Professional Documents
Culture Documents
Dracunculiasis (Guinea Worm Disease) : Review Synthèse
Uploaded by
JeromeOriginal Title
Copyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
Dracunculiasis (Guinea Worm Disease) : Review Synthèse
Uploaded by
JeromeCopyright:
Available Formats
Review
Synthèse
Dracunculiasis (guinea worm disease)
Chris Greenaway
Abstract The guinea worm and its life cycle
DRACUNCULIASIS (GUINEA WORM DISEASE) IS A PARASITIC disease that is The nematode D. medinensis belongs to the order of
limited to remote, rural villages in 13 sub-Saharan African coun- Spirurida, which are tissue parasites that produce eggs
tries that do not have access to safe drinking water. It is one the containing larvae or release free larvae and that require
next diseases targeted for eradication by the World Health Orga- arthropods as intermediate hosts. The best-known exam-
nization. Guinea worm disease is transmitted by drinking water
ples of this order are the filariae, which include the impor-
containing copepods (water fleas) that are infected with Dracun-
tant human parasites Wuchereria bancrofti and Brugia
culiasis medinensis larvae. One year after human ingestion of in-
fected water a female adult worm emerges, typically from a lower
malayi (causes of elephantiasis), Onchocerca volvulus (cause
extremity, producing painful ulcers that can impair mobility for of river blindness) and Loa loa (the eye worm). The mature
up to several weeks. This disease occurs annually when agricul- female guinea worm is one of the longest nematodes, mea-
tural activities are at their peak. Large proportions of economi- suring up to 1 m in length, but is only 1–2 mm thick.
cally productive individuals of a village are usually affected si- There is no known animal reservoir of infection (even
multaneously, resulting in decreased agricultural productivity and though this has not been conclusively disproved), which
economic hardship. Eradication of guinea worm disease depends makes eradication much more probable if safe drinking
on prevention, as there is no effective treatment or vaccine. Since water can be ensured.
1986, there has been a 98% reduction in guinea worm disease A summary of the guinea worm’s life cycle is depicted
worldwide, achieved primarily through community-based pro- in Fig. 1. People become infected when they drink water
grams. These programs have educated local populations on how containing copepods (water fleas) that harbour infective
to filter drinking water to remove the parasite and how to prevent larvae. The ingested copepods are killed by the digestive
those with ulcers from infecting drinking-water sources. Complete juices of the stomach. The released larvae then move to
eradication will require sustained high-level political, financial the small intestine, where they penetrate the intestinal wall
and community support.
and migrate to the connective tissues of the abdominal
CMAJ 2004;170(4):495-500 wall and the thorax. Male and female larvae mature and
mate 60–90 days after infection. The male worm dies
D
racunculiasis (guinea worm disease), caused by shortly after mating, and the female matures over the sub-
the nematode parasite Dracunculiasis medinensis, sequent 10–14 months, slowly migrates to the surface of
is a painful, disabling disease of impoverished, the body and emerges through the skin. When affected
rural villagers in 13 sub-Saharan African countries that body parts are submerged in water, the female worm re-
do not have access to safe drinking water. The disease has leases larvae, which are ingested by copepods, thus com-
a low mortality rate but causes an enormous amount of pleting the life cycle.2
morbidity and is often economically devastating for af-
fected villages.1–7 The Global Dracunculiasis Eradication Epidemiology
Campaign, which began in 1981, has reduced the number
of afflicted people by 98%. In 1986 there were 3.5 mil- The seasonal variation of guinea worm disease is closely
lion cases in 20 different countries in Asia and Africa. In related to rainfall. In arid areas, transmission usually co-
2002 fewer than 55 000 cases were reported, from 13 incides with the rainy season, when surface water is avail-
African countries. The greatest burden of guinea worm able. In wet areas, transmission is most intense in the dry
disease today occurs in Sudan, Ghana and Nigeria. These season, when sources of drinking water are limited. Stag-
3 countries together account for 93% of all cases world- nant sources of drinking water, such as ponds, cisterns,
wide, with Sudan reporting 73% of the cases.8 The goal pools in dried up riverbeds, temporary hand-dug wells and
of the Global Dracunculiasis Eradication Campaign is to step-wells, commonly harbour populations of copepods
stop transmission of guinea worm disease outside of Su- and are the usual sites where infection is transmitted.2,9
dan by the end of 2004, and within 4–6 years after peace Guinea worm disease is limited to villages that depend
and stability have been achieved in the disease-endemic on contaminated sources of drinking water. In disease-
areas of Sudan. endemic villages the prevalence ranges from 15% to
CMAJ • FEB. 17, 2004; 170 (4) 495
© 2004 Canadian Medical Association or its licensors
Greenaway
70%.3,7,10–12 The incidence of disease varies by sex, age and Clinical manifestations
occupation, and by village and country. This variation is
likely due to how and where people of different ages and Infected people remain assymptomatic for about
sex obtain their drinking water.2,7 1 year after infection, at which time the mature female
Only a few cases have been imported to the Americas, worm approaches the skin and forms a papule in the der-
although all countries are required to report all cases to mis (Fig. 2) that is painful because of the host reaction.
