Professional Documents
Culture Documents
Assignment No 4
Uploaded by
sharad0 ratings0% found this document useful (0 votes)
9 views1 pageThis document contains assignment questions about phase diagrams and copper-nickel alloys. Question 1 asks about heating a 70% nickel-30% copper alloy and determining (a) the temperature of first liquid formation, (b) the composition of the first liquid, (c) the temperature of complete melting, and (d) the composition of the last solid. Question 2 asks if an alloy can exist in equilibrium with a 37% nickel-63% copper solid phase and a 20% nickel-80% copper liquid phase, and if so what the approximate temperature would be.
Original Description:
Original Title
Assignment No 4.docx
Copyright
© © All Rights Reserved
Available Formats
DOCX, PDF, TXT or read online from Scribd
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentThis document contains assignment questions about phase diagrams and copper-nickel alloys. Question 1 asks about heating a 70% nickel-30% copper alloy and determining (a) the temperature of first liquid formation, (b) the composition of the first liquid, (c) the temperature of complete melting, and (d) the composition of the last solid. Question 2 asks if an alloy can exist in equilibrium with a 37% nickel-63% copper solid phase and a 20% nickel-80% copper liquid phase, and if so what the approximate temperature would be.
Copyright:
© All Rights Reserved
Available Formats
Download as DOCX, PDF, TXT or read online from Scribd
0 ratings0% found this document useful (0 votes)
9 views1 pageAssignment No 4
Uploaded by
sharadThis document contains assignment questions about phase diagrams and copper-nickel alloys. Question 1 asks about heating a 70% nickel-30% copper alloy and determining (a) the temperature of first liquid formation, (b) the composition of the first liquid, (c) the temperature of complete melting, and (d) the composition of the last solid. Question 2 asks if an alloy can exist in equilibrium with a 37% nickel-63% copper solid phase and a 20% nickel-80% copper liquid phase, and if so what the approximate temperature would be.
Copyright:
© All Rights Reserved
Available Formats
Download as DOCX, PDF, TXT or read online from Scribd
You are on page 1of 1
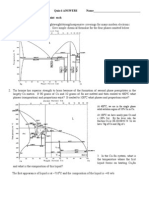
Assignment No: 4
NOTE: Explain using Phase Diagram.
Q1. A copper-nickel alloy of composition 70 wt% Ni-30 wt% Cu is slowly heated
From a temperature of 1300°C.
(a) At what temperature does the first liquid phase form?
(b) What is the composition of this liquid phase?
(c) At what temperature does complete melting of the alloy occur?
(d) What is the composition of the last solid remaining prior to complete melting?
Q2. Is it possible to have a copper-nickel alloy that, at equilibrium, consists of an
α phase of composition 37 wt% Ni-63 wt% Cu, and also a liquid phase of composition 20 wt
% Ni-80 wt% Cu? If so, what will be the approximate temperature of the alloy? If this is not
possible, explain why.
