Professional Documents
Culture Documents
Different Ways of Expression of R
Uploaded by
Arbaz ChilwanOriginal Title
Copyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
Different Ways of Expression of R
Uploaded by
Arbaz ChilwanCopyright:
Available Formats
What is gas constant R?
The gas constant is a physical constant denoted by R and is expressed in terms of units of energy per temperature
increment per mole. It is also known as Ideal gas constant or molar gas constant or universal gas constant. The
constant is also a combination of the constants from Boyle's law, Charles's law, Avogadro's law, and Gay-Lussac's law.
It is a physical constant that is featured in many fundamental equations in the physical sciences, such as the ideal gas
law, the Arrhenius equation, and the Nernst equation. Physically, the gas constant is the constant of proportionality
that relates the energy scale in physics to the temperature scale, when a mole of particles at the stated temperature
is being considered.
Gas constant R value can be given as, R = 8.3144598(48) J⋅mol−1⋅K−1
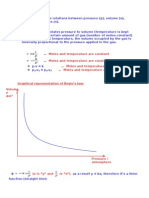
Different ways of expression of R
R=PVnT
Where,
P is the pressure.
V is the volume,
n is the number of moles of the given substance
T is the temperature.
Thus, writing pressure as force per unit area we can derive the dimensional expression for R as-
R=(force/area)×(volume)(amount)×(temperature)
Area and Volume can be expressed in terms of length as volume=(length)^3 and area = (length)^2
Thus we get-
R=(force/length^2)×(length)^3(amount)×(temperature)=(force)×(length)(amount)×(temperature)
Since force times length is work we get-
R=Work x Amount × Temperature
Thus gas constant can be interpreted as work per degree per mole.
Derivation of Gas Constant, R
Let us take any gas:
1 mole of Argon = 39.948 g Ar = 22.4 L Ar @ STP
STP = Standard temperature (0oC or 273 K) & Standard Pressure = 1 atmosphere
Assume the gas acts ideally. Then PV = nRT
Solve for R:
Substitute into equation:
Solve: R = 0.0821 atm L / mol K
If R is needed in units of pressure (kPa):
= 8.314 kPa L / mol K
If R is needed in units of pressure (mm Hg)
= 62.396 mm Hg L / mol K
R = 0.0821 atm L / mol K.
Gas Constant In Different Units
The gas constant has many applications. It is expressed in many units. Some of the gas constant value in different
units are listed below-
Gas Constant Value Units
8.3144598(48) J⋅K−1⋅mol−1
8.3144598(48)×103 amu.m2.s-2.K-1
8.3144598(48)×10−2 L.bar.K-1.mol-1
8.3144598(48) m3.Pa.K-1.mol-1
62.363577(36) L.Torr.K-1.mol-1
1.9872036(11)×10−3 kcal.K-1.mol-1
8.2057338(47)×10−5 m3.atm.K-1.mol-1
0.082057338(47) L.atm.K-1.mol-1
R Constant For Atm
You might also like
- A Modern Course in Statistical PhysicsFrom EverandA Modern Course in Statistical PhysicsRating: 3.5 out of 5 stars3.5/5 (2)
- Gas Laws (Schools - Nbisd.org-Page-Open-16436-0-Ideal PDFDocument14 pagesGas Laws (Schools - Nbisd.org-Page-Open-16436-0-Ideal PDFbinzNo ratings yet
- Chapter 4 States of Matter 2021Document24 pagesChapter 4 States of Matter 2021suh mey chongNo ratings yet
- Notes Solutions Chapter 05Document22 pagesNotes Solutions Chapter 05steveislaryNo ratings yet
- Topic 11-4 Ideal GasDocument3 pagesTopic 11-4 Ideal GasHinata CosaNo ratings yet
