ans,
Note: Qu23a1 = -Wi2341
241 The enthalpy change of the water = work done.
M := 20:kg q 4.18 id ‘At := 10-degC
‘ ee eg dest
M-Cp-At
eS Ans,
Wdot
2.12 Qs 75k —12KI W:= AU-Q
i J Ans,
AU ss -12-40 Q= AU Ans,
2.13. Subscripts: e, casting; w, water; t, tank. Then
me AUy + My-AUy + mrAU, = 0
Let C represent specific heat, C= Cpecy
Then by Eq. (2.18)
meCerAty + My-Cw Aty + meCr At
kg my = 40k my = Skg
50: ia Cte 05
te = 500-degC ty = 25-degC ty :=30degC (guess)
Given =meCer(t2- te) = (my-Cw + meC)-(t2- tr)
to = Find(t) wy SR7TeaeBe Ans.
7
215
@
)
©
247
2.18 (a)
(b)
mass-g
AE := AUt
Azi= 50m
Di=2m
mdot := p-wA
Wdot := mdot-g-Az. Wdot. =:
ur = 7620-8
kg.
Hy Ur +P
Uo = 27844
kg
Hg = Ug + P2-V2
‘Aus 0022.44
ee
cy = 4
AUt = mass-Cy-AT
gk
AEp := AUt
AULA 4 180 Ans.
291433" Ans,
3
>
3
Az "Dp? A=3.42m
4
mndot = 1.571 x 10882
3
L697 1OKW Aus.
Py = 1002.7-kPa Vy
ene
Hy #76313 b—— Ans.
Tae
a
Po = 1500-kPa V2 = 169.7.
gm
AU :2 U2- Uj AH = HQ-Hy
Ans, OH 2275.3 Ans.
222 Di
Sem. uy
m
5
Dz = Sem
(a) Foran incompressible fluid,
mdot = constant = u,A4p = uyA,9.
stant, By a mass balance,
(b)
2.23 Energy balance: mdots-H3 — (mdoty-H + mdotz-H2) = Qdot
Mass balance: mdot3 — mdot; ~ mdotz = 0
Therefore: mdoty-(H3 ~ Hr) + mdoty-(H3 - Ha) = Qdot
or mdot-Cp-(T3 - T1) + mdoty-Cp-(T3 ~ T2) = Qdot
T3Cp-(mdoty + mdotg) = Qdot + mdoty-Cp:Ty + mdoty-Cp-T2
Sdeg® —mdoty = 0.882 Ty = 75degC
s
1g
kg-K
Qdot + mdot;-Cp-T; + mdotz-Cp-T2
‘ty WWD238degC Ans.
fmdoty + mdot)Cp a 43.295 deEC
2
2.25 By Eq. (2.32): AH+ x
By contim eu
incompressibility eieinltaS
Dp = 3.8m
AT S.0.019degC Ans.
ATE 0023degC Ans.
226 Ty = 300K T2:= 520K w= 10
5
Wsdot = 98.8kW —ndot = soit
AH := Cp(T,-T1) AH = 6.402 109
Kemal
By Bq. 2.30):
a ye)
Qdot = | AH + oe +) no ndot - Wsdot Qdot =+9.904kW Ans.
2.27 By Eq. (2.32b): also
By continunity, ee eee
uy = up
constant area T Pe
atts cpar = LR(t—1)
10
Given ZR (T-T))
T2 *= Find(T2) ‘Ty = 578-821 rankine Ans,
(119.15-degF)
KI
up = 200-2 Hy = 3492 Hb = mse
8 B es
2.28 uy
aaa
uy? — uy
By Eq. (2.32a: Q:= Ha-Hi + Ans.
Hyp 31125-
kg
(guess)
By Eq. (2.32a): Given Ho =Hy ug := Find(uy)
uyes7R36% Ans.
Sg 3
3
Vy = 388.61
om
ap V2
Continuity: D2 := Dy
wv
"
Ans.
230(a) ty = 30-degC ty = 250-degC -mol
J
Cv i 208
By Eq. (2.19): Q:= n-Cy-(t2-t) Q=13738kI Ans.
‘Take into account the heat capacity of the vessel; then
100-kg, cy = 05+ i.
kg-degc
(my-cy + n-Cy)-(2- 11) Q= 1101410 Ans.
© 00-dege ty = 40edegC n= 4emol
Cp = 29.1 _
mol-degC
By Eq. (2.23): Q:= mCp-(.-ty) 18.62k] Ans,
231 (a) t = 70-deg? tz = 350-degF 3-mol
BIU
es 5 By Eq. (2.19):
V = Se deg? by Eq. (2.19):
Qe nCy(t2-ty) Q=4200BTU Ans.
Take account of the heat eapacity of the vessel:
BTU
ay == 200+ Ib = 0.12-————
ey ~ Bardeu?
Qs (myey +n-Cy)-(t2-11) O920BTU Ans.
(b) ty = 400-degF tz = 150-degF = 4-mol
BIU
Cp = By Eq. (2.23):
aan ty Bq. (2.23):
Qs Cp(ty -t1) Ans.
2.33 Hy = 1322.6—— Ha == 1148, oe up ot
Tb Tbs s
a 8°
Vy = 3.058-—— Vo= 7814 y= Bin Dy 10in
ibm Ib
En?
yor 4 Ib
rade a ndot = 3.463 « 10°
M
u2= n997
see
22
=u
Fg, (2.328): Wy := Hp-H) +2 W, = -173.99 BTU
2 Ib
Wot :=-Wemdot Wat 2:39.52 hp Ans.
BTU BTU ft gm
234 Hp 3075 — Hy = 330 2520-5 — molwe:= 4482
: ae ge eee mmol
a 3
f f
Vp 9255. Vy = 0.28 Dps4in Dz Tein
1 pe 7 nin Da
dot = 679.2632
br
We = 5360. BEY
ibmol
Ws
Q = 98.9 BU
mohwt Ibm
5g ATU
Qdot := mdor-Q —Qdol, = =67128——= Ans.
13
2.37
kg.
bar n = 34.602mol
28.9.2
mol
Vy = 24942
‘mol
Whence —nP-2V4 Ans.
V2
Given: Tee Tye = Tr3 Whence Tp := 3-Ty
1
Cp = 29-ioule AMs=Cp(T2-T1) A= I Ans.
molK PoP
Q:=0AH QS 602,081) Ans.
QeW
AU ss
n
Work exactly like Ex. 2.10: 2 steps, (a) & (b). A value is required for PV/T,
namely R.
J Ty = 293.15-K 333.15-K
molK P} = 1000-kPa Pp = 100-kPa
R
8.314
(a) Cool at const VI to P2
(b) Heat at const P2 to T2
cy=2R
fe
Tasty Ta = 29315K
ATp:=T2-Tag ATp = 303.835K AT,:=Ty-Ty AT, = -263.835K
14
AH, = Cp-ATp, AH = 8.841 10°
mol
: aed
AU, = Cy-AT, AU, = 5.484 10° —
mol
3m RT)
Vi = 2437x109 Vp vp = 0.028
mol Pp ‘mol
AUg+Vi-(P2—P1) Hy = -7.677% 10°
f 5 oat mol
AH ~ P2(V2- Vi) Uy = 6.315% 10°
mol
Ans.
18
3
34
35
Chapter 3 - Section A - Mathcad Solutions
ifa_)
petite)
pM) p
At constant T, the 2nd equation can be written:
eae ml 22] = xaP 44.1810 bar! pz = 1.01-p
P Pi
na ‘
AP = 225.2bar Py 3 226.2bar Ans.
em
bi=2700-bar =e := 0.125- Pys= Lbar Pp = 500-bar
em
V2
Since Work = -| PdV a bit of algebra leads to
P2
Work:=e] ——ap
P=b
P,
Alternatively, formal integration leads to
(ren sf
KeatbP a: 3.910 %atm | 0.1107 ?-atm?
3000-atm Vi Left? (assume const.)
Combine Eqs. (1.3) and (3.3) for const, T:
P2 :
Work = vf (a+ bP) PaP Work =1665aim-° Ans.
Py
16
Mis S-kg
3
Lom
yea P= Lebar t= O-degC ty = 20-dege
1°" 1550 ke acide i 7
With beta independent of T and with P=constant,
i iy
iz B-dT Vo i= Vi-exp|B-(t2-t1)] AV = V2-V)
AVotal = MAY AV poral = 7.638% 107 ‘Ans,
Work :=-P-AVyoigt (Const. P) Ans.
Q:= MCp-(t2- ty) Ans.
Agta °= Q Ans.
AUjotal = Q+ Work Ans.
2
38 pi Bbar Pps bar = T= 600K Cp c= ZR
(a) Constant V: WHO and SUS Q=Cy.AT
AT:=1)-T, AT = -525K
AU = Cy-AT Qand av sio91 ns.
ano
i ai
AH := Cp-AT ‘AH = =15.28—— Ans.
er
(b) Constant T: AU = AH=0 and Q=Ww
P2) Bias eg
Work := R-Ty-in| 22 | Q and Werk 2103722 ans
Pi) ‘mol
(©) Adiabatic: Q=0 and AUS WCyAT
7
and
Step 41: Adiabatic
P2 = 3bar T
Step 12: Isothermal
12
{2
PL)
War:
00K
AU
wt
18
:
AH = Cp-AT
a= -792t5 Ans
‘mol
3
3m
Vp = 4.988% 107
mol
Tq = 378.831K
Aug) = 4597 10
mol
AHgy = 6.436 1082.
mol
f
Qs) = 0—
mol
War = 4.597% 10°
mol
3
Vo = 0.01720
mol
aU, = 0
J
mol
AK »
AH =
Pe 3 J
Qi -R-TrInj =| Qi = 6.006% 10° —
Pi mol
6.006 10°
mol
Wiz -Qi2 Wi2=
P3-V:
X03 15 = 400K
P3:=2bar V3 = V2 Ts
R
Cy(T}-T2) AUg3 = 4.157% 103
Step 23: Isochoric U3
mol
Allg = Cp(T3—T2) AHg3 = ~5.82x 10°
mol
J
Qx3 = Cy(T3-T) — Qx3 = 4.157 10° —
mol
Was = 0 Wo = 0-4
mol mol
3
P4 = 2bar T4 = 378.831K V4 = 0.016
mol
‘Step 34: Isobaric AU3g = Cv(T4- 13) AU34 = 439. on
mol
J
Cp(T4-T3) AH3g = -615.996—
mol
Qs4 = 615.996
mol
W54 = 175.9994,
mol
3.10 For all parts of this problem: T2=T, and
AUS AH=0 Also Q = -Work and all that remains is
to calculate Work, Symbol V is used for total volume in this problem,
19
2m Vo i= em!
P2) Pp)
@ Work = a-R-T-In] = Work := P)-Vj-In]
Pi Pi)
Work = 2982k Ans.
(b) Step 1: adiabatic compression to Py
(intermediate V)__Vj = 2.702m°
Wy = 3063kI
a
Step 2: cool at const P, to V2
Wo = -P2-(V2- Vi) W = 2042kJ
Work := Wy + W2 Woik= $105k7 Ans.
(©) Step 1: adiabatic compression to V,
fy,yt
Pi =P} | (intermediate P) —_P; = 62.898 bar
(V2,
Wy = 7635k
Step 2: No work.
Work = 7635ki Ans.
(a) Step 1: heat at const V, to P,
Step 2: cool at const Py to V2
~P2(V2-Vvi)
W: }3200k) Ans.
20
(©) Step 1: cool at const P, to V2
Wy = -P1-(V2- Vi) W, = 1100k3
Step 2: heat at const V; to P; W220
Work := Wy ‘Wor itdoia A"
3.17 (a) No work is done; no heat is transferred.
AU'S AT=O T= Ty = 100-degC —_Not reversible
(b) The gas is returned to its initial state by isothermal compression.
(Vv
Work = n-R-Tn 2) but MRT = Po-V>
ae
Vian? Vo
Work := P2:V>_In
Work = 878:9K1 Ans.
re
sis
3.18 (a) Py := 100-kPa Pp = 500-kPa Ty = 303.15-K
5 Cp
cy=2
ves bead
Adiabatic compression from point 1 to point 2:
KE
Qin = 0 AU,2 = Wi2 = Cv-aTi2
mol
AU,2 = Cy{T2-T1) AH)2 = Cp(T2-Ti) Wig AU)2
ak
AHy2 = 5.13
eT Same)
24
Cool at P, from point 2 to point 3:
= AHD3
AHQ3 Ans.
kK
Qag = 5.15 Ans.
mol
Isothermal expansion from point 3 to point 1:
AU) = AH) = 0
FORTHE CYCLE: AU = AH = 0
Q= Qin + Q3+Q51 Work := Wy2 + W23 + W3i
Qaa oo Work vo04 2
SSS Snel 5 ‘mol
(b) [each step that is 80% efficient accomplishes the same change of state,
all property values are unchanged, and the delta H and delta U values are
the same as in part (a). However, the Q and W values change,
2
Step 12: Wiz = —
P 12 08
Qu2 = AU 2 ~ Wir
Ww:
Step 23: Wo3 52
0.8
2
Qg3 == AUp3 ~ Wo3
Step 31: W3) = W31-08
Qs = -W31
FOR THE CYCLE:
Q = Qi2+Qz3+Q31
pene
192 —
mol
3.19 Here, V represents total volume,
vi
(a) Isothermal: y= Ppt
V2
eT z= 600K P= 200kPa = Ans.
v es
Work = PV; ma ‘SOR) Ans.
Wa
vi Po V2
b) Adiabatic: Pep tee Tye?
(0) Adiabati: 2 @) eT BY
P25 69.65kPa Ans.
994.4k) Ans,
23
(c) Restrained adiabatic: Work = AU = Pex AV
100-kPa Work = ~Pexr(V2- Vi) Work'=400KI Ans.
Pext
PrVi
AU = neCy-AT.
T2SARTIK Ams
P2= 147:57KPa Ams.
3.20
‘T3 := 323.15-K
Step 12:
R-Tyln@)
Qi Wi 2.50244
mol
KI
a AUp3 == Cy-(T3 -T2)
Q23 == AU23 Als
ki
Qa3 = -2.079— AU23 = 2.070
mol mol
Process: Work := W)2 + Wo3
Qi Qi2t G3 Age
24
AH == AH}2+AFy3
AU = AU 2 + AUa3
321 By Rq. 2.328), unitemass basis: motwt = 28-2 ane Lau? =o
mmol 2
But AH = Cp-AT Whence AT =
322 3 = 403.15-K
AU = 20792 Ans,
mol
Each part consists of two steps, 12 & 23.
Work := W23
wok 67608. Ans.
= = mob
2
Q:= AU — Work
Qe e463 Ans.
= mol
Tre Ts AU)? = Cy(t2-T)
cp(t2-N) Qu2 = AH
ky
2 = -Q12 W)2 = -0.831—
Wi2 = AU}2- Qua 12 sy
P3 kJ
W 3 := R-To-Inj — W323 = 7.718 —
93 := RT (2) 23 Ral
Work := Wi2+ Was Work = 6.986 Ans.
EE pene es eA
i
Q = AU - Work Q=-4808— = Ans.
Pinal
os
© bet P= P3 Ty (2)
Pi
AHp3 = Cp-(T3-T2) 03 := AH3
AUp3 = Cy-(T3 - Ta)
Work := Wi2 + W23 Woks Ans.
‘mol
Q = AU - Work Q=-2.894
For the second set of heat-eapacity values, answers are (kJ/mol):
AU = 1.247 AU = 2.079
(a) Work = 6.762 Qe
26
51S
3.23
3.24
(b) Work = 6.886 = Q= ~5.639
© Work = 4.972 Q= -3.725
Ty = 303.15-K Toi=Th T3 = 393.15-K
Py = I-bar P3 := 12-bar
Forthe process: AU:= Cy(T3-T)) AH = Cp(T3-T1)
av= isnt
mol
Step 12: P AS a
cep 12: Pa i= Pye
eect
KI
Wi2 = 5,608-—
mol
Step 23:
For the process:
Q= Qin+ Qs Ans.
Work = W3 = -P2(V3~ V2) = -R.(T3 -T2)
But T3=T) So. Work = R-(T2-T;)
>)
Also WERT) “(| ‘Therefore
1
(p\ 12-T
In} ae sf 50-K Py = 4-bar
PB) OT
27
328
3.26
™2-T aa
P Pre == | PS227obe Ans.
1
Va t= 256-em> Define:
ae
Py
Assume ideal gas; let V represent total volume:
P)-Vp= P2(Va+Vp) From this one finds:
ap__-Va VatteD 4
Vp Vp 37503cm? Ams.
Py Vat+VB r
T)=300K Pj Cy =Cp-R
‘The process occurring in section B is a reversible, adiabatic compression. Let
P(fina) = P2 Ta(final) = T Tp(final) = Ty
nA = ng, Since the total volume is constant,
2naRT1 _ nyR(TA+Tp)
u
= = o
(a) Pp := 1.28-atm Q)
Pr
Ta c= 2-Ty Ae Q=na(AUa + Ug)
Define = 2 q=Cy(Ta+Ta-271)
A
Ta = 43025K Ans,
(b)
©
@
Combine Eqs. (1) & (2) to eliminate the ratio of pressures:
Ta 23 425-K (guess) Tp = 300-K
Given
Ans.
TatTh
Poss P| qa Ans.
21
cy(Ta+Ta-2T71) Ans.
Tp = 325K By Fa. 2),
fe)
Pp = Py (2) Pee 182Fatm Ans.
1
Tas 2Tpo-Ty @ Ans.
ar 2T rT
= Cy(Ta+Tp- 2-4) Ans.
Eliminate Ty +Tp from Eqs. (1) & @):
P,
= ip Ans.
2TCy
Q) Ti =319.06K Ans.
a Ans.
373.15-K
bax?
Guess: yet
Given
Guess: vo:e RT
P2
Given ie < V2:=Find(V2) V2
Va v2
Eliminate P from Eq, (1.3) by the virial equation:
“ar Bc
1
[eS geeeas
fork = 12.62
MseNe/ ° mmol
ML
(b) Eliminate dV from Eq. (1.3) by the virial equation in P:
fel) Poy
ave RT) = +C ar W:=-RT ere | a
Cs a
30
3
Vi i= Find(Vi) Vy, = 30780"
mol
mol
Ans.
Note: The answers to (a) & (b) differ because the relations between the two
sets of parameters are exact only for infinite series.
332 Te = 282.3K THDBISK Tye Ty = 1.056
Pe P, = 0.238
Pe
(guess)
3
va ®t v= 2066S
mol
Given PVC
RT
“agem
V := Find(V) v= 1919 Zz Ans.
thol
(b) By := 0.083 - oe Bo = 0.304
*
By = 039-217 By = 2.262% 10>
742
Pr ee
Z=1+(Bo +o: Bie £20932
(©) For Redlich/Kwong EOS:
0 Qe
0.08664 Table 3.1
Table 3.1 a(T,) =
Eq. 3.51)
ET
2-P,
B(Tr.P,) = a
r
Eq. (3.50)
Calculate Z Guess:
Given Eq. 3.49)
Z-B(T,.Pr)
+ Blt e) a0) PP) ESE, Ri (ererBl PD)
gery
Find(Z) Z = 0.928 v ee 9165 Ans.
(@_ For SRK EOS:
e=0 2 = 0.08664 ¥ = 0.42748 Table 3.1
r ne
| 2 ( 3] ‘Table 3.1
(Tyo) =[1 + (0.480 + 1.5740 - 0.17607) UT)
¥-0(Tr0) :
a(t) eal) Eq.@51) — B(Tr,Ps) Eq. (3.50)
Calculate Z Guess:
09
Given Eq. (3.49)
Z= 1+B(Te,P,) —a(Tr)-B(tr Ps) ETN aR
Zi=Find(Z) Z = 0.928 a
Ans.
(©) For Peng/Robinson EOS:
wi
o
1-¥2 Q:=007779 = 045724 Table 3.1
iN
a(Tr,o)
[ 1+ (037464 + 1.542260 - 0.269020") (
11,7)
Table 3.1
2
Eq. (351) 8(T,.P; 2 or Eq. (3.50)
or 4. Bs a(t. eee 4. (3.50)
Calculate Z Guess:
Given Eq. 3.49)
Z= 1+B(Tr,Py) ~a(T,)-B(Tr,Ps) Ges
Zi=Find(Z) Z= 0.92
333 Ty 3053K 9 -T=32ISK Tye z Tp = 1.058
oe
= 4B. T2bar P= 1Scbar Pae P, = 0.308
Pe
© = 0.100
vei
mol
Z=0907' Ans.
Bo = -0.302
33
3
ERT y= e142 ans.
fi EES OL
P,
1+ (Bo +B) Za0ai? Vv
(©) For Redlich/Kwong EOS:
Q = 0.08664 WY = 0.42748 Table 3.1
Wa(T,)
Table 3.1 (ts) = walt) Eq. @.51)
QT
Eq. 3.50)
Caleulate Z Guess: 2-09
Given Eq. (3.49)
2Z-B(Tr.Ps)
Z= 1+ B(TrPe) alt) BOP B(r.P))(Z + o-B(T.Ps))
Zi=Fink@) Z y= ZRT
P
(@)_ For SRK EOS:
o=l 5:0 2 = 008664 = YY = 0.42748 Table 3.1
f yp Table 3.1
3 able 31
o):=|1 + (0.4804 1.5740 - 0.1760)-(1— 1,2
¥-0(T,.0) ap,
a(t) elte2) Eq.@51) — (T;,Py) 7 Eq. (3.50)
Caleulate Z Guess 250.9
Given Eq. (3.49)
zeal +8(T.P) a(t) BCC) Ea ee reptinP)) a Bia)
4
Les
ind(Z) Z = 0.907 Ve
(©) For Peng/Robinson EOS:
142
o 1=$2 Q:=0.07779 = Y= 0.45724 Table 3.1
2
1
a(Tr.a) = [ + (0.37464 + 1.542260 - ol - Jl oa
q(t) = ¥a(T.0) Bs) B(T,,P)) = ae Eq. (3.50)
OT : Tr
Calculate Z Guess: Z=09
Given Eq. (3.49)
Z~B(Tr.Pr)
Z= 1+B(Tr.P,) —4(T;)-B(Tr.Pal
Find(Z) Z = 0.896
334 Te = 318.7K T= 348.15-K T, = 1.092
1S-bar =— Pr = 0,399
Pe
(guess)
3
vekT ye i939
mol
Given
35
V = Find(V)
0.422
() Bo == 0.083 - ==
+
0.172
By = 0.139- >
Tr
Pr
Zi=1+(Bp+o-By 7
Z= 0893
Ans.
Bo = -0.283
By = 0.02
20,899.
(©) For Redlich/Kwong EOS:
o W = 0.42748 Table 3.1
w-a(T,)
‘ oe Eq. (3.51)
a(Tr) q(T) am 4.
B(T;,P,) = Eq. (3.50)
Calculate Z Guess: 0.9
Given Bq. (3.49)
Z= 1+B(Tr.Ps)-a(Tr) BrP)
Z = Find(Z)
(a) For SRK EOS:
1
Z = 0.888
2 := 0.08664 =P = 0.42748 Table 3.1
36
Table 3.1
of ae
W-a(T;,@)
OT;
a(t Eq. (3.51) B(tr,P,)
Eq. (3.50)
Calculate Z Guess: Z=09
Given Eq. (3.49)
2 148) a0) C8 ae
Z=Find(Z) Z = 0.895
(©) For Peng/Robinson EOS:
ozity2¢
-¥2 007779 -W = 0.45724 Table 3.1
a(Tr,@) = [ 1+ (0.37464 + 1.542260 ~ 0 ct ata
v-a(Tro
T,) » ~~ Eq. (3.51 TrP,
eC) Saat 4-51) B(Tr.P,
Eq. (3.50)
Calculate Z Guess: Zi=09
Given Eq. 3.49)
Z= 1+B(Tr.P.) ~a(T,)-B (TPs) Ea Ea
Find(Z)
ZRT
P
0.882 v
37
335 T= $23.15K 1800-kPa,
Tr = 0.808
Py = 0.082
By = -0.281
Z= 0.939
(©) Table F.2:
molwt := 18,0155
mol
or
337
Z=0931 Ans.
= 124.99-.molwt
gm
. HS ea
Y= 22525 Ans.
eS nol
cm?
D=5000-— — n:= mol
mol?
Ts 273.15-K
Given ae lte tse £(P,V) =
[ 4 1
0858 O86
2906 a@86
0858 [oes
Pi=[- a0) bar oars] asia aaa
720) o782 0765 0822
140) 0.757) 0718 0.5+0.1781
10 O74 aati 05-0281)
180] O78 624 D503
~_ 200) 0735 losr7 | 050.4161]
O74 388 0540 468i
Note that values of Z from Eq, (3.38) are not physically meaningful for
pressures above 100 bar.
39
BP;
Zij= 1+ Eq. (3.37) 23;
0 7 Be Ze 2 =
20 7 7 1
40 0583] 0858 O86
60 0.306] 0.506; 0885
89 D867 0.888) 088
yoo] bar foar9| 0812 0749
120 0764 id oa
140 O7er a 050178
160) ‘O74 O87 05+028i1
120) 0733 0628 O5+O.366i
200 0735) 577 DE+OaT6I
‘o743| oss 0590. 468)
Note that values of Z from Eq, (3.38) are not physically meaningful for
pressures above 100 bar.
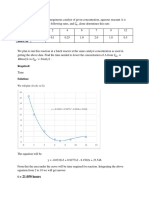
Eq. 8.38)
09
ZG 08
06
30 100 150 200
Pj-bar 2
3.38 (a) Propane: 169.8-K 42.48bar = 0.152
T := 313.15-K 13.71-bar
T, = 0.847 Pris 2 P, = 0.323
c
o & Q := 0.08664 = 0.42748 Table 3.1
w-a(T,)
Table 3.1 kg. @.
oT,
Eq. (3.50)
Caleulate Z for liquid by Eq. (3.53) Guess: Z:= 001
40
Given
Z= B(T;,Pr) +(Z + 6-B(Tr.P,)}-(Z +0: atta any
ZRT cin
Zs= Find(Z) Z = 0.057 veo O81 Ans.
P <2 anol
Calculate Z for vapor by Eq. (3.49) Guess:
Given
Tr ,Pr]
22 19 Blta) at OCP) eee
ZRT 55 em
Z:=Find(Z) — Z = 0.789 var Ne 092 Ans,
Rackett equation for saturated liquid: T,
Ans.
For saturated vapor, use Pitzer correlation:
0.422
#0 Os Gore: Bg = -0.468
Ty
0.172
Bl eee By = -0.207
ve Verse Ans.
at
Parts (b) through (t) are worked exactly the same way. All results are
summarized as follows. Volume units are cu.cm./mote.
RIK, Lig, R/K, Vap. Rackett Pitzer
(a) 108.1 1499.2 94.2 1537.8
(b) 14.5 1174.7 981 1228.7
(©) 122.7 920.3 102.8 990.4
(@) 133.6 717.0 109.0 808.0
(@) 148.9 1516.2 125.4 1877.0
(1583 1216.1 130.7 1296.8
(g) 1704 971.1 1374 1074.0
(a) 1871 768.8 146.4 896.0
(i) 153.2 1330.3. 133.9 1405.7
@ 1642 1087.9 1403 11543
(kK) 179.1 835.3 148.6 958.4
@ 2014 645.8 160.6 795.8
(m) 61.7 12525 535 12769
(n) 64.1 1006.9 55.1 1038.5
(0) 66.9 814.5 57.0 853.4
(p) 703 661.2 59.1 707.8
(@ 644 1318.7 54.6 1319.0
(r) 674 1046.6 56.3 1057.2
(8) 70.8 835.6 58.3 856.4
(t) 748 669.5 60.6 700.5
42
3.39 (a) Propane Ty = 369.8 Po = 42.48-bar
T = 313,15K
P, = 0.323
o-l & Q. := 0.08664 WY = 0.42748
’)
a(t.) [| 1-+(0.480 + 1.5740 ~ 0.1760
W-a(Tr.0) i OP, a
a(T;) == ¥-a(tro) Eq. (3.51) B(Ty,Pr) = Eq. (3.50)
OT;
Calculate Z for liquid by Eq. (3.53) Guess: Z=001
Given
Z= p(ty,P,) +(2+68(te.P.)}{2 +08 (Tr.P9) aha
(Ts) B(Te-Pr) )
Z=Find(Z) = Z= 0.085 -V sS Ans.
Calculate Z for vapor by Eq. (3.49) Guess: Z=09
Given
Z-B(TrP,)
Z= 1+B(T:.Ps) —a(T): 8(1P) eat Zs eT)
ind(Z) Z = 0.78 vet
Parts (b) through () are worked exactly the same way. All results are
summarized as follows. Volume units are cu.cm./mole.
SRK, Lig. SRK, Vap. Rackett Pitzer
(a) 104.7 1480.7 94,2 1537.8
(b) 110.6 1157.8 98.1 1228.7
(©) 118.2 904.9 102.8 990.4
(@) 1285 7033 109.0 805.0
(©) 1421 1487.1. 125.4 1577.0
(150.7 1189.9 130.7 1296.8
(g) 1618 9478 1374 1074.0
(i) 1771 747.8 146.4 896.0
(146.7 1305.3. 133.9 1405.7
@ 156.9 1035.2 140.3. 1154.3
(i) 170.7 815.1 148.6 955.4
(@ 191.3 6285 160.6 795.8
(m) 612 12489 535 1276.9
(n) 63.5 1003.2 55.1 1038.5
(0) 663 810.7 57.0 53.4
(p) 69.5 59.1 7078
(@) 614 1296.8 54.6 1319.0
(r) 63.9 1026.3 56.3 1057.2
(3) 669 817.0 58.3 856.4
(0) 70.5 652.5 60.6 700.5
3.40 (a) Propane T, = 369.8-K Py = 42.48-bar = 0.152
T= 404273.15)K T= 31315K P= 13.7I-ber
ee Ty = 0.847 Bish Py = 0.323
Te Pe
From Table 3.1 for PR:
2
1
a(T;,0) al + (037464 +1 542260 — 0.269920?) ( ll
:= 0.07779 ‘Y = 0.45724
QP,
B(Tr.Pe) = Eq. (3.50)
Calculate Z for liquid by Eq. (3.53) Guess: Z = 0.01
Given
2 p(te,P))+ (2-+e-6(ty,P2)}(2+ 6:8 (Te.P) (eet)
=FindZ) 2 = 0.049 V
Calculate Z for vapor by Eq. (3.49) Guess: Z:=06
Given
Z= 1+ (t-.P) ~a(t:)6(T;.P,) 2-p(TE Fi)
(Z+6-B(T;,P))(Z+0-B (Ty Py)
Zi=Find(Z) Z = 0.766 v
45
Parts (b) through (t) are worked exactly the same way. All results are
summarized as follows. Volume units are cu.cm./mole.
PR, Lig. PR, Vap. Rackett Pitzer
(a) 922 1454.5 94,2 1537.8
(b) 97.6 1131.8 98.1 1228.7
(©) 104.4 879.2 102.8 990.4
(@) 113.7 678.1 109.0 805.0
(®) 125.2 1453.5 125.4 1877.0
( 132.9 1156.3 130.7 1296.8
(g) 143.0 915.0 137.4 1074.0
(b) 1571 7158 146.4 896.0
@ 1294 1271.9 133.9 1405.7
@ 138.6 10023 1403 1184.3
(K) 1512 7828 1486 9554
@ 1702 5973 160.6 795.8
(m) 54.0 1233.0 53.5 1276.9
(n) 56.0 9873 58.1 1038.5
(0) 584 794.8 57.0 853.4
(P) 614 641.6 59.1 707.8
(@ S41 12802 546 1319.0
() 363 1009.7 563 1057.2
() 589 800.5 583 856.4
() 622 636.1 60.6 700.5
46
3.41 (a)Forethylene, — molwt : 28.054 oe To = 282.3-K
mo
@ 2= 0.087 T = 328.15-K
‘T, = 1.162
From Tables E.l & E.2: Zo = 0.838
Zg+@Z, 2 = 0.841
i8-ke
molwt
(b) T= 323.15-K 115-bar
r P
hres Ty = 1.145 Pes Py = 2.282
Te Pe
From Tables E.3 & E.d: Zp = 0.482 126
P-Viotal
Z=%+oZ Z= 0493 A 2171 mol
ZRT
mass := nemolwt mass =60,898kg" Ans.
3.42 Assume validity of Eq. 3.37).
3
V, = 23000
7
, ‘mol
Prev RT 3 om?
Z; = 0.922 B= —(Z)-1 B = -1.942x 10° —
=a 2 p 1) =
With this B, recalculate at P,
a7
0.422
Bo := 0.083 — —s Bo = -0.146
Tr
Bi ore By = 0.104
a
Ty
Te
+ (Bo +o-B)) R— Ans.
For an ideal gas:
34 T= 320K P
v
tek
Te
1028s? 3
“i mi
Vig = Vere | Viig = 96.769
mol
08,
Vionk = 035:m? mig i= tg = 49 7'SB4R Ans.
ig
mmolwt
0422p = 0.449
16
48
3 em!
Veap = 1318x100
mp = 2341kg Ans.
tr T, = 0.701
P, = 0.064
T
Te
P
Pris
Po
molwt := 58.1232
mol
V = 9.469 10°
mol
Ans.
3.46 (a) T 1, = Ty = 1.091
©
P
Prem e Py = 2.874
motwt := 30,0722.
mol
0.100
ZixLo+o2) = 90.87
mol
Viotal
Methane °= —F
‘molt
o P= 20000kPa = PVE ZRTS LRT pT
where a a = 29.5482
kg
at 4.105
This equation giving T, as a function of Z, and Eq, (3.54) in conjunction with
Tables E.3 & E.4 are two relations in the same variables which must be
satisfied at the given reduced pressure. The intersection of these two
relations can be found by one means or another to occur at about:
Tyi= 1.283 and 7,
0.693
Whence T= TrTe
T=391.7K or {185-deg Ans.
298.15-K
Po 50.40-bar —— @ = 0.087
PY = PpPeV = ZRT
R
where a= a = 4.675
2
<
50
T
= Tr = 1.056
Te
Whence P= 4675-2 at Ty»
This equation giving P, asa function of Z and Eq. (3.54) in conjunction with
Tables E.3 & E.4 are two relations in the same variables which must be
satisfied at the given reduced temperature. The intersection of these two
relations can be found by one means or another to oceur at about:
PL79-73 bar Ans.
582
Yio: °
3.48 myater == 15-kg ve v= 26.667
Mater em
Interpolate in Table F.2 at 400 degC to find: P.#.9920:kPa Ans.
3.49 Ty = 298.15-K Tr = 0.977
P) = 2200-kPa Pri = 0.452
Veotat = 0.35:m? o:= 0.100
From Tables El & E.2: Zo
Zs lgt+oL,
Ti = 1615
493.15-K.
61
303.15-K Te i= 304.2-K T= oe
Veotat = 0.5-m? Pe 73.83:bar a := 0,224
Bo ~ 0.083 - 2422 Bo = -0.341
16
Tr
172
By = 0.139 — om By = —0.036
Ans.
T, = 0.613
Po := 34.0:bar Pr = 0.03
i 3a 3
molwit == 28.0145 Vig = 342-62
Bo = -0.842
62
T,
Z = 0.987
Rapor Aiyapor = 5.718 10” mol
ZRTp ee
Final conditions:
anol 2-Viig
iota “= T-mol + Myapor —
i Dtotal
Tre Ty = 2.363
359.2bar
Use Redlich/Kwong at so high a P.
Q = 0.08664 W:=042748 a(T) = Th a(T;) = 0.651
Wea(ty)-R-Te QRTe
—_—_———._ (3.42) Sess (3.43)
Pe ©
o.o01m> been oe)
Peat 26.737
mol mol
Ka A (3.44) = dsoli bar Ans.
“VIB Vive)
For isobutane: Te = 408.1-K,
3.52
Py = 4-bar
6.48-bar
Po
‘Tp = 415K
From Fig. (3.17): Pry = 245
‘The final T > Tc, and Fig. 3.17 probably shoutd not be used. One can easily
show that
_ Pe with Z from Rq. @.54) and
Pr ORT ‘Tables E.3 and E.4. Thus
0.181
Z=032
Eq. 3.68): Vo = Vp et Ans.
Pr
3.53 For n-pentane: Po i= 33.7-bar py = 0.63-22
em?
Ty = 291.15K AIB.ISK Pp t= 120-bar
P2
Pa =
euuiPe
Ty = 0.62 Pri = 0.03 Trg = 0.88 Pra = 3.561
From Fig. 3.17): py = 2.69
i P12
By Eq. 3.65), py = pp 2
Prt
Ans.
3.84 Forethanol: Ty := 513.9-K
= 6148-bar P= 200-bar
3
Ve = 167.
‘mol
From Fig. 3.17:
Pr
Pp Ve
imolwt
3.55 For ammonia:
Te = 405.7-K T= 293.15K
Pe r= 112.8-bar 857-kPa
em?
Ve = 2.5 Ze = 0.242
mol
0.2887)
1-1)
Eq. (3.63): Viiquia = Ve all ) |
Bo = 0.627
By = 0.534
x
Viiguia = 27-11
ed mol
Pre Pp = 3.253
® = 3.25:
Ans.
Tet Tr= 0.723
Te
P, = 0.076
3
3
om
Vyapor = 2616
: mol
Ans.
Alternatively, use Tables B.1 & E.2 to get thevapor volume:
Ze 0.929 Zj = -0.071 ZLot@Z, Z=0911
Vv, _2ZRT
fapor =
AV i=
apor~ Viguid
10 gal. of gasoline is equivalent to 1400 cu ft. of methane at 60 degF and 1
atm, Assume at these conditions that methane is an ideal gas:
f-atm
Ibmolrankine
R = 0.7302
519.67-rankine P= atm
Vv
Ve= 1400-8? -
nso n= 3.6891bmol
RT
For methane at 3000 psi and 60 deg:
Te: 190.6-1.8-rankine T=
519.67-rankine
T, = 1515
Pe = 45.99-bar 3000-psi P, = 4.498
0.012
From Tables E.3 & E.4:
Zo = 0819 Zy = 0234 ZL ko+o2 Z = 0.822
56
Ans.
3.39.T
Calculate the effective critical parameters for hydrogen by equations (3.55)
and (3.56)
43.6
ee Te = 30.435K
21.8K
L+
2.016T
Pos — 5 __sbar P. = 10.922bar
44.2K
1+
2.016T
P, = 0.294
guess of volume:
mol
Use the generalized Pitzer correlation
0.422
Bo := 0.083 -
Tr
Bp = 0.495 By := 0.139
Z= 0823 Ans. Experimental:
2 = 0.08664 Y= 0.42748 ‘Table 3.1
W-a(T)
Ta
Table 3.1 a(t;) = Eq. G51)
87
QP,
B(Te,Pr) 2= a Bq. 3.50)
Calculate Z, Guess: Zi 09
Given Eq. (3.49)
Z(2 + B(Tr.Pe))
Ans. Experimental:
Z= 1+B(TesPr) ~a(Ts}B(Tr.Pe}
1.7787
0.012 Te = 190.6K Po := 45.99bar
Atstandard condition: T= [to = sy + oe T = 288.706K
Pitzer correlations: P:= latm
ral T= 1515 es P, = 0.022
i Pe
a 7
Bo := 0.083 - 2422 2 139-9172 By = 0.109
1 1
Pr Py
Zo 1+ Boe %q = 0.998 1 = Bre 24 = 0.00158
ZRT m
Z=Zo+o21 Z=09% Vps a Vy = 0.024
P mol
an 5 | 5
(a) At actual condition: T =| (50- 325 + ila P := 300psi
Pitzer correlations: eee
Tel T, = 1486 P, = 0.45
Te
Bo = -0.141
58
0.172
By = 0.139--— By = 0.106
Tr
Pr Pr
Zo = 1+Bo Tr Zo = 0.957 = Bee Zy = 0,0322
3
Z = 0.958 V2 = 0.00109 7
mol
V2 ete
a2 915 10°. Ans.
24 Vi an day
© D = 22.624in A= 0.259m"
wis Ans.
ole
3.62
0.012
0.087
O41
0.140
0.152
0.181
0.187
0.19
0.191
0.194
0.196
02
0.205
021
021
0.212
0.218
0.23
0.235
0.252
0.262
0.28
0.297
0.301
0.302
0.303
0.31
0.322
0.326
0.286)
0.281
0.279
0.289
0.276.
0.282
0.271
0.267
0.277
0.275
0.273
0.274
0.273
0.273
021
0.272
0.275
0272
0.269
027
0.264
0.265
0.256
0,266
0.266
0.263
0.263
0.26
0.261
Use the first 29 components in Table B.1
sorted so that «o values are in ascending
order. This is required for the Mathead
slope and intercept functions,
tope(w.Zc):= (-0.091)
intercept(«,Zc) = (0.291)
R i= cor(w,Zc) = (0.878) R*= 0.771
Chapter 4 - Section A - Mathcad Solutions
4.1 (a) Ty = 473.15-K T= 1373.15-K n= 10-mol
ForS0%: A= 5699 B=0801107 C=
icpHi(473.15,1373.15,5.699, 0.801103,
ICPH := 5.654-10°.K AH:=RICPH Q:= AH
GaN Ans.
(b) To := 523.15-K T }473.15-K ‘= 12-mol
For propane: A 1213 B= 28785107? C= -8.824-10"°
rep (523.15, 1473.15, 1.213, 28.785-107
=8.824-107 °,0.0) = 1.947-10°
ICPH := 1.947-10%K AH := R-ICPHL Q:=n-AH
QS 194K 107K Ans.
42 (a) To = 473.15-K n= 10-mol — Q:= 800-K
3 6
Forethylens Ass 1424 pe AAROEIOT™ 4.39210"
K ev
1 = 2 (guess) Given
Bo 2(2 C133
Qe {| A-t06 a1) Bape? | +S (2- |
= Find(t) = 2.905 T:=tTo
13745K Ans,
(b) Tg := 533.15-K n= 15-mol Qs 2500-KI
31.630-10°*
K
For I-butene: A :=
967 iB c
61
3 (guess) Given
Qen {| a-to6-0 +E ne -]-Snie ee )
Find(x) 7 = 2.652
3.8K Ans.
(©) To == 500-degF n= 40-lbmol Q:=10°BTU
Values converted to SI units
To = 533.15K 1.814x 10'mol Q = 1.055 x 10°ks
14.394:10- 4.392.107
K KR
For ethylene: A
424
c= 2 (guess) Given
t= Find(t) 1 = 2.256 T:=tTo
Convert given values to SI units Vv = 7.079m9
= (T— 32degF) +273.15K To = (To ~ 32degh) +273.15K
T = 1187.37K To = 323.15K
66.985 mol
A= 3355 B= 057510°° 0.0 =D=-0.016-10°
1ePh{323.15,773.15,3355,0.575-10" 3,0.0,-0.016-108) = 1688.702
@
ICPH := 1648.702-K AH:=R-ICPH Q
465% 1 BTU Ans.
4.4 molwt := 1001-22 To =.323.15-K 153.15-K
mol
0000:
z= 20000-kg n= 9.99 10'mol
mohwt
For CaCO3: Ais 12572 B=263710° =D = -3.120-10°
repHi(323.15, 1153.15, 12.572,2.637-107°,0.0,-3.120-10°) = 113554
ICPH := 11355.4K AH ss
-ICPH Qs mal
4314% 10K) Ans.
4.7, Let step 12 represent the initial reversible adiabatic expansion, and step 23
the final constant-volume heating.
Ty = 298.15K T3 = 298.15-K Py = 121.3-kPa
P.
Pp = 101.3-kPa P3 == 104.0-kPa T2 = T3 =
3
cp = 30.bule (ques) T) = 290.41K
molK
Ans,
33.70 309.2
30.25 341.9
48.98 | 353.2 |K
41.06 es
40.73 353.9.
i AH yp is the given Peers ee a
SH Me valet NPA emurmal ty x EK gy Te
boiling point. Te Te
366.3 357.2 0.635 0.658,
366.1 336.7 0.587 0.674
AH = | 433.3 [3939/2 1 -| 053 | Te =| 0.028
aaa) 3632.) 0.504 0.649
392.5 358.2, 0.539, 0.639
(a) By Eq. (4.13)
(b) By Bq. (4.12): AH, =|
4.10. The In P vs. 1/T relation over a short range is very nearly linear. Our
procedure is therefore to take 5 points, including the point at the temperature
of interest and two points on either side, and to do a linear least-squares fit,
from which the required deriva Eq, (4.11) can be found, Temperatures
are in rankines, pressures in psia, volumes in cu ft/bm, and enthalpies in
Btu/lbm. The molar mass M of tetrafluoroethane is 102.04, The factor
5.4039 converts energy units from (psia)(eu ft) to Btu.
(a) T := 459.67+5 AV := 1.934~0.012 +e
18.787 7
21.162 0
1
Data: P= | 23.7 = =— y= h(P}
ais P= [23767] t=] S| x= oe = h(n)
26617 10
29.726 Is
slope = slope(x,y) slope = ~4952
(-P).
3
aPaT =: slopdPdT = 0.545
_ Tay-dPaT
“3.4039
AH = 90,078" Ans.
‘The remaining parts of the problem are worked in exaetly the same
way. All answers are as follows, with the Table 9.1 value in ():
(a) AH = 90.078 (90.111)
(b) AH = 85.817 (85.834)
(©) AH = 81.034 (81.136)
(@) AH = 76.007 (75.902)
(©) AH = 69.863 (69,969)
65
4a 119377 3364 34.72 334.3
32.082 |-SE ty 1=/ 5126 |K Py =| 80.97 bar Ty =| 397.9 |
153.822 356.4 45.60. 349.8
AlTis the valueat “Hep is the given pony
O deg value at the normal Typ = 223:15K
boiling point. Te
2109 246.9 0.509 0.623
J
AH =| 118952 Atle :=| 10995 |. ty =| 0533 | Ti =| 0.659
gm
2178 1942 0491 0.629
(a) By Eq. (4.13)
AHexy
— 100% This is the % error
‘AHexp
( -2,732.016 J
(g) -105,140 J
(hy -38,292 J
() 164,647
(i) -48,969 5
(&) 149,728 F
() -1,036,036 J
(m) 207,436 J
(s) -492,640 5
(0) 109,780 J
(u) 235,030 J
(v) -132,038 J
(05) -1,807,968 J
(x) 42,7205
(y) 117,440
@) 175,305 J
4.22 ‘The solution to each of these problems is exactly like that shown in
Example 4.6. In each case the value of AH029g is calculated in
Problem 4.21, Results are given in the following table. In the first
column the letter in () indicates the part of problem 4.21 appropriate to
the AH? value.
TK 4A | 103 4B | 106 ac | 105 Ap | IDCPH | anos
(@ [873.15 | -5.871] 4.181 | 0.000 | -109,795
(6) [773.15 1.861] 3.394 | 0.000 -900,739 |
(p_| 923.15 6,048 | -9.779 | 0.000 “2,716,381
(i) [973.15 9.811 | -9.248 [2.106 189,87
@_| 583.15 9.523 11,355 | -3.450.
_| 683.15 0.441 0.004 | 0,000.
(mj | 850.00 4.575 -2.323 | 0.000
(| 1350.00] -0.145| 0.159 | 0.000
fo) | 1073.15 -1.011 -1.149 | 0.000
@ [M315 | -1424| 1.601 | 0.
()_| 733.15 4.016 | -4.422 | 0.991 117,204
fw) | 750,00 7.297 | -9.285 | 2.520 72] 247,202 |
‘6y {900.00 2.418 | -3.647 | 0.991 34] 128,504 |
(w) | 673.15_| 2.586} -4.189 | 0.000 “1,803,784
| 648.15 0.060} 0.173 | 0.000 22;
6) _{ 108: 4.766 129,628
78
ition of a combination of Eqs. (4.18) & (4.19) with
evaluated parameters. In each case the value of AH(9g is calculated in Pb.
4.21, The values of AA, AB, AC and AD are given for all cases except for
Parts (e), (g), (h), (k), and (2) in the preceding table. Those mi
follows:
[PartNo.[ AA [103.ap [1064c | 10-S.ap |
© -7.425| 20.778 | 0.000 3.737
@ 3.629 | 8.816 | 4.904] 0.114
(h) -9.987| 20.061 | -9.296 1.178
®) 1.704} -3.997 | 1.573] 0.234
@ -3.858| -1.042] 0.180 [ 0.919]
3
150.1082
day
424g
atm
(60-32) Sk +27, ISK T= 288.71K P
‘The higher heating value is the negative of the heat of combustion with water
as liquid product.
Calculate methane standard heat of combustion with water as liquid product:
CH, +20, > CO, +28,0
Standard Heats of Formation:
i
Ajo? =
02 = Orel
= 285830.
mol
A co2 + 2-AH p201ig - AH icra — 2-AH p02
HigherHeatingValue := -AH, AH, = -8.906x 10°.
mol
Assuming methane is an ideal gas at standard conditions:
P gol
Sq 1.793 x 10°——
nae a a
7
4.25 Caleulate methane standard heat of combustion with water as liquid product
Standard Heats of Formation: CH, + 20; ~> CO; +2H,0
3
AHigonig = -74520——
J
Alcon = ~393509
Allgcue = AHyco2 + 2AM pong ~ AHscHs ~ 2-AH 02
J
AHecH4 = ~890649—
mol
Calculate ethane standard heat of combustion with water as liquid product:
Standard Heats of Formation: C,H, + 7/20; -> 2CO, +3H,0
Heong == ~83820——
mol
AllecaHs = 24H ico2 +3-AHpH201iq ~ AHicaHs ~
1560688
‘mol
ABecoH6 =
Calculate propane standard heat of combustion with water as liquid product
Standard Heats of Formation: CsHs + $0; ~> 3CO, +4H,0
1046802
mol
ABicsus
AH osHe = 34H coz + 4 AH DOI ~ AHacsHe ~ SAH yo?
ease
Gas b) has the highest standard heat of combustion.
78
4.26
4.28
2H2 + 02=2420()
C+ 02=CO2%g)
N2(g)+2H2(g)+C(s)+1/202(g)=(NH2)2CO()
AHy99 == AH, + AH + AH ‘ANog
On the basis of 1 mole of C10H18
(molar mass = 162.27)
Q = -43960-162.275 Q=-7.133x 10°F
This value is for the constant-volume reaction:
C1OHI8(1) + 14,502(g) = 10CO2(g) + 9H20()
Assuming ideal gases and with symbols representing total properties,
Q= AU = AH-A(PV) = AH-R-T-Angas
T= 208.15-K Angas ‘= (10 14.5)-tnol
-7.145 x 10°F
AH = Q4R-T-Atggs AH
This value is for the constant-V reaction, whereas the STANDARD
reaction is at const. P.However, for ideal gases H = f(T), and for liquids H
is a very weak funetion of P. We therefore take the above value as the
standard value, and for the specified reaction:
CIOHI8(1) + 14.502(g) = 10C02(g) + 9H20) AH
9H20(\) = 9H20(g) AHyap = 9-44012-5
‘C1OH18(1) + 14,502(g) = 10CO2(g) + 9H20(g)
yap, AHbog = 6748436) Ans.
79
4.29
FURNACE: Basis is 1 mole of methane burned with 30% excess air.
CHa + 202 = CO2 + 2H20¢@)
Entering: Moles methane =n = 1
Moles oxygen ng = 2-13 ng = 2.6
Moles nitrogen ny 15 26 2 ng = 9.781
1
Total moks of dry gases entering n= my +m. +15 13.381
At 30 degC the vapor pressure of water is
4.241 kPa, Moles of water vapor entering:
4.241
—_ 42H 13.381 = 0.585
4 701.325 - 4241 7
Leaving: C021 mol a)
120 ~ 2.585 mol @)
2.6-2= 0.6 mol @)
Nz --9.781 mol @
By an energy balance on the furnace:
Q = AH = AHy9g + AHp
For evaluation of AHy we number species as above.
1 5.457 1.045
3AT0| | 1490 | A
e 7 10"
06 3.639 0.506
9.781 3.280. 0.593
4 Ris 8.314
Swi pe YB
A= 48.692 B = 0.010897
80
‘The TOTAL value for MCPH of the product stream:
Mcpit(303.15, 1773.15, 48.692, 0.897.107, 0.0,-0.5892.105)
MCPH
AHp := R-MCPH-(1773.15 — 303.15)
From Example 4.7: AHgog := ~802625
Q = AHp + AH29g QF
9.895 11
9.89511
“10,61 Ans.
HEAT EXCHANGER: Flue gases cool from 1500 degC to
50 degC. The partial pressure of the water in the flue gases leaving the
furnace (in kPa) is
m2
pp. 101.325 pp = 18.754
ny tn +n3 ng
‘The vapor pressure of water at 50 degC (exit of heat exchanger) is 12.34
kPa, and water must condense to lower its partial pressure to this value.
Moles of dry flue gases: n
nengtng n= 11.381
Moles of water vapor leaving the heat exchanger:
1234
101.325 — 12.34
‘Moles water condensing: An
mi ng = 1.578
2.585 1.578
Latent heat of water at $0 degC in J/m«
AMEgg := 2382.9-18.015
‘Sensible heat of cooling the flue gases to 50 degC with all the water as
vapor (we assumed condensation at 50 degC):
MCPH(323.15,1773.15, 48.692, 10.897. 10" >, 0.0, -0.5892-10°) = 60.01086
MCPH := 60.01086
Q = R-MCPH-(323.15 ~ 1773.15) - An-AHs9 Q''=766;677:] Ans.
at
4.30
4NH13(g) + 802(g) = 4NO(Q) + 6H2O0(@)
BASIS: 4 moles ammonia entering reactor
Moles 02 entering = (5)(1.3) = 6.3
Moles N2 entering = (6.5)(79/21) = 24.45
‘Moles NH3 reacting = moles NO formed = (4)(0.8) = 3.2
‘Moles 02 reacting = (5)(0.8) = 4.0
‘Moles water formed = (6)(0.8) = 4.8
ENERGY BALANCE:
AH = AHg + AHg9g + AHp = 0
REACTANTS: 1=NH3; 2-02; 3-N2
4 3,578 3.020 0,186
65 | A]3.039) B=} 0506|-10? D=| -0.227 j-10
24.45 ) 3.280, 0.593, 0.040
ea: A Svai B yn Bi D Yi
7 i i
A= 118.161 B= 0.02987 D = -1.242x10°
TOTAL mean heat capacity of reactant stream:
McpH(348.15,298.15, 118.161, 0.02987, 0.0,-1.242-10°) = 126.61632
MCPH := 126,61632 AHg := R-MCPH-(298.15 ~ 348.15)
The result of Pb. 4.21(b) is used to get
AHggg = 0.8-(-905468)
PRODUCTSi=NH3; 2:
2; 3-NO; 4-20; 5=N2
os 3.378 3.020 0.186
25 3.639 0.506 0221
32 A= |3.387 0.628 |-1073 0.014 |-10°
48 3.470 1.450 0.121
24.45 3.280. 0.593) 0.040
82
A Yiwai
A= 119.65 B= 0.027 D= 8.873% 10"
Yai D Yer
By the energy balance and Eq. (4.7), we can write:
To:= 298.15 t= 2 (guess)
Given
tisFind(t) 9 1328300 Tis Ty K-t Ans,
431 C2H4(g) + H20(@) = C2HSOH()
BASIS: 1 mole ethanol produced
Energy balance: ayy = Q = AHg + AHgog
AHp9g := 277690 — (52510 - 241818) AHy9g = -8.838x 10"
Reactant stream consists of 1 mole each of C2H4 and H20.
1.424 14394) 4.392
As B H10-* C=
3.470 1.450 0.0
A ae Yai Yee
=-4322x10° D
S 00
os > jh’
0.121
Yd
Lixo
A=4894 B= 0.01584
McPH(298.15,593.15,4.894,0.01584,~4.392-10"§,0.121-109) = 11.1192
83
MCPH := 11.1192
Ag := R-MCPH:(298.15 ~ 593.15) AH = -2.727« 10"
Q = (AHR + AHo98}-5 £5115683) Ans.
4.32, One way to proceed is as in Example 4.8 with the alternative pair of reactions:
CH4+ H20=CO+3H2 ‘AlLyogq = 205813
CH4 + 28120 = CO2 + 4112 AFLgogp += 164647
BASIS: 1 mole of product gases con!
CO; & H20 0.6275 mol H2
g 0.0275 mol CO2; 0.1725 mol
Entering gas, by carbon & oxygen balances:
0.0275 + 0.1725 = 0.2000 mol CH4
0.1725 + 0.1725 + 2(0.0275) = 0.4000 mol H20
AHg9g := 0.1725-AHz9gq + 0.0275: AH 9gb AHgog = 4.003 x 10"
02
0A
1.102 9.081 2.164 x
( ) oe jo cs 10° ® b= (.°° Ji’
3.470 1.450. 0.0 \o.121)
A ya AUB = Bi Yc D yn Dj
LR8 B= 2396x107? C= -4.328x107 Da 484% 10?
‘The energy balance is written
Q= AHR + AHp9g + AHp.
REACTANTS: 1=CH4;2=H20 i
1cPHH(773.15, 298.15, 1.728,2.396-10
4.3310 ,4,84-10°) = -1377.435
137.435 AHR := RICPH AH = -1.145x 104
ICPH =
PRODUCTS:
CO; 3=H20;
=H?
4
B= 6.397% 10 D = 3.579% 10°
rcpi{298.15,1123.15,3.370,6397-10"4,0.0,3.579-108) = 3164.293
ICPH := 3164.293 ‘AHp := RICPH ‘AHp = 2.631 10"
Q = (Hp + 4Ha98 + AHp)-J QE 54886) Ans.
4.33 CH4 +202 = CO2 + 2H20(g)
C2H6 + 3.502 = 2CO2 + 3H20(@) AFg9gp = — 1428652
BASIS: 1 mole fuel (0.75 mol CH4; 0.25 mol C2H6) burned completely with
80% xs. air.
02 in = 1,8§(0.75)(2) + (0.28)3.5)] = 4.275 mol
N2 in = 4.275(79/21) = 16.082 mol
Product gases: CO: 1S + 2(0.25) = 1.25 mol
H20 = 2(0.75) + 3(0.25) = 2.25 mol.
02 = (0.8/1.8)(4.275)
N2= 16.082 mol
AH 9g != 0.75-AHl9gq + 0.25-AHpogp Q:=-8:10°
9 mol
Energy balance: Q = AH = AHz9g+AHp AH = Q= Ay 3
PRODUCTS: 1=CO2; 2=H20; 3=02; 4=N2
1.25 5.457 1.045 “1.157
2.25 3.470 1450} 3 o121 | 5
= A B K10-* D 10"
19 3.639 0.506
16.082 ) 3.280 0.593,
85
4.34
As Sima B Yves
A=74292 B= 0015 D = 9.62% 10"
By the energy balance and Eq. (4.7), we can write:
To = 303.15 t= 2 (guess)
) t= Find(t)
eee
Given Q=AHg9¢ = RY A-To-(t = 1) + To (:
= L788 TH542K Ans.
BASIS: 1 mole of entering gases containing 0.15 mol SO2; 0.20 mol
02; 0.65 mol N2
S02 +0.502=S03 Conversion = 86%
S02 reacted = SO3 formed = (0.15)(0.86) =
02 reacted = (0.5)(0.129) = 0.0645 mol
Energy balance: AH773 = AHg + AH29 + AHp
Since AHy and AHy cancel for the gas that passes through the converter
unreacted, we need consider only those species that react or are formed.
Moreover, the reactants and products experience the same temperature
change, and can therefore be considered together. We simply take the
number of moles of reactants as being negative. The energy balance is
then written: AH773 = AHy9g + SH yet
AHg9g =
~395720 = (~296830)}-0.129
1: $02; 2: 02; 3: SO3
0.129 5.699 0.801 =1.018
~0.0645 | A=] 3.639] B:=10506}10-9 D3=| -0.97 |-10°
0.129 8.060. 1.056 ~2.028
A= 0.06985 B= 2.58% 10" D
McPH(298,15,773.15,0.06985,2.58-10" 7, 0.0,-1.16-104) = 0,019666
MCPH := 0.019666 AFIngt == R-MCPH(773.15 ~ 298.15)
AH773 = (AH295 + AHpet)-J Ans.
4.35 CO(g) + H20(g) = CO2g) + H2g)
BASIS: 1 mole of feed consisting of 0.5 mol CO and 0.5 mol H20.
moles H20 reacted = moles CO2 formed = moles H2
(0.6)(0.5) = 0.3
Product stream: moles Ct
moles CO2 = moles H2
formed
= moles H20 = 0.2
3
Energy balance: Q= AH = AHR + AHg9g + AHp
= 0.3:[-393509 — (-110525 ~214818)] __ AHy9g = -2.045 x 10°
CO 2: B20
( .)
As
3.470.
0557) 3 0.031) 5
Bs -10 Ds 10
1.450, 0.121
D -> Di
004x107 D = 4.5x 10°
McPH(298.15,398.15,3.423, 1.0035-1073,0.0,0.045 10°)
MCPH := 3.8103
3.8103
Aly := R-MCPH.(298.15 ~ 398,15) AH = ~3.168« 10°
Products: 1:CO 2: H20 3:CO2 4:H2
87
02 3.376) 0.557 =0.031
B 3.470 1.450 os 0.121
a As B ho Des -10°
5,457 1.045 1.157
03 3.249 (oa 0.083
Ac x -Ay OB YB D ya Dj
i i 7
A= 3.981 B=84isxi0* —D=-3.042x104
McPH(298.15,698.15,3.981,0.8415-10"°,0.0,-0,3042-10°) = 4.25405
MCPH := 4.25405
‘Alp = R-MCPH- (698.15 ~ 298.15) AHp = 1415 10"
Qs (Alig + AHp9g + AHp)- ‘Ans,
436 BASIS: 100 tbmol DRY flue gases containing 3.00 Ibmol CO2 and 11.80
Umol CO x Ibmot 02 and 100-(14.8-x)= 85.2-x Ibmol N2. The oil therefore
contains 14.80 tbmol carbon;a carbon balance gives the mass of oil burned:
12.011
0.85
14.8. Ibm = 209.133 Ibm
The oil also contains H20:
ZO O0} pmol = 0.116ibmot
18.015
Also 20 is formed by combustion of H2 in the oil in the amount
209.133-0.12
Ibmol = 12.
sig tbe! 48 lbmol
Find amount of air entering by N2 & 02 balances.
N2 entering in oil:
0.02
© pmol = 0.149 bmol
tbmol N2 entering in the air=(85.2-x)-0.149 85,
88
Imo! 02 in flue gas entering with dry air =
3.00 + 11.8/2 + x + 12.448/2 15.124 + x Ibmol
(C02) (CO) (02) (H20 from combustion)
‘Total dry air =N2 in air + O2 im air = 85.051 - x + 15.124 + x = 100.175 Ibmol
Since air is 21 mol % 02,
= ISM +x
100.175
0.21 (0.21-100.175 ~ 15.124)-Ibmol__x = 5.913 Ibmot
02 in air = 15.124 + x = 21.037 Ibmols
N2 in ai 5,051 - x = 79.138 Ibmoles
N2in flue gas = 79.138 + 0.149 = 79.287 Ibmols
(CHECK: Total dry flue gas
= 3.00 + 11.80 + 5.913 + 79.287
= 100.00 Ibmol]
Humidity of entering air, sat. at 77 degF in Ibmol H2O/Ibmol dry air,
P(sat)=0.4594(psia)
0.4594
14.696 — 0.4594
Homo! H20 entering in air:
= 0.03227
0.03227-100.175-Ibmol = 3.233 Ibmol
If y = Ibmol H20 evaporated in the drier, then
Ihmol H20 in flue gas = 0.116+12.448+3.233+y
5.797 + y
Entering the process are oil, moist air, and the wet material to be dried, all at
77 degF. The "produets” at 400 degF consist of:
3.00 lbmol CO2
11.80 Ibmol CO
5.913 Ibmol O2
79.287 Ibmol N2
(15.797 + y) Ibmol H20(g)
Energy balance: Q = AH = AHy9g + AHp
where Q = 30% of net heating value of the oil:
a9
= -03-19000- 22. 209.13:1bm
Ibm
1.192 10°BTU
Reaction upon which net heating value is based:
OIL + (21.024)02 = (14.8)CO2 + (12.448 + 0.116)H20(g) + (0.149)N2
=19000-209.13-BTU ‘AHg9gq = -3.973 x 10°BTU
‘To get the "reaction" in the drier, we add to this the following:
(11.8)CO2 = (11.8)CO + (5.9)02
AH 988
AHgggp = 11,8:(-110525 + 393509)-0.42993-BTU.
(yH20(g) Guess: y = 50
Allggge(9) := 44012-0.42993-y-BTU.
(y)H2000
(The factor 0.42993 converts from joules on the basis of moles to Btu on the
basis of lbmol.]
Addition of these three reactions gives the "reaction" in the drier, except
for some 02, N2, and H20 that pass through unchanged. Addition of the
corresponding delta H values gives the standard heat of reaction at 298 K:
AHp9g(y) #= AHo9gq + AH29gp + AH29ge(¥)
For the product stream we need MCPH:
1:CO2 2: CO 3:02 4:N2 5: H20
To = 298.15 R= 1,986 T wee T = 477594
3 5.457 1.045 1.157
18 3376 0.587 0.031
ny) =| 5.913 A=] 3.639 0.506 |- 0.227. |-10"
79.218 3.280 0.593 0.040
15.797 +y. 3.470, 1.450 0.121
LS AQ)= Vawra; BO) =YawrB: Do = Yor;
1
= t= 1602 Cply) = R [aoe Pao +420
2
Ty
90
Te = 562.2K 8.98bar — Z_ 25 0.271
353.2K That
Ty = 0.628 Pe 0.02
Generalized Correlations to estimate volumes
Vapor Volume
0.422
PO OURS eet Eq. (3.61)
Ty
By = 0.139 - By = -1.073 Eq. (3-62)
Py Pr
Ze lsBox obra Z= 0967
ZRTy 3
Ves V = 2.838% 19°
- mol
Liquid Volume
257 3
Vogt = Vere) Eq. 3.63) Vat = 96.807"
mol
‘Combining the Clapyeron equation (4.11) AH = T-AV-4 Pye
a
eae
with Antoine's Equation Pay =e ™C
gives AH ® T-AV-
(toy
67
5
AV=V=Veap AV = 2.829 19°
mol
B= 277378 C= 53.00
Answers for parts (b)-(e)
() Ant = 36.262 ©
mol
( an=32.278
mol
(@) an=28948 2
mol
(© aH =33.838
mol
4.13, Let P represent the vapor pressure.
T = 348.15-K
100-kPa
(
Given hl ee 5 48.157543 -
kPa}
ind(P) dPdT := P.
Ree
P = 87.396KPa ‘AH = 31600.
68
5622.7-K
((5622.7K _ 4.70504)
(guess)
4.70504: a
T
joule
mol
Clapeyron equatio
V= vapor molar volume. V = Viq+ SY
T-apaT
F439: Bn v{2¥ 4) Ans.
38): et }
4.14 (a) Methanol: Ty = 512.6K 0.97bar Ty = 337.9K
AL: 13.431 -81.28-10~ = 131.1310" ©
fee q
CpL(T) =| AL+ 14 = js
eRe
Ay =2211 By := 12216-1077
Cpy(T) = (megeres re yee
Trsat =
ki
AHg = 38.301 —
mol
eikd
Ally = 35.645
mol
69
12
Text 7
encnet vany+ | cey(naT alt= 49.38"
1 Tost a
nes oo mel Q=n-AH ‘Ans.
ir
(b) Benzene: aH, = 28.273. 55.296 2.
moi mol
J
Ky
(© Toluene AH, = 30.625— AH = 65.586 —
mol mol
4.15 Benzene T.:= 5622K Pg
Tisat 2 451.7K — Tosat = 358.7K Cp
Estimate AHy using Riedel equation (4,12) and Watson correction (4.13)
Tosat
Ten = 0.628 Trasat Trasat = 0.638
30.5882
mol
aly = 30.285.
mol
is adiabatic and isenthalpic.
Assume the throttling proces:
Guess vapor fraction (x): x := 0.5
Given Cp-(Tisat— Tasat) = XAHy x= Find(x) KE 04982 Ans.
189.4-K
308.3-K
4.16 (a) For acetylene: T, 61.39-bar Ty,
T= 298.15-K
70
Th T
rut Tv ned erat T= 0087
AH, = 1691
mol
any = 6638-2.
mol
(b) For 13-butadiene: Agog = 88.5
mol
ig
(©) For ethylbenzene: AHy5g = —12.3——
mol
Ws
(@) For n-hexane: AH5g = -198.6—
mol
KI
(©) For styrene: AFg9g = 103.9-—
mol
4.17 Ist law: dQ = dU -dW = CydT+P-dV A)
Ideal gas: PV=SRT and PdV+V-dP = RdT
Whenee V-dP = R-dT- P-dV @)
Since p-V? = const then P.8-VO"Lav.
from which V-dP
—Pb-dV
‘Combines with (B) to yield: Pav = So
1
4.18
R
Combines with (A) to give: AQ = Cyat + a
or dQ= Cpar-Rars RH
which reduces to
Since Cp is linear in T, the mean heat capacity is the value of
Cp at the arithmetic mean temperature. Thus Tam ‘= 675
Cem 85+ 0.57:10"?-Tam)
Integrate (C): 50K
Ans,
For the combustion of methanol:
CH3OH(g) + (3/2)02(g) = CO2g) + 2H20(g)
AHQ98
393509 + 2-(-241818) — (-200660)
AH 9g = ~676485
For 6 MeOH:
Ans,
For the combustion of 1-hexene:
(COH12(g) + 902(g) = 6CO2(g) + 6H20(g)
72
4.19
AHg9¢ := 6-(~393509) + 6(-241818) - (-41950)
27 Ams.
AHg9g = ~3770012 AHao@ ='=3,770,01
‘Comparison is on the basis of equal numbers of C atoms.
C2H4 + 302 = 2CO2 + 2H20(g)
Bog = [2-(-241818) + 2-(-393809) ~ $2510]. —
mol
mple 4.7. However, with
simpler to proceed differently.
Parts (a) - (d) can be worked exactly as
Mathead capable of doing the iteration,
Index the product species with the numbers:
1= oxygen
2= carbon dioxide
3 = water (g)
4= nitrogen
(a) For the product species, no exeess air:
0 3.639 0.506) ~0.227
2 5.487 1.045 | 10-3 “137
2 3.470 1.450 | K oat
11.286, 3.280, 0.593, 0.040.
4 A Yai B: Sg
A = 54.872 B= 00125 D = ~1.621«10°K
oT
cp
For the produets, Ape R | FedT Tos 298.15K
Jig
The integral is given by Eq. (4.7). Moreover, by an energy balance,
Algo + AHp = 0
3
t= 2 (guess)
B
Given ~AHg9g = R- [4 To(t-1)+ ; 92
Find(z) += 8497 Tis Tot = EE 25335K Ans.
Parts (b), (¢), and (d) are worked the same way, the only change being in the
numbers of moles of products.
6) nq, = 14.107 198.6K Ans.
© ho, = Rn, = 16.929 T= 1950.9K Ans.
(a) no, = 3.0 Ng, = 22.571 T= 1609.2K Ams.
(©) 50% xs air preheated to 500 degC. For this process,
Alb + AH 299 + AH
AHgir = MCPH-(298.15 ~ 773.15)
For one mole of air:
McPHi(773.15,298.15,3.355, 0575-10
For 4,5/0.21 = 21.429 moles of air:
R-MCPH-AT
[aie = 21.429-8.314-3,65606-(298.15 — 773,15)
mol
b
=
i
AH gi, = -309399
mol
‘The energy balance here gives: AHyog + Agi, + AHp =
%
1s 0.227
2. 1,157
a Ds -10°-K?
2 0.121
16.929, 0.040
Ds domi
A= 78.84 Be oie D ==1.735 x 10°K?
t= 2 (guess)
Given —AH998 - AHair = R} A-To-(t ~ 1) 2 tele? — 1)...
D
+P
To
tis Find(z) 1 = 7.656 Tis Tp Kt
420 n-CSH12 + 802 = 5CO2 + 6H20(1)
By Eq, (4.15) with data from Table C.4:
Ans.
4.21 The following answers are found by application of Eq. (4.15) with
data from Table C4,
(a) -92,220 5 (n) 180,500 5
(b) -908,468 J (0) 178,321 5
(c) -71,660 3 (p) -132,439 J
(a) -61,980 5 (@) 44,3703
(©) -367,582 J () -68,910 J
75
437
4.38
Given Cp(y)-(400 - 77)-BTU = Q- AH99(9) Find(y)
y = 49.782 (Ibmol H20 evaporated)
= 4388. (lb H20 evap. per Ib oll burned)
DORIS oe ‘a
Whence
One mole of product gas containing 0.242 mol HCN, and
= 0.379 mol each of N2 and C2H2. The energy balance is
Q= AH = AHa93 + AHp
‘AHy9g := (2-135100 — 227480). a AHiggg = 5.169% 10°
Products:
0.242) 4.736 1.359 0.725
0379| A= /3.280] B:=/.0.593 10> D =| 0.040 |-108
I 6.132, 1.952. =1,299
3 A
yn Dj
Sra B ye Bi D
i
R:= 8314 A=47133° B= 12934x10> Dp = -6.526x 10"
McPH(298.15,873.15,4.7133, 12934-1073, 0.0, -6.526-104)
22010
MCPH := 5.22010
AHp := R-MCPH-(873.15 ~ 298.15)J AHp = 2.495 x 10
Q := AHgog + AHp GQ S304] Ans.
BASIS: 1 mole gas entering reactor, containing 0.6 mol HCI, 0.36 mol 02,
and 0.04 mol N2.