the World Health Organization (WHO). Because of the The initial blister is accompanied by redness and indura-
1-year incubation period, these few cases have occurred in tion and is preceded by a slight fever, an urticarial rash
recent immigrants or refugees from Sudan or other dis- with intense pruritus, and systemic symptoms of nausea,
ease-endemic countries.13 vomiting, diarrhea and dizziness. Over the course of up
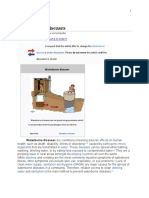
1. Human drinks unfiltered
water containing copepods
(water fleas) harbouring
infective third-stage
guinea worm larvae
Stomach
2. Gastric juices kill the
copepods; the released larvae
penetrate the host's stomach
and intestinal wall and enter
7. After 2 weeks, and 2 the abdominal cavity and
molts within the water fleas, retroperitoneal space
the larvae develop into
infective larvae. Ingestion of
the infected water fleas by
humans completes the life cycle
3. Fertilized female worms
migrate toward the skin surface,
usually on the lower extremities
(Male worms die soon after mating)
4. A year after infection,
the female worm induces
a painful blister and
begins to emerge
through the skin
6. The immature larvae are
ingested by water fleas
5. When the lesion comes into contact with water, the
Chesley Sheppard
emerging worm releases larvae into the water source.
Free-living larvae survive ony 3 days until they find a host.
Fig. 1: Life cycle of the guinea worm.
496 JAMC • 17 FÉVR. 2004; 170 (4)
Guinea worm disease
to several days the blister enlarges and ruptures to expose Economic impact
the adult worm (Fig. 3). The pain and systemic symp-
toms decrease with the rupture of the blister, and the Guinea worm disease occurs almost exclusively in iso-
worm usually is manually extruded by winding it onto a lated rural areas. Although rarely fatal, it causes a major
stick over several weeks. economic burden on affected villages. In Nigeria it has
More than 90% of worms emerge from the lower ex- been estimated that infected people lose 100 days of work
tremities, usually below the knees.2,7,11,14 Infected people per year2 and that children are absent from school for 25%
commonly have a few worms emerging at the same time of the school year.6 The cost in lost revenue for the individ-
(1.8 per person on average), with as many as 14 reported in ual and the community can be very high. In a study in
one individual.10,12 Secondary infection of the ulcers occurs Benin, the annual cost of guinea worm disease was esti-
in most cases, because the rural populations have little ac- mated to be 16 000 CFA francs per patient (US$60).5 From
cess to medical care. In addition to local cellulitis, wound a survey of 87 households in southern Nigeria, the esti-
complications may lead to abscess formation, tetanus, sep- mated annual loss in 3 rice-growing states in southern
tic arthritis or systemic sepsis. Infection of joints may sub- Nigeria was US$20 million. The Dogon people of Mali
sequently result in deformities or contractures.2,14 The pro- have referred to guinea worm disease as “the disease of the
longed incapacity per episode of guinea worm disease is empty granary.” 2
due to the emergence of worms from weight-bearing parts The World Bank has estimated that the economic rate
of the body and high rates of secondary infection. Continu- of return on the investment in guinea worm eradication
ing pain for 12–18 months after the emergence of worms will be about 29% per year once the disease is eradicated.
was experienced by 28% of people with guinea worm dis- This rate is based on very conservative estimates of the
ease in one study, and permanent physical impairment in average amount of time infected workers are unable to
the form of “locked” knees or other joints occurred in
0.5% of this population.2,15
During their migration, guinea worms may occasion-
ally end up in aberrant locations such as the pancreas,
lung, periorbital tissue, testes, pericardium and the spinal
cord, causing local compressive symptoms and local ab-
scess formation.14
There is little or no acquired immunity to this disease,
so it is not uncommon for an individual to develop guinea
worm disease several times during their life, and as often
The Carter Center / S. Fitzgerald
as annually.2
Treatment
As the guinea worm emerges through the dermal le-
sion, the affected person pulls it out slowly and carefully
(to minimize inflammation and pain) by winding a few Fig. 2: Foot blister induced by the female guinea worm in a
centimetres of the worm each day onto a stick. This person with dracunculiasis (guinea worm disease).
painful process may take several weeks, as the worm may
be up to 1 m long. The pain can be relieved with wet
compresses on the lesion and the use of an oral analgesic.