- Ideal Gas LawDocument4 pagesIdeal Gas LawAbdul RaufNo ratings yet
- Revision Notes On Kinetic Theory of GasesDocument6 pagesRevision Notes On Kinetic Theory of Gasesvermarashmi2780No ratings yet
- CHEM103 LW4 AnnotatedDocument25 pagesCHEM103 LW4 AnnotatedOmar MatarNo ratings yet
- Gas Laws & ConversionsDocument8 pagesGas Laws & Conversionsvarun283100% (1)
- Class-11 Chemistry Chapter-5 States of Matter Part-IIDocument7 pagesClass-11 Chemistry Chapter-5 States of Matter Part-IINevin ShajiNo ratings yet
- Study Guide Gas LawsDocument3 pagesStudy Guide Gas LawsAdamNo ratings yet
- Ideal Gas Law LectureDocument46 pagesIdeal Gas Law LectureIkooyyNo ratings yet
- Gas PropertiesDocument54 pagesGas PropertiesAli AhmedNo ratings yet
- Topic 4 States of MatterDocument43 pagesTopic 4 States of MatterJowyn SeetNo ratings yet
- Ideal Gas LawDocument10 pagesIdeal Gas Lawchaotictrust28No ratings yet
- Chem 105-8, Sections 8.1, 8.2, 8.5 NotesDocument36 pagesChem 105-8, Sections 8.1, 8.2, 8.5 NotesKhang TrầnNo ratings yet
- Gas PropertiesDocument9 pagesGas PropertiesReza Gustarani DaneswariNo ratings yet
- M17 Wolf57139 03 Se C17Document24 pagesM17 Wolf57139 03 Se C17c.s.kalkmanNo ratings yet
- CH 10 Gases StudentDocument48 pagesCH 10 Gases StudentTrọng NguyễnNo ratings yet
- Ch01-Slides-1 Gas LawsDocument60 pagesCh01-Slides-1 Gas LawsDoreen BenezethNo ratings yet
- Chem 181 Chemistry of GasesDocument15 pagesChem 181 Chemistry of GasesJoey PooleNo ratings yet
- TutorialDocument9 pagesTutorialaravindNo ratings yet
- Learning About: The Ideal Gas Law: Read The Textbook, Pages 383 - 385, and Answer The Following QuestionsDocument2 pagesLearning About: The Ideal Gas Law: Read The Textbook, Pages 383 - 385, and Answer The Following Questionscamilo atiluaNo ratings yet
- Ideal Gas Law ProblemsDocument4 pagesIdeal Gas Law ProblemsJopie Aranda0% (1)
- Investigative Activity of Ideal Gas LawDocument7 pagesInvestigative Activity of Ideal Gas LawShaina AdralesNo ratings yet
- Meterological ParametersDocument47 pagesMeterological ParametersRDWSD SedamNo ratings yet
- Kinetic Theory of Gases CH13Document17 pagesKinetic Theory of Gases CH13Rishab SharmaNo ratings yet
- Ideal Gas Law Derivation For The Equations of The Ideal Gas LawDocument4 pagesIdeal Gas Law Derivation For The Equations of The Ideal Gas LawIan Kirk VillanuevaNo ratings yet
- 1 Wiki Phys Gas ConstantDocument6 pages1 Wiki Phys Gas ConstantTari PuspitaNo ratings yet
- Module 3 Ideal Gases and Ideal Gas LawDocument12 pagesModule 3 Ideal Gases and Ideal Gas LawHazel AdoNo ratings yet
- 1-Ideal Gas LectureDocument60 pages1-Ideal Gas LecturemahmoudNo ratings yet
- Chapter 9 Gases After-Lecture-NoteDocument30 pagesChapter 9 Gases After-Lecture-NoteElvis ChanNo ratings yet
- Bab 1 SolutionsDocument37 pagesBab 1 SolutionsNgọc HuyềnNo ratings yet
- CHM 121 - Lecture Note 7 - Kinetic Theory of Gases, Gas Laws, EquationsDocument35 pagesCHM 121 - Lecture Note 7 - Kinetic Theory of Gases, Gas Laws, Equationssomide kayodeNo ratings yet
- Physical Chemistry Chapter 2 ProblemsDocument37 pagesPhysical Chemistry Chapter 2 ProblemsS. GreenNo ratings yet
- Properties of Gases - 220308 - 154934Document28 pagesProperties of Gases - 220308 - 154934Dhruvi PadmaniNo ratings yet
- ME REVIEW ThermodynamicsDocument65 pagesME REVIEW ThermodynamicsKhate ÜüNo ratings yet