HC reacted = (0.6)(0.75) = 0.45 mol
4HCKg) + 02(g) = 2H20() + 2CL(g)
For this reaction,
31
Agog #= 2-(-241818) ~ 4-(-92307) AHg9g = -1.144 x 10°
Evaluate AHgp3_ by Eq. (4.21) with
To s= 298.15 Ts 823.15 R:= 8314
1:20 2: C2 3: HCI
2 3.470 145 0.121
4.442 0.089 3 0.344} 5
A= Be 10 10"
3.156 0.623 0.151
=I 3.639 0.506, 0.227
“4 AA > a AD = SnD;
5 a
AA =-0439 AB = 8x 10 AD = -8.23 x 10
8.23.10) = -0.72949
MDCPH(298.15,823.15,-0.439,8.0-107
MDCPH := ~0.72949
AHgp3 := AHy93 + MDCPH-R-(T - To) AHg93 = 117592
Heat transferred per mol of entering gas mixture:
AHg23°5
Q = ‘0.45 ‘Ans.
4
439 CO2+€=2C0 @
2C + 02=2C0 &
Eq. (4.21) applies to each reaction:
For (a):
2 3.376 0.557
nef-1] Acs/ 1771 o7m|io> Dp -10°
ah 5.457, 1.045
AA nA; AB Bj AD » “Dj
a
92
AA=-047 = AB=-7.02x10* AD = 1.962% 10°
MpcpHi(298.15,1148.15,-0.476,-7.02-10"*,0.0,1.962-108) = -0.410505
0.410505
AH29gq + R- MDCPHe-(1 148.15 ~ 298.15) SH 148q = 1.696% 10°
MDCPH,
SH 148
3.376 0557 0.031
3.639} B:=| 0.506 |-107* — D:=| -0.227 |.10°
L771 0.771 0.867 ,
AA
De aD = Sn Dj
AA = 0.429 AB=-934x 1074 AD = 1.899 10°
MDCPH(298.15, 1148.15,-0.429,-0.934.10"3,0.0, 1.899.10°) = -0.549680
MDCPHp := -0.549680
AH} tagh °= AHoogp + R-MDCPH (1148.15 — 298.15)
AH tap = 2.249 10°
The combined heats of reaction must be zero:
nco,'AHy 48a +0,°AH) 1480 = 0
Define: r r= 1327
For 100 mol flue gas and x mol air, moles are:
Flue gas, Air Feed mix
coz 12.8 0 12.8
co 37 0 37
93,
oz 34 o21x 54+ 0.21x
N2 781 0.79. 78.1 + 0.79x
128
Whence in the feed mix: p= ——__
5.44021
19.155mol
Flue gas to air ratio= Ans.
Product composition:
neo #= 3.7 +2-(12.8 + 5.44 0.21-19.155) ng = 48.145
ON, = 78.1 +.0.79-19,155 ON, 93.232
Mole % CO=
Mole % N2=
4.40 CH4 +202 = CO2 + 2H20(@) AHg9gq = ~802625
CH4 + (3/2)02 = CO + 2H20(g) AH 9gp *= ~519641
BASIS: L mole of gas consisting of 0.94 mol CH4 and 0.06 mol N2
Air entering contains:
1.35-2.0,94 = 2.538 mol O2
mol N2
‘Moles CO2 formed by reacti 0.94-0.7
1.058
Moles CO formed by reaction = 0,94-0.3 = 0.282
J
AHa9g = (0.658-AHa994 + 0.282-AHaog5} AHo9g = -6.747 x 10°
mol mol
4
Moles 1120 formed by reaction = —_0,94.2.0 = 1.88,
Moles 02 consumed by reaction= 2.0.658 + 3 0.282 = 1.739
Product gases contain the following numbers of moles:
0.658
0.282
1.880
2.538 - 1.739 = 0.799
9.548 + 0.060 = 9.608
0.658 5.457 1.045.
0.282 3.376 0.557
n:=| 1.880 As=|3470] B=] 1.450 |-1077
0.799 3.639 0.506
9.608. 3.280 0.593
R:=831
A= 454881 B= 9,6725x 10
mohK
McPH(298.15, 483.15,45.4881,,9.6725-107?,0.0,-3.396-10") = 49.03001
MCPH := 49,03091
J
Ap := R-MCPH-(483.15 ~ 298.15)-K AHp = 7.541 x of
mol
Ki
Energy balance: Adley = Alz99 + AH AFlyy = ~599.252-—
mol
kg
AH yp9-mdoty20 + Sy ndotjgel = 0 mdoty20 = 34.028.
see
kT
From Table C.1: AH29 '= (398.0~ 104.8).
‘B
AH} 29'mdotr20
ah
alin dott = 16.635
6.635—
see
dott
95
Volumetric flow rate of fuel, assuming ideal gas:
ndotfyet"R:298.15-K
101325-Pa
Ans.
441 C4H8(g) = CAH6(g) + 12(@) 1os780-—
mmo
BASIS: 1 mole C4H8 entering, of which 33% reacts.
‘The unreacted C4H8 und the diluent H2O pass throught the reactor
unchanged, and need not be included in the energy balance. Thus
1 To 2= 298.15:K T= 798.15K
1 Evaluate AHy9g__ by Eq. (4.21):
A 1: C4H6 2: H2 3: C4H8
2.734 26.786 8.882 0.0
3.249.) B=] 0.422 |-107? 0.0 |-107® D =} 0.083 |.10°
1.967 31.630 9.873 00
AA
Yovd awe Pmw ae mc, AD Sad;
AA = 4.016 AB =~4.422 107 aC = 991x107" pd = 83% 10°
MDCPH(298.15,798.15 ,4.016,~4.422-10" 3,991.10" 7,0.083-105) = 1.94537
MDCPH := 1.94537 {
‘AHi29g + MDCPH-R-(T ~ To) Agog = 1.179 108
mol
AH795
Q = 0.33molAH 7g Q
Chapter 5 - Section A - Mathcad Solutions
5.2 Let the symbols Q and Work represent rates in kJ/s. Then by Eq. (5.8)
[Work]
[Qn]
Te
98.15-K Qu = 250-
Ans.
By Eq. (5.1), Qe = [Qu] - [Work]
5.3 (a) Let symbols Q and Work represent rates in kJ/s
Ty := 750K
By Eq. (5.8):
But
(b) ny == 0.35
84 (a) Te := 303.15-K Ty = 623.15-K
Te
fee oe == 0.55-1Carot 20282 Ans.
TH
7
By Eq. (5.8), Tu *
5.7 Let the symbols represent rates where appropriate, Calculate mass rate of
LNG evaporation:
P= 1.0133-bar 198.15-K
in PV
molwt := 1722 miNG = —~-molwt mung = 62548
‘mol RT s
Maximum power is generated by a Carnot engine, for which
[work] _ |Qr] - [Qc|
[ee] [ee]
Ty = 308.15-K
Ca
Qc = 512 -mpxo
kg
i (Ta
Work = Qe] — Ans.
(Te
Qu = Qc + Work Ans.
ASrorat = ASH20 + ASres
is the same as in (a), and the total
ivided into two halves.
(b) The entropy change of the water
heat transfer is the same, but
(sche
AStotal “= AS res + ASH20
(©) The reve -s an infinite number of heat
reservoirs covering the range of temperatures from 273.15 to 373.15 K,
each one exchanging an infinitesimal quantity of heat with the water
and raising its temperature by a differen
Ty = 500K
Pv
ns n= 1.443mol
RT;
(a) Const.-V heating; AU = Q+W=Q=n-Cy-(T)-T))
Tre Ti+ T= 1x 10K
acy
T) Po)
By Eq. (6.18), $= tn| — | - Reta]
ty Eq. (5.18), AS ofcr (®) nn(Z}
ai
cin 2) a8 = 20.7042 An
Syn] = 1794 Ans.
n-Cyln| ti AS: ! Mie
‘Whence
PT)
(b) The entropy change of the gas is the same as in (a). The entropy
change of the surroundings is zero. Whence
AStotal = 10. 794: 2 Ans.
The stirring process is irreversible.
99
5.10 (a) The temperature drop of the second stream (B) in either
case is the same as the temperature rise of the first stream Cp
(A), ie, 120 degC. The exit temperature of the second
stream is therefore 200 degC. In both cases we therefore
have:
(463.15)
463.15 473.15
Sq = Cptn| SO Sp = Cprln
Ane Sas 15) a ie 8)
Ans.
(b) For both eases:
AStotal ‘= AS + ASB Ans.
(¢) In this case the final temperature of steam B is 80 degC, ie., there is
a 10-degC driving force for heat transfer throughout the exchanger.
Now
co 463.15) in( 353 3)
Pe 343.15 ) \ 473.15
Ans.
AStotal “= ASq + ASB Ans.
ag
5.16 By Eq. (5.8), AW = dQ= To
Since dQ/T = dS, AW = dQ - To-dS
Integration gives the required result.
T= 400K To = 300K
582x100
mol
100
J
molK
Th
AS := Cp-In| — AS = ~11.799-
T
Work := Q—Tg:AS Ans.
Qe = |Q| ~ [Work] Ans.
Qo
ASreservoir = = Ans.
Te
AS + ASreservoir Process is reversible,
SAT Tyr := 600K = Tey := 300K = Tyg = 300K = Tea := 250K
For the Carnot engine, use Eq. (5:8): Jw} _ Ty - Tea
[Qui] Tar
The Carnot refrigerator isa reverse Carnot engine. Jw] _ Ty2—Te2
‘Combine Eqs. (5.8) & (5.7) to get: _—
a (G-7) tog Jecsp
Tez
Equate the two work quantities and solve for the required ratio of the heat
quantitic eee
Tex fin=Te1)
Tu (TH = Tee
Ans,
7
S48(@) Ty = 300K Py L2bar Tp = 450K Pp = Ghar Cp == GR
AH
AS
©) onze ase 1.484
mol mol-K
101
( aH= 3.8108
mol
@ ane
mol
@ als 6651-1080
mal
5.19 The cycle is the same a
identificaitons C=1; D=
page 298:
For the given data:
5 ( 973.15 - 473.15
7 (1973.15 — 773.15,
J
mokK
J
molK
J
mobK
AS = 4.953,
AS = 2.618
AS = -3.607
shown by Fig. 8.10 on page 298. With the
; A=3; and B=4, the efficiency is given by Eq. (A) on
Work = 3.6x 10°
mol
Whenee Tp = T) + YOK
ov
AN 374K
Ans.
102
T= 800
Step 1-2: Volume decreases at constant P.
Heat flows out of the system. Work is done on the system,
Wioe {P(V2-vi)J= 4R-(12-T1)]
Step 2-3: Isothermal compress
out of the system.
W: R-Ta-h = R-T2h ba)
= R-Tyla| 2] = R-TyMa
PruCaare (Po ley
Step 3-1: Expansion process that produces work. Heat flows into the
system. Since the PT product is constant,
‘Work is done on the system. Heat flows
P-dT+T-dP = 0 (A)
PVERT PdV + V-dP = R-dT
PdV = R-dT- V-dP = RAT roe
In combination with (A) this becomes
PAV = RT + RdT = DRT
Moreover,
V1
-| Pave
V3
Q5I = AUs1 — W31 = Cy-(T) - T3) #2-R-(T1 = Ts)
Ws
2R-(T1- 13) = -2R(T1 -T2)
Qi = (Cv+2R)(T1 Ts) = (Cp+R)(T1~T2)
[Woe] _ [Wi2-+ Was + Wai]
1
Qa1
1
Cpe gR 1 = 700-K 350-K
403
Py = LS-bar
3 J
={R(m2-1 Wiz = 2.91x 10 —
Wr = {R(T -T1)] 2 =
P3) 3_J
Wo3 = R-Te in| > W3 = 2.017 10° —
(Pi) mol
3]
v31 = -2R(T) -T. Wai > -5.82x 10° —
War = -2R(T1-T) 1 mal
J
Q31 = (Ce +R)-(T1-T) Q31 = 1.309 104
[Wiz + Was + Wai]
@i
5.26
403.1SK Py
6.Sebar Tres =.298.15-K
By Eq. (5.18), AS
With the reversible work given by Eq. (3.26), we get for the actual W:
(Po
Work := 1.3-R-T-ln| =] (Isothermal compresion) Work = 4.163 10° —
(Pt
Q:=-Work Q here is with respect to the system.
So for the heat reservoir, we have
Ans,
Ans,
5.27 (a) 10PS(473.15,1373.15,5.699,0.640.10"3,0.0,~1.015-10°) = 6448223
ICPS
448223,
104
By Eq. (5.14) with P = const. and Eq. (5.15), we get for the entropy
change of 10 moles
0-mo) AS = n-R-ICPS
(by 10Ps(523.15,1473.15,1.213,28.78
ICPS := 20.234265
By Eq. (5.14) with P= const. and Eq, (5.15), we get for the entropy
change of 12 moles
2-mol AS = n-RICPS 4s
5.28 (a) The final temperature for this process was found in Pb. 4.2a to be 1374.5 K.
‘The entropy change for 10 moles is then found as follows
0.0) = 10.835459
10PS (473.15, 1374.5, 1.424, 14394-1073, ~4,392-107
ICPS := 10.835459 nn s= 10-mol
AS == mR-ICPS Ass 900 o> Ans,
(b) The final temperature for this process was found in Pb. 4.2b to be 1413.8 K.
‘The entropy change for 15 moles is then found as follows:
630-107, -9.873-10" §,0.0) = 21.309212
eps(s33.15,1413.8, 1.967
ICPS := 21.309212
AS = 1 RCPS
(c) The final temperature for this process was found in Pb. 4.2c to be 1202.9 K.
‘The entropy change for 18.14 kg moles is then found as follows
icps(s33.15, 1202.9, 1.424,14.394-10" 3, ~4,392:107§,0.0) = 8.245741
ICPS := 8.245741 n:= 18140-mol
AS = mRICPS aS= 4296x102 Am.
105
5.29 The relative amounts of the two streams are determined by an energy
balance. Since Q= W = 0, the enthalpy changes of the two streams must
cancel. Take a basis of 1 mole of air entering, and let x = moles of chilled air.
Then 1 - x= the moles of warm air.
To = 298.15-K ‘Temperature of entering air
Tr
Tz
x€p(Ty = To) + (1 =»)Cp-(T2-To) = 0
248.15-K ‘Temperature of chilled air
48.15-K ‘Temperature of warm air
Given
Thus x = 0.5, and the process produces equal amounts of chilled and warmed
air. The only remaining question is whether the process violates the second
law. On the basis of 1 mole of entering air, the total entropy change is as
follows.
Pp
-bar
1; Ty
AStotal = * con(Z) +(1=x)-Cp (2 \- nu)
\To, 0) Po
Ans.
Since this is positive, there is no violation of the second law.
5.30 Ty := 523.15-K
Cyi=Cp-R Q = AU - Work
108
Q
Q=-1.733% 10
ASpes “= ASres = 5.718
Tres mokK mol
T Pp J
pin] — | ~ R-ln} AS = -2.301
TT PL molK
ASjoil = AS +AS;e —-“ASigg = 34=EE_ PROCESS IS POSSIBLE.
mol
5.33 For the process of cooling the brine:
pa aTi=-40K —— mdot:= 20-8
kek sec
Ty := (273.15 + 25)-K Tr = 298.15K
T2:= Q73IS-15-K Tz
To = (273.15 +30)-K Ty = 303.15K
Cp
258.15K
I
AH = -140—
kg
as = -0.501 2.
kek
Eq. (5.26): Wdotideal “= mdot (AH —Tg-AS) Wdotigeal = 256.938kW
By Eq. (5.28): wjoy . dotideat
mM
Wdot.=951.6kW Ans.
534 E = 110-volt 9.7-amp
Wdotmech != -1.25-hp Wdoteicct = FE Wdotetect = 1.067 x 10° W.
At steady state: Qdot + Weoteject + Wdotmech = fu! =0
it
Qdot
+Sdotg =
To dt
107
Qdot := -Wdotetecr~ Wdotmech __ Qdot = -134.875W
Ans,
Sdowg
O-amp = Ty = 300K
Waotetect = 2.5% 10° W
At steady state: Qdot + Wdoteleat = LU
dt
Qdot
S +sdotg
5.38 mdot = 10——— Ty = (25+273.15)K Py 2= 10bar
Ans.
(©) Sdotg := mdot-AS
(@) Ty =
20+273.15)K — Whost = To AS
108
5.39(a) Ty = 500K Py == Gbar = Tz = 371K Pp s= 1.2bar
To = 300K Basis: 1 mol
AH = n-Cp(T2-T1) W,:= AH ‘Ans.
1s a(con(2)-n0()
Eq.(527) —— Wideal = (AH = To-AS) Ans.
Eq. (5.30) Wiost = |Wideal ~ Wsl Ans.
Eq. (5.39) Ans.
Ww, Widest Wise
(a) -3753.8) ~5163) 1409.37 4, 8
(b) ~2460.93 -2953.91 493) 16432
(©) -3063.75 -4193.71 11300 a672
(a) -3853.5) 4952.4) 1098.8) 30632
(© -3055.45 4119.21 1063.81 3462
Py := 2500kPa Po = 150kPa To = 300K — mdot = 20!
(P2) I =
as
~Ren| = AS = 0.023.
Py
Sdotg = mdot-AS
Waotiost *=
109
$42 Quis IK) Wee 045K) Ty = (2504273.15)K Ty = 523.15K
Te = (25+273.15)K = 208.15K
Mactuat = 045
Nmax = 0.43
sv the process is impossible.
Qi = 50K
Ty = 350K
Q@
T2
Ty = GIS +273.15)K Te 204273.15)-K
Ty = 588.15K To = 293.15K
Ans,
Qdoty := LWeotl Qdote = Qdoty ~ [Waot]
Qdote =:745.297MW (minimum value)
| waot!
(b) = 0.6-Nmax Qdoty, -= aot! Qdoty = 2.492 10° W
n
Qdote == Qdoty - |Weot} (MW (actual value)
River temperature rise: Vdot := 165:
8
1 re
oar eee Ans.
emK Vdorp-Cp
110
67
6.8
Chapter 6 - Section A - Mathcad Solutions
At constant temperature Eqs. (6.25) and (6.26) can be written:
dS = -B-V-dP and dH = (1 ~B-T)-v-dP
For an estimate, assume properties independent
of pressure.
1200-kPa
2095-10 2." !
Te = 408.1-K 28
emK
gm om?
mobwt + 58,1232 262.7.
mol ‘mol
Eq. (8.63) for volume of a saturated liquid may be used for the volume of a
compressed liquid if the effect of pressure on fiquid volume is neglected.
359 088 |
T
T =| 360 /-K Tr Tr = | 0.882
|
361) fe
(The elements are denoted by subscripts 1, 2, & 3
oF 131.601
2857 3
v =| vere 1] [ee Fiscal
| mol
(132.68
Assume that changes in T and V are negligible during throtling. Then Eq.
(6.8) is integrated to yield:
1
AH = T-AS4+V-AP but
-vr(P2-Pi)
Tr
AH= 0 Then at 360K,
AS
el a
We use the additional values of T and V to estimate the volume expansi
3
AV = V3-V, AV = 1.079. AT = 13-7, AT=2K
mol
-1.Av B = 4.098755 103K"!
Vi AT
Assuming properties independent of pressure,
Eq, (6.29) may be integrated to give
AS = cet BV-AP AP :=Po-Py AP = -2x 10°kPa
Ty (a8 +8-VraP)
Whence t= 10S +B-¥r AP)
TORK Ans.
Cp molwt :
69 T= 208.15-K Pj = Lbar Pp = 1500bar
3
250-107 87! bar”? Vip = 1003
kg
em?
By Eq. (3.5), Vo := Vyexp[-1-(P2~Pi)] Va = 937. 54
: 8
Vi tV. 7
Vave "= = Vave = 970.2875 By Eqs. (6.28) & (6.29),
AH = Vave-(1- B-T)-(P2- Pi) AU := AH ~(P2-V2~ PV)
B AU 8 ns.
ke
Qs TAS Work := AU-Q
112
6.10 For a constant-volume change, by Eq. (3.5),
f(T2-T1)—K-(P2—P))= 0 Ty = 298.15-K Ta: 323.15-K
1 Py bar
= 442-10" °-bat™
36.210 °K"!
Ans.
6.14 --- 6.16 Vectors containing T, P, Te, Pe, and @ for Parts (a) through (n):
300 40 308.3 61.39 (187)
115 15 150.9 48.98 000 |
575 30 562.2 48.98 210
500 50 425.1 37.96 200
325 60 304.2 73.83 24
175 60 132.9 34.99 048
piel? lag pel? tour tere | 2954 {5 oar vc 18
650 30 553.6 40.73 210
300 35 282.3 50.40 087
400 70 373.5 89.63 094
150 50 1262 34.00 038
575 Is 568.7 24.90 400
375 25 369.8 42.48 152
475, 75. 365.6 46.65, 140)
? >
P
eee Pras
113,
6.14 Redlich/Kwong equation: © := 0.08664 := 0.42748
-_
Bs {e 2) (3.50)
‘Guess: z=l
Give ~ 4B 3.49)
Given 1+B-aB oy G49
2(B,q) = Find(z)
( Gs
fs hl4 k in{ 206i) +B: (6.62b)
\Zl6:.a)
1)-1.5-qi]] (6.64) The derivative in these
T3{(2(
SR; = R-(in(Z(B;,q))) —fi—0.5-qr) (6.65) equations equals -0.5
FRy SRi
2.302103) 76.218)
“poee108} mol [7.976]
SSit03
Ans.
116
6.15 Soave/Redlich/Kwong equation:
= 008664 = Ye oarTas c= (0.4804 15740-01760")
aeeaseeeae saa es
os\P Pr wa
(1 —Tr = — (3.50) — (3.51,
a=[ireG-n)P pe (2% G50) 4 (4) Bsn
Guess: z
Given 2=1+P-q8-—~—2, B49) 2(B,q) := Find(2)
2(2+B)
os
t
‘The derivative in the following equations equals: (2)
ai
i= of Zoe
2\Bi ai
HR; = RR: nal.) -1 [a (=y" + | wleso
a x|w2.a)-01-a() “w| (6.65)
CF
isl.l4 } (6.62b)
i
(0.37464 + 1.542260 ~ 0269020?)
6.16 Peng/Robinson equation: ¢ := 1+/2
2 = 0.07779 Y= 0.45724 c
pee eeAeeee
2
2
os’ Pr ea)
veLi+e(-Te ={2-—]| G50) q:=|——| 651)
ai-[i+el )| B ( *) 4 (2 <
Guess: 2
z-B iz
Gi Fl+p— 7———— (349) Z(B,q) := Find
ven 8-98 Caieren) (B.q) := Find(z)
(tr )\°>
‘The derivative in the following equations equals: sale
ai)
2(Bi,ai) + 0-Bi)
be tald tin Lo 2b) +0 (6.62b)
2Y2 \Z(Bi.ai) +e-Bi
HR =n 0.9 ot (6.64)
]
| (6.65)
tas 103 “mol 38.054) miolK
“3.861103 |:
2505-109]
2.EB5-103 a e
768 109
435105] | ae
Bia 103
“877103
208 109
ABSA}
AS57108} * 3
2103]
3.947.403 55:78}
116
Lee/Kesler Correlation — By linear interpolation in Tables E.
HR)? car)! HR
hO equals ~ hI equals = h equals
0 ety
0 equals a sl equals “ s equals &
686 093 950 1.003
590 ASS =1.709 A471
778 024 ~705
675 ons -1319
ms 008 993
744 165 =1.265
108 =019 962
699 ao | | -1200
710 ~.001 ~.770
742 007 =875
651 a4 1.466
167 =.034 = 73
716 032 ~701
746. «154 1.216
Z:=(Z0+o21) B54) h(h0+ohl) 676 UR:
117
71 =961
1.10 ~492
-497 ~.549
=.829 -A43
-631 ~.590
-110 ~296
-.674 ~.700 eee
50:5 (S0+os1) SR
~750 441
517 509
587 555
97 -429
~sil 589
~491 =.563,
=.688 ~287
216.107
- * [aaaas08
O78) SpE
‘01689 ‘A.87403)
aver) Be
0.752] 21.407;103|
[0.707 4
or 6.952:103)
nT) H1.G2- 105}
0748) 2.802103]
ose) Fas 708
0765} Az]
ores BRED
0.768 | 53786:103|
118
617
:23.15-K, ts
‘The pressure is the vapor pressure given by the Antoine equation:
(50) = 36.166
2788.51
+ 220.79,
PY) eo(s 8858 —
Spa = 1375 6.166KPa Pd = 1375-828
dt K
(a) The entropy change of vaporization is equal to the latent heat divided by
the temperature, For the Clapeyron equation, Eq. (6.69), we need the
volume change of vaporization. For this we estimate the liquid volume
by Eq. (3.63) and the vapor volume by the generalized virial correlat
For benzene:
Pe = 48.98-bar Zoi 0.271
P
Pris Pr = 0.007
P
Bo = -0.941 Bi = 139 — 2172 By = -1.621
a2
Tr
Voap = 7.306 x 10°
mol
02887 3
-n)
By Eq. (8.63), Viig = veal! ed Vig = 93.15
mol
Solve Eq. (6.69) for the latent heat and divide by T to get the entropy change
of vaporization:
AS := dPdt-(Vvap- Viig) Ans.
(b) Here for the entropy change of vaporizati
R-T
AS = 7 dPdt Ans.
119
6.20. The process may be assumed to occur adiabatically and at constant
Pressure. It is therefore isenthalpic, and may for calculational purposes he
considered to occur in two steps:
(1) Heating of the water from -6 degC to the final equilibrium temperature
of 0 degC,
(2) Freezing of a fraction x of the water at the equilibrium T.
Enthalpy changes for these two steps stim to zero:
Cp-At +-AHfsion = 0
The entropy change for the two steps is:
Ty = 273.15-K Ty = (273.15 - 6)-K
(T- x-AH, nm
cptn{ 22), XA Hfison
TT T2
The freezing process itself is irreversible, because it does not occur at the
equilibrium temperature of 0 degC.
6.21 Data, Table F. 1156,3- Bry Ho = 1533.4 BYU
Ibm Ibm
TT
$1 := 1.7320BMU_ g, BM.
lbmrrankine lbp
AH := H- Hy AS =
ate
+ te He Fei Ans.
For steam as an ideal gas, apply Eqs. (4.9) and (5.18). [t in degF]
ty = 227.96 t= 1000 Py := 20psi Pp = S0-psi
21 .
Eee eeer a 7 ty, 2382017 The ee 87 1, = 10.9028
(ketvins) (kelvins)
120
Mecpui(382.017,810.928,3.470, 1.450-10"3,0.0,0.121-108) = 4.373944
Mcps(382.017,810.928,3.470, 1.450-10"%,0.0,0.121-10°)
1339091
MCPH := 4.373944 MCPS := 4.339091 molwt := 18. b
Ibmol
R-MCPH: wnkii
AH = (2-4) rankine BTU ans,
molwt
( (T2 Po)
Be MePsnl ~ bl
oii ences 1 eeaEAE I
molwt
6.22 Data, Table F.2 at 8000 kPa:
Viig = 1.384 Fig = 1317.1 Stig = 3.2076
gm gm amK
om? J J
Veap = 23.525 Hyg = 2759. Syap i= 5.74712
gm gm-K
15-108 5
em
mig = ————— myap ‘= —
Mig
Hott = mig Hig + Map Hyap
Stotal = Mig: Stig + Mvap'Svap
124
6.23 Data, Table F.2 at 1000 kPa:
Viig
6.24 Data, Table F.3 at 350 degF:
3
Vig := 0.01799.
Ibn
Hig = 321.762
Mig + Map = 3-Ibm
3.1m
mig = ——"—
30-Vji
1+
Vvap
Map '= 3-lbm— Mig
Heal = Mig’ Hig + ™ap"Hyap
Hiotal
I
Sig 2.1382
3
f
Voap = 3.342-——
‘vep bp
BTU
Hyap = 1192.3-——
ae Ibm
S0-miig Vig _
Map: Wap = 50-mig:Viig Mig +? = 3g
ap
Imig = 2.36410
yap = 0.63616
=15191BTU Ans.
122
6.26
Data, Table F.1 at 230 degC:
90.3 Siig #= 2.6102.
em gm-K
(1 = ¥)Hig +x Fyap
xe 0582 Aas.
Viotal = miotal: Viig + Myap'AV|y
pe) Table F.l, em
Viotal °= 0.15 130 degC: Vvap >= 392.4- a
3
Table F.l, om
30 degC: AV iy = 32930-
Veotal Veotal ~ Motat Vi
Meal = 2 Mygp = 2 Mota Vita
wap AV
Moral = 0.382kg Myap = 4.543 x 10° ?kg
Mlig ?= Mrotal ~ Myep
Ans.
123
6.27 Table F.2, 1100 kPa:
Interpolate @101.325 kPa & 105 degC:
Const.-H throttling:
Hy 5 Huig + %+(Hyap - Htiq)
H - Hig
Fivap = Hig XS 01953 Ans.
6.28 Data, Table F.2 at 2100 kPa and 260 degC, by interpolation:
2235-2. 81 = 6.5640 motwt := 18.0152
Hy
gm emK mol
Hy == 2923.5 Final state is at this enthalpy and a pressure of 125,
em kPa.
By interpolation at these conditions, the final temperature is 224.80 degC and
7.93162 AS := 82-8} 58 3
mk = emk
For steam as an ideal gas, there would be no temperature change and the
entropy change would be given by:
Py = 2100-kPa
Pp = 125-KPa
6.29 Data, Table F.4 at 300(psia) and 500 degF:
Hy = 1257.7. BTU 3703. B TU
lb Ibpytankine
BIU ; oe
Hp := 1257.1. Final state is at this enthalpy and a pressure of
Thm 20(psia).
By interpolation at these conditions, the final temperature is 438.87 degF and
BIU BTU
8606-———_. = S)- 7
oe AS :=S2~S; AS = 0.29
Ses
Igy tankine
124
6.30
631
For steam as an ideal gas, there would be no temperature change and the
entropy change would be given by:
Ib
Py = 300- molwt := 18-
a Ibmol
P
-R o( 2)
oe 1) Ans.
molwt
Data, Table F.2 at 500 kPa and 300 degC
Sy := 7.4614- 4 Phe final state is at this entropy and a pressure of
gm 50 kPa. This is a state of wet steam, for whi
Stig = 0912-4 Svap = 1.5947.
mK mK
i
Hig *= 340.564—
gm
S2= $1 = Stig +%-(Svap ~ Stig) x= 058
Ho = Hig + X-(Hyap ~ Mig) ‘Ans,
Vapor pressures of water from Table F.1:
At 25 degC: Pat 2= 3.166:kPa
P= 101.33KPa — Xwater !=
At 50 degC: Psat
Psat :
vater = Riyater 05122" Ans.
125
6.32 Process occurs at constant total volume:
Vaotal “= (0.014 + 0.021)-m?
mass ‘= Mig + Myap
x= 4158x104 (initial quality)
_ Veotal ee cm? This state is first reached as
2 Tass i" gm Saturated liquid at 349.83 degC
For this state, P = 16,500.1 kPa, and
I J
U; 1641.7-— Uy = Ulig + X(U yap — Unie Uy = 419.868 —
a 1 Ulig+%(Uvap ~ Utig) =
Qs U2-Ur Ans.
3
6.33 Veotat := 0.25mi
Data, Table F.2, sat. vapor at 1500 kPa:
3
Vj = 131.66 U) = 2592.4
gm
Of this total mass, 25% condenses making the quality 0.75 x = 0.75
Since the total volume and mass don't change,
we have for the final state:
Vo = Vi = Viig +x-(Vvap— Vig) ‘Whence:
Vi~ Vtig : 5 :
x= +4 (A)__Find P for whieh (A) yields the value x = 0.75
Vvap- Vig for wet steam
126
6.34
Since the liquid volume is much smaller than the vapor volume, we make a
preliminary caleulation to estimate:
af ag
iz Voap = 175.547
Yvan.
‘This value occurs at a pressure a bit above 1100 kPa. Evaluate x at
1100 and 1150 kPa by (A). Interpolate on x to find P = 1114.5 kPa and
J
Utig += 782.41 ale Uyap *=.2584.9-—
gm am
ii
= Uti yap ~ UI Uz = 21343—
Up = Utig + (Uvap Urig) 2 a
Q = mass-(U2- Ui) QE=869.9kI Ans.
3
Table F.2,101,325 kPa: Vyap = 1673.0
‘am
Urq = 418.959-— Uyap #= 25065:
ig 418.959 nap = 25085
198m?
yap = otal = Mig + Map
Vvap
Vi = Vig + (Wvap~ Vtig) Vi = 98.326
Ur = Ulig+ ¥-(Uvap~ Uti), = 540.421
ee
Since the total volume and the total mass do not change during the
process, the initial and final specific volumes are the same. ‘The final state
is therefore the state for which the specific volume of saturated vapor is
98.326 cu em/gm. By interpolation in Table F.1, we find t= 213.0 degC and
127
6.35
6.36
Data, Table F.2 at 800 kPa und 350 degC:
em? 3 3
Vy = 354,34 Uy == 2878.9-— Voota #= 0-4!
‘em. =m
The final state at 200 degC has the same specific volume as the initial
state, and this occurs for superheated steam at a pressure between 575
and 600 kPa. By interpolation, we find P = $96.4 kPa and
J Veotal
Ug '= 2638.7-— U2 -U Ans.
a aoa 2 2 (u-u)
Data, Table F.2 at 800 kPa and 200 degC:
Uy = 2629.9 51 = 68148——
gm gmk
(a) Isothermal expansion to 150 kPa and 200 degC
Ups 2656.3 So = 7.6430
am emK
Q = mass-T-(S)~$)) Q=30220K Ans.
Also: Work := mass-(Uz—U})-Q
(b) Constant-entropy expansion to 150 kPa. The final state is wet steam:
729345
gmk
Svap
3
Uyap = 2513.4
= 0.929
Uhiq + % (ap ~ Utig)
Wis m(U2-Uy) W= 262527 ig Ans.
128
6.37 Data, Table F.2 at 2000 kPa:
6.38
x12 0.94 Hig 2= 908.589-——
gm
‘i ad
Hy = Hig + -(Hyvap- tig) Hi = 2.684 10° — mass := I-kg
am
For superheated vapor at 2000 kPa and 575 degC, by interpolati
Q := mass-(H2- Hy) ‘Ans,
First step: Qi2=0
Second step: W235 0
For process: Q=U3-Un
Table F.2, U, = 2601.8: z
Wap = 2601.82
2700 KP: gm
xp = 0.9
Table F.2, 400 kPa:
Svap = 6.2244)
gmk
Up = Utig + 1-(Uvap — Unig) Uy = 2.439% 109.
em
3 mi
81 := Siig + X1-(Svap- Stig) Sy = 5.861 x 10 a
Stig = 1.7764
‘K
Uyap = 2552.7.
am
3
Vvap 2= 462,22.
gm
129
Since step 1 is isentropic,
$1 ~ Stig
So = $1 = Stiq + ¥2-(Svap ~ Stiq) a x = 0.798
'vap ~ Stig
ee
Ua := Utiq + x2-(Uvap ~ Uliq Uz = 2.159 10° —
gm
f om?
Vo = Viiq + x2"(Vvap— Vig) Vp = 369.135—
gm
V2 __ and the final state is sat. vapor with this specific volume.
Interpolate to find that this V occurs at T = 509.23 degC and
U3 = 2560.7 Whence Q::= U3-Up Work := U2 Uy
Ans.
J
6.39 Table F.2, 400 kPa & Si = 7.0548-——
175 degC: gm:
Table F.1.sat. vapor, J
‘Sq = 6.6221-——
175 degC 2 mk
mass := 4-kg T r= (175 +. 273.15)K
Q = mass-T-(S2- $1) W = mass-(U2-Us)-Q
Q=-775.66k] Ams. = W=66766k] Ans.
6.40 (a) Table F.2, 3000 kPa and 450 degC:
Hy = 3344.6 si = 7.0854
gm gm-K
Table F.2, interpolate 235 kPa and 140 degC:
J
Hp = 2744.5.— Sp i= 7.2003
130
Ans.
Tp w= (140-4 273.15)-K
Ty = 413.15K
= 235-kPa
i 3 =
tcpal(723.15,413.15,3.470,1 450-1079, 0.0,0.121-10°) = -1343.638
ices(723, 15,413.15, 3.470, 1, 450-107? 0. 0,0.121 10°) 2.415901
ICPH := -1343.638-K ICPS := -2.415901
Eqs. (6.86) & (6.87) for an ideal gas: molwt = et
P))
R, ICPS - (|
As, Vu
baie molwt
Ans,
Ta = 1.11752 Pry = 0.13602 Tr = 0.63846 Pz = 0.01066
‘The generalized virial-coefficient correlation is suitable here
HIRB(1.11752,0.13602,0.345) = -0.13341 HRB, := ~0.13341
SRB(1.11752,0.13602,0.345) = -0.08779 SRB; := -0.08779
HRB (0.63846 , 0.01066, 0.345) = -0.04422 HRB2 == ~0.04422
‘SRB(0.63846 ,0.01066,,0.345) = -0.05048 SRB2 := ~0.05048
131
641 Data, Table F.2 superheated steam at 550 kPa and 200 degC:
7.0108
2640.62
gmK
gm.
Up i= 2963. pb Soe 757922 Qi2 = U2=Uj)
gm gmK
Qn = 32252
Step 2-3: Isentropic expansion to initial T. gm
: I
Qo3 = 0 835 7.5782———
em-K
‘Step 3--1: Constant-T compression to initial P.
T= 4BASK Q3) = T(S- $3)
For the cycle, the internal energy change ~0.
—Weycle
n
Qi
Ans.
642 Table F.4, sat.vapor, 300(psi):
Ty = (417.35 + 459.67)-tankine Hy i295
mn
Ty = 877.02rankine
1= 1.51052
ibprrankine
432
Superheated steam at 300(psi) & 900 degk
BIU BTU.
= Sates 1,7591-
Hy = 1473.6- Tm 2 Bar ine
BTU
Qi2:= Ho-Hi Q31 = Ti(81~- $3) Qs = -218.0075—
For the cyele, the internal energy change =
Woyele = —Qeycle = ~Q12~ Q31
Q31
neste
Qi2
6.43 Data, Table F.2, superheated steam at 4000 kPa and 400 degC:
81: 67733-— For both parts of the problem:
gm
(a) So we are looking for the pressure at which saturated vapor has the given
entropy. This occurs at a pressure just below 575 kPa. By interpolation,
572. Ans,
ees
(b) For the wet vapor the entropy is given by
x= 095 S2= Stig + X-(Syap~ Stig)
So we must find the presure for which this equation is satisfied. This
occurs at a pressure just above 250 kPa. At 250 kPa:
J J
son Svap = 7.0520:
Stiq
S2= Stig + x-(Svap ~ Stig)
So = 6.77982 Slightly > 6.7733
gm-K
By interpolation Boy
133
6.44 (a) Table F.2 at the final conditions of saturated vapor at 50 kPa:
ky
Sp = 7.5947 — 1 = 2646.0 S122
kek ke
Find the temperature of superheated vapor at 2000 kPa with this
entropy. It occurs between 550 and 600 degC. By interpolation
Ko
4
59.16 (degC) Hy = 3598.0-—
kg
Superheat: At = (359,16 ~ 212.37)-K.
kg
(b) mdot =
Wot := |mdot-(H2-H1)|
6.45 Table F.2 for superheated vapor at the initial conditions, 1350 kPa and 375
degC, and for the final condition of sat. vapor at 10 kPa:
kJ kK kd
205.4 $1 = 7.2410-— Hy := 2584.8-—
kg kgK kg
If the turbine were to operate isentropically, the final entropy would be
S2= 8)
Hy
Table F.2 for sat. liquid and vapor at 10 kPa:
ky
Shiq :=:0.6493-—_
kgK
Mig := 191.832 = Hyap '= 2584.8
S2- Stig
x Ban Sue x2 = 0.879 H = Hig + X2-(Hyap — Hig)
H=2294x 10°
kg
ay = 0.681 Ans.
134
6.46 Table F.2 for superheated vapor at the initial conditions, 1300 kPa and 400
degC, and for the final condition of 40 kPa and 100 degt
aas0.7-0 s) = 73404 Hy = 2683.82
kg kg-K kg
If the turbine were to operate isentropically, the final entropy would be
H
Hy
Table F.2 for sat. liquid and vapor at 40 kPa:
kD KD
Stig = 1.0261-—— Syap = 7.6709-——
‘a kek pT OO
Ky Ky
Hig = 317.16-— Hyap = 2636.9-—
kg kg
S2- Stig
Ses Sha x2 = 0.95 Hyiq + x2-(Hyap - Htig)
Ws 2.522% 198
32.85- H = 2856.3
gm. gm
Table F.2 (ideal-gas values, 1 kPa and 225 degC)
J
10.0681-—— Py = 1-kPa.
em!
T = (225 +.273.15)-K 498.15K vei-v-_1
molwt P
‘The enthalpy of an ideal gas is independent of pressure, but the
entropy DOES depend on P:
Hp = H-Hig
135
6.48
@ = 0345 Pe
T, + = 0.76982 Pret P, = 0.072546
Te Po
‘The generalized virial-coefficient correlation is suitable here
. 0.172
By = 0.083- 2822 ay =-0.558 By = 0.139- By = -0377
16 a2
T T
By Eqs. (3.58) & (3.59) along with Eq. (6.40)
Pr RT
Z = 1+(Bp+@-By)-— Z=0,935 Veo Z-1
(Bo+ oi} z R* Bmowwt
HRB (0.76982,0.072546 ,0.345
~0.178580 HRB := -0.178580
SRB(0.76982,0.072546,0.345) = -0.167101 SRB := ~0.167101
T= (179.884273.15)K oT
(Table F.2) molwt := 18.015 2%
mol
om? em?
Vy 1.127 194,29. AViy =v
gm
J
Hy := 762.605-— AHy = Hy - Hy
am
Aly = Sy~ S)
136
3
AVyy = 193.1639 AH = 2.014107. ag, = 4.445
em em gmK
(@) Gy: Hi TS) 6 =-206.062. Gy = Hy TSy Gy = 206.01
gm gm
J i
(b) AS, = 4.445 —— 4.445 ——
amK em-K
© Vrs Ans.
For enthalpy and entropy, assume that steam at 179.88 degC and 1 kPa
is an ideal gas. By interpolation in Table F.2 at 1 kPa:
Hig = 2841. p= 9.8834
em emK
kPa
The enthalpy of an ideal gas is independent of pressure; the entropy
DOES depend on P:
(@) Assume In P vs. 1/T linear and fit three data pts @ 975, 1000, & 1050 kPa.
975 178.79
Data: pp := | 1000 |-kPa 179.88.) (degC) i= 1.3
1050 182,02
1 iy n( 2) Slope = slope(x,y) Slope = -4717
4273.15 (kPa)
ae ‘Slope-K dPdT 22.9848
pdt
437
AVjy-dPdT.
Reduced conditions: « := 0.345
0.7001
The generalized virial-coefficient correlation is suitable here
o4 0172
Bo = 0.083- 2972 y= -0664 By = 0130-842 By = 063
16 42
Tr T,
By Kgs. (3.58) & (3.59) along with Eq. (6.40)
Pr
R-T
Z:=1+(Bo+o-B1) Z=098 VR = ———(Z-1)
es P-molwt
HRB(O.7001 0.0453 0.345) = ~0.1444
‘SRB(0.7001 ,0.0453,0.345) = -0.1493
RT.
HR =
molwt
6.49 Ts (358.43 + 459.67)-rankine T = 818.Jrankine
(Table F.4)
BTU
Ibprrankine
S1:= 0.5141
ae BTU
AVy = 2.996-— AHy = 863.45——
Ibn Ibm
@) G:=H|-T-S) Gy = Hy-TSy
Bru
Gy = -89.94
BTU SH
(b)aSjy = 1.055——__
Ibprrankine T
RI
© Va W- iz
. molwt P
bia
For enthalpy and entropy, assume that steam at 358.43 degF and 1 psi
is an ideal gas. By interpolation in Table F.4 at 1 psi:
1222.6 Sig 21497 pgs
Ibn Ibyetankine
Hi psi
‘The enthalpy of an ideal gas is independent of pressure; the entropy DOES
depend on P:
Hp = Hy ~Hig Ans.
BIU
Ibrrankine
(d) Assume In P vs. 1/T linear and fit threedata points (@ 145, 150, & 155
psia)
14s 355.77
Data: pp:=/ 150 |-psi_ t= | 358.43) degF) 1.3
155, 361.02
139
Slope ‘= slope(x.y)
Slope = -8.501 x 10°
aPaT == —-Stope-rankine dPdT = 1.905
2 rankine
este on eee og
Spy = AV)-dPdT Siy = 1.056: i Ans.
ASiy = AViy AS = (000
Pos 220.55-bar
T
T, = 0.7024 P, = 0.0469
Tr
‘The generalized virial-coefficient correlation is suitable here
0.422 0.172
Bo = 0.083-——— By = -0.65 By = 0.139-——= By = -0.62
1,6 782
By Eqs. (3.58) & (3.59) along with Eq. (6.40)
Pr RT
Z=1+(Bp+oB)— 72-0982 Z-
Boros R= Bimota 7D
FIRB (0.7024, 0.0469,0.345) = -0.1482 HRB := ~0.1482
SRB(0.7024 0.0469 0.345) = -0,1526 SRB == -0.1526
140
6.50
For propane: Te
69.8-K Po i= 42.48-bar 0.152
T= (195 +273.15)-K T= 468.15K P= 135-bar
T Pp
= = 3.178
To Pe
Use the Lee/Kesler correlation; by interpolation,
639
0.6141 Zy:= 01636 Z = Zt oLy
Ans.
Hay = ~0586-R-Ty
Higo = ~7.674« 10° Hy = -1.802% 10? 2
mol ‘mol
Spo = -1463-R Sai = -O.717-R
l
Spo = -12.163. Spt = 5.961
aa molK ey molK
Hp = Hgo + o-Hpy Sa = Sro-+o-Se1
3 J
Hp = -7.948 10° Sp = -13.069
ee mol . molK
07 3,-8,824-10" 0.0) = 1766.023
1cPH{308.15, 468.15, 1.213,28.785
1ops(308.15,468.15, 1213,28.785-10"°, -8.824-107 6,0.0) = 4.56487
ICPH := 1766.023-K ICPS = 4.56487
AH := RCPH + HR Ans.
As Ans.
{ices - «(F)) +8R
141
6.51
6.52
369.8K Pos 42.48-bar ew = 0.152
For propane: Te
T=343.1SK Pg := 101.33-kPa P
500-kPa,
T= (10+273.15)-K
Tet T, = 0.92793
Te
P, = 0.35311
‘Assume propane an ideal gas at the initial conditions. Use generalized virial
correlation at final conditions.
HRB(0.92793 0.35311 0.152) = 0.465534 HRB := ~0.465534
SRB(0.92793 ,0.35311,0.152) = —0.346693 SRB 0.346693
AH := R-Te HRB Ans,
{se uf - ) ge
4S = R+| SRB— In| fe Ans.
(Po) AS 5 OT oR a
For propane: 0.152
Tei 36.8K 42.48-bar 200.0.
mol
If the final state is a two-phase mixture, it must exist at its saturation
temperature at 1 bar. This temperature is found from the vapor pressure
equation
P= |-bar A := 6.72219 B = 1.33236 2.13868
Dis 1.38551 x(T) r Guess: T= 200-K
Given 7
=P, onl 220 +B(n)!S +c) +D-4cD)*]
E 1-1) |
Find(T) T = 230.703K
The latent heat of vaporization at the final conditions will be needed for an
energy balance. It is found by the Clapeyron equation. We proceed
exactly as in Pb. 6.17.
142
onl +(1) +B-(1(1D)'* + €-(2¢)§ +d. (cn)*]
>)
1-1(T)
T = 230.703-K 4 pry = 44g hPa dPdT 4.428124. 428
aT K K
P
Pree Py = 0.024 Ty Tr = 0.624
0.422
Bo := 0.083 — Bo =-0.815 By = 0.139
16
Tr
RT Pr
Vvap a) +(Bo+o-Bi) . Vig
om 3
Viap = 1.847% 1045 Viig = 75.5452
mol
Abliy := T-(Vvap ~ Viig)-4PaT Ay = 1.879 we
imo
ENERGY BALANCE: For the throttling process there is no enthalpy
change. The calculational path from the initial state to the final is made
Up of the following steps:
(1) Transform the initial gas into an i
(2) Carry out the temperature and pressure changes to the final T & Pin
the ideal-gas state.
(3) Transform the ideal gas into a real gas at the final T & P.
(4) Partially condense the gas at the final T & P.
‘The sum of the enthalpy changes for these steps is set equal to zero, and
the resulting equation is solved for the fraction of the stream that is liquid.
For Step (1), use the generalized correlation of Tables E.7 & E.8, and let
0 1
-(2) aa pace
OF RT. 1 Rh
143
Pe P, = 4.708
Pe
1 = -3.773 1 = 3.568
J : at
By Eq. (6.76) AH; Te (ro + 11-0) AH, = 1.327x 10° —
mol
For Step (2) the enthalpy change is given by Eq. (6.86), for which
3
icpHi(370, 230.703, 1.213,28.785-10 3, -8.824-10" 0.0) = -1260.405-K
AH := R-(-1260.405-K) AH) = - 1.048 « wie
mol
For Step (3) the enthalpy change is given by Eq. (6.78), for which
703
tye BUR ona
HIRB (0.6239, 0.0235 ,0.152) =
P, = 0.0235
0.07555, 0.07555,
AHg := R-T- HRB For Step (4), AHy = -x-AHyy
J
‘AH = -232.28——
mol
For the process, AH) + AHg + AH ~ x-AHy =
‘AH, + AH + AH3
AHyy
136° Ans.
653 For 1,3-butadiene: @ := 0.190 425.2-K
3
Po: 42.77-bar 267 20.4 Ty = 268.7-K
mol
T= 380K 1919.4-kPa 27B1S-K Po = 101.33-kPa
= 0894 P P, = 0.449
Pe
144
Use Lee/Kesler correlation, HOWEVER, the values for a saturated vapor
lie on the very edge of the vapor region, and some adjacent numbers are for
the liquid phase. These must NOT be used for interpolation. Rather,
EXTRAPOLATIONS must be made from the vapor side. There may be
some choice in how this is done, but the following values are as good as any:
Zo: Z=0718
7442 Ze
ZRT
P
Vvap
Hpo i= -0.689-R-Te
Heo = ~2.436% 10°
mol
Spo = -0.540R
J i
Spo =—4.49 Spi = 7.383
RO mokK RI molK
Hro+@HrRt Sr == Spo + @-SRi
J
Sp = -3.892
x molK
1cPH(273.15,380,2.734,26 786-10" >, -8.882-10" 60.0) = 1124.694
0) 3.45478
ICPH := 1124.694.K ICPS := 3.45478
icps(273.15,380,2.734,26.786-10">,-8,882.10" 6,
Hyap = RICPH + HR
Svap := R- [reps - o(F)) +SR
Po})
145,
For saturated vapor, by Eqs. (3.63) & (4.12)
{a-1)87] 3
cm
jeZet 7 om Ans.
Viiq = VeZe Mia 5 10088 rol
AG (®)-195) 7) ’
enna OM Se AH, = 22449—
AH, = R-Ta! To \ mol
930 -
Tea
r 0032
By Eq. (4.13) = AHy| — AH = 14003
Hiig = Hyvap ~ A Aus.
cas Ans.
Stiq = Svap ~ st
ig = Svan
6.54 For n-butane:
37.96bar Ze = 0.274 Ty t= 272.7-K
70K 1435-kPa Po = 101.33kPa
Tr, T. = 087 0.378
Te
Use Lee/Kesler correlation. HOWEVER, the values for a saturated vapor
on the very edge of the vapor region, and some adjacent numbers are for
the liquid phase. These must NOT be used for interpolation. Rather,
EXTRAPOLATIONS must be made from the vapor side, There may be
some choice in how this is done, but the following values are as good as any
146
J
2.733% 10° —
Hr =
mol
1epH(273.15,370, 1.935,36.915-107*
reps(273.15,370,1.935,36.915-10"
ICPH := 1222.048-K
§ -11.402-10
Z= 0742
ZLakoroLy
Hpy := -0.83-R-Te
Her = -2.937 10°
mol
Spi = -0.835-R
Sri = -6.942
dau mol K
SR = Spo +@-SRi
J
Sp = -5.421
morK
11,402.107,0.0) = 122.088
$0.0) = 3.80735
3.80735
ICPS
»)
(P
Svap := R-| ICPS — In| ae ) +Sz
(Po,
[(r-90*7
Viiq= VeZe
( (P. \
1.092,{ in ©] — 1.013
AH, = R-To peieseh SERS etna
.
0.930-—*
Te
147
AH, = 22514
|
aot | = mol
038
ast = 152952.
mol
By Eq. (4.13) AH
Hig = Hyap ~ AH Ans,
AH
Sig == Svan * Ans.
6.55 (a) The final-state volume of acetylene is
(750~33:5)-em? Vegas = 58Sem> T= 398.15-K
= 0.187 3083K Pe = 61.39-bar
T= 1291 Guess: Zi 0.65
P
P = 183.9bar Py = BR Py = 2.996
Pressure is clearly high, and requires use of Lee/Kesler correlation.
Solution is by trial, because P is unknown but is required to find Z.
Start with reduced pressure from guess value above. The eventual result
for a reduced pressure of 3.07 is:
Zo := 0.6298 Zy = 0.1948 Zo traZy, Z = 0,666
Z0R-T oe a
p= ot Peis P, = 3.07 188.5bar Ans.
Ves Pe
() By the first law, Q = n-AU = n-(AH — A(PV))
Q= n(4H-V-AP) = n-AH ~ Veag-AP
The enthalpy change is evaluated by a three-step process:
(1) Reaction at 298.15 K
(2) Change in T for products in std. states
@) Transformation to state of real gas
From these,
148
AH = AH9g + AHp + H®
Step (1): From data of Table C.4
AH p9g ‘= [ -986090 + 227480 - (~59800) ~ 2-(~285830) } oat
mol
J
127180 —
mol
Step (2), Table C.1 data, for acetylene(g):
McPH(298.15,398.15,6.132, 1.952-1073,0.0,-1.299.105) = 5.71731
Table C.2, for caleium hydroxide(s):
McPHt(298,15,398.15,9.597, 5.43510
AH} 98 =
0.0,0.0) = 11.48920
For the products,
MCPH := R-(5.71731 + 1148920) MCPH = 143.055.
motK
AHp := MCPH-100-K AHp = 143055
mol
Step (3), from Tables E.7 & E.8 at the reduced conditions of Part (a) for
acetylene:
Hpo = -2.340-R-Te Ai = -0.384-R-Te
Fro = -3.998% 10° 2. Bar = -984.271
mol mol
Hp = Hro + @-HR1
AH := AHagg + AHp + Hg
Q = MAH ~ Vgas(P ~ I-bar)
6.56 Propylene: @ := 0.140
400.15-K P= 38:bar Po: I-bar
The throttling process, occurring at constant enthalpy, may be split into
two steps:
(@) Transform into an ideal gas at the initial conditions, evaluating property
changes from a generalized correlation.
(2) Change T and P in the ideal-gas state to the
final conditions, evaluating property changes by equations for an ideal gas.
Property changes for the two steps sum to the property change for the
process. For the initial conditions:
T P
as Ty = 1.095 Pes Py = 0815
Te. : Pe
Tre
Hp := Ho + @-Hy
Hy = -1.623« 85x10
mol
S] = -0.496-R Sr = So+o-S}
J
0 = 4.697 $y = 4.124!
mol-K molK
Step (2): For the heat capacity of propylene,
22.706-10"
K
Solve energy balance for final T. See Fq, (4.7).
Acs 1637 B
t=1 (guess) Given
H=R (fa vee 2ar(e— )] + £ P(e. |
t= Find(z)
.908 Tit T286327K Ans.
Icps' (400 15,363.27, 1.637,22.706-10" ~ 6.91510 0.0) 0.898338
Ices ).898338
Po
ASig = n{tces ae (3) ASjg = 22.774: 2
\P ig mol K
150
6.57
The throttling process, occurring at constant enthalpy, may be split into
two steps:
(1) Transform into an ideal gas at the initial conditions, evaluating
property changes from a generalized correlation.
(2) Change T and P in the ideal-gas state to the
final conditions, evaluating property changes by equations for an ideal gas.
Property changes for the two steps sum to the property change for the
process. For the initial conditions:
a Ty = 1.144 Pee Pr = 0.518
Te Po
Step (1): Use the generalized virial correlation
Tr
HRB(1.14386, 0.51789, 0.152) = -0.444283
SRB(1.14386,0.51789,0.152)
Hg := R-TeHRB Hp = -1.366% 10°
mol
Sr := R-SRB Sp = ~2.284
zi . molK
Step (2): For the heat capacity of propane,
-8.824-107%
2
28,785-10_
K
Solve energy balance for final T. See Eq, (4.7).
Avs L213 B
K
t= 1 (Guess) Given
Hp = wffa Tle +37
v= Find(t) +
6.58
6.59
(
ICPS = 0.394958 ASig = R {ices »()) ASig =
1cps(423, 408.91 1.213,28.785-10"°,-8.824-10" °,0.0) =
AS == -Sa + ASig
For propane: T
T= (100 +273.15)-K
373.1SK Po = I-bar P= 10bar
Ty = 1.009 Pa P, = 0.235
Pe
Assume ideal gas at initial conditions. Use virial correlation at final conditions
HRB(1.00906, 0.23540, 0.152) = ~0,260821 HRB := ~0,260821
SRB(1.00906 ,0.23540,0.152) = -0.179862 SRB := ~0.179862
AH i= R-Te HRB
ssa(oe-0(F)}
Pri = 0.056 Ty = 1.606
152
Use generalized virial-coefficient correlation for both sets of conditions.
HRB(1,07095, 0.05578 ,0.094) = -0.052991 HRBj := -0.052991
SRB(1.07095 , 0.05578, 0.094) = -0.034156 SRB := 0.034156
HRB (1.60643, 0.27892, 0.094) = 0.119727 HRB := -0.119727
'SRB(1.60643 , 0.27892, 0.094) = -0.056506 SRBp := ~0.056506
1cPH{400 600, 3.931 ,1.490-107°,0.0,-0.232-10°) = 915.867
1cPs(400,600,3.931, 1.490-10"$,0.0,-0.232-108) = 1.85161
ICPH := 915.867-K ICPS := 1.85161
Eqs. (6.82) & (6.83) are written
AH := R-ICPH+ R-Te(HRB2- HRB})
( P2)
AS = tors -ble|]+ R-(SRB> - SRB})
uv
6.60 Carbon dioxide: @ t= 0.224 To B042K — Po := 73.83-bar
Tis 31815K P= 1600-kPa_—- Py := 101.33-kPa
Throttling process, constant enthalpy, may be split into two steps:
(1) Transform to ideal gas at initial conditions, generalized correlation
for property changes.
(2) Change T and P of ideal gas to final T & P.
Property changes by equations for an ideal gas.
Assume ideal gas af final T & P. Sum property changes for the
process. For the initial T & P:
T P
T= Tr = 1.046 Pp = — Py = 0.217
Te ue
Step (1): Use the generalized virial correlation
HRB(1.04586 , 0.216714 ,0.224) = —0.232491 HRB := ~0.232491
SRB(1.04586,0.216714 0.224) = -0.157944 0.157944
183
I
Hp = R-TeHRB Hg = -587.997— Sp :=RSRB Sp = -1.313
e mol molK
Step Q): For the heat capacity of carbon dioxide,
pensast we MEI pe suasnadta?
Solve energy balance for final T. See Eq. (4.7).
ti= 1 (guess) Given
E 2(2_,),D(t!
Hp eR} AT(t- += -1)+2.
7 [ (Bork 2(=)]
c= Find(t) 5 = 0.951 TstT T= 30271K Ans.
icps(318.15,302.71 5.487, 1.045-10" 3, 0.0,-1.157-105) = -0.227818
Re
ICPS := -0.227818 AS, = (10°59 21) AaSig = 21.087
f P | mol-K
AS = -SR+ASig AS 52
Ans.
661 To = 523.15K Po = 3800-kPa P= 120-kPa
J :
For the heat capacity of ethylene:
molK
14.394-10- > 4,392.10"
A= 1424 Be co
K e
(2) For the entropy change of an ideal gas, combine Eqs. (5.14) & (5.15)
with D=0:
104 (guess) Given
nds) t= 0.589
senfainG)+[ arson? (3 }e-a- oz |
: 08,
9K Ans
icp1i(s23.15, 308.187, 1.424, 14.394-10°3, 4.30210
IcPH
RICPH AHig
(b) Ethylene: o
To Po
To = To = 185317 Py = Pyo = 0.75397
© :
Tree = 1.0917 Py = 0.02381
Use virial-coefficient correlation,
‘SRB(1.85317 ,0.75397,0.087) = ~0.10426 SRBo = -0.10426
SRB(1.09170 0.02381 ,0.087) = -0.01374
The entropy change is now given by Eq. (6.83):
5 (guess) Given
ASS RIA in(d)+[Betore To (: = ) lt = v-u(F) ist
0.
| +SRB— SRBp
tau(SRB) := Find(s) T = tau(SRB)-To T = 303.08K
At final P and thi
r, reevaluate SRB, then T:
Ty = 1.0736
‘SRB(1.07360 0.02381 ,0.087) = -0.01440 SRB := -0.01440
T = tau(SRB)Tp PSBORK Ans.
The work is given by Eq. (6.82):
155
3
1cpH(523.15,303.11, 1.424, 14.394-1073,-4.392:107 60.0) = -1452.982
1.208 10°
ICPH := -1452.982-K AHig = RICPH — AHig =
HRB(1.85317, 0.75397, 0.087) = -0.24094 HIRBo := ~0.24094
HRB(1.07360, 0.02381 ,0.087) = -0.02242 HRB == ~0.02242
Hig + R-Te- (HRB - HRBo) \
Ba
Po = 30-bar P:=2.6bar
For the heat capacity of ethane:
3 —
19,225-1 =
0 Ce SIO
K Pe
(a) For the entropy change of an ideal gas, combine Eqs. (5.14) & (5.15)
with D=0:
B
t= 0.4 — (guess) Given
/
AS=R [s n(e+] Ty +C. w()} (¢-1)~ if 3]
Posi
t= Find(x) t=005 Ti=tTy 367.59K Ans.
cpHi(493.15,367.592, 1.131, 19.225.10 4-5.561-10"§,0,0) = -1050.616
ICPH := -1050.616-K RICPH Alig = -8.735x 103
mol
We = Aig
(b) Ethane: 48.72-bar
fe Po
Ty = 1.6153 Po 5 Po = 0.61576
6
156
At final conditions as calculated in (a)
TAT) = TCT) = 1.20404 Py = 0.05337
Use virial-coefficient correlation.
SRB(I.61530,0.61576,0.1) = ~0.12314 SRBo ‘= -0.12314
SRB(1,20404 ,0.05337,0.1) = -0.02370 SRB
The entropy change is now given by Eq. (6.83):
.02370
T= 0.5 (guess) Given
AS= RIA Ce a[ Tp+C re(24} fe -1)- »(2) a
2 Po
+ SRB —SRBg
tau(SRB) =
‘ind(z) T s= tau(SRB)-To = 362.69K
At final P and this T, reevaluate SRB, then T:
T,(T) = 1.18798
‘SRB(1.18798,0,05337,0.1) = -0.02459 SRB
T = tau(SRB)-To
T,(T) = 1.18813
The work is given by Eq. (6.82):
HRB(1.61530,0.61576,0.1) = -0.26131 HRBo
HRB(1.18813,0.05337,0.1) = -0.04158 HRB = -0.04158
10PH(493.15, 362.74, 1.131, 19.25.10" 3, -5.561-107 80.0) = -1086.568
9.034 10°
‘mol
ICPH := ~1086.568-K RICPH AH;
ig
157
6.63 n-Butane: — = 0.200 Te 425K Pe = 37.96-bar
Ty=3B1SK — Pos
A-bar P
J
moFK
For the heat eapacity of n-butane:
36.915-10
A= 1.935
HRB (0.76017 , 0.02634 0.2) = -0.05679
‘SRB(0.76017 0.02634, 0.2) = -0,05210 SRBo
The entropy change is given by Eq. (6.83) combined with Eq. (5.15) with D = 0:
r= 04 (guess) SRB := 0.0 (starting value)
Given aS = RIA n(x) -[» Ty +C. na (* . ‘)| (c-1)- “() ist
be SRB - SRBg
tau(SRB) := Find(z) T <= tau(SRB)-To, T = 375.17K
At the final P and this T, evaluate SRB:
T
t= T, = 0.88254 Pe = 0.20548
Te
Use virial-coefficient correlation.
‘SRB (0.88254 , 0.20548, 0.2) = -0.24876
Ts
w(SRB)-To T= 381.77K.
Although it will make little difference, we recaleulate SRB at this temperture:
T
eT
SRB(0,89806 ,0.20548,0.2) =
Tr T, = 0.89806
0.23533 SRB := ~0.23533
188
Ts tau(SRB)-Tp Ans.
Tr : T, = 0.89722
Te
HRB(0.89722 0.20548 , 0.2) = —0.30330 HRB := -0.30330
‘The work is given by Eq. (6.82):
repH(323.15,381.41, 1.935,36.915-1073, -11.402-10" 60.0)
787.744
ICPH := 787.744-K AHjg = RICPH Mig = 6.549 103
Ee be mol
We := Aig + R-Te(HRB ~ HRBo) Ans,
6.64 — The maximum work results when the 1 kg of steam is reduced in a
completely reversible process to the conditions of the surroundings,
where it is liquid at 300 K (26.85 degC). This is the ideal work.
From Table F.2 for the initial state of superheated steam:
Hy = 3344.62 81 = 7.0854
kg kg
From Table F.1, the state of sat. liquid at 300 K is essentially correct:
Hy = 112.5- 00-K
By Eq, (8.27),
Wideat = (H2 - Hi) - Te-(S2- $1) Ans.
6.65 Sat, liquid at 325 K (51.85 degC), Table F.1:
Hyig 22 217.02 Stig = 0.7274 Viig s= 1.013
ke kK gm
169
Pyat = 12.87-kPa For the compressed liquid at
325 K and 8000 kPa, apply
Py ‘= 8000-kPa Eqs. (6.28) and (6.29) with
325-K p= 46010 SK
KD
Hy = Hig + Viig:(1 ~ B-1)-(P1 ~ Psat) Hy = 223.8815
kJ
$1: Stig B-Viig-(P1 - Psat) 8) = 07m
For sat. vapor at 8000 kPa, from Table F.2:
Hp := 2759.98 Ss: san. Toi 300-K
ke kek
a 4 KI
Heat added in boiler: Q=H)-Hy Q= 2536
8
Maximum work from steam, by Eq. (5.27):
Wideat = (Hi —Ha) —To-(S1 Sa) Wiseat = -102982
"i
Work as a fraction of heat added:
Wi
el sa Frac 04058 Ans,
‘The heat not converted to work ends up in the surroundings.
+ Widea! 7
Sdotg gure = 2M 9, AB Sdotg sun = 50.2344
Te seo Ke
kg kW
Sdote system *= (81 — Sp)10-<2 SdotG system = ~50.234——
Jot aystem *= (Sy — Sp) 10-2 Ot syst :
Obviously the TOTAL rate of entropy generation is zero. This is because
the ideal work is for a completely reversible process.
160
6.66 Treat the furnace as a heat reservoir, for which
Qdot
= (6004+273.15-K T= 873.19K
Sdotg Ans,
By Eq. (5.34)
Te = 300-K — Wdotiost := TorSdotg Ans.
6.67 For sat. liquid water at 20 degC, Table F.1:
3.8682 S} == 0.2963: —
kg kg:
For sat. liquid water at 0 degC, Table F.1:
Ho = 0.04
kg
For ice at at 0 degC:
Hg =Ho-3334 y= 5-284
kg 273.15 keK
Hy = 333.44 sy =-121
kg kek
kg
Ty = 293.15-K mdot
ne = 0.32
By Eqs. (5.26) and (5.28):
Weotigeat = mdot] H2-Hi-To-(S2-$1)] — Weotideai = 13.686kW
Wdotideal “
‘Wdot = ——— Wdot'=42:77kW Ans.
mk
161
6.68 This is a variation on Example §.6., pp. 175-177, where all property values
6.69
are given. We approach it here from the point of view that if the process
is completely reversible then the ideal work is zero. We use the notation
of Example 5.6:
J kt Kw
26760 $75 73856 Hy = 00
Hy = 2676055 1 : n= OOF
WW
$2 1= 0.0 = 273.15-K
7 ig K
The system consists of two parts: the apparatus and the heat reservoir at
elevated temperature, and in the equation for ideal work, terms must be
included for each part.
Wideal AHapparatus reservoit ~ To ASapparatus eservoir
AHapparatus.reservoir = H2— Hi ~ Q’
Sanparats reservoir = S2~ St kg
50K (Guess)
KI i f
0-= = Ho- Hi ~ Q'- To} $2- $1 —
ke \
T= Find(T) 7 Ans.
(136.64 degC)
From Table F.4 at 200(psi):
BTU BTU
Hy = 1222.6-—— $8) = 1.5737,——_—_— it 420 degF)
: bn t Darrinkine "120 de8F)
BTU BTU (Sat. tig.
Hy 55.51 Hyap = 1198.3-——
hig bas p = 1198.3. be and vapor)
BIU BTU
Stig = 0.5438-—————_— Sap = 1.5454.
i Fprankine Sip = 15458 0.96
162
Hy = Htiq + x-(Hyap — Hiiq) $2 := Stig + %-(Svap— Siig)
3 BTU eters
Hp = 1.165 x 10 s eal
Tin Iby-rankine
Neglecting kinetic- and potential-energy changes,
on the basis of 1 pound mass of steam after mixing, Eq. (2.30) yields
for the exit stream:
H = 0.5-H; + 0.5-H2 H= 1193.65 (wet steam)
Ans.
BTU
Stig + x-(Svap ~ Stig) ——
tig + (Svap ~ Siig) Ibprrankine
By Eq. (5.22) on the basis of 1 pound mass of exit steam,
6.70
3
1.3496 ©
Ibm
Bru
118.0=—
bm
VOL tig = miig?Viig VOLtig = 79.796 8°
VOLyap := Viank ~ VOL tig, VOL yap = 0.2048°
VOL vay
Myap = a mMyap = 0.151 Ibm
Vyap
163
ig Uiig + Map Uv
eae U; = 406.726 BU
+ Myap Ibm
By Eq. (2.29) multiplied through by dt, we can write,
(Subscript t denotes the contents of the tank.
Hand m refer to the exit stream.)
d(m;-U,) +H-dm
Integration gives: my-Up—m-Uy +f Hdm=0
0
From Table F.3 we see that the enthalpy of saturated vapor changes
from 1203.9 to 1203.1(Btu/Ib) as the temperature drops from 430 to 420
degF. This change is so small that use of an average value for H of
1203.5(Btu/Ib) is fully justified. Then
my-U2—my-Us + Have'm = 0 Haye= iana.s STC
Pee ee ‘mp(mass) := my ~ mass
Property values below are for sat. lig. and vap. at 420 degF
3
Vvap = 1.4997.
BTU
Uyap == HIT ——
sp be
7 . =
Vo(mase) = Van Ema) seo cee
mp(mass) Vvap - Vig
Up(mass) := Ulig + x(mass)-(Uyap — Utiq)
mass = 50-Ibm (Guess)
Gaetan teem)
Haye ~ Uz(mass)
mass := Find(mass) 6 Ib Ans.
164
6.72
The steam remaining in the tank is assumed to have expanded isentropically.
Data from Table F.2 at 4500 kPa and 400 degC:
$1 = 6.7093-——
amk
ee J By interpolation in Table F.2
S2= Si = 6.7093 at this entropy and 3500 kPa:
3
Vo 1= 78.726.
gm
Vtank
my =
Vi
This problem is similar to Example 6.7, where it is shown that
Q= A(meH) ~ H-Am,
Here, the symbols with subscript t refer to the contents of the tank,
whereas H refers to the entering stream.
We illustrate here development of a simple expression for the first term on
the right. The1500 kg of liqui ly in the tank is unchanged during the
process. Similarly, the vapo in the tank that does NOT condense
is unchanged. The only two enthalpy changes within the tank result from:
1. Ad
ion of 1000 kg of liquid water. This contributes an enthalpy change of
tig Am,
2. Condensation of y kg of sat. vapor to sat. liq.
‘This contributes an enthalpy change of
(Hig — Hvap) = -y-SHyy
Thus A(mpH) = Hig: Am,—y-AHyy
165
A(meV,) = Viger = ¥-AViy
Whence Q= HhigrAm, = y-AHyy — H-Amy
‘Am, != 1000-kg Required data from Table F.1 are:
At 50 degC: n= 2093-8
kg
At 280 degC: Hig = 1085.8
kg
nay
kg
Vig’
hee 5.641 kg,
Vy
Q = Amp(Hiiq- H) —y-AHiy Qs Ans
6.73 Given: Veonk = 0.5m?
fa
CH04— Ty = 295K Mink *= 30-kg
kgK
Data for saturated nitrogen vapor:
80 1.396 0.1640
85 2.287 0.1017
90 3.600 0.06628
95 |-K 5.398. |-bar V =| 0.04487
100 1.718 0.03126
105 10.83 | 0.02223,
10, 14.67 0.01598
168
lB
8.9
82.3 At the point when liquid nitrogen starts to
85.0 accumulate in the tank, it is filled with saturated
ky Vapor nitrogen at the final temperature and having
H=| 86.8 eg properties
| 87.7,
Map, Tvaps Vvap>Hyvap»U vay
|e 3: Tvap» VvapHyap,Uvap
85.6
By Eq, (2.29) multiplied through by dt,d(ny Ut) — H-dm = dQ
Subscript t denotes the contents of the tank; H and m refer to the inlet
stream, Since the tank is initially evacuated, integration gives
MWvap Uvap ~ Hin-Myap = Q = mtank-C-(Tyap-T1) (a)
v,
Also, Map = @®)
Voap
Calculate internat-energy values for saturated vapor nitrogen at the given
values of T:
(56.006
—.