You might also like
- Chapter N9 Proplems HW - Ch9Document1 pageChapter N9 Proplems HW - Ch9xuantoni1112003No ratings yet
- HW 8 TrustedDocument20 pagesHW 8 TrustedEldeniz AliyevNo ratings yet
- Assignment 4 Phase DiagramDocument4 pagesAssignment 4 Phase DiagramAhmedAhmedNo ratings yet
- Assignment PhaseDiaDocument5 pagesAssignment PhaseDiaAnshu Kumar GuptaNo ratings yet
- Soal Diagram FasaDocument1 pageSoal Diagram FasaDecy Putri EfhanaNo ratings yet
- CH 09Document8 pagesCH 09Antoinette ChuaNo ratings yet
- Tutorial 2 HelperDocument3 pagesTutorial 2 HelperDedy SaputraNo ratings yet
- 9.1. Consider T-WPS OfficeDocument4 pages9.1. Consider T-WPS OfficeTia AgustinNo ratings yet
- CH 10Document9 pagesCH 1000gp1200r100% (1)
- Tutorial - Phase Transformations 2 - SolutionsDocument2 pagesTutorial - Phase Transformations 2 - SolutionsNaveen MeenaNo ratings yet
- Assignment 8 SolutionDocument6 pagesAssignment 8 SolutionBrishen Hawkins100% (1)
- Closed-Book Practice-Ch 09 (2017!08!07)Document9 pagesClosed-Book Practice-Ch 09 (2017!08!07)Juan100% (1)
- Homework 5Document4 pagesHomework 5Ferhat PeynirciNo ratings yet
- HW5-Phase Diagram-2023Document5 pagesHW5-Phase Diagram-2023nanniedefiNo ratings yet
- ENSC 3313, Fall 2012 Quiz 6 Answers Name - Answer Any 4 Questions Only, 1 Point EachDocument2 pagesENSC 3313, Fall 2012 Quiz 6 Answers Name - Answer Any 4 Questions Only, 1 Point EachterratempestNo ratings yet
- Practica 2 - Cap. 2 Diagrama de FasesDocument4 pagesPractica 2 - Cap. 2 Diagrama de FasessulayNo ratings yet
- Đề 2Document1 pageĐề 2Phạm Minh CườngNo ratings yet
- Engineering Materials - Manufacturing Engineering and TechnologyDocument12 pagesEngineering Materials - Manufacturing Engineering and Technologyhans vNo ratings yet
- Sheet 2 (Material)Document6 pagesSheet 2 (Material)Omar AssalNo ratings yet
- PSet 8 FL13Document5 pagesPSet 8 FL13cacer009No ratings yet
- Phase Diagram - : Dr. Aneela WakeelDocument21 pagesPhase Diagram - : Dr. Aneela WakeelHassan KhanNo ratings yet
- DMCB 2423 - Tutorial CHPT 9 - AnswerDocument6 pagesDMCB 2423 - Tutorial CHPT 9 - Answerzul hilmiNo ratings yet
- 9 SolutionsDocument31 pages9 SolutionsLaurertan TavaresNo ratings yet
- Week 11Document12 pagesWeek 11lduran_63No ratings yet
- Tutorial Problems 5Document8 pagesTutorial Problems 5Majak MarialNo ratings yet
- Assignment 1: DEADLINE: Monday, 17 January 2022Document10 pagesAssignment 1: DEADLINE: Monday, 17 January 2022Syafiqah RusdiNo ratings yet
- Che 3330 - Spring 2012 HW 5Document5 pagesChe 3330 - Spring 2012 HW 5Brett CasserlyNo ratings yet
- Phase DiagramDocument47 pagesPhase DiagramMuhammed MansNo ratings yet
- 02 Phase Diagram of AlloysDocument60 pages02 Phase Diagram of AlloysBudi HerwantoNo ratings yet
- Assignment 2Document1 pageAssignment 2Anonymous toFx4nRNo ratings yet
- Assignment 7 SolutionsDocument11 pagesAssignment 7 SolutionsIsaiah Qe LiewNo ratings yet
- Sheet 5 - Phase Diagrams IIDocument2 pagesSheet 5 - Phase Diagrams IIroro98syamNo ratings yet
- Tutorial SolutionDocument8 pagesTutorial SolutionBukti NegalNo ratings yet
- Recitation 2 QuestionsDocument14 pagesRecitation 2 QuestionsfzfwsbyxrhNo ratings yet
- HW 9 2011 SolutionsDocument15 pagesHW 9 2011 SolutionsParamesh Anugu100% (1)
- Composition Solidus Temperature Liquidus Temperature: (WT% Si) (°C) (°C)Document7 pagesComposition Solidus Temperature Liquidus Temperature: (WT% Si) (°C) (°C)Muhammad Ibkar YusranNo ratings yet