- Ideal Gas Equation of StateDocument18 pagesIdeal Gas Equation of StateAditya SuryawanshiNo ratings yet
- Fizik Nota9Document17 pagesFizik Nota9Dyatie DkNo ratings yet
- Gases Tutorial 2Document4 pagesGases Tutorial 2Idil WarsameNo ratings yet
- Test Supplement 13Document16 pagesTest Supplement 13jimmys45No ratings yet
- MARTINEZ Ideal Gas and Polytropic ProblemDocument25 pagesMARTINEZ Ideal Gas and Polytropic Problemyeng botz0% (1)
- 01 1stlaw ExessolsDocument29 pages01 1stlaw Exessolsblanca.pegueraNo ratings yet
- GASESDocument101 pagesGASESdwyquishNo ratings yet
- Lecture 5 GasesDocument78 pagesLecture 5 GasesHiep NguyenNo ratings yet
- 18 - 70 Heppy Yessya PutriDocument6 pages18 - 70 Heppy Yessya PutriHeppy YessyaNo ratings yet
- CHM 101 Lecture Note-Gas LawsDocument11 pagesCHM 101 Lecture Note-Gas LawsMichael DanielsNo ratings yet
- 11 GasesDocument17 pages11 Gasespuja ritongaNo ratings yet
- RT PV: Chem151 (Kua)Document8 pagesRT PV: Chem151 (Kua)Pink PigNo ratings yet
- Chemistry - Mccord - Exam 1Document9 pagesChemistry - Mccord - Exam 1Miguel MartinezNo ratings yet
- Gas Laws:: P V K VDocument18 pagesGas Laws:: P V K VFarah Zu'biNo ratings yet
- Avogadro's Law: Examples1Document11 pagesAvogadro's Law: Examples1liennev02No ratings yet
- Phy Kinetic Theory of GasesDocument16 pagesPhy Kinetic Theory of GasesSachinNo ratings yet
- 5.0 States of MatterDocument106 pages5.0 States of MatterTasya KassimNo ratings yet
- Gas Law: (Continuation)Document33 pagesGas Law: (Continuation)Ghienel Surla NaguitNo ratings yet
- Tutorial Q1Document2 pagesTutorial Q1Mihai MirceaNo ratings yet
- 1 23 Gas Calculations PDFDocument6 pages1 23 Gas Calculations PDFschool adressNo ratings yet
- Behaviour of Gases - PDFDocument7 pagesBehaviour of Gases - PDFNasih AhmadNo ratings yet
- Chapter 5 Lecture NotesDocument8 pagesChapter 5 Lecture NotesAhmad KamalNo ratings yet
- BK Dutta Heat Transfer 3pdf PDF FreeDocument272 pagesBK Dutta Heat Transfer 3pdf PDF FreeArbaz ChilwanNo ratings yet
- Petro - Google Drive 2Document1 pagePetro - Google Drive 2Arbaz ChilwanNo ratings yet
- Frydendal PHD Thesis PDFDocument234 pagesFrydendal PHD Thesis PDFArbaz ChilwanNo ratings yet
- Resistor ChartsDocument5 pagesResistor ChartsmarlonfatnetzeronetNo ratings yet
- 5 Most Popular Inspection Techniques For The Oil and Gas IndustryDocument1 page5 Most Popular Inspection Techniques For The Oil and Gas IndustryArbaz ChilwanNo ratings yet
- Chapter3 ImppppppppppppppppppppppDocument52 pagesChapter3 ImppppppppppppppppppppppdhavaleshNo ratings yet
- 1.2 CTDocument58 pages1.2 CTUditChoudharyNo ratings yet
- H2 ReportDocument5 pagesH2 ReportArbaz ChilwanNo ratings yet
- The AICHE Student Pocket HandbookDocument68 pagesThe AICHE Student Pocket Handbookpapaas100% (1)
- Heating SystemDocument5 pagesHeating SystemPiyush MoradiyaNo ratings yet
- h2 Production Roadmap 2009Document134 pagesh2 Production Roadmap 2009Arbaz ChilwanNo ratings yet
- Engineering Thought Intelligence Fictional Intellectual Behave Exercise 1 ComputationDocument4 pagesEngineering Thought Intelligence Fictional Intellectual Behave Exercise 1 Computationposgas aceNo ratings yet
- 30 Tips For Indesign Users enDocument38 pages30 Tips For Indesign Users enMoo MNo ratings yet