U-=(H-P-V) 59.041
61.139
U =| 62.579 z
63.395 :
63.325
(62.157)
Fit tabulated data with cubic spline:
Us := Ispline(T,U) Vs := Ispline(T,V)
Uyap(t) == interp(Us,T,U,t) Vyap(t) = interp(Vs,T, Vt)
Tyap:= 100K (guess)
167
6.74
Combining Eqs. (A) & (B) gives:
Given
MankC-(T1 ~ Tap): Vvap( Toa
Uvap Trg) ~Bin = spam To) Yo)
Tyap = Find(Tyap) Tyap = 97.924K
Viank
ae ap Tvap vane
TA82lkg Ans.
‘The result of Part (a) of Pb. 3.15 applies, with m replacing n:
my-(U2-H)-mj-(U)-H) = Q 50
Whence my-(H - Ua) = m-{H - Uy)
Also Un = Ujig.a + X2-AUyy.2
Hi : Veank
V2 = Viig.2 + X2°AViy.2 Ve
™
Eliminating x, from these equations gives
: ie
St Vig
m2
mo} H = Uliq2 = —a—— AU wa] = me(H = Ui)
we
which is later solved for my
my 3 16000-kg
Vy = 3.125% 107 3
kg
Data from Table F.1 @ 25 degC:
3 a:
Viig.t := 1.003- AVyy1 = 43400
gm gm
168
6.75
AU yy. = a0.
Ur = Utig #x1-AU jy.
x1 = 4.889% 1075 Us = 108.913
Data from Table F.2 @ 800 kPa:
3
Viig2 = 1.115- Uiiga
am
cm I
AViy.2 $= (240.26 ~ 1.115) 2 = (2575.3 ~ 720.043)-—
gm kg
mi 3 kl
AViy.2 = 0.239 — AUjy2 = 1.855 x 10°
ke ie kg
: 7 i
Data from Table F.2.@ 1500 kPa: H:» 2789.9-—
2
(AUiw2
my-(H = U)) + Veaak la
ee my = 2.086% 10°kg
H-Ulig2 + Viig2'| ws)
~ Ulig2 + Viig2
\A¥v2
steam = my ~ my Ans.
The result of Part (a) of Pb. 3.15 applies, with ny = Q
Whence U2 =H
From Table F.2 at 400 kPa and 240 degC
169
= 2043.94, _ Interpolation in Table ¥.2 will produce values of t
kg and V fora given P where U = 2943.9 ki/kg.
1 384.09 ) 303316
100 384.82, 3032.17
200 tz =| 385.57 V2 := | 1515.61
300 386.31 1010.08
400. 387.08 757.34
5 Veank = 1.752
6.76
x= 01
3
Vi = 7957x103 my = 257.832kg
kg
170
‘The process is the same as that of Example 6.7, except that the stream
flows out rather than in, The energy balance is the same, except for a sign:
Q= A(me Hi) + H- Amant
where subscript { denotes conditions in the tank, and H is the enthalpy
of the stream flowing out of the tank. The only changes affecting the
enthalpy of the contents of the tank are:
1. Evaporation of y kg of sat. lig.:
y-(Hvap— Hliq)
2Exitof 0.6mi-ke of
-0.6-my "Hig
‘Thus
(mH) = ¥ (Hyap ~ Hig) - 0.6m): Hhig
si
ilarly, since the volume of the tank is constant, we can write,
A(my Vi) = ¥(Vvap~ Viig) ~ 0-6-1 Viig = 0
0.6-my-Vij
Whenee y= LO Via
Wap ~ Vig
0.6-m1-Viig
-(Hivap — Htig) ~ 0.6-m)-Hiig + H-Amanic
Veap~ Vig
But H=Hiq and 0.6-m, = Aman
and therefore the last two terms of the energy equation cancel:
0.6-m1-Viig
= “(Hyap ~ Hic
Yop Vig tet)
171
6.77
Data from Table F.1 for sat. liq.
Thy = 1006-42 @4 degey Hy = 3559-2 (85 dege)
ke kg
Data from Table F.2 for sat. vapor @ 400 kPa:
Hy 273768
kg
By Eq. (2.30), neglecting kinetic and potential energies and setting
the heat and work terms equal to zero:
H3-mdots ~ Hy-mdoty ~ Ha-mdotz = 0
Also mdot, = mdot3 ~ mdot mdots :
yhenes mdots-(Hi - Hs)
Whence dot := (Hi - #3)
Hi —H2
Ans.
Table F.2, superheated vap., 3000 kPa, 375 degC:
kJ
Ho = 3175.64 Sys
ig
Table F.1, sat. liq. @ 50 degC:
kg
Hy Stig ?= 0.7035-—.
a a kek
172
Find changes in H and S caused by pressure increase from 12.34 to 3100
kPa. First estimate the volume expansivity from sat. lig, data at 45 and
55 degC:
3
AV = (1015-1010. a= 10K P= 3100-kPa
gm
3m? 4
avesx1g 3S B= 4941x104!
gm
Apply Eqs. (6.28) & (6.29) at constant T:
Hy := Hig + Viig-(1 ~ B-T)-(P ~ Psat) Hy = 2119268
8
ki
$1 Stiq- B-Viig:(P ~ Psat) 51 = 0702
By Eq. (2.30), neglecting kinet
and work terms equal to zero:
c und potential energies and setting the heat
Hg-mdot3 ~ Hy-mdoty ~ Hy-mdot = 0
Also mdotg = mdot3 — mdoty
mdot3-(H3 - Ho)
Whence —mdoty == maoty $8922 Ans.
Hy =H SLES see
kg
mdoty = mdot3 ~ méaty mdoty = 13.1152
see
For adiabatic conditions, Eq, (5.22) becomes
Sdotg := S3-mdots ~ Sy-mdoty ~ Sz-mdoty
I.
Sd 193 — Ans.
eT eK
The mixing of two streams at different temperatures is irreversible.
473
6.19 Table F.2, superheated vap. @ 700 kPa, 200 degC:
w i
Hy == 2844.2. $3 = 6.8859
3 ke 3 kek
Table F.2, superheated vap. @ 700 kPa, 280 degC:
ny = 30177-2 $1 = 7.2250 dot = 50-48
ke ek see
Table Ft, sat. ig. @ 40 deg
J
Siig = 0.5721-
ig ard
By Eq, (2.30), neglecting kinetic and potential energies and setting the heat
and work terms equal to zero:
Hp
Alig Hg-mdots ~ Hy-mdot) — H2-mdot = 0
Also mdot; = mdotz + mdot;
mdot)-(Hi - Hs)
Hy — Hp a
For adiabatic conditions, Eq. (5.22) becomes
mdoty ==
S2:= Siig dots := mdotz + mdoty
Sdotg
mdoty ~ Sz-mdotg
Ans.
‘The mixing of two streams at different temperatures is irreversible.
6.80 Basis: 1 mol air at 12 bar and 900K (1)
+28 mol air at 2 bar and 400K (2)
=3.5 mol air at T and P.
174
6.81
Ty == 900K
Ssmol
ms tmol ms
Ist law: T := 600-K (guess)
Given ny-Cp-(P- Ty) +n2-Cp-(T-T2) = 0-3
T = Find(T)
2nd law:
Given
Find(P)
i
molwt := 28.0142 cp = 0.248 BTU _
Tbmol Tbqyrankine
M, =steam rate in Ibm/see
Ibm
My = nitrogen rate in Ibm/see Mp = 40-—2
sec
(1) = sat. fig. water @ 212 degF entering
(2)= exit steam at 1 atm and 300 degF
@)= nitrogen in at 750 degF Ty = 1209.67-ankine
(4) = nitrogen out at 325 degF Ta = 784.67-rankine
Hy t907-BTS St (Table F:3)
6.82
Hg := uia¢ BE sy = 181582 (rabte 4)
1m mrankine
Eq. (2.30) applies with negligible kinetic and potential energies and with the
work term equal to zero and with the heat transfer rate given by
Ibm
My:= fbn (guess) Qe -60.BTU M,
_ Tbin
Given Mp(Hp~H1)) + My:Cp-(T = Ts) = ~60-
7
M; := Find{Mg) Ans,
Eq. (8.22) here becomes
Sotg = My(So ~S1) +Mo(S4~S3)- 2
To
Ts
S4-S3 = Cp-ln| —
T3,
Te = 529.67-rankine
mobwt :=
mol PD molt
M, = steam rate in kg/see
M, = nitrogen rate in kg/sec Mg := 20-
(1) = sat. lig. water @ 101.33 kPa entering
(2) = exit steam at 101.33 kPa and 150 degC
G)= nitrogen in @ 400 degC
(4) = nitrogen out at 170 degC Tar 443.15-K
176
ki
Hy 2 419.064 Sus lek (Table F.2)
wu i
Ha: me a— S2- 1.6075 (Table F.2)
ic and potential energies and setting
By Eq, (2.30), neglecting ki
the heat transfer rate given by
the work term to zero and wi
(guess)
‘Ms ‘= Find(Ms) Ans.
Eq, (6.22) here becomes
Sdotg = Ms (S2-S1)+Ma(Se~ $3) - 2
Ts kl
Sa~S3 = Cpin| == Tg *298.15K — Q = -80- My
8
6.86 Methane =1; propane =2
Ti=363.15K P= S500-KPa yy = 0.5 yel-y
© = 0.012 oy = 0.152 Ze = 0.286 2g = 0.276
Tel = 190.6K T= 3698K Pop = 42.48-bar
The elevated pressure here requires use of either an equation of state or
the Lee/Kesler correlation with pseudocritical parameters. We choose the
latter.
Tpe *= yr Tet + y2°Te Poe = yirPet + y2"Pea
The = 280.2K Pye = 44.235 bar
T Pp
Tpr = = Tpr = 1.296 Por = ——
maT ot Pcie Por = 1.243,
By interpolation in Tables E.3 and E.4:
ZO := 0.8010
=yraytyz@2 — @ = 0.082 W+@Zl Z=08l
For the molar mass of the mixture, we have:
molt := (y1-16.043 + yo 44.097) molwt = 30. oe
mol mol
3
T
v= 14.799 mdot ==
Pemoiwt am
Vemdot Vdor = 2.07% 10S a= YE = G 901m?
sec a
D=2964em Ans.
178
6.87 Vectors containing T, P, Te, and Pe for the calculation of Tr and Pr:
500 425.2 20 42.77)
400 304.2 200 73.83
450 552.0 60 79.00
600 6177 20 21.10
620 617.2 20. 36.06 a
Ts Piz Poss an
250 190.6 90, 45.99 te Te
150 154.6 20 $0.43 fo
500 469.7 10 33,70 Pres ee
450 430.8 35 78.84 Pe
400, 374.2, 15 40.60.
Ta7e O86
1308 2708
Dai 0780)
0.871 0948
pe = [1008 pr = [55
7312 1957
037 0387|
7,065) 0287
"as oad]
7,060) 0.369)
Parts (a), (2). (h).
‘500 20 425.2 42.77 190)
150 20 154.6 50.43 022
500} 33.70 |-bar @ ==] 252
450 35 430.8 78.84 245
400. 15, 374.2 40.6 327,
Tre
179
1.176 0.468
0.97 0397
Tr =| 1.065 Pr =| 0.297
1,045 oad
1.069 0369
———=
172
0139-2 (3.62)
re?
—
722
DBO (6.80) pBi = & (6.81)
Te
(0.253 ae ) (0.483 0311)
0.37 see 0.73 oass|
Bo =|-0309} Bi =| 6.718 10° | DBO =) 0574} DBI =} 0.522
| 0.321 4217x1073 | | 0.603 0.576 |
‘ie {
\-0.306 009x107? } (osex Losi)
Combine Eqs. (3.58), (3.59), and (6.40) and the definitions of Tr and Pr to get:
VR i= [k Te (Bo +0 Bl)
Pe
HR :=[R-Te-Pr| BO-TrDBO+o(B1-TeDB)]) 78)
ai teueieieecneet
SR :=[-R-Pr(DB0+o-DB1)} (6.79)
180
-1377x10° fags
(-200.647 ae
94.593 |, aq ae 2.469 i
| cm HR =| -1226x10° | sr =| 1.74
vr = |-355.907 | =a] mob
| 146.1 =1.746% 10° eles
| 3) a
\9282.454 =1.251« 10) fae
Parts (b), (c), (d), (@), and (£) -- By Lee/Kesler correlation:
By linear interpolation in Tables E.1—K.12:
9 1
(HR) (HR) HR
DEFINE: h0 equals hi equals “Sh equals TR
: RTe ae RTe fe RTe
0 1
(sR) (sR) SR
0 equals ———— si equals ——— s equals ——
‘q R eq R ql R
( 663 0.208 ‘2.008 228)
124 =050 $121
278| Zi =| -.088 2.970
783 =.036 0,671 0.596
707 0.138 = 1.486 0.169
—1.137 0.405
4.381 5.274
2.675, 2.910
0.473 0.557
0.824 0.289,
400 200 304.2) 224
450 60 552.0 au
T:=| 600 |-K P=} 20 |-bar Te :=| 617.7 |-K @ =| 492
620 20 6172 303
250 90 (isa6 012
181
oe — +
Z0+oZ1) B54) h:=(hO+a-h1) (6.76) s:=(s0+as1) (6.77)
——
heTorR)
O71
0.118
Z=| 0.235
0.772
0. 700)
AR
|| 597396
67.284
‘The Lee/Kesler tables indicate that the state in Part (c) is liquid.
6.8 Vectors containing T, P, Tel, Te2, Pel, Pe2, wl, and 2 for Parts (a) through (h)
650 60 562.2 553.6
300: 100 304.2. 132.9
600 100 304.2 568.7
ss /K oP bar Tel = pie /K Te2 oa K
200 15 1906 126.2
450 80 190.6 469.7
250 100, 126.2 154.6
182
48.98 40.73 210 210)
73.83 34.99 224 048
B83 24.90 | 224 400
48.72 50.40 .100 .087
29.63] P| gs.00 094 *| 012
45.99 34.00 012 038
45.99 33.70 012 252
\ 34.00. 30.43, 038, 022,
——__— > ——
Tpe i= (5:Tel +.5-Te2) Ppo := (.5-Pel + .5-Pe2) S-ool + 5-092)
T -
Tpr:= ra Ppr:= oe
557.9 44.855 0.21
218.55, ( 54.41 0.136
436.45 | 49.365 | 0312
293.8 49.56 | 0.094
Tpe = K Ppe = bar o=
282.05 | 67.81 0.053,
330.15 | 39.845 | 0.132
\ 140.4 } lanais/ (0.03
(1.165 (1.338
1.373 1 “|
1.375 2.026
1.191 1.913
Tr) aig Pers) 912
1,263 1.875
1.363 2.008
(1.781 2.369
183,
Lee/Kesler Correlation --- By linear interpolation in Tables E.1—-.12:
6543 219 ~1.395 ~461
7706 .1749 -1217 =116
7527 .1929 -1346 097
‘ws 6434 : 1501 hose —ESLO hi: ~400
7744 1990 1.340 049
6631 1853 1.623 254
7436 1933 “1372 =.110
9168. 1839, 0.820, 0.172
=.890 466
658 235
-n9 2402
944, 430,
=.704 224
965 =348
750 250
=.361, .095,
0 equals ca? hi equats cue), equals :
RTpe Ripe RTpe
sO equals (sx? sl equals feqae
R R
(20 + w-Z1) (3.54)
(no+e-h1) (6.76)
SS
(s0+@-s1) (6.77)
WA
14
18
Chapter 7 - Section A - Mathcad Solutions
28922 cp
mol
With the heat, work, and potential-energy terms set equal to zero and
with the initial velocity equal to zero, Eq. (2.32a) reduces to
uz
aH+—-=0 But AH = Cp-AT
Whence aT
From Table F.2 at 800 kPa and 280 deg
Hy = 30149 8) = 7.1595
kg keK
Interpolation in Table F.2 at P = 525 kPa and S~ 7.1595 kd/(kg*K) yields:
3
k
Hy = 2855.2. Vo = 531.21 mdot :='0.75-—=
ke gm sec
With the heat, work, and potential-energy terms set equal to zero and
with the initial velocity equal to zero, Eq, (2.32a) reduces to:
Whence
Ans.
mdot- V2
By Eq. (2.27), a= Ans.
up
ing problem may be earried out for a
series of exit pressures until a minimum cross-sectional area is found.
The corresponding pressure is the minimum obtainable in the
converging nozzle. Initial property values are as in the preceding
problem.
186
wy
Sy = 7.1595-—— S)= 8}
kgk
Interpolations in Table F.2 at several pressures and at the given
entropy yield the following values:
‘400 2855.2 S312
425 2868.2 307.12
P:=| 450 |-kPa asso.7 | 435.45 |
kg em.
475 2892.5 465.69
500, 2903.9 447.72
—
eee mdot-V2
uy = f—2(Ho - Hy Ags=
uw
565.2 7.05
541.7 7.022
uy =| 518.1 )™ ‘Ap =| 7.028 fem?
see
| 494.8 7.059
lan 2 7.127
Fit the P vs, A2 data with cubic spline and find
the minimum P at the point where the first
derivative of the spline is zero,
iss pis Pi 3, = Aa,
s := cspline(P,A) A(P) = interp(s,p,az,P)
00-kPa (guess)
a cm
Given 4 _A(pyain) = 0 =
ae (Poi) = 0 Pmin ?* Find( nin)
Ans. \ (Big) 7.021em? Ans.
187
Show spline fit graphically: := 400-kPa,401-kPa...500-kPa
23 tt 1
7 /
LE
Boo 7.07
A@)
om 7.05
7.9 From Table F.2 at 1400 kPa and 325 degC:
Hy = 3096.52 $1 = 700499. $2 =8}
ke kg-K
Interpolate in Table F.2 at a series of downstream pressures and at S
7.0499 kdi(kg*K) to find the minimum cross-sectional area.
800 2956.0 294.81
715 2988.5 30212 |,
ki com
750 |-kPa Hp =| 2940.8 | Va =| 309.82
725 2932.8 | 317.97
700. 2924.9, 326.69.
188
740
Since mdot is constant, 5.561
the quotient V2/a2 is a
measure of the area. Its a
‘minimum value occars very 5.552
close to the value at a
5577
vector index i=3.
At the throat, Ag = Gem?
Agua,
imdot = Logi t& Ans.
noes ae
V:
25
At the nozzle exit, P= 140 kPa and S = S1, the initial value. From
Table F.2 we see that steam at these conditions is wet. By
interpolation,
ky
4098-— s
ke-K vee
From Table F.4 at 130(psi) and 420 degF:
Hy v= 1233.68 Sy = 1.63102
ba Ibpyrankine
242
uP aw
By Eq. (2.322), ce eau aH =-7338"
2 Ibn
Hy +H Fy = 1154828
From Table F.4 at 38(psi), we see that the final state is wet steam:
1167-8
Din
Btu
ibgrrankine
Hyap
Svep = 1.6872
189)
TAI
712
0.987 (quality)
BTU
82 = Stig + ©(Svap — Si 82 = 1.67
a+ (Svap~ Stig) a Ibpyrankine
Sdotg := $2 S} Ans.
wick
2 molwt
By Eq. (2.320),
But AH= Cp-AT Whence
AEST
Initial t= 15 + 167.08 = 182.05 degC Ans.
Values from the steam tables for saturated-liquid water:
3
AIS degC: V := 1.001 288.15-K
em
Enthalpy difference for saturated liquid for a temperature change
from 14 to 15 degC:
(67.13 ~ 58.75) At 2K cp = a
em ‘At
I
stam P49
Apply Eq. (7.25) to the constant-enthalpy throttling process. Assumes
very small temperature change and property values independent of P.
190
ar BT) AP AT = 0.093K
cp (a BOD od =|
‘The entropy change for this process is given by Eq. (7.26):
AS = Cp T+AT) _py.ap AS = 1.408 x 1073
tT) gmk
Apply Eq. (5.36) with Q=0: To = 293.15-K
Wlost «= To-AS
TAB~TAS R= 8.314 P2:= 1.2
350 80 304.2 73.83 224)
350 60 282.3 50.40 087
The Pls Tes Pe = os
250 60 126.2 34.00 038
400, 20 369.8 42.48 152
5.457 1.045 “1.157
1.424 0.0
14394]
10 c
3.280 a)
1.213, 28.785.
As in Example 7.4, Eq, (6.84) is applied to this constant-enthalpy
Process. If the final state at 1.2 bar is assumed an ideal gas, then
(A) of Example 7.4 (pg. 258) applies. Its use requires expressions
HR and Cp at the initial conditions.
0.040
0.0
Eq.
for
List 1.084
a
Tl 124 1g
Tes Tr= Pr=
Te 1.981 1.765
(1.082, 0.471}
191
7.13 Redlich/Kwong equation: Q := 0.08664
_—
Bes (2 2) (3.50)
Guess: I
Zz
Given = 1+B-a8
2(6.,q) == Find(z)
+B;)
isl I =o 200 *) (6.62b)
ZABi Gi,
Rj == R-THi{(Z(B;.qi) ~ 1) ~ L.S-qrli] (6.64) The derivative in these
SRi == R-(In(Z(Bi,4)) Bi 0.5-qi;) (6.65) equations equals -0.5
The simplest procedure here is to iterate by guessing T2, and then
calculating it.
Guesses
2(61.a) (2.681 10° “8.177
9773) 2.253 10° etee racaa
oaee| |
\ 233 J
=1.396 x 10°)
192
7.14 Soaye/Redlich/Kwong equation: Q := 0.08664 W = 0.42748
—_______
(0.480+ 1.5740 - 0.1760") a
2B 3.49) 2(8,4) = Find(2)
; s) (6.62b)
26.4) :
if 20.8)- I af (my a | «| oot)
(ai) 4
SR = x) meto.a) -B 6.65)
(Tr)?
‘The derivative in these equations equals: ~«(3]
Oy
Now iterate for T2:
Guesses
2(6;,ai) = 3
O75 2.936 x 10° eee) 2
O79 3 3
oe x 10° 4.769 | 300
tos] «HR =| 7956 SR= Ths
0868] 526.246 -1.789 | 232
Fi a |
-1.523% 10° ) 2.679. (384)
Pe
-[rardeene Eni DB |
7 rT’ ||
193
TAS
37464 + 1.54226-0 — 0.269920")
— =
Pr Wea
=[QA2] 650 =|) osr
: ( " cen ‘ ) :
Guess: zl
Given 2=1+B-gB- ney (349) z(,q) == Find(z)
id (a ol Seale ) (6.62b)
22 ZIBi.ai) +e
ein)
SR = neta) aa »(2}'s 1 (6.65)
HR; =
(15)
The derivative in these equations equals: ele
4
104
Now iterate for T2: cae
26: (-so41x 10 6.152)
one 3 4.784
| 2.459% 10° ~
- HR =| -7459* 19] gp
a 600.376 ~1.847
O35 i ~
1.581 « 10" fee
ae see sceeseseeseset
12
748
Hy = 3462.9.
By Eq. (7.13),
mdot := an Ans.
For isentropic expansion, exhaust is wet steam:
Stig 2= 0.8321 ie Svap = 79094
x= 0.92 (quality)
195
cps] RAs Bice + S244 1) +2
z - oT?
270
297
229
383,
T2=
Ans,
S2= 8)
ks
Hyap := 2609.9-—
ms kg
Hp = Hig + (Hyap- Hig) H'g = 2.421 x 10° 2
Hp- Hy
H2-H)
7.19 The following vectors contain values for Parts (a) through (g). For intake
conditions:
kJ Ky
32743. 65597
kg kek
sso.e 2 esis
. = 0.80
3634.5. 6.9813 0.7
kg kek
0.82
3161.2: Ag. 6.4536- pis
0 kek 0.75
0.75
id
oan psa
Bu 0.75
1.6000
Tbgerankin€
Buu Buu
1389.6.— 1.5677-——_—
Dm Ibprrankine
196
For discharge conditions:
ki J
0.9441-—_ 7.7695: ——
kgk ( kek
0.8321. 19094
ek eK
i ig
0.649: 8.1511-——
kek
Ww J
w=| bos =| 7507
Stiq = kek Syap = kek
15301 7. 68S.
igk kek
Bu Bu
0.175024 1.92002
Tbyerank ibjrankine
0.220024 1.8625.
bavrankine Tarrankine
2893022 2625.44 80- ke
kg kg s
251.453 2609.9.
kg kg
191.832 2584. gt 70 48.
ke ke
ky kK
Hig =| 3405645 | Hap =| 2086057
504. ore 2706.3-4
ke kg
Bu Btu Ib
94,03-— 16.15 — |.
Bm Bin 10
120.99.B% 1273-34 100.0"
bm bm see
197
S1= Stig
——____ >
NQis Hp := [ Hlig + ¥’2-(Hyap ~ Hig) }
© Syap~ Siig
aimee >
AH := [n-(H’2-Hi)] Hp =H) +AH Wot := (AH-mdot)
———
ow 2 Mtg Tonawls..os,.))
0 Hig S2:= [Stig + x2-(Svap ~ Stig) J
K Ans
bar R= 8314 He
mole-K
J
molK
720° T:242315K Po?
For isentropic expansion,
For the heat capacity of nitrogen:
198
721
0.593-10*
K
1D = 0.040-10°-K?
A:= 3.280
For the entropy change of an ideal gas, combine Eqs. (5.14) & (5.15)
with C=0. Substitute:
T= 05 (guess)
Given
AS= nA mies]
Ans.
Find(t) To=
‘Thus the ini
Ty 1223.15-K
J
mol
Cp ss 32+
Eqs. (7.18) and (7.19) derived for isentropic compression apply equally well
for isentropic expansion. They combine to give:
Ws = -15231
mol
Ans.
Ans.
199
Te = 408.1-K 36.48-bar —@ := 0.181
Po = 5000-kPa =P == 500-kPa
For the heat capacity of isobutane:
37,853-10"> crn rilegass107®
K e
Tyo = 1.282 Pro = 1.3706
Use generalized second-virial correlation (borderline at TO, but good enough):
HRB(i.282, 1.3706,0.181) = -0.940291 HRB := ~0.940291
SRB(i.282, 1.3706,0.181) = ~0.534176 SRBo := -0.534176
The entropy change is given by Eq. (6.83) combined with Eq. (5.15) with D =
T= 05 — (guess) SRB = 0.0 (starting value)
Given
AS = Rj A-In(t) 4 [» To +C-To- ()]« —1)-1n if) uJ
42 Po)
+SRB - SRBo
tau(SRB) := Find(z) T := tau(SRB)-To 443.37K
At the final P and this T, evaluate SRB:
TCT) z T,(T) = 1.0864 P, = 0.1371
SRB(I,0864,0.1317,0.181) = ~0.082852 SRB := -0.082852
Do a further iteration for T:
T := tau(SRB)-To T = 445.65K T,(T) = 1.09202
SRB( 1.09202, 0.1317,0.181) = ~0.081601 SRB := -0.081601
T:= tau(SRB)-To T = 445.62K T-(T) = 1.0919
200
‘The enthalpy change is given by Eq. (6.82):
HRB(1.0919,0.1317,0.181) = -0.126003
1p H(523.15,445.62, 1.677,37.853:10 >, -11.945-10" §,0.0
RICPH — AHig = -1 1.089
ICPH := -1333.811-K Aig nal
$326.5
mol
AH! = AHig +R-Te-(HRB ~ HRBo) AH
The actual enthalpy change from Eq. (7.16):
08 — ndot= 700M ate aH AH = ~66612—
‘sec. mol
Wot = ndot-AH Woot $'=4662:8kW Ans.
‘The actual final temperature is now found from Eq. (6.82) combined with
Eq, (4.7), written:
t= 0.7 (guess)
Given
AH R. Ato(s=1)+ Brel? $a? (3 J
+Te(HRB — HRBo)
tau(HRB) := Find(z) T := tau(HRB)-To T = 457.88K
Although it makes little difference, we recalculate HRB at this temperature:
Te(T) = 1.122
HRB(1.1220,0.1317,0.181) = -0.119069 HRB == -0,119069
T = tau(HRB)-To
7.23 From Table F.2 @ 1700 kPa & 225 degC:
851.02 81 65138
ke ek
201
7.24
For
At 10 kPa:
Hig = tora
dot := 05-48
sec
Hy := Mig + X2-(Hvap- Hlig) AH = H)-Hy
Ep = 2.465 10° an = -385.g48
kg kg
(a) Qdot := mdot-AH — Wdot Ans.
(b) For isentropic expansion to 10 kPa, produ
S1~ Stig
19 wet steam:
Hhig + ¥'2-(Hyap ~ Hig)
wap = Stig
2 aera Hy = 2.063% 10° =
kg
Wdot' := mdot-(H'- Hj) Ans.
To = 673.15-K I-bar
For isentropic expansion, I
motK
For the heat capacity of carbon dioxide:
A= 5.457 Bs D ss -1.157-10°K?
the entropy change of an ideal gas, combine Eqs. (5.14) & (5.15) with C= 0:
1.5 (guess)
1
Given
AS= n{Ants[are + a ; (=) (c-1)- (n
a }
728
1 = 0,693,
.= Find(s) = 1-To
ICPH(673.15 466.456, 5.457, 1.045-10”*,0.0,-1,157-1
ICPH := -1174.8¢-K AH = RICPH
0.78 Work := AH’
(AH := Work AH = -7. 326-8
mol
For the enthalpy change of an ideal gas, combine Eqs. (4.2) and (4.7)
with C= 0:
Given
r Hi
SHER Atole-)+Bagel?-i)-2{2 ‘)
2 Toe
t= Find(z) += 0.772 wT T= 519.9K Ans.
Thus the final temperature is 246.75 degC
Vectors containing data for Parts (a) through (¢):
500 6 371 12 3.5
450 3 376 20 40
Th=] 525] Pri=]10] t2:=|458] P2:=]3.0] cp=|55/-R
475 7 32 15 45
580 4 403 12, 2.5
—
Cp(T2- TD] Ideal gases with constant heat capacities
see ee eee
[ R
(7.22) Applies to expanders as
q
cpTl (2) ° || wellas to compressors
Pl
203
7.26
ndot:= 175 T1:= 550 PL?
Guesses: = 0.75 Wdot := 600
Given.
[ a
&
anendot-3.5-R-TL (@) -1
Wadot = ——— ee 7 = 0.065 + 0.08-In(Wdot)
10
( Wdot
[ Waoe!
) = Find(Waot,n)
n
For an expander operating with an ideal gas with constant Cp, one can
show that:
T2 = 433.213
By Eq. (5.14):
as=r{& (2) - (2) AS = 6.435
R TI \Pi}}
By Eq. (5.37), for adiabatic operation :
Sdotg = ndot-AS ‘ Ans.
204
7.27 Properties of superheated steam at 4500 kPa and 400 C from Table F.2,
p. 714:
HI := 3207.1 Sls
5.7093
saturated vapor, then
ig. 7.4) must produce
If the exhaust steam (Point 2, Fig. 7.4) is "dry,
isentropicexpansion to the same pressure (Point 2',
"wet" steam, withentropy:
S2=
1 = 6.7093 = (x)(Svap) + (1-x)(Sliq) [xis quality]
A second relation follows from Eq. (7.16), written:
AH = Hvap - 3207.
(N)AH) = (0.75)] (2)(Hivap) + (1-x) (Big) - 3207.1]
Each of these equations may be solved for x. Given a final temperature
and the corresponding vapor pressure, values for Svap, Sliq, Hvap, and
Hlig are found from the table for saturated steam, and substitution into
the equations for x produces two values. The required pressure is the one
for which the two values of x agree, This is clearly a trial process. For a
final trial temperature of 120 degC, the following values of Hand S for
saturated liquid and saturated vapor are found in the steam table:
503.7 Hy = 2706.0
Sl = 1.5276 Sv = 7.1293
‘The two equations for x are:
_ Hy 801.7-.75-HI
75-(Hv — HD)
XH
The trial values given produce:
These are sufficiently close, and we conclude that:
t=120 degC; P=198.54 kPa
If were 0.8, the pressure would be higher, because a smaller pressure drop
would be required to produce the same work and AH.
208
7.29
7.30
PLs=
atm, PQ:=tatm = Ths 15C n= 0.55
Data in Table F.1 for saturated liquid water at 15 degC give:
Eqs. (7.16) and (7.24) combine to give: AH = -V-(P2—P1)
Ws i= AH (7.14)
AH - V-(P2 -P1)
Eq. (7.25) with B=0 is solved for AT: AT. a
P
Assume nitrogen an ideal gas. First find the temperature after isentropic
expansion from a combination of Eqs. (5.14) & (5.15) with C= 0. Then
find the work (enthalpy change) of isentropic expansion by a combination
of Eqs. (4.2) and (4.7) with C= 0. The actual work (enthalpy change) is
found from Eq. (7.20). From this value, the actual temperature is found
by a second application of the preceding equation, this time solving it for
the temperature. The following vectors contain values for Parts (a)
through (e):
(7s 6-bar I-bar
673.15 S-bar I-bar
TTBAS |-K 7-bar P:= | L-bar
723.15 8-bar 2-bar |
758.37 95-psi 1s-psi)
200 0.80
150 0.75
adot 175 ies 0.78
100 a 0.85
0.5-453.59 0.80 Ri Bale
206
For the heat capacity of nitrogen:
3
0593.10 "= 0,040-10%.K?
Tau(To.Po,P) := Find(t) 11 = Tau(To, Po, Pi)
(460.67
431.36
= | 453.48 |K
494.54
455.14,
T= To,ti
rcphi( 753.15, 460.67,3.280,0.593-10" 3,0,0,0.040-105) = —1167.969
1cpHi(673.15,431.36,3.280,0.593-10" 3,0.0,0.040. wa 875.585
1cpr(773.15,453.48,3.280,0.593-10" 3,0.0,0.040-105)
IcPH(723.15, 494.54, 3.280, 0.593-10”°,0.0,0,040-10°) = 834.936
1CPH(755.37,455.14,3.280 0.593.107? ,0.0,0.040-10°) = -1096.005
=1168.427
-1167.969)) (- 9710.3)
875.585 -7279.6 7
ICPH := } -1168.427 /K AH’ := R-ICPH AH’ = | -9714,3 (inal
834.936 | 6941.
-1096.005 ) \-91 aio
207
AH = | ~7577.2 | —
AHS [stole +8 rele JP
Tau(To,AH) = Find(t) 71 = Tau(To,
| 529,34
‘AS16.28
7.31 Property values and data from Example 7.6:
Hy 3301.64 Si= 6.6858 mdot = 59.02.48.
kg kek see
Hp := 2436.0. 82 = 7.6846. Wot := -56400-kW
ke kek
Ty = 300-K By Eq. 6.26)
Weotigeat = mdot| H2~H1-Te-(S2-Si)] Wotideat = -74084 KW
Wot
nes mes0.761. Ans.
‘dotideal
The process is adiabatic; Eq. (5.33) becom
Sdotg := mdot-(S2 - $1) Sdotg, 149 - Ans,
Wabotjost = To Sdotg ‘Waottoct = 17685k\ Ans.
208
7.32 For sat, vapor steam at 1200 kPa, Table F.2:
Hp = 2782.7. $2 = 65194
kg kek
The saturation temperature is 187.96 degC.
‘The exit temperature of the exhaust gas is therefore 197.96 degC, and
the temperature CHANGE of the exhaust gas is -202.04 K.
For the water at 20 degC from Table F.1,
= 02963
Hi $1
kek
‘The turbine exhaust will be wet vapor steam.
For sat. liquid and sat. vapor at the turbine exhaust pressure of 25 kPa, the
best property values are found from Table F.1 by interpolation between 64
and 65 degC:
72.084 Hy = 2346.3.
kg kg
i WI
08932 Sy = 6.9391
kek : kek
For isentropic expansion of steam in the turbine:
S'3- Stig
Ss: 8; : eee
nae - avaeicsiy
83-6519 x3 = 0811 Hy = 2.17410
kg
AH; == n-(H3 ~ Ha) Hy i= Ho + AHp3
Alig = ~437.964 2345x1094
kg ke
H3 ~ Hig
a= 83 = Siig + x3'Siy
x3 = 0.883, Soca eR.
209
Y
25.
see
(273.15 + 197.96):K
For the exhaust gases:
Ty = (273.15 + 400)-K
Ty = 673.15K 12 = 47L11K
McPH(673.15,471.11,3.34, 1.12:1073,0.0,0.0) = 3.9808
McPs(673.15.471.11,3.34,1.12-10 $,0.0,0.0) = 3.9741
MCPH =
9808 MCPS := 3.9741
molwe:= 1s 22
mol
{To
AH ggs °= R-MCPH-(T2 ~ T1) AS gas == R-MCPS: Hla
3k
‘AH gas = ~6.687 x 10° —— = -11.791
ae kmol ad kmoFK
Energy balance on boiler:
=ndot-AHgas
mdot = ———_* mdot = 0.30971 £2.
Ho —Hy sec
(@) Wot := mdot-(H3 — Hz} Wadot = S135,65KW Ans.
(b) By Eq. (5.25): To = 293.15-K
Wabtideal ‘= ndot-AH gas + mdot-(H3 - Hj)
4 -Tar{ ndot-ASgag + mdot-(S3 — si)}
_ Wit g
Waotigeat
Webtideal = -314.302kW 1
(©) For both the boiler and the turbine, Eq. (5.33) applies with Q
210
Boiler: Ans,
For the turbine: Sdotg = mdot(S3 ~ $2)
Turbine:
SEW!
1565 Ans.
K
w ;
(@) Waotisoitr = 0453457 “Widjsnbsie = 192014
W
Waottoserurbine = 0.1560-——
Woottostboiter
~ [Waotideal
Fractionpoite
Wdotiost turbine
[Waoticeal|
Fractionurbine ‘=
Note that: ny + Fractionpjjer + Practionrurbine = 1
7.34 From Table F.2 for sat. vap. at 125 kPa:
Hy = 2685.22 si = 72847
kg kgK
For isentropic expansion, Sip = Sy = 7.2847
Interpolation in Table F.2 at 700 kPa for the enthalpy of steam with this
entropy gives
H
78H = 1 an = 46935982
n kg
any
Hy := Hy +H Wyss Ans.
an
Interpolation in Table F.2 at 700 kPa for the entropy of steam with this
enthalpy gives
mdot Wdot
7.35 Assume air an ideal gas. First find the temperature after isentropic
compression from a combination of Eqs. (5.14) & (5.15) with C= 0. Then
find the work (enthalpy change) of isentropic compression by a
combination of Eqs. (4.2) and (4.7) with C=0. The actual work (enthalpy
change) is found from Eq. (7.20). From this value, the actual temperature
is found by a second application of the preceding equation, this time
solving it for the temperature. The following veetors contain values for
Parts (a) through ():
298.15, 101.33-kPa (375-kPa
353.15 375:kPa 1000-kPa
Tye Le 100:kPa 500-kPa
373.15, 500-kPa 1300-kPa
299.82 14.7-psi 55-psi
\s3e.71 S5.psi 135:psi
100 0.75
100 0.70
150} mot 0.80
50 | see 0.75 :
0.5-453.59 0.75
0.5-453.59 0.70,
For the heat capacity of air:
0575-10 °
A=3355 B D := ~0.016-10°K?
0.5 (guess)
212
Given
AS= raw “fen + ea (: . I (e~1)- HF]
Tau(To,Po,P) := Find(t) Ws Tau(To,.Po,,Pi)
(451.06)
464.5
476.19 |
oti
| 486.87
| 434.74
(435.71
1cPH(298.15,431.06,3.355,0.575-10"?,0.0,-0.016:105) = 472.123
rcpH(353. 15 ,464.50, 3.355 ,0.575-10 ~,0.0,-0.016- 10°) = 398.669
1CPH(303.15, 476.19, 3.355,0.575-10" °,0.0,-0.016 105) = 617.403
1cPH(373 15,486.87, 3.355 ,0.575-10~ ,0.0,-0.016- 10°) = 408.647
ropti(299,82,434.74,3.355,0.575.107°,0.0,-0.016 10°) = 479.494
icpHi(338.71 ,435.71,3.385,0.575-1073,0.0,-0.016-105) = 345.980
472.123 ) (3925.2
398.669 | 3314.5
617.403 5133.1] J
ICPH := K AH’ = R-ICPH AH'= _
408.647 3397.5 | mol
479.494 3986.5
345.980 5233.6 (2876.5
te 4735
an := [SH 6416.41 3
7 AH = ae
4530 | mol
53154
41093
213
7.36
T= 1S (guess)
Given site n[ tol ye roel?) P(E)
To
Tau(To, AH) := Find(t) Gi Tau(To, AHI)
——>
Wadbot := (ndot-AH)
Ammonia: — Ty = 405.7-K Pore 1128bar = 0.253
To = 294.15-K Py = 200-kPa P= 1000kPa
For the heat capacity of ammo
0.186-10°-K?
Tro = 0.725
Tro
Pro = 0.0177
Te
Use generalized second-virial correlation:
HRB(0.7250,0.0177,0.253) = —0.045843 0.045843
SRB(0.7250,,0.0177,0.253) = ~0.044781 SRBo := ~0.044781
The entropy change is given by Eq. (6.83) combined with Eq. (5.15); C= 0:
Tis 14 (guess) SRB := 0.0 (starting value)
214
Given
AS= nls nta[naae t or ee) (r= 1)- (¢) .
T = tau(SRB)-Tp T = 416.907K
At the final P and this T, evaluate SRB:
tT = Te) = 1.028 P, = 0.0887
°
SRB(1.0276, 0.0887 0.253) = ~0.069844 SRB := -0.069844
Iterate for T:
T := tau(SRB)-Tp T = 423.094K T,(T) = 1.043
B( 1.0429, 0.0887 ,0.253) = -0.066701 SRB := -0.066701
tau(SRB):To T= 422814K T,(T) = 1.042
SRB(1.0422, 0.0887 ,0.253) = ~0,066841 SRB := ~0.066841
‘T := tau(SRB)-To = 422.83K Te(T) = 1.0422
A further iteration is clearly not warranted. The enthalpy change for
this final T is given by Eq. (6.82), with HRB at the above
HRB(1.0422, 0.0887, 0.253) = -0.097461 0.097461
1cpHi(294.15,422.83,3.578,3.020-10" ?,0.0,-0.186-10°) = 580.487
ICPH == $80.487-K AH,
sICPH
AH’ := AHig + R-Te (HRB — HRBo)
The actual enthalpy change from Eq. (7.17):
AH
aH = 56732.
q mol
n= 0.82 AH
218
‘The actual final temperature
Faq. (4.7), written:
now found from Eq. (6.82) combined with
14 (guess)
Given
AHER Ato (s— 1) +B-10"
+ Ty (HRB - HRBo)
Find(t) T := teu(HRB)-To T = 448.44K
1.1054, Iterate for final T:
HRB(1.1054 , 0.0887 , 0.25: 0.085587 HRB := --0.085587
T = tau(HRB)-To T= 44745K Ty(T) = 1.1029
HRB(1.1029, 0.0887 0.253) = —0.086005 HRB := —0.086005
T := tau(HRB)-To T,(T) = 1.103 T =44748K Ans.
SRB(1.1030,0.0887 0.253) = -0.056033
1.521
as , -2.168-1076,0.0) = 1.7134
ICPH := 636.059-K ICPS := 1.77134
AH := RICPH ait = 52882
mol
P.
as z= r{ 100s -tn| 22 as = 3201
(Pi molK
Since the process is adiabatie: Sq = AS
221
Ans,
Ans.
72 Tl = G5+273.15)K TI = 308.15K
‘T2 := (200 + 273.15)K T2 = 473.15K
Cp 3.5K
v- RT ndot = 19.775 22
PL see
With compression from the same initial conditions (P1,T1) to the same
final conditions (P2,12) in each stage, the same efficiency in each stage,
and the same power delivered to each stage, the applicable equations are:
1
P2) N where r is the pressure ratio in each stage and N is
Pi) the aumber of stages.)
Eq, (7.23) may be solved for T2prime: T2 :=[(T2-T1)-n +T1]
12 =4154K Eq. (7.18) written for a single stage is:
RL
pa \NCp
T2=T1 (@ | Put in logarithmic form and solve for N:
(a) Although any number of
N’= 3.743 stages greater than this would
serve, design for 4 stages.
222
7.44
p2\N
(b) Calculate r for 4stages: Nix 4 or (2) r
Power requirement per stage follows from Eq. (7.22). In kW/stage:
(©) Because the gas (ideal) leaving the intercooler and the gas entering the
compressor are at the same temperature (308.15 K), there is no enthalpy
change for the compressor/interehanger system, and the first law yields:
Qdot, := -Weot, ‘Qdote = 975
Heat duty = 87.94 kW/interchanger
(@) Energy balance on each interchanger (subscript w denotes water):
With data for saturated liquid water from the steam tables:
Aly = (1884-104. any = 83.642
kg kg
{Qdot,| 7
mdotyy := (in each interchanger)
‘Aly
R= 8314
464 6 35
347 5 25
12/455) P2=|6| Cp=l4s]R
505 8 55
496 7, 4.0,
AH := [Cp-(T2~T1)] Ideal gases with constant heat capacities
223,
Ans.
7.47 The following vectors con
conditions first:
298.15 100-kPa.
363.15 200-kPa
333.15 |-K Py =| 20-kPa
294.26 L-atm
366.48, 15-psi
'2000-kPa 0.75
fevers 0.70
5000-kPa n= |0.75
20atm 0.70
1500-psi 0.75
224
(7.22) AHs =| 4.745 x 10°
(3.219% 108
3.729% 10°
3
5.959 10°
4.765 x 10°
values for Parts (a) through (e). Intake
20-kg
30kg
mdot :=} 15:kg }-
50-b
80-16
t
sec.
257.2
6962)
523:1.4-——
2173
714,3.
From the steam tables for sat.liq. water at the initial temperature (heat
capacity calculated from enthalpy values):
1.003 45
1.036} 420
V:=| 1017 eo 4.20 ek
1.002 4.185
1.038 (420
ByEq.(724) — AHs =[V-(P2
1.906
an v(i=een}(P2= i)
By Eq. (7.25) AT :=
Cp
(298.338)
363.957
T2:=(T1 +47) Tz =| 333.762 |K
(367.986,
225
7.48 Results from Example 7.10:
i
AH usr
To i= 300K Wideat
Since the process is adiabati
Sq = AS
Whost := To AS
ki
3622.7-—— 6.90!
kg aK
KD
i 6.9485-——
3529.6 ae
cl
ky a
3635.4-—~ 6.9875: kek
7 = 7
Mp? ki 2 ‘
3475.6.— oats
kg
BTU
STU ..
1507.0. BTU 16595
Ibm
BIU
TU a
1558.8. 222 1.6759. ere
bm
Sat. liq. and sat. vap. values from Tables F.2 and F.4 @ P3 = P4:
i
= 2584.82
332.4
tons e
WI
u 2609.95.
453
pare is
ia
H i
i912 saan
e Ene 2616.0
0644 ot
419.064= =
Bru 11505.BTU
7 Se .
tso17 se
BTU
BTU :
69.73.50 105.8.
Tn 7
229
Chapter 8 - Section A - Mathcad Solutions
8.1 With reference to Fig. 8.1, SI units,
At point 2: Table F.2, Ha 8p == 6.9636
At point 4: Table Fl, Hg :=
At point 1:
At point 3: Table Fil, Hig := Ha AB := 2382.9
x3 = 0.96 Hg i= Hiq+%sAHyy — H3 = 2496.9
Stig * AS = 7.3241
For isentropic expansion, $'3 = S2
83-8)
x= la x33 = 0.855
ASiy
[= Hig +X3AHy Hs = 2246
H3~ Ha
Maubine = Fe $05. Ans
Wy = Hs ~ Ha Qu ss Ha- Hy
W, = -1.035x 10° Qu = 3.322 10°
noce = Ha Ans
a Qu
8.2 mdot:= 1.0 (kg/s)
‘The following property values are found by linear interpolation in Table F.1:
State 1, Sat, Liquid at TH: HI =
60.7 Si
3482 PL
State 2, Sat. Vapor at TH: H2 := 2792.0 S2:= 6.4139 P2
533
533
2ar
83
State 3, Wet Vapor at TC: Hligq
= 125
State 4, Wet Vapor at TC: Sli = 0.3929
(a) The pressures in kPa appear above.
(b) Steps 2-3 and 4--1 (Fig, 8.2) are isentropic, for which $3=S2 and S1=S4,
Thus by Eq. 6.73):
82-5)
rte SS gear
Svap — Sliq
Svap - Sliq
(©) The rate of heat addition, Step 1
Qdotl2 := mdot-(H2 - H1) (isis)
(A) The rate of heat rejection, Step 3.
H3 := Hliq + x3-(Hvap—Hliq) 4 := Hliq + x4-(Hvap — Hliq)
HB = 1.919% 10° HA = 699.083
Qdot34 := mdot-(H4 - H3) (kS/s)
(©) WdotI2 = 0 Wadot34 := 0
Waot23 := mdot(H3 ~ H2) t
Wdot4] := mdot (HI - H4) Waoid = 161.6
Wdot23 + Weotdl | ¥
a ee 0.368
ee Qdotl2 | :
Note that the first law is satisfied:
ZQ = Qdotl2 + Qdot34. EW s= Wdot23 + Wdot4]
TQ+IW=0
‘The following veetors contain values for Parts (a) through ().
Enthalpies and entropies for superheated vapor, Tables F.2 and F.4 @ P2 anc
T2 (see Fig. 8.4):
28
0.6493 sisi
kgK keK
0.8321. 7 9094.-K.
kek kek
i iw
0.6493-—— 8.1511-——
kek kgK |
Syap =
1.3069. 73854
kek kek
: ;
o31a1-—BtU _ 1.756821 _
Tbpy-rankine mrankine
0.1326. — BTU 1.97812
Tbpyrankine Ibpyrankine
em?
1.010-——
gm
cm?
1.017
am 0.80 ( 0.75
? 0.75 0.75
0.80 0.80
Manbine | 0.78 Mpume | 9.75
0.78 075
(oso, 0.75
230
2 10000-kPa 10-kPa
100 7000-KPa 20kPa
1m | 5 8500-kPa. 10-kPa
1O-KW PL Pass
50 6500-kPa 101.33-kPa
30 950-psi 14.7-psi
80 (i2s-psi_) (psi
Ha = Hig Hi = Ha+ Wpump
—
S2~Stiq
S'3 = So x’
wap — Stig
esaegeeeeLeaeaeeeeEeee
Hy = [Ho + neubine:(H'3 ~ Ha) |
See:
wai
mdot = ——~Set
Wrurbine + Wpamp
Qdoty
Qdote = Qdot + Waot
Answers follow:
240705)
395111 | ag
sec
231
8.4
~ Qdoty
Subscripts refer to Fig. 8.3.
Saturated liquid at 50 kPa (point 4)
om? KI
Va == 1.030- Hq == 340.564-—
am kg
Saturated liquid and vapor at 50 kPa:
ky
Hiig := H. Hyap = 2646.0-—
hig = Ha ” ie
ki ky
Stiq == 1.0912-— Syap = 7.5947-——
ig wk ap .
: i
By Eq. (7.24), Woump := Va-(P4-P1) Woump = 3.348 ie
Hy = Ha+ Wpunp m=sa918
8
The following vectors give values for temperatures of 450, 550, and 650 degC:
3340.6 7.0373
Hp =| 3568.3 | So =| 7.3282 | EL
kg kg-K
3792.9 7.5891
232
85.
13 := Hig + x'3-(Hyap ~ Htig)
S'3—Stiq
ap ~ Stig
Wrurbine = H'3 - Ho
eee
__ |Weurine + Wump|
Qu = (H2- Hi)
Qu
(0.297
0314}: Ans.
0.532
Subscripts refer to Fig. 8.3.
Saturated liquid at 30 kPa (point 4)
as
Va = 1.022 Hy s= 289.3022
am kg
Saturated liquid and vapor at 30 kPa:
. = 5000
Hig = Ha Hyap ‘= 2625.4 P4:=| 7500 |-kPa
10000.
By Eq. (7.24),
Hy = Ha + Wpump
294.381
296.936 2
299.491 }
Hy =
233
86
The following vectors give values for pressures of $000, 7500, and 10000 kPa
at 600 degC
3664.5 7.2578
Hp =| 3643.7} So =| 7.0526 ae
3622.7 6.9013
$'3 = Sz
H's == Hiig + 3-(Hyvap — Hi
Hp- Hi)
£5 (0.925
x3 =| 0.895 Aus.
0.873
From Table F.2 at 7000 kPa and 640 degC:
Hy = 376642 $1 = 7.2200 82:58)
kg kg-K
For sat. lig. and sat. vap. at 20 kPa:
ky
Hlig i= 251.4532 Hyap = 2609.9-—
kg kg
ky Ky
Sig = 0.6321 Svap = 7.9094
The following enthalpies are interpolated in Table F.2 at four values for
intermediate pressure P2:
725 3023.9
750 3032.5
Pais “kPa u i
TIS 3040.9 | kg
800, (3049.0
234
n= 0.78 Wr = n-(H2-Hi) Hy +Wi2
-579.15 (38? 3) 7.4939)
572.442 | Ky 3194 | Ky 7.4898 | kd
Wr= — F= — Se —
565.89 | ke 3200.5 | kz 74851 | kg-K
(559.572, 3206.8, \TAT97,
where the entropy values are by interpolation in Table F.2 at P2.
S2~ Stig
xy H’3 = Hiig + 8'3-(Hyap ~ Hi
Syap~ Siig ao
Was = n-(H3 ~ Ha) 20.817
-7811 | ky
AW = Wi2- W23 AW= ne
man 3.073 | ke
17.723
The work difference is essentially linear in P2, and we interpolate linearly to
find the value of P2 for which the work difference is zero:
timerp a ,P2,0.0| = 765.16kPa (P2)
(a)
Also needed are values of H2 and S2 at this pressure. Again we do linear
interpolations:
linterp(P2,H2,765.16-kPa) = 3197, oe Hp = 3197.9.
2 Z
linterp(P2, 82, 765.16-kPa) = 7. 4369 Sp i= 7.4869.
kek kg
‘We can now find the temperature at this state by interplation in Table F.2.
‘This gives an intermediate steam temperature 2 of 366.6 degC.
235
‘The work calculations must be repeated for THIS case:
So-
Wio = H2-Hy eo
Svap ~ Stig
Wr = -s68.54 x3 = 0.94
kg
= Hig + ¥3-(Hivap ~ Hig)
kJ
2.469% 10° Waa = ~568.46—
kg
ks
Work = Wiz + W23 Work = 1137
8
For a single fsentropie expansion from the initial pressure to the
final pressure, which yields a wet exhaust:
$1 - Sti
x3 aaa Bs = Myig +¢3-(Hyap — Hig)
wap ~ Stig
‘4 3k
x3 = 0.903 Hy = 238% 10)
Wis H3- Hy w= 1386.22
kg
‘Whence the overall efficiency is:
Work i
overall = 8202, Ans.
He lh w
236
8.7
From Table F.2 for steam at 4500 kPa and 500 degC:
i J
3439.3. 8) = 7.031
kg . kg-K
By interpolation at 380 kPa und this entropy,
Fh
n= 028
Hg = 2.918% 10°
Isentropic expansion to 20 kPa:
Sig = S2 Exhaust is wet: for sat. liq. & vap.:
KI
Hyiq == 251.453-— Hyap = 2609.9. zo
kg kg
WD Ky
0.8321. Svap = 7.9094.
kgK 7 kek
237
Wi= (Hs - Ha)
w= -sa.sseK
ke
Aig + x'4-(Hvap ~ Hig)
Hy = 2317x10°
ke
56x 10°
H.
af ke
Vs-(P6~P.
Wpump elt) Hg != Hs + Wpurap
ks i
Wamp = 5.841 = iis = 257.2947
For sat, lig, at 350 kPa (Table F.2):
24.270. ty
kg
We need the enthalpy of compressed liquid at point 1, where the pressure is
4500 kPa and the temperature is:
Hy 38.87 (degC)
138.87 — 6 Thm (tr 273.15)-K ty = 15287
temperature, 132.87 degC
ferpotation in Table F.1 gives:
3
559.5. Psat = 294.26-kPa Veatstig i= 1.073
kg gm
Heattig
Also by approximation, the definition of the volume expansivity yields:
Pr: Po
1 (soto om
Veatlig 20 J gmK
p=932x 104+
238
88
By Eq. (7.25),
KI
Hy = Hoattig + Vsattig(1-B-T1)(P1—Psat) Hi = 561 305
By an energy balance on the feedwater heater:
1-H
H3— Hz
mass
«mass =0,13028kg Ans.
Work in 2nd section of turbine:
Wir = (ekg — mass)-(H4~ Hs) Wh = ~307.567k)
Wret °= (i+ Wpamp)I-kg + Wir Wret = ~823.3K)
Que: (Ho ~ Hi)-1-ke
W, .
Qu = 2878kI ns [Wn n= 0.2861 Ans.
Qu
Refer to figure in preceding problem.
Although entropy values are not needed for most points in the process, they are
recorded here for future use in Problem 15.8.
From Table F.4 for steam at 650(psia) & 900 degF:
Hp = 1461.22
Ibm,
By interpolation at S0(psia) and this entropy,
BIU
Hs = 1180.4-—— Wi:= n-(H'3 - He)
m
Hg i= H+ Wy = 12422810 ~219.0420U
Ibm bm
BIU,
83:5 1.7431.
- Tb, Tankine
Isentropic expansion fo 1(psia)
Exhaust is wet: for sat. lig. & vap.:
BTU
Hg #= 69.73-— Hyap 11053228
Ibm Sm
239
BTU
0.1326-————__
Ibryrankine
Hy +n-(H4~ Ha)
Hy - Hiig
yap — Hig
x4 = 0.944
Ps = I-psi
Vs(P6 - Ps)
Woump ==
‘pump
650:psi
For sat. lig. at S0(psia) (Table F.4):
Hy = 250.21 220
Ibm
BTU
Syap = 1.9781-———_—_—
a Ibe rankine
Aig + X4(Hyap ~ Hig)
31.204 BEL
tom
Hg = 1097957
$4 := Stig + X4:(Svap ~ Stig)
Sq = 1.874g—BU_
Ib rankine
3
#
Vii V5 = 0.0161-—
Hig. 3 Ibm
BTU
Woump = 2.489
cae I
Ho = Hs + Wpump He = raise
ty := 281.01 S87 == 0.4112. em
Toy tankine
We need the enthalpy of compressed liquid at point 1, where the pressure is
{650(psia) and the temperature is
ty = 281.01-11
Ty (t1 +459.67)-rankine ty = 270.01
At this temperature, 270.01 degF, interpolation in Table ¥.3 gives:
Poot = 41.87-psi
Bru
238,96-—— ;
bn BrU
Hes
240
of the volume expansivity yields:
(cotmsootre) a
———— |———_ _ Piss
Veattiq 20 7) Tone rankine
B= 495 10° J
rankine
By Eq. (7.25) and (7.26),
, BTU
Hy = Hsatiiq + Vsataig:(1 - B°T1)-(P1 ~ Psat) Hy = 257.65 —
BTU
S81 == Ssatliq + Vsat.ig’B-(P1 Ps. S} = 0.397-———_—_
1 liq + Vsat.tig'B-(Pi = Psat) 1 iaankne
By an energy balance on the feedwater heater:
H) - He
See ret Ey tte ‘mass =0-18687 Ibm, Ans.
Work in 2nd section of turbine:
War: (I-lbm— mass)-(Hg ~ H3) Wy = -158.051 BTU
Wret = (Wi+ Wpump)1+Ibm + War Wret = -374.586BTU
Qu = 1.204% 10°BTU
241
89
Turbine
Steam at 6500 kPa & 600 degC (point 2) Table F.
Hy = 365212 Spi= 7.1258 Pp := 6500-KPa
kg kgK
AC point 3 the pressure must be such that the steam has a condensation
Aemperature in feedwater heater 1 of 195 degC, 5 deg higher than the
temperature of the feed water to the boiler at point 1. Its saturation pressure,
corresponding to 195 degC, from Table F.1, is 1399.0 kPa. The steam at point 3
is superheated vapor at this pressure, and if expansion from P2 to P3 is
isentropic,
ky
His := 3142.6— Wy:= n-(H3 -H:
3 ig i= n-(H3— Ha)
kK
= Ho + Wy Hy = 3.244 1 Wy = ~407.6=
kg kg
From Table F. Hyo = 8290-4
kg
Simitar eal
lations are required for feedwater heater I
242,
At the exhaust conditions of 20 kPa, the properties of sat. liq. and sat.
vap. are:
KI ’
jig = 251.453-— Viig = 1.017-——
hig ke bg em
kr
§ = 0.8321-——
Sig = 08321
we find 17, then 18 is the mid-temperature hetween t7 and t(190 degC), and
‘that fixes the pressure of stream 4 so that its saturation temperature is 5 degC
hhigher. At point 6, we have saturated liquid at 20 kPa, and its properties from
Table F.2 are:
‘sat +273.15)K
Vo := Vig
Ikq. 7.24)
KI
Wpanp = 8.238 — AHg7 *= Wpump
‘B
We apply Eq, (7.25) for the calculation of the temperature change from point 6
to point 7. For this we need values ofthe heat capacity and volume expa
of water at about 60 degC. They can be estimated from data in Table F.
., . 272.0-2302 kd
Cp i= ar
10 kg-K
—4 1
B= 5.408 10° 4 cp = 41s
K kek
Solving Fg. (7.25) for delta T gives:
AH67 ~ Viig-(1 ~ B-Tsai)-(P2~ Po)
ATs San aeg ee rae ATg7 = 0.678K
ATe7 190 -t7
cat + tors + to+5
17 = 60.768
243
tg = 130.38 From Table F.1: Hg ss 54797
2
Hy = Hyig + AH67 ty = 125.38 To 2= (273.15 +t9)-K
kr
Hy = 259.691 —
kg
Atpoints 9 and 1, the streams are compressed liquid (P=6500 kPa), and we
find the effect of pressure on the liquid by Eq. (7.25). Values by
interpolation in Table Fl xt saturation temperatures 19 and f1:
om?
Veat9 = 1.065--
gm
mm
Vogt) = 1.142 Pyatst = 1255.1-kPa.
em
om om?
AVg = (1.075 — 1.056)-—— AV, = (1.156 ~ 1.128)
em em
1 AVo 1 4v,
Bo Tao aT Voat AT
et —31
= 892x104 = 1.226% 103
Bo z Bi 7
KI
Ho = Hsa.9 + Vsat9-(1 ~ Bor Ts){(P2 ~ Psat.9) Hy = 530.97
Ty 273.15 + 190)-K T) = 463.15K
AS
Hy := Hg, V. 1-By-Ty)}-(P2—Poat Hy = 810.089—
1 = Hgaa.1 + Vsat.i-(1 ~ Br T1)(P2~Psat.t) 1 =
Now we can make an energy balance on feedwater heater Ito find the
‘mass of steam condensed:
Hy) - Ho
my = —
H3 — Hig
O.11563Kg Ans.
24a
‘The temperature at point 8, #8 = 130.38 (see above) is the saturation temperture
in feedwater heater II. The saturation pressure by interpolation in Table F.1 is
273.28 kPa.
Isentropic expansion of stcam from the initial conditions to this pressure results
in a slightly superheated vapor, for which by double interpolation in Table F.2:
Then Hg = Ho + 9-(H's — Ha)
oat x 10
kg
‘We can now make an energy balance on feedwater heater II to find the mass of
steam condensed:
Hy =
(Hy = Hy)-1-kg ~ my (Hip - Hs)
Ha- Ag
ii’ 0.09971 Kg Ans.
my c=
‘The final stage of expansion in the turbine is to 20 kPa, where the exhaust is wet.
For isentropic expansion,
i S82-Stiq
xsi5 Hig + ¥'5-(Hyap ~ Hig)
‘ap ~ Stiq
x'5 = 0.889 2349.10
ke
‘Then Hy = Ho + n-(H's — Ha)
The work of the turbine is:
Weurbine = Wy-I-kg + (1-kg — my). (Hg - Hg)...
+(1-kg ~ my~ my)-(Hs— Ha)
Wrarbine = -936.2kI Qu (H2-Fi}rke Qu = 2.842« 105g
/Wrurbine + Wpump' Ike}
32¢ ns.
tH aM
248,
810
Isobutane: Te
8.1K Pe = 36.48: bar 0.181
For isentropic expansion in the turbine, let the initial state be represented by
symbols with subscript zero and the final state by symbols with no subseript.
Then
To = 533.15K Po
J
800-kKPa P= 450-kPa
As For the heat capacity of isobutane:
morK
6
Azle B ass 10"
To Po
Tos Tro = 1.3064 Pro = = Pro = 1.3158
w= 0 0=B 0
Use generalized second-virial correlation:
HRB(1.3064, 1.3158,0.181) = -0.868161 HRBo := -0.868161
SRB( 1.3064, 1.3158,0.181) = ~0.486134 SRBp := -0.486134
‘The entropy chang:
given by Eq, (6.83) combined with Eq. (5.15) with D
T= 0.8 (guess) SRB := 0.0 (starting value)
Given
r y 26 P
AS = RL A-tn(s) +] BT + C-T02| E*4) |e —1) nf 2),
L a) Po)
_+SRB- SRB
tau(SRB) := Find(z) T := tau(SRB)-To T= 452.5K
At the final P and this, evaluate SRB:
T P
= T,(T) = 1.1088 Pes Py = 0.1234
Te Pe
SRB(J_1088 , 0.1234,0.181) = ~0.073105 SRB := ~0.073105
T = tau(SRB)-To T = 454.52K TT) = 11137
Although it will make little difference, we now recalculate SRB at this femperature:
SRB(1.1137,0.1234,0.181) = -0,072162 SRB = ~0.072162
T == tau(SRB)-To T = 454.49K TT) =
137
246
A further iteration is clearly not warranted.
‘The enthalpy change for this final temperatu
the above T:
given by Eq. (6.82), with HIRB at
HRB(1.1137,0.1234,0.181) = -0.113300 HRB := -0,113300
repHi{s33.15,454.48, 1.677,37.853:1073,-11.945-107 60.0) = -1372.822
5
1372.822-K Abig := RICPH — AHig = -1.141x 10°
1cPH
mol
AB turbine “= AHig + R-Te(HRB ~ HRBo)
J
AHturbine = ~8852.4—— Wrurbine #
AHourbin
mol ‘urine
‘The work of the pump is given by Eq, (7.24), and for this we need an estimate of
the molar volume of isobutane as a saturated liquid at 450 kPa. This is given by
Eq. (3.63), The saturation temperature at 450 kPa is given by the Antoine
‘equation solved for t degC:
VP := 450-kPa_
Ayp = 1457100 Byp = 2606.775 — Cyp s= 274.068
Byp i
ts v = = (t+
fe i: t=34 T= (14273.15)-K
kPa T= 30715K
cm’?
Vo = 262.7 0.753
mol
02857 3
it)
Vig» ver a Vig = 112.36
mol
Wopump = Viig:(Po - P) a z
ipamp *= Viig-(Po Woump = 488.8—
mol
The low rate of isobutane can now be found:
ee 1000-kW ce
[Weurbine + W pum]
247
‘The enthalpy change of the isobutane in the cooler/condenser is calculated in
two steps:
a. Cooling of the vapor from 484.48 to 307.15 K
b. Condensation of the vapor at 307.15 K
Enthalpy change of cooling: HRB at the initial state has already been caleulated.
For saturated vapor at 307.15 K:
aus T, = 0.7526
Te
HRB(0,7526, 0.1234, 0.181) = ~0.265848 ARBsat := ~0.265848
1ePH454.48, 307.15, 1.677,37.853-107§, 11,945.10" 6,0.0) = ~2112.435
ICPH := ~2112.435-K Abiig := RAICPHL Alig = 1.756% it
mol
AH, = AHig + R-Te(HIRBygi ~ HRB) ‘AH, = ~18080
For the condensation process, we estimate the latent heat by Eqs. (4.12) and (4.13):
Tq = 261.4K Tn = 0.641
( (Pe
RT 1.092 inf =) = 1.013 ;
AH == SPs Ay = 2.118 10*§ >
0.930 = Tm mol
J
AH, AH, = -18378——
mol
Qdotout := mdot (AH, + AHp)
1000-kW
Qdotin = |Weurhine + Wpumg| mdot + |Qdoto, —
[Weursine + Wpume] | Qdotou] Span
Qdotou, = =4359KW Qdotin' = 5359kW Ans.
248,
8.11 Isobutane: T, := 408.14K. Po 15.36.48: bar o = 0.181
For isentropic expansion in the turbine, let the initial (inlet) state be
represented by symbols with subscript zero and the final (exit) state by
symbols with no subscript. Then
3400-kKPa P= 450-kPa— molwt =
For the heat capacity of isobutane:
pp ew 37.853:10 1.945.107
K
Po
Tyo = 1.0124 Pome Pro = 0.932
Use Lee/Kesler correlation for turbine-inlet state, designating values
by HRLK and SRLK:
HRLKo := ~1.530 SRLKo = -1.160
‘The entropy change is given by Eq. (6.83) combined with Eq. (5.15) with D = 0
t= 0.8 (guess) SRB = 0.0 (starting value)
Given
14+1)]
AS=R AnG)4[ ey ror? a | (c-1)-wof 2)
2), Po,
+SRB~SRLKo
tau(SRB) := Find(t) T := tau(SRB)-To 329.31K
At the final P and this T, evaluate SRB:
T
THE) = TCT) = 0.8069 Py = 0.1234
SRB(0.8069, 0.1234,0.181) = -0.19472 SRB := ~0,19472
T = tau(SRB)-To T= 33431K THT) = 0.8192
Although it will make little difference, we now recalculate SRB at this
temperature:
‘SRB(0.8192, 0.1234,0.181) = -0.18538 SRB := ~0.18538
T= tau(SRB)-Tp T = 334.07K Ty(1) = 0.8186
249
A further iteration is hardly warranted. The enthalpy change for this final
temperature is given by Eq. (6.82), with HRB at the above T:
HRB(0.8186, 0.1234 0.181) = -0.21941 HRI
0.21941
3
1cpH(413.15,334.07, 1.677,37.853:10" 3, -11.945-107§,0.0) = —1118.642
ICPH s= ~1118.642-K AHigi= RICPH Hig = -9.3x ws
mol
Abiurbine ‘= AHig + R-Te(HRB - HRLKo)
J
Alyurbing = ~4853.6— Wrurbine = AH turbine
mol
The work of the pump is giv
molar volume of saturated-
calculated in Problem 8.10:
Eq. (7.24), and the required value for the
isobutane at 450 kPa (34 degC) is the value
J
Wy = Viiq (Po - P, W, = 331.462 —
pamp= Vig (POP) Wyamp = 3814625
For the cycle the net power OUTPUT is:
75 KS Waot z= -mdot (Warne + W pump)
Wedot = 5835KW Ans.
The enthalpy change of the isobutane in the cooler/eondenser is calculated in
‘two steps:
a, Cooling of the vapor from 334,07 to 307.15 K
b, Condensation of the vapor at 307.15 K
Enthalpy change of cooling: HRB at the initial state has already been
calculated. For saturated vapor at 307.15 K it was found in Problem 8.10 as:
HRBgat = -0.2658
icphi(334.07,307.15, 1.677,37.853-10",-11.945-10" §,0.0) = 338.775
ICPH := ~338.775-K AHig = R-ICPH AHig = -2817 4
mol
AHg := AHjg + R-Te(HRBsa ~ HRB)
250
For the condensation process, the enthalpy change was found in Problem
8.10:
‘AH = -18378- Qdotout := mdot-(AF, + AHp)
Qdotou = -27552KW Ans.
For the heater/boiler:
Qdotin = Waot + |Qdotoutl ‘Qdotin = 33387KW Ans.
Weot ge
— FONTS. Ans.
Qdotin thet
‘We now recalculate results for a cycle for which the turbine and pump each
have an efficiency of 0.8. The work of the turbine is 80% of the value
calculated above, ie,
Wiurbine := 0.8 Wrurbine Wiarbine = ~3883—
mol
‘The work of the pump
Wpump J
Wopump = Wamp = 414.3—
aa 8 mol
Waot := -mdot(W'turbine + W'pamp) - Wedot =-4476KW Ans,
The decrease in the work output of the turbine shows up as an increase
in the heat transferred out of the cooler condenser. Thus
Qdotout = Qdotout + (Wrurbine~ Wrurbine)-mdot
Ans.
The increase in pump work shows up as a decrease in the heat added in the
heater/boiler. Thus
Qdotin = Qdotin~(Wump— Wpump}-mdot Qt, = 33280kW. Ans,
Wdot
Qdotin
25
8.13 Refer to Fig. 8.10.
8d
Pe := I-bar Pp := 5-bar a
By Eg. 8.290):
=
Vo (Pp)"
Ne ¥o) Ans
Vo \Pe}
Eq. G.29b): Qpa = 1500-500"
mol
Qpa = Cp{Ta Ta = 515.845K
Ratio
Tue
cy
9
Ans.
252
8.16
heating
. (combustion)
P (par)
A
cooling
v
Figure shows the air-standard turbojet power plant on a PV diagram.
Ta := 303.15-K Te = 1373.15-K
By Eq. (7.22)
a
(pp)? 3
Wan= Cp-Ta||[—= | -1] = Cp-Ta-ler? 1)
(Pa)
R
G 2
Pp)" 7
Wen ® CeTe}]— | - 1} © Cp-Teler? = 1
Pe,
where cr is the compression ratio and er is the expansion ratio. Since the
two work terms are equal but of opposite signs,
5 (guess)
(3 ) 62)
i 2 7
Given Toler” — 1) = -Taler 1) er = Find(er)
er = 0.552
253,
cp
(7.18): Tp2 Te!