- Important Terms and Concepts: ReferencesDocument7 pagesImportant Terms and Concepts: ReferencesABDELRHMAN ALINo ratings yet
- Eng Mat Chapter 4Document126 pagesEng Mat Chapter 4VC Chua Yee LeongNo ratings yet
- Ch2 MCQ PDFDocument6 pagesCh2 MCQ PDFPratibha BhondeNo ratings yet
- Assignment # 1 Metallurgy (ME-410-B) VIII Semester (B. Tech Mechanical Engg.)Document3 pagesAssignment # 1 Metallurgy (ME-410-B) VIII Semester (B. Tech Mechanical Engg.)NourTarekHassenNo ratings yet
- Chemistry TestDocument1 pageChemistry TestL.ABHISHEK KUMARNo ratings yet
- Mse230 E2 Practice SolDocument20 pagesMse230 E2 Practice SolAkshit PandeyNo ratings yet
- UEME 1122 Material Science Tutorial 1Document2 pagesUEME 1122 Material Science Tutorial 1JamesNo ratings yet
- Chemistry TestDocument2 pagesChemistry TestL.ABHISHEK KUMARNo ratings yet
- TDF Q - Assignment Week2Document10 pagesTDF Q - Assignment Week2Ayu Sekar TunjungNo ratings yet
- Chapter 9: Phase Diagrams: Issues To Address..Document41 pagesChapter 9: Phase Diagrams: Issues To Address..Faiz AkhtarNo ratings yet
- CH3 Questions: Fig. 10-20 Unary Phase Diagram For Carbon. (Region For Diamond Formation Is Shown With A Dotted Line)Document4 pagesCH3 Questions: Fig. 10-20 Unary Phase Diagram For Carbon. (Region For Diamond Formation Is Shown With A Dotted Line)Mohammed IbrahimNo ratings yet
- Chapter 4-Phase DiagramDocument16 pagesChapter 4-Phase Diagramtky96No ratings yet
- Chapter 4Document19 pagesChapter 4Diệu ThiệnNo ratings yet
- Phase Diagram: Development of MicrostructureDocument9 pagesPhase Diagram: Development of Microstructuresenthilkumar tsNo ratings yet
- TYPD ExercisesDocument10 pagesTYPD ExercisesConstance Lynn'da GNo ratings yet
- HW SolutionsDocument16 pagesHW SolutionsoerbilNo ratings yet
- I. MULTIPLE CHOICE QUESTIONS (7.5 Marks/15 Questions 0.5 Marks Per Question)Document5 pagesI. MULTIPLE CHOICE QUESTIONS (7.5 Marks/15 Questions 0.5 Marks Per Question)Đỗ ĐăngNo ratings yet
- Tugas LiburDocument5 pagesTugas LiburAnonymous AjrDxGNo ratings yet
- Ans GP-Spec Heat WSDocument4 pagesAns GP-Spec Heat WSOthello64No ratings yet
- Phase Diagram Exercises - Worked Answers - CorrectedDocument14 pagesPhase Diagram Exercises - Worked Answers - CorrectedHayden Reeves75% (4)
- Chemistry Olympiad 2015 Tutorial Worksheet (ACJC Prelims 2014/P3/2 (B), (C) )Document5 pagesChemistry Olympiad 2015 Tutorial Worksheet (ACJC Prelims 2014/P3/2 (B), (C) )Timothy HandokoNo ratings yet
- Tutorial 8Document2 pagesTutorial 8Nbl KlfNo ratings yet
- Solution Assignment Ch9Document5 pagesSolution Assignment Ch9Julian GarcíaNo ratings yet
- Module 2 - 11-02-2020 Phase DiagramDocument57 pagesModule 2 - 11-02-2020 Phase DiagramsharadNo ratings yet
- Sardar Patel College of EngineeringDocument2 pagesSardar Patel College of EngineeringsharadNo ratings yet
- Lighting System of AutomobileDocument20 pagesLighting System of Automobilesharad100% (2)
- Energy Requirement - of India and The World General Concepts & Various Forms of Renewable EnergyDocument50 pagesEnergy Requirement - of India and The World General Concepts & Various Forms of Renewable EnergysharadNo ratings yet
- Steering SystemDocument57 pagesSteering Systemsharad100% (2)
- Pressure Cookers and Balloons: TL DR (Too Long Didn't Read)Document3 pagesPressure Cookers and Balloons: TL DR (Too Long Didn't Read)sharadNo ratings yet