- Word Formation - Open Cloze Exercises Unit 1Document2 pagesWord Formation - Open Cloze Exercises Unit 1Fatima Luz RuizNo ratings yet
- Chapter 2 PDFDocument21 pagesChapter 2 PDFMahdi BanjakNo ratings yet
- Lesson From Seveso ItalyDocument48 pagesLesson From Seveso ItalyBilal ZamanNo ratings yet
- Basic Science AssigDocument5 pagesBasic Science AssigdomromeoNo ratings yet
- MCR3U Unit #1 NotesDocument12 pagesMCR3U Unit #1 NotespersonNo ratings yet
- Illness Narratives - Positioned IdentitiesDocument35 pagesIllness Narratives - Positioned IdentitiesAnaNo ratings yet
- Mendelian & Modern Genetics: General Biology 2Document51 pagesMendelian & Modern Genetics: General Biology 2sannsannNo ratings yet
- EKJERP IPPF Document Eng v1.2 250819Document63 pagesEKJERP IPPF Document Eng v1.2 250819ahmad yaniNo ratings yet
- Test - Reading Explorer 3 8A - QuizletDocument4 pagesTest - Reading Explorer 3 8A - QuizletTatjana Modea SibulNo ratings yet
- Product Data Sheet: Product Description Technical SpecificationDocument1 pageProduct Data Sheet: Product Description Technical SpecificationYASHICA VAITTIANATHANNo ratings yet
- Tata CRMDocument39 pagesTata CRMRoger Brown100% (1)
- ITEM ANALYSIS - MATH 9 EmeraldDocument5 pagesITEM ANALYSIS - MATH 9 EmeraldLiz Rabaya IntiaNo ratings yet
- Free Downloads: Type Matters!Document3 pagesFree Downloads: Type Matters!Shinko Art Studio9% (11)
- Practice For Group Work Week 2 Case 2.1 - Assigning Plants To Products (Better Products Company)Document2 pagesPractice For Group Work Week 2 Case 2.1 - Assigning Plants To Products (Better Products Company)Thu TrangNo ratings yet
- VERITAS Cluster Server For UNIX, Fundamentals: Lesson 6 VCS Configuration MethodsDocument50 pagesVERITAS Cluster Server For UNIX, Fundamentals: Lesson 6 VCS Configuration MethodsNirmal AsaithambiNo ratings yet
- Participatory Irrigation ManagementDocument11 pagesParticipatory Irrigation ManagementSidNo ratings yet
- Split Valuation SAPDocument7 pagesSplit Valuation SAPPramod ShettyNo ratings yet
- Seasons and WeatherDocument2 pagesSeasons and WeatherОлінька СтецюкNo ratings yet
- PCS-9613S X DataSheet en Overseas General X R1.10Document57 pagesPCS-9613S X DataSheet en Overseas General X R1.10parveen115No ratings yet
- 003 Users Manuel of Safir 2016 - MechanicalDocument74 pages003 Users Manuel of Safir 2016 - Mechanicalenrico_britaiNo ratings yet
- Edit Resume - My Perfect Resume 1Document1 pageEdit Resume - My Perfect Resume 1Ferid MemmedzadeNo ratings yet
- LEPTOSPIRADocument31 pagesLEPTOSPIRADinar NastitiNo ratings yet
- Project Work Class 11Document17 pagesProject Work Class 11DipeshNo ratings yet
- Sciencedirect: Development of Battery Monitoring System in Smart Microgrid Based On Internet of Things (Iot)Document6 pagesSciencedirect: Development of Battery Monitoring System in Smart Microgrid Based On Internet of Things (Iot)KiranNo ratings yet
- Mathematics 9 DLPDocument10 pagesMathematics 9 DLPAljohaila GulamNo ratings yet
- Radiation Protection in Dental RadiologyDocument52 pagesRadiation Protection in Dental Radiologyivan dario ardila martinezNo ratings yet
- Study of PomeloDocument30 pagesStudy of PomeloKyle Cabusbusan75% (4)
- Aptitude Tests For Job ApplicantsDocument30 pagesAptitude Tests For Job ApplicantsMboowa YahayaNo ratings yet