(7.18); D of )
By Ee
This may be written: Tp := Te-er
TSbar y= 0.8
817 Ta:=305-K = Pass LOSbar Py
Assume air to be an ideal gas with mean heat capacity (final temperature by
iteration):
McpHi(298.15,582,3.385, 0.575.10" 3,0.0,-0.016-109) = 3.5998
Cimgir = 3.5998-R Cpr = 29.929 ——
254
Compressor:
fr _R
Comayts |( Pp) 5
Wai 22 | 1) Woyie = 8291 10°§
7 IPa mol
Tate tp = 582.009K
Tass 7
B At Comair B
Combustion: CH4+202=C0O2+2H20
Basis: Complete combustion of 1 mol CH4. Reactants are N mol of
air and Imol CH4,
Because the combustion is adiabatic, the basie equation is:
AH + AHy98 + AHp = 0
For AH_R, the mean heat capacities for air and methane are required.
‘The value for air is given above. For methane the temperature
change is very small; use the Value given in Table C.1 for 298 K:
427 R.
The solution process requires iteration for N. Assume a value for N
until the above energy balance is satisfied.
@) Te
Aly = Cpmgie-N-(298.15 = 582.03)-K + 4.217-R-(298.15 — 300)-K
1000K oN
7.638 (This is the final value after iteration)
AHp = ~4.898 x 10°
mol
The product stream contains:
1 mol CO2, 2mol H20, 0.79N mol N2, and (0.21N-2) mol 02
(oa 5.457 1.045 L137
3.470 1.450 0121 |
-10 10°
3.280 0.593 0.040
3.639, 0.506 0.227,
255
8.638 A: Yai B
>
A= 198517 B= 0.036 D
B,D yw
~1.387% 10°
Mcphi(298.15, 1000.,198.517,0.0361 ,0.0,-1.3872-10°) = 221.483338
Cpmp = 221.4833-R AHp := Cpmp:(To - 298. 15K)
AHp = 1.292 10°
mol
From Ex. 4. Hao = -802625
mol
AH + AH29g + AHp = 3. os (This result is sufficiently close to zero.)
mol
Thus, N = 57.638 moled Of aif per miole of methane fadk “Ans.
Assume expansion of the combustion products in the turbine is to 1(atm),
iLe., to 1.0133 bar:
Pp = 1.0133bar Po = 7.Sbar
The pertinent equations are analogous to those for the compressor, ‘The
mean heat capacity is that of the combustion gases, and depends on the
temperature of the exhaust gases from the turbine, which must therefore be
found by iteration, For an initial calculation use the mean heat capacity
already determined. This calculation yields an exhaust temperature of about
390K. Thus iteration starts with this value. Parameters A, B, and D have
the final values determined above.
mcpti( 100,343.12, 198.517, 0.0361 ,0.0,1.3872-105) = 222.356026
Cpm := 222.356-R For 58.638 moles of combustion product:
one
58.638-CpmT see
Ws astonty| 2] : ws = 1.214% 10°
” LVPe, mol
Ws .
To:=Tet Gm Tp 2343123K° (Final result of iteration.) Ans,
nl
256
Wane = WS + W5aieN
(J per mole of methane)
Parts (b) and (¢) are solved in exactly the same way, with the following
results:
()) Te = 1200 N= 3748 Winet = -7.365:10° Ty = 343.123
7519-10° Tp = 598.94
(©) Te 1500 N= 24.07 Wepet =
287
Chapter 9 - Section A - Mathcad Solutions
92 TH
(204.273.15)K 293.15K
S3.15K,
To = (-20+273.15)K Tc
Odore := 1250002.
day
Te
Scam = 03) 0 0.6-0¢anot
dot
BNE 9.2) Wot = 0.381 kW
°
twat Cost = 267183583 ns,
. °
9.4 Basis: 1 Ibm of tetrafluoroethane
The following property values are found from Table 9.1:
0.09142
State 1, Sat. Liquid at TH: HI := 44.943 $1
State 2, Sat. Vapor at TH: H2 := 116.166 $2 »
State 3, Wet Vapor at TC: Hliq := 15.187 Hvap := 104.471
iq := 0.03408 Svap := 0.22418 P4
(a) The pressures in (psia) appear above.
(b) Steps 3-2 and 1-4 (
Thus by Eq. 6.7:
State 4, Wet Vapor at TC:
ig. 8.2) are isentropic, for which $3=S2 and S1=S4
— Siig
Svap— Sliq
971
(©) Heat addition, Step 4
HB := Hliq+33-(Hvap—Hlig) 4 <= Hig + x4-(Hvap — Hlig)
H3 = 101.888 H4 = 42.118
Q43 := (H3 - Ha) 043 = $9.77 (Bau,
258
(d) Heat rejection, Step 2-1:
Qai = (HI -H2) (Bru/lb,,)
© Wiss 0
W32 := (H2 - 13)
Wi4:= (H4-HI)
43
oe
a. W14 + W32
a= $219
Note that the first law is satisfied:
2Q = Q21 +.Q43 EW i= W324 Wid TQeEW=0
Te = 298.15-K Ty = 523.15:K (Engine)
Te = 273.15-K ‘Ty = 298.15:K (Refrigerator)
Te
By Eq. (5.8): x
u
Ncamot = 0.43
By Eq. (9.3):
By definition:
But
Whence Quy
Given that: 4) = 0.6-NCamoe
Qc
Qu=
269
(a) Qc
mk Ws 1LSkW
see
ee
© 5 2.667- Ans.
Ww .
Ans.
©) Qu = Qc+W Qu ee
(© oF Ty = (40+273.15)K Ty = 313.15K
Ta-Te
o \
To := Ti ; | Fe = 227.75K Ans.
ori) -
or -45.4 degC
‘The following vectors contain data for parts (a) through (e). Subseripts
refer to Fig. 9.1. Values of H2 and S2 for saturated vapor come from
Table 9.1.
(489.67 0.79 600
479.67 0.78 500
469.67 |-rankine y=) 0.77 Qdote =| 400 Be
459.67 | 0.76 300 a
449.67, (0.75 200,
107.320 0.22244)
105.907 0.22325
H =| 104.471 | BY s2=| 022418 |B
I Top tankine
103.015 0.22525.
101.542 tase
From Table 9.1
: Bu
Ty s= $39.67: rankine Hyg = 37.978. — for st. liquid
Tom
S528) (isentropic compression)
260
‘The saturation pressure at Point 4 from Table 91 is 101.37(psia). For
isentropic compression, from Point 2 to Point 3', we must read values for
the enthalpy at Point 3° from Fig, G.2 at this pressure and at the entropy
values $2. This eannot be done with much accuracy. The most
satisfactory procedure is probably to read an enthalpy at S~0.22 (H=114)
and at S=0.24 (H#=126) and interpolate linearly for intermediate values of
H, This leads to the following values (rounded to 1 decimal):
115.5
116.0
nes Be Hg s= Ha + AHy3
bee 1
1172 Hy = Hy
TERE (24.084) 273.711
30,098 276.438
KW k
Hy = 883372 V3 =|36337 (© ty =| 279.336 |
kg kg kg
43.414 283.026
50.732. 286.918,
—
mdot := xtc Ans.
= aon Ans.
Qdoty Ans,
——>
Wdot := (mdot-AH3) Ans,
261
Qdote
Wdot
©Camot Ans.
Subscripts in the following refer to Fig. 9.1. All property values come from
Tables F.1 and F.2.
Tp = (44273 15)-K 14 = G44273.15)-K 11 = 0.76
Qdote 1200. Hp = 2508.94 sp = 9.0526 4.
sec kg kek
kr : ;
Hae 2.4 (isentropic compression)
The saturation pressure at Point 4 from Table F.1 is 5.318 kPa. We
must find in Table F.2 the enthalpy (Point 3') at this pressure and at the
entropy S2. This requires double interpolation. The pressure lies
between entries for pressures of J and 10 kPa, and linear interpolation,
with P is unsatisfactory, Steam is here very nearly an ideal gas, for
which the entropy is linear in the logarithm of P, and interpolation must
be in accord with this relation. The enthalpy, on the other hand, changes
very little with P and can be interpolated linearly. Linear interpolation
with temperture is satisfactory in either case.
‘The result of interpolation is
Hy - Hi
wy = 28147- AHo3 : Hy = Ha
kg a
Hos = 402.368 2
bs ie
262
91
3k
Hy == Ho + AHos Hy = 2911x107
KE ns,
see
mdot= 0:
it
Qdotjy := mdot-(H4- H3) Qdoty = <1404 = Ans.
Weot = 204kW Ans.
ow Sete 55,881 Ans.
Wdot
sae rae canst 39238 Ans.
amet 'T4-T2 seria ~
Parts (a) & (b): subscripts refer to Fig. 9.1
5 (Btuy(s)
At the conditions of Point 2 {1
P = 14.667(psia)} for sat. liqui
5 degF and
and sat. vapor from Table 9.1:
Btu Btu
505 Hyap = 100.799" Hy = Hyap
ibm bn
Hig 2=
283
Bu Btu
Stig = 0.01733 = 0.22714.
ig Tbprrankine Svap 7 Tprrankine
For sat, liquid at Point 4 (80 deg):
Bu Bu
Ha := 37.9782 S4:= 0.07892.
‘ ibm ibmrankine
(a) Isenthaipic expansion: Hy := Hy
Buu Qdote aes
dot — mdot 0.0; Ans.
Qin 5 Ho-Hi os
(b) Isentropie expansion: Si=S4
BTU
Hhig + x1-(Hyap— Hig) Hi = as
(©) The sat. vapor from the evaporator is superheated in the heat
exchanger to 70 degF at a pressure of 14.667(psia). Property values
for this state are read (with considerable uncertainty) from Fig. G.2:
75-3
Thin
Qdote
Hoa - Ha
(@)_ For isentropic compression of the sat. vapor at Point 2,
S3 = Syap and from Fig. G.2 at this entropy and P=101.37(psia)
1133-2 Eq. (9.4) may now be
Ibm applied to the two cases:
In the first case Fi1 has the value of Hd:
Hg
Hp - Hy
H3 Hp
In the second case H1 has its last calculated value [Part (b)]:
Hy)
He
3:7659° Ans.
In Part (c), compression is at constant entropy of 0.262 to the
final pressure. Again from Fig, G.2t
Bu ie (Last caleulated
H3 := 138 ca Wot := (H3 - H2,)-mdot value of mdon)
Wdot = 1.289 ou
see
dot
y= Jeote| 31871 Ans.
Waot se
9.12 Subscripts: see figure of the preceding problem.
‘At the conditions of Point 2 [sat. vapor, t = 20 degF and P = 33.110(psia)]
from Table 9.1:
Btu Buu
Hi := 105.907 $y = 0.22325-—B
7 om 7 Ibgrankine
At Point 2A we have a superheated vapor at the same pressure and
at 70 degF. From Fig. G.2:
Bu Bu
Hoa = 116-2 Spq = 0.2435.
1 fbn ae Ibjetankine
For sat. liquid at Point 4 (80 degF):
Br
Ha = 37.9782 Sa = 0.078922
Thm nt
Energy balance, heat exchanger:
Hy = Hy~ Haq +H H = 2788550
Qdote
Ibm
mdot = 25.634—
H2-Hr sec
mdot
9.13
For compression at constant entropy of 0.2435 to the final pressure of
101.37(psia), by Fig. G.2:
Bu H3—H2a
Hy = 127-—— AH om,
tm a n
Bru
Woot := mdot-AH comp AHoomp = 14.6677 —
m
396.66KW Ans.
If the heat exchanger is omitted, then H = H4.
Points 2A & 2 coincide, and compression is at a constant entropy of 0.22325
to P= 101.37(psia).
Qdate
mdot
Ho -Ha
Btu
Weot := mdot-AHeomp AHlcomp = 1345755
ila Bp
mdot = 29.443 =—= = 4i8.032kW Ans,
Subscripts refer to Fig. 9.1.
At Point 2 [sat. vapor @ 10 degk] from Table 9.
Ho= rosa 2 = 022418 BY
S2
H values for sat. liquid at Point 4 come from Table 9.1 and H values
for Point 3° come from Fig. G.2. The veetors following give values for
condensation temperatures of 60, 80, & 100 degF at pressures of
72.087, 101.37, & 138.83(psia) respectively.
31.239 1133
Bu tu
Hy =] 37.978 | Wy s=] 1168 aa Hy = Hg
44.943) ™ n93)™
266
(@) By Eq. (9.4):
a ‘Ans.
an = BoB Si AH = H3-H;
= ince = H-
») OTs 3 — Ho
Eq. (9.4) now becomes
—>
-H
paar ‘Ans.
AH
9.14 WINTER Ty = 293.15
WINTER Waot := 1.5
Qdoty = -0.75-(TH- Te)
House :
Ty 7293.15 K iQui Wdor_ _ Ta-Te
[Qdots] Ta
Te :=250 (Guess)
Given
Wdot Tu-Te
267
SUMMER Te = 298.15
SUMMER Qdote == 0.75-(Ty = Fc)
Wdot _ TH-Te
Qaote c
Tu i= 300 (Guess)
Given,
Weer _ Ta~Te
0.75-(TH-To) Te
Ty := Find(T 4)
House
293.15 K
Maximum t= 49.42 degC
9,15 and 9,16 Data in the following vectors for Pbs. 9.15 and 9.16 come from
Perry's Handbook, 7th ed.
1033.5 1186.7
fe. bck Ho := 284.7. = His = ey
785: kj 1056.4) kg
By Eq. (9.8): 2
ly Eq. (9.8): 0351
0.17 )
Ans.
9.17 Advertized combination unit:
TH == (150+ 459,67)-rankine Te := (30 + 459.67)-rankine
‘Ty = 609.67 rankine Te = 489.67 rankine
Btu Ta-Te Btu
Weasnot = 12253—
$= 50000. Weamot '= Qc
Qc te ‘Camot ‘= Qc" Tc "
268
9.18
919
Bu
S:Weamot Wi = 18380
Wis
This is the TOTAL power requirement for the advertized combination unit.
‘The amount of heat rejected at the higher temperature of
150 degF is
Btu
Qu = Wr+ Qe Qu = 683805"
For the conventional water heater, this amount of energy must be supplied
by resistance heating, which requires power in this amount.
For the conventional cooling unit,
Ty = (120 + 459.67)-rankine
Bi
Weamot = QC Wow = 919052
Work := 1.5-Weamot Work = is7ss St
t
The total power required is
Wit = Qu + Work
i ee NO CONTEST
To := 210 Te = 255 Ty := 305
By Eq. (9.3):
c ‘c
oy #= 0.65: oy = 0.65.
Ty-Te 7 Ta-Te
Qe
wie ce
1 on
Define r as the ratio of the
actual work, WI+ WIL, to the
Carnot work:
This problem is just 2 reworking of Example 9.3 with different values of x.
It could be useful as a group project, i
269
Chapter 10 - Section A - Mathcad Solutions
10.1 Benzene: Ay i= 13.8594 By == 2773.78 Cy = 220.07
Toluene: Ag = 14.0098 By = 3103.01 C2 = 219.79
BL Bo
AH Ap
nas i
Psaty(T) =e 8 -
KPa Psatp(T) :=€ kPa
(a) Given: xj = 0.33 T:= 100-deg@ Guess: yy := 0.5 P= 100-kPa
Given xj-Psaty(T) +(1- x1)-Psatg(T) =
xy-Psaty(T) = y-P
Find(y, ,P) y= 0545 Ans. B=109.119kPa Ans.
(b) Given:
OO-degC Guess: xy := 0.33 P= 100-KPa
Given xy-Psat) (T) + (1 —x1)-Psatg(T) =
xyPsaty(T) = y-P
XL
:= Find(xy ,P)
() (oF)
(© Given: x)
Ans. P= 92,042kPa Ans.
0.33 P= 120-kPa Guess: yj i= 0.5 T
(00-degC
Given xy-Psaty(T) +(1 —xy)-Psatg(T) = P
xr-Psaty(T) = yP
ind(v1,1)
Ans, ‘T's 103,362dekC Ans.
(@) Given: yy
33 P= 120-kPa Guess: xy 100-dege
Given xp-Psaty(T) + (I~ x1}-Psatg(T) = P
xp-Psaty(T) = ypP
(x1)
x .
{ : | = Find(xy ,T) Xp=0.173. Ans. T= 1090.161degC Ans.
TT) i
(©) Given: l0S-degC P := 120:kPa Guess: xj = 0.33 yy = 0.5
Given xp-Psaty(T) + (1 —x1)-Psatg(T) = P
xy-Psaty(T) = ypP
Find(xy.y1) 3} =0383° Ams. 20.485 Ans.
oO 2 = 0.33 x1 = 0.283 yi = 0.485
Guess: L:=035 Vir0s
Given 22 Lx t Ve
L+Vel
ind(L,V) Vapor Fraction: = =.0.93] Ans.
(i
()-
(g) Benzene and toluene are both non-polar and similar in shape
and size. Therefore one would expect little chemical interaction
between the components. The temperature is high enough and
pressure low enough to expect ideal behavior.
Liguid Fraction: 1, = 0.769. Ans.
Fa]
10.2 Pressures in kPa; temperatures in degC
(a) Antoine coefficients: Benzene=1; Ethylbenzene=2
3.8594 By = 2773.78 Cy == 220.07
4.0045 Bp = 3279.47 C2 = 213.20
Psat) (T) Aye]
sat (T) := exp] Ay — a
(7) PAI — a qi)
Psatz(T) := exp} A2 - —
oe
P-x-y diagram: = T= 90
P(x1) s= xy-Psaty(T) + (1 —x))-Psat(T)
T-x-y diagram: — P' := 90
‘Guess t for root function: t := 90
1(xp) == root{_x1-Psaty (t) + (1 — x1)-Psata(t) - Pi]
Xy-Psaty(T(x;))
Bani) e Uw) Bastia)
x1 3= 0,0.05..1.0
ts
140
130
120
ho
no os 1 5 05 1
xtyi(u) xryi(s)
272
(b) Antoine coefficients: 1-Chlorobutane=1; Chlorobenzene-2
Ay == 13,9600 C1
C2
Ag = 13.9926
= xy Psat (T) + (1 —x4)-Psata(1)
‘T-x-y diagram: —P':= 90.
Guess t for root function: t := 90
T(x) = root] xy-Psaty(t) + (1 x1)-Psatgit) ~ P’,t]
xa Psat (T(x)
beats Gey)) (xy) Psaty
0,0.05..1.0
(rs)
160
11333)
P(x)
P(x)
66.67 |
20 05 | 0 05 1
xtoyu(xa) x1 ¥ihs)
273
10.3 Pressures in kPa; temperatures in degC
(a) Antoine coefficinets: n-Pentane=1; n-Heptane=2
Ay = 13.8183 Bi cy
‘Ag i= 13.8587 Bos
Psaty(T) = ofa -
Ti 55 P= 101418
Psaty(T) + Peat)
one
Since for Raoult's law P is linear in x, at the specified P, x1 must be
7 xy-Psaty (T)
apes oo
For a given pressure, z1 ranges from the liquid composition at the bubble
point to the vapor composition at the dew point. Material balance:
xp(i-V)typV
V is obviously linear in z
vila) os
‘ot
274
(b) At fixed T and 21, calculate x1, y1 and P as functions of fraction vapor (V)
( Psat (T) + Psat2(T)
v ae 7]
Given Three equations relate x1, y1, & P for given V:
p= x-Psaty (T) + (1 ~ x)-Psatz(T)
yp = Psat; (T)
215 (1-V)x+ Vey
F(V) == Find(x,y,p)
xi(V) = £1 iV) PCV) = £(V)3
Plot P, xt and yl vs. vapor fraction (V) V:=0,0.1..1.0
100 -
10.4 Each part of this problem is exactly like Problem 10.3, and is worked in
exactly the same way. All that is involved is a change of numbers. In
fact, the Mathcad solution for Problem 10.3 can be converted into the
solution for any part of this problem simply by changing one number, the
temperature.
10.7 Benzene: Ay ss 13.8594 By := 2773.78 Cis
Ethylbenzene Ay := 14.0045 By := 3279.47 C2
Bi
AI
icy
deel kPa Poatg(T) =e OS kPa
10.8
10.9
7 Guess: T
(afGiven: x)= 035 yt N6-degC P= 132-kPa
Given xy-Psat}(T) + (I —x1)-Psata(T) = P
x1-Psaty(T) ® yy-P
©) sa Find?)
= Find(P, P
i :
For parts (b), (e) and (d) use the same structure. Sef the defined
variables and change the variables in the Find statement at the end of the
solve block.
34.01dexC Ans.
-206.95kPa Ans.
a
) 111,65-deg_C P= 117.84-kPa
(© T= 91.15-deg_C P= 65.66-kPa
@) T= 72.12-deg_C P= 35,57-kPa
‘To calculate the relative amounts of liquid and vapor phases, one must
know the composition of the feed.
To increase the relative amount of benzene in the vapor phase, the
temperature and pressure of the process must be lowered. For parts (c)
and (d), the process must be operated under vacuum conditions. The
temperatures are well within the bounds of typical steam and cooling water
temperatures.
13,8594 2773.78 220.07
(1)= benzene
Q)=toluene A z= | 14.0098] B= / 3103.01) C:=| 219.79
(3)= ethylbenzene 14,0045, 3279.47 213.20
Psat(i,T)
P
276
1 Eq. (10.17)
0.441)
(b) T= 110-deg_C v y=] 0.333
J
P= 100-kPa aaa
0.509)
() T= 110-deg C V= 0349 0.312
P= 110-kPa ane
0.573
(@ T* 110-deg_C V = 0.143 y= | 0.283
1
P= 120-kPa —
10.10. As the pressure increases, the fraction of vapor phase formed (V)
decreases, the mole fraction of benzene in both phases increases and the
the mole fraction of ethylbenzene in both phases decreases,
14.3916 B 2795.82 c (23° 00
14.7258 3271.24) ~~ (241.85
a7
10.11 (a) (1) = acetone
(2) = acetonitrile
= 115:kPa,
_ Psat(i,T)
eee.
Eq. (10.17)
‘V:=Find(V) -¥ =,0,056 Ans.
irk;
Eq. (10.16) ic Ans.
bee _ 1+V-(ki- 1)
yeP
x= Ans.
Psat(i,T)
ae Ans.
a
(0) x) = 0.286 yt = 0.678 V = 0.545 r= 0.739
(© x1 = 0.182 yi = 0317 Vv = 0.504 r= 0.638
(@) x1 = 0.351 yi = 0.706 v= 0.420 r= 0.594
10.13 Hy = 200-bar Psatg = 0.10-bar Piss L-bar
Assume at 1 bar that the vapor is an ideal gas. The vapor-phase fugacities
are then equal to the partial presures. Assume the Lewis/Randall rule
applies to concentrated species 2 and that Henry's law applies to dilute
species 1. Then:
278
10.16
yrP = Apxy yorP = x2-Psaty PE ypP+ypP
P= Hy-xy +(1~ x1) Psat
Solve for x1 and yt:
EE P — Psatz Hpxy
1 A — Psat ms
5450201077
WERE Detar oe y= 09, Ans.
Pressures in kPa
Psaty := 32.27 Psaty := 73.14 A: 067 21 = 0.65
11 (s1.%2) = exp( Anz?) rab 2) = exp(Axr))
(x12) == x1-y(x189)Psatn + x3-73 (01 .x9)-Psaty
(a) BUBL P calculation:
2
(x12)
Pout
DEW P calculation:
Psaty + Psat
2
Guess:
Given yeP" = xyyj (xr, -x1)-Psaty
P= xy-yi(xq,1 —x1)-Psaty
(1 —x1)-¥9(x1 1 ~ x1)-Psaty
(x
= Find(x;,P’) Puew = 43.864 © Ans.
\Paew,
The pressure range for wo phases is from the dewpoint to the
bubblepoint: From 43.864 to 56.745 kPa
279
(b) BUBL P ealeulation:
XI
xeri(ais) x) Pst
The fraction vapor, by material balance is:
u-X1
yilsi) = x1
(©) See Example 10.3(¢).
892 Ans.
v=0379 PR
71 (0, 1)-Psat Paty
= O21 =
721 ,0)-Psat
Since alpha does not pass through 1.0 for 0 At
jen Fo =Find(T) T2°376:433 ns.
xp-yyPsaty (1)
serene 0.137bar Ans.
‘The romaining probloms for this chaptor have boon eclved on MS-EXCEL 2000
We givo the reculting spreadsheets.
Problem 10.25
a)BUBLP — T=S0F(S1.41C)
P=200 psia ——~P=250psia—«Pa215 psia (14.824 bar) ANSWER.
Component oxi Ki yieKitxi KI yisKitxl Ki yiPKined
methane 0.100 5600 0560 4800 0400 5150 0515
‘ehylene 0500-0700 0250 0575 0.288 0650 0.225
‘ethane 0400 0445 0178 0980 0152 0420 0168
SUM= 1.088 SUM= 0900 SUM= 1.008 close enough
bDEWP TOF ESH.ATC)
Pat90 pela P=200 psia (19.79 bar) ANSWER.
Component yl Ki xiii KI xy
mettere 500 $800 0.085 S600 0089
ethylene «0250 0730 0342 0700 0.367
ebane 0250 0460 0543 0445 0562
SUM= 097! SUM=1.008 close enough
)BUBLT —P=250 psa (17.24 bar)
T=50F T=60F T2587 F (49.44 0) ANSWER
Component xi Ki yiekixi Ki Ki yiekixd
mettane 0.120 4.900 0582 4600 0552 4700 "0564
etyiene 0400 08800272 ozs 0818 0246
ethane 0.450 0450 0216 0182 o4c5 0108
SUM= 1.076 0962 SUM= 1.004 close enough
@)DEWT — +250 psia (17.24 bar)
Te40F Ts 50F T= 45 F (27336) ANSWER
Component yi Ki xeyiKi Ki xinyKI KI xieyiki
menane 0430 6200 0.082 4800 0083 5050 0.085,
ayene 0360 0.800 0450 0680 0520 9740 0486
thane 0210 0820 0404 0450 0467 0485 0433
SUM= 0937 SUM= 1.084 SUM= 7.005 close enough
284
Protlern 10.25,
a)BUBLP — T=600(140F)
200 psa 60 oslo x80 psia (5516 bar) ANSWER
Component xi Ki yikrxi KI yleKitei Ki yieKitad
ethane 10 2015 0202 6800 0880 4.950 0405
propane 020 0620 0124 2050 0410 1475 0205
Tsobutene «030-0285 GOTT 0780 0234 0.560 0.168
Bopentene 0400071 0028 0205 0.082 0.12 0088
SUM= 0430 SUM= 1405 SUM= 1.006 close enough
byDEWP —T=60.C (140 F)
P80 psia P80 psia psla (3.585 bay ANSWER
Component yi Ki xisylKi KI XIYUKI KI xinyiK
‘thane 048 4950 0097 6.800 0071 8600 0073
propane: 025 1475 a169 2050 0.122 2.000 0126
Isobutane «0150580 0.268 0780 012 0760 0107
isopentare «0.12«012«1.000 «0.208 0585 0195 0615
SUM= 1.534 SUM= 0970 SUM= 1.010 close enough
yBUELT 5 bar (217.55 psa)
220F Ta150F 145 F (62.78 C) ANSWER
Component xi Ki yiskitxi Ki yieKi'xi Ki yinkital
ethane o14 5350 "0749 3.800 0532 3700 0518
propane 073 2500 0325 1525 0108 1475 0.102
ssobutane «02514750368 O78 0.180 O70 0180
ssopentare «048057 ORTH «OFT 01300250120
SUM 1.716 SUM= 1050 SUM= 1.010 close enough
) DEWT 15 bar (217.58 psia)
Te150F TeUd5F TH1eB F (64.440) ANSWER
Component yi Ki Ming Ki eyK Ki xinyikh
ethare 042 3800 0111 3700 0114 amon Ott
propane 030 4525 0197 1475 0.203 1.500 0.200
isobutane «O15 0760-0187 0.720 0.208 0.740 0203
‘sopentane «0.19«O.27OaB1 028 0.820 026 0500
SUM= 0.986 SUM= 1045 SUM= 1.013 close enough
285
Problom 10.27,
FLASH T=80F (14.81 C) 250 pala (17.24 bar)
Fraction condensed
855 120145 ANSWER
Component zi vi xiFyimi
ethane 050 0875 0.058,
ehane 010 0.508, 0.052
propane 020 0787 0275
‘butane 020 0.529 08:6
41000 SUM= 1.001
Problem 10.28,
First calculate equilibrium composition
T=95.C (203 F)
P=80 psia P65 psia P69 psia (4.83 bar) ANSWER.
Component xi KI oyirKinxi KI ikital KE yiFKx
butane 025 225 0565 27 067s 26 08s
nhewre «O75 O48 0.3375 081 0385 0.49 0.3675
‘SUM= 0.9000 SUM= 1.0575 SUI
= 110005 Clase enough
Now calculato liquid fraction from mole balances
yi= 0633,
ANSWER L= 0.347
Problor 10.29
FLASH P= 200 atm (29.39 psta)
T= 200 F (99.3.0)
Fraction condensed
v= 0.360 [50st ANSWER
Component zi Ki yi ai=yuK
pentane 0.252.250 0378 0.168
ntwxene 0.45 1.000.450 0.450
mepiane §— 0.300.450 0.172 0.382
SUM= 1.000 SUM= 1.000
286
Problem 10.30
FLASH Te40 c (104 F}
Fraction condensed
v=0.0
Pet10 psia
Component zi Ki yt
thane 015 8400 0220
propane 035 1900 0482
butane 080 510 0398
SuM= 1.053
Problem 10.31
FLASH TsTOF (21.110)
Fraction condensed
v= 0.20 te oo
P50 psia P40 psi
Component zi Kyl xieyiK KI yl
ethane 001 7400 0032 0004 9200 0035
propane 0.05 2400 0.084 0038 3000 0.107
outane 050 0925 0470 0508 1150 0.558
butane 0.44 080 0312 0472 aid 0370
SUM= 0907 1023 SUM= 1071
287
xy
0.045,
0248
oot
os82
xisyih
‘0.004
0036
0.485,
0487
082
ANSWER
P=120 pala (8.274 bar)
Ki yl ey
4360 0210 0047
11620 0413 0255
0525 0357 0.800
SUM= 0.999 1,001
ANSWER
Pag psia (3.024 bar)
Ki yl dey
500 00% 0.008,
2700 0101 0.037
41080 0524 0494
074 0343 0484
‘SUM= 1.002 1.000
Problom 10.32
FLASH
Component
methane
ohare
propane
eutane
‘Component
methane
ethane
propane
rebutane
‘Component
methane
ohare
propane
butane
T=156(5F)
P=300 pela
‘Ve 0.1858
aK oy
030 6600 0,908
040 0820 O08
030 0200 o070
030 ood 07
‘SUM= 1.078
0.30
o10
030
030
Target: yi-0.8
Leases
xinyiki
162
0.103
0382
036
SUM= 0.962
Ls 0.6850
alsyiKi
0.973
088
0.376
0424
suM= 0.960
‘P=ZTO psia (18.616 bar)
Ve 0.2535,
Kyi
8200 0.02
0.900 0.082
0.230 0.088
0.0405 0.020
SUM= 1.000
L=07465 ANSWER
xeyiKi
429
0103
0373
0.395
SUM = 1.000
288
Problem 10.33,
First cafculate vapor composition and temperature on top tray
BuBL. P=20 psia
rear Ts60F 1°69 F (20.86.C) ANSWER
Component xi KI yiekitxl KI ying) KI yikitet
p-butane 050 1575 0788 1350 0875 1550 O75
pentane «95004500225 0.360 0.180 0440 0220
SUM= £013 SUM= 0855 SUM 0.896 close enough
Using calculated vapor composition from top tray, calculate composition out of condenser
Flash calculation P=20 psia (1.378 bar)
60 Le 0.s0
T=70F ANSWER
Component zi Kyl xisyiK
n-butane 078 1575 0843 0402 1350 0890 0.680
pentane «02204500137 0308 0360 0115 0328
SUM= 1.085 0.905 SUM= 1.007 0.983
Problem 10.34
FLASH T40 6 (104F)
v= 0.80 Le oso ANSWER
P2280 sia 'P=325 psia (7.928 bar)
Component 2i yl xing KE yi aeyK KI liegt
methane 00 7.900 0768 0.097 11000 o7a6 aor! e400 O772 082
rebutane 050 0235 0217 08a D200 0253 OB7! 0205 O24 914
SUM= 0985 7.021 SUM= 1038 0.943 SUM= 0996 1.006
close enough
289
Chapter 11 - Section A - Mathcad Solutions
For an ideal gas mole fraction = volume fraction
€02 (1): x1 0.7
N2(2): x2 3503
AS = R/S" xirln(s)
For a closed, adiabatic, fixed-volume system, AU =0. Also, for an ideal
gas, AU = Cv AT. First calculate the equilibrium T and P.
ny? i= 4-mol Twa = ((75 +273.15)-K]
nar := 2.5-mol Tar = (130+273.15)-K
TN2 = 348.15K Tar = 403.15K
AN?
Mtotal ‘= NN2 + NAr XL 2 —
Total Dtotal
x1 = 0.615 x2 = 0.385
Cyn2
Cpar=CvartR — Cpn2=CuntR
Find T after mixing by energy balance:
T2+Ta
pie Dna Tar
z (guess) Given
nz Cen2-(T~ Tr2) = narCvar(Tar~T)
T := Find(T) T—273.15-K = 90degC
Find P after mixing:
Pro + Par
2
(guess)
280
3
Given
(ny2 + nar)-R-T
P
P sx Find(P) P= 24.38bar
Calculate entropy change by two-step pat!
1) Bring individual stream to mixture T and P.
2) Then mix streams at mixture T and P,
ASyo != nN2 (omon( 4) - we P }) ASxq = 11 806-2
2) Pra,
iG (ame (ery
Sqr = mar] CParln| | — Rel = }\ ASa, = -9.5474
\ Par}
. I
ASmnix = Mrotat( RS? xi-In(xi)) = 36.006
Smniy = Mt wo yx (3) Sax = 36.0065
Gens
AS := ASy2 + ASay + ASprix Ans.
mdoty2
molwtng ‘= 28.014-2— molwty = 2.01622
mol mol
mdot mot
molarflowyy2 me molarflowyg = ———2
molwiy? mola,
molarflowsotal := molarflowy2 +molarflowy2 molarflowrotal = 319.409 22
see
molarflowy2 molarflown2
yey 0224 yg 0.776
molarflowiotal molacfloweotat
Ans.
~R-molarflowjota!: yy ta(y3)
291
4
115
Ty = 448,15-K Tz = 3081S-K | Py ss 3-bar
For methane:
McPH(448.15,308.15, 1.702,9.081-1073, -2.164 107 §,0.0) = 4.82200
,-2.164:10" 0,0) = 4.79051
McPS(448.15,308.15, 1.702,9.081-10
For ethane:
McPH(448.15,308.15, 1.131, 19.225-1075, ~5.561-10" 6,0.0) = 7.59664
eps 448.15,308.15, 1.131, 19.225-10" 3,s.561-10"®,0.0) = 7.53108
MCPH := 0.5-4.82200 + 0.5-7,59664 MCPH = 6.209
MCPS := 0.5+4,7905I + 0.5-7.53108 MCPS = 6.161
AH := R-MCPH-(T2 -T;) aH = 72274.
mol
(T Pa)
AS = R-MCPS-In} — | ~ R-Inj | + R-2-0.5-In(0.5)
Th Pry
‘The last term is the entropy change of UNmixing
J
moh
AS = -15.813.
Wigeat := AH - Tg-AS
Basis: 1 mole entering air.
0.21 y2i= 0.79
Assume ideal gases; then AH = 0
The entropy change of mixing for ideal gases is given by the equation
following Eq. (11.28). For UNmixing of a binary mixture it becom
300-K,
vie
J
= Re(y-In(y,) + yeh AS = -4273
8 = R-(yr-ln(y1) +y2-In(y2)) morK
He
By Eq. (5.27): Wideal := ~To-AS Wideat = 1.282 WS
voy Wide! papel
By Eq, (5.28): Work := Morn 20a Ane
mM <: fi
292
11.16
0 1.000
10 0.985
20 0.970 od
40 0.942
0913
bar Z =| 0.885
end := rows(P)
0.869
0.765 is=2..end
0.762
0.824
500 0.910
Fiis a well behaved function; use the trapezoidal rule to integrate Eq.
(11.34) numerically.
Fi+F;
ERP) Indi = ng + Ai
41 = exp(ingi) fis
orPi
Generalized correlation for fugacity coefficient:
For CO2: Ty = 304.2-K 3.83-bar © = 0.224
T = (150 + 273.15)-K = 1391
4
BO = o.0g3 — 2422 BO = -0.166 BI = 0.139 ~ 2172 Bl = 0.096
TS 42
BREE Ty
2 7]
Pe |
Ga(P) == exp] [-(B0 +0 By fo(P) = OG (PP
283
bar bar
Calculate values: |” a 8.25]
20 oar 79.555
20 [ome] 37978
| 9} a | 88382
20 o8%6| T1676,
100] 0872! 187
200) O77)
300] [0696
| 400) 0656
500) ‘836
, 8
oe
4a(P i
eee 6
i ee
ice riaoten
Pi
‘bar
Agreement looks good up to about 200 bar (Pr=2.7 @ Tr=1.39)
11.17 For S02: 8.84-bar = 0.245
Po
P = 300-bar
Pr = 3.805
For the given conditions, we see from Fig. 3.15 that the Lee/Kesler
correlation is appropriate,
Data from Tables E-15 & E.16 and by Eq. (11.64):
204
11.18
11.19
090°
GRRT := In(6)
6 6 = 0.724
fS27 bar GRRE 0.323. Ans.
Isobutylene: Te: 4179-K Pe = 40.00-bar = 0.194
a) At 280 degC and 20 bar: T := (280 +273.15)-K P= 20-bar
Tn) = TT) = 13236 PCP) = PP) = 0.5
r(T) 2= ke (7) = 1.323 1! Po rf
At these conditions use the generalized virial-coeffieient correlation.
PHIB(1.3236,0.5,0.194) = 0.9379 PHIB = 0.9379
f= PHIBP =18.76bar Ans.
b) At 280 degC and 100 bar: T := (280 + 273.15)-K. P= 100-bar
Te(T) = 1.3236 P,P) = 2.5
At these conditions use the Lee/Kesler correlation, Tables E15 & 1.16 and
Eq. (11.64).
60 = 0.7025
$1:5 12335 ¢ = 90919
oe
oP
£2 73.469bar Ans.
The following vectors contain data for Parts (a) and (b):
(a)=Cyclopentane; (b) = I-butene
_ (0.196
~ \o.19t
S118 45.02
/K Poe bar
420.0, 40.43.
0.273 258 ) om? 322.4
ze(2?) ve ( Ble as x
0277 2393 ) mol 266.9,
383.15 215 5.267
Ts -K P= bar Psat = -bar
393.15, 34 25.83
>
T in
at 0.117
= T; Psat = Psat, =
Te (0.9361 Pe 0.6389
Calculate the fugacity coefficient at the vapor pressure by Eq. (11.6:
Tr
(a) PHIB(0.7486,0.117, 0.196) = 0.9000 (° |
PHIB :=
(b) _ PHIB(0.9361,0.6389,0.191) = 0.7596 0.7596
Eq. (3.63), the Rackett equation:
=
Th fia )
Tor = Tor =
Te {0.635
Eq. (11.41):
97.092 \ om?
van = (700? at
91.426 } mol
Leh oe
£ f be Ans.
20.07
molwt = 18-2
24
mol
P = 150-bar
cm
Veat = 18.342
mol
Equation preceding Eq. (11.41), pg. 387:
Vear(P —P. seat
rom exp Lael Pos) r=1079 rec S079 Ans.
L RT eae
11.22 The following vectors contain data for Parts (a) and (b): | molwyt := te
mol
Table F.2: (a) 9000 kPa & 400 degC; (b) 1000(psia) & 800 degF:
296
[ (400+273.15)-K |
ny :
(800 + 459.67)-rankine
3121.2. el 629152
gm emK
AL Bu Sis Btu
1399.64 1.3677
3 Tbprrankine
Table F.2: (a) 300 kPa & 400 degC; (b) S0(psia) & 800 deg
1
8.0338-—
gorK
Bu
Heer eee eee
Ibn rankine
Eq, (A) at the top of page 388 may be recast for this problem as:
—————>
[ molwe | H2- Hr | (0.0377),
rou] Rig ten (2-5) = (o.0sa2)
TY
@ =2s00n o «
aes
11.23 The following vectors contain data for Parts (a), (b), and (c):
(a)=n-pentane —_(b) = Isobutylene (© = 1-Butene:
469.7 33.70 0252
Te =| 4179 -K 40.0 |-bar 0.194
420.0 40.43 0.191
0.270 3130), 309.2
Ze =| 0.275 2389/27 tas} 266.3 |-K
oan 2393) ™! 2669
200 1.01325
300 |-bar Psat :=] 1.01325 |-bar
150, 1.01325
297
> (0.6583 ) — 0.0301
Ta
lax 1, = | 0.6372 | p= Pt ad 2s
Te i Pe
0.6355 ) 0.0254
Caleutate the fugacity coefficient at the nbp by Eq. (11.65):
(a) PHIB(0.6583 0.0301 ,0.252) = 0.9572 0.9572
(b) PHIB(0.6372,0.0253,0.194) = 0.9619 PHIB = | 0.9619
(©) PHIB(0.6355,0.0251, 0.191) = 0.9619 \0.9619
EEE
3857
Eq. (3.63): —_Vsat | vexe! 7) |
‘Vsat-(P — Ps
Eq. (1.41): ¢ [pata aten
11.24 (a) Chloroform: T, := 536.4K @ = 0.222
Psat := 22.27-bar
Ze = 0.293
Tis 47B.15K T,= 0.882, Tm = 0.623
0257 3
Eq. (3.63): Vsat = Verret tm) Vsat = 94.409
mol
Calculate fugacity coefficients by Eqs. (11.65):
0.422
Bt 0139-42
42
Tr
BO := 0.083 -
715
BO = -0.433, BI = -0.152
298
P(P)
re) = 6) = of (90-080)
£(P) = af << Psat, 9(P) Pera aten| oy Psat) ||
uJ
he Peat __ [Vane ~ Pat)
#(F) =i Ps Pat 9), HP el SE Sa
P := O-bar,0.S-bar.. 40-bar
(b) Isobutane = 36.48-bar@
181
Ze 2 0.282 261.4-K 5.28-bar
a Th
T= 313.1S-K Ty:= — Ty = 0.767 Tm = Tr = 0.641
Te Te
0.2487 3
i Ip)
Eq. (8.63): Vsat = Vezg(tm) Vest = 1021052
‘mo
Calculate fugacity coefficients by Eq. (11.65):
0.422
BO cre aerisl BI = 0.139 - 2172
Tr
299
BO = ~0.562
>
PAP) = 2
= 5
£(P) if S Psat, (P)-P,6(Psat)-Psat ox
oP)
sat | Wsat-(P — eo]
Ps
P< Psat,6(P), oo a
if 'sat O(P) (Psat) pe Rr
O-bar,0.5:bar...10-bar
os
uP)
06
od
aed Bee
bar’ bar bar
11.25 Ethylene = species 1; Propylene = species 2
282.3 (50.40 0.087
KK Pe := ‘bar wes
365.6, 46.65 0.140
0.281 131.0) em?
Ze= ve =( 22) 22%
0.289, 188.4) mol
T = 423.15-K y= 035
n=? HF
300
By Eqs, (11.67) through (11.71)
(ite i)
1317 1.157,
131 157.966) em? _ 8 aes
“(157.966 188.4} mol °° (48.189 46,627
0.087 0.114 282.3 321.261 0.281 0.285
o= Te= K Z=
(oid 0.14 321.261 365.6 0.285 0.289
By Eqs. (3.61) and (3.62):
BO; j *= 0.083 — on 6 unm
() Ty
le! 138 nd a _ (0108 ae)
0.189 -0.251 (0.085 0.046 }
892 -99.181 | om?
( 99.181 -159.43) mol
( 0 20.96) a
~\2096 0 }
cp
301
fhaty := ghaty-P phat pens A
ak = phate ab ns,
ke OBIS)
For an ideal solution, $id = pure species
: Po, _ (0595 . i Pr (
Pac = Loa) tide = ox i, BOq,k + Ok Blk,
fhatig, = Gide
Alternatively,
P
Pry po idk = exp) (B04, +04 eB)
7 Lica
11.27 Methane = species 1
Ethane = species 2
Propane = species 3
021 0.286
0.43 Ze.=| 0.279
0,36, 0.276,
190.6 98.6 3
305.3 |-K ve =| 145.5 |
mol
(369.8 200.0
ns3 i 1
302
1.958 1.547 1.406
Tr=| 1547 1.222 1.411
1.406 L111 1.009
98.6 120.533 143.378)
Ve =] 120.533 145.5 171308 |
mol
143.378 171,308 200
(45.964 47.005 43.259) 0.012 0.056 0.082)
Pe= | 47.005 48,672 45.253 jbar @=]00s6 1 0126)
(43.259 45,253 42.428 ) (0.082 0.126 0.152)
( 190.6 241.226 265.488) (0.286 0.282 0.281)
Te= | 241.226 3053 336.006 |K 0.282 0.279 0.278
265488. 336.006 369.8 ) 0.281 0.278 0.276)
By Eqs. (3.61) and (3.62):
0.172
(t,)
Blj, j = 0.139-
By Eq. (11.61):
eran 0 30.442 107.809
em
=| 30442 0 23.482 |
mol
107.809 23.482 0
Ghat. = vos {* vata D Daal 2-85, k- 8) JT
hat := ghaty-P
6), j = 2B, )~Bi,i~
11.28
For an ideal solution, did = 6 pure species
. 0.761 ) 2 +
Pros py Pee ozi8| idk 2= ex = (B +0, kBlk,¥)|
0.824 ) bk J
“(oan
fata, := éidkeP 0.88 Ans.
Give: = (2.6.x; -1.8:x2)x1-92
RT
() Substitute x)= 1 - x),
a
=.8-x1 — 1.8)-x)-(1—x1) = -1.8-x) +72 + 0.8x)>
RT
Apply Eqs. (11.15) & (11.16) for M=GE/RT:
(a)
S (nm)
‘dx
5
2
Zo -1842-n +24x17
dx)
fy, © 1.842814 TA x
Ing = 3x1 — 16x)?
(b) Apply Bq. (11.95):
(©) Divide Gibbs/Duhem eqn. (11.96) by dxt:
These two equations sum to zero in agreement with the
Gibbs/Duhem equation,
(a) When x1 =1, we see
from the 2nd eq. of
Part (¢) that
QED.
When xi = 0, we see
from the 3rd eq. of
Part (c) that
QED.
(©) DEFINE: g= GE/RT
e(x1) = 18x) 4x17 +0.8-x13
In (x1) = 1.8421 + 14x — 16x
tnya(x1) = 9? 1.6.x.
‘nye
Inypel)
x15 0,0.1..10
11,32
Xi?
0.02715
0.09329
0.17490
0.32760
0.40244
0.56689
0.63128
0.66233
0.69984
| 0.72792
0.77514
0.79243
0.82954
0.86835
0.93287
0.98233.
xi = 0,001.1
*) a {3448 10°
infit(x1,VE,F)}8} =} 3202,¢10°
ec} \ 244.613
Ans.
600
ve} 400
xian Larbatie st") 509
0 02 04 06 O08 1
xxl
By definition of the excess properties
VE = xpxplatbx; re(xs)?]
Bs —t.0-(xy) + 3(e —b)-(xy)? + 24b—aday ta
1
(Vbary)® = (x9)? a+ 2-b-xy + 3-e-(x1)*|
(vara) = (x1)?[ b+ 246-9.) +3-e(%)?]
(b) To find the maximum, set dVE/dx; = 0 and solve for x). Then use x, to
find VE nae
Guess: x1 :
Given
4o(xl) +3-(—b) (Xl)? +2(b~ a)-xl $a = 0
x1 := Find(x1) X1= 0353" Ans.
307
VE mac = x1-(1 = x1)-(a+ bext +0-x1?)
(©) vebary(xi) = xt)? [a+ 2-b-xt + 3-6-(x1)? |
2 27
Vebara(xl) = (x1)? a~b+ 2(b~)-xt + 3-6-6419? }
x1 = 0,001.1
4000
Years xt) 70
Vebara(x!) é
Discussion:
a) Partial property for species i goes to zero WITH ZERO SLOPE as x,-> 1.
'b} Interior extrema come in pairs: VEbar min for species 1 oceurs at the
same x, as VEbar max for species 2, and both occur at an inflection point on
the VE ys. x, plot.
¢) At the point where the VFbar lines eross, the VE plot shows a maximum.
11.33 Propan
1; n-Pentane=2
= (75 +273.15)-K
yi 0s
276-466 :
islam jedan
466-809
By Eq. (11.57): Be yey Bi, j epi ne om
, B = 504.25
22 a
308
Use a spline fit of B as a function of T to
find derivatives:
331), 558),
bil =| -276 |. bits] 466}
mol ‘mol mol
=235. (399,
( 50) (323.15
us | 15 |+27as K | s4a1s K
(00) \s73.15
vsiL := Ispline(t,b11) B1I(T) := interp(vsl!,t,b11,1) BIICT) = 276
moi
3
ys22 := Ispline(t,b22) B22(T) := interp(vs22,t,b22,T) B22(T) = 809
mmol
3
vs12 := Ispline(t,b12) Bi2(T) := interp(vs!2,t,b12,T)B12(T) = -466
mol
{anc “Leia
aT aT 1.92 3.18) em?
abdT = aBat = son Ga
5.92.) mol
“arm “par :
\at ar
3
Differentiate Eq. (11.57): dBdT := yryydBdT), jdBaT = 3,55
'q. (11.57) DVD vasa ard
vi
BP
By Ba. 37:2 = 14 Z = 0.965
P(B
By Eq. (6.54): HRRT == fe ~ sat} HRT = -0.12 HR := HRRT-R-T
J
By Eq. (6.55): SRR -dBdT SRR = -0.085 SR == SRR-R
11.34 Propane =1; n-Pentane=2
T= (954 273.15)-K Pis2bar = yp 05 ais -y
: _ {276 -466) om? a2
~ \-466 -809) mol ari
8),j = 2-Byj- Bi i-Bj,j
By Eqs. (11.59) and (11.60):
iy
es [aed -wer]
P 2,
hat2(yl) := exp) ——-(Bo > +y!
thataCe0 = exp 35 (B2,2+91 81.)}
ghati(yt) »
yl: 0,0.1..1.0
0.99
0.98 +
ghatl (yt)
— "0.97
gha2(y1)
0.96 F
09s b
oe ocrarei02 oa 06 08 1
yl
310
11.36
x=
(a) Guess:
0.0426
0.0817
0.1177
0.1510
0.2107
0.2624
0.3472
0.4158
0.5163,
0.6156
0.6810
0.7621
O8181
0.8650.
0.9276
0.9624
xr(I~x1)
F(x) :=} x1-(1-x1)
x1°-(1-x1)
HE;
x(t) [asbaxteo(xty?]
n= rows(x)) i
.0.01..1
“191.7 xls
b= 100 c:= 0.01
ree
linfit(x1 ,HE,F) Eb
bey
°
i
100
200
0 02 04 06 08
xppxl
an
By definition of the excess properties
HE = xpxofarbor te(n)']
a
xy
(Hoary)? = ()°2 + 2-b-x1 +3061)" }
Aee(xi)3 4 3(e~b)(x1)? +2 a)ext ba
(Ltbara)® = (x1)*[ a= b+ 24b-e)en1 +3-e(x)*]
() To find the minimum, set AHE/dx; = 0 and solve for xj. Then use x, to
find HE gig.
Guess: x12 0.5 EG) = x1-(1-x1)-(a4 bal tex
Given 4-c-(x1)> + 3-(e—b)-(x1)? + 2-(b— a) xl +a = 0.
xl = Find(xl) xl
51 Ans.
HE min == x1-(1—x1)(a + bxt +x?) Ans.
(©) HEbarj(x1) = HEQal) +(1~x1)S-- TEC)
ix
HEbarg(x1) := HE(s1) - x1 (:. 00)
x1 0,001.1
00 ——
HEbari(x1) ©
HEbarp(x1)
500
312
Discussion:
a) Partial property for species i goes to zero WITH ZERO SLOPE as x,-> 1.
b) Interior extrema come in pairs: HEbar min for species 1 occurs at the same
x, as HEbar max for species 2, and both oceur at an inflection point on the HE
vs. x; plot.
©) At the point where the H¥bar lines cross, the H® plot shows a minimum.
11.37 (a) (1)=Acetone (2) = 1,3-butadiene
T= (60+273.15)K P= 170-kPa
ua) vif ) cm?
0.267 ©" (20.4) mol
( 0.307 0.2485 0.082
Eq. (11.67) 0.2485 0.19 0.126
0,082 0.126 0.152,
( 5082 464.851
Eq. (11.68) Te =| 464.851 425.2 |K
369.8 0
0.233 0.25
Eq. (11.70) Ze=| 0.25 0.267
0.276 0
( 209 214.65 Fi
Eq. (11.7) ~|[21465 204 |
“| mol
200 }
(47. 104 45.013
re {esos 42.826 |bar
(42480
Note: the calculated pure species Pe values in the matrix above do not agree
exactly with the values in Table B,1 due to round-off error in the calculations,
Faq. (11.69) Pej, j =
313
0.036 0.038
0.656 0.717)
Pr=|0.038 0.04
717 0.784}
0.824 0
a
Fq.@.61) —BO;,; = 0083-22
(t5;,))'*
0.74636 -0.6361 -0.16178
BO =| 0.6361 0.5405 -0.27382
0.16178 -0.27382 -0,33295
Bq.6.62) Bij = 0139-2
(a)
0.874 -0.558 0.098 )
Bl =|-0.558 -0.34 0.028
0.098 0.028 -0.027
Eq, (11.66) *(B0;, | +: jBli,3)
3 (7210278 “est em’
(665.188 -499.527) mol
= om?
Eq. (11.57) = yi i eae
q. (11.87) B DY DY weve B= 598.524
i jm
BP
Eq. 3.37) Z:= 1 +} —
4g. RT
Ans,
314
an Eq. (681) dBldTy;,j = 2
TH i)
Differentiating Eq, (11.57) and using Eq, (11.66)
en : [i
ist j=
Eq. (6.80) dB0dTs;,j
Eq. (6.54) HR := P. (2 - ana Ans.
Eq. (6.85) SR := -P-dBdT Ans.
Eq. (6.53) GR:= BP Ans.
3
v= 13694. HR = -450322-—
mol mol
7 OR = -125.1-—.
SR = ~1.006:
mobK mol
HR = -175.666-—
cm
© ‘V & 24255: rT mol
aaa eee oR 333.2,
molK mol
cm J
(d) V = 80972-—— HR = -36.48-—
mol mol
sr = 0.097 oR 1
molK mol
om J
() V = 56991-— HR © -277.96-—
mol mmol
SR = ~0.647.—_ 252
molK mol
315
Pbs. 11.X1 --- 11.X3
11.XI_ Calculate ¢ and f bar by the Redlich/Kwong equation of state
for one of the following, and compare results with values taken from a
suitable generalized correlation:
(a) Acetylene at 325 K and 15 bar.
(b) Argon at 200 K and 100 bar.
(©) Benzene at $75 K and 40 bar.
(d) Carbon dioxide at 350 K and 35 bar.
(e) Ethylene at 300 K and 50 bar.
( n-Hexane at 525 K and 10 bar.
(g) Methane at 225 K and 25 bar.
(h) Nitrogen at 200 K and 75 bar.
11.X2 Calculate @ and f bar by the Soave/Redlich/Kwong equation of
state for the substance and conditions given by one of the parts of
Pb. ILLX1, and compare results with values taken from a suitable
generalized correlation.
11.X3_ Calculate 6 and f bar by the Peng/Robinson equation of
state for the substance and conditions given by one of the parts of
Pb. 11.X1, and compare results with values taken from a suitable
generalized correlation.
‘Vectors containing T, P, Te, Pe, and @ for Parts (a) through (h):
325 15 308.3 61.39 187
200 100 150.9 48.98 000
575 40 562.2 48.98 210
350 35 304.2 73.83 224
300 50 223) °° | soo 087
525 10 507.6 30.25 301
225 25 190.6 45.99 012
200. 15 126.2. 34.00 | 038
316
1.054) 0.244
1.325 2.042
1.023 | .817
> >
oes List | te 0.474
Te 1.063 Pe 0.992
1,034 0.331
118 0.544
1.585, 2.206,
11.X1 Redlich/Kwong Equation: Q
0.08664 W = 0.42748
0.02 ) 4.559
0.133 3.234
0.069 4.7
7 0.036 3.998
fia (2 2) CS) B= ost, 680 ae | 504
0.028 | 4.691
0.04 | 3.847
0.121 eas
Guess: z:= 1
Given 1+B-qf-——— 3.49) 2(B.,q) == Find(z)
z(z+p)
Bisai) +
ce Ky hn 6.62b)
(ay Bia ) ;
4; = exp(Z(Bi,ai) ~ 1 in(Z(8;,qi) - Bi) - gr4)(41.36)
Pi
317
2(Bi.a) =
a5}
722 |.
“0668
9987):
9630}
891]
O88
0.888)
11.X2 Soave/Redlich/Kwong Equation 2 = 0.08664 = 0.42748
x
*)
© = (0.4804 1.574-0 - 0.1760 a
(0.02 4.49
| 0133 3.202
| 0.069 4737
| 0.036 (v0) 3.19
Pr x Wo i
=|] @.s0) -( G51) q=
a Ge cag FP
0.028 4.62
0.04 3.827
\o.21 2.304 )
z-B ,
Given 2=14+B-q-B- (3.49) 2(B,.q) == Find(z)
z-(z+B)
Z(Bi,ai) + Bi
vers nels) oan
Z(8i.a)
4; = exp(2(Bi,ai) ~ 1 = In(2(Bi,)) - Bi) - ar) 11.36)
fj
OrPi
318
2(Bi.a
aoa
L729.
on73).
Case]
686
683}
6.862)
0.884
11,X3 Peng/Robinson Equation
Lay2 Q := 0.07779 Y= 0.45724
—_—>
c= (0.37464 + 1,54226-0 ~ 0.269920?) a=[ieeli-1°)F
0.018) 5.383
0.12 3.946
0.062 5.658
a eal sy ga {48
0.073 5.359
0.025 5.527
0.036 | | 4.646
0.108, ] (asa
Guess:
Given z= 1+B-q-B. (3.49) -2(B.q)
2-8
+e-p)}@+op)
Ly 2{8i.a) +06:
242 Z(Bi.4i) +68
) (6.62b)
319,
exp(Z(Bi.ai) - 1 - in(2Z(B;,03) - Bi) ark) (41.36)
@ BY GENERALIZED CORRELATIONS
Parts (a), (d), (f), and (g) — Virial equation:
325 308.3 15 61.39 187)
350 304.2 35 73.83 224)
Te := P= Pe := |
$25 507.6 10 30.25 301
225 190.6 25 45.99. ia)
=
Tek
Te
Evaluation of 6:
eee
BO (os cad 2 | (3.61)
\ Tr
ppo:= 2°75 680)
ws
[Pr (a)
fa 11.6!
om (B0 + @-B1)| (41.68) ®
(3)
320
Parts (b), (©), (€), and (h) —- Lee/Kesler correlation:
Interpolate in Tables E.13 - E.16:
1.1842
0.9634
7454
7517,
40= ls
7316
8554,
—>
os (051°) (11.64):
0.9883
1.2071
321
‘0.000
0210
~ | 0.087
0.038
(b)
©
©
@)
Chapter 12 - Section A - Mathcad Solutions
12.1 Methanol(1)/Water(2)- VLE data: T= 333.15K
39.223 0.1686 0.5714)
(ose 02167 0.6268
| 48.852 03039 0.6943
52.784 0.3681 0.7345
56.652 0.4461 0.7742
= -kPa x ys
60.614 0.5282 0.8085
63.998 0.6044 0.8383
67.924 0.6804 0.8733
70.229 0.7255 0.8922
(72.832 0.1776, 09141
Number of data points: := rows(P) n=10
Calculate x2 and y2: xg = (1-x1)
Vapor Pressures from equilibrium data:
Psat) := 84.562-kPa Psatg = 19.953-kPa
Caleulate EXPERIMENTAL values of activity coefficients and
excess Gibbs energy.
— —
yeP yoP face nese
ney ~— R= Shay CENT (x1-n(v1) +2-4n(y2))
322
u,= inf)= h(x) = Gert, =
7] (ee Tom] | [base aos O07
2| [aay 026} [0.85 ci28 0.104
3] [ee 7078] ([Gz7e aT 0.185
a] [286 712 oz 0106 O76]
3] aes v7} fast 31468 ‘a8
6] [x07 cs) 308 0.148
7] [05] qa] [oo oat 0786)
3] [ai vas} [081 03 on7
3] [ras ee aaa O08
vo] [row rai] [oor o348 0.086
os r 7 r 1
oab : 4
(a) Fit GE/RT data to Margules eqn. by linear least squares:
GERT;
X12,
Slope := slope(VX,VY) Intercept := intereept(VX, VY)
Slope = -0,208 Intercept = 0.683
Slope + Ai
Ans,
‘The following equations give CALCULATED values:
1 Gs1 22) = enpl 22[ Ata +2-(Aa1 - Ara} |]
y2Gx1 ,x2) := exp] x? Ag) +2-(Ar2- Ari) x2}
i
O1-j~ 01 Xa,
1101 Xi
i i
Peale, = Xa 71 (X1, X25) Psatn + X,72(X1, .Xa,) Psat
HB aeed eels alia ie ec
x 11 (1 .Xa, | Psat
Yiecate,
i Peale,
5
P-x,y Diagram: Margules eqn. fit to GE/RT data.
90 ;$——_—______——
o|
Pj
m |
soa
|
Pa
atc, 59
nan, *|
a 40]
Peale
4 30
Wa
20
04 06 08
+ X15 Yicate,
ot)» Yheale,
500 Px data
° Py data
P-x caleulated
— Pycaleulated
324
Peale, xa¥l (a1, .82)) Psatr +x2,72(x1,.82,)
x1 71(31,0%2,) Psat
Yicale;
\ Peas,
RMS deviation in P:
RMS = 0.399kPa
(b) Fit GE/RT data to van Laar eqn. by linear least squares:
X1 99,
VXi5= x1, VYj:
GERT;
Slope := slope(VX,VY) Intercept := intereept(VX, VY)
Slope = 0.641 Intercept = 1.418
1 i
ay2 = ——— a=
Intereept (Slope + Intercept)
ap = 0-705. ag
i |
apy x2
21 ne
YG x2) = exp] apy 1 EY
a2"
0485 Ans.
a
A(X1,x2) ~ af +
101 Xi, = 01, ~ 00999 (To avoid singutarities)
X,
-x
1
325
Peale, = Xy 41 (Xi .Xo,} Psat 4X2, 2(X1,X2,} Psata
Peale, = x1, 71 (81, .%2,}Psaty +29,-72(1,.X2,) Psata
Xt V(X
Pea
X2,} Psat x11 (x1, %2,) Psaty
Yleale, =
Yicale, =
id Peale
i
P-xy Diagram: van Laar eqn. fit to GE/RT data.
0 ~
80
qe)
iPa
coo
Pi 60
kPa
ic)
Peale;
a 4g
Peale; de
Wa 30 a al
20
109 02 a4 06 08 L
XTX,» Yea;
coc Px data
° Py data
—— P-x calculated
—— P-y calculated
RMS deviation in P:
RMS = 0.454kPa
326
(©) Fit GE/RT data to Wilson eqn. by non-linear least squares.
Minimize the sum of the squared errors using the Mathcad Minimize funetion.
Guesses: Agu = 1.0
SSE(A 49,431) != ze + (ron +412) : I
i Seapine, +1821
ig
11(Xi,0%2,) Psat + x9, 72x12, } Psatz
Veale,
ca ale,
Peale; 1
327
P-x,y diagram: Wilson eqn. fit to GE/RT data,
90,
0
Pi
im 7
2 6
is
Peale; a
i 40
a ao
we
20
ue 0 coor 0.6 08
¥1 XA» Yieate,
000 P.x data = -
data
x calculated
— Py calculated
RMS deviation in P:
RMS = 0.48kPa
(4) BARKER'S METHOD by non-linear least squares.
Margules equation.
Guesses for parameters: answers to Part (2).
yi (xt x2, Aq2, Aan) i= exp| (2) Ata + 2-(Agt - Aa): xt]
‘yo (xl x2,A12,Aan) == exp] (xt)? Aor +2-(At2- Aoi) 2]
328
mize the sum of the squared errors using the Mathcad Minimize function.
Guesses: An
$ Agi = 10
\ 2
1,74 (1,092, -412,Aat)} Psats .. |
1,992, >A12,A21)} Psat
X171(%1,.Xa,.A12,A21) Psat
+ X2,-12(%1 Xo, Ar2,Aat) Psata
gL wate ate
Xx
7a( XX Az, Aa) Psaty
Peale,
x17 1(81 982, A12,A21) Psat.
+2,72(m1,, 2 »Ai2,Aai} Psaty
(19%, Ai2,Aat} Psaty
Yicale,
Peale
RMS deviation in P:
RMS : RMS = 0.167kPa
329
P-xy diagram, Margules eqn. by Barker's method
0 —
80
Bi
‘KPa 70.
aco
eeiaried
ira
Sc30,
Peale;
a 40
Pealey 39
wa
20
0
q CSR a 1
sapedIyeXty- Ylcaey
aco P-x data Ed
°° P-y data
P-x calculated
— P-y calculated
Residuals in P and yt
PP, os . :
KPa o Xe
¥1,-Yteale;} 100 tte i.
br : AEE
v2 0406 08
x1
Xx % Pressure residuals
yl residuals
330
(e) BARKER'S METHOD by non-linear least squares.
van Laar equation.
Guesses for parameters: answers to Part (b)-
1.101 X1, = OL) ~ 00999 Xa, f
/ 2
ayaxl
= exp) ay2:] 1+
mp x2
ey?
= exp) agy-| 1+
alaxl
‘Minimize the sum of the squared errors using the Mathcad )
vist .x2,a;2,a21)
‘7a(x1_.x2,a12.a21)
mize function.
Guess
"| ay2 = 0.5 ayy = 1.0
1) ELF (earn) ee : ]
i L +0,°72(%1 982, 412-821) Psat
Minimize(SSE..a12,a21) (
SSE(a12
fa)
(arr)
Pea = X1y11 (Xo Xay 12a} Paty
+Xo.-yo(X1.,.X2,.012,821)-Psatz
7X1 -Xoy. 012,21)
X17a(X1,X2,,a12, 421) Psat
Beg ed eee ee era aE
leat, fa
ale,
Pate, = X1;71(%1).%2,,412,821) Psat
+2, 2(1,82,,012,821} Psata
x4, 71( 1,582, 08120801) Psat
Ycale,
Peale,
331
RMS deviation in P:
RMS = 0.286kPa
P-xy diagram, van Laar Equation by Barker's Method
90
80
10
aaa 60
50
Peale; 40
20
15 02 oa oe 08 1
Na 9191) Vooal
900 P-x data a
© Pry data
— P-x calculated
— P-y calculated
332
Residuals in P and y1.
Pi-Peate; os
ke
(¥1,-¥teate;} 100 ‘i
oo 02 04 06 08
4
200% Pressure residuals
© yl residuals
(®) BARKER'S METHOD by non-linear least squares.
mn equation.
Guesses for parameters: answers to Part (c).
j= .1ot Xi, = Ol j-.01
J
ya (x1 .x2.A 12,821) == exp -In(xt +x2-A 42)
rin sts)
| !
y2(xt.x2.A12-Aa1) #= exp) -In(x2 + x1-A1)
(_ -An Aa }
+
fei) ee
J#X2AqQ xX2#X1-AQ
Minimize the sum of the squared errors using the Mathcad Minimize function.
Guesses: AjQ = 0.5 Ag, = 1.0
333
1 (1.8, r20Aau)Psaty i
2,2 Aai) Psaty
i + x27
S9E(A42,A21) “rte
Ca ee
4198)
(N27)
(‘*) := Minimize(SSE,Ay2,A24) ( e
Agi) Aa.
Peale, = X17a{X1,oXa,Ay29Azi) Psat
Xa. X1,,.X2.,Ay2,A21)-Psata,
X171(%1,-%2, i242) Psat
Yieale, *=
ea Peale,
Peale, = 51,71 (81,982, -125.Na1} Psat
+%, *72(¥1,9%, 412-429) Psat
x7 (%4 92, A 2,21) Psat
¥ieale;
RMS deviation in P:
be RMS = 0.305kPa
334
P-xy diagram, Wilson Equation by Barker's Method
iPa
ooo
Peale;
kPa
Peale;
90
80
20
10
0
Residuals in P and yl.
Pi-Peal,
Pa
wx
(X1,-Meale;) 100
02 oa 06 0
tpt pX1y Viole
280 Px data
© Pry data
P-x calculated
— Py calculated
q =
eo
os x 4
x s :
“05 *
so 02 oa 06
x,
**% Pressure residuals
© yi residuals
08
12.3. Acetone(1)/Methanol(2)-- VLE data: T := 328.15-K
72.278 0.0287 (0.0647
75.279 0.0570 0.1295 |
77.524 0.0858 0.1848 |
78.951 0.1046 0.2190 |
82.528 0.1452 0.2694 |
86.762 0.2173 0.3633 |
90.088 0.2787 ose |
93:206 0.3579 04719 |
95.017 0.4050 0.5133
96,365 0.4480 0.5512
Ps -kPa x yess
97.646 0.5052 0.5844
98.462 0.5432 0.6174
99.811 0.6332 0.6772
99.950 0.6605 0.6926
100.278 0.6945 0.7124
100.467 0.7327 0.7383
100.999: 0.7752 0.7729
101.059 0.7922 0.7876
99.877 0.9080 0.8959
99.799 0.9448 0.9336
Number of data points: n == rows(P)
Calculate x2 and y2: oy
Vapor Pressures from equilibrium data:
Psat; = 96.885-kPa Psaty = 68.728-kPa
336
Calculate EXPERIMENTAL values of activity coefficients and
excess Gibbs energy.
—? —
yeP —.—_
Ui eee GERT := (xy-in(y)) + x2"In(2})
i 1 72, Infra.) = lafr2,) GERT,
7] [ew Tom Om 0.75] O27]
2] [i765 oH 0588 oot ‘04s
DB) pz 7005] [0544] 5a 105 082
al [766 1002] [ose 7975107 0.068
5 158 To28) [048 0.028) 0.089
8) aar 1027 0.404 0.027 0-108
7] F386 087 0.384 0.055) O78
@| [izes 1.103} 025 0.086 0782
3} [aaa 773] [ote 0725) 0161
To} [az va] fozae 073 0.163
Ti] 66 iiss} [0753 oa77 0.105
v2) ats5| 72 o.taa] 07182 O76
73] [toe 1278 0087 O88 O64
7a] 83] 1317 ore 0275 rd
7] prose 1378 0.06 oa] = [ors
76} [hos Tas Doe [oss 0-128]
7] [ioe 7a] [0059 0385 oni9|
v3] [057 rar] [0056 ‘o407 ons
193] [ao yeaa] [o0i7| cd ‘oe
30] [rove sar] — foots] 0558 048
337
06. '
T
Sof 4) o4
tnfy2i) .
GERT 95
VXi'=
Slope := slope(VX, VY) intercept(VX , VY)
Slope = 0.018 Intercept = 0.708,
Aa := Intercept Agy = Slope + Ara
0 Aus.
AS
Alas
The following equations give CALCULATED values:
4G 5x2) = expl 27 Arn +2¢(421 - Arg)xt |
ania)
j= 1.101 X1,:=01j-.01 Xo i= 1-Xt
i
22Cxt ,x2) = exp] x17 Agy +2-(Ar2
i
Peale, = X71 (Xa, .Na,) Paty +X, 12(X1,X2,} Psat
X1y71(%,.X2)) Psaty
Yicale,
j Peale,
338
P-xy Diagram: Margules eqn. fit to GE/RT data.
10s
100
95
90
85
° 02 04 06 08
14.91 -%1j» Yreale;
B00 P.x data
© Py data
P-x calculated
— Pry calculated
RMS = 0.851 kPa
339
(b) Fit GE/RT data to van Laar eqn. by linear least squares:
xi,
VX} = x1 VY =
i GERT;
Slope := slope(VX, VY) Intercept := intercept(VX, VY)
Slope = 0.015 Intercept = 1.442
1 L
ans a=
Intercept (Slope + Intercept)
21 = 0.686 Ans.
r .
' anrx2)
y2(x1 x2) := exp) apy 1+
L airxl)
j= 1.101 X41, = .O1-j — 00999 (To avoid singularities)
X: -X,
35 4
= Xt; 1 (X1,.X2,)-Psaty +X, ¥2(X1,.X2)} Psat
x1, 71(x1,.%2)} Paty +2, 72(x1,%2,) Psat
irl x
Yieale, =
340
P-x.y Diagram: van Laar eqn. fit to GE/RT data.
105
100
Pi
ime %
cao
a)
kPa
Beate
Peale;
KPa 9
Peale,
fe
10 y
02 Y 06 08 1
saa P.x data
© Pry data
— P-xcalculated
— Py calculated
RMS deviation in P:
RMS = 0.701kPa
(© Fit GE/RT data to
imize the sum of the squared errors using the Mathcad Minimize function.
‘on eqn. by non-linear least squares.
Guesses: Aqg = 05 Agi := 10
SSE(A12-Az1) = °[GERT + (x1,-Infxr, + 92,412)
fi +X; Inf +21) }
3M
Ar
= Minimize(SSE ,A 2,421)
Aa
egjo(—M2 Aan
“Usd #x2-Aqg x2 +x1-Agy
1
ee (14x20
Ag
exp] —x1.| ——>__
eae xL+x2A1
dates (x2 +x1-Agi)
101 X= 01 j-01
i
1-71 (Xr Xa} Psats + Xo,-¥2(X1, Xo} Psat
111 (1,099,) Psat + x9, P(s1,.92,} Psata
Xt A(X sX,
ees
Peale,
P-xy diagram: Wilson eqn. fit to GE/RT data,
105
100
Bi
ipa 95
eso
fi 90
iP
o 85
Peale,
Wa a0
Peal; 95
Wa
2
6s
0 02 04 06 08
x17 %y» Yee,
909 Px data
© Pry data
— Px calculated
— Py caleuiated
RMS deviation in P:
RMS := RMS = 0.361 kPa
(a) BARKER'S METHOD by nor
Margules equation.
ear least squares.
Guesses for parameters: answers to Part (a).
exp] (02° [A12+2(Aat~Ana}xt |
tral} x2. Aiz,Aa1) = exp], (1)? Aor +2-(At2 — Aor 2] |
vast x2,A12,A21) #
343
Minimize the sum of the squared errors using the Mathead Minimize funetion,
Guesses: A= 0.5 Aa = 1.0
S8E(Ar2,A21) = Do]Pi~ (x4p7a(%1,.¥2,,A12, Az) Posty T
(exepra(syn9,-ArzsAai) Psat)
(Ai2\
= Minimize(SSE,A12,A21) Ans,
(Aor
Peale, Xj 71(%a, Xo, -Ar2,Azs) Psat
+ X2,-72(X1, Xo, A12,Aar) Psate
dilate
X1y71(X1,X2,,A12, Aas) Psat
y uss ieeeltteeee
teal,
Peale,
Pate, = ¥1;71(81, 2, ,A12,Az1) Paty
+ 82,°72(1,.%2,,A12,Azi) Psata
1,71 (41,082, A12,A21} Psat
Neale, # as
RMS deviation in P:
(Pi-Peat,)?
RMS := od RMS = 0.365kP:
[= 65kPa
i
P-x-y diagram, Margules eqn. by Barker's method
Pi
kPa
By
Pe
Peale;
kPa
Peate,
Pa
105
100
95
90
85
80
0 a
9 02 oa 06 08 1
X, YI,» X1, 5 ¥leale,
coq pxdta | | Ft
° Py daa
— P-xcaleulated
calculated
x
200% Pressure residuals
© yl residuals
345
(e) BARKER'S METHOD by non-linear least squares,
van Laar equation,
Guesses for parameters: answers to Part (b).
101 XI 01-j — .00999
(xt x2 ) wae (1228)
(xt ,x2 012,021) = exp, 212
" i ara)
r -2
agrx2)?
ya(x1x2,4)2,021) ova (1+ a ) |
ayzxl
Minimize the sum of the squared errors using the Mathcad Minimize function.
Guesses: aya i= 05 ay = 10
SSE(a12,221) ay ae Psat. } r
i #¥0,-7(%1,.%9, 28125821) Paty
sag
Ans.
0.672)
Pt
) := Minimize(SSE.,a)2,a21) (2) 2
= Xj -71(X1,,X2,.a12,a91} Psat
ale 1X2, ri) Psat
+Xo,-y2(X1,.X2, .a12, 421) Psat
2,°¥2(X1, °X2, -a12,a21)-Psaty
X4-7a(X4, Xz. 012,021} Psat
171%, .%2,-a12,421) 1
Peale;
up7 (1, 9%2,412-821) Psat)
$x, ¥2(%1, 82,412,221) Psat
x4,71(,0%2, 412,821 Paty
Yieale, ==
7 Peale,
RMS deviation in P:
P-xy diagram, van Laar Equation by Barker's Method
105
100
90
85
80
15
70|
cd 0 0.2 04 0.6 08
sep X1j-Yreae,
000 P-x data
© P-y data
— P-x calculated
— P-y calculated
347
Residuals in P and y1.
1s
xxx
(v1,-¥1eaK6;} 1000
of 06 (OS 1
x,
*%* Pressure residuals
© yl residuals
() BARKER'S METHOD by non-linear least squares.
Wilson equation.
Guesses for parameters: answers to Part (c).
101 X1, = 01 j-.01
j
ya(xt.x2,Ag2sAar) = exp) -In(xt + 22-42)
(An An)
+x2/
XI HXZAqg x2#xL-AQ,)
yo(x1 ,x2,A42,A21) = exp| —In(x2 + x1-A21) 1
(_-An Migee al
Xl4x2-AjQ 24KI-AQ) |
‘Minimize the sum of the squared errors using the Mathcad Minimize function.
Guesses: Ay = 05 Ag = 10
348
SSE(Aj2.A21) aoe Pi— (171 (¥1,.%9,.A.20Aau) Psat =
+X, 2( 81, %2,.12-Aa1) Psat
(Ar
eee = Minimize(SSE,A.12,A2))
reale, = Xi 71(X1,.X2,.Ay2,Agt)-Psaty ..
Peale, ayn 1X2 Ara 21) Psat)
+X2.-72{X1,.X2,4A12,Agq)-Psaty
2,72 1).%2, Ar 2} 'satz
Xx
(%, %2AraeAay) Psat
Yicale, =
a Peale,
Peate, = x1,-11{1,83,-A12.A2i) Psat.
+ rar(m, oe 2421} Psat
x] a(x 190, -Ar2, Aoi) Psat
Yesle, = ———
Pes
RMS deviation in P:
ee RMS = 0.35kPa
349
J
P-x,y diagram, Wilson Equation by Barker's Method
los -
100 2
Pi
ia 9s|
ooo
zal 0)
Pa
Peale, 85
ia 80
Reales 95
ia
— 7»
—
oO 02 04 06 08 1
MpypX oY)
101% Yieale,
900 P-x data
© Py data
— Px calculated
— Py calculated
Residuals in P and y1.
{Piya} 100
xy
xxx Pressure residuals
© yl residuals
350
12.6 Methyl t-butyl ether(1)/Dichloromethane-VLE data: T= 308.15-K
‘83.402 0.0330 ota
82.202 0.0579 0.0254
80.481 0.0924 é0416
76.19 0.1665 i804
72.442 0.2482 anise
68.005 0.3322 0:1975
65.096 | swe | Oat? 022457
59.651 0.5036 oY) 63686
56.833, 0.5749 04564
53.689 0.6736 aseed
51.620 0.7676 07176.
50.455 0.8476 09038
49.926 0.9093 5002
49.720 0.9529 0.9502
——
(x ya = (1-y:)
Psaty 2 49.624-KPa Psatg := 85.265+kPa
Calculate EXPERIMENTAL values of activity coefficients and excess
Gibbs energy.
—
yoP
xpPsaty 7? Gp.Psatg
—>
GERT
GERTx1x2
n= 14
XP-X2
351
(a) Fit GE/RT data to Margules eqn. by nonlinear least squares.
Minimize sum of the squared errors using the Mathead Mini
Guesses: Aqz = -0.3 Cc
ize function.
SSE(A12,A21,C) = [oerti (Aaixa, # Aang, ~ C-x1,99, a
Ar)
Aa, | = Minimize(SSE,A12,Az1,C)
te
Ans,
(b) Plot data and fit
GeRT1x2(x1 ,x2)
(Agpxd + Aizx2 - Cx1-x2)
GeRT (xt ,x2) := GeRTxIx2 (x1 ,x2)-x1-x2
tpl (x x2) == 32? Ay +2(Azt Arz Cal +3-Ca"|
Iny2(x1 x2)
xi? [aa +2-(Ai2~ Ag - C)x2+3-C 22°]
108 Xi i= 01 j-.01 Xo I= Xi
i
GERTX1x2; e
GeRTx132( Xr, X:
Ppa) -0
i)
ty (% 9)
ing)
sa)
os 8a OSs
nipX
ape eXtp XI AI XI,
362
(c) Plot Pxy diagram with fit and data
y1@c1 ,x2) := exp(Iny1 (x1 ,x2))
y2(xl x2) := exp(Iny2(x1 ,x2))
Xpyl(X1,. Psaty + X2-¥: »X2,
111 (%1,oX2,} Psat + X2,-72(X1 0X:
Peas ) Psat
X171(X1,.Xo, Psat
Peale
‘ale,
P-xy Diagram from Margules Equation fit to GE/RT data,
40
° 02 oF 06 08
XI ¥1jo%1 Veale;
900 P-x data
© Pry data
P-x calculated
— Py caleulated
3GERT; : GeRT(X1,0%2, GERT;
n
In] —
12,
353
0.004 T
° Po6 0
8GERT; 0 fee ooo Blny1y
. a X= 9.005
1
a: 05 1 “005
Xi,
Calculate mean absolute deviation of residuals
+ 7 _——»
mean [SGERT| ) = 9391 x 10 smean([sinyiy2}) = 0.021
(e) Barker's Method by non-linear least squares:
Margules Equation
yi (xt x2.A12,A21C) = exp] (x2)°-[A12+2-(Aai - Ai2- C)-x1
L+3-C-x12
yo(xt x2,412,A21,C) -)
[Ao1 +2-(A12 Any C)-x2 ‘|
|ys0007
Minimize sum of the squared errors using the Mathead Minimize function.
Guesses: Ay = 03 Agi = C02
}
4
88E(A12,Aa1,0) = S/Pi~(aa,71(%1,.¥2,,A12,A01 ,C}Poaty ...P
Pa 12(m1,.%2, Aiz Agi ,C)-Psaty
AQ
Agr | := Minimize(SSE,A2,A21,C)
c
Ans.
354
Plot P-x,y diagram for Margules Equation with parameters from Barker's
Method.
Pea X1p71(X1 Xo, »Aiz Aan C} Psat
Hacetipaeed
+X2 2(X1;-%2;-A12,421 0) Psatz
x a1i(X Xoj-Ar2,A2t .C} Psat
Yiealey = Pas
i
90
80
60
so
49
0 02 04 06 Os 1
X14 X1, Yale
19¥1j X15 ¥reale;
900 P-x data
° Py data
—— Px calculated
xy calculated
Peale, = X1;°71(%1,9%2,-A12+A21 .C) Psat
+ N2,-¥2(X1, 9X2, -Ai2,Aa1.C} Psat
x1,°79(%1,.%2, -A12,A21,C) Psat
Yieale
E Peatc,
355,
Plot of P and y1 residuals.
Peale;
kPa
XxX
(rvteay
en 05 1
x
XXX Pressure residuals
© yl residuals
RMS deviations in P:
RMS = 0.068kPa
366
12.8
(a) Data:
(0.0523 1.202 1.002
0.1299 1307 1.004
0.2233 1295 1.006
0.2764 1.228 1.024
0.3482 1.234 1.022.
O.4187 1.180 1,049
x1 =| 0.5001 ‘L329 y=] 1.092
0.5637 1.120 1.102
0.6469 1.076 1.170
0.7832 1.032 1.298
0.8576 1.016 1.393
0.9388 1.001 1,600
0.9813, 1.003, 1.404, J
sz rowel) im lan
GERT; = x1,In(y;,) +32;n(72,)
(>) Fit GE/RT data to Margules eqn. by linear least-squares procedure:
GERT;
Slope = slope(X.Y) Intercept := intercept(X, Y)
Slope = 0.247 Intercept = 0.286
Ajg = Intercept Agi := Slope + Ai
A Ag) =0534° Ans,
YIG&1.x2) s= exp] x27 Aa +2-(Ag) —Ardac ||
P2681 x2) += exp x17 Aas +2-(Ar2— Ar) ] |
GeRT (XT x2) = x1 -In(y1 (x1, x2)) + x2-In (y2(x , 2)
357
Plot of data and correlation:
GERT;
aac
GeRT(x1,.%2)) 0.4
mea)
infr(er, 22)
oa
0
x,
{c) Calculate and plot residuals for consistency test:
8GERT; = GeRT(x1,.%2,) - GERT}
Biny 172 = In| = In|
(PQ) Le,
368
8GERT)= — Sly 1y2i =
on
EES aes
|-2.264-10 3] |-9.153-10 “5
Sos a
sao 30
2.87410 -3) -0.019) sted
[22210 3| 5.93410 >
aie wom] rh
|-1.553-10 -3| 9.5910 3!
|-8.742-10 + 9.139-10 3} °
6.96210 5) l 0.011
'9.025:10 5 9.028
423640 + 188
Calculate mean absolute deviation of residuals:
rmean(|SGERTI) = 1.615« 107? mean [Biny 12 ) = 003
Based on the graph and mean absolute deviations,
the data show a high degree of consistency
12.9 Acetonitrile(1)/Benzene(2)- VLE data T= 318.15-K
31.957 0.0455 0.1056
33.553 0.0940 0.1818
35.285 0.1829 0.2783
36.457 0.2909 0.3607
36.996 0.3980 04274
37.068 0.5069 0.4885
Ps kPa x= we
36.978 0.5458 0.5098
36.778 0.5946 0.5375
35.792 0.7206 0.6157
34.372, 0.8145 0.6913
32.331 0.8972 0.7869
30.038 0.9973 0.8916
359
—
ty)
Paty = 27.778-KPa Psaty = 29.819-kPa
Calculate EXPERIMENTAL values of acti
Gibbs energy.
a= (om
coefficients and excess
> —>»
yee yo? —_——-
ae GERT = (xy-n(y}) + x2-In(72
" yppeat, 1? Sp Paty (xrin(ry) +92trGi9))
—
GeRTaix2:= SERT se rows) an = 12
xVx2
(a) Fit GEART data to Margules eqn. by nonlinear least squares.
Minimize sum of the squared errors using the Mathead Minimize function.
Guesses: Ag=-03 Ane -05 C02
SSE(A12,A21.C) = Si [sern (Aat x1, F A12x2, — Cor x
(1.028
1.155 Ans.
cP Nosy
Minimize(SSE ,Ay2,A21,C)
(b) Plot data and fit
GeRTH1x2(X1 ,x2) 25 (Azi-xl + Arzx2 - C-x1-x2)
GeRT(x! ,x2) = GeRTX1x2(x1,x2)-xT-x2
byl (xt x2) = x2? [Arg +2(Aaq —Ar2—C) xt $3-C-x1|
Iny2(x1 ,x2) = x1? [ani +2-(Ai2— Ag -C}x2+3-Cn2?|
js t.lol Xp, = Ol j-.01 Xo, = 1-X,
1, i 2, 4,
360
GERTx1x2)
GeKtrsha(%.%2)
ta(ru)
‘wrt(Xi,.X2)
Infra)
n(x, %2,)
0 02 04 06 O08 1
X1oX1) 84X11 XI
1X, XI oI XI
(©) Plot Pxy diagram with fit and data
‘YAGI ,x2) := exp(Iny1 (xt .x2))
72(x1 ,x2) 2= exp (Iny2(x1 .x2))
ie = Xp -71 (Xp, ,.Xz_}-Psaty + X2,-y2(X1,,X2_}-Ps
ale, = Xayrtl(X1, Xo,}-Psats + Xoy-y2(Xa, Xo} Psats
Xi (x
Yieale, =
‘i
361
P-x,y Diagram from Margules Equation fit to GE/RT data,
38
36
0 02 oF 06 o8 1
x1pe¥te Xt, Yeale
1M Xt; eae;
P-x calculated
— Pry calculated
(d) Consistency Test: SGERT; == GeRT(x1,.¥2,) - GERT}
(ri ha, 2) y fy.)
Sly 1y2j = a Eee No iG ra
(1,2) J
ont ; ~
| - : of x we x
SGERT; of. Pee eco any ty2; o
Per Pea
ae oO 05 1 0.05
Calculate mean absolute deviation of residuals
es
smean( |scert!) = 6.237104 mmean|[Siny1y2}) = 0.025
(©) Barker's Method by non-linear least squares:
Margules Equation
ya (xl x2,A12,A21,C) = exp) (x2)*[ Ain +2-(Aai — Ai2—C)-x1
[43-C-x1? |
als 32.A12.A21.€) a. +2(Ai2~ Ai ~ O)2 |
+3-C-x2?
Minimize sum of the squared errors using the Mathead M
Guesses: Aigi= 0.3 Agi = -0.5 Cs
SSE(Az.A21.C) |
2
x71 (,o%9,-A12,A21 C} Psaty .. YP
2(xi 82,-A12,Aat C) Psat
An
Agi b= Ans.
Cc
Plot P-x,y diagram for Margules Equation with parameters from Barker's
Method,
Peate; = Xa,7i(X1 Xa, .A12,Aa1 ,C)-Psat «.
Hacer one
+X2y-72(X1,.X2,,A12,Aa1 C)-Psata
X171(%1,-X2, A122) .C}Psaty
esadewvaedeal
Yieale;
Peale
ey
363
38 OO
36
34
32
30
28
6G 02 oa 06 08 1
Xp YI, 0X1, ¥teate
p00 Px data 2 H
2 Pry data
P-x calculated
— Py caleulated
Peate, = x1, 71[¥1,9%2, A12,A21 ,C} Psaty
+9,-72(%1,0%2,-A12,A21,C}-Psata
Nip 11 (81,999, -A12,A01,C)-Psaty
Yieale, oa
364
Plot of P and yl residuals.
06
04 peace
Pi-Peate
i 02 .
~2} ,
eo 05 1
x,
XXX Pressure residuals
© yl residuals
RMS deviations in P:
RMS = 0.04kPa
365
12.12 It is impractical to provide solutions for all of the systems listed in the
table on Page 460; we present as an example only the solution for the
system 1-propanol(1)/water(2). Solutions for the other systems ean be
obtained by rerunning the following Mathcad program with the
appropriate parameter values substituted for those given. The file
WILSON.med reproduces the table of Wilson parameters on Page 460
and includes the necessary Antoine coefficients.
Antoine coefficients:
1-Propanol: Al := 16.0692 BI Cl := 204.00-K
a
‘Water: AD := 16.2620
Psaty (T) s= ex al 7
Psata(T) =
B2
xp] A2 - ————=___ | kPa
T — 273.15:K) + C2
Parameters for the Wilson equation:
3
75.14
vi
mol
al = 775.48-2a
mol
v2 ~al2 { -a2
AIQ(T) Boo 22) A21(T) wer 21)
vi RT RT
x1+x2-Al2(T) x2 +x1-A21(T)
(x1 +32-A12(1))
| ( AI2(T) AZT) |
exp|-x1{ AZO) __ A)
xL+x2-A12(1) x2 #x1-A2I(1),
(x2 + x1-A21(1))
evfo[ A12(T) AQT) I
yl (xl ,x2,T) = a
201, x2,1) =
366
P-xy diagram at Tos (60427315) K
Guess: P := 70-kPa
Given P= xb-yl(xl 1 = x1,1)-Psat (1)
+ (1 = x1)-y2(x1, 1 + x1, T)-Psato(T)
Peq(sx1) := Find(P)
xI-y1 (1,1 —x1,7)-Psati(T)
yeq(x!) x:= 0,0.05.. 1.0
Peq(xi)
Peq(x) _
yes) = ke
a 0 9.024
0.5; o3i7
oa 0.368
[ors 0384
oz 0398
035 oa08
[es oata
[035] [042s]
oa 032
[045] aa |
os] 084
O58 O68
08) O84
[bas] 0503}
o7 om?
[7s] O587
03 0598
O85 OBA
0a] O71
0.85 0825;
t 7
04 06 08 1
x. yea)
12.13. It is impractical to provide solutions for all of the systems Yisted ta the
table on Page 460; we present as an example only the solution for the
system I-propanol({)water(2). Solutions for the other systems can be
obtained by rerunning the following Mathcad program with the
appropriate parameter values substituted for those given. The file
WILSON.med reproduces the table of Wilson parameters on Page 460
and includes the necessary Antoine coeffi
Antoine coefficients:
L-Propanok = Al :=
6.0692 BL == 3448.66K Cl
104,09.K
Water: 16.2620 B2 == 3799.89-K C2. = 226.35-K
Bi |aPa
Psat; (T) ——<$—$—_——
ae (T-273.15-K) + Cl
B2 i)
Ag eceeceeee DCE
(T= 273.15-K) +C2 | .
368
Psata(T)
Parameters for the Wilson equation:
3 3
75.14. v2 = 18.07:
‘mol mmol
vie
ad = 775.48 aa = 1351.90.28
mol mol
oxf o( A201) an) YI
ate xi+ x2 +xPA21(T) }]
at (x1 #x2-A12(T))
wfn{ tm so)
(xl +x2-A12(1) xd Deararicn)}
(x2 + x1-A21(T))
y2G1,,x2,T) =
‘T-xy diagram at P == 101.33-kPa
Guess: T= (90 +273.15)K
Given p= xteyl(x1,1 —x1,7)-Psaty(T) .
+ (1 = x1)-y2¢x1,1— x1, 1)-Psatg(T)
Teq(xl) := Find(T)
xl-y1(x1 1 — x1, Teq(x1))-Psaty (Teq(x1))
=
yeq@l) 7
x= 0,0.05..1.0
369
Teg)
x= yeq(x) = K
o a ag
oa 0305 364133
on 0368 Bear
O15 0383| 361805)
02 0.308 189)
025 0.408 31251
03 ‘oai9| 364.067
035 08) 35095
oa aaa 360876
Zs oasa se08aa
os 0.463] 360,068
055 0.486] 350549
oe ‘508 wat
065 0528 e371
o7 0.555 BeL7e7
‘ors 089 362.208
08 0633 365.073
085 0687 364.173
a9] o768 a
095 0859) 67 RB
7 7 arom,
‘T.xy Diagram at P
315,
Teal). 370
x, yogi)
370
12.14 It is impractical to provide solutions for all of the systems Tisted in the
table on Page 460; we present as an example only the solution for the
system -propanol(1)/water(2). Solutions for the other systems can be
obtained by rerunning the following Mathcad program with the
appropriate parameter values substituted for those given. The file
NRTL.med reproduces the table of NRTL parameters on Page 460 and
includes the necessary Antoine coefficients.
Antoine coefficients:
1-Propanol: Al :
16.0692 BI = 3448.66-K CI = 204.09-K.
Water: = A2:= 16.2620 B2 := 3799,89-K
226.35+K
Psaty(T)
Psatp(T) := exp] A2- 82 ___ | xpa
(P= 273.15K) +2
Parameters for the NRTL equation:
biz z= 500.4024 ba i= 1636.57. 5081
mol mol
+12(T) bar
RT
G12(T) := exp(-a-412(1)) G21(T) = exp(-c-121(7))
2 an
A(t ,x2,T) == exp} x2" £21(7)| ————___|_
xi +x2-G21(1))
4 G12(1)-2121) i
(24 x1-G12(T))? |
72(x1 32,7)
| ene { = S2@ y
x2 +x1-G12(T),
ee G2L(T)-21(7)
(x1 +x2-621()"
3m
P-xy diagram at T := (60+273.15)-K
Guess: P := 70-kPa
Given pm xteyl(xt 1-1, 1)-Psat (1) «.
+ (1 = x1)-y2(x1, 1 = x1,T)-Psatg(T)
Peq(xl) := Find(P)
xl-yl (x1, 1 ~ x1, 1)-Psaty(T) 0,0.05.. 1.0
vyeq(xl) :=
0.05;
ar
a5
a2 |
[ozs]
03]
035;
oa
035
05
055)
(8)
08s
Or
a76
08.
035
[09]
095)
372
Pxxy Diagram at T = 333.15K
35
x. yeats)
12.15. It is impractical to provide solutions for all of the systems listed in the
table on Page 460; we present as an example only the solution Tor the
system f-propanol(1)/water(2). Solutions for the other systems can be
obtained by rerunning the following Mathead program with the
appropriate parameter values substituted for those given. The file
.med reproduces the table of NRTL parameters on Page 460 and
ludes the necessary Antoine coefficients,
Antoine coefficients:
1-Propanol: Al 2
6.0692 BL = 3448,.66-K = Cl = 204.09-K
Water: 6.2620 B2:=3799.89-K C2. = 226.35-K
Psat) (T) SSH BUELL
L (T= 273.15-K) +C1 |
Psat(T) me |
xp] A2~ le:
@-273.15K) +2
Parameters for the NRTL equation:
S081
wa
RT
G21 (T) = exp(-a-221(1))
w21(T) =
r \2
| G21) ]
YUGL x2, T) = exp) x2’ | im So) “||
| G12(T)-12(T)
|
(2431-61201)? JJ
of (_ 6am _Y |
AGI Y2,T) = ep] a) e120) S|
,_21(1)21) |
(xl +32.621(1))” J
T-xy diagram at P := 101.33-kPa
Guess:
= (90 + 273.15)-K
Given p= xleyl(xd,1 = x1, T)-Psath(T)
+(1=x1)-y2(x1, 1 ~ x1, T)-Psaty(T)
Teq(xl) := Find(T)
xL-y1 (XE , | — x1, Teq(x1))-Psaty (Teq(x1))
———eoo
yeq(xl) =
374
0,0.05...1.0
Teq(x)
x= yea(x) = K
a ° a8
0.05 oer 363.579
[23} 0.379) Set 715
0: 0.308 Fae
02 ‘0403 1035
025 ‘o.a08 30S
03: oai6' 308
036: 0.35 ore
04 0.436) 350882
045) 0888 380.64
05 0.68 360677
O55: O61 350767
08 ‘0507 35083
065 0828 Sa
07] 0865 SII
O75 0588 362.18
08 083 Ba
[08s] | 0.683) | 363.043]
08] o764 355.361
0.985 ‘0.888 357353
a 1 370.282
T.xy Diagram at P := 101.33-kPa
375
x, yeq(x)
375
12.16 It is impractical to provide solutions for all of the systems listed in the
table on Page 460; we present as an example only the solution for the
system J-propanol(I)Avater@). Solutions for the other systems can be
obtained by rerunning the following Mathead program with the
appropriate parameter values substituted for those given. The file
WILSON. med reproduces the table of Wilson parameters on Page 460
and includes the necessary Antoine coefficients.
Antoine coefficients:
1-Propanol: Al == 16.0692 BI = 344866K Cl = 204.09-K
Water: A2:= 16.2620 B2 = 379989K C2 = 226.35-K
Bl ]
Psat (T) := exp) Al - 51 __
fe ox @-m.isH+o1 |”
Psata(T) = ex A - = lea
(T= 273.15-K) + C2
Parameters for the Wilson equation:
cm’ °
Vi = 75.14- v2 = 18.07
mol ‘mol
aid 775.48 a2 = 1351.90.22
mol mol
ny we N2_ of -al2 V1 oof 221
AL2(T) = exp] SEE A21(T) := exp! St
: vi of 32) ey v2 RT
exo AIT) ary YY
G1 2,7) = SX2-A12(T) x2 #x1 = x-A21(T)) |
Gt +32-a127))
oof-at{ AP Aan)
2O«L,x2,7) x14x2-A12(T) x2 +x1-A21(T))]
(2 +x1-A21(T))
376
(a) BUBLP: 1 := (60+273.15;K xl: x2i= 1 ~xl
Guess: P= 101.33-kPa, yl: y= i-yl
Given yl-P = xl-yl(xt ,x2,T)-Psaty(T)
ylty2=1
y2P = x2-y2(x1 ,x2,T)-Psat2(T)
{Poul
yl | := Find(P,yl,y2)
31,27kPa yl= 044 y2.= 0/586 Ans.
()DEWP: —T := (60+273.15)K yl
P= 101.33-kPa xl = 0.1
Guess:
Given yl-P = xt-yL(x1,x2,T)-Psaty(T)
xl 4x25
y2P x2+y2(x1 ,x2,T)-Psatz(T)
x1 = 0,042 x2= 0,958 Ans.
(© P,T-flash Calculation
Padew + Poubl
= T:s (604273.15)K zl = 03
Guess: viens Ol x2i=1-yl
yrs 0d yl t-xl
Given x1-y1 G41, x2, T)-Psaty (TD)
yl= 2 xt SL
P
x2-72(x1 ,x2,T)-Psata(T)
ye tty
377
xI(1-V)+yl-V= zl Eq. (10.15)
xl
x2
yl | = Find(xt ,x2,y!,y2,V)
y2
Vv
x130.08 x2=092 -y1-=.0.351_ y2= 0.649 -V = OBL
(@ Azeotrope Calculation
‘Test for azeotrope at: T= (60 +273.15)-K
71(0,1,T) = 21.296 y2(1,0,T) = 4.683
«1129 = woes 112g = 21.696
Psat (7
a2} se xi) = 0.218
Since one of these values is >1 and the other is <1, an azeotrope e
See Ex. 10.3(e)
Gu P= 10133kPa x1 = 0.3 xis l-yl
yl 03 ye
Given yL-P = x}-71 (x1 ,x2,T)-Psat(T)
y2P = x2-42(x1 x2, T)-Psaty(T)
xl4x2=1 0 yl+y2=
x2
yl ind(x1 ,x2,y1,y2,P)
y2
\Paz
Pag = 31,453kPa x15 04403 yl =0.4403 Ans.
378
12.17
Its impractical to provide solutions for all of the systems listed ia the
table on Page 460; we present as an example only the solution for the
system I-propanol(1)/water(2). Solutions for the other systems can be
obtained by rerunning the following Mathead program with the
appropriate parameter values substituted for those given. The file
NRTL.med reproduces the table of NRTL parameters on Page 460 and
includes the necessary Antoine coefficients.
Antoine coefficien
L-Propanol: Al := 16,0692 BI := 3448,66-K Cl = 204.09-K
Water: A2 == 16.2620 B2 = 3799,89-K C2 = 226.35-K
BI ]
Psaty(T) := exp] Al — ——_—____ ‘kPa
a“ o[ (1 —-2B.15K) + Cl |
B2
82 lips
(P= 273.15-K) + C2
Psat(T) = cl 22 -
Parameters for the NRTL equation:
biz = 500.4024 bar = 1636.57 2
‘mal mol
bi2
1121) = Ot 217)
G12(T) = exp(-a-t12(T)) G21(T) 35 exp(-a-+21(T))
r 2 7
yl (x1 x2) := exp! x27 221¢7) (ae) wl
| x1 +x2-G21(T)) “| |
i 4 G12 7127)
| (2+x1-612(1)"
= exp| x12] x12¢7) (22m }
x2 +x1-G12(T),
» @2L)-21(7)
J
Loa +22-G21(1))°
379
(a) BUBL P: T= (60-+273.15)-K xis030 0 = I=x1
Guess: P = 101.33-kPa yl: 04 yl
Given yl-P = xt-yl(xl ,x2,T)-Psat(T)
yleyel
y2sP = x2-y2(x1 ,x2,T)-Psatz(T)
Pubt
yl Find(P, yl ,y2)
Ly
Phub) = 30.98KPa yt $.0:397 y2= 0.603. Ans.
(b) DEW P: T= (60+273.15)-K yl y2i=h-yl
Guess: Ps= 101.33+KPa x20) Qe 1-x1
ee yL-P = x1-yl (X1,x2,T)-Psati(T)
xl+x2=
y2:P = x2-y2(x1 ,x2,T)-Psaty(T)
Puew
x ind(P,.x1 ,x2)
x2
Page 2 207kPa xl = 0.037 x= 0963 Ans.
(c) P,T-flash Calculation
Pdew + Publ
= Ts (604273.15)-K 21-03
Guess: Vv =05
yl 0.1
a xi-71 (x1 ,x2,T)-Psaty(T)
Given eee xL+x22 1
P
x2-Y2(x1 x2, T)-Psat2(T)
ye ylay2=1
xb-(I-V)+ylV= zt Eq. (10.15)
380
xl
[2
yl | := Find(x1 ,x2,yl,y2,V}
|
ww
xls
059 xQAO9I y= 0.345. y2= 0.655. V = 0.841
(@) Azeotrope Calculation
‘Test for azcotrope at: T := (60 + 273.15)-K
IQ, 1,T) = 19.863 ¥2(1,0,T) = 4.307
1(0,1,T)-Psaty (7
aly = OED ESD e129 = 20.236
Psatz(T)
Psat) (T)
al2y =
20,0, T)-Psata(T)
Since one of these values is >1 and the other is <1, an azeotrope exists.
See Ex. 10.3(¢).
Guess: P= 101.33-kPa xl
yl
x2
0. y2
Given yl-P = xl-yI(xl x2, T)-Psati (1)
y2:P = x2-72(x1 x2, T)-Psatg(T)
xl4x2=1 ylty2 xl=yl
(x1
| x2
yl | = Find(xt ,x2,y1,y2,P)
y2
(oa
XI = 0.4204 yl = 0:4204 Ans,
381
12.18
It is impractical to provide solutions for all of the systems listed in the
table on Page 460; we present as an example only the solution for the
system I-propanol(1)/water(2). Solutions for the other systems can be
obtained by rerunning the following Mathead program with the
appropriate parameter values substituted for those given. The file
WILSON. med reproduces the table of Wilson parameters on Page 460
and includes the necessary Antoine coefficients,
Antoine coefficients:
L-Propanol: Al := 16.0692 BI = 3448.66-K C1 := 204.09-K
Water: A2:= 16.2620 C2 = 22635-K
Psat) (T) = exp) Al a
Be
Psat2(T) := exp} A2 - ————_—_______ |p
ae. of 273.15 al .
Parameters for the Wilson equation:
3
Vie 75:14
mol
al2
ALT) = a on 2
ror y
one AID) | aaa
x1+x2-A12(1)_ x2 +x1-A21(T) }}
YUGKL x2, 1) =a
(x1 +x2-A12(7)
eof{ 82 sic
201,27) = XL +x2-A12(T) x2 + x1-A21(T)}}
(x2 + x1-A21(1))
382
(a)BUBLT: Pp
01.33-kPa xl :=03 x2:=1-x1
Guess: T= (60+ 273.15)K yl=03
yL-P = xI-yl (xt ,x2,T)-Psat;(T)
yl+y2=1
y2P = x2-y2(x1 x2, T)-Psaty(T)
oe
jvt |= Find(T. 1.92)
\ v2
Thub = 36L07K yis04i9 -y2= 0.581 Ans.
(b)DEWT: P= 101.33-KPa yl 03 y
Guess: T = (60+273.15)K xls 0.1 2
Given yl-P = xl-yl(x1,x2,T)-Psati(T)
xl #32
y2-P = x2-y2(x1 ,x2,T)-Psatz(T)
Taew |
xl | := Find(T,x1,x2)
2)
Tew = 364,29K xh = 0.048 32= 0.952) Ans.
(©) PyT-flash Calculation
Tew + Tou
T oe P= 10133kPa 21 := 0.3
Guess: v=05 ee
yl=01
x1-y1(x1 ,x2,T)-Psat(T)
(Given fete y| tee eereen ne ep ee
P
x2-y2(ott x2, T)-Psaty(T)
ee yl+y2=1
XL(I=V)+yl-V = zl Eq. (10.15)
383
2
yl | := Find(xl ,x2,yl,y2,V)
y
v
x1 = 0.089 x2=0911 yl= 0.35)
(d) Azeotrope Cal
‘Test for azeotrope at: 101,33-kPa
ro =[(—21_ cr) s27.15
Pp)
Al = In| —
KPa)
Th2 := Ee = C2 | +273.15-K)
10,1 ,Tb2) = 16.459
71, 1,T)-Psat (Tb2)
Pp
PB
2,0,T)
a2; :
2(Tb1)
y2= 0.649
V = 0.805,
TbI = 370.232K
Th2 = 373.15K
y2(1,0,Tb1) = 3.781
al2p = 19.598
ail2, = 0.282
Since one of these values is >I and the other is <1, an azeotrope exists.
See Ex. 10.3(¢), Guesses:
T= (60+273.15)K xl = 04 x25
yli=04 y2:=1-x1
Given yL-P = x1-y1(x1,x2,T)-Psaty(T) x1 #x2 = 1
y2P = x2-42(K1,x2,T)-Psaty(T) yl +y25 1 xleyl
384
12.19
(xt)
&
YL | = Find(xt ,x2,y1,y2,7)
Tez,
Taz = 360,843K xls 0.4562 yl= 0.4562' Ans.
It is impractical to provide solutions for all of the systems listed in the
table on page 460; we present as an example only the solution for the
system I-propanol(1)/water(2). Solutions for the other systems can be
obtained by rerunning the following Mathcad program with the
appropriate parameter values substituted for those given. The file
NRTL.med reproduces the table of NRTL parameters on Page 460 and
includes the necessary Antoine coefficients.
Antoine coefficients:
1-Propanol: Al := 16.0692 BI = 3448.66-K Cl := 204.09-K
Water: A2:= 16.2620 B2 = 3799.89-K C2 = 226.35-K
seeneeiDleaaaaea |e
(T=273.15-K) + C1 }
a
Psaty (T) = ol -
B2 claee
Psat(T) := exp] A2~ 2° __
a of (P= 273.15K) + 2 |
Parameters for the NRTL equation:
cal
bi2 = 500.40 bat = 1636.57 0.5081
mol mol
bi2
+12(T) = 2 b21
221(T) = ——
RT Mae T
G12(T) := exp(-a-112(T)) G21 (T) = exp(-a-r21(T))
385
2 ou)
AGEL x2.T) 2 exp) 32 a(S
|, gi2m-12c)
L g2+xr-ciany?
GIT)
x1-G12(1)
G21(T)-21(1)
Eee Ate
(xl 442-421 (1)?
(@)BUBLT: P
01.33-kPa xl
Guess: T= (60+273.15)-K yli= 03
Given yl-P = xl-yl(X1,x2,T)-Psati(T)
yl+y25 1
y2eP © x2-y2(x1 ,x2,T)-Psatg(T)
( ee
yl | := Find(T,yl,y2)
( ye
Toobt = 360:81K yi= 0.416 y2= 0584 Ans
(b)DEWT: P= 101.33-kPa yl = 03 y2:= 1-xl
Guess: T = (90 + 273.15)-K x1 = 0.05 x2izl-yl
Given yl-P = x1-7i(x1 ,x2,T)-Psaty(T)
y2P = x2-72(x1,x2,T)Psaty(T) EZ I
(Taew
xl t= Find(T, x1 ,x2)
x2
Taow = 364.28 xt = 0.041 Ans,
386
(© P,T-flash Calculation
Taew + Toubt
101.33-kPa zl := 0.3
2
Guess: x= 01 x2 1-yl
yl = 0.1 y2:=1-xh
I-y1. Gel ,x2,,1)-Psat (T)
Given ys Sere re alee
x2-y2(x1 ,x2,T)-Psato(T)
ee yleyoet
ey
xI-(1-V)+yl-V = zl Eq. (10.15)
x1)
2
yl | = Find(x1 ,x2,yl,y2,V)
y2
v
xf=0,069: x2=0.931 yl = 0353 y2=0647 V= 0814
(d) Azeotrope Calculation
Test for azeotrope at: P := 101.33-kPa
Tel = a = 0 +2nsx] ‘Vol = 370.232K
Al-|h |
. ( yo)
‘Tb2 = 373.13K
V1 (0,1, 162) = 14.699 (1,0, Tb1) = 4.051
387
yl (0, 1,T)-Psat} (Tb2
aly = P a2; = 0.271
21,0, 1)-Psata(Tb1)
Since one of these values
See Ex. 10.3(¢). Guesses:
T= (904273.15)-K x1 =0.4 x2
>I and the other is <1, an azeotrope exists.
yl yliz 04 y2
Given yL-P = xl-yl(x1 ,x2,T)-Psaty(T) x1 +92 = 1
y2P = x2-y2(X1,x2,T)-Psatg(T) yl +y2= 1 xleyl
(x
x2
yi ind(X1 ,x2,y1 2,7)
y2
(Taz
Taz = 360.641K xb = 0.4477 yl= 0.4477. Ans.
12.20 Molar volumes & Antoine coefficients:
74.05 14,3916 2795.82) (230.00 )
40.73 16.5938 3644.30 C =| 239.76
18.07, 16.2620, 3799.89, 226.35
Bi
Psat(i,T) := exp} Aj 7 lira (65 +273.15)K
y
73. S)+ci |
0 161.88 291.27
583.11 0 107.38 |S
i
l4ago1 46955 9) ™
Wilson parameters:
388
(a) BUBL P caleulation: No iteration required.
x03 x04 ag 1x19
ix, T) = ex9{ 1 (“e vain) fy
| XpA@.1,7)
LF aiid
| (eden |
-y ,x, T)-Psat(i,T)
Publ = 7 XY, 1) Psat(i,T)
Pub
Phubi = H17.6kPa Ans.
(b) DEW P calculation:
yi 03 y2= 0.4 yse byl nye
Guess: xp 0.05 x02 © ages ta xpo ar P= Phun
Given
Poy = xy-7(1,x,T)-Psat(I, 7) Peyg = xg-7(2,x,T)-Psat(2,T)
Peyg = x3-y(3,x,T)-Psat(3,T) Dx 1
{ox i
\
x2
| = Find(xy ,x2,x3,P)
| %
(Paew
389
0035:
0.188 Poy = 68.05KP2 ang
Xo
Paew +P
(© PyT-flash calculati T= 33815K
403 04 23:2 1~2-2
Guess: V=05 Use x from DEW P and y from BUBL P as initial
guess.
Given
Pry © x-y(1 xs T)-Poat(1 1) w= VWtyreV =
Peyp © xo-y(2.x,T)-Psat(2, 1) xp(1~ V) #y2-V 5 20
Prys = x3-y(3,X,T)-Psat(3,T) x3-(1- V) +y3-V = 23
Dae
V=0.677 Ans.
380
12.21 Molar volumes & Antoine coefficients:
Antoine coefficients:
14.3916 2795.82 230.00
16.5938} B:=| 364430] C:=| 239.76
16.2620 3799.89 226.35
Bi
T= (65+273.15)K ——Psat(i,T) == exp] Aj- kPa
(Z 2. is}+c,|
NRTL parameters: K
0 03084 0.5343 0 184.70 631.05
0.3084 00.2994 222.64 0 -253.88 =
mo
0.5343 0.2994 0 (1197.41 845.210
a
Gi, = exp(-c, 5+
ae jena)
(a) BUBL P calculation: No iteration required.
x03 x= 04 xg = 1x1 x2
[ou G5,i¥)
VG,x,T) == exp} 4
: ae YG 51
Prost =) si7G,x,T)-Psat(i,T) ys sree rene)
Publ
(0,525
[oar |: Phubl = 115:8kPa Ans.
391
(b) DEW P calculation:
yi03 yee 04
Guess: xj 5005 x2 Pout
Given
Poy = xp-y(1,x,T)Psat(1,T)Peyp = xo-y(2,x,T)-Psat(2,T)
PaesrGaDPaGy Yael
(x :
x2
2= Find(x, ,x2,x3,P)
%
\Paow.
0.038
x= |.019 Pay 2689KPa ang
072)
Pew +P
(©) PyT-flash calculation: ~ bub
me03 mi04 32 1-2-2
Guess; V:= 0.5 Use x from DEW P and y from BUBL P as initial
guess.
Given P-y, = xp-y(1,x,T)-Psat(1,T) xi(l-V)+yr-¥ 3 zy
Pryp = xory(2,x,T)-Psat(2,7) x(1-V) +y2V = 2p
Pryg = x3-7(3,x,T)-Psat(3,T) xa(1-V) +y3-V
1 yee
a
392
XL
x2
X3
yi | 2= Find(xy ,x2,x3,1.92,93,V)
y2
33 |
i)
foils 039i
x=] 0346 y=} 0427 V-=0668 Ans.
- \0.536. A0.182
12.22 Molar volumes & Antoine coefficients:
74.05 14.3916 2795.82 230.00
V:=|40.73| A :=| 16.5938] B= | 3644.30] C=] 239.76
18.07, 16.2620 3799.89, 226.35
Psat(i,, 1OL.33kPa
0 161.88 291.27
Wilson parameters: 583.11 010738 =
mol
1448.01 469.55 0
AG,1,T) = —Lexp| Sed 3 3
4,1) @ of RT ) 7
(a) BUBL T ealeutation:
x27= 0.4 X35 T= xy xg
393
y(i,x,T) = expl 1 2
oy ete xpA(,i,7)
rd jA@.j.1)
Guess! T= 300K yy = 030 y= 03 yy= 1 -yl-y2
Given
Pry = xp-y(1,x,T)-Psat(1, 7) Pey2 = xa-y(2,x,T)-Psat(2,T)
P-y3 © x3-7(3,x,T)-Psat(3,T) Sx Psat(i,T)
fm
ve
= Find(y1 y2,93,T)
v3
Tub! )
(0.537
y=} 0.362 Tyuot = 333.97K Ans,
0.102.
(b) DEW T calculation:
y= 03 yo
Guess: x1 1= 0.05 xp s=
wietmyn
xg 1-xy-x2 T= Thubt
Given
Poy = xy-y(1,x,T-Psat(1,T) Py = xp-7(2,x,T)-Psat(2,T)
P-y3 = x3-y(3,x,T)-Psat(3,T) Dae 1
394
f
x1
© | c= Find(x1,x2,%3,7)
fee.
(teew
‘0.042
x= 0.203 Teow = 3474K Ans.
(0.755
Tew + Tt
(©) P.T-flash calculation: =e
M03 m=02 yxl-y-m
T = 340.694K
Guess: V:= 0.5 Use x from DEW P and y from BUBL P as initial
guess.
Given Peyy = xpy(1,x,T)-Psat(1,T) xp(l-V)+ypv = 21
Peyg = xz-y(2,x,T)-Psat(2,T) x2-(1- V) +y2V § 22
P-y3 = x3-y(3,x,T)-Psat(3.T) x3-(1- V) +y3-V = 23
Ds Py
x1
x2
x3
Yt | = Find(x1,x2.3,91,9293-V)
395
0,124) 0:537
0.169 y=} 0.241 V.= 0.426 ‘Ans.
0.706, 0.222
12.23 Molar volumes & Antoine coeffici
Antoine coefficients:
14.3916. 2795.82, 230.00
16.5938, B 3644.30 239.76.
16.2620. 3799.89, 226.35,
P := 101.33kPa Psat(i,T) Ain i(epeacccatet) ate PkPa
lk ~ 273, 33] +C
NRTL parameters: K
0 0.3084 0.5343 0 184.70 631.05 )
a:=/0308 0 02994] bi=| 22268 0-23.88 |
0.5343 0.2994 0 1197.41 845.210 J ae
i1.3 3 1 Pe
1i,j,T) =
3 GGT) = exp(-ai, 520.5.)
(a) BUBL T calculation:
x03 x04 x3 x1-¥2
YG... GGT 4
664.0%
yGi,x,T) := exp]
2k. .-Gk.i,D)
GG4,7) ( i |
VS aD -
pa 905.91 J
7
396
Guess: T= 300K yp 0.30 yg 03 ysis ly -y2
Given
Peyy = xpy(1,x,T)Psat(, 1) P-y2 = x2-7(2,x,T)-Psat(2,7)
Pey3 = x3-7(3..x, T)-Psat(3,T) SY xvGx.7 Psat(i,T)
(fu)
yp
= Find(y1,y2.¥3-1)
Thubl = 334.4K Ans,
(b) DEW T caleulation:
y= 03 -yiny2
Guess: x1 = 0.05 xg = 1-x1-42 T= Thubi
Given
Py, = xpyCx,T)-Psat(l.T) Py: 2,8, T)-Psat(2,T)
Pey3 = x3-7(3,x,T)-Psat(3,T) yx
x1 i
2
= Find (x1 ,x2,x3,T)
3
Few.
Tew = 347:5K Ans.
297
Teew + Tbubl
(©) P,T-flash calculation: T 5
T =.340.955K.
ms03 202 1-2-2
Guess: V:= 0,5 Use x from DEW P and y from BUBL P as initial
guess.
Given P-y) = xy-y(1,x,T)-Psat(t,T) xp(l-V) +yrV = 21
Peyp = xo-7(2,x, T)-Psat(2,T) x(1-V) +y2-V = 2
Peys = x3-y(3.,T)-Psat(3.T) xx(1- V) +93-V 5 23
Sse! a
ind (x1 ,x2,%3,y1,y2.95.V)
(0.132 (0.537.
*= | 0.173 0.239 V= 0415 Ans.
0.695, 0.224
398
12.26
x xg 1x]
ee VE = 7.92.
mol mol
VE 2= xy-x2-(45-x1 + 25-9]
By Bg. (2227): Vie VE+xpVi txV2 Y= 1059250
‘mol
By Eqs. (11.15) & (11.16):
Vbar) := V+x24_v Voar) = V-x;|¢-v
ox ax;
om om?
Var; = 105,022 Vbary = 105.925 Ans
a ‘mol ‘mol
Cheek by Eq. (11.11):
3
V r= xp Vbary + x9: Vbar v=10592 ok
mol
moles) == eae em moles
1 82
moles; + moles? moles = 25.455mol
moles),
“1 x1 = 0.503 xzis d=
moles:
on? iy
VE == xp-xp{ 1.026 + 0.220-(x} - x3) J VE = -0.256 5
mol mol
By Eq. (1227), V5 VE+xy-Vy+x9-V2 V = 88.1369.
mol
Vioal ‘= Vemoles Vigat = 2243¢m° Ans.
399
12.28
For an ideal solution, Eq. (11.78) applies:
Veoeal = (x1-Vi +X2:Va)-moles Vital = 2250em? Ans.
LiCL2H20 --> Li+ 1/2 C2 +22 +02 (1)
Li+ 12 C12 + 10 H20 —> LiC\(10 H20) 2)
2(H2 + 1/2 02 —> H20) 8
LiC12H20 + 8 H20() --> LiCI(10 H20)
1012650)-3_ (Table C.4)
441579-5 (Pg. 444)
(-285830-J) (Table C4)
AH] + AH2 + AH3
5895 (On the basis of 1 mol of solute)
Since there are if moies of soiution per mole of solute, the result on the
HCW.S H20)
2.44 di mad 5 with sign changed
12.30 Calculate moles of LiCl and H20 in original solution:
0.1-125 0.9-125
a= kmaol nyL0 kmol
BUC "72.39 20 18.015
nic) = 0.295kmol ny20 = 6.245 x 10° mol
20 ;
Moles of LiCl added: nic s= Gag'kmol ici = 0.472kmel
nu
Mole ratio, original solution: “22 — 91.18
ric
0
Mole ratio, final solution; = —~@°__ 8.15
ALicl+ 8LiCL
ALicl-+ AL ic = 0.7667kmo!
0.2949(LiCI@1.18 H20) -—> LiCl + 21.18 H20) (1)
0.7667(LiCI + 8.15 H20 —-> LiCI(8.15 H20)) (2)
0.2949 LiCL(21.18 H20) + 0.4718 LiC
> 0.7667 LiCl(8.145 H20)
Au = nuci(3 ai (Fig. 12.14, n=21.18)
mol)
AH2
(aici + atic) (= ») (Fig. 12.14, n=8.15)
mol,
Q:= AHI + AH2 Q=S14213kI Ans.
12.31 Basis: 1 mole of 20% LiCl solution entering the process.
Assume 3 steps in the process:
1. Heat M1 moles of water from 10 € to 25. C
2. Unmix 1 mote (0.8 moles water + 0.2 moles LiCl) of 20 % LiCl solution
3. Mix (MI + 0.8) moles of water and 0.2 moles of LiCl
401
Step 3: Guess M1 and find AHS solution from Figure 12.14. Calculate AH
for process. Continue to guess M1 until AH =0 for adiabatie process.
(0.8:mol+ My) oy
My := 1.3-mol 13 = ———___+ AHG = -33.16—
0.2-mol mol
3 = 10.5
AH := My-AHj ~ 0.2-mol-AH3 + 0.2-mol-AH3
AH = -0.061kI Close enough
soem Ans,
M+ T-mol
12.32 H20@5C 120 @25C€ (1)
LiCIG H20) LiCl+3#20 @)
LiCl+ 4120 LiCl(4 H20) GB)
aH = 1.509
mol
AH := 20756 From p. 444 (AH LiCl(s) - AH LiCl in 3 mol HO)
mmol
255-1 Krom Figure 12.14
mol
‘AH := (AH) + AH + AH3}-0.2-mol ‘AH =:7646.9051 Ans.
402
12.33. (a) LiCl+ 41920 —~-> LiCK4H20)AH := 2558 From Figure 12.14
‘mol
0.2-mokAH =-5.1k) Ans.
(b) LiCl H20) => LiCl +3 H20 (1)
LiCIG H20) + 120.
(s) - AH LiCl in 3 mot 11,0)
cl
AH = 20.756" From p. 444 (AH Lit
-25.5-AL From Figure 12.14
ak
2 mol
AH := 0.2-mol(AH; +A4Hp) AH =+0.949kJ Ans.
Li+172. C2 +H2+ 1/202 (1)
(c) LiC*H20
H2+ 1/202 @)
Lit 12CR 8
@
LiCl + 4820
LiCK4 H20)
YH20+3H20 -
Li
AH, = 712.58: x From p. 444 for LiCt-H,0
mol
From Table C.4 AH, H,0()
From p. 444 for LiCl
AH From Figure 12.14
AH := 0.2-moh(AH; +AH)+AH3+AHg) AH =/-1472k) Ans.
iC(4H20) (1)
+9120) (2)
(a) LiCl + 4420
4/9 (LiC(9 H20)
5/9 LiCl + 4/9 LiC9 H20) ——> LiCK4 120)
403
ass From Figure 12.14
mol
Ay = 4.324) From Figure 12.14
9 mol
AM,
AH := 0.2-mol(AH) +AHj) AH $-2.22k) Ans.
LiCl +3 120) (1)
LiCl +9 120) @)
LiCI(4 H20) (3)
\9 H20) > LiCl(4 H20)
(©) 5/6 (LiCK3 H20)
1/6 (LiCI(9 H20)
LiCl+ 420
5/6 LiC(3 H20) + 16
-20:156)- From p. 444 (AH LiCi(s) - AH LiC1 in 3 mol H,0)
mol
32.4) From Figure 12.14
mol
AN; := -25.5 From Figure 12.14
mol
“U561K) Ans.
AH := 0.2moh(AH; +4Hj+4H3) AH =
® 58 Lic*H20 LiH12 C2 +H2+1202) (dy
5/8 (H2 + 1/2 02, 20) @
3/8 (LiC(9 H20) LiCl + 9 820) 8)
5/8 (Lit 12 C2 Lich @
iCl + 4 20 LiCK(4 120) 6)
'H20 + 3/8 LiC\(9 H20) —~-> LiCl(4 H20)
58.
Aly = 2712.58) From p. 444 for LiCH1O.
8 mol
(285.89) From Table C.4 AH, H,0()
mol
AH; = 2.02.4 From Figure 12.14
3 mol
404
a :
Hy = 5:¢-408,61y-AL From p. 444 for Lic
8 mol
AHs := -25. 5 From Figure 12.14
mol
AH := 0.2 mol (AH; + 4H) +4H3+AH,+AHs) AH =-0.403K Ans.
12.34 BASIS: 1 second, during which the following are mixed:
(J) 12 kg hydrated (6 H20) copper nitrate
Q) 15 kg H20
15 kmol
m=
BL 395.61 se0 2
ny = 0,041 Kel
sec
‘Mole ratio, final solution:
6(H2 + 1/2 02 > H20()) o
Cu + N23 02> Cu(NO3)2 @
‘Cu(NO3)2.6H20 --> Cu+N2+602+6H2 — @)
Cu(NO3)2 + 20.81 H20 -—> Cu(NO3)2(20.51 H20) (4)
120() —> Cu(NO3)2(20.51 120)
‘Cu(NO3)2.6420 + 14.
AHL := 6-(-285.83-k)) (Table C.4)
AH2 t= -302.9.K AH3 2110.8-K3) AHA = =47.84-kI
AH := AHI + AH2 + AH3 + AH4 AH = 45.08kJ
This value is for 1 mol of the hydrated copper nitrate. On the basis of 1
second,
Qn Ae Ans.
mol
12.35
12.36
LiC13H20 —> Li+ 172 C2 +3H2+3/202 (1)
3(H2 + 1/2 02 --> F200) @
2Li+ 1/2 C2 +8 H20 —> LiCIGH20)) 3)
LiCK7H20)---> Li+12C2+7H20 (4)
LiC\(7H20) + LiC13H20 —> 2 LiClSH20)
BLL Sd AH2 := 3.(-285.83-k)) (Table C.4)
(439.288) (Pg. 444)
(-436.805-kI) AH
AH = AHI+AH24AH3 +AH4 AH = 19.488KI
Q=AH Q=19488k) Ans.
Li+ 1/2 C2 + (0+2)H20 —> LiCn+2 H20) (1)
2(H2 + 1/2 02 --> H20) @Q)
LiCL2H20 —> Li+ 1/2 CI2+2H2+02 — @)
LiCL2H20 + n H20 —> LiCl(n+2 H20)
-(-285.83-KJ) AHS := 1012.65-k} (Table C.4)
Since the process is isothermal, AH = AHI + AH2 + AH3
Since it is also adiabatic, AH = 0
Therefore, AHI := ~AH2 - AH3 AHL = 440.991)
Interpolation in the table on pg. 444 shows that the LiCl is dissolved in
8.878 mol H20,
XLic 0.4012' Ans.
12.37 Data: ne ~te-74
15 867.85
20 870.06
25 -871.07
nis] 50 ‘AH == | -872.91 |-kI
100 873.82
300 ~874,79
500 -875.13
1000. 875.54
Ca+ C2 +n H20 ---> CaC2(n H20) AHy
Cals) --> Ca + C2 ~AH fea
CaCh(s) + 0 H20 > CaCl H20) — AHtilde
From Table C.4: AB goacy * ~795.8:KI
~-Tows(n)
(“en
Boe
407
1238 CaCl > Ca+ CR w
2(Ca + C2 + 12.5 H20 ---> CaC2(12.5 H20) (2)
CaC2@S H20)—> Ca+ C2 +2520 3)
CaCRQ25 H20) + CaC2 --> 2 CaCi2(12.5 H20)
AHI = 795.84) (Table C.4)
AHO = 2(-865.295-K3) AHB := 871,07-K)
AH := OHI + AH2 + AH3 Q=4H =
Ans,
12.39 The process may be considered in two steps:
Mix at 25 degC, then heat/cool solution to the final temperature. The two
steps together are adiabatic and the overall enthalpy change is 0.
Calculate moles H2O needed to form solution:
18015
e 34.911 Moles of H2O per mol CaC2 in final solution.
170.986
Moles of water added per mole of CaCl2.6H20:
n-6 = 28911
Basis: 1 mol of Cacl2.6H20 dissolved
CaC2.6H20(s) —> Ca + C2 +6 H2+302 (1)
Ca + C2 + 34.991 H20 --->CaCI2(34.911 H20) (2)
6(H2 + 1/2 02 —-> H20) 3)
CaCR.6H20 + 28.911 H20 > CaC2G4.911 H20)
AHI := 2607.94 AH =
6-(~285.83:k)) (Table C4)
AH2 := -871.8:45 (Pb. 12.37)
‘AHpog ‘= AH] + AH2 +AH3 for reaetion at 25 degC
AHgog = 21.123 Moin 2= (110.986 + 34.911-18.015)-gm
motn = 739.908¢m
408
ry
Cp = 3.28:
Kg-degC
AT = -8.702degC
12.43 mj = 150-b (E2804)
Hye 2
Tb
100: %rmy + 25-%-m2
m +m),
m3 = mj +m
Q
12.44 Enthalpies from Fig. 12.
AHg9g + Cp-AT
T= 25-deg + AT
3-H ~ (my-Hy + my Hp)
AH 298
aT:
Msoln' Cp
he
6:298degC Ans.
mg := 350-lb (25% soln.)
Hy = -23-222 (rig. 12.17)
Ibm
= 47.5% — (Final soln.)
(Fig. 12.17)
n= 05 1x, (60 % sotn)
Hy = 20- me (pure #2804) Hy = 1082 (pure H20)
a _
HE := H~(xi-Hi + xH) Ans,
12.45 (a) my == 400-16
sm = 178-lbq,
Hys= 100. BEY
Ibm
35-%my + 10-%mg
my +m
m3 := my +m
(85% soln. at 130 degF)
(10% soln. a¢ 200 deg)
Hy i= 152-200
irs
m
(Fig. 12.19)
= 27.39% (Final soln)
BTU
ben
Hy = 41 (Fig. 12.19)
409
12.46
12.47
Q:= ms-Hg ~ (m-H) + mH)
(b) Adiabatic process, Q= 0.
BO2SBTU Ans.
mH] +mzH2 B
(epee ai Hy = 1isa55TS
5 mn
Prom Fig, 12.19 the final soln, wit
about 165 degF.
this enthalpy has a temperature of
(feed rate)
(Fig. 12.17 at 20% & 80 degF)
55:
(Fig. 12.17 at 70% and 217 degF)
Ibm {Slight extrapolation]
x2 = 0.7
Hy = 1157.7-222 Crable Fe, 1.5(psia) & 217 deg]
Ibn
xpm Ib Ibn
ig et pigs mete eo eee yy gaye
x sec see
Sania
Qs my Hz + m3-H3 ~ my-H} = 208802 Ans.
Mix m1 Ibm NaOH with m2 ibm 10% soln. @ 68 degF.
BASIS: mg =
Ibm, x3 = 0.35
my = Lb (guess)
i
Given my +my= mz my txymy = xp-m5
(im
ums.
ind(my ,m3) my = 0.385 Ibm m3 = 1.385Ibm
410
12.48
From Example 12.8 and Fig. 12.19
Hy 478.7220
Bim
my-Hy + myH,
Bee
m3
From Fig. 12.19 at 35% and this enthalpy, we find the temperature to be
about 205 degF.
First react 1 mol S03() with 1 mol H20()) to form 1 mol H2S04{):
$03()) + H20()) --> H2S04())
With data from Table C.
Alggg ‘= [-813989 — (441040 - 285830)]-J_ AH 9g = -8.712« 10°
Mix 1 mol or 98.08 gm H2SO4() with m gm H20 to form a $0% solution,
MH2S04
mp2so4 ‘= 98.08-gm so = os
MH20 = Msoln ~ MH2S04
Data from Fig. 12.17:
Hupsos = am [pure acid @ 77 degF (25 degC)]
mn
Hao = 45 ~ [pure water @ 77 degF (25 degC)]
Heoin 0.5 (80% soln, @ 140 degF (40 deg C)}
AH mix = Msoin Hsoin ~ ™H2S04-Hi2804 — MH20°HH20
AFyig = -18.145 kg BLU
Ibm
AH 9g + AH nix
Msoin
an
12.49 my = 140-lbm xp 0.1S mp :=230'lbm
65 Te (Fig. 12.17 at 160 degF)
-1022e (Fig. 12.17 at 100 deg)
myx + my-x2
m3 = my +m x3 = 55.4%
m
Q + (pH) +mg-Hy
Q:=-20000-BTU 3 _ G+ (myth + meta)
m3
From Fig, 12.17 find temperature about 118 degF
12.50 Initial solution (1) at 60 degh; Fig. 12.17:
BrU
my 2 1500:Ibn xi = 0.40 93. TU
Ibm
Saturated steam at (atm); Table F.4:
1m3(imy) == my + m2 Hp = 1150.5 202
Ibm
xy-my Hy + my-H
x3(m) = Ha(m) aaa
my +m m3{ma,
mg := 125:lbm x3(mg) = 36.9% H3(mg) = -2 * y
mn
‘The question now is whether this result is in agreement with the value read
from Fig. 12.17 at 36.9% and 180 degF. It is close, but we make a second
calculation:
salma) 5.37%
This is about as good a result as we can get.
412
12.51
12.52
Initial solution (1) at 80 degF; Fig. 12.17:
mj := Flbm xy = 0.45
Saturated steam at 40(psia); Table F.4:
mg(mg) := my + mp Hy := 1169. BI
Ibm
xpmy
som) = 2
mg := 0.05-Ibm x3(mg) = 42.9% Ha(m) = -34, sa
‘The question now is whether this result is in agreement with the value read
from Fig, 12.17 at 36.9% and 180 degF. It is close, but we make a second
calculation:
BIU
gle) = 5
.048-Jbim x3 (m) #42.
This is about as good a result as we can get.
Initial solution (1) at 80 degF; Fig. 12.19:
my: Flbp: x1 = 040 Hy = 77-——
Saturated steam at 35(psia); Table F.
Hy = 1161.12 x3 = 0.38 m2 =
Ibm
ms := my +m m3 = 1.053 ibm,
my-Hy + m2-Hp
m3
BTU We see from Fig. 12.19 that for this enthalpy
at 38% the temperature is about 155 degF.
413
12.53. Read values for H, Hi, & H2 from Fig. 12.17 at 100 degF:
BTU
Hz := 68-——
- Tom
12.54 BASIS: 1(bm) of soln.
Read values for H1 & H2 from Fig, 12.17 at 80 degF:
BTU BIU
— HH = 48 =04 =1-
Tbs 2 be x x2 x1
Q2 AHS H-xy-H)-xH)= 0
H = xy-H) +xz-H Hea
See
From Fig, 12.17, for a 40% soln. to have this enthalpy the temperature is
well above 200 degF, probably about 250 degF.
12.55 Initial solution: —3¢j;= ———298.08 xy = 0.421
2.98.08 + 15-18.015.
3-98,08
3-98.08 + 14-18.015
Final solution
x2
x2 = 0.538
Data from Fig. 12.17 at 100 degF:
414
Unmix the initial solution:
AB unix = [*1-Hin2sos + (1 ~ x1) He20 J - Ha
AMypmig = 118.185 222
Ibm
React 1 mol $03(g) with 1 mol H20()) to form 1 mol H2S04@), We
neglect the effect of Ton the heat of reaction, taking the value at 100
degF equal to the value at 77 deg¥ (25 degC)
395720 -285830-—
mol mol
AH s03
J
AH gaaso4 ‘= ~813989-—
mol
AH,x °= AHguaso4 — AHip10 - 4Hp503 AH, = -1.324x we
mol
Finally, mix the constituents to form the final solution:
137.23 BIL
bm
AH pi != Ho ~[s2Hipsoa + (1 x2)Haz0] AH mic
Q
Hynix’ (2-98.08 + 15-18.015):Ib ...
+ Llbmol- AH, ..
+ AH nig (3-98.08 + 14-18.015)-Ib @
26809BTU Ans.
12.56 Read values for H(x1='
1.65), HI, & H2 from Fig. 12.17 at 77 degF:
Hex -125.BTU Hy = 0 BTU
Tas Bin
65
415
12.57
12.58
From the intercepts of a tangent line drawn to the 77 deg? curve of Fig.
12.17 at 65%, find the approximate values:
Heer ee bag 103272 Ams.
as ? Tbs
Graphical solution: If the mixing is adiabatic and water is added to bring
the temperature to 140 degF, then the point on the H-x diagram of Fig.
12.17 representing the final solution is the intersection of the 140-degF
isotherm with a straight line between points representing the 75 wt %
solution at 140 degF and pure water at 40 degF. This intersection gives
x3, the wt % of the final solution at 140 degF:
x3 = 42-% my := 1-Ib
By a mass balance:
0.75-m} 0.75-m} tay i
mem gO el
(2) my = 25-lbyy im := 40-1 Serer
x= 0 » eee
Enthalpy data from Fig. 12.17 at 120 degF:
Hy = 93 BU = 14.8 BTU
Din bm —
mg =m) +m +m3 mg = 1401bm
xpmy + xyM2 + x3M3
xq = 0.42
(Fig. 12.17)
Qs my Hy-(mpHy+me-H2+m3H3) QS=11055BTU Ans.
16
(b) First step: mj :=40-1b
mg == 75elb
From Fig. 12.17 at this enthalpy and wt % the temperature is about 100
degF.
BASIS: 1 mol NaOH neutralized.
For following reaction; data from Table C.4:
NaOH() + HCl(g) —> NaCl(s) + H20()
AHg9g := [411153 ~ 285830 ~ (425609 - 92307)]-J
AHagg = -1.791 x 10°5
NaOH(s) + HCl(g) —> NaClis) + H20()
NaOH(inf H20) —> NaOH) + inf 20 (2)
HCK9 H20) —> HCKg) +9 H20() 3)
NaCl(s) + inf H2O -—> NaCl(inf H20) (4)
NaOH (nf H20) + HCK9 H20) ---> NaCi(inf H20)
AH) := Alg9g AH := 44.50-K3 AHs ‘= 68,50:KI
AH = 3.88-kI AH
= AH + AH + AH + AH,
Qs AH
621871 Ans.
47
12.60. First, find heat of solution of 1 mole of NaOH in 9 moles of H20
at 25 degC (77 degF).
Weight % of 10 mol-% NaOH soln:
Seas x1 = 19.789%
1-40.00+ 9-18.015
Hypo = sate (Table F.3, sat. lig. at 77 degF)
Hola = asa 12.19 at x1 and 77 degF)
Hyaou = 478. an [Ex. 12.8 (p. 455) at 68 degF]
Correct NaOH enthalpy to 77 degF with heat capacity at 72.5 degF
(295.65 K); Table C.2:
295.65-K molt = 40,00-
mol
Rr [ 16316-1075 BTU
Cj 0.121 + BSN cp = 0.245
molwt | K Tbayrankine
\
Hyaon = Hao + Cp-(77 ~ 68)-rankine HyaoH *= 480.91 x
0
AH := Hyoin ~ [x HNeor + (1 - x1)}-Hi20 |
AH =
224 Thisis for 1 gm of SOLUTION.
am
However, for 1 mol of NaOH, it becomes:
AH = 45.259
mol
a8
Now, on the BASIS of 1 mol of HCI neutralized:
NaOH(s) + HCl(g) —> NaCis)+ 20) 4)
HCl(inf 120) —> HC\(g)+inf #202)
NaOH(@ H20) —> NaOH(s) + 9 H20 8)
NaCl + inf H20 —>NaClinfH20) (4)
HCIGinf 120) + NaQH@ H20) ---> NaCi(inf H20)
AH == -179067-J. (Pb. 12.59)
AH) 2=74.5:k) (Fig. 12.14 with sign ehange)
AH := 45.259-kJ (See above; note sign change)
AH4:= 3.88:kI (given)
AH := AH) +AH)+AH;+AH; — Q:= AH
4049) Ans.
419
Chapter 13 - Section A - Mathead Solutions
Note: For the following problems the variable kelvin is used for the ST
unit of absolute temperature so as not to conflict with the variable K
used for the equilibrium constant
13.4 Hg) + CO2Ag) = H20(g) + CO)
ve De
By Eq.(13.5), yn, = yoo,
-l-l+1+1=0 ng 1+
yH20 = yCO =
By Eq, (A) and with data from Example 13.13 at 1000 K:
T = 1000-kelvin
e
) (395790) + £.¢-192420 - 200240)-—.
mol! 2
ja
fe.
2.082, po
2.084
ale)
to®
2.086
2.088, '
02, 03 06
420
13.5 (a) H2(g) + CO2g) = H20(g) + CO)
i
By Eq. (13.5),
+t
T= 1100-kelvin
(1-8) Jos I
a = | —— ]-(-395' —— + =-(-187000 — 209110)-— ..
G(e) \F Ic 395960) . 5 5
Peasy
+RT otal
ear)
Guess: ee 0.5
Ans.
Given
0.35,0.36..0.65,
2,102,
2.103
2.104
ole)
10°
—2.105
2.106
213 O55 04 043 05055806 (0.65
at
(by H2(g) + CO2(g) = H20(g) + CO(g)
By Eq. (13.5), YH, > YCO, =
T-1+1+180 ngs 1+152
=yco® =
7 yH20 = Yoo F >
By Eq. (A) and with data from Example 13.13 at 1200 K:
T = 1200-kelvin
dey
.4,0.41..0.7
-2.121
2122
2.123,
Gle)
42
(c) Hg) + CO%g) = H2O(g) + CO(g)
» ¢D;
AA=
542 AB = ~4.409x 10°? AC = 1.169%10°° AD = 83x 10°
:= 1100-kelvin ‘To := 298.15-kelvin
310°) = 897.285
wcrH(298.15,1100,3.542,-4.409.10" 9, 1,169-10
wwcrs(298.15,1100,3.542,-4.409:10"°, 1.169-107°,8.3:10°) = 1.78716
IDCPH := 897.285:kelvin — IDCPS := 1.78716
r
AG = AH — Fy oss ~ AGpog) + R-IDCPH ~ R-TDCPS
432
13.18
AG
2 -5.429% 10° Ken 2) K = 81048
mol
By Eq, (13.28), = 0.5 (guess)
a
ie,
(1s +6)-(.-8)
By Eq. (13.4), noone
Given ind(s) 5 = 0.83506
lte
by = negHg = &
ane yea = yo2Ha =
5H Te ee Vee Ite
yeni yoons = 04551. ya 50.4552. Ans.
(C2H5CH:CH2(g) = CH2:CHCH:CH2(g) + H2(g) vel
oy (2) @)
Number the species as shown. Basis is 1 mol species 1 + x mol steam.
ng = 1+x
1
Texte
From data in Table C.4,
By Eq. (13.5), yi =
09730
mol
AH p98 ©
‘The following vectors represent the species of the reaetion in the
order in which they appear:
“1 1.967 31.630
=| 1 A= | 2.734 =| 26.786 |-10"7
1 3.249 0.422
873 0.0
Css | -8.882 |-107§ Di=| 00 |-10°
0.0 0.083.
end := rows(A) i
AA Dvesi AB = virB AC Yee AD =P vidi
i i 7 i
i! az 3x 10°
AD
BA = 4.016 AB = 4422x107? AC = 9.91x 107
‘T := 950-kelvin ‘To := 298.15-kelvin
iwcptt(298.15,950,4.016,-4.422-1073,0.991.1078,8.3-10°) = 1112511
10°) = 2.21675
wePs(298.15,950,4.016,-4.422-103,0.991-10"*
IDCPH := 1112.51 1-kelvin IDCPS := 2.21675
AG := AHoo - 7 (AF29g — AG9g) + R-IDCPH - R-T-IDCPS
8G = 4.896% 10° K = 053802
mot
By Eq. (13.28), ne
Since 0.10-(1+x+e) = 0.843
f@)
Ans.
(b) Ans,
434
13.19 C4H10(g) = CH2:CHCH:CH2(g) + 2H2(g)
a @) @
Number the species as shown. Basisis | 1 4
1 mol species 1 + x mol steam entering. a
8
= —— = 0.12
14x42e ee ext de
By Eq. (13.5), yy =
y3 = 2y2 = 0.24
From data in Table C.4,
i I
= \— AG5g := 166365-—
‘Alagg = 235030-—— G298 =
The following vectors represent the species of the reaction in the order in
which they appear:
=I 1,935 36.915
1 A= | 2734 B := | 26.786 |-107*
2 3.249. 0.422 j
11.402 0.0
6 5
-8.882 |-10 0.0 |-10
00 0.083,
end := rows(A) iss Lend
AA vv
297 AB = -9.285x10 9 AC = 252x107
+B
Yc AD yy viDj
6
AD = 1.66% 10°
925-kelvin ‘To == 298.15-kelvin
incpn(298.15,925,7.297,-9.285-10" 3,2.52-10" 1.66.10") = 1694.861
weps(298.15,925,7.297,-9.285-10"5,2.52-10" 5, 1.66-108) = 3.49109
IDCPH := 1694.861-kelvin IDCPS := 3.49109
AG
T
AHo9g — 7g (tase = AGo9) + RIDCPH - R-T-IDCPS
435
13.20
AG = 9242 10°
mol RT
By Eq. (13.28), compa (inte) =K
-e
K
Because O.12(1+x4+20) Be se
K+(0.24)
xh 20 x= 43151 € = 0.839
oR
1
@) y=
Yaeam= 0.812 Ans.
)
1/2N2(g) + 3/2H2(g) = NH3(g) ve-l
Basis: 1/2 mol N2, 3/2 mol H2 feed np = 2
‘This is the reaction of Pb. 4.21(a) with all stoichiometric coefficients divided
by two. From the answers to Pbs, 4.21(a), 4.22(a), and 13.7(a) ALL,
DIVIDED BY 2, find the following values:
J
~16450-——
‘mol
AB = 2,0905-107> AD = -0.3305-10°
@ To s= 298.15-kelvin
iocPH298.15,300,-2.9355,2.0905-10" 3,0.0,-0.3305-10°) = ~4.9576
imces(298.15,300,-2.9355,2.0905-10
40.0,-0.3305-10°) = ~0.016577
4.9576-kelvin IDCPS := ~0.016577
IDCPH :=
i
AG := AHg9— Fy (Ass ~ AGo9g) + R-IDCPH ~ R-T-IDCPS
436
aG = 1.627% 108 K = 679.37
mol
Pl PO=t
From Pb. 13.9 for ideal gases:
pros
e=l -(i + 1.299.K- F) 9664
PO}
Yui = ya. = 0.9349 Ans.
2-6 °
5 by the preceding equation
(b) For ynH3
Solving the next-to-last equation for k with P = PO gives:
K = 6.1586
1.299
Find by trial the value of T for which this is correct. It turns out to be
99:S-kelvin Ans.
(©) For P= 100, the preceding equation becomes
- K = 0.06159
129.9
Another solution by trial for T yields. “T+
(@) Eq. (13.27) applies, and requires fugacity coefficients, which can be
evaluated by the generalized second-virial correlation. Since iteration
will be necessary, we assume a starting T of 883 K for which:
ForNu3Q): Tp= 8p, 21437 Pp Ip coger
405.7 12
PHIB(..43 ,0.887,0.253) = 0,924
437
13.21
583
ForN2@y 0 Tes 335
PHIB(4.596, 2.941 ,0.038) = 1.034
For H2(), it is at such a large reduced temperature,that it may be
assumed ideal; @ = 1.
‘Therefore, i 1.3
0.924) 1
=| 1.034 TT" = 0.909
1.000 i
‘The expression used for K in Part (c) now becomes:
= K = 0.05598
[1299
(0.909
Another solution by trial for T yields T- 9K Ans.
‘Of course, the INITIAL assumption made for T was not so elose to the
final T as is shown here, and several trials were in fact made, but not
shown here, The trials are made by simply changing numbers in the
given expressions, without reproducing them.
CO() + 2H2(g) = CH30H(@) vez
Basis: 1 mol CO, 2 mol H2 feed n= 3
From the data of Table C.4,
24791.
mol
This is the reaction of Ex. 4.6, Pg. 139, from which:
AA
7.663 AB = 10,815-10°* AC = -3.45-10 © AD = -0.135-10°
438
(@) T= 300-kelyin To = 298.15-kelvin
incrti(298.15,300,~7.663, 10.815-10" 3, ~3.45-10" °,-0,135-105) = 9.043
weps(298.15,300,~7.663, 10815.10"?,~3.45-107, -0.135-10°) = ~0.03024
IDCPH := -9.043-kelvin IDCPS ‘= -0.03024
AG := AHQ98 ~ + (AH 29g - AGoog) + R-IDCPH ~ R-T:IDCPS
0 )
(ac
ad AG 4
AG = -2.439% 108 — K = exp) K = 1.762% 10
e Peal owl aa J
P =1
By Eq. (13.5), with the species numbered in the order in which they
appear in the reacti
e:=Find(e) © = 0.9752
(b) yz = 0.5
333
eis
2y3+t
Solution of the equilibrium equation for K gives
Find by trial the value of T for which this is correct. It turns out to be:
T Aikelvin Ans.
439
(c) For P= 100 bar, the preceding equation becomes
100°? K = 2.7% 10"
4(1-e)
Another solution by trial for T yields T= 51648-kelyin Ans.
(d) Eq. (13.27) applies, and requires fugacity coefficients, Since
iteration will be necessary, assume a starting T of 528 K, for whiel
100
34.99
For COQ): T= Py = 2.858
PHIB(3.973,2.858, 0.048) = 1.032
100
Tr= 1.03 Py
512.6 80.97
For CH30H(3): T; := Py = 1.235
By Eq, (11.64) and data from Tables E.15 & E.16.
45 := 0.6206.0.97630°% 3 = 0.612
For H2(2), the reduced temperature is so large that it may be
assumed ideal; 0 = 1.
‘Therefore:
1.032
4 =| 1.000
0.612 1
The expression used for K in Part (¢) now becomes:
3
(3-26)
4a(1—s)
Another solution by trial for T yield
100" .0.593 K = 1.6011 x 107
S28 7ikelvin Ans.
40
13.22 CaCO3(s) = CaO(s) + CO2%g)
Each species exists PURE as an individual phase, for which the activity is
£10. For the two species existing as solid phases, fand {0 are for practical
purposes the same, and the ai If the pure CO2 is assumed an
ideal gas at I(atm), then for CO2 the activity is (/10 = P/PO =P (in bar), As
a result, Eq, (13.10) becomes K = P = 1.0133, and we must find the T for
whieh K bas this value.
From the data of Table C.4,
Agog = 178321 a AGaog = todo)
the order
‘The following vectors represent the species of the reacti
which they appear:
vl 12.572 2,637 3.120
1 6.104 | B= | 0,443 -107° Dd =| -1.047 }-10°
1 3.457 1,045, 1.157,
1.3 SA > iA B= SviBi AD =: Yer
3 4
AA = -LO1L AB = -1.149x 10 AD = 9.16« 10
298.15-kelvin
151.83-kelvin To
ioceH(298.15,1151.83,-1.011,-1.149-1073,0.0,9.16-104) = ~1346.495
IDcps(298.15,1151.83,—1.011,-1.149-10"9,0.0,9.16- 104) = =1.86655
IDCPH := -1346.495-kelvin IDCPS := ~1.86655
Ee oi
AG := AHy9g — Fy (Hoss ~ AGo9g) + R-IDCPH ~ R-T-IDCPS
J ~AG)
G = -126.353— K = exp( 2 =
on( ar) Ke 1013
Thus Pes RR Ans,
Although a number of trials were required to reach this result, only the
final trial is shown. A handbook value for this temperature is 1171 K.
4st
13.23, NH4CI(s) = NH3(g) + He)
‘The NH4CI exists PURE as a solid phase, for which the activity is 110.
Since f and 10 are for practical purposes the same, the activity is unity. If
the equimolar mixture of NH3 and HCl is assumed an ideal gas mixture at
1.5 bar, then with {0 = I bar the activity of each gas species is its partial
pressure, (0.5)(1.5) = 0.75. Asa result, Eq. (13.10) becomes K
(0.75)(0.75) = 0.5625 , and we must find the T for which K has this value.
From the given data and the data of Table C4,
176013 Gogg = 91121
AHp§
mol ‘mol
‘The following vectors represent the species of the reaction in the order in
which they appear:
a 5.939 16.105 0.0
1 3578} Bi=| 3.020 }10°? D:=| -0.186 |-10°
1 3.156. 0.623 0.151
3 AA Deri AB yes ad =} vidi
AA = 0.795 AB =~0.012462 AD = -3,5x 10°
T= 623.97-kelvin Ty
298.15-kelvin
wepH( 298.15 ,623.97 0.798 -12.462-10°3,0.0,-3.5-10°) = -1619.117
wcps(298.15,623.97,0.795 ,~12.462:10"3,0.0,~3.5-10°) = ~3.48845
IDCPH := -1619.117-kelvin IDCP!
-3.48845
AG := AHg9g (AH29g - AGz9g) + R-IDCPH ~ R-T-IDCPS
AG = 2.986 x 10° — K = 0.5624
Thus 7 $623.97K Ans.
Although a number of trials were required to reach this result, only the
final trial is shown.
482
13.25 NO(g) + (1/2)02(g) = NO2(g) =-05
aeons 5 K 298.15-kelvin
ywor(yo2)"* — ywo-(0.20)
j
From the data of Table C.4, AGaog #= ~35240-——
‘mol
(-a0
Keep ar K= 1.493% 10°
yo = 10°? ywor = 107° (guesses)
: 05 ee
Given yno2 = (0.21)""-K-yno-ywo2 + ¥NO = 5-10)
YNO Same
( } Find(yv0.yN02) io?
\YNO2
This is about 7:107° ppm (a negligible concentration) Ans.
13.26 C2HA(g) + (1/2)02(g) = <(CH2)2>0(g) v= -05
See Example 13.9, Pg. 492-494, From Table C4,
3 J
‘AHg9g = -105140-— AGoqg <= -81470-—
a9 eal 298 al
Basis: 1 mol C2H¢4 entering reactor.
Moles 02 entering: ng2 = 1.25-0.5
Moles N2 entering: BN? != No? 2
nig := 1+ng2+ nN? ng = 3.976
Index the product species with the numbers:
trogen
‘The numbers of moles in the product stream are given by Eq. (13.5).
403
For the product stream, data from Table C.1:
Guess: g = 0.8
Ire 1.424 14.394
nog 0.5 3.639 0.506 |. 1973
B i
le) e A= | 9385 23.463 | kelvin
aN2 3.280 0.593
4,392 0.0 -1
0.0 6 227 0.5
12 D= -10° kelvin” =
-9.296 | elvin? 0.0 1
0.0 0.040, 0
a4 Ale) = Yin): AG B(e) = > a(e):-Bi
Cle) =F ale)ici DE) = ¥ale)edi
n(e)
os K(e) = 15.947
Me) on ogs ©)
‘The energy balance for the adiabatic reactor is:
‘AHg9g+AHp = 0 For the second term, we combine Eqs. (4.3) & (4.7).
‘The three equations together provide the energy balance.
For the equilibrium state, apply a combination of Eqs. (13.11a) &
(13.18).The reaction considered here is that of Pb. 4.21(g), for which the
following values are given in Pb. 4.23(g)
‘AA := -3.629
.114:10°kelvin? To <= 298.15-kelvin
Guess:
B
IDCPH : aato(e~1)+ Brg? -1) .
+2fay 3.(3 ii wie)
To \ 7
I
IDEPS == AAdIn(t) +) AB-Ty +] ACT” ee ) -(r-1)
aD
(Ty |
IDCPH = ~130.182kelvin IDCPS = ~0.417
Given
Ble) *
“AHlgog = R} Ale) To(e - 1) + 22
cle) DOs)
+ oOne le d. ig
K(e) = exp| | S298 86298 | A298 ) | ncps -
| RT) RTyt
(ca) ©) _ (oss24s
u aa F o) ares
Ti=tTo ‘T= 949,23kelvin Ans.
13.27 CH4(g) = C(s) + 2H2(g)
= 1 (gases only)
The carbon exists PURE as an individual phase, for which the activity is
unity. Thus we leave it out of consideration.
405,
From the data of Table C.4,
i 4
AH}9 := 74520-— A = 50460-—
D9 a Case an
‘The following vectors represent the species of the reaction in the order in
which they appear:
1 f 1.702 9.081
1 As] 1771 orn }-10-%
2 io 0.422
2.164 0.0
6 s
3 0.0 |:107 D:=| 0.867 |-10"
0.0 0.083
Desi AB Yves ac Svcs ad = S vidi
: 7.01 x 10°
AA = 6567 AB =-7466x 107? aC = 2.16410 AD =
Ts 98.15-kelvin
23,15-kelvin Ty
1DcPHi(298.15,923.15,6.567,~7.466-10">,2.164-10°°,-7.01-108) = 1644.114
701-10) = 3.22848
wcPS{(298.15 923.15 ,6.567,~7.466-107>,2.164-10
ic
= 1644.114-kelvin IDCPS := 3.22848
T
AG := AHggg — Fp (olla = AGg9g) + R-IDCPH = R-TDCPS
0
-aG)
aG=-11o9xwtt Ks onl 7 J} K=42302
mmol RT
By Eq. (13.5), ng = 1
Ite
(a) By Eq, (13.28),
448,
0.7173 (fraction decomposed)
2e z:
ae yeua = 0.1
ie Ans.
() For a feed of 1 mo! CH4 and 1 mol N2, ng = 2
By Eq. (13.28),
Given iat ats K
Q+e)(-2)
250; (fraction decomposed)
13.28 1/2N2(g) + 1/202(g) = NO(g) veo @
‘This is the reaction of Pb. 4.21(n) with all stoichiometric coefficients
divided by two. From the answers to Pbs. 4.21(n), 4.22(n), and 13.7(n)
ALL DIVIDED BY 2, find the following values:
AH9g != 90250-—— AGagg = 86550-—
mo mol
‘AA = ~0.0725 AB = 0.0795-10"* AD = 0,1075-10°
T= 2000-kelvin Tp = 298.15-kelvin
wcPHi(298.15,2000,-0.0725,0,0795-10">,0.0,0.1075-10°) = 62.763
icPs{298.15,2000, 0.0725 0.0795-10" 3, 0.0,0.1075-10°) = 0.036429
IDCPH
62.763-kelvin IDCPS == 0.056429
fr
AG = AHa9g~ —~-(AFlagg ~ AGagg) + R-IDCPH ~ R-TIDCPS
0
“47
aa =6s01xit@2 Ky ki x00( 29) Kj = 0.02004
mol \RT
1/2N2(g) + O2(g) = NO2g) 5 @
From the data of Table C.4,
AH: = 33180- : AG: = 513102
298 °= mol 298 = mmol
The following vectors represent the species of the reaction in the order in
which they appear:
0:5 3.280 0.593 0.040
-t.| as|3039| B=} 0.506 |10°° 0.227 }-10
1 4.982 1,195, 0.792.
ie.3 BASS vrAi AB =P viBi AD a Di
AA = ~0.297 AB = 3.925% 10"*
:= 2000 -kelvin To := 298.15-kelvin
wwcpH(298.15,2000,-0.297,0.3925.10
wwcps(298.15,2000,-0.297,0.3925-10" $,0,0,-0.585-10°) = -9.21904
IDCPH := 95.145-kelvin IDCPS := -0.21904
T
AG := AHo9g — a (Hogg - AGz98) + R-IDCPH ~ R-T-IDCPS
5
AG = 1.592 10°
mol
) Kp = 6.9373 x 10
With the assumption of ideal gases, we apply Eq. (13.28):
JNO JNO
5 (@.7)95.(0.05)°%
a)
=k)
02)"
448
13.29
yno #= K1-(0.7)°5.(0,05)"% Ans.
@ Pont 200
yno2 -(t)"
ei Ieee Ay GL a
(yn2)"*-yo2r 0.7°4.0.05) PO
ry 05 eepiaeneateo aes
yor = (2) Kr (0.7005) non 4104 1Q"F Ans
2H2S(g) + $02(g) = 38(s) + 2H20(g)
The sulfur exists PURE as a solid phase, for which the activity is £110. Since
fand {0 are for practical purposes the same, the activity is unity, and it is
omitted from the equilibrium equation. Thus for the gases only,
From the given data and the data of Table C4,
AH 98 #
145546 Gao = ~89830-2.
mol mol
The following vectors represent the species of the reaction in the order in
which they appear:
2 3.931 1.490 0.232
5.699, og01 | -1.01
B :10"? Db * ha08
4.114 —1.728 0.783
2 3.470, 1.450 0.121
Sai AB Yves AD Ye
AA = 5,721 AB =
065% 10°? AD = ~6.28x 10°
23.15-kelvin To := 298.15-kelvin
409,
wmcpHi(298.15,723.15, 5.721 ,-6.065-10°°,0.0,-6.28-104) = 991.371
wees (298.15, 723.15,5,721,-6.065-10" 3,0.0,-6.28-10°) = 2.19811
IDCPH := 991.371-kelvin IDCPS »
2.1981]
T
AG = AHg9g — fs (AH298 ~ AGz9g) + R-IDCPH - R-T-IDCPS
-1538x 108K exp
AG
mol
By Eq. (13.5), gases only: ng = 3
ei=Finde)
Percent conversion of reactants = PC
niga)
uJ “100
Tio mio
PC 100 [By Eq. (13.4)]
Since the reactants are present in the stoichiometric proportions, for each
reactant,
100 PE76867" Ans.
nip =v; Whence
13.30 N204(g) = 2NO2g)
@ ()
Data from Tables C.4 and C.1 provide the following values:
vel
030.
mol
J
AH9g = 57200 AGaog
To 2= 298.15-kelvin T = 350-kelvin
450
AA = =1.696 AB = 0,133-107 AD := 1.203-10°
ioceHi(298.15,350,~1.696,0.133:10" 3, 0.0, 1203-108) = -25.929
tHcPs{298.15,350,-1.696,0.13310 3,0.0,1.203-108) = -0.079402
IDCPH := -25.929-kelvin IDCPS := -0.079402
Be
AG := AHy9g -
To
3d
AG = -3.968 x 10° — K=3911
mol
=
2 = 0.4044
© = 0.7031
By Eq, (4.18), at 350 K:
AH := AHggg + R-IDCPH. ait = 5694
mol
This is Q per mol of reaction, which is
.7031 — 0.4044
Whence Qs AltA
451
xara | (1-xa)ya
SATA SAYA
1331 By Eq, (13.32),
In(ya) = O.1-xn” Infyp) = O.t-xa? Whence
ae xp(0.1-x47 2
(seed Stl
— XA “At
a -exp[0.1-(2-x4 — 1)] K= eo 22)
1000.
mol
(guess)
i (-
1 copfo.t(2xa—1)] = owl ee
) xa i= Find(xq)
XA
x, = 03955 Ans.
For an ideal solution, the exponential term is unity:
XA RT
Given
Find(xa) -#4'$'0:4005
‘This result is high by 0.0050, Ans.
13.32 H20(g) + CO(g) = H2(g) + CO2%g)
From the the data of Table C.4,
AHg99 = ~41166-—
mol
To := 298.15-kelvin
482
IDCPH := 539.735-kelvin [DCPS := 1.00107
AG = AHg93~ Flt = AGo9g) + R-IDCPH ~ R-TIDCPS
AG = -9.668 10° K = exp 46 K = 4.27837
mol RT
(a) No. Since v= 0 ,at low pressures P has no effet
(b) No. K decreases with increasing T. (The standard heat of reaction is
negative.).
(c) Basis: 1 mol CO, 1 mol H2, w mol H20 feed.
From the problem statement,
f
2s ggn
Tico + NH2 + NCOZ
By Eq, (13.4), mpeite Neo =e
Sere en Eg Oo Cte gig ae 6 = 0941
l-e+ltete 2+e 1.02
Let z= w/2 = moles H20/mole "Water gas”.
By Eq. (13.5),
yung = No# = 22=8 veo = 122 alte
OS Daw 24dz Woo" oa 2e ye Free
nes By Eq. (13.28) (guess)
2420
Given _#-(1+8)
ao eK a Find
Gia) z= Find(2) Ans.
(@) 2C0@®) = COr®) +c) V==1 (gases)
Data from Tables C4 and C.1:
i
‘Aiggg := =172459.——— ‘AGy5g == ~120021-—2-
mol mol
453
13.33
AA= 0476 AB=0.702-10°> AD = -1,962-10°
1cPHi( 298.15, 800,0.476, 0.702-10" 3,0.0,-1.962:10°) = 19.511
wers{298. 15, 800, 0.476, 0.702: 10> 0,—1.962: 10°) = -0.12817
IDCPH := 19.511 -kelvin IDCPS := ~0.12817
AG := AH298 — % (F129 - AGz93) + RIDCPH ~ R-T-IDCPS,
AG = -3.074 10" K= 1017
‘mol
By Eq. (13.28), gases only, with P= PO =1 bar
yeor
K= 101.7 for the reaction AT EQUILIBRIUM.
(veo)?
If the ACTUAL value of this ratio is GREATER than this value, the
reaction tries to shift left to reduce the ratio. But if no carbon is present,
no reaction is possible, and certainly no carbon is formed. The actual value
of the ratio in the equilibrium mixture of Part (e) is
yoo2 W232 yco = edn
yeo2 = 0.092 yoo = 5.767 1075
x SiS psa
RATIO := 2 RATIO = 2.775% 10°
(sco)?
No earbon can deposit from the equilibrium mixture.
CO(g) + 2H2(¢) = CHBOH() ve-2
This is the reaction of Pb. 13.21, where the following parameter values are
given:
Aiggg == ~90135-—- AG 9g == -24791-—
mol mol
550-kelvin To #5 298.15-kelvin
454
6
AA = -1663 AB = 10815-1073 aC = ~3.45-107° AD = -0.135-10°
3
345-107 §,-0.135-10°
~3.45-10"°,-0.135-10°
= ~956.435
2.39055
ICP H(298.15,550,~7.663, 10.815-107
swcrs(298.15,550,=7.663,10.815-10
IDCPH := -956.435-kelvin DCPS := -2.39055
AG := AHo9g ~ F(Attoos ~ AGg9g) + R-IDCPH ~ R-TIDCPS
0
ad -AG a
= 3.339 10 — = exp, SO Ky = 6.749 x 10
AG = 3.339% 10" Ky oo( 2) 1
H2(g) + CO%g) = CO(g) + H20() v=0 (2)
From the the data of Table C.4,
I J
‘AHy9g := 41166-— G99 := 28618 —
at, mol ae mol
T= 550-kelvin To = 298.15-kelvin
‘The following vectors represent the species of the reaction in the order in
which they appear:
1 3.249 0.422 0.083
-1 5.457 104s |g -hIS7} os
vs 10"? D
i 3.376 0887 =0.031
1 3.470, 1.450, 0.121
+4 AAS YS vidi a8 Svs AD = Y vidi
AA =
86 AB=5.4x10°* AD = 1.164% 10°
0.0, 1.164105) = 231.996
0,1.164-10°) = ~0.540599
wcPH(298.15,550,-1.860,0.540-10
mDcPs(298.15,550,~1.860, 0.540-10~
IDCPH := -231.996-kelvin _ IDCPS := -0,540599
455
t
AG '= AHy9g— Fy (Atos = AGagg) + R-IDCPH - R-T-IDCPS
0
AG = 1.856% 108 Ky:= oo 2) K> = 0.01726
mol RT)
Basis: 1 mole of feed gas containing 0.75 mol H2,
0.18 mol CO, 0.08 mo] CO2, and 0.05 mol N2.
Stoiel
metric numbers, vj;
i= 2 CO CO2 CHBIOH = 20
i
1 2 al oO 1 0
2 a 1 -l 0 1
Eq. (13.7)
0.73 -2e)-€ 0.15 -e1 +22
eerste Seer
= 2) 1
0.05 -€2
yoon® <> YeHOH = TE
Pi= 100
By Eq. (13.40), 62:= 0.1 (guesses)
Given
e1-(1-2€;) >
See ee (2)
(0.7526) ~52)"(0.18-e,+e2)
0.15 ~ 1 + €2)-€ a
( 22 = Kp == Find(e; 29)
“£1 ~ 8}-(0.05 -&) £
5) = 0.1186 62 = 8.8812 10>
486
O.1S—e; +82
0 Rey
e)
YCH3OH
28
~ YH2~ YCO ~ Yoo? = YCH3OH ~ YH20
vis os
13.34 CH4(g) + H20(g) = CO(g) + 312g) v=2 @)
From the the data of Table C.4,
J J
205813-—— AGp9g := 141863-—
mol 298: mol
The following vectors represent the species of the reaction in the order in
which they appear:
AH29g
1 1.702 ‘9,081
= 3.470 1430] a
1 3.376 0.557
3 3.249, 0.422
2.164 0.0
29 ha ON 08 i l.4
00 ~0.031
0.0 0.083
AA meee AB ze
AA=7.951 AB = ~8.708x 10"?
*
AD = 9.7% 10°
1300-kelvin
1ocpHi(298.15,1300,7.951,-8.708:10"°,2.164-10° °,9.7-108) = 2585.215
rcps(298.15, 1300,7.951,-8.708.10°3,2.164.10"8,9.7-10°) = 4.76801
IDCPH := 2585.215kelvin DCPS == 4.76801
AG := AHygg - Z(sit08 = AGooq) + R-IDCPH = R-T-IDCPS
0
3 J -aG
G = 1,031 10° Ky = exp 3S Ky = 13845
femal 7 ox #8) :
H120(g) + CO(g) = H2(g) + COX) v=0 @
‘This is the reaction of Pb. 13,32, where parameter values are given:
roceri( 298.15, 1300, 1.860,-0.540-1073,0.0,—1.164-108) = 1130.273
scPs(208.15, 1300, 1.860, 0.540: 107 ,0.0,-1.164:10°) = 1.577615
IDCPH := 1130.215-kelvin IDCPS.
1.577615
AG <= AHy9g - Fy lone — AGyog) + R-IDCPH — R-T-IDCPS
AG = 5.891 x 10° Ko on 22)
mol RT
(a) No, Primary reaction (1) shifts left with increasing P.
(b) No, Primary reaction (1) shifts left with increasing T.
(©) The value of K1 is so large compared with the value of K2 that for
all practical purposes reaction (1) may be considered to go to
completion. With a feed equimolar in CH4 and H20, no #20 then
remains for reaction (2). In this event the ratio, moles H2/moles CO
is very nearly equal to 3.0.
458
(@) With 120 present in an amount greater than the stoichiometric
ratio, reaction (2) becomes important. However, reaction (1) for
all practical purposes still goes to completion, and may be
cousidered to provide the feed for reaction (2). On the basis of 1
mol CH4 and 2 mol 120 initially, what is left as feed for reaction
(2) is: 1 mot H20, 1 mol CO, and 3 mol H25 Thus, for
reaction (2) at equilibrium by Eq, (13.5):
CO = YH20
By Eq. (13.28),
one 2048 a
(1-ey
YH?
Ratio = Ans.
Yoo
(@) One practical way is to add CO2 to the feed. Some H2 then reacts
with the CO2 by reaction (2) to form additional CO and to lower the
H2/CO ratio.
(D 2CO(g) = CO2%g) + Cis) vy =-I (gases)
‘This reaction is considered in the preceding problem, Part (d), from
which we get the necessary parameter values:
120021
mol
For T= 1300 K,
1300-Kelvin Ty := 298.15-kelvin
wwcpH(298.15, 1300,0.476,0.702-10"3,0.0,-1 962.105) = 531.734
iwcps(298.15, 1300,0.476,0.702-10"3,0.0,-1.962-105) =
35870
IDCPH := $31.734-kelvin IDCPS := 0.35870
T
AG = AHyo3 — Ps (A#295 - G98) + R-IDCPH ~ R-T-IDCPS
0
489
oN :
aG=s6nxitL Ks onl SG) Ke 52sscoux 1073
mmol RT)
As explained
Problem 13.32(d), the question of carbon depo:
IAL compositions the value of this ratio is greater than
the equilibrium value as given by K, there can be no carbon deposition.
‘Thus in Part (@), where the CO2 mole fraction approaches zero, there is
danger of carbon deposition, However, in Part (d) there can be no carbon
deposition, because Ratio > K:
eS
Ratio Ratio = 0.924
(2?
13.37 Formation reactions:
C+ 282 = CHS
C+ (12)02 = CO
C+02=CO2
Elimination first of C and then of O2 leads to a pair of reactions:
CH4 + #20=CO+3H2 (1)
CO+H20=CO2+H2 @)
‘There are alternative equivalent pairs, but for these:
Stoichiometric numbers, v5,
CH4 #20 CO COP 2 Vy
For initial amounts: 2 mol CH4 and 3 mol H2, n0 = 5, and by Eq. (13.7):
2-8 3-e)-e) 81-8,
YCH4 = 5426) YH20 = Fe2e, Yco = TeDe
ne Be) +82
Pete Eo gu mae Sd
By Eq. (13.40), with P = PO=1 bar
3
yoor(yna)" yori _
ae ki om ek
YCH4-YH20 YCO"YH20
From the data given in Example 13.14,
J
‘AG, = =27540-— 8G; = 31304 1000-kelvin
mol mol
: -AG, } : -AG2
ieee aT) 2S OPT
Ky = 27.453 Kp = 1.457
eels eg i= (guesses)
Given
(e1=#2)-(52) 403)?
~81)(3-e) -82)(5 +21)
£9-(3€1 +22)
=K
(r= 8) 08-2)
1)
| = Find(, ,e) e) = 1.8304 82 = 0.3211
£2)
e~e e182
YH20 yco
5+ 26 3426)
461
£2 eine
a
5428 S42e)
yous = 00196. 0.098 yoo = 0.1743
oo = 0.0371 yp = 0.671)
‘These results are in agreement with those of Example 13.14.
462
14.1
(b)
Chapter 14 - Section A - Mathcad Solutions
Aya = 0.59 Agi = 142 T= (55 +273.15)-K
Margules equations:
rast) = expl (1-1) Aig +2(Aar - Ara)xr] |
2 1
sraler) = expL x1? Aas +2-(Ai2 ~ Aas)(1-x1)]]
Psaty := 82.37-kPa Psatz = 37.31-kPa
BUBL P calculations based on Eq. (10.5):
Pauta(s1) := x1-7a(x1)-Psats + (1 x1} 72(s1)-Psata
rvs} Pa
Js
nba = ae
xp = 0.25 sisi):
xy = 0.50 wba)
xy 075 Phubilti) = 85.70 kPa vile) = 0.808
BUBL P caleulations with virial coefficients:
3
om
3
pe Baa = -1523-
By == -963-
mol mol
612 = 2-Bi2— Bui ~ Baa
fr 2.
| Bi-{P = Psat) + P-
04(P.7 91.7) = em zl “) ~
(PT. 91,92)
1
Bop-(P ~ Psat) + P-y1?-8 4 |
ae RT
463
Psat; + Psat
2
xy = 0.25 Given
yt-® (PT, y1,92)-P = xr-yi(xi)Psaty
yo@2(P,T,y1.y2)-P = (1 x1} 72(xs)- Psat
Guess:
yrs 05
yeloy
y
yz | = Find(y1.y2.P)
bas
x1 = 0.50 Given
yi) (P.T.y1.y2)-P = x74 (x1)-Psaty
y2@2(P.T y1sx2)P = (1x) ra(ui) Psst
nel-y
vt
yo | = Find(y1 .y2.P)
P
xp=0.75 Given
yr@ (PT, ys.y2)}-P = x1-74(x1)-Psati
y22(PT yty2)}P = (1 x1) va(a1)Psate
yeloy
He
iW ei
y2 | == Find(yy .y2,P)
P
464
14.3
14.4
T= 200K
Hy = 200-bar
Assume Henry's law applies to methane(1) in the liquid phase, and that
the Lewis/Randall rule applies to the methane in the vapor:
fhaty! = Hy-xy ‘that;
By Eq. (11.35): os on(22) $1 = 0.827
\
Equate the liquid- and vapor-phase fugacities and solve for x1:
yee? :
ee oni’ Ans.
Ai
Pressures in kPa.
a
Data: 0,000 12.30 0.000
0.0895 1551 0276
0.1981 1861 0.4565
0.3193 21.63, 0.5934
0.4232 2401 0.6815
0.5119 25,92 0.7440.
0.6096 27.96 0.8050
0.7135, 30.12. 0.8639
2..r0ws(P) xg: 1-x1 Psatz := Py
(a) It follows immediately from Eq, (12.10a) that:
ii) = Az
‘Combining this with Eq. (12.10a) yields the required expression
465
(b) Henry's constant will be found as part of the solution to Part (c)
(c) BARKER'S METHOD by non-linear least squares.
Margules equation,
‘The most satisfactory procedure for reduction of this set of data is to find
the value of Henry's constant by regression along with the Margules
parameters.
‘yal x2,A12,Aa1) = expl 62)? Ana +2-(Aat ~ Aia)xt]
oli x2,A12,Aa1) = expL_(1)2L Ani +2(Ai2 ~ Aoi)? ||
Guesses: 02 A048
Mininize the sums of the squared errors by
setting sums of derivatives equal to zero.
Given
‘ fel Hy 2]
0 | sale-(o (x1, Aa. A2I) Tae) :
L (439; %2(%1, 3,12, A01)-Psate
Hy |
Pim] mira (a,o%
Ai2,A21)
#92, *72[81,0%2, ,A12,Aal) Psaty
paris, siete) ty r
sae exp(Aiz)
+4) 2(81,%2,,A12,Aa1) Psat
Ans,
466
(a) y1(x1 x2)
nol 2?[ Ar2 +2-(Adi- Av)xt]]
4220x1522) = exp x17 Agr +2-(At2 ~ Ant] }
Hy
Peas, = 1, 71( 1,984) AD +2 72(m1, 99,}Psata
02
°
Pious,
_ “02
{oi vet) 100
-04
“06
° 02 oa 06 08
x
Xx Pressure residuals
© yl residuals
Fit GE/RT data to Margules eqn. by least squares:
i= 2..rows(P)
=>é
Lia
Given
“expla
¥2;Pi
+x9,Inj ———
ne,
AQ
Ans.
Agr | s= Find(Ai2.Az1 Hi)
Hy
YUG&L x2) = exp] x2 Ar2 +2¢(Ag1 = Arz}ext
Y2G.1 x2) = expl x1? Ag) #2-(Ai2~ Ani }2]]
H,
Peal © x1;71(1,-82)) ale) 2,721.
My n(x Hi
- a ‘Ai2)
Peale,
Yieale,
468
0 02 04 06 08
x1
XX Pressure residuals
© yl residuals
145 Pressures in kPa
0.3193 21.63 0.5934
Data 0.4232 24.01 0.6815
0.5119 25.92 0.7440
0.6096 0.8050
0.7135 30.12 * | 0.8639
0.7934 31.75 0.9048
0.9102 34.15 0.9590
1,000 36.09 1.000
i= 1.7 ” xi Psaty = Pg
(a) It follows immediately from Eq. (12-10a) that:
Combining this with Eq. (12.10a) yields the required expression.
{b) Henry's constant will be found as part of the solution to Part (c).
468
(©) BARKER'S METHOD by non-linear least squares.
Margules equation.
‘The most satisfactory procedure for reduction of this set of data is to find
the value of Henry's constant by regression along with the Margules
parameters.
ta(xl.92,A12,Aai)
sp (2) Aiz + 2(Aa1 ~ Ata) xt}
5
roll .x2, Az, Ann) = exp 1) Agr +2-(A12- Ani) x2] |
Guesses: Ho:= 14 An = 0.148 Ayz = 0375
Mininize the sums of the squared errors by
setting sums of derivatives equal to zero,
Given 0= 2. =
12
! |
0 Elsa Pi = (11 (¥1,.%9,.412,A2i} eat |
Heat Sapnafsypss Aina Ty
d
7 | Pi- (xara (s1,.%2,-A12,Az1}-Psaty ..
Taf fan foyrpaiots ra
H2
so n(s.9, Ara Ane ||
Ad
(Ar Az) 0.469
| Agr | := Find(Ay2,A21,Hp) | Aoi} =| 0.279 | Ans,
(ie He}, 3487
(8) yl0at x2) = expl x2°[ Ara +2-(Aot~ Avz}xt] |
y2(x1,,x2) = exp| x12. [Aa +2-(At~ Aat)22]]
470
} Hp
pPssts +9339(81, 2) Sota
: i'l! exp(Aat,
xi (x 2¥2)) Paty
Peat, = 1,71 (X1,.%2,
Veal =
cua Peat
‘The plot of residuals below shows that the procedure used (Barker's
‘method with regression for H2) is not in this case very satisfactory, no
doubt because the data do not extend close enough to x1 = 0.
1
oe
Pi-Peale, 4 °
(21,-¥1teate)} 100, .
3
“oz 04 06 08
a
xx Pressure residuals
& yl residuals
Fit GE/RT data to Margules eqn. by least squares:
i. yo t-y
( vi;Pi )
xin] ——} — (Aatx, raya,
Tl xp -Psaty | i 172
me) FAD,
4am
viyPi
; ~ (Aare,
oii sym }- ;
S|) X1
0 Dia ex Psat ven
' 32, Pi
Ha
expan
+39,
An -
Agi | = Find(Aj2,Az1,H2)
Ho
Get x2) = exp 2? Ara +2(Aa1 = Arg}! |
12Gs1 x2) = expl_ x17 Any +2412 ~ Ari)92] |
Hz
1 11 (10%, Psat) +9,77(4,) Sota)
x1 719% Psaty
Yleale, = Peak,
472
07
x
%ox Pressure residuals’
© yl residuals
‘This result is considerably improved over that obtained with Barker's method.
14.6 Pressures in kPa
Data: 15.79 0.0 0.0
17.51 0.0932 0.1794
18.15 0.1248 0.2383
19.30 0.1757 0.3302
19.89 0.2000 0.3691
P x
21.37 0.2626 0.4628
24.95 0.3615 0.6184
29.82 0.4750 0.7552
34.80 0.5555 0.8378
42.10 0.6718, 0.9137,
i 2..rows(P) x= 1-1 Psat = Py
(a) _ It follows immediately from Eq. (12.10a) that:
inn?) = Ai
Combining this with Eq. (12.108) yields the required expression
473
(b) Henry's constant will be found as part of the solution to Part (c)
(©) BARKER'S METHOD by non-linear least squares.
Margules equation.
The most satisfactory procedure for reduction of this set of data is to
find the value of Henry's constant by regression along with the
Margules parameters,
Ayo +2(Ao1 ~ Arg)xt J]
An}2 |]
127 Ay? = -0.70
yi (xt x2,A12.Aa1) = exp (22)
sa(61 x2, Ar2,Aag) 2 exp 1)? Agr + 2-(A2
Guesses: Hy = 35 Art
Mininize the sums of the squared errors by
setting sums of derivatives equal to zero.
Given
> | rtm
[ +439, 7o(81,0%2,.A12, Aa): Ponts
oe Hy "
nadie dal)
(+2, Y2(X1, 982, Ar2,Azi) Psat
Az) (0.731).
AQ | = | 1187} Ans.
Hi) (32.065
474,
(8) yiGxt x2) == exp] x2 LAr +2-(Ao1 - Ars) xt ]}
{20% x2) = expl x12 Ay +2(Ar2 ~ Ant)x2]}
Peale, = s(t + x2,72( x1, »%2,} Psata
Hy
a, ¥1(s1,.%9,) zolan)
Yieale,
: Pose
Wie)
0 01 02 03 04 05 06 029
x;
X** Pressure residuals
© yl residuals
GEIR data to Margules eqn. by least squares:
i= 2..rows(P)
Given
d
aan
475
fale Een ina .
“I Sxp(Aia)
13;
+x2,"Inl =
x2,-Psatz
Al
Aal
AL
y(n) x2) = exp] 2°{ Ai +2(An1 - An)xt]
72(K1,,x2) := exp xi? Ag) +2.(A12~ Adi) 2]]
aati ee + x2-72(x1_.x2, -Psaty
TNO) Say tT)
476
PjPeate, es
Gat
0 01 02 03 04 05 06 07
x,
Xx Pressure residuals
© yi residuals
14.7 Pressures in kPa
0.1737 1930 0.3302
Data: 0.2000 19.89 0.3691
0.2626 21.37 0.4628
0.3615 24.95 ools4
0.4750 29.82 0.7552
MS) 95555 34.80 "| 08378
06718 42.10 0.9137
0.8780 60.38 0.9860
0.9398 6539 0.9945
1.0000, 69.36, 1.0000,
i= 1.9 xg i= 1-xy Psaty := Pig
(a) It follows immediately from Eq, (12.10a) that:
Combining this with Eq. (12.10a) yields the required expression.
(>) Henry's constant will be found as part of the solution to Part (c).
a7
(©) BARKER'S METHOD by non-linear least squares.
Margules equation.
‘The most satisfactory procedure for reduction of this set of data is to
find the value of Henry's constant by regression along with the
Margules parameters.
ult x2,A12,Aa1) = exp], 22 Aja +2-(Aat ~ Aig} xi]
sraletx2A12, Anu) = exp] (x17? Ani +2(Ai2~ Aai)22 J]
Guesses: Ho=4 0 Agy=-137 Ai = 0.68
Mininize the sums of the squared errors by
setting sums of derivatives equal to zero.
Given
x4, 70 (81,992, ,A12-Azr) Psat
y S-/ vr
| dang i |
l P va(*i, 2x2, A125 Ani) ) plan) }
478
(@ 71Cxt x2) = exp| x2? A1+2-(Aat- Aa)xt]]
20st x2) := exp] x17[A21 +2-(Ar2 = Azi) x2] |
Hp
Peate, = X1p71(X1,.%2,) Psat + ¥9,72(%1,0%2,) onland
xiv (11,2) Psat}
Yicale, =
' Prato,
1
Pi-Peate Osea ce oe
xx x
Gives) 190 eee
a
ae oO 02 04 06 08 1
x,
**<* Pressure residuals:
© yl residuals
Fit GE/RT data to Margules eqn. by least squares:
i 1.9 yost-yt
[(( ow \ ?
xi tn Paar
||| Pst |
+39,-ln| — |
», |
\ ) J
473
yiyPi | : it
xia fa fAara, «ap,
i al
ee .
yaP
x2
Aa
Ho
AG x2) = exp) 2A +2(A21-Arz)at ||
2061 32) = exp] x1°[ Any +2(Ara~ Ani) ] |
u,
Peat 2= 8111 (s1,.¥2,) Psat 939/765, Ta
xii
Yteale,
x;
Xxx Pressure residuals
© yl residuals
14.8 (a) Data from Table 12.1
15.51 0.0895 0.2716 (1304 1.009
18.61 0.1981 0.4565 1.188 1.026
21.63 0.3193 0.5034 4a 1,050
24.01 0.4232 0.6815 1.071 1,078
25.92 |kPa xj =| 0.5119] yi =| 0.7440} yy =| 1.044) yp := | 1.108
27.96 0.6096 0.8050 1.023 1.138
30.12 0.7135 0.8639 1.010 1.163
31.75 0.7934 0.9048 1.003 1.189
34.15 0.9102 0.9590, 0.997 1.268,
ncsrows(P) oo n=9 i= lan
Psat; = 36.09KPa Psat == 12.30KPa T's (50-4 273.15)K
Data reduction with the Margules equation and Eq. (10.5):
vii ¥2,Pi
Pe Y:
yi xy Psat 3 Xo Peat
481
on GERT; = x1;ln(11,) 32, nfra,
Guess: AO A= 03
= 7 [GERT- (Aarxi, + Ata-x2,) «1,2,
£(A12,Aar
Ai2)
( | :=Minimize(f,A12,421) Aig = 0374 Aap $0197 Ams.
Agi)
y | GPRT}—(Aarm, tara
RMS Error: RMS =:
(RMS = 1,033 107 x1:= 0,001.1
GERT,
goo 0.05
[Aarxt+aye- (xt) Jot (at)
Data reduction with the Margules equation and Eq. (14.1):
3 3
Bu=-18402- By =-18008. By
mol mol
812 #= 2-Bj2- Bi — Bao
flex = Psat) + Po(va)*9r2 |
Er esi ta) a
RT i
482
Guess: A= 01 Ag = 03
£(A12,Az1) = 3 [GERT:~ (Aor x1, +Anz
ist
(Aw
:= Minimize(f,A}2,A21) ANZ
(4a
RMS Error: RMS :=
GERT;
goa
[aorxt+aqa-(-xt) }xt-l-xt)
‘The RMS error with Eqn. (14.1) is about 11% lower than the RMS error
with Eqn. (1
483,
Note: The following problem was solved with the temperature (I) set at the
normal g point. To solve for another temperature, simply change T to
the approriate value.
14.9 (a) Acetylene: Ty 2=308.3K Pg = 61.39bar Ty = 189.4K
ie
les T, = 0.614
2 = 0.08664 Y = 0.42748 Table 3.1
= 2a?
Table3.1— a(T,) = ¥ atte < kg. (3.42)
c
Wa(T) Q
a(t) = a7) bg sn) B(TrsP,) := Eq. (3.50)
aT;
Define Z for the vapor (Zv) Guess:
Given Eq. 3.49)
zw~B(Tr,Pr)
zw ® 14B(T,.P,) -a(T,)-B(Ty,P, WreBiP)\@veo8cr)
Zx(Tr.Py)
Define Z for the liquid (ZI) Guess: zl:= 0.01
Find(zv)
Given Eq. (3.53)
a= B(T,,P,) + (21+ 6-B(Tr,Pe) {el 0-8 (Ty.Py gate)
To find liquid root, restrict search for zl to values less than 0.2, zi < 0.2
“il, ,)
Pind(zl)
484
Define I for liquid (11) and vapor (Iv)
1 ZA(T, Pr) +o-B(TesP,)
1(Tr.P, a (a = ee
(a9) ee-(t- 2) Eq. (6.62b)
Z(T,.P,) +o-B(T;,P,)
WP) = of ees ay
Ingi(T; Pr) = Zi(Ts,Pr) — 1 - In(Zi(Tr,Pr) — B(Tr.Pr)) ~ a(Tr}-1U(Tr.Ps)
Eq. (11.36)
Ingv(T, ,P,) := Zv(T;,P,) ~ 1 ~ In(Zv(Ty,Py) ~ B(TrsPr)) —a(Te}Iv(Tr, Ps)
Guess Psat: Psaty := =
Given Ingl(T,,Psat,) = Ingv(T;,Psat;) Paty := Find(Psat;)
3
Psat, = 0.026 — ZI(T;,Psat;) = 4.742 107° Zv(T;,Psat,) = 0.965
Prat = PsatyPe
Vebir Ans.
The following table lists answers for all parts. Literature values are interpolated
from tables in Perry's Chemical Engineers’ Handbook, 6th ed. The last column
shows the percent difference between calculated and literature values at 0.85Tc.
‘These range from 0.1 to 27%. For the normal boiling point (Tn), Psat should be
1.013 bar, Tabulated results for Psat do not agree well with this value,
Differences range from 3 to > 100%.
Ta (Ki) [0.85 Te (K)| Psat (bar) | Psat (bar) [% Difference
@Tn @ 0.85 Te[Lit. Values|
[Acetylene [ea\~ 160 | et | 2027 | 1878 25%
largon a3 {068 | 128s [2023 | 1870 8.2%
[Benzene 3532) 160 | 4779 | 16028 | 15.52 32%
In-Butane 27a7| 152 | 361s [| 1435 | 12.07 78.9%
[Carbon Monoxide | 817 | 092 | 1730 | 162 | 1291 17.7%
In-Deca 4473) 2.44 | 6250 | 6633 | 521 27.3%
[Ethylene vena} 103 | 2400 [irri | 1769 0.1%
In-Heptane 3716] 208 | 4592 | 76o1_| 759 13%
[Methane fiat—ort | 1620 | 10.30 | 17.33 11.9%
Nitrogen 73) 08s | 1073] 1467} 12.57 16.7%
485
14.10 (a) Acetylene: @ = 0.187 Ty = 3083K Po i= 61.39bar Ty 2= 189.4K
T:=T, Note: For solution at 0.851, set T:=0.85T,. T; =
For SRK EOS: Ty = 0.614
ol e=0 2 = 0.08664 = 0.42748 Table 3.1
ayP
( 2) 2
(Tro) =| 1+ 40.480 + 1.5740 ~ 0.17601 - Ty Table 3.1
a(To}R Te
a(t) = ¥— Ba. (8.42)
c
: T..0)
a(T,) = (" ) Fq. 351) B(T;,Pr) Eq, (3.50)
Define Z for the vapor (Zv) Guess:
Given Eq. (3.49)
w= 1+ (TesPs)4()-B(TesP) ae
Zv(Ty, Py) == Find(zv)
Define Z for the li
Given Eq. (3.53)
id (ZI) Guess: z1:= 0.01
21> B(T;,Pr) + (21 +e-B(Ty, Py) (zl +0: otra ayy reer -2)
att} B(TeP)
‘To find liquid root, restrict search for zI to values less than 0.2, zi < 0.2
Zi(T,,Pr) = Find(zl)
486
Define I for liquid (M1) and vapor (Iv)
icon (ate) +0-B(Tr,Py)
© (ZT,P,)+e-B(TPr)
1 uf 2v(Tr,Pr) ce
o~6 | 2o(T;,P:) +6-B(TrPr)
Eq. (6.62b)
Ingl(T,Pr) = ZI(Te.Pr) 1 = In(Z4(Te,Py) ~ B(Te,Ps)) — a(Ts)-U(Tr, Pr)
Eq. (11.36)
Inbv(Ty,P,) := Zv(Te,Pp) — 1 - In(Zv(Ty,P:) ~ B(Tr.P)) - a(Tr)Av(T,Ps)
Guess Psat: Psat, = 222
Given Ingl(T,,Psat,) = ingv(Ty,Psat;) Psat; := Find(Psat,)
Psat; = 0.017 ZI(Ty,Psat,) = 3.108% 10> Zv(T,,Psat,) = 0.975
Poot = PoatyPe —-Pyat = 1.073 bar Ans.
The following table lists answers for all parts. Literature values are interpolated
from tables in Perry's Chemical Engineers' Handbook, 6th ed. The last column
shows the percent difference between calculated and literature values at 0.85Tc,
‘These range from less than 0.1 to 2.5%. For the normal boiling point (Tn), Psat
should be 1.013 bar. Tabulated results for Psat agree well with this value.
ferences range from near 0) to 6%.
[in (K)] Psat (bar)| 0.86 Te (K)] Peat (bar) | Psat (bar) |% Difference
~@tn @ 0.85 Te] Lit. Values:
[Acetylene Tea] 1073 | 26a | 20016 | 1378 2%
[Argon 673 | 0976 | 1283 | 1878 | 1670 0.5%
[Benzene 3532| 1.007 | 4779 | 18658 _| 15.52 0.5%.
in-Butane 2727| 1008_| 3613] 12238 | 1207 1.4%
(Carbon Monoxide [817 | 1019 | 1130 | 12e71_| 1291 0.3%
in-Decane mars} 1014 | 6950 | 5394] 5.21 2.1%
{Ethylene ea4] 004 | 2400 | 479i6_| 17.68 13%,
In-Hoptane 3716{ 1011 | 4592 [7.779 | 7.59 2.5%
[Methane Talos | 1620 | 1746 [17.33 08%
Nitrogen 773} 0e2 | 1073 | 12617 [1257 0.3%
487
14.10 (b) Acetylene: @ := 0.187 Ty = 308.3K Py :=61.39bar Ty
T:=Ty Note: For solution at 0.85T,, set T= 0.85T,. Ty =
For PREOS: Tr = 0614
or 1492 e:= 1-J2 Q = 007779 Y = 0.48724 Table 3.1
1 t
@(T;,0) a +-(037464 + 1.542260 - aooont hl -12)| eee
a} er?
a(T,) = (Tre) RTS Eq. 3.42)
"
w-a(Tr.0) QP,
aft, = tee) F951) p(T;,P,) =! Bq. B50)
OT,
Define Z for the vapor (Zv) Guess: w= 0.9
Given Eq. (3.49)
z- B(TrsPy)
zv = 1 +B(TesPy) - 9(Tx)-B(Tr.Py) re (t,,P)- (ert on)
Zv(Tr Py) = Find(zv)
Define Z for the liquid (ZI) Guess: zi:= 0.01
Given Eq. 3.53)
21> B(T,.Pr) + (21+6-8(Tr,P,)) (21+ 0 otraayy( eter) 2)
To find liquid root, restrict search for 2 to values less than 0.2 21 < 0.2
ZI(TesPz) == Find(zl)
488
Define I for liquid (11) and vapor (Iv)
1 of ergot)
ZT,.Pr) + ©-B(T,.P,)
U(Tr.Pr)
o
€
Eq. (6.62)
Iv(Te.Py)
o-e
1 ; w(t TpP,) +6 —
Ingl(T,,Pe) == ZI(T,,P,) - 1 ~ In(Zi(T;,P,) — B(Te.Py)) — a(Ts) (Tr. Pr)
Eq. (11.36)
inov(Ts,Py) := Zv(Te.Pe) — 1 - In(Zv(Te,Ps) ~ B(Te.Pe)) ~ a(Te}Iv(TesPr)
Guess Psat: Psat; ae
Given ingi(T, ,Psat,) = Inov(T, sats) Psat, = Find(Psat,)
Psat, = 0.018 ZA(Te, Psat) = 2.795 x 10) Zv(T; ,Psate) = 0.974
Poot = Prato Phay = 109har Ans.
‘The following table lists answers for all parts. Literature values are interpolated
from tables in Perry's Chemical Engineers’ Handbook, 6th ed. The last columa.
shows the percent difference between calculated and literature values at 0.85Te.
These range from less than 0.1 to 1.2%. For the normal boiling point (Tn), Psat
should be 1.013 bar. Tabulated results for Psat agree well with this value.
Differences range from near 0 to 7.6%.
‘Tn (K)[ Peat (bay [0.88 Te (K)] Peat (bar) | Peat (bar) | % Difference)
@™m ‘@ 0.85 Te| Lit. Values
(Acotylene Teaa| 1090 [2627 | teres [1978 [01%
‘Argon 73 {1015 | 1283 | 18676 | 1870 | 0.1%
[Benzene 3532] tote | a7r9_| 15.457_|_15.52 “04%
in-Butane 2727] 1.016 | 3613 | 12084 | 12.07 0.1%
[Carbon Monoxide | 817 | 1.041 | 1130_|tz76a_[_ 1201 “2%
In-Decane aars 7.016 | 250 | 6260 [6.21 05%.
[Ethylene 768.4] 1.028 [2400 _| tv 7aa_|_ 17.89 0.3%
in-Hoptane arte] tote 4592 [767i 7.58 11%
Methane Tia] 099 162047342 | 17.33 0.1%
[Nitrogen 73 {1016 | 1073 | 12817 | 1257 | _-0.4%
489
14.12
(a) van der Waals Eqn. Tr:= 0.7
o=0 s0 apie pees (Tr) = 1
“8 oe
walt Pi
ate = 209 acre,py = SPE zw := 0.9 (guess)
OTe Tr
(Tr, Ps
Given = zv = 1+ B(Tr,Pr) — 4(T0)-p(Te, Py PEP Eq. 3.49)
(wy
Zv(Tr,Pr) = Find(zv)
zi=.01 (guess)
2 1+ BT, Pr) = zl
a(t) BCT, PH)
Given 21 = B(Tr,Pr) + (2) Eq. G53) zl< 0.2
Zi(Tr,Pr) += Find(2t)
BcTr, Pr) Hictr,P8 = B(Tr,Pr)
Zo(Tr, Pr) Z(Tr,PD)
By Eq, (11.36):
Ingv(Tr, Pr) = Zv(Tr,Pr) — 1 ~ In(Zv(Te, Pr) ~ (Tr, Pe) ~ q(Tr)-Lv(Tr,PY)
I(T, Pr) = Bottom pg. 214
Ingl(Tr,Pr) == Zi(Tr,Pr) ~ 1 — In(Zi(Tr, Pr) — B(Tr,Pr)) = q(Tr)-1i(Tr, Pr)
Psate := 1
Given Indl Tr,Psatr) ~ Ingv(Tr, Psatr) = 0 Psatr := Find(Psatr)
‘ZW(Tr,Psatr) = 0.839 Zi(Tr,Psatt) = 0.05 Psatr = 0.2
Infl(Tr,Psatr) = -0.148 Ingv(Tr,Psatr) = -0.148 B(Tr,Psatr) = 0.036
= log(Psatr) 302° Ans.
(b) Redlich/Kwong Eqn Tr := 0.7
o=l e=0 Q = 0.08664 W := 0.42748
a(t) = Tr *
Pah
ST) gcresey = SF Guess:
OTe Tr
a(t
490
zv— B(Tr,Pr)
z-(zv + B(Tr,Pt))
Given zy = 1 + B(Tr,Pr) ~ 4(T:)-B(Tr, Pr) Eq. (3.49)
Zv(Tr,Pr) := Find(zv)
Guess: zl:= 0
Given z= B(Tr,Pr) + 2h(z1+ B(Tr,Pp)-
2< 02 Zi(Tr,Pr) = Find(2l)
7
i( 2vCTe,Pe) + BCT PD) ree =
\ Zv(Tr,Pr)
Iv(Tr,Pr) ==
Z\(Tr, Pr)
By Eq. (11.36):
Ingv(Tr,Pr) s= Zv(Tr,Pr) — 1 - In(Zv(Tr, Pr) ~ B(Er, Pd) — q(Tr)-Iv(Tr, Pr)
Ingl(Tr,Pr) := ZMTr,Pr) — 1 - in(Zi(Tr, Px) — B(Tr,Pr)) — q(TH)-IMTr, Pr)
Psatr = 1
Given _Ingi(Tr,Psatr) = Indy(Tr,Psatr) Psatr := Find(Psatr)
Zv(Tr,Psatr) = 0.913 ZI(Tr,Psatr) = 0.015 Psatr = 0.087
Inv(Tr,Psatr) = 0.083 Ingl(Tr,Psatr) = ~0.083 B(Tr, Psatr) = 0.011
= -1-log(Psatr) @ £0,058 Ans.
1415 (@) xlai=01 9 da=t-xla—xIP0.9 2B 1-xIB
oaaie Ais 2 Ag =2
la A21.A12) exp| x20? Ar2-+2-(Ani ~ Az} xia]
YAB(Agn Ata) = exp] x26°{ Aig +2(Ani - A2)x18] |
ya Aor Ay2) = exp] xter{ Ag) +2-(Ar2 ~ Ani) x2e |]
28(Ag1.Ar2) = exp] x18 {Aa +2-(Ar2— Ai) 328 ||
431
Given xlavylar(Aaq,Ai2) = x1B-718(Aot.Ar2)
x2owy2ee(Agi ,A12) = x28-72B(Agy .A12)
far)
= Find(A12,A2 Agy= 2-747. Aj2= 2.747 Ans.
Aa)
(b) xla = 0.2 Wast-xla xIB:=09 x2B:= 1-xIp
aces Ai2=2 An =2
yle{Aay Ata) = exp] x20 Ara +2-(Aa1 ~ Ara}xlee |
11B(Aa1.Ata) = exp] x26 [Aig +24Aa1 ~ Ara}x1p))]
2a(Agy tz) = exp| xta?[ Agi +2(Ar2 ~ Ani)20] |
28 (Aot Ara) = exo] x16 Ani +24A12—An)}926)]
Given xlaryler(Azy,Ai2) = x1B-y71B(Az1 Az)
x2a-y2a:(Agq,A12) = x2B-72B(Ap1 ,A12)
Ajys 2.148) Ag, = 2.781. Ans.
en
© xla:= 0.1 Rac I-xla — xIB=08 = x2Bs= 1-x1B
Guess: Az = 2 Agi =2
yla(Agi Aya) = exp[ x20? Ato + 2(Ao1 -An)xta]]
‘AB(Aa1.Ar2) = exp] 326°[ Ara +2-(A21 ~ Ayz} xi]
‘a(Aay 412) = exp] xto( Any +2-(Ar2~ Api)}20] |
Tor
¥2B(A21 Ara) = exp| xIB"{ Agr +2-(Aj -An)29]]}
492
Given xloryla(Agi Ai2) = x1B-y1B(Ag1,Ai2)
x2a-y2a.(Az1 Ata) = x2-y2B(Az1,A12)
14.16 (a) xla:= 0.1
Guess:
Given ae
fi ayyxlay? , if
exp) arz| 1+ xloc® exp. a
aia a 7
/ 2 2!
ag x20, 21x28
exp| apy | 142] x20 = exp) azi-| + x28
aj2-xla, ay7-xIB
az ae :
( ) := Find(ay2,a21) aj = 2.747 aor Ans.
\aa1 f
Given apata)?} ( apxip?
exp] ay2] 1+ xlot ® exp) aja b+ = x1 B
(0 agnx2a., \ aarx2B,
ap 1-x2e)
exp| arr 1+
("") = Find(a12,a91)
a2
xip
Given ee
exp) aig} 1+——— | xia =
L 2
2
a12°x1B)
2} 1 1
of ran fos
aa)? agrx28) >
\ " aipxia) axip)
a2)
ind(a12,a21) aj = 281 ag, = 2199 Ans.
agi )
14.18 (a) a= 975
T = 250.450 A(T)
24 T 7 T
Parameter A = 2 at two temperatures. The lower one is an UCST,
because A decreases to 2 as T increases. The higher one is a LCST,
because A decreases to 2 as T decreases.
Guess x= 0.25
Given A(T)-(L= 2.) = (=) Eq.) Ex. 14.5
x20 xs05 xI(1) = Find(x) x21) = 1=x1(1)
494
UCST = 300 (guess)
Given A(UCST) = 2 UCST := Find(UCST)
LesT
Given A(LCST)
00 (guess)
LCST := Find(LCST)
Plot phase diagram as a function of T
nM 225.1. UCST
500 T T i r T
Tl 40F 4
wi
2 300F 4
an \ 1 1 \ 1.
0203. 04 05 06 07 08
AU(TH) x2(T1) ,x1(72) 22(12)
(b) a:= 540 bis-I7.1
T := 250.450 A(T)
L
250300350 400450
T
495
Parameter A =2 at a single temperature. It is
a LCST, because A decreases to 2.as T decreases.
Guess: x = 0.25
Given A(T)-(1-2-x) & In|
Eq. (), Ex. 14.5
x20 x05 x1(T) := Find(x)
LCST := 350 (guess)
Given A(LCST)= 2 LCST = Find(LCST) LEST. 3.346")
Plot phase diagram asa function of TT := LCST. 450
450
400
ale
m= 350 F —
ieee sce epien seperate ee
397 02 03 04 OF 06 07 08
xi(1), 1x1)
©
500
=19.9
T = 250..450 A(T) =
(T) T
Parameter A =2 at a single temperature. It is
an UCST, because A decreases to 2 as T increases.
Guess: x = 0.25
Given A(T)-(1-2-x) = uf! =] Eq. (E), Ex. 14.5,
x
x20 x505 — xI(T): Find(x)
UCST := 350 (guess)
Given A(UCST)= 2 UCST == Find(UCST)
Plot phase diagram asa function of TT := UCST..250
350
= 300
T
a o 02 04 06 08 1
X(T), 1x)
14.20 Guess: xla:
5 IB = 05
Given Write Eq. (14.70) for species 1:
xlovexp| 0.4(1 - xla)?] = xtf-expL o.8-(1 - x18) |
xla_ | _xIB
Ixia 1-xIp
el (Material balance)
ind(xla.,x1B) xia 20.371 x18 ='0.291 Ans.
497
14.22 Temperatures in kelvins; pressures in kPa.
5363.7
Pisat(T) == ox 9.1478- =) water
P := 1600
P2sat(T) s= cn 14651 1- a =!) SF6
1d 3-phase eq)
574-5 of text):
rium temperature and vapor-phase composition (pp.
Guess: T = 300
Given P= Plsat(T)+P2sat(T) —Tstar := Find(T) star = 281.68
Plsat(Tstar)
ylstar ylstar-10° = 695
P
Find saturation temperatures of pure species 2:
Guess: T := 300
Given P2sat(T) = P 12 = Find(T)
TIL := Tstar,Tstar + 0.0001... 12 yII(T) <= 1 -
THe Totar, Vstar + 0.01 . Tstar +6 yitcry = PCD
P
Because of the very large difference in scales appropriate to regions I
and II [Fig. 14.20(a)], the txy diagram is presented on the following page
in two parts, showing regions I and II separately.
yutcrny-10® yimcrap: 108
498
1
Tstar
50 700 750~800 850 ~—900 9501000 1050
yliTny- 108, yt1¢r9- 108
14.24 Temperatures in deg. C; pressures in kPa
Plsat(T) = exp 14.0098 - pee Toluene
\ T+219.79
ee P:= 101.33,
P2sat(T) = oa( 16 2620 - =~ Water
T+ 226.35,
Find the three-phase equilibrium T and y:
Guess: T:=25
Given P-= Plsat(1) +P2sat(T)Tstar
ind(T) Tstar = 84.333
sat(Tst
vylstar := one yistar = 0.444
For z1 y1*, first liquid is pure species 1.
499
14.25
yl=0.7 Guess: Tdew := Tstar
Pisat(Tdew) gow := Find(Tdew)
Tdew = 98.53 Ans.
Given yl
In both cases the bubblepoint temperature is T*, and the mole fraction of
the last vapor is y1*,
‘Temperatures in deg. C; pressures in kPa.
Plsat(T) := oo 13.8887 723)
a 101.33
P2sat(‘T) + oon{to2ma0- 228) water
T+ 226.35
Find the three-phase equilibrium T and y:
Guess:
= 50
Given P= Pisat(T)+P2sat(T) Tstar := Find(T) star = 82.91
Pisat(Tstar)
Istar = 0.474
: y
ylstar
Since 0.35 Tdew,ylo,yl(T)) T = 100,99.9.. Tstar
500
Path of mole fraction heptane in residual vapor as temperature is
decreased. No vapor exists below Tstar.
100
95
85
Shs 036 038 04 042 044 046 048
ylpath(T)
14.26 Pressures in kPa. Pisat:=75 Pasa = 110A
cat) == exp x1)? lal) := exp(A-x1?)
Find the solubility limits:
Guess: xla:= 0.1
Given (1-281) w =u) xla:= Find(xta)
xla
xla = 0.224 xIB —xlo. xB = 0.776
Find the conditions for VLLE:
Guess: Pstar := Psat ylstar := 0.5
Given Pstar = x1B-y1(x1B)-Plsat + (1 — xlo)-y2(xla)-P2sat
yistar-Pstar = xlo-y1(xIa)-Plsat
Pstar
| = Find(Pstar,ylstar) star = 160,699 ylstar = 0.405
ylstar )
601
Calculate VLE in two-phase region.
Modified Raoult’s law; vapor an ideal gas.
Guess: x1 0.1 P= 50
Given P= xi-yl(x1)-Plsat + (1 —x1)-y2(x1)-P2sat
P(xl) == Find(P) loa) := XETLGD. Psat
Pol)
Plot the phase diagram.
Define liquid equilibrium line:
PL(x}) == if(P(x1) < Pstar, P(x), Pstar)
Define vapor equilibrium line:
PV(x1) := if(P(X1) < Pstar,P(X1) ,Pstar)
Define pressures for liquid phases above Pstar:
Pliq := Pstar.. Pstar + 10
x1 = 0,0.01..1
200
ns
PLE 50
PV(x1),
Pig
Pliq 100
5
50
0 02 04 06 08 1
xl yIGrl) xla, x18
802
14.27
x1 := 0,0.05..0.2
at BES PLOd) iG"
2 Ofers. eA Fe enol
foos} P4956) O24
OF 147.658 G3t4
O48: 165525. 0368)
ee 158.508 0387
x1 := 1,0.95..0.8
ee: PLO) yfQ)
i Te). eal
085 T3656 ‘063
23] 137.098 0808]
oa5). 760907). aaa]
os) 156505 oat
xia=0224 x1 = 0.776 ylstar =-0.405
Temperatures in deg. C; pressures in kPa.
3799.89
‘Water: Pi: I HRP ee rea
a) on( T+ 20
477.07)
n-Pentane: sat(T) := exp| 13.8183 - 247707
T+233.21
( 2991.32
n-Heptane: —P3sat(T) := exp] 13.8587 — pe)
\ T + 216.64,
30 23 1-21-22
Ps
(a) Calculate dew point T and liq
assuming the hydrocarbon lay
0133 zl 045
100 Qa Ba 1 ~ Xo
Guess: Tdewl =
503
Given P= x2a-P2sat(Tdewl) + x3a-P3sat(Tdewl)
23-P = x3a-P3sat(Tdew!)
Ra+x3a0 5 |
Ga, Find(x2a,x3a,Tdew!)
Calculate dew point temperature assuming the water layer forms first:
xIB
Given _x1B-Plsat(Tdew2) = z1-P Tdew2 := Find(Tdew2)
Guess: Tdew2 := 100
Tew.
79.055
Since Tdew2 > Tdew1, the water layer forms first
(b) Calculate the temperature at which the second layer forms:
Guess: Tdew3 = 100 a= 22 x3o.
= x20
ylieal ylis2d pao
Given P = Pisat(Tdew3) + x2u-P2sat(Tdew3) + x3a-P3sat(Tdew3)
yL-P = Plsat(Tdew3)
5 2 yl+y2+y3=1
y2:P = x2a-P2sat(Tdew3) x2a+ x30 = 1
yl
y2
y3
ae ‘= Find(y1 ,y2,y3, Tdew3 ,x20.,x301)
x2a
xB,
14.28
yh s0348
‘Tdew3 = 72.886
(©) Calculate the bubble point given the total molar composition of the
two phases
‘Toubble := Tdew3 xa —2 Ba 2
R+23 2+3
x2a = 0.545 x30 = 0.455
Given
P = Plsat(Tbubble) + x2a:P2sat(Thubble) + x3a-P3sat (Thubble)
‘Toubble := Find(Tbubble)
_ Pisat(Thubble)
yl As 0,114
y P ¥
x2a-P2sat(Toubble) Sao
P
x3or-P3sat(‘Thubble) eee
a. ae a
‘Temperatures in deg. C; pressures in kPa.
Water: Plsat(T) := exp| 16.2620 — pee)
T+22635
477.07
n-Pentane: P2sat(T) := ea(3 8183 - P25]
T + 233.21
2991.32
-Heptane: P3sat(T) := SRA eee
|
P i= 101.33 20320 OAS Biz L-zl-2
(a) Calculate dew point T and liquid composition
assuming the hydrocarbon layer forms first:
505
Guess; Tdew! := 70 22a:
Given P = x2a-P2sat(Tdew!) + x3a-P3sat(Tdew!)
23-P = x3u-P3sat(Tdewl) Xa+x3a= 1
xa)
x3a_ | = Find(x2a,x3a.,Tdew!)
Tdew! J
Tdewl = T1018: x30 = 0.733. x2q°= 0267
Calculate dew point temperature assuming the water layer forms first:
xip Guess: Tdew2 = 70
iven xP sat(Tdew2) = 2i-P ‘Tdew2 := Find(Tdew2)
‘Tdew2 = 70.91
Since Tdew1>Tdew2, a hydrocarbon layer forms first
(b) Calculate the temperature at which the second layer forms:
Guess: Tdew3 x3a xa
yli=zl yen
Given P = Plsat(Tdew3) + x2a-P2sat(Tdew3) + x30-P3sat(Tdew3)
y2_ 2
yL-P = Plsat(Tdew3) yl+y2+y3= 1
3 2B
y2-P = x2a-P2sat(Tdew3) X2a+x30= 1
Find(yl ,y2,y3,Tdew3,x2a,,x30)
506
yi = 0292 y= 069 ysS 0.339.
‘Tdew3 = 68/793" x2 = 0.1738.
(c) Calculate the bubble point given the total
molar composition of the two phases
‘Thubble := Tdew3 a= —2 a= 3
+B 2B
x2at = 0.662 xa = 0.338
Given p= Plsat(Toubble) + x2c-P2sat(Thubble) + x3ce-P3sat(Tbubble)
‘Tbubble := Find{Tbubble) ‘Toubble = 44.361
Plsat(Tbubble)
P
x2a-P2sat(Tbubble)
P
x3o-P3sat(Tbubble)
0872
y3 = 0.037
0. 302) Tea ( 84
0.224) ~ (304.2
P:= 10,20..300 (bar)
3
‘40.51
73.83,
83.14 (bar em*3/mol K)
1432 ©
ny
7
Te
TH33I5 (KW) 1
Use SRK EOS
From Table 3.1, p. 99 of text:
c= 0 = 008664 Y= 042748
Gasipsaneniinec satura carrey
: 2
14+ (0.480+1.5740~0.1760°)(1— 14") |
507
—
O-R-Te
(14.34) b Pe (14.35)
(6.842 10 133.076)
a=
os 10) 29.679
_ b2P (14.32) aeartraagiae
Bat) = = ae 80
z= 1 (guess)
Given
i 22 ~ Ba(P)
BEBO PPO ata
Zo(P) == Find(z2)
22(P) + Ba(P)
1n(P) = In] ——————_| (6.62)
- ( Za) Z
For simplicity, let 0, represent the infinite-dilution value of the fugacity
coefficient of species 1 in solution.
Eq. (14.99): lia = 0.088
by qj
9 y(P) == exp] | | -(Za(P) ~ 1) ~In(Za(P) ~ Ba(P))} |
auee
rea ~ a) (2-2 |
Psat) == 0.0102 (bar) Vj = 124.5 (em3/mol)
Eqs. (14.94) and (14.95), with gsatl=1 and (P-Psatl) =P, combine to git
fe Psaty ( v1)
wih) = exp]
Poy (P) RT)
08
ou
oor Vi
yi(P) A
rao? FX
110 a
0 sd 100 150 200 250° 300
P
(es)
126.2,
as
T
Te
0.302
14.33 @ =
0.038
10,20..300 (bar)
40.51
34.00,
R = 83.14 (bar em?/mol K)
308.15 (K) 0 Trs=
Use SRK EOS
From Table 3.1, p. 99 of text:
ol c= 0 Q = 0.08664 W = 0.42748
a
[1 +-(0.480 4 1574-0 —0.17602).(1 — 2° sf
>
ORTe
__ Weak
: 14,34) b (14.35)
Pe se Pe
(7.298% 10 133.076
as) b
\6.713 10 26.737
509
= (1432) = (14.36)
m= 1 (guess)
Given
22 - Ba(P}
22 = 1+ Bo(P) ~ q2-B2(P)- (o=68xP)o+eBP) (14.31)
Za(P) := Find(z9)
(Z2P) +82)
fete (6.62b)
1a) = (6.62b)
For simplicity, let © represent the infinite-dilution value of the fugacity
coefficient of species 1 in solution.
1y23=0.0 Bq. (14.99):
i] by
91(P) == exp, [2 (Z2(P) ~ 1) - In(22() — ne] :
Coa
Ly
4-2} 2(1- wy(2) em
4
Psaty := 2.910" (bar) Vi z= 125 (em3/mol)
Eqs. (14.94) and (14.95), with osatl =1 and (P-Psatl)=P, combine to
give:
(
vy we oat PV,
1) + ool ser
Pop (P)
510
30 100~«1S0~=«0D~~S=O«S0S«0D
Note: y axis is log scale.
st
Chapter 15 - Section A - Mathcad Solutions
Final state: lee at 32 degF.
BIU BIU
(0.02 — cee eae ibptankine
(70+ 459.67)-rankine
@ Cooling
water @70(F))
Cond.
8 €
w
s 4) comp: Throttle
A D
ice @ 32) | "| Water @ 70(F)
<2 e2O jg Water @ 70
Point A: sat. vapor at 32 degF.
Point C: sat. liquid at 70 deg. P
35.79(psia).
Point D: Mix of sat. liq. & sat. vapor at 32 degF with the enthalpy of Point C.
Point B: Superheated vapor at 85.79(psia) and the entropy of Point A.
Data for Points A, C, & D from Table 9.1. Data for Point B from Fig. G.2.
512
Wiaeal := Hz ~ Hi - To-(82~ $i)
BIU
Wideal = 12.466.
we Tom
Wedotigeat ‘= mdot- Wigeal Wdolideat <13.15kW Ans.
(b) For the Carnot heat pump, heat equal to the enthalpy change of the
water is extracted from a cold reservoir at 32 degF, with heat
rejection to the surroundings at 70 degF.
BTU
Hy-Hy Qc = “18137
491.67-rankine Ty =
Qc:
BIU
THT
x <) Work = 14.018
Woot := mdot Work
Waotigeat
Wot
nee
‘The only irreversibility is the transfer of heat from the water as it cools
from 70 to 32. degF to the cold reservoir of the Carnot heat pump at 70
deg.
(©) Conventional refrigeration cycle under ideal conditions of operation:
Isentropic compression, infinite flow rate of cooling water, &
minimum temp. difference for heat transfer =
For sat, liquid and vapor at 32 degF, by interpolation in the table:
BTU BTU
Ha := 107,60-—— Sa c= 0.2223-———___
- Dm be Ibgrrankine
For sat. liquid at 70 degF:
TU
He := 34.58-——— Hp = He
Ic es p= He
For superheated vapor at 85.79(psia) and S = 0.2223:
BIU
‘Hg = 114——
' Tom
513
rent circulation rate:
Ibm
(Hp - Hy) :
= mdot = 2.484—"
mdot
Ha-Hp see
Wdot := mdot-(Hg ~ Ha) Waot=16.77kW Ans.
Wdotigeat
2 nye 0784 Ans.
Th Wdot It.
The irreversibilities are in the throttling process and in heat transfer in
both the condenser and evaporator, where there are finite temperature
differences.
(4) Practical eyele.
Point A: Sat. vapor at 24 degF.
Point B: Superheated vapor at 134.75(psia).
Point D: Mix of sat. lig. and sat. vapor at 24 degF with H of point C,
Point C: Sat. Liquid at 98 degF.
(Note that minimum temp. diff. is not at end of condenser, but it is not
practical to base design on 8-degF temp. diff. at pinch. See sketch.)
8@)
90) ye
id and vapor at 24 degF:
For sat,
e
514
For sat, liquid at 98 degF, P=134.75(psi
He = 44.24- BIL S¢ 25 0.0902-—BIU_
mm ar Fankine
For isentropic compression, the entropy of Point B is 0.2229 at
P=134.75(psia). From Fig. G.2,
Hp-H,
Hye
1
i" BTU The entropy at this His
He So aneeib read from Fig. G.2 at
P=134.75(psia)
Hp- Hi
0228 BY yee xp eo up = 0.288
Tbgyrankine Heap ~ Fig
BTU
Sp = Stiq + ¥D-(Svap ~ Sti Sp = 0.094
1D = Siiq + xD-(Syap ~ Stig) D Roe
Refrigerent circulation rate:
Ibm
-(Hp-Hy).1-28
mdot := ————_S° ndot = 2.91428
ecceeHAe Ap) see
Webot := mdot-(Hp ~ Ha) Waot
AT22KW Ans.
Wdotigeal
Wdot
eH nig = 0.979" Ans.
THERMODYNAMIC ANALYSIS Tg: (70+ 459.67)-rankine
Wadotjost.compressor *= mdot-Ty+(Sp- S4)
Qdoteondenser ‘= mdot-(Hc - Hp)
Weottost.condenser °= mdot-Tg-(Sc ~ Sg) ~ Qdotcondenser
515
Waotjostthrottle = mdot’Tg-(Sp - Sc)
Weotjostevaporator °= To-/ mdot(S4— Sp) .. 1
bm
+L—(S2-s))
L sec J
Welotigcay = 13.152kW 27.85%
Weotiost compressor = #.305kW 17.59%
‘Wadotjostcondenser = 14A78KW 30.02%
Waitiost:throttle = 6.621 kW 14.02%
Weloticstevaporaior = 4.968kW. 10.52%
The percent values above express each quantity as a percentage of the
actual work, to which the quantities sum.
516
15.2. Assume ideal gases. Data from Table C.4
282984. AGg9g := 2571903
Allo
AH298 ~ AGo9g
as
298 398.1 5K
J
ASo9g = ~86.513
1298 K
BASIS: 1 mol CO and 1/2 mol 02 entering with
accompanying N2=(1/2)(79/21)=1.881 mol
nico = Iemol — Myip = 2.381-mol —ngoy == I-mol nz = 1.88]-mol
(a) Isothermal process at 298.15 K:
2.38 air
2.88
ar 5!
Since the enthalpy change of mixing for ideal gases is zero, the overall
enthalpy change for the process is
AH := AHy9g For unmixing the air, define
aN?
n= N=079 yp
By Eq. (12.35) with no minus sign:
Sunmixing = Pair R-(yr-In(y1) +y2-In(
AS,
i
ruining = ~2017
5t7
For mixing the products of reaction, define
02
BN2 + ACO?
y= 0347 2
neo2 + AN2}-R-(yr-ln(y1) + yo-In(y2))
ASmnixing = 15. 465. —
AStnixing “=
AS °= ASunmixing + AS298 + AS icing -81 m3
300-K Wideal ‘= AH - Tg-AS Ans,
Te
(b) Adiabatic combustion:
1 mol CO,
1.88 mol N
sie
298.15 K
1 mol CO AFapx
0.5 mol O»
1.88 moi No
Heat-capacity data for the product gases from Table C.1:
n02'5:457 + nyz'3.280
coe A= 11.627
mol
nco2' 1.045 + my2-0.593.
5 oo B= 216x109
mol
ncog'— 1.157 + nyz-0.040
eee 0° D = -1.082% 10°
mol
518
1
C1
For the products, AHp® «| = ar To = 298.15-K
To
‘The integral is given by Eq, (4.7). Moreover, by an energy balance,
AHy9g + AHp = 0
Guess <
Given
For the cooling process from this temperature to the final temperature of
298.15 K, the entropy change is calculated by
10P3(2622.6, 298.15, 11.627,2.160-10"$,0.0,~1.082-10°) = -29.701
ICPS := -29.701 AS := R-molb ICPS AS = ~246.934 2
Wideal.cooling = AH - Tg-AS
AH = -2.83 x 10°) Wigeai séoiing = ~2089043 Ans.
Wideal.coolins
ny = ne ne= 0.8078 Ans.
Wideat
‘The surroundings increase in entropy in the amount:
Qo
(AH298- Wideatcooting) —ASg == x wee
The irreversibility is in the combustion reaction. Ans.
519
15.3 1 Saturated vanor
2.700 kPa
H,= 26017 BOK OF te
f swam, / vival Saturated Squid
S,= 8.2244 ‘TRICKS | 1.000 kPa oa 479.88 C; 45303 K
2_ Saturated vapor
275 kPa
Hy» 27207
S,= 7.0201
For the sat, steam at 2700 kPa, Table F.2:
ks KI
Hy = 2801.7 Sp = 62244
1 z 7 ek
For the sat. steam at 275 kPa, Table F.2:
ky kJ
H == 2720.7-- Sa := 7.0201-——
2 ie 2 kek
For sat. liquid and vapor at 1000 kPa, Table
kJ ki
‘jig *= 762.6-— Shi 2.1382-——
Hig kg Ha kek
KI kJ
Hyg; nd Syap *= 6.5828 —— Trot *= 453.03K
p= 262 =p ek 7
(a) Assume no heat losses, no shaft work, and negligible changes in kit
and potential energy. Then by Eqs. (2.30) and (5.22) for a completely
reversible process:
ic
Ag(H-mdot) = 0 Ag(Semdot) = 0
We can also write a material balance, a quantity requirement, and relation
between H3 and S3 which assumes wet steam at point 3.
‘The five equations (in 5 unknowns) are as follows:
Guesses: mdoty = 0.1-88 ——mdoty s= mdotymdoty += mdoty + mdoty
s
Hy +H) H3— Hig
$3 = Sgt ——
2 ecetnaceaer ee
H3
520
Given
Ki
Hy-mdots — Hy-mdot) ~ Hy-mdot = 0
8
lots ~ Sy-mdoty ~ Sp-mdot = 0
sK
ki
mdots = mdoty + mdoty (H3 ~ Hijg)-mdots = 300
Hg ~ Hii
83 * Stiq + _
sat
mdot;
mdoty
mdots | := Find(mdot , mdotz,mdot,H3,83)
Hs
83
‘mdots poor
Hy= 27679
Ee Re
Steam at Point 3 is indeed wet.
(b) Turbine: Constant-S expansion of steam from Point 1 to 1000 kPa
results in wet steam of quality
: S1-Stig tHe .
Xb = Sig Hiturb = Hig + Xturo-(Hyap — Hig)
3 ky
Xturb = 0.919 Hiusp = 2614107
Neurb *= 0.78 Hrs = Hy + Meurb-(Hturb~ Hi)
Hyyrb = 2.655 x 10°
kg
921
quip = Sturb == Stig + Xturb (Svap~ Sig)
Ky
turd = 0.94 Sub = 65167
Compressor: Constant-S compression of steam from Point 2 to 1000 kPa
results in superheated steam. Interpolation in Table F.2 yields
Heomp ?= 2993, se Neomp ?= 0.75
( Hreomp ~ Ha KI
Heomp ‘= Ha ema Hoomp = soe
Jeomp
; Pa
By interpolation:
Scomp *= 7.1803:
kK
‘The energy balance, mass balance, and quantity requirement equations of
Part (a) are still valid. In addition, The work output of the turbine equals
the work input of the compressor. Thus we have 4 equations (in 4
unknowns):
Guesses: mdot, == 0.086 8 dot := 0.064 88
s s
mdots := 0.1588 Hy := 2770.82
5 kg
Given
(Heomp ~ Ha)-mdoty = -(Htars — Hs)-mdoty
Hy-mdot3 — H-mdoty ~ Hy-mdotg = 082
$
any KI
mdots = mdot) + mdot (H5 - Huig):mdots = 300
5
mdoty
mdot
= Find(mdoty .mdot2,mdot3, H3)
mot
Hs }
522
Steam at Point 3 is slightly superheated.
ki
By interpolation, 83 65876 Fe
THERMODYNAMIC ANALYSIS — Tg := 300K _—_ (assumed)
By Eq. (5.25), with the enthalpy term equal to zero:
Wdotigeal ‘= To:(mdots-$3 ~ mdoty-$1 — mdotz-S2)
Wdotigeat = 6.014kW
Waottostturb := Tormdoty-(Sturo~ Si)
Wotiost.comp := Te'mdot- (Scomp ~ S2)
Weotost.mixing
To-| (mdots-$3 ~ mdoty-Sturb) ~ mdotz-Scomp |
48.2815%
34.1565%
17.5620%
The percent values above express each quantity as a percentage of the
absolute value of the ideal work, to which the quantities sum.
623
Some property values with reference to Fig, 9.1 are given in Example 9.1.
Others come from Table 9.1 or Fig. G.2.
For sat. liquid and vapor at the evaporator temperature of 0 degF:
BTU
Hig = 12.0905" Hyap = 103,015 222.
BTU BTU
8, = 0.22525-———__—_—— Stig := 0.02744 ——___
tied Ibarrankine : Tbrankine
For sat. liquid at the condenser outlet temperature of 80 deg:
Hy = Hq
Hi - Hii
xs $1 = Stiq + x1-(Svap - Stiq)
yap — Hig
: BTU
x1 = 0.285 eles Tope tankine
From Example 9.1(b) for the compression step:
Hg := Hy + AH Hy = 120.5220
Ibm
From Fig, 9.3 at H3 and P = 101.37(psia):
i BU
83 = 0.231 ine aot t= 1845.15
7 if
Weot := mdot-AH Waot = 3.225 ot
F
524
‘The purpose of the condenser is to transfer heat to the surroundings.
Thus the heat transferred in the condenser is Q in the sense of Chapter
15; ie.,it is heat transfer to the SURROUNDINGS, taken here to be at a
temperature of 70 degF.
Internal heat transfer (within the system) is not Q. The heat transferred in
the evaporator comes from a space maintained at 10 degP, which is part of
the system, and is treated as an internal heat reservoir.
‘The ideal work of the process is that of a Carnot engine operating
between the temperature of the refrigerated space and the temperature of
the surroundings.
To = (70+ 459.67)-rankine TH=To
Qdote : 120000 272 Tes (10 + 459.67)-rankine
uote 4 BTU
Weotigea) = |Qdot Weotideal = 1.533 x 10° ——
ideal = |Qdotc| s tideal =
Wotjost comp *= Tormdot-(S3—S2)
Hq — H)-mdot Qdot = -1.523 x 2Te
Waotios.cond = Te-mdot-(S4— $3) — Qdot
Wdotiost throttle “= Temdot-(S - Sa)
Wedotjost evap = Te-mdot (82 - $;)
Hy ~ Hp
Te
+To° -mdot
525
The final term accounts for the entropy change of the refrigerated space
(an internal heat reservoir).
Wot ™15309 jae
Webticsisomp.
Waotioe cana 3625: 2a
BTU
4730.2——
obits
Waotlost.trattie 2
Wabotpsccép = 2047 “—
47.53%
17.42%
11.24%
14.67%
9.14%
‘The percent values above express each quantity as a percentage ofthe
actual work, to which they sum:
Weor = 32282: 3a
The discussion at the top of the second page of the solution to the
preceding problem applies equally here. In each case,
To = (70+ 459.67)-rankine
TH = Te
‘The following vectors refer to Parts (a)-(e):
(40
459.67)-rankine
526
600
500
Qdote := | 400 | BTL
see
300
200
>
TH
Walotigeat = {loi
For sat. liquid and vapor at the evaporator temperature, Table 9.1:
21.486 107.320
18318 105.907
Hyap 2= | 104.471 Ie
103.015
101.542
0.04715 0.22244
0.04065 0.22325
Stig := | 0.03408 0.22418 aes
0.02744 0.22525
0.02073. 0.22647,
For sat. liquid at the condenser temperature:
‘Hyg := 37. on 84 = 0, 0190 Hy = Hy
Hi-H pee eee
xis ing Hn Stig + xt-(Svap ~ Stig) |
From the results of Pb. 9.9, we find:
AIT From these values we must find the
118.9 ‘corresponding entropies from Fig. G.2.
0.1 [Bt ‘They are read at the vapor pressure for
he 80 degF of 101.37 kPa. The flow rates
121.7 come from Problem 9.
123.4
0207 8.653
0.229 7361
BTU.
$315] 0.231 | ——_ mdot := | 6.016
Ibyrtankine
34] ais |
0.237, 3.146,
927
ae
[To-mdot-(S3 - $2)
(Hg - H3)-mdot]
—_
[To-mdot-(S4- $3)] ~ Qdot
——___»>
[To-mdot-(: - 5,
Wdotjost throttle
—~——__—_>
Webotiosievap = [Te-mdot-(S2 - $;)]
gE Nets
Hy) - Hp
+) [ToS fmdor
‘The final term accounts for the entropy change of the refrigerated space
(an internal heat reservoir).
528
15.6
In cach case the ideal work and the lost work terms sum to give the actual
work, and each term may be expressed as a percentage of the actual work.
The discussion at the top of the second page of the solution to Problem
15.4 applies equally here.
Tq == (70-4 459.67)-rankine
Te = (30+ 459.67)-rankine
TH
Wadotigegi = 163.375——
Waotieal
see
(lave re BTU
For sat. liquid and vapor af the evaporator temperature, Table 9.1:
sais BU
by,
lbm-rankine
7.978. BEY
Ibm
From Problem 9.12,
= 116-BTU Soa s= 02435.—BTU
Hoa Us: Tbarrankine
529
\
Hy = Haq + 14,667.51 Hy = 130.67 BTS
From Fig. G.2 at this enthalpy and 33.11(psia):
BTU
byrrankine
S3 = 0.2475-
Energy balance on heat exchanger:
Hy == H4- Hoa +H2 Hy = 27885220
Ibm
$1 := Stiq + x1-(Svap — Stig)
BIU
8} = 0.061 >
Tbgytankine
Upstream from the throttle (Point 4) the state is subcooled liquid with
the enthalpy:
Haga = Hy
The entropy at this point is essentially that of sat. liquid with this
enthalpy; by interpolation in Table 9.1:
0.059621 _
Tbprrankine
Saa ?
Ibm,
From Problem 9.12: mdot := 25.634-—=
sec.
Wotios..comp = Tormdot-($3 ~ S24)
Qdot := (H4- H3)-mdot
Weotjos.cond °= To'mdot-(S4— $3) - Qdot
530
Weotiostthrottle = Te-mdot (8; - S4a)
Wabotiost.evap ‘= Te-mdot-(S2 - $1)
Hp)
oo | mdot
Te }
The final term accounts for the entropy change of the refrigerated space
(an internal heat reservoir).
Weotjostexchanger *= Ta-mdot-(S2q - So +S4a— $4)
Wot := mdot-(H3 - H2a)
3.45%
14.45%
23.16%
11.99%
4.30%
‘The figures on the right are percentages of the
actual work, to which the terms sum.
531
15.7
0.5 kg/s
2
¥
i
3
1_ Feed Slury
Toe | \ 100 C
Compression to a pressure at which condensation in coils occurs at
110 degC. Table F.1 gives this sat. pressure as 143.27 kPa
Meomp '= 0.75
Hy 4io.ne (sat, liquid)
kg
i ki
Hy = 2676.0-5 $2 1= 7.3554 (sat. vapor)
2 z 2 eR ah eapo)
For isentropic compression (o 143.27 kPa, we find by double interpolation in
Table F.2:
- Hp
5 := 2737.02 Hy = Hp+ : Hy = 275732
kg Neomp kg
By more double interpolation in Table F.2 at 143.27 kPa,
$3 = 7.4048
By an energy balance, assuming the slurry passes through unchanged,
KI
Hag i= Hy + Hy -Ho Hy = 500.45—
g
532
158
This enthalpy is a bit larger than that of sat, liquid at 110 degC; find quality
and then the entropy:
a Cy ‘ a
461.3-— Hyy := 2230.0-— Stic 1.4185-——
ke ke ma iek
kt Hy — Hig
$203-— a = 0018
kek eee 2
S4= Stig + X4°S] Sq = 1.5206 ue mdot == 0.5:
14 Sig’ X4-Stv S84 = 1.520055 = 0
To = 300K
Weotideas = mdot{ Hg - Hi ~ To-(S4 ~ $1) ]
Weottosrevap = mdat-Te-(S4-$3+S2-S1)
Weotiostcomp = mdot-To-(S3 - S2)
‘Wot := mdot-(H3 — Ho)
21.16%
60.62%
18.22%
The figures on the right are percentages of the
aM actual work, to which the terms sum.
‘Wadot =
A thermodynamic analysis requires an exact definition of the overall
process considered, and in this ease we must therefore specify the source
of the heat transferred to the boiler.
Since steam leaves the boiler at 900 degF, the heat source may be
considered a heat reservoir at some higher temperature. We assume in
the following that this temperature is 950 degF.
‘The assumption of a different temperature would provide a variation in
the solution.
533
‘The ideal work of the process in this ease is given by a Carnot engine
operating hetween this temperature and that of the surroundings, here
specified to be 80 degR.
We take as a basis 1 bm of H2O passing through the boiler. Required
property values come from Pb. 8.
Ty '= (459.67 + 950)-rankine Tc := (459.67 +80)-rankine Ta == Te
Subscripts below correspond to points on figure of Pb. 8.7.
AY) (2576 S1\ (03070
He 1461.2 & 1.6671
1282.2] aru Ss} }i74a31} pru
1047.8 | Ibm sa|* | 1.9748 | Ibperankine
Hs 69.7 s5| | 0.1326
Hy} \2502 s;) (oat
Widea
For purposes of thermodynamic analysis, we consider the following 4 parts
of the process:
The boiler/heat reservoir combination
‘The turbine
‘The condenser and throttle valve
‘The pump and feedwater heater
Te: is Si}t te St]
Wiost.boiler.reservoir
m := 0.18688:lbm (From Pb. 8.8)
‘o-L m-(S3 - $2) + (I-lbm=m)-(S4 ~ S2) |
‘The purpose of the condenser is to transfer heat to the surroundings, The
amount of hea
Wiostturbine
1 Ibpy Hs — (-lbm ~ m)-Ha ~ m- Hy
-829,045BTU
534
15.9
g'[l-lbpr$s — (I-lbm~m)-S4—m-$7}~Q
Wiostcond valve
Wiostpurap heater “= Tor[,I-lbmr(S1 - Ss) + m-(S7~ $3) ]
The absolute value of the actual work comes from Pb. 8.8:
Wabsvanue = 374.6FBTU 50.43%
30.24%
13.30%
4.90%
119%
42.82111U ‘The numbers on the right are percentages of the
absolute value of the ideal work, to which they
(absolute value) sum,
Refer to Fi
the liquefaction section to the right of the dashed line. Enthalpy and
entropy values are those given in Ex. 9.3 plus additional values from
the reference cited on page 323 at conditions given in Ex. 9.3.
re 9.7, page 322, The analysis presented here is for
Property values:
‘Hg := 1140.0- oo
kg
i
Hg := 1009,7-— S: = 83894
kg 7 kek
i Ki
Hy := 719.8-— 144
a kg kg-K
Ho := 285, add 28.
kg kek
ki
Sto 5 9.521
a kek
535
Hg = Hs S6:= Ss
Hi
Hio S12 = Sio
To i= 295K
The basis for all calculations is 1 kg of methane entering at point 4. All
work quantities are in kJ. Results given in Ex. 9.3 on this basis are:
Fraction of entering methane that is liquefied:
Fraction of entering methane passing through the expander:
On this basis also Eq. (5.26) for Ideal Work, Eq. (5.33) for Entropy
Generation and Eq. (5.34) for Lost Work can be written:
Wiost = Ta SG
al = A(H-n)gs Tor (Sem)p, Sq = A(S-m)g,—
Wideal = [His:(1 =z) + Ho-z~ Ha] -To[S15-(1 - 2) + S¢-z- $4]
Re
(b) Heat Exchanger Il: So,p := [ (S7- S6)-(1 - x) + (Sia - Si3)-(1-2) ]
S0313A- — Wosth = Te Se. = 92.24
Soo Mea lostb Sab Wiostb eg
536
(@) Throttle:
Soa'=[Soz+ Syo(1-2-x)-Sr~%)]
Wiostd
Entropy-generation analysis:
SGy
SG
S_Ge
$4
x
= TeSad
kiMkeg-K
0-044
0.313
0.157
0.964
1478
Work analysis, Eq. (15.3):
Note that:
kaikg
$3.20
92.24
4624
28430
489.00
Es |Wigeall
837
Percent of ¥
2.98%
21.18%
10.62%
68.22%
100.00%
Percent of
10.88%
2.66%
18.86%
9.46%
58.14%
10.00%
Chapter 1 - Section B - Non-Numerical Solutions
1.1 This system of units is the English-system equivalent of SI. Thus,
Be = (by )()(poundal)~
1.2 (a) Power is power, electrical included. Thus,
energy
‘ime
(®) Blectric current is by definition the time rate of wansfer of electrical charge. Thus
Power [=]
Charge [=] (electric current)(time) [=) A-s
(0) Since power is given by the product of current and electric potential, then
current As
(@) Since (by Ohm’s Law) current is electric potential divided by resistance,
werk
Electric potential [=] P°** a
electric potential
current als
kgm?
Resistance [=] =
(6) Since electric potential is electric charge divided by electric capacitance,
charge, Ast
Capacitance [=] = [=]
‘apacitance ] ieSrie potential gg?
1.3 The following are general:
Inx = In10 x logigx Cy
100_ kPa
P#/kPa = P™/tom x —
z On * Rss wo
(PC =T/K~ 273.15 ©
By Eqs. (B) and (4),
100
In P*/kPa = In 10 logy P™/ es
ie B10 PM tor + In 756 Ger
The given equation for logiy P* /torr is
logy) P*/torr
Combining these last two equations with Eq. (C) gives:
tno (a7,
in Pika,
a + ina
TK 27 750.061
3
3026 (a~ pe) -
\ WK) en
‘Comparing this equation with the given equation for In P™/kPa shows that:
A= 2.3026 a — 2.0150 B= 230266 ce
=273.15
539
1.9 Reasons result from the fact that a spherical container has the minimum surface area for a given interior
volume, Therefore:
(@ A minimum quantity of metal is required for tank construction.
(6) The tensile stress within the tank wall is everywhere uniform, with no sites of stress concentration.
Moreover, the maximum stress within the tank wall is kept to a minimum,
(©) The surface area that must be insulated against heat transfer by solar radiation is minimized.
its of mass-velocity®. Its fundamental units are therefore:
1.17 Kintic energy as given by Eq. (1.5) has u
Ex Leg? [=] Nm (JS
Potential energy as given by Eq. (1-7) has units of mass-length-acceleration. Its fundamental units are
therefore:
Ee f)kemms
1.20 See Table A.1, p. 652, of text,
‘© (atm) * 1 bar = 1/0.986923 = 1.01325 bar
© (Btu) © 1 kJ = 1/0.947831 = 1.05504 ks
© I(hp) = 0.75 kW = 1/1.34102 = 0.745701 KW
‘# 1(in) = 2.5 om = 2.54 om exactly, by definition (see p. 651 of text)
# Iifbq) = 0.5 kg = 0.45359237 kg exactly by definition (see p. 651 of text)
‘= l(mile) ~ 1.6 kan = $280/3280.84 = 1.60934 km
(quart) © 1 liter = 1000/ (264.172 x 4) = 0.94635 liter (I Titer = 1000 em?)
yard) ~ 1m = (0.0254)(36) ~ 0.9144 m exactly, by definition of the (in) ané the (yard)
An additional item could be:
© Moile)(i)-!~ 0.5 m s“!= (5280 /3,28084) (1/3600) = 0.44704 m st
1.21 One procedure here, which gives results that are internally consistent, though not exact, is to assume:
1 Year [=] 1 Ye [+] 364 Days
This makes 1 Year equivalent to exactly 52 7-Day Weeks. Then the average Month contains 30! Days
and 4} Weeks. With this understanding,
1 Year [=] 1 Yr [=] 364 Days [=] (364)(24)(3600) = 31,449,600 Seconds
Whence,
#1 Se [=] 31.4496 Second 1 Second [=] 0.031797 Se
1 Mn [=1314.496 Second 1 Minate [=] 60 Second [=] 0.19078 Mn
#1 Hr (=] 3144.96 Second 1 Hour [=] 3600 Second {=] 1.14469 Hr
#1 Dy [=]31449.6 Second 1 Day [=] (24)(3600) Second [=} 2.74725 Dy
#1 Wk (=1314496, Second 1 Week (=] (7)(24)(3600) Second (=] 1.92308 Wk
#1 Mo [=] 3144960 Second 1 Month =} (4)(7)(24)(3600) Second[=] 0.83333 Mo
The final item is obviously also the ratio 10/12.
540
umerical Solutions
Chapter 2 - Section B - Non-!
2.3 Equation (2.2) is here written: AU" + AEp+ AEK=Q4+W
(@) In this equation W does not include work done by the force of gravity om the system. This is
accounted for by the AEy term. Thus, W’ = 0
(&) Since the elevation of the egg decreases, sign( AE) is (~>)
(6) The egg is at rest both in its initial and final states; whence AEx = 0.
@ Assuming the egg does not get scrambled, its internal energy does not change; thus AU" = 0.
(©) The given equation, with AU" = AE, = BV = 0, shows that sign(Q) is (—). A detailed exam-
ination of the process indicates that the kinetic energy of the exg just before it stikes the surface
ppears instantly as internal energy of the egg, thus raising its temperature. Heat transfer €0 the
surroundings then returns the internal energy of the egg to its inital value
26 If the refrigerator is entirely contained within the kitchen, then the eleciricsl energy entering the re
fiigerator must inevitably appear in the kitchen. The only mechanism is by heat transfer (from the
condenser of the refrigerator, usually located behind the unit or in its walls). This raises, rather than
lowers, the temperature of the kitchen, The only way to make the refrigerator double as an air condi-
tioner isto place the condenser of the refrigerator outside the kitchen (outdoors)
2.7 According to the phase rule (Fg, (2.7)], F = 2~ 1 +N. According to the laboratory report a pure
‘material (= 1) isin 4-phase (x = 4) equitibrium. If this is true, then = 2~4 41 = —1. This is
not possible; the claim is invalid,
2.8 The phase rule (Eq. (2.7) yields: F =2—x +N =2-2+2=2. Specification of T and P fixes the
intensive state, and thus the phase compositions, ofthe system. Since the liquid phase is pure species
1, addition of species 2 to the system increases its amount in the vapor phase. Ifthe composition of
the vapor phase is £0 be unchanged, some of species 1 must evaporate from the Liquid phase, thus
decreasing the moles of liquid present.
2.9 The phase rule [Eq (2.7)] yields: F = 2— 3 +N =2-243-=3. Withonly 7 ond P fixed,
cone degree of freedom remains. Thus changes in the phase compositions are possible for the given
T and P, [ethanol is added in a quantity that allows 7 and P to be restored to their initial values,
the ethanol distributes itself between the phases so as to form new equilibrium phase compostions and
altered amounts of the vapor and liquid phases, Nothing remains the same except 7 and P.
2.10 (a) Since F = 3, fixing T and P leaves a single additional phase-rule variable to be chosen,
(©) Adding or removing liguid heving the composition of the liquid phase or adding or removing
vapor having the composition of the vapor phase does not change the phase compositions, aid
does not alter the intensive state ofthe system, However, such additions or removals do alter the
‘vera composition of the system, except forthe unusual ease Where the two phase compositions
aze the same, The overall composition, depending on the relative amounts of the two phases, cat
range from the composition of the liquid phase to that ofthe vapor phase.
2.14 Ifthe fuid density is constant, then thc compression becomes a constant-V’ process for which the work
is zero. Since the cylinder is insulated, we presume that no heat is transferred, Equation (2.10) then
shows that AU = 0 for the compression process.
S41
2.16 Electrical and mechanical irreversibilities cause an increase in the internal energy of the motor, man-
ifested by an elevated temperature of the motor. The temperature of the motor rises until a dynamic
equilibrium is established such that heat transfer from the motor to the stroundings exactly compen-
sates for the ireversibilites. Insulating the motor does nothing to decrease the irreversbilities in
the motor and merely causes the temperature of the motor to rise until heat-transfer equilibrium is
reestablished with the surroundings. The motor temperature could rise toa level high enough to cause
damage.
2.19 Lot symbols without subscripts refer to the sotid and symbols with subscript w refer to the water.
Heeat transfer from the solid to the water is manifested by changes in internal energy. Since energy is.
conserved, AU" = — AU, If total heat capacity of the solid is C' (= mC) and total heat capacity of
the water is Ci, (= mCu), then:
CUT = To
~ChTe = Ton)
or
Teo ~ Ee(T ~ Ta) 4)
equation relates instantaneous values of 7, and 7. Itcan be written in the alternative form:
TC! — T9C! = ToC, ~ ToC
or Tag, + TC! = Ty, TC! (By
‘The heat-transfer rate from the solid to the water is given as Q = K (Ty — 7). [This equation implies
that the solid is the system.] It may also be written:
ar
1 = Kha c)
OF =Kte-D «
In combination with Eq, (4) this becomes:
i:
«[tm-Go-m-7]
where both @ and # are constants. The preceding equation may now be written:
aT
dt
a 1d(a~ AT)
Br B a-aT
-pr
at
Rearrangement yields:
Integration from Ty to T and from 0 to r gives:
aT
a Blo
which may be written: xp(—Bt)
When solved for ' and rearranged, this becomes
rage(n- %) exp(-Pr)
3
@ Tah + ToC!
whereby the definitions of « and p, = TT
When + = 0, the preceding equation reduces to 7 = To, as it should, When r = 00, it reduces to
T = a/f. Another form of the equation for af is found when the numerator on the right is replaced
by Eq. (B)
a _TCh4TC
By inspection, T—=a/8 when T=, the expected result.
220 The genoral equation applicable here is Eq. (230):
al(i+$ o+W,
(a) Write this equation for the single stream flowing within the pipe, neglect potential- and kinetic.
energy changes, and set the work term equal to zero. This yields:
(Alyn = @
(®) The equation is here writen for the two streams (Land I} lowing in the two pipes, again neglecting
‘any potential- and kinetic-energy changes. There is no work, and the the heat transfer is internal,
between the two streams, making Q = 0. Thus,
(AM + AM = 0
(©) Fora pump operating on a single liquid stream, the assumption of negligible potential- and kinetic-
energy changes is reasonable, as is the assumption of negligible heat transfer to the surroundings.
Whenee,
(Am =
(@) Fora properly designed gas compressor the result is the same as in Part (¢).
(©) Fora properly designed turbine the result is the same as in Part (c).
() The purpose of a throttle is to reduce the pressure on a flowing stream, One usually assumes
adiabatic operation with negligible potential- and kinetic-energy changes. Since there is no work,
the equation is:
AH=0
(g) The sole purpose of the nozzle is to produce a stream of high velocity. The kinetic-energy change
must therefore be taken into account. However, one usually assumes negligible potential-energy
‘change. Then, for a single stream, adiabatic operation, and no work:
A[(i + x) m] = 0
The usual case is for a negligible inlet velocity. The equation then reduces to:
AH+ lu =o
543
2.21 We reformulate the definition of Reynolds number, with mass flowrate m replacing velocity w
ty = udp =u =D?
p= uD? p
Solution for w gives:
‘Whence, Re
(@) Clearly, an increase in results in an increase in Re.
(6) Clearly, an inetease in D results in a decrease in Re.
2.24 With the tank as control volume, Eqs. (2.25) and (2.29) become:
dm dent)
pa ang Sent)
dt a . dt
Expanding the derivative in the second equation, and eliminating ri by the first equation yields:
+H
Multiply by dr and rearrange:
Substitution of #7’ for AT requires the assumption of uniform (though not constant) conditions through
‘out the tank. This requires the absence of any pressure or temperature gradients in the gas in the tank.
2.32 From the given equation: P
rs oRT
By Fq.(13), [Pav = f por
Whence,
2.38 Recall: aPV)=Pdv+¥aP and = dW = Pav
Whence, = dW =VdP—a(pv) and [Wa fPdP APY)
ByEg.@24), dQ =dU -aw
ByEg.Q.10, U=H-PV and dU =dH~Pd¥—VvaP
With div =—P dV the preceding equation becomes dQ = dH ~ VdP
Whence, [Q=aH-fVaP
54g
umerical Solutions
Chapter 3 - Section B - Non-
3.2 Differentiate Eg. (3.2) with respect to P and Bq. (3.3) with respect to
(Ge), =~95 (5), Ge), +7 (sear) = * ear)
(ir), = 7 (Sr), Gs), & Caran) =P (
Addition of these two equations leads immediately to the given equation.
x
is not covered until Chapter 6.
h(i Ei
sia)
where Vo, A, and B are constants. Application of Eq, (3.3), the definition of x, requires the derivative
of tis equation:
av A, _4P_]_ an
(),-% B+P” (B+PP] B+P
“Multiplication by —1/¥ in accord with Eq, (3.3), followed by substitution for Vo/ ¥ by the Tait equa-
tion leads to:
AB
@+ PBFA API
3.7 (a) For constant T, Eq, (3.4) becomes: * = «dP
3.3 The Tait equation is given as:
Integration from the initial state (P), V;) to an intermediate state (P, V) for constant « gives
¥ 4
ina etP PD)
Whence, V = Moexpl-«(P — A)
‘exp P) expe P,)
If the given equation applies to the process, it must be valid for the initial state; then, A(T) =
FrexpicPi), and
KT) exp(—KP)
(0) Differentiate the preceding equation: dV =~ A(T) exp(-«P)dP
Therefore, wa_f' par
A
=KA(r) [ Pexp(-«P)dP
de,
A(T),
AMP + Dexp-eP) = (Pet Despre PJ]
345
With ¥) = A) exp(—e Pi) and Vy = A(T) exp(—KeP;),_ this becomes:
eae
o |
3.11 Differentiate Eq. (3.34e) with respect to 7:
1=8) pra-nara a? 4 pa-aye AT _ (18) PUPA AP pune aT
ge ae ers area
Algebraic reduction and substitution for d P/dz by the given equation yields:
Tie
PVG
ar
as lo
ye Mrs) +
Foran ideal gas T'p//P = 1/R. This substitution reduces the preceding equation to:
3.12. Example 2.12 shows that U2 =H’ If the gas is ideal,
HaU'S PV
"RT! and Uy—U"= RT!
Forconstant Cy, U;—U" =Cy(Ip- 1’) and Cy(Ty~T") = RT”
Whence,
When Co/Cy is set equal to y, this reduces to
yt
This result indicates that the final temperature is independent of the amount of gas admitted to the
tank, a result strongly conditioned by the assumption of no heat transfer between gas and tank.
3:13 Isobarie case (5 = 0). Here, Eqs. (3.35) and (3.36) reduce to
W =-RT0" = 1) and 2 rahe —1)
Both are indeterminate. The easiest resolution is to write Eq, (3.35) and (3.36) in the altemative but
equivatent forms:
Z @-YRT_(h
) and = By Gi ')
from which we find immediately for = 0 that
“R(h-T) and Q ia -m= cr 1
546
3.44
Isothermal case (5 = 1). Equations (3.35) and (3.36) are both indeterminate of form 0/0, Application
of PHopital’s rule yields the appropriate results:
P,
Wa Ro and
voctinit p(B)
Adiabatic ease (5 = y). In this case simple substitution yields,
2)" ae
Isochoric case (8 = 00). Here, simple substitution yields:
W=0 md @Q An (2-1) = (2-!)-oa-m
~My
] men
A
What is needed here is an equation relating the heat transfer to the quantity of air admitted to the tank
and to its temperature change. For an ideal gas ina tank of total volume V7 at temperature 7,
ay oe Py!
RF aT
my
The quantity of air admitted to the tank is therefore:
ve Pi)
n = w
‘The appropriate energy balance is given by Eq. (2.29), which here becomes:
AAO ant
Got WH = 0
where the prime (') identifies the entrance stream of constant properties, Multiplying by dé and inte-
arating over the time of the process yields:
aU, = m0, — n'H! = O
With n
=ms mW: — H) —m(U — H') = 0
Because Uz = Hz— RT and Ui =H — RT, this becomes:
my — H! ~ RT) >> Cy, then Ts = Tj, If mC =0, then we
recover the isentropic expansion formulas.
3.27 Foran ideal gas, AU =CyaT PV = RT APY) = RAT
Whence,
But
3.28 Since Z = PY/RT the given equation can be writen: Steer
RT
Differentiate atconsiant 7: dV =~ “Tap
6 my
‘The isothermal work is then: W -f pav=rr flap
m sy
Whi =
ence, Wa RTh Compared with Eq, (3.26)
3.29 Solve th 3 ae
.29 Solve the given equation of state for V Pa 60
P Rr
350
Whence,
By definition (Bq. (3.3)}
Differentiate:
By the equation of state, the quantity in parentheses is RT/P; substitution leads to
[@,-F) G-#)
3.31 When multiplied by ¥/RT, Eq. (3.41) becomes:
v aTyV/RT
FV OY +00)
a(D)V {RT
oa THe Fayev Feab
L__arye L
stitute P= sel Sa) a
Substimte P= Ver 255, RT Th eb ayoa teat
1
1+ bp + (bor +--
= bp
Expressed in series form, the first term on the right becomes:
The final fraction of the second term becomes:
fesse Ieee a
1+ (€+0)bp + €a (bp) Soper
—(€ tobe +(e +o)
Combining the last three equations gives, afer reduction:
a. _atry (<+0)a(T)]
Pato GP)ae [PTE
Equation (3.12) may be written: Z= 14 Bp-+ Cp? +--
Comparison shows:
$81
For the Redlich/Kwong equation, the second equation becomes:
bal) _ ary
oF olor )
=P
cCah+ a RF
Values for a(7) and b are found from Eqs. (3.42) and (3.43), with numerical values from Table 3.1
_ 0.08664 RT a(T) _ 0A2748RT.
. Pe RT TP
‘The numerical comparison is an open-ended problem, the scope of which must be decided by the
instructor,
V+ 2C'P+3D'PP +.
eae
roo
Equation (3.12) with V=1/p: Z=1+ Bp -+Cp?+ Dp +
az
336 DitfremtateRa.(.1I (32) =
1,
Whence,
Z
Differentiate: (2) =B+2Cp+3Dp? +
a),
Whence, (2),
8 Jao
3.56 ‘The compressibility factor is related to the measured quantities by
Pvi_ MPY!
2 = RT ~ mT
By Eq. 6.38), Boye = Sober w
dz_dM dP dv! dm dT
(a) By B4.(4), aa ada ae
i 2 MTP TR WF o
Thus Max 52% |Y5M + MS 4PL+ [%eSV"| + 1%5m + N87]
Assuming approximately equal error in the five variables, a ++1% maximum error in Z requires
errors in the variables of <0,2%,
© By Eq. (8),
dbz (ap
BECO, 2 Za(F-4)+ (
‘Therefore
952
Max |% 5B) an |(w6se1+ reerr)
2z-1
FT| (m8 + 5841 + 5m)
For 2 0.9 and for approximately equal error in the five variables, a 1% maximum error in B
requires errors in the variables of less than about 0.02%. This is because the divisor Z— 1 * 0.1
Inthe limit as 2 —> 1, the error in B approaches infinity
3.57 The RedlielvKwong equation has the following equivalent forms, where a and 6 are constants:
Vv +b)
Wile a
Poh RPRO +H
z
From these by differentiation,
aZ\ _ a(V— by — RTP + by
( ) BU De OATES w
a, RTAV BP + bP
(3) a QV + bY — by ~ RTP PY + by? oy
ay, TPP — bY + bY
In addition, we have the mathematical relation
aZ\ _ 2 /aV)r
@), ~ GP/AM 2)
Combining these three equations gives
(2) aV(V — by? ~ bRDPYUV + by
Tr
OP), aRTQV FOV — by PAV + bP heal
For P + 0, ¥ -> co, and Bq. (D) becomes: | lim
(2 b= a/RTP
, RT
For P > 00, ¥ > b, and Bg, (D) becomes:
3.60 (a) Differentiation of Eg, (3.11) gives:
az
4 20% pe aie
(#), +2C'P +3D'PP+ hence Sim,
Ifthe limiting value of the derivative is zero, then B’ = 0, and
(®) For simple fuids, «o = 0, and Eq, (3.59) becomes B° = BP./RT.. If B = 0, then by Bg. (3.61),
0.422
o
= 0.083 - Tr =0
553
1 -(18
ba 0.085
3.X1 Figure 3.3 suggests that isochores (paths of constant volume) are approximately straight lines on a
PT diagram, Show that the following models imply linear isochores:
(a) Constant-. « equation for liquids. (b) Ideal-gas equation. (¢) Van der Waals equation.
SOLUTION
Linear isochores require that (8P/8T)y = Constant,
we
(o byt Gdapietwacosenr ces: (22) 2
w termites rr area (2) 8
o
In each case the quantities om the right are constant, and so therefore is the derivative,
Chapter 4 - Section B - Non-Numerical Solutions
4 For consistency withthe problem statement, we rewrite Eq. (4.8) as:
B c
(Co) = A+ SNH DESH HEED
where
Then
2/T\. Define Cry as the value of Cp evaluated a the arithmetic mean temperature Tx
Chg = A+ Tam) + CTiy
Te+T Tet)
hth
2 2 2
and
where
1
a Patsarty
B Cae
Whence, sie A+ ghee N+ GME +2 +h)
Define & as the difference between the two heat capacities:
Peetl P4204
3 4
This readily reduces to:
Making the substitution r = 73/7) yields the required answer,
46
For consistency with the problem statement, we rewrite Eq, (4,8) as
(Cr)
B D
AtSTetD+a
Zhe D+ ae
where
‘Then
Ta) Ty, Define Cr as the value of Cp evaluated at the arithmetic mean temperature Toy,
D
Chg = A+ BTan +
As in the preceding problem,
Trt aeEeeTe
Fog = EOE et = Beet gee
4
8 4D
Whence, Cr = AF ST +1) $y
2NE+D+ rae ED
Define 6 as the difference between the two heat capacities:
(ict
wr" aeaet
D (z-1)?
Te (eer,
Making the substitution + = 72/7; yields the required answer.
This readily reduces to:
4.8 Except for the noble gases [Fig, (4.1)], Cp increases with increasing T. Therefore, the estimate is
likely to be low.
4.27 (a) When the water formed as the result of combustion is condensed to a liquid produet, the resulting
Jatent-heat release adds to the heat given offas a result of the combustion reaction, thus yielding a
higher heating valuc than the lower heating value obtained when the water is not condensed.
(6) Combustion of methane(g) with H)O(g) as product (LHV):
Cis) + O2(g) + CO2x(g) A Hing = —393,509
2Ha(@) + Onle) > 242018) Aig = Q)(-241,818)
CHa(g) > Cs) + 2H2(g) AH = 74,520
CH(g) + 203(g) > COn(g) + 2H:0(g) | AHS, = 802,625 3 LEV)
Combustion of methane(g) with H,O() as product (HHV):
(CHalg) + 202(g) > COx(g) + 220g)
2H:0(g) > 2H,0(1)
CHa(g) + 20212) > COa{e) +2H200) | A Hig = ~890,649 J (HRV)
(0) Combustion of n-decane() with H2O(g) as product (LH):
10.C(s) + 1002{g) > 10C02(g) AHigyg = (10)(~393,509)
11 Hx(g) + 5409(@) > 11 H,0(@) Hig = (11Y(-241,818)
CioHa2() + 100(5)+ 11H2@) A HSgg = 249,700
6,345,388 J (LHV)
CoHian(h) + 15403(@) > 10COx(g)+ 11 HO) | AHiy
Combustion of n-decane(?) with H0(0 as produet (HAV):
CioHaa(0) + 15L02(g) > 10 COs(g) + 11 HeO(e) AMq = ~6,345,388
11 HhO(e) > 1100) (1)-44012)
CioHtaa( + 15}0:(¢) > 100%) +1100 [AMS 520 1 (HEV)
556
Sal
56
SAL
Chapter 5 - Section B - Non-Numerical Solutions
Shown to the right isa PV diagram with two adi-
abatie Hines | > 2 and 2 — 3, assumed to inter-
sect at point 2. A cycle is formed by an isothermal
line from 3 —> 1. An engine traversing this cycle p
would produce work. For the eycle AU = 0, and
therefore by the first law, O + 1 = 0. Since
W is negative, Q must be postive, indicating that
heat is absorbed by the system. The net result
is therefore a complete conversion of heat taken
in by a cyclic process into work, in violation of
Statement 1a of the second law (Pg. 156). The
assumption of intersecting adiabatic ins is there-
fore false.
Imtersecting
adiabatics|
‘The energy balance for the over-all process is written: Q— AU" + AEx + ABp
Assuming the egg is not scrambled in the process, its intemal-energy change after it returns to its initial
temperature is zero. So too is its change in kinetic energy. ‘The potential-energy change, however, is
negative, and by the preceding equation, so is Q. Thus heat is transferred to the surroundings.
The total entropy change of the process is: ASioai = AS' + AS!
Just as AU’ for the egg is zero, so is AS". Therefore,
Qesuce
AS = BShag = =
Since Q is negative, A Siu: is positive, and the process is irreversible.
By Eq, (5.8) the themmal efficiency ofa Camot engine is:
re : one eae
Differentiate: (ino _OM
Since To/Ti is ess unity, the efficiency changes more rapidly with Te than with Ty. So in theory itis
more effective to decrease Te. In practice, however, fc is fixed by the environment, and is not subject,
to control. The practical way to inctease m is to increase Ty. Of course, there are limits to this to.
For an ideal gas with constant heat capacities, and for the changes T, > Ts and P, > Ps, Eq, (5.14)
can be rewritten as a e
AS=Crin(2) — rin(2
c »(2) en(2)
(if PR =P, AS; =crin (2) Ir h=h,
i
h h ty
Whence, Asy =Cpln(2)—rin( 2) =cym(2
a nF) -en(F) = "(7)
Since Cp > Cy, this demonstrates that ASp > AS,.
a (Pt x
Of B=T, ASp=—Rb ( ') If he toe
Whence, AS =Cpin(2) ~ rin (®) = 0, n(2
: a (3) : (2) non
‘This demonstrates that the signs for ASp snd A Sy are opposite
5.12 Start with the equation at the top of Page 167:
as CK aT CHaT dP
RoR TR Te
Foran ideal gas PY = RT, and InP+InV¥ =InR+InT. Therefore,
aya dry ap ar av
i Pee i ape v
Whence,
Because (Cif/R)—1=CHE/R, this reduces to
car
char y
Raritan
AS plc#ar | ¥
tnegtin yi sf $n)
As an additional part of the problem, one could ask for the following proof, valid for constant heat
capacities. Return to the original equation and substitute dT/T —dP/P + d¥/¥:
43 _ char, chav ap_ citar , car
y Yeresey Tau steevaty Jeet Aetete) JeentedSeet diatads Saat 2
Integration yields
13 As indicated in the problem statement the
basic differential equations are:
dW —dQy ~dQc (4)
Qn Te
0c 7 Te (8)
where Qc and Oy refer to the reservoirs.
558.
(@ With dQx=CydTy and dQc = ChdTc, Eq, (B) becomes:
CydTy TH
CedTe Te
Whence, din Te = Wd In Ty
Integration from Ty, and To, to Ty and Te yields:
To _ (tu) * Hee ere
m= Gg) [e=m(H)
©) With dQy—ChdTy and dQ = ChaTo, Eq. (A) becomes:
AW = CydTy + Ch Te
Integration yields: W = Ci(Tn ~ Tag) + Co(Te ~ Tey)
Eliminate T- by the boxed equation of Part (a) and reatrange slightly:
~
W = Cit (FE -1) + 08% [(%) : |
(©) For infinite time, Ty = Te = T, and the boxed equation of Part (a) becomes:
ron (i)-a((9)
From which: TH 2 Tes (Tig)*
T= (Teg (T yee se Tes MOD (Ty OVA
Because ¥W/(W + 1) — 1 =—1/( + 1), then:
yr ae
Because Ty = 7, substitution of these quantities inthe boxed equation of Part (6) yi
ae [(@)"" ‘| +C6Tey iq” = ]
las:
w
5.14 As indicated in the problem statement the TH
basic differential equations are:
aW —dQy~dQc=0 (4) aw
4Qu Tn
dQc Te i)
where Qc and Qy refer to the reservoirs.
(@) With dOc =CedTe, Eq. (B) becomes
dQn __™
ChaTe ~~ Te
Substitute for dQ, and dQc in Eq. (4):
or
aw
Integrate from To, to Te:
Te Te Zl
We=—CLTyIn = + CL(To To.) oF w= C6 (twin + r0~ 1)
Tey Te
(6) For infinite time, Te = Ty, and the boxed equation above becomes:
rR
w= c¢ (ruin 2 + Ty ~ Be)
Tr
5.15 Write Eqs. (5.8) and (5.1) in rate form and combine to eliminate | |
Te VA ig
Fair Top TW 1Gl where r=
With 1Ocl = kA(Te}! = kA(r Tu)", this becomes:
A(Tyy* ual
kA(Tyys of A=
APh(Tyy A [aes
Differentiate, noting that the quantity in squate brackets is constant:
df Ww 3 1 _ | ar —3
a | kaa [Sr wl- Kt | La=rPA
Equating this equation to zero, leads immediately to: 4r =3 or 75
5.20 Because W = 0, Bg. (2.3) here becomes
= AU! =mCyar
‘A necessary condition for AT to be zero when Q is non-2er0 is that m = 90. This isthe reason thet
natural bodies (air and water) that serve as heat reservoirs must be massive (oceans) oF continually
renewed (rivers)
5.22 An appropriate energy balance here is: Q = AH' =0
Applied to the process described, with Tas the final temperature, this becomes:
mT + mT
mCrT ~T)) +mCp(T — Ts) =0 whence
my +m
w
msm, T= i+ ty/2
560
3.23
5.24
531
‘The total entropy change as a result of temperature changes of the two masses of water:
r r
= myCrln = +m Crin 2
AS =mCeln ge +mCeln ®
Equations (1) and (2) represent the generat case. If m= mz =m
. r
AS = mOpn=e or A
Because T= (T+ 13)/2> VTS, AS" is positive
Isentropie processes are not necessarily reversible and adiabatic. The term isentropic denotes a pro-
cess for which the system does not change in entropy. There are two causes for entropy changes in a
system: The process may be internally irreversible, causing the entropy to increase; heat may be trans:
ferred between system amd surroundings, causing the entropy of the system to increase or decrease.
For processes that arc intemally irreversible, itis possible for heat to be transferred out of the system.
in an amount such that the entropy changes from the two causes exactly compensate each other. One
can imagine irreversible processes for which the state of the system is the same at the end as at the
beginning of the process. The process is then necessarily isentropic, but neither reversible nor adia-
batic. More generally, the system conditions may change in such a way that entropy changes resulting
from temperature and pressure changes compensate each other. Such a process is isentropic, but not
necessarily reversible. Expansion of gas in a piston/cylinder arrangement is @ case in point. It may be
reversible and adiabatic, and hence isentropic, But the same change of state may be irreversible with
heat tranafer tothe surroundings. The process is still isentropic, but neither reversible tor adiabatic
‘An isentropic process must be either reversible and adiabatic or irreversible and non-adiabatic.
GCedt [Cod
T-% h-T
By inspection, one sees that for both T > T) and 7; > T the numerators and denominators of the
above fractions have the same sign. Thus, for both eases (Cp), is positive.
tod
Coe SRO
WTR ~ Inche/TY
By definition, (Coly
larly, (Cols
By inspection, one sees that for both T > Ty and Zo > T the numerators and denominators of the
above fractions have the same sign, Thus, for both cases (Cp), is positive.
When T = Tp, both the numerators and denominators of the above fractions become zero, ard the
fractions are indeterminate. Application of ! Hopital’s rule leads to the result: (Cp)yy = (Cp)s = Cp.
The process involves three heat reservoirs: the house, a heat sink; the furnace, a heat source; and the
surroundings, « heat source. Notation is as follows:
|Q| Heat transfer t the house at ermperature T
\Qr| Heat transfer from the furnace at Ts
1Q.| Heat ransfer from the surroundings at T,
‘The first and second laws provide the two equations:
| = |Or| + 1Qah and. eae
361
‘Combine these equations to eliminate |Q,1, and solve for |r!
~ jo ( Pom) Te
\ori=10(2—= %)
With T=295K Tr = 810K T, = 265K and |Qj = 1000 k}
The result is: 1Qe| = 151.1410
Shown to the right is a scheme designed to ac-
complish this result. A Carnot heat engine op-
erates with the furnace as heat source and the
house as heat sink. The work produced by the en- Tori
sine drives a Carnot reftigerator (reverse Carnot
engine) which extracts heat from the surround-
ings and discharges heat to the house. Thus the
hreat rejected by the Carnot engine (|Q1]) and by
the Camot refrigerator (|Q21) together provide the
heat |Q| for the house, The energy balances for
the engine and refrigerator are
Wiegiae = |e! — 1Qrl
\hetis = 102] ~ 1x1
Equation (5.7) may be applied to both the engine 1el
sand the refrigerator: SURROUNDINGS @ T,
Wel te It _
FURNACE @ Tr
ial 7 1a 7
Combine the two pairs of equations:
Whom = 108% — )
Since these two quantities are equal,
Tr- tr
10x17 = 10a or Oa = 10a
Because the total heat transferred to the house is |Q| = 1011 + 1Qal,
a Te-T Te-T\ _ 9 Te-Te
|Ql = lal + Onl on (1+ Fe) 0
r T (Te
But 101 = |Orlzz whence 10l= one (2)
Solution for [Or yields the same equation obtained more easily by direct application of the two laws,
of thermodynamies to the overall result ofthe process.
8.32 The process involves three heat reservoirs: the house, @ heat source; the tank, a heat source; and the
surroundings, a heat sink. Notation is as follows:
$62
|Q Heat transfer from the tank at temperat
|Q'| Heat transfer from the house at 7”
[Q| Heat transfer to the surroundings at 7,
‘The first and sevond laws provide the two equations:
11+ |O1= 106 and
‘Combine these equations to eliminate |Qq|, and solve for |Q:
With = T=448.15K 7 306.15 K
2w7IsK =,
The result is: 1Q| = 143.38 ks
ture T
Izl_ 19 121 _y
and |Q"| = 1500 83
TANK@T |
‘Shown tothe right is a scheme designed to accom-
plish this result. A Camot heat engine operates
with the tank as heat source and the surroundings
as heat sink. The work produced by the engine
drives a Camot reftigerator (reverse Camot en
gine) which extracts heat |Q'| from the house and
discharges heat to the surroundings, The energy
191
balances for the engine and refrigerator are: SURROUNDINGS @ T,
(Fleagine = 12| — 1Qe,1
[Whesis = |Qx| ~ 10'1
Equation (5.7) may be applied to both the engine
and the refrigerator:
Woy _ Te IQal _ To
le!
lel T oi 7
HOUSE @ T'
Combine the two pairs of equations:
(Weexe = 101(1 #)-10*
Since these two quantities are equal,
10-5 ® ~oiB5* or 12.-101(
T
537
$40
where Q; is here redefined to refer to the system rather than to the surroundings. Nevertheless, the sec=
cond term accounts forthe entropy changes of the surroundings, and can be written simply as dSty,/d:
a(mS)ev — Shae ASy Sher _ g
ar ninaaat a Sar detasl tee
Multiplication by d end integration aver finite time yields
ASS, + MS, = 0 or AS = 0
The general equation applicable here is Eq, (5.22):
(@) Fora single stream flowing within the pipe and with a single heat source in the surroundings, this
becomes
(asi 2s, 20
z
(0) The equation is hete writen fortwo steams (I and I) flowing in two pipes. Heat tans is
internal, beloeon the two sueams, making Q — 0. Ths,
(Sun + (AS)urin = Se = 0
(©) For a pump operatiing on a single stream and with the assumption of negligible heat transfer to
the surroundings
(AS = So 20
(@) For an adiabatic gas compressor the result is the same as for Part (c).
(©) For an adiabatic turbine the result is the same as for Part (c).
(D For an adiabatic throttle valve the result is the same as for Patt (c)
(g) Foran adiabatic nozzle the result is the same as for Part (c)
‘The figure on the left betow indicates the direct, irreversible transfer of heat |Q| from a reservoir at 7;
(o a reservoir at 7;. ‘The figure on the right depicts a completely reversible process to accomplish the
same changes in the heat reservoirs at T; and T:
ni R
Hl lal
564
The entropy generation for the direct heat-transfer process is:
80 ~101(3 - 7) -101(452)
For the completely reversible process the net work produced is Wise
(2%
N-h
ant = Wh Hi} Te101(S SB)
This is the work that is lost, Wig, in the direct, irreversible transfer of heat |. Therefore,
nat
wim o\(2EB) ant ins
plas
Rh
Wea = To| Ql TeSc
Note that a Carnot engine operating between 7; and 7; would not give the correct Wigeat Of ios.
because the heat it transfers to the reservoir at 73 is not Q.
5.45 Equation (5.14) can be written for both the reversible and irreversible processes:
bead ar ie Tw aT ee
eS it yh =f cen?
asmn= [FS ng AS f c#Z -0t
By difference, with AS = 0 ce -
Since A Sipe, must be greater than Zero, Tiney must be greater than Try
565
Chapter 6 - Section B - Non-Numerical Solutions
6.1 By Eq. (6.8), and isobars have positive slope
ite terse satis (22) = (22)
coninwinreccin: —[( Cates tee sae ces
wo
6.2 (a) Application of Eq, (6.12) to Eq. (6.20) yields:
aCe) _ fav —TEr/ar»)
oP), | ar E
. (22),-(0),-108), Gr
Whence,
For an ideal gas’ and (Fe =0
>
(2) Equations (6.21) and (6.33) are both general expressions for dS, and for a given change of state
both must give the same value of dS. They may therefore be equated to yield:
(ar a),
iB) [P
reviswamane — [er=erar(22) (2)
Hoy ma (22) 8
By Eqs. (3.2) and (6.34): (#) =» and (3), .
Contin watered sauton: [cy 6r =ar7 (2)
6.3 By the definition of H, U = H — PV. Differentiate:
au) _ (aH ar
Gr), @), °(% ete
366
Substitute forthe final derivative by Eq. (3.2), the definition of B:
ar.
(22) -ce-arv
Divide Eq. (6.32) by a7 and restrict to constant P. The immediate result is:
OO A®,
Solve for the two derivatives by Eqs. (6.34) ond (3.2); substitution gives:
6.4 (a) In general,
By the equation of state,
tees seesEaCE
(F), Crt Leer a |
aaa
“¥=6b
Substituting this derivative into Eq. (6.32) yields dU = Cy dT, indicating that U = f(T) only,
(6) From the definition of H,
From the equation of state,
dH =dU +a(PP)
RdT + bdP
dPV)
Combining these two equations and the definition of part (a) gives:
dH = Crd? + RaT + bdP = (Cy + RAT + baP
‘Then,
(#
ar),
=Cy4R
By definition, this derivative is C'p. Therefore Cp = Cy +R. Given that Cy is constant, then
80 is Cp and so is
CriCv,
(0) For a mechanically reversible adiabatic process, dU = dW. Whence, by the equation of state,
CydT =—PdV =
But from part (b), R/C)
av
T
dinT =~(y —I)dinly ~ 6)
From which:
aT.
rae
Rm
Bday ~ 6)
(Cr —Cr/Cr
or
Then
dint +diny ~ oy! =0
TY — by = const
Substitution for 7’ by the equation of state gives
PU ~ 6) ~ by
R
= const.
367
or
68 It follows immediately from Eq. (6.10) that:
ac
v¥=(— and
Gr),
Differentation of the given equation of state yields:
RT arr)
pet TO) ainp
v P and Ss ar
Once V and (as well as G) are known, we can ppl the equations
H=G+Ts and U=H-PV=H-RT
‘These become
" anny eae ar(T) a
H=r TT and u=T(7) TF RT
By Eqs. (2.16) and (2.20),
aw
ud pee
c= (5),
Because Fin a faneton of temperature only, these become
er é
cu oe
‘The equation for ¥’ gives the ideal-gas value. The equations for A and U’ show these properties to be
functions of 7 only, which conforms to ideal-gas behavior, ‘The equation for $ shows its relation to
P to be that of an ideal gas. The equations for Cp and Cy show these properties to be functions af T
only, which conforms to ideal-gas behavior, as does the result, Cp = Cy + R, We conclude that the
given equation of state is consistent with the model of ideal-gas behavior.
66 It follows immediately from Eq. (6.10) that:
aG
”- (5), _
Differentation of the given equation of state yields:
aG
at),
K and S=
Once V and $ (as well as G) are known, we can apply the equations:
H=G4+TS and U=H-PVY=H-PK
These become:
a :
eo) and v= Fr)- TE
Ha F(T) +KP—
(y+ KP 1 i-
By Bgs. (2.16) and (2.20),
co=(#8) at
568,
Because F isa function of temperature only, these became:
@P
C= and
‘The equation for V shows it to be constant, independent of both T and P. This is the definition of
‘an incompressible fluid. H is seen to be a function of both T and P, whereas U, S, Cp, and Cy are
functions of T only, We also have the result that Cp = Cy. All of this is consistent with the model of
an incompressible uid, as discussed in Ex. 6.2.
6.11 Results for this problem are given in the text on page 213 by Eqs. (6.59) and (6.60) for G® and #7
Bq, (6.45), page 206, then yields 5
6.12 Parameter values for the van der Waals equation are given by the first line of Table 3.1, page 99. At
the bottom of page 214, itis shown that J = f/Z. Equation (6.63b) therefore becomes:
GF
RT
a6
Zz
= 1-1 =p) —
For given T and P, Z is found by solution of Eq, (3.49) for a vapor phase or Eq. (3.53) for a liquid
phase with o = =0. Equations (3.50) and (3.51) for the van der Waals equation are:
Be 2
E ond -
With appropriate substitutions, Eqs. (6.64) and (6.65) become:
He 9B
RT fas Zz
7
and | = mz—A)
{6.13 This equation does not fall within the compass ofthe generic cubie, Eq. (3.41): so we start anew. Fist,
‘multiply the given equation of state by /RT:
Substitute: z
‘Then,
z
Because p = P/ZRT,
Given T and P, these two equations may be solved iteratively for Z and &
Because 4 is a constant, Eys. (6.57) and (6.58) may be rewritten as:
569
Gt
S [@-vE+2-1-mz @)
HR _ ft (az) ag
wo f (BB+ ©
In these equations, Z is given by Eg, (4), from which is also obtained:
az
Inz=—mn1—g)—9& and (3) 7 nes exp(-48)
I
The integrals in Eqs. (B) and (C)) must be evaluated through the exponential integral, E(x), @ special
function whose values are tabulated in handbooks and are also found from such software packages as
MAPLE®. The necessary equations, as found from MAPLE®, are:
5 dl
fe nF = ep-atet-et—91- 2-9) £48 Ines) — 7
where y is Buler’s constant, equal ro 0.57721566.
" az) st
and at) (FZ) F=aecaei-gd—p-B-9}
har)
Once values for G2/RT' and H®/RT are known, values for $*/R come from Eq. (6.45). The diffi-
euler of integration here are one reason that cube equation have found pester vor.
6.18 Assume the validity for purposes of interpolation of Fg. (6.70), and write it for Ts, and 7)
mre aa 2 (a)
inp aa ; (8)
inp =a ©
samaicrimay nla (2
samen —wher(L 1) asf =1
‘The ratio of these two equations, upon rearrangement, yields the required result
6.19 Write Eq, (6.70) in logyg form log P= A “
Apply at the critical point log P. =
(8)
i
sie
570
By difference, log P= B (; eS 7) -B (@ a *) ©
Whence,
Equation (C) then becomes
fee G *) (
Apply at T; = 0.7: IogtP) 5
By Ea. 0.49), = -1.0— log?) e027
Whence,
(fa)men=
6.83 The slopes of isobars and isochores on a T'S diagram are given by Eqs. (6.17) and (6.30):
ar) _ : ar) _ 7
B5)_~ Ce a 35 )y Sy
Both slopes are necessarily positive, With Cp > Cy. isochores are steeper.
An expression for the curvature of isobars results ftom differentiation of the first equation above:
(z 21 (a) 7 (ace) 7 _ 1 (aCe) (aT) _ Py, 7 (aCe
as), Cre\aS), CRIS} 3 Var ),\as),~ CRU” Ce Var J.
acer __or a
aT), ae BT ator
win craaser, (35) =m and 1-£(
Because this quantity is positive, so then isthe curvature ofan isobar.
6.84 Division of Bq. (6.8) by dS and restriction to constant yields:
an a
(22) <7+7(%2),—aweavas,
@
as),
Therefore,
sm
Also,
29)(3) 80) G8)
Whence,
By Fas. (3.2) and (3.37)
ay
(F), and
av) _R dB
wi a) 54S ani
hence, (3), Bt id
Differentiation of the second preceding equation yields:
ap R R dB\ 1 (av
(B)--s- GB) AG)
av) __Rr
From the equation of state, ( 3), --2
®)__ Rk BRT OR
Whence, (35), <- yet he Ret)
Clearly, the signs of quantity (BT — 1) and the derivative on the left are the same. The sign is deter-
mined from the relation of f and ¥ to B and dB /dT’
1 (R, 4B >
pr -F(5+F)-1=
In this equation dB /dT is positive and B is negative, Because RT/P is greater than |B, the quantity
BT ~ 1 is positive. This makes the derivative in the first boxed equation positive, and the second
derivative in the second boxed equation negative.
Since a reduced temperature of 7; = 2.7 is well above “normal” temperatures for most gases, we
expect on the basis of Fig. 3.11 that B is (—) and that dB/dT is (+). Moreover, d?B/dT? is (~).
By Eqs. (6.53) and(6.55), G®=BP and. S*# = —P(dB/dT)
Whence, both G* and $? are (). From the definition of G8, H® =G"+TSS, and H¥is(-)
By Eqs.337) and (640, Y= B, and V*is(-).
‘Combine the equations above for G*, 8, and H*:
ata P(a—1 2) whence, (3 )
ar. anes
Therefore, C2 = ( es
*). is(4). (See Fig. 65.)
Non-Numerical Solutions
Chapter 7 - Section B
7.2 (a) Apply the general equation given in the footnote on page 260 to the particular derivative of interest
here:
ar (2 as.
oP}, a5), \oP J,
The two partial derivatives on the right are found from Eqs. (6.17) and (6.16); thus,
(2) Tr (2)
For gases, this derivative is positive. It applies to reversible adiabatic expansions and compressions
in turbines and compressors.
(6) Application of the same general relation (page 260) yields:
(5), Go), Gr),
‘The two partial derivatives on the right are found from Eqs. (2.16) and (6.31); thus,
(or), =e-[? *(),]
For gases, this may be positive or negative, depending on conditions. Note that itis zero for an
ideal gas. It applies directly to the Joule expansion, an adiabatic expansion of gas confined in a
portion ofa container to fill the entire container.
7.3. The equation giving the thermodynamic sound speed appears in the middle of page 250. At written,
it implicitly requires that V represent specific volume. This is easily confirmed by a dimensional
analysis. If V is to be molar volume, then the right side must be divided by molar mass;
v? (ap
¢=-x la), ‘a
Applying the equation given in the footnote on page 260 to the derivative yields:
Gr), ~~ (3s), (3),
(), 1G), GG). (1G), 1G), @),]
Division of Eg. (6.17) by Eq, (6.30) shows that the first product in square brackets on the far tight is,
the ratio Cp/Cy. Reference again to the equation of the footnote on page 260 shows that the second
product i square brackets on the far right is ~(@P/@V)r, which is given by Eq. (3.3).
‘Therefore, 3) -2 (3) ce (3)
Is Cy NAV Jy Cy NeW.
W,
573
vCp
~ MCye
Substitute into Eq, (4)
(a) For an ideal gas, Y = RT/P and x = 1/P. Therefore,
@®) For an incompressible liquid, ¥ is constamt, and x = 0, leading to the result: ¢ = oo. This of
course Icads to the conclusion thatthe sound speed in liquids is much greater than in pases.
16
1
1 : :
CCritical-pressure
ratio
Po/Py
‘As Pr decreases fom an initial value of P; = Pi, both up and sr steadily increase until the critical~
pressure ratio is reached. At this value of Pp, zp equals the speed of sound in the gas, and further
reduetion in Pz does not affect 2 oF
7.7 The mass-flow rate rn is of course constant throughout the nozzle from entrance to exit,
‘The velocity u rises monotonically from nozzle entrance (P/P; = 1) to nozzle exit as P and P/P,
decrease.
‘The area ratio decreases from 4/4; = 1 at the nozzle entrance to & minimum value at the throat and
thereafter increases to the nozzle exit,
7.8 Substitution of Eq. (7.12) into (7.11), with m; = 0 gives:
2 ( 2 ) ( 2 )
eeat = 1-——) = van (—
thea = pat) 7h (Sa
and P; isin Pa. The units of ty ae then
where V1 is specific volume in m?-kg™
‘akg m-s
With respect to the final term in the preceding equation, note that Pj V has the units of energy per unit
‘mass, Because I N-m = 1 J, equivalent units are J-kg™!. Moreover, P.¥) = RTi/M; whence
With 2 in units of J-(kg mol)"-K-!, RT;/M has units of F-kg~
S74
7.46 It is shown at the end of Ex, 7.5 that the Joule/Thomson inversion curve is the locus of states for whieh
(8Z/9T)p = 0. We apply the following general equation of differential calculus
),-@).+@),@),
(),= Ga), G(r),
Yn (37), - Ga), @), Ge),
ae P a) Pf -l az
mone paper, pn zhao (8) -2(c2.[eur(%)
Setting (0Z/0T)p = 0 in each of the two preceding equations reduces them to:
(BG), Ronit
(),-- (3), G2), a7), "za F
Combining these two equations yields:
&)
a),
(@) Equation (3.41) with van der Waals parameters becomes:
ara
“V¥=6b FF
P
Multiply through by /R7, substitute Z = PV/RT, V = 1/p, and rearrange:
1 ae
1b RT
In accord with Bq, (3.48). define q = a/bRT. In addition, define § = bp, Then,
Zz 9 (ay
ae
: az ‘az
ee (=),- (GF),
By Eg. (3.43) with @(T,) = | for the van der Waals equation, q
a V(t) ar
nal)
(=),
W/QT,. Whence,
Then,
In addition,
‘Substitute for the two partial derivatives in the boxed equation:
rit _be
Pape a
Whence, @
By Eq, (3.43), Pe = QRT./b. Moreover, P = ZpRT. Division of the second equation by the
first gives B, = ZpbT'/ QT... Whence
zer,
7 ©
These equations allow construction ofa T; vs. P; inversion curve as in Fig. 72. For a given value
of 7,, calculate q. Equation (B) then gives &, Eq, (4) gives Z, and Eq. (C) gives P,.
(©) Proceed exactly as in Part (a), with exactly the same definitions. This leads to a new Eq. (4):
R
eee
CITE Tee a.
By Eq. (3.43) with a(T;) — 7,95 for the Redlich/Kswong equation, ¢ = Wi/S27;!5, This leads to:
dq __1Sq : az) _ _1Sg
ated be at),~ Ta+5
Moreover, (2) ees
op), G-sF + kP
Substitution of the two derivatives into the boxed equation leads to a new Eq, (B)
14ey) 1 )
(4) (are @
‘As in Part (a), for a given 7, calculate q, and solve Eq. (B) for &, by trial ora by @ computer
routine. As bofore, Eq. (4) thon gives Z, and Eq. (C) of Part (@) gives 2,
7.A7 (a) Equal to, (6) Less than. (c) Less than. (a) Equal to. (e) Equal to
7.28 When a saturated liquid is expanded in a turbine some of the Liquid vaporizes. A turbine properly
1, the derivative is negative (try some values). Thus, the
work of compression decreases as C’p increases and as the molecular complexity of the gas increases.
74S. The appropriate energy balance can be written: W = AH —Q. Since Q isnegative (heat transfer is
‘out of the system), the work of non-adiabatic compression is greater than for adiabatic compression.
Note that in onder to have the same change in state of the ai; ie, the same AH, the irreversibilities of
operation would have to be quite different for the two cases.
7.46 There is in fact no cause for concem, a adiabatic compression Sends the steam further into the super-
heat region.
S78
Chapter 8 - Section B - Non-Numerical Solutions
8.12 (a) Because EQ. (8.7) for the efficiency noice includes the expansion ratio, re
this quantity to the compression ratio, r = Vc/ ¥p, and the Diesel cutoff ratio. re
Vo = Vay r= VciVs. Whence,
1 Vg, we relate
141 Vo. Since
r _VelVo _ Ya
2 Ve[Va Vo
Equation (8.7) can therefore be written:
3 [ewe aver) _ Ltn (a
Toes
vb rear yr
| 1") ot
7 | mea =1~ (5) wa
(®) We wish to show that
ease
ae)
end
7-0”
1
I or more simply
Taylor's theorem with remainder, taken tothe Ist derivative, is written
g=all)+2'0)-@-)+R
eT +6@—1)
where, @-1) @ Land x > 1, R > 0. Therefore:
x >14a-(e-1)
bas ve)
(@ Ity = Land = 8, then by Ea. (8.6
yes
-(3) and
yh guy :
ty Sand [vines = 0.4904
(i) caer mt [tome 0
et yey
- (3) Geos [oe = 04007
f-1>a-(e-1)
5647
i
8.15. See the figure below. In the regenerative heat exchanger, the air temperature is raised in step B > B*,
while the air temperature decreases in step D > D*. Heat addition (replacing combustion) isin step
Bac
Bip Wen
ition, ”
a One
where, Was = (He ~ Ha) = Co{T's — Ta)
Wen = (Hp — He) = Cp(Tp — Te)
Oarc = Cp (Te — Ty) = Co(Te ~ Tp)
Ty—Ta+Te~To Tr -Ts
Whence, peeesstDy ty a
Te~To Te
Tp
By Eq. (3.296),
mi nan (2) =n (2)
‘Then, ql
Multiplication of numerator and denominator by (Ps/P.)("~” gives:
el)
580
Chapter 9 - Section B - Non-Numerical Solutions
9.1 Since the object of doing work |H’| on a heat pump is to transfer heat [Qy7/ to a heat sink, then:
What you get = |u|
‘What you pay for = {77
1Qu!
Whence @ = 20
Wi
For a Camot heat pump,
Qa Ta
°
Iai 1Oci~ Tw Te
9.3 Because the temperature of the finite cold reservoir (contents of the relfigerator) is @ variable, use
differential forms of Carnot’s equations, Eqs. (5.7) and (5.8):
eae and awe
4c Te
In these equations Oc and Oy refer to the reservoirs. With dn = Cl dTo, the first of Carot’s
equations becomes:
)
E)a0n
Ty be
ae
4g, are
i Te
Combine this equation with the second of Camot’s equations:
~CTy
ae
aW = -C'Ty +0 dTe
Te
Integration from To=Ty to Te = Te yields:
tee eee tate
W = Cty WE + Ce Ty) 8 ct (1 B+k |
9.5 Differentiation of Eq, (9.3) yields
Be Te
buy aeiacirgt
(=) i (in). Ty TP
Because Ty > Tc, the more effective provedure is to increase Te.
For a real refrigeration system, increasing Te is hardly an option if refrigeration is equired at a patic-
ular Value of Tc. Decreasing Ty is no more realistic, because for all practical purposes, Ty is fixed by
environmental conditions, and not subject to conto
Ty Te OF
9.6 Fora Camot refrigerator, a is given by Bq. (9.3). Write this equation for the two cases:
Te
e and
Tw -Te
Because the directions of heat transfer require that Tyr > Tay and Tc < Tyo, a comparison shows that
© < @, and therefore that « is the more conservative value,
58
Chapter 10 - Section B - Non-Numerical Solutions
10.5 For a binary system, the next equation following Eq. (10.2) shows that P is linear in x}. Thus no
‘maximum or minimum can exist in this relation. Since such an extremum is required for the existence
‘of an azeotrope, no azeotrope is possible.
10.6 (a) Because benzene and toluene are chemically simitar and the pressure is only l(atm), this system
can be modeled by Raoult’s law to a good approximation,
(®) Although n-hexane and n-heptane are chemically similar, a pressure of 25 bar is too high for
‘modeling this system by Raoult’s law.
(©) At 200 K, hydrogen is supercritical, and modeling the hydrogen/propane system at this temper
ture by Raoult’s law is out of the question, because no value of P “for hydrogen is known.
{d) Because isooctane and n-vetane are chemically similar and at a temperature (373.15 K) close 10
their normal boiling points, this system can be modeled by Raoult’s law to a good approximation,
(e) Water and n-decane are much too dissimilar to be modeled by Raoult’s law, and are in fact only
slightly soluble in one another at 300 K.
10.12 For a total volume of an idesl gas, PI = RT. Multiply both sides by y;, the mole fraction of
species jin the mixture:
mm;
LRT
mM
yPVi =m RT or p=
where m; is the mass of species /, M, is its molar mass, and p, is its partial pressure, defined as
i =P. Solve for m;
RT
Applied to moist air, considered a binary mixture of sir and water vapor, this gives
ng a MiOPHOK' ag Mucpas¥!
= RT
(a) By definition,
= Tie or ha
(#) Kfairis in equilibrium with liquid water, then the partial pressure of water vapor in the air equals
the vapor pressure ofthe water, and the preceding equation becomes:
Pet
wpe — Mino Fit.
Ma %
582
(c) Percentage humidity and relative humidity are defined as fatlows:
and
A _ pag PRs
—= oe == (100)
Fe PR Pro
Combining these two definitions to eliminate pro gives:
ps
PPR Gra/100)
10.14 Because the vapor space above the liquid phase is nearly pure gas, Eq. (10.4) becomes P = x7
For the same mole fraction of gas dissolved in the liquid phase, P is then proportional to 1. Values
aiven in Table 10.1 indicate that were air used rather than COz, P would be about 44 times greater,
‘much too high a pressure to be practical
10.15 Because Henry’s constant for helium is very high, very little of this gas dissolves in the blood streams.
Of divers at approximately atmospheric pressure.
10.21 By Eq. (10.05) and the given equations for In y; and In y2,
AP = xy exp(txd) Pt and yeP =x explaad) Pg
‘These equations sum to give:
Px exp(and) PS xp exp(axt) Py
Dividing the equation for y; P by the preceding equation yields:
srexplar Pmt
a expCA) PS + mz expla PS
_xz this equation obviously reduces to:
p=
pot
PR
10.23 A little selection should convince anyone that there is no other way that BOTH the liquid-phase and.
vapor-phase mole fractions can sum to unity
10.24 By the definition of a K-value, y; = Kixr and yz = K>x2. Moreover, y; + y2 = 1. These equations
combine to yield:
Kurt Kon = 1 or Kyxy+ Ka(l =) =
i-k
Solve for xy x
Substitute for.x; in the equation yy = Kix
Ki — Ka)
583,
Note that when two phases exist both x, and yy are
dependent of 2
By a material balance on the basis of | mole of feed,
silty
=z or x -Vy+ nV
Substitute for both xy and yy by the equations derived above:
Kil = Ka)
i= ky
Rett
Ven
(Ki
Solve this equation for V Da
Note that the relative amounts of liquid and vapor phases do depend on 21.
584
Chapter 11 - Section B - Non-Numerical Solutions
11.6 Apply Eq, (11.7)
iaecleatet ee) Fa lien"? Cah,
11.7 (a) Let m be the mass of the solution, and define the partial molar mass by:
Let My be the molar mass of species k. Then
m= DmMia=nMit DM; G4i)
i Pr"...
Whence, rie = M, |
(Bane (oom
ee aaa eee
(3st!
i= (Tr), oe
If.M, is the molar mass of species 7,
"TA
Whence,
119 For application of Eg. (11.7) all mole fractions must be eliminated from the given equation by the
relation x; = mf
nM = My + 1nMz + 3Ms +
weet, (ACIDY ea tne 83)
td Peaaas eNO rma,
Because n =m; 4m +, (#) 1
Tr Pasay
= My+ xml] ~ 21
Mrtxsfl—2m}C| and | My = My txyan[1 —225]C
ence, = M+ 2 [1
wi mB
uM,
Similarly,
‘One can readily show that application of Eq, (11.11) regenerates the original equation for Mf. The
infinite dilution values are given by.
Me =M+xmuC kD
Here-x; and xy are mole fractions on an i-free bass.
11.10 With the given equation and the Dalton’s-taw requirement that P = S>) p,, then:
pa yn,
For the mixture, P= ZRT/V. These two equations combine to give Z = Ty y1Zi.
11,11 ‘The general prineple is simple enough
Given equations that represeat partial properties &%,, Sf", or SP as functions of com
position, one may combine them by the summability relation to yield a mixture property
Application ofthe defining (or equivalent) equations for partial properties then regenerates
the given equations if and only ifthe given equations obey the Gibbs/Duhen equation.
11.12 (a) Multiply Eq. (4) of Ex. 11.4 by m (= mj +2) and eliminate xy by xy = m/(n1 + na)
nfl = 600(m, + m2) ~ 180m ~ 207 —
Form the partial derivative of mf with respect to m at constant ny
an ant
cn ao ao
Gy tmyP tm)
00 - 180-20] ]=s20- 00 + 40
Whence, 60x +40x}
Form the partial derivative of mH with respect to nz at constant
or
(8) Im accord with Eq. (11.11),
H = (420 ~ 60.5? + 40.x}) + (1 — 22)(600 +4029)
Whence, H = 600 180x; ~ 20x}
ath
(©) Write Ba. (11.14) fora binary ystem and divide by days PE 4 4 SP
ay
Differentiate the the boxed equations of part (@)
dh ae ath 7
dey PHD} = 120.29 and FT = 12049
‘Multiply each derivative by the appropriate mole fraction and add:
1200382 + 120891 = 0
(@) Substitute x, = 1 and x, = 0 in the first derivative expression of part (c) and substitute xj — 0
in the second derivative expression of part (c). The results are:
(Si), (@),
ath
©
11.13 (@) Substitute x2 = 1 — x; in the given equation for V and reduce:
:
Apply Eqs. (11.15) and (11.16) to find expressions for Fs and Vs. First,
av
O = 58-Im, — 2a?
dx; se
Then, 2oxt tax} | and +x |
587
(b) In accord with Eq. (11.11),
rp
1128 — 2x1 — 2047 + 4x9) + 1 —41)(00 4x3 + 14x)
10
Whence, ¥ 8.41
which is the first equation developed in part (a),
(6) Write Bq, (11.14) for a binary system and divide by dy
Differentiate the the boxed equations of part (a):
27) gage + 4rxt and Oe Max +4237
dx
“Multiply each derivative by the appropriate mole fraction and add
xi(-2 = 40x, +427) + (1 —11)(2x) +427) =0
TThe validity of this equation is readily confirmed.
(@) Substitute x; — 1 in the first derivative expression of part (c) and substitute x; = 0 in the second
derivative expression of part (c). The results are:
©
UL14 By Eqs. (11.15) and (11.16)
aH 7 aH
and Th = Hay
f= H+nZ
588,
Given that: H = x(a) + bx) + xa(aq + baa)
Then, after simplification, a“ 1a) + 2bix1 ~ (az + 2bax2)
Combining these equations gives after reduction:
Fi, =a, + bi tx2eibi—x2ba) and fh = a2 + baz
x1 (ejb1 mba)
These clearly are not the same as the suggested expressions, which are therefore not correct. Note.
that application of the summability equation to the derived partial-property expressions reproduces
the original equation for #7. Note further that differentiation of these same expressions yields results
that satisfy the Gibbs/Duhem equation, Eq. (11.14), written:
diy
“e
The suggested expresions do not obey this equation, further evidence that they cannot be valid.
11.15 Apply the following general equation of differential calculus:
(3)-@)+@),@).
ead al ed al
Bm dram Ltr den, L OF doy KOM) 2m,
(tM) (av aM) (av
+057), Seen : "(F), Gee.
By definition,
MY) 2 ( o n() anor
( Peg aes Tnem . Inte
Therefore,
oy (aM
fnew Pa)
11.20. Equation (11.55) demonstrates that [n 6, is a partial property with respect to G*/RT. Thus Indy
G,/RT, The partial-property analogs of Eqs. (11.53) and (11.54) are:
ae ‘i ainds AE
RF = |r),
‘The summability and Gibbs/Duhem equations take on the following forms:
ot ‘ 3 #
ap Dvd and Dednd (const 7, >|
589
11.26 For a pressure low enough that Z and In are given approximately by Bs. (3.37) and (11.35):
BP BP
Zal+e and ng= ap
then Ing© 2-1
11.28 (a) Because Bq. (11.92) shows that In y is a partial property with respect to G#/RT, Egs, (11.15)
and (11.16) may be written for M = G#/RT:
GE adeG*sRT) GE dG*RT)
y= gRte nn ap 7 ae
Substitute xp = 1 —x1 in the given equaiton for G#/RT and reduce:
7 7 aGRT.
ge 18x: +27 +082} whence sein 18425, + 24x?
Thea, fing =—18+2m+14e7—16x7| and [inn =—x? 165
(8) In accord with Eq. (11.11),
ap TA tale = C18 42m + 1.4x? = 1.653) + (1 —)(-2? - 1.623)
Whence, Fe
18x) +47 +0.8x}
‘which isthe fist equation developed in part (a)
(©) Write Bg, (11.14) fora binary system with
diny | dinys
Iny; and divide by dx):
eCaanat uaal
Differentiate the the boxed equations of part (a)
ain, dinys >
Te = 24+28x1—4.8xF and ae, 2x) — 48x}
Multiply each derivative by the appropriate mole fraction and add:
21 +28.) 4.847) + = 21-21 = 4.8.47)
‘The validity of this equation is readily confirmed,
(d) Substitute x1 = 1 in the first derivative expression of part (c) and substitute x; = 0 in the second
derivative expression of part (c). The results ere:
e
04 06
xy
11.29 Combine definitions of the activity coefficient and the fugacity coefficients:
xP. &
n
Hie
Note: See Eq. (14.54),
11.30 For C£ = const, the following equations are readily developed from those given ia the last cotueaa
of Table 11.1 (page 404):
=
‘Working equations are then
HE-G
a
se and
HE
For Ty = 298.15, Tr = 328.15, (T) = 313.15 and AT = 30, results forall parts of the problem are
given in the following table:
HE +CBar and Gf = Hf ~ 1 SF
W.For CE
S$ uf oof
GF oF Sf ok oF
=622 1920 4354 42
109815951677 3.3
407-984 «1.935, -2.7
“sosi 1794 1974 | 4384 1020. 4914
1995 14oh 10399 Ler? 159510047
err (903 ‘3s28} 1935984348
@| 632 -208 -2817 23.0) -0.614 482 683.5) -2817 -208 716.5
(3 | 144s as “817 tol nt96 93818137 | "2817 40s isons
| Ts 416 —3as7 t10|-2503 “85 ‘a339| "3057-6 ‘gua
@| 759 1465 2.368 -8.0| 1.602 1225 699.5 2.368 1465 688.0
@
®
o
591
11.31 (a) Multiply the given equation by m (=) + m2), and convert remaining mole fractions to ratios of
‘mole numbers: ee
nt mom, 4 mits |, mans
Be An A dy
RT a n ”
Differentiation with respect to m; in accord with Bq, (11.92) yields [(8n/11)u,.n) = U
= Agm(1 ~ a1) + Aig 1) = Avex
Similarly, Iny2 = Arr (1 = 23) — Aiea + dagen ~ 2)
In yy = Ayana + Apsei (1 — a5) + Anvxa( — 3x3)
(0) Bach In y; is multiplied by x,, and the terms are summed. Consider the first ‘erms om the right of
‘each expression for In ,. Multiplying each of these terms by the appropriate x, and adding gives:
AaQs¥2 ~ x82 4X20) — x}ay — MaeaXs) = Anaxixa(1 1 + 1 — x2 — 3)
= Apxiaal2 — (ty +2 +33)] = Aamir
An analogous result is obtained for the second and third terms on the right, and adding them
yields the given equation for G2/RT
(©) For infinite ditution of species 1, InA@y = 0) = Arpxe + Arsxs ~ Apseexs
For pure species 1, Inv =1) =0
For infinite ditution of species 2, In yes = 0) = Aisx?
For infinite dilution of species 3, In yr(as = 9) = Aine}
11.38 By Eq. (11.84), written with M = G and with x replaced by y: G® =G¥ — S}yGF
Equations (11.31) and (11.35) together give Gf = BjP. Then for a binary mixture:
GF = BP ~ yBuP — yBnP or GF = P(B— yBu — Ba)
Combine this equation with the last equation on Pg. 394: | G¥ = 812Pyyn
;
Frome tcohamerTite 11 ageso: st = (82)
7
Because 5i2 is a function of T only:
Priya
Again from the last column of Table 11.1: CE (3)
‘This cquation and the preceding one lead directly to:
592
on-Numerical Solutions
Chapter 12 - Section B -
AP
12eives: Pain Pt anh
an dyn )
a PS (ao? -n
Ben)e a (o?? n
12.2 Equation (12.1) may be written: yi?
‘Summing for /
ap
Differentiate at constant 7: = Py (
Apply this equation to the limiting conditions:
dn
For x; =0 m nayr wal an
4
For x1=1 = nay eo
ayy
‘Thea,
ap wtyoc_pet op = (SP
(a),.-rr eG
(Bt of (i)
Sine both "and 7 are always positive definite, it follows that:
nl
pes
12.4 By Eqs. (12.15), Inn=ag and n= As?
‘Therefore, in 2! = AG} — af) = AG —x) = A280)
”
A _yaPS (nln) (PM) _ a,
By Eq. (12.1), BO ynP a GE) (Fs) ee
‘Whence, In(@j27) = ACL — 2x1)
fan azeotrope exists, yz =1 at O is accompanied. by tas
opposite extremum in In 72, Thus the difference ny; ~Iny2_ is also an extremum, and Bq. (12.8)
becomes useful:
: &
iy — ny = nt = MORE
pn de
‘Thus, given an expression for G°/RT = g(x), we locate an extremum through:
@GYRT) _ dinn/n)
det dx
593
For the van Laar equation, write Bq. (12.16), omitting the primes (’):
where As Anxy + dns
Moreoves, and
me dd
Then eae
a@(G°/RT) 2x, dd x
as? a dx;
2andal eo xa 4 im (UA)
ale te ad ran]
2Andn aA aA
24 (4 rntt) (nd 4
‘This equation has a zero value if either 4); or 42) is zero. However, this makes G/RT everywhere
zero, and no extremum is possible. If either quantity in parentheses is zero, substitution for 4 and
dAjdx; reduces the expression to diz = 0 ot 42) = 0, again making G*) RT everywhere zero. We
conclude that no values of the parameters exist that provide for an extrecaure iw bavi /9)
‘The Margules equation is given by Fq, (12.9b), here written:
oe aa v4
Sean wl A= Ags, + An eee a
rT where Aa + Ane Tay = A A ae
aGF/RT) dA
‘Thea, “om Aan) braze
#GYRT) da dA a4
OC = 24 or) S40 A py
ax} lnm angy, tn a0 ge, hee
da aa
= 244g = 2] ry — xy
dea — FE afer 2 4]
‘This equation has a zero value when the quantity in square brackets is zero. Then:
aa
oR
(x2 ~ x1)(Aat — Ain) — Aaixt — Aiaea = Aavta + Avex 2A + AD)
Substituting xp
— x1 and solving for x1 yields:
Ay = 2d 2
34a — Ai) ” *=36=H
594
12.7
When r= 2, x1 =0, and the extrema in In y; and Iny oveur at the left edge of a diagram sucht
as those of Fig, 12.9, For values ofr > 2, the extrema shift to the tight, ceacking a limiting value fe
1 =00 at x1 =1/3, For positive values of the parameters, in all of these cases dai > Ay2, and the
intercepts of the In, curves at xy = 1 are larger than the intercepts of the In yy curves at x1 = 0.
When y= 1/2, x = 1, and the extrema in In y; and In y» occur at the right edge of a diagram
such as those of Fig. 12.9. For values of r < 1/2, the extrema shift to the left, reaching a limiting
value for r = 0 at x1 = 2/3. For positive values of the parameters, in all of these cases day < uz.
and the intercepts of the Iny1 curves at x1 = 0 are larger than the intercepts of the in ys curves at
m=
No extrema exist for values of r between 1/2 and 2.
Equations (11.15) and (11.16) here become:
GE dG*RT) GE dGFRT)
lon= pte amd nya Be
(@) For simplicity of notation, omit the primes that appear on the parameters in Eqs. (12.16) and
(12.17), and write Eq. (12.16) as:
oF ay
pp = Ae aty + Aaa
$F = Avene where Ansty +4:
ze
a aGFRT)
ax,
and tay, =
rare
D— Angry + Anis) = BEE dau, + Aix)
Aux (42 + Ane)?
D ‘Anita
Anx
inn = An(1
neta)
‘The equation for In yz is derived im analogous fashion.
:
©) wnt To Pca, tan = [ACHAT
su 2286 us et
nF _ A
== where aD = Ayn + Ayn
RT
595
Differentiation in accord with the first equation gives:
a 1 _m_ (amp)
2g) = ota
‘The remainder ofthe derivation is the same as in Part (a).
12.16 This behavior requires positive deviations from Raoult’s law over part of the composition range and
negative deviations over the remainder. Thus a plot of G* vs. x starts and ends with GE = 0 at
x1 = Oand.x; = 1 and shows positive values over part of the composition range and negative values
over the remainder, with an intermediate crossing of the x: axis, Because these deviations are usually
‘quite small, the vapor pressures P;*" and P," must not be too different, otherwise the dewpoint and
bubblepoint curves cannot exhibit extrema.
1211 Assume the Margules equation, Eq. (12.9b), applies:
e
Ferd + Ann) — and Zequimola) = bain + An)
em anda + Ann Fteauimolar) = Edin + Aa
But [see page 424, just below Bq. (12.106): Ae
ly? Any = ny
& equimolar) = Hin y +t SF equimota = 1
SF tequimolan = Ftiny® sing) or |S equimotr
HY)
xX}(0.273 + 0.096 x1) + x2¥9(0.273 ~ 0.0962)
¥1x2(0.273.¥2 + 0.096.522 + 0.273 x1 — 0.096122)
= x120.273)0) +2)
Ge
Fp tran
(6) The preceding equation is of the form from which Eqs. (12.15) are derived. From these,
Inn = 0.27383 and nye = 027337
(©) The equations of part (6) are not the reported expressions, which therefore cannot be correct. See
Problem 11.11
12.28 Write Bg. (11.96) for a binary system, and divide through by «xy
din din in
dete 0 whence = AA __ dian _ din
_ a dx, m2 dx; 2 dr
596
x
Integrate, recalling that In yp = 1 for x = 0:
x dina if a dinyy
in as a ae
ae mdf de ym dx ™
: ann
(@) For Inj, = Ax, Ot =24n
Whence ny =2a [ nde or inna as
By Eq. (12.6),
(0) For Inyy =
Whence nya 2A [mide +39 [nde 38 [xan
I to I
: alae b
inp = ar} + 2xt— ax} or inna (44% an) =a [a4 04200]
sane 3B
Apply Bq. (12.6): & aad(d + Bx) + wax} + > ~ Bu)
Algebraic reduction can lead to various forms of this equation; e.g,
Ge B
F oaafa Fara]
(ec) For Inyy = x}(4 + Bxz + Cx3),
PON ood + Beat Cx) +B +20n) =2Ae +3Bd 4d
Ie
= Dax; + 3Bxg(1 —m) +4001 — 4)?
Whence ay 24 f eat +38 f Mal andny sac [sya
I hb A
or nye =Qd +3a+acy [ side, 08480) [stan tac [sax
5 fi
A+ 3B +4C 3B+EC) yas
ZY i> CEES) a vent
Inys
3B
mnasi[er Perc (ar 2) cu]
997
7 wand [arfesais Gasm+%8]|
‘The result of application of Eq. (12.6) reduces to equations of various forms; e.g.
o a c
are [4 + pita) + Fd tat ap
and AH = AH +A)
12.40 (a) As shown on page 444, x1 =
oy Oe
Eliminating 1 +7 gives: ane? my
dan Ally ds,
Differentiation yields: pote rare a
where
Whence,
Combining this withthe result of Par () gives: FA =
From which, i= 8H - anf
Substitute: Py pie gage nate
mH
598
fxg Hea mfif
Whence, pot’ Rye oe
area
AE
fi
However, by the summabilty equation, #7 — sf
‘Then, T= HE
12.41 Combine the given equation with Eq. (A) of the preceding problem:
Sif = sais + Arve)
With xp =1—x) and xy =1/U+A) (page 444): x
‘The preceding equations combine to give:
ki-— (¢
+H
(a) It follows immediately from the preceding equation that: tim AH = 0
(0) Because fi/(1 +i) + 1 for fi —> 00, it follows that:
(©) Analogous to Eq. (12.10b), page 424, we write:
Eliminate the mole fractions in favor of
: 1 ji
AE (=) [arta aot]
In the limit as i? — 0, this reduces to Aa. From the result of Part (a) of the preceding problem,
it follows that
ast
av dit
Ant
12.42 By Eq, (12,29) with MF
) SH =H ~ Dix. Differentiate:
(22),-@,-2@,
With (¥) =Cp, this becomes (3) =Cp~ SisiCn = ACr
ai
Therefore, f aaH.
i [lecea [ans ame fh acrar
599
Chapter 13 - Section B - Non-Numerical Solutions
134 (@) ANH (@) + 502(g) + 4NO(g) + 6HLO(®)
ee
T B
By Bq, (13.5),
er ~5e 4e b=
DMmy= FE, A= FE MOTE MMO=Zye
® 2HS(g) + 302(@) + 2H1:0(g) + 280218)
By Eq. (135),
: _ 2 _ 2
ste OO ere MORTEM BEE
© 6NOn(g) + 8NHs(@) > TNo(e) + 12H20(2)
ve Da -6- 84741255 m= P= 344H1=8
By Eq, (13.5),
_ 3-60 4-86 _ 1446 12s
wor ay5e OM BES. OM Bese HOT RE Se
132 Coal) + }Or(@) + ((CHs)2)1@) wo
CaHg(g) + 302(g) > 2CO2(g) + 2H20(g) Q)
‘The stoichiometric numbers v,,, areas follows:
By Eq. (13.7),
Jet =
133 CO2(g) + 3H2(g) + CH;OH(@) + H20(g) oO
COz(g) + Hz) > COG) + H:0) Q
‘The stoichiometric numbers v,; sre as follows:
Oz Hh CHOCO HO
Bens oye
ite 1 1-3
ai ° eee
m= Sa2454128
ByEg.(13.7,
ee & l+e Ste
pete e gee HON Fe, POO Bde, MO SIDE,
13.7 The equation for AG*, appearing just above Eq, (13.18) is:
an T ace T ACZaT
AHS - = a — AG +R Pa? ~RT re
AG IS ane fa fi z |, RT
To calculate values of AG*, one combines this equation with Eqs. (4.19) and (13.19), and evaluates
parameters. In each case the value of Aflj) — Afiigg is tabulated in the solution to Pb. 4.21, In
addition, the values of 4.4, AB, AC, and AD are given in the solutions to Pb. 4.22. The required
values of AG} = AGieg in J mol"! are:
(a) -32,900; (/) ~2.919,124; (i) 113,245; (n) 173,100; (*) 39.630; (0) 79,455; (u) 166,365;
(s) 39,430; (9) 83,010
13.8 The relation of K, to P and K is given by Eq. (13.28), which may be concisely written:
o-()"s
(@) Differentiate this equation with respect to T snd combine with Eq, (13.14):
Ge),-G)
‘Substimute into the given equation for (86, /97)
(b) The derivative of K,. with respect to P is:
() (FY
601
Bg
‘Substitute into the given equation for (3/3 P)r:
(F),
(© With Ky= F100", ky = Devin, Pilleremiation then yet:
Ldky yy
Ky dee Sy, de
(A)
dy Vide om dn
de, nde. mde,
Because yy = m/m,
But nrg b Ube and n= My + vee
Whenee,
‘Therefore,
Substitution into Eq. (4) gives
teas vw (aye
Ede be (2)
In this equation, both Ky and ng-+ vee (= n) ate positive. It remains to show thatthe summation
term is positive, If m = 2, this term becomes
f 8 o
4 ye, typ 2 mg toy =
y » ya
where the expression on the right is obtained by straight-forward algebraic manipulation. One
can proceed by induction to find the general result, which is
Dy ey? <2
vive
All quantities in the sum are of course positive
3N2@) + jG) > NH)
For the given reaction, v = —1, and for the given amounts of reactants, ng = 2.
jase)
By Eq. (13.5), Nk
By Eq. (13.28),
13.10
17 ay p >
Whence, Goat 6) (3) Ka 1299 55
This may be written: red —2rep+(r—-1)=0
where,
‘The roots of the quadratic are:
Because 6 <1, = 1—r-¥?,
‘The reactions are written:
Mary: 2NH; + 3NO — 3H;0 + $Np 4)
Paul: ANH: + 6NO > 61:0 + 5Nx @
Peter: 31,0 + §N; > 2NH, +3NO ©
Each applied Eqs. (13-116) and (13.25), here written:
IK =-aGyRT ond K = (PY TCA"
For reaction (4), AG = 3AG%,, ~ 28%, — 3G ig
For Mary's reaction » = }, and
A pn i
sy! fueling and nk, = 294
FhusTia a
For Paul's reaction v = I, and
Shao Fin ~2A6%
Ka=@Py! we and = InKe 4
Fius Sie oe
For Peter's resetion » = —4, and
Ko = (PY
Mary: (py
Paul:
Peter:
‘Taking the reciprocal yields Mary’s equation
13.24 Formation reactions: INo + Whe > Nis w
@
°
Hy +40; + 120 ®
Combine Eq, (3) with Eq, (1) and with Bq, (2) to eliminate Nz
NO» + }H; > NH + 02 5)
NO» > $0; +NO 6
Te set now comprises Eqs. (4), (5), and (6); combine Eq, (4) with Eg, (5) to eliminate Hy
NOp + $10 NH +1302 o
Equations (6) and (7) representa set of independent reactions for which r = 2. Other equivalent sets
of two reactions may be obtained by different combination procedures. By the phase rule,
Fa2-xtn- 2-145-2-0
wo (PY pie
(a) Equation (13.28) here becomes: t.(<)K=K
13.35 (a) Equation (13.28) x (FB)
Whence, ="
Kcr) |
(6) The preceding equation indicates thatthe equilibrium composition depends on temperature only.
However, application of the phase rule, Eq. (13.36), yields
Fo2+2-1-
2
This result means in general for single-reaction equilibrium between two species A and B that
two degrees of freedom exist, and that pressure as well as temperature must be specified to fix the
equilibrium state ofthe system. However, here, the specification that the gases are ideal removes
the pressure dependence, which in the gencral case appears through the ds.
13.36 For the isomerization reaction in the gas phase at low pressure, assume ideal gases. Equation (13.28)
then becomes:
ye _ (2 y lay,
-) K=K whence YA = KD)
ma & mK
604
Assume that vapor/liquid phase equilibrium can be represented by Raoult’s law, because of the low
pressure and the similarity of the species:
APD) =nP and
(1 xa) PENT) = = ya) P
(a) Application of Eq. (13.36) yields:
(6) Given T, the reaction-equilibrivum equation allows solution for ya. The two phase-equilibrium
‘equations can then be solved for x, and P. The equilibrium state therefore depends solely on T-
13.38 (a) For low pressure and a temperature of 500 K, the system is assumed to be a mixture of ideal
‘gases, for which Eq. (13.28) is appropriate, Therefore,
oa (B)'« aK (2) «= () Ku
(@) These equation equations lead 0 the folowing set
na Kivox | (1) [ox = Knox] (yen = Kinyox | @)
‘The mole fractions must sum to unity, and therefore;
vox + Kiyox + Kuyox + Kmyox = Yox(1 + Ki + Ku + Kun) = 1
i
Jox = TPE ET Kw ®
(©) With the assumption that AC'z = 0 and therefore that Kz = 1, Eqs. (13.20), (13.21), and (13.22)
combine to give:
s = exp (—AGis8 aces) Altay (, _. Te
K=KoK, ~e( Fe)? | ee (l- F
298.15
att (i= 28) — a
Whence, (8.314) 298.15)
K exp
‘The data provided lead to the following property changes of reaction and equilibrium constants
at 500 K;
Reaction | AHZq AGig K
1 | -1,750 2.8470
=1,040 ~1,000 1.2637
Mm | 10920 8690 0.1778
(@ Substitution of numerical values into Eqs. (1), (2), (3), and (4) yields the following values forthe
mole fractions:
vox = 0.1891 nox = 0.5383 Yox = 0.2390 rw = 0.0336
605
Chapter 14 - Section B - Non-Numerical Solutions
14.2 Start with the equation immediately following Eq, (14.49), which can be modified slightly to read:
s (nG"/RT inZ) ainz
where the partial derivatives written here and in the following development without subscripts are
understood to be at constant T', n/p (or p/n), and n,. Equation (6.59) after multiplication by n can
be written:
nck PY 32 ey
Tar = 2mm) (2) + sntincy (2) —ninz
Differentiate:
a@GYRT) _ 42 ay.3
(Eee endo + 5 (E
nG* fi 3
or BOC TED) a8 4.) + 39220 46) Inz
Sn
amBy cae
Gm br
The equation of state, Eq, (3.39), can be written:
By definition,
Zant Bo+C oe n2mnt nds (2) +02) (2)
(nz)
an;
Differentiate
+ (2) entry + (2) ante +2
1+ p(B+B)+eQC+E))
When combined with the two underlined equations, the initial equation reduces to:
Ing,
14 9B +B) + PAC +)
‘The two mixing rules are:
2By
B=yiBy + 2iy2Bo
C=C + SyfmCi2 + 3ryFCm + 2Cae
Application ofthe definitions of Band C; to these mixing rues yields:
12 —yi)Br +29} Bu
2G = 2yndCin + 6y123Cu12 + 3y3(1 ~ 2y1)Ciza — By} Caz
{Bir + 2yf Bia + y2t2 — ya) Bro
2yi Cin + 3¥FC1 — 2ydCii2 + 6y1¥3Cina + 2973 — 2y2)Cax2
606
14.1
In combination with the mixing rutes, these give:
B+ B, = 201 + Bu)
2C + Cy = 307Ciu + 2ymCra + 93Cin)
B+ Bz = 2G2Bu + y1Bi2)
2C + Cy = 303Can2 + 2y192Ciza + Y7Ci2)
In combination with the boxed equation these expressions long with Eq, (3.38) allow calculation of
Indy and Inge,
For the case described, Bgs. (14.1) and (14.2) combine to give: yy P = xP)
If the vapor phase is assumed an ideal solution, 9; =), and yjP =x;P)
When Eq, (3.37) is valid, the fugacity coefficient of pure species jis given by Eq, (11.38):
— — BaP _ Ble PY
For small values of the final term, this becomes approximately:
of, umn
— snp [i BEEP
aT
Write this equation for species 1 and 2 of a binary mixture, and sum. This yields on the left the
difference between the actual pressure and the pressure given by Raoult’s law:
P— Pony = HE Py esBals (Ppt = P)
Because deviations from Raoult’s law are presumnably small, P on the right side may be replaced by
its Raoul’s-law value. For the two terms,
Pt P= PM 2PM Pt
PS a2) PM — Pt = (PPS
PS ~ P= PS PM PM = PS PM x) PA = (PS Be)
Combine the three preceding equations
ut Br (PM — PPM xx
RT
eae (Bo — Bes pt
= psy
(Bn Pi ~ BaP)
607
Rearrangement yields the following:
snp Pe — PE)? (By BY — Bea PM
P—PIRL) = ane es (aoe)
RT PS — PF
xn(Pet ~ PS [oy + ~ Bu) PS
7) 7A |
Clearly, when Bz: = Brs, the term in square brackets equals 1, and the pressure deviation from the
Raoult’s-law value has the sign of Bi; this is normally negative. When the virial coefficients are not
‘equal, a reasonable assumption is that species 2, taken here as the “heavier” species (the one with
the smaller vapor pressure) has the more negative second virial coefficient. This has the effect of
‘making the quantity in parentheses negative and the quantity in square brackets < 1. However, if this
latter quantity remains positive (the most likely case) the sign of By, still determines the sign of the
deviations.
14.13 By Eq. (11.87), the definition of, Iny, = In fy ~ Inxy ~ Inf
wens Ta ayaa
‘Combination of this expression with Eq. (14.67) yields: jas” 0
pecawse za, | 2450 coos. r)
By Eq. (11.42), the definition of fi, =
Combination with Eq. (14.68) yields:
14.14 Stability requires that AG <0 (see Pg. 556). The limiting case obtains when AG = 0, in which
event Eq, (12.30) becomes:
oF
aT Dx inxy
Foran equimolar solution x; = 1/N where Nis the number of species. Therefore,
GF (max
eel 1
a atat <= InN = RT InN
RTS in gy = AT Ey WN = aT
and GF (max) = RT In2
For the special case of a binary solution, N
608
14.17
M19
1421
14.23
According to Pb.11.38, G&=8,2Pyy2 or
Ge
This equation has the form: S— = Axx
his equation has RF
for which itis shown in Examples 14.5 and 14.6 that phase-splitting occurs for 4 > 2. Thus, the
formation of two immiscible vapor phases requires: 12P/RT > 2.
Suppose 7 = 300 K and P = 5 bar. The preceding condition then requires: 612 > 9977 em? mol!
for vapor-phase immiscibility. Such lange positive values for 8:2 are unknown for real mixtures.
(Examples of gas/gas equilibria are known, but at conditions outside the range of applicability of the
two-term virial EOS.)
GF
Consider a guadratic mixture, deseribed by: =. = Axyep
It is shown in Example 14.5 that phase splitting occurs for such @ mixture if A > 2; the value of
A = 2 corresponds to a consolute point, at x: = x2 = 0.5. Thus, for a quadratic mixture,
phase-spliting obtains i
of oat tL eraosar
This is @ model-dependent result. Many liquid mixtures are known which are stable as single phases,
eventhough G¥ > 0.SRT for equimolar composition.
‘Comparison of the Wilson equation, Eq, (12.18) with the modified Wilson equation shows that
GRD )m = C(GE/RT), where subscript m distinguishes the modified Wilson equation from
the original Wilson equation, To simplify, define g@ = (G4/RT);, then
Bm = CE gm = Cg. Inn = Clay,
where the final equality follows from Bq, (11.92). Addition and subtraction of Inx; on the left side
of this equation and of C In; on the right side yields
ln(siyie = ney = CIn(eyy) — Clan,
or In@eiyi)n = C Inger) — (C= Nina
: dingayn _ dine -
Differentiate: a > eee
As shown in Example 14.7, the derivative on the right side of this equation is always positive. How-
ever, for C sufficiently greater than unity, the contribution of the second term on the right ean make
dingy
dx,
over part of the composition range, thus violating the stability condition of Bq, (14.67) and implying
the formation of two liquid phases,
<0
@ Refer to the stability requirement of Eq. (14.66). For instability, ie., for the formation of two
Tiguid phases,
@UGE RT) 1
dd =
609
14.29
1430
1431
over part of the composition range. The second derivative of G® must be suliciently negative so
as to satisfy this condition for some range of x). Negetive curvature is the norm for mixtures for
which G* is positive; see, eg., the sketches of G* vs, x; for systems (a), (2), (), (e), and (f) in
Fig. 11.4. Such systems are candidates for liquid/liquid phase splitting, although it does not in
fact occur for the cases shown. Rather large values of G* are usually required,
(b) Nothing in principle precludes phase-splitting in mixtures for which G* < 0; one merely re-
quires that the curvature be sufficiently negative over part of the composition range. However,
positive curvature is the norm for such mixtures. We know of no examples of liquid/liquid phase-
splitting in systems exhibiting negative deviations from ideal-solution behavior.
The analogy is Raouit’s law, Eq. (10.1), applied at constant P (see Fig. 10.12): ypP = 11,
Ifthe vapor phase in VLE is ideal and the liquid molar volumes are negligible (assumptions inherent
in Raoult’s law), then the Clausius/Clapeyron equation applies (see Ex. 6.5)
dine _ Ani
@ R
Integration from the boiling temperature T, at pressure P (where P,** = P) to the actual temperature
T (where PB = P,** gives:
Combination with Bq, (10.1) yields:
which is an analog of the Case SLE equations.
Consider binary (to-species) equilibrium between two phases of the same kind. Equation (14.70)
applies:
xtytaafyf = 1,2)
Irphase pis pure species 1 and phase is pure species 2, then xf = yf =1 and xf
md oyfaahyP=t
Henee, atyt =afyt =
‘The reasoning applies generally to (degenerate) N-phase equilibrium involving ’ mutually immis-
cible species. Whence the cited result for solids.
The rules of thumb are based on Case II binary SLE behavior. For concreteness, lt the solid be pure
species ! and the solvent be liquid species 2. Then Eqs. (14.89) and (14.88a) apply:
)
AH?!
n= vsey Sl (
ax AH
(a) Differentiate: Spa Sp
Thus dxi/d7 is necessarily positive: the solid solubility x; increases with increasing 7.
(6) Equation (14.88a) contains no information about species 2. Thus, to the extent that Eqs. (14.89)
and (14.88) are valid, the solid solubility x is independent ofthe identity of species 2.
610
(c) Denote the two solid phases by subscripts 4 and B. Then, by Eqs. (14.89) and (14,88a), the
solubilities x4 and xp are related by:
XA gp | SH Tins = Ta)
facet Tas Pa
‘where by assumption, An] = Hj = ant
Accordingly, x4/xp > 1 ifand only if Ty 7 (See Fig. 14.21b). Then x4/tq > 1 ifandonlyif AH < SHG, in
accord with the rule of thumb.
14.34 The shape of the solubility curve is characterized in part by the behavior of the derivative dy, /4P
(constant 7). A general expression is found from Eq. (14.94), »; = PSP /Fi, where the enhance-
ment factor F; depends (at constant T) on P and y;. Thus,
#(),-@)2|
“BenQ, C59) 3]
in (4), 1 ]
ap (ain Ae
a ay
This is a general result. An expression for Fis given by Eq, (14.95)
pat
Whence,
oe PP)
exp
A
RF
From this, after some reduction:
in Fi) aindy WE
(Ch }, -() Rr (
Whence, by Eq. (4), (8)
‘This too is a general result. If the two-term virial equation in pressure applics, then Ing, is given by
Eg, (11.59), from which:
ands 1 aindy 2yabi2P
(0B!) = frases we (ORB) =
Vix Bu - yu _ 1
dy af RT P
SaP
RT
Whence, by Eq. (B),
The denominator of this equation is positive at any pressure level for which Eq. (3.37) is likely to be
valid. Hence, the sign of dy, /d P is determined by the sign of the group in parentheses. For very low
pressures the 1/P term dominates and dvi /dP is negative. For very high pressures, 1/P is small,
and dy;/aP can be positive. If this is the case, then dy /dP is zero for some intermediate pressure,
and the solubility yy exhibits a minimum with respect to pressure. Qualitatively, these features are
conisistent with the behavior illustrated by Fig. 14.22. However, the two-term virial equation is only
valid for low to moderate pressures, and is unable to mimic the change in curvature and “fattening”
of the »; vs. P curve observed for high pressures for the naphthalene/CO; system,
14,35 (a) Rewrite the UNILAN equation:
=F inie+ Pet) info + Pe] Ca
As s+ 0, this expression becomes indeterminate. Application of PHépita’s rule give
imn = tim™ (Pe Pew
sa" = 807 oe Pet” oF Pee
=4( ae
“ TNCEP eH
or
Which is the Langznuir isotherm,
() Henry's constant, by definition: — = fim
jeury’s constant, by definition; = fim SE
dn_m(e e
Ditferentiate Eq. (a): “(8
ce a 5 (cine er Pe
Whence,
2465) «
(©) All derivatives of n with respect to P are well-behaved in the zero-pressure limit:
m
™ sinh s
dn
fim = Zosinhs
612
14.36
1437
Bee.
Numerical studies show that the UNILAN equation, although providing excellent overall corre-
lation of adsorption data at low-to-moderate surface coverage, tends to underestimate Henry's
constant
Start with Eq. (14.105), written as:
ingP/m) =—ink + f'@— 2-1
With 2=148n+Cn?-+--, this becomes:
In(P/n) = ~Ink +2Bn + det +
Thus a plot of In(P/n) vs. m produces —Ink as the intercept and 2B as the limiting slope (for
n> 0), Altemstively, a polynomial curve fit of In(P/m) inm yields —Ink and 2B as the frst
two coefficients
For species i in a real-gas mixture, Eqs. (11.42) and (11.48) give:
bh = UD) + RT inyidyP
RT dinydP
At constant temperature, aut
With dy,
uf Eq, (14.101) then becomes:
Feat dln + Yxdinysds = 0 (const 7)
For pure-gas adsorption, this simplifies to:
a
fan= (cons
FpaadinP+ding (const 7) ey
which is the real-gas anslog of Eq. (14,103). On the left side of Eq, (4), introduce the adsorbate
compressibility factor through = =: Ta/RT = I14/nRT:
a dn
gpa a de (8)
where n is moles adsorbed. On the right side of Eq. (4), make the substitution:
ap
ding=@- D> ©
which follows from Eq. (11.34). Combination of Eqs. (4), (B), and (C) gives on rearrangement (see
See. 14.8)
ne dn dP
ding = 0-9 dee ZS
which yields on integration and rearrangement:
vairen fe -y'2-o9[ fa
This equation is the real-gas analog of Eq, (14.105).
613
14,39 & 14.40 Start with Bq. (14.105). With z= (1 —bm)-', one obtains the isotherm:
n= EPC — br)exp (- w
bn
For bn sufficiently small, exp (>) a1
kp
Whence, by Bq. (4), © EP(L~ 2bm) eon
tee eee T+ 35kP
which isthe Langmuir isotherm
With 2= 1+ Bn, the adsorption isotherm is: KP exp(—2Bn)
from which, for Bn sufficiently small, the Langmuir isotherm is again recovered
Adm dP
1441 By Fa, (14.103) with a= A/m, S6H- SE
yg, (14.108) with @ = dfn, SEP a WZ
The definition of y and its derivative ar:
aan
and ai
Whence, wo
@)
By general differentiation,
» ”
n= Hey ©
ay Pav pet Lge? ©
The equation, ¥;x; = 1, is an approximation that becomes increasingly accurate asthe solution
procedure converges. Thus, by rearrangement of Fg, (B),
pe
LF,
With P fixed, Eq. (C) can now be written in the simple but approximate form:
aP
@Du=5
Buss
Equation (4) then becomes:
dyandDx or sy =na( Ex)
‘where we have replaced differentials by deviations. The deviation in J°, x is known, since the true
value must be unity, Therefore,
i
bDn =P -1
ae
61a
14.42
1
Leusnsy
By Eq, (14.128),
Combine the three preceding equations
yi
PY ae
iy = ST _
YS ei/n)
P/PS, the Raoult’s law anslogy, is substituted the required equation is reproduced:
bye
"ee
Multiply the given equation for G#/RT by n and convert all mole fractions to mole numbers:
ay a
‘Apply Eq. (11.92) for #
1) pam) a
tny, = diom (= 22) 4 dim ($= 8) — antat
= Ajaa(l — 4) + Ars = x1) = Aaysnxs
Introduce solute-free mole fractions:
% wd xe
te *
‘Whence, day = Aor} — x1)? + Araxs) — x1)? — Ansxdx$ (1 — x1)?
Fors 0, ln yf? = Anaes + Aisxs — Aasxixh
Apply this equation to the special case of species 1 infinitely dilute in pure solvent 2. In this case,
xsl, xh=0, and
my = Ai Also Iny = Ais
Whence, In yp? = xh 723 4 x5 In yf — Aasxtas
|In logarithmic form the equation immediately following Eq, (14.24) on page 536 may be applied to
the several infinite-dilution cases:
In =n fitiny® — Ay a=InFitiny twas
nf +iny?s
‘Whence, In Ay = In fy = x4 (ln Hy 2 — In fi) + 44m Hy 3 — In ft) — Arsxixh
or In
yin Mia + x4 InAs — Aastix)
61s
14.43 For the situation described, Figure 14.11 would have neo regions like the one shown from « to f.
probably one on either side of the minimum in curve I.
ty
aT
1444 By Eq. (14.123) with Py = Vp Incr)
Represent in y» by a Taylor series:
tnralnno + 222] a
ny eines
me] ate
But at x; = 0 (x2 = 1), both In y and its first derivative are zero. Therefore,
1 (any i
‘ if at di i
Also, Ingy Ind =) =x) -
1/@inyg
13 ast ye
1 1 (@loyy
at p aa ( as ) od: *
Iny
Therefore, In(xzy3) = +Inx2 + In»
and.
B
616
Chapter 16 - Section B - Non
umerical Solutions
16.1 The potential is displayed as follows. Note that K is used in place of & as a parameter to avoid
confusion with Boltzmann’s constant.
‘Combination of the potential with Eq, (16.10) yields on piecewise integration the following expression
for B
2
B= aN [4K =H
ey PREM 1)
BBN og yee tT 4 Reeth
From this expression, SF = £5 [-(A? ~ nge¥7 4 (P — K*yee"7]
according to which dB/d7 —0 for 7» co and also for an intermediate temperature Ty
e+e
ol Ge)|
‘That Tr, corresponds to a maximum is readily shown by examination of the second derivative d*B /dT?,
16.2 The table is shown below. Here, contributions to 2 (long range) ate found from Eq, (16.3) [for U(et)},
Bq, (16.4) [for Mind], and Bq, (16.5) [for (disp)}. Note the following:
1, As also seen in Table 16.2, the magnitude of the dispersion interaction in all cases is substantial
2. U(el), henec fe!), is identically zero unless both species in a molecular pair have non-zero
permanent dipole moments,
3. As seen for several of the examples, the fractional contribution of induction forces ean be sub-
stantial for unlike molecular pairs. Roughly: (ind) is larger, the greater the difference in polarity
of the interacting species.
617
Molecular Pair = Cg/10- Jm® fel) find) f(disp) fed F(disp)
CHUCHH ig 498
oo 1000 0
CHyYCHCh 343 0 0,008 0.992 a
CHa(CHs).CO 249 0 0088 0.912 8
CHYUCH,CN 2.1 0 0188 0812 °
GHeCHCh 161.9 0 0008 0.992 0
CHHye(CHS):CO 119.1 0 0096 0.904 8
CHyg/CH3CN 106.1 0 0.205 0.795 0
CHCly/(CH3)2CO 95.0 0.143 0.087 0.770 0.186
CHC1;/CH3CN. 983 0.263 0.151 0,586 0.450
(CH)COICHSCN 27030805. 0.052 0.142——5.680
16.3, Water (#,0), a highly polar hydrogen donor and acceptor, is the common species forall four systems;
in all four cases, it experiences strong attractive interactions with the second species. Here, interactions
between unlike molecular pairs are stronger then interactions between pairs of molecules of the same
kind, and therefore A is negative. (Sec the discussion of signs for H in Sec. 16.7.)
16.4 Of the eight potential combinations of signs, two are forbidden by Eq. (16.25). Suppose that H® is
negative and $® is positive, Then, by Eq. (16.25), G* must be negative: the sign combination G¥ @,
H® 9, and S® @ is outlawed, Similar reasoning shows that the combination G® ©, H® @, and S© ©
is inconsistent with Eq. (16.25). All other combinations are possible in principle.
16.5 In Series A, hydrogen bonding occurs between the donor hydrogens of CHCl: and the electron-rich
benzene molecule. In series B, a charge-transfer complex ocours between acetone and the aromatic
benzene molecule. Neither cyclohexane nor n-hexane offers the opportunity for these special salvation
interactions, Hence the mixtures containing benzene have more negative (smaller positive) values of
H® than those containing cyclohexane and n-hexane. (See Secs. 16.5 and 16.6.)
16.6 (a) Acetone/eyclohexane is an NAINP systeon; one expects G¥ ®, H® ©, and S® &.
(®) Acetoneidichloromethane is a solveting NA/NA mixture. Here, without question, one will see
GF 6, H® 2, and S¥ 8,
(©) Aniline/eyctohexane is an AS/NP mixture. Here, we expect either Region I or Region II behavior:
GF @ and H® ©, with SF @ or. [At 323 K (50°C), experiment shows that S® is @ for this
system]
(A) Benzene/carbon disulfide is an NP/NP system, We therefore expect G* &, H® ©, and S* @,
(©) Benzenetn-hexane is NP/NP. Hence, G* @, H® ©, and S® @.
(A) Chloroform/i,4-dioxane is a solvating NA/NA mixture. Hence, G® ©, H® ©, and S® ©.
(2) Chloroforin/a-hexane is NA/NP. Hence, G® @, H® @, and S* ©,
(A) Bthanol/n-nonane is an AS/NP mixture, and ethanol is a very strong associator. Hence, we expect
Region Il behavior: G* @, H® ®, and S® 6.
16,7 By definition, 2[By —$(6u+By)]
At normal temperature levels, intermolecular attractions prevail and the second vial oeticents are
negative. (See Sec. 16.2 for a discussion of the connection between intermolecular forces and the
second virial coefficient) ITinterations between unlike molecvar pairs are weaker than interactions
between pairs of molecules of the same kind,
Byl < HB + By|
618
and hence (since each B is negative) 4; > 0. Ifunlike interactions are stronger than like interactions,
[Byl > HB + Byl
and hence 6 <0. [interactions are identical for all molecular pairs, By = Bu = By and
by =0
‘The rationalizations of signs for H of binary liquid mixtures presented in See. 16.7 apply approxi
mately tothe signs of 2 for binary gas mixtures, Thus, postive isthe norm for NP/NP, NAJNP, and
ASINP mixtures, whereas diz is usually negative for NA/NA mixtures comprising solvating species
One expects 612 to be essentially zero for ideal solutions of real gases, e.g, for binary gas mixtures of
the isomeric xylenes
619
You might also like
- Coulson and Richardson’s Chemical Engineering: Volume 2A: Particulate Systems and Particle TechnologyFrom EverandCoulson and Richardson’s Chemical Engineering: Volume 2A: Particulate Systems and Particle TechnologyNo ratings yet
- Chemical Engineering Review For PE ExamDocument274 pagesChemical Engineering Review For PE Examkrishnaswamy9100% (8)
- Solution Manual for an Introduction to Equilibrium ThermodynamicsFrom EverandSolution Manual for an Introduction to Equilibrium ThermodynamicsNo ratings yet
- ACM Examples GuideDocument240 pagesACM Examples GuideSurya Budi Widagdo100% (8)
- Phase Equilibria in Chemical EngineeringFrom EverandPhase Equilibria in Chemical EngineeringRating: 4 out of 5 stars4/5 (11)
- Fundamentals of Chemical Reaction EngineeringFrom EverandFundamentals of Chemical Reaction EngineeringRating: 2.5 out of 5 stars2.5/5 (3)
- Aspen Custom Modeler 2004.1: Dmcplus Controllers Interface GuideDocument42 pagesAspen Custom Modeler 2004.1: Dmcplus Controllers Interface GuideSurya Budi WidagdoNo ratings yet
- Aspen MUSE GettingstartedDocument27 pagesAspen MUSE GettingstartedSurya Budi WidagdoNo ratings yet
- Energy Conversions ChartDocument3 pagesEnergy Conversions ChartSurya Budi WidagdoNo ratings yet
- IEEE STD 367-1996 - Determining The Electric Power Station Ground Potential Rise and Induce Voltage From Power Fault PDFDocument131 pagesIEEE STD 367-1996 - Determining The Electric Power Station Ground Potential Rise and Induce Voltage From Power Fault PDFMuhammad SyarifNo ratings yet
- Porter Easter Ling SolutionsDocument74 pagesPorter Easter Ling SolutionsKrishna Mahajan60% (10)
- VaporizerDocument13 pagesVaporizerSurya Budi Widagdo100% (1)
- Aspen-Getting Started Using Equation Oriented ModelingDocument90 pagesAspen-Getting Started Using Equation Oriented ModelingSurya Budi WidagdoNo ratings yet
- 04 Reactive DistillationDocument16 pages04 Reactive DistillationJaJar Yaneeporn PatcharavorachotNo ratings yet
- Chemical Engineering Thermodynamics Solution Manual PDFDocument725 pagesChemical Engineering Thermodynamics Solution Manual PDFNaveen100% (1)
- Fundamentals of Momentum, Heat, and Mass Transfer 5th Edition Welty Solutions ManualDocument328 pagesFundamentals of Momentum, Heat, and Mass Transfer 5th Edition Welty Solutions ManualDylan Johnson89% (56)
- Natural Gas Storage TankDocument10 pagesNatural Gas Storage TankSurya Budi WidagdoNo ratings yet
- ACM Polymer Simulations With Polymers PlusDocument64 pagesACM Polymer Simulations With Polymers PlusSurya Budi Widagdo100% (1)
- Thermodynamic Models for Chemical Engineering: Design, Develop, Analyse and OptimizeFrom EverandThermodynamic Models for Chemical Engineering: Design, Develop, Analyse and OptimizeNo ratings yet
- Separations Processes SolutionsDocument495 pagesSeparations Processes Solutionsgreg_mclawnen80% (5)
- Solution Manual For Chemical Engineering ThermodynamicsDocument4 pagesSolution Manual For Chemical Engineering ThermodynamicsBayu Prayudi Wibowo8% (36)
- Fixed-Bed Reactor Design and Diagnostics: Gas-Phase ReactionsFrom EverandFixed-Bed Reactor Design and Diagnostics: Gas-Phase ReactionsRating: 4 out of 5 stars4/5 (5)
- Coulson and Richardson’s Chemical Engineering: Volume 1B: Heat and Mass Transfer: Fundamentals and ApplicationsFrom EverandCoulson and Richardson’s Chemical Engineering: Volume 1B: Heat and Mass Transfer: Fundamentals and ApplicationsRating: 3.5 out of 5 stars3.5/5 (3)
- Kinetics (Gjjkkkgty)Document5 pagesKinetics (Gjjkkkgty)Chrysler Kane DepnagNo ratings yet
- Customizing RADFRAC ConvergenceDocument22 pagesCustomizing RADFRAC ConvergenceSurya Budi Widagdo75% (4)
- Introductory Chemical Engineering Thermodynamics, Elliot & Lira PDFDocument690 pagesIntroductory Chemical Engineering Thermodynamics, Elliot & Lira PDFAndrea Castillo86% (42)
- Modeling in Transport Phenomena: A Conceptual ApproachFrom EverandModeling in Transport Phenomena: A Conceptual ApproachRating: 3 out of 5 stars3/5 (2)
- Solution Manual For Fluid Mechanics and Thermodynamics of Turbomachinery 7th Ed Sydney Lawrence Dixon Cesare Hall PDF FreeDocument10 pagesSolution Manual For Fluid Mechanics and Thermodynamics of Turbomachinery 7th Ed Sydney Lawrence Dixon Cesare Hall PDF FreePIYUSH PORWALNo ratings yet
- Che 05012 Chemical KineticsDocument8 pagesChe 05012 Chemical Kineticstri anggraini arifNo ratings yet
- Introduction To Statistical Physics Solution Manual: Kerson HuangDocument105 pagesIntroduction To Statistical Physics Solution Manual: Kerson Huangsumivrindavan100% (7)
- Veljko Milkovic Two Stage Mechanical OscillatorDocument39 pagesVeljko Milkovic Two Stage Mechanical OscillatorAbigaile2009No ratings yet
- Maintenance and Operation of Electric PL PDFDocument233 pagesMaintenance and Operation of Electric PL PDFsani priadiNo ratings yet
- Aspen-Getting Started Modeling Processes With ElectrolytesDocument60 pagesAspen-Getting Started Modeling Processes With ElectrolytesSurya Budi Widagdo100% (1)
- Linear Control of Nonlinear ProcessesDocument23 pagesLinear Control of Nonlinear ProcessesSurya Budi WidagdoNo ratings yet
- ACM Modeling Language ReferenceDocument193 pagesACM Modeling Language ReferenceSurya Budi Widagdo100% (1)
- Solution Manual LevenSpiel by 13BCH@ITNUDocument370 pagesSolution Manual LevenSpiel by 13BCH@ITNUManishMakwana87% (156)
- Paper Thermo Mechanical EngineeringDocument14 pagesPaper Thermo Mechanical EngineeringAdif HerawanNo ratings yet
- Thermodynamics (Prausnitz) Manual SolutionDocument195 pagesThermodynamics (Prausnitz) Manual SolutionColin Zhang78% (9)
- Statistical Physics Solution ManualDocument105 pagesStatistical Physics Solution Manualgeorgeattack100% (4)
- Reaction Kinetics for Chemical Engineers: Butterworths Series in Chemical EngineeringFrom EverandReaction Kinetics for Chemical Engineers: Butterworths Series in Chemical EngineeringRating: 4 out of 5 stars4/5 (3)
- Thermodynamic Manual SolutionsDocument665 pagesThermodynamic Manual SolutionsFövqi Efendi86% (21)
- Differential DistillationDocument36 pagesDifferential DistillationShruti100% (1)
- John Mark R. Allas Bs Che-3Document11 pagesJohn Mark R. Allas Bs Che-3John Lloyd De CastroNo ratings yet
- Chapter 2 - Section A - Mathcad Solutions: M 35 KG G 9.8 M S Z 5 M Work M G Z Work 1.715 KJ U Work U 1.715 KJDocument11 pagesChapter 2 - Section A - Mathcad Solutions: M 35 KG G 9.8 M S Z 5 M Work M G Z Work 1.715 KJ U Work U 1.715 KJFaris Naufal100% (1)
- Prob 4.44 Table Lookups Given Values: R and V Are Constant So..Document5 pagesProb 4.44 Table Lookups Given Values: R and V Are Constant So..tkshoeNo ratings yet
- Chapter5 ADocument21 pagesChapter5 ANic BlandoNo ratings yet
- MMAN2700ThermoProblemSheet6Solutions - 1st Law Steady FlowDocument21 pagesMMAN2700ThermoProblemSheet6Solutions - 1st Law Steady Flowgrandw9524No ratings yet
- CH 10Document34 pagesCH 10hirenpatel_universalNo ratings yet
- Chapter 1 - Section A - Mathcad SolutionsDocument4 pagesChapter 1 - Section A - Mathcad SolutionsdjeofitraNo ratings yet
- Self Assessment Solutions Tutorial 7 Self Assessment Exercise No. 1Document5 pagesSelf Assessment Solutions Tutorial 7 Self Assessment Exercise No. 1Alexander MugabeNo ratings yet
- Zeroth Law and Introductory ConceptsDocument13 pagesZeroth Law and Introductory ConceptsMohd Danial Muhd AliNo ratings yet
- Thermo Homework 6Document7 pagesThermo Homework 6Danny BoyleNo ratings yet
- Casa de Dos Pisos EtabsDocument19 pagesCasa de Dos Pisos EtabsYuber Tacuri CristobalNo ratings yet
- Herrera Alisson Deber3 EjerciciosBraytonEESDocument11 pagesHerrera Alisson Deber3 EjerciciosBraytonEESAly HerreraNo ratings yet
- Lohith PDFDocument8 pagesLohith PDFBoddupalli Lohith KumarNo ratings yet
- Chapter 5 - Section A - Mathcad Solutions: 5.2 Let The Symbols Q and Work Represent Rates in Kj/s. Then by Eq. (5.8)Document21 pagesChapter 5 - Section A - Mathcad Solutions: 5.2 Let The Symbols Q and Work Represent Rates in Kj/s. Then by Eq. (5.8)light2618No ratings yet
- School of Chemical Engineering: Chem 251 Assignment 1 and 2Document16 pagesSchool of Chemical Engineering: Chem 251 Assignment 1 and 2Keevani NaidooNo ratings yet
- Solution To Exam No. 2Document5 pagesSolution To Exam No. 2mozam haqNo ratings yet
- Tutorial 3 - Question 8Document2 pagesTutorial 3 - Question 8DiablofireZANo ratings yet
- Engineering Mechanics: Second PartDocument9 pagesEngineering Mechanics: Second Partاحمد سلمان عزيز , مسائيCNo ratings yet
- Introduction To Convection: Flow and Thermal ConsiderationsDocument29 pagesIntroduction To Convection: Flow and Thermal ConsiderationsChris MustacchioNo ratings yet
- Bio Process Engineering Principles (Solutions Manual) - P. Doran (1997) WWDocument170 pagesBio Process Engineering Principles (Solutions Manual) - P. Doran (1997) WWAmm Roeh78% (23)
- CH 11Document22 pagesCH 11Ingenio MetalurgiaNo ratings yet
- Repaso de QI MartesDocument8 pagesRepaso de QI MartesCriss TorresNo ratings yet
- Ten-Decimal Tables of the Logarithms of Complex Numbers and for the Transformation from Cartesian to Polar Coordinates: Volume 33 in Mathematical Tables SeriesFrom EverandTen-Decimal Tables of the Logarithms of Complex Numbers and for the Transformation from Cartesian to Polar Coordinates: Volume 33 in Mathematical Tables SeriesNo ratings yet
- Tables of Coefficients for the Analysis of Triple Angular Correlations of Gamma-Rays from Aligned NucleiFrom EverandTables of Coefficients for the Analysis of Triple Angular Correlations of Gamma-Rays from Aligned NucleiNo ratings yet
- Absolute Pipe Roughness - Enggcyclopedia PDFDocument1 pageAbsolute Pipe Roughness - Enggcyclopedia PDFSurya Budi WidagdoNo ratings yet
- MPCDocument250 pagesMPCPrathak JienkulsawadNo ratings yet
- AbsorberDocument15 pagesAbsorberShri VardhanNo ratings yet
- A-S-P-E-N-O-N-E V7.1Document14 pagesA-S-P-E-N-O-N-E V7.1Surya Budi WidagdoNo ratings yet
- MPC 3Document165 pagesMPC 3Surya Budi WidagdoNo ratings yet
- MPC 2Document141 pagesMPC 2Surya Budi WidagdoNo ratings yet
- OVERVIEW OF PROCESS CONTROL Part 1Document80 pagesOVERVIEW OF PROCESS CONTROL Part 1NOUREDINE MorsliNo ratings yet
- Transport Phenomena 2nd Ed by Bird Stewart Lightfoot (Solution Manual)Document761 pagesTransport Phenomena 2nd Ed by Bird Stewart Lightfoot (Solution Manual)Adibah Hani Azit90% (42)
- An Introduction To Nonlinear Model Predictive ControlDocument23 pagesAn Introduction To Nonlinear Model Predictive ControlSohibul HajahNo ratings yet
- Aspen-Summary File ToolkitDocument208 pagesAspen-Summary File ToolkitSurya Budi WidagdoNo ratings yet
- AspenTech Physical Prop MethodsV7 - 1-RefDocument234 pagesAspenTech Physical Prop MethodsV7 - 1-RefGary McKayNo ratings yet
- Aspen-Getting Started Building and Running Process ModelDocument101 pagesAspen-Getting Started Building and Running Process ModelSurya Budi WidagdoNo ratings yet
- Aspen-Getting Started Modeling Petroleum ProcessesDocument90 pagesAspen-Getting Started Modeling Petroleum ProcessesSurya Budi WidagdoNo ratings yet
- Aspen-Getting Started Modeling Processes With SolidDocument96 pagesAspen-Getting Started Modeling Processes With SolidSurya Budi WidagdoNo ratings yet
- Elce221 Lab 1Document6 pagesElce221 Lab 1Little VoiceNo ratings yet
- B23CS1044 (Neeraj Kumar) Chemistry AssignmentDocument12 pagesB23CS1044 (Neeraj Kumar) Chemistry Assignmentb23cs1044No ratings yet
- Kimo HD200 Thermo Hygrometer DatasheetDocument5 pagesKimo HD200 Thermo Hygrometer DatasheetbrendaparkerudkNo ratings yet
- Schedule 2.0 Product Description: GE Frame PG9171 Technical Description(s) General Operating ConditionsDocument3 pagesSchedule 2.0 Product Description: GE Frame PG9171 Technical Description(s) General Operating ConditionsnboulegrouneNo ratings yet
- Observation Report UPSDocument3 pagesObservation Report UPSr.k.velashNo ratings yet
- Complete NEW BINDING FILE WORK 2Document43 pagesComplete NEW BINDING FILE WORK 2Md Mudassir ShaikhNo ratings yet
- holley-DT-862A Three Phase Electromechanical Watthour MeterDocument4 pagesholley-DT-862A Three Phase Electromechanical Watthour MeterKin Ming ChungNo ratings yet
- Design - Construction of A Vertical Axis Wind TurbineDocument4 pagesDesign - Construction of A Vertical Axis Wind TurbineWinggi ArfaNo ratings yet
- Dynamics: BDA 20303 SEMESTER 1 SESSION 2017/2018 By: Khairulnizam NgadimonDocument22 pagesDynamics: BDA 20303 SEMESTER 1 SESSION 2017/2018 By: Khairulnizam NgadimonShaktivell LetchumananNo ratings yet
- Well-Control Operations in Horizontal Wells: O.L.A. SantosDocument7 pagesWell-Control Operations in Horizontal Wells: O.L.A. SantosJulian Felipe Noguera CruzNo ratings yet
- Heating Ventilation and Air Conditioning The Heart of Modern BuildingsDocument8 pagesHeating Ventilation and Air Conditioning The Heart of Modern Buildingsstynedale18No ratings yet
- WL3 Filled OutDocument8 pagesWL3 Filled OutBraden WellsNo ratings yet
- Chapter 6: The Second Law of Thermodynamics: η net wor k output total heat output W Q Q QDocument4 pagesChapter 6: The Second Law of Thermodynamics: η net wor k output total heat output W Q Q QMariana CaballeroNo ratings yet
- Cvmk2: Three-Phase Power Analyzer, Assembly On Panel or Din RailDocument2 pagesCvmk2: Three-Phase Power Analyzer, Assembly On Panel or Din RailRodrigo PardoNo ratings yet
- Resistive Transducer NotesDocument3 pagesResistive Transducer NotesqwertyNo ratings yet
- Chapter5 EnergyDocument42 pagesChapter5 EnergywonghjNo ratings yet
- BE1-47N Negative Sequence Voltage Relay: AdvantagesDocument8 pagesBE1-47N Negative Sequence Voltage Relay: AdvantagesEmmanuel EntzanaNo ratings yet
- @bohring Bot 15 02 2024 SR Super60 Jee Adv2021 P2 GTA 16 Key &Document18 pages@bohring Bot 15 02 2024 SR Super60 Jee Adv2021 P2 GTA 16 Key &vaibhav.singh.theking88No ratings yet
- RFF Ib 19 04 - 4Document8 pagesRFF Ib 19 04 - 4dm mNo ratings yet
- Leader DLP Unit Test-04 03 Sept QPDocument48 pagesLeader DLP Unit Test-04 03 Sept QPSonamNo ratings yet
- Module 1 Backbencher - ClubDocument23 pagesModule 1 Backbencher - Clubsamanth0404No ratings yet
- Diffusion CoefficientDocument5 pagesDiffusion CoefficientAbhishek Kumar0% (1)
- DE Midterm ExaminationDocument3 pagesDE Midterm ExaminationHades Vesarius RiegoNo ratings yet
- Advanced DerivativesDocument2 pagesAdvanced DerivativesMelody CairelNo ratings yet
- 11 2 Center of GravityDocument18 pages11 2 Center of GravityluisynonNo ratings yet
- CAMSCODocument12 pagesCAMSCOraulNo ratings yet
- 6619163561165b001828a08a - ## - Chapter 01 - Electric Charges and Fields Study ModuleDocument55 pages6619163561165b001828a08a - ## - Chapter 01 - Electric Charges and Fields Study Modulegaganbanshiwal6No ratings yet