The risk of bacterial superinfection can be reduced with
the use of topical antiseptics or antibiotic ointment.16 In
one study the mean duration of disability from guinea
worm disease was 50% shorter among patients who had
been given antibiotics as well as instructions and supplies
to clean and dress their wounds than among those who
did not receive any intervention.3 No anthelminthic med-
ication is effective against the disease, and there is no vac-
cine.2,17 Chippaux18 found that treatment with mebenda-
zole was associated with aberrant migration of worms,
which were more likely than usual to emerge in places
other than the lower limbs. Prevention is the only effec-
tive intervention to reduce the incidence of guinea worm
disease. Fig. 3: Guinea worm emerging from foot ulcer.
CMAJ • FEB. 17, 2004; 170 (4) 497
Greenaway
work.19 The benefit of eradication will be limited almost and Prevention (CDC) proposed that guinea worm disease
exclusively to the villages and countries in which the disease would be the ideal indicator with which to measure the suc-
is endemic. The benefit of guinea worm eradication for the cess of this global initiative. By the mid-1980s, however, it
global community will be the knowledge that it has partici- was evident that it would not be possible to gather all of the
pated in reducing the suffering of some of the world’s most resources needed to achieve the intended goals of the
underprivileged populations and that these populations will IDWSSD. As a result, in 1986 The Carter Center led a
be provided with safe drinking water and community-based movement, in collaboration with the CDC, UNICEF and
health volunteers who are capable of delivering other basic the WHO, to increase advocacy and funding for the Global
health services.20 Dracunculiasis Eradication Campaign. High-level advocacy
has been an important aspect of the campaign, with sus-
Eradication tained support by former US president Jimmy Carter and
2 former African heads of state, Amadou Toumani Touré of
Guinea worm disease has been reported in writings from Mali and General Yakubu Gowon of Nigeria.2,21–23 As the
India, Greece and the Middle East since antiquity, and campaign gained momentum, transmission of guinea worm
dead female worms have been found in 3000-year-old disease decreased in several countries, and in 1991 the
Egyptian mummies. Historically, the disease occurred in World Health Assembly passed a resolution calling for the
Algeria, Egypt, the Gambia, Guinea Conakry, Iraq, Brazil eradication of guinea worm disease by 1995.
and the West Indies but died out spontaneously in these ar- Since the beginning of the Global Dracunculiasis Eradi-
eas. Guinea worm disease was eliminated from Uzbekistan cation Campaign, 7 countries have eradicated the disease,
in 1932 and from southern Iran in 1972 through case iden- including Pakistan (1993), Kenya (1994), India (1996),
tification and containment and through elimination of the Cameroon, Senegal and Yemen (1997) and Chad (1998)23,24
parasite from water reservoirs.2 (Fig. 4). Because of the campaign, the number of reported
The United Nations declared 1981–1990 to be the Inter- cases decreased from 3.5 million cases in 20 countries in
national Drinking Water Supply and Sanitation Decade Asia and Africa in 1986, to fewer than 55 000 cases in 13
(IDWSSD), raising the possibility that guinea worm disease African countries in 2002 (Sudan, 41 493; Ghana, 5606;
could be eradicated by improving the quality of human Nigeria, 3820; Togo, 1479; Mali, 857; Burkino Faso, 580;
drinking water. In 1981 the US Centers for Disease Control Côte d’Ivoire, 266; Niger, 233; Benin, 157; Mauritania, 42;
1993
1996
1998 1997
WHO Collaborating Center for Research, Training and Eradication of Dracunculiasis
1997
1997
1994
Currently disease-endemic (≥ 100 cases)
Currently disease-endemic (< 100 cases)
Formerly disease-endemic
1998 = Last indigenous case reported
Fig. 4: Map of current and former dracunculiasis-endemic countries, 2002.
498 JAMC • 17 FÉVR. 2004; 170 (4)
Guinea worm disease
The successful implementation of these strategies has
led Global Dracunculiasis Eradication Campaign to target
the eradication of guinea worm disease in all countries ex-
cept Sudan by the end of 2004. Sudan has the greatest bur-
den of disease in the world, primarily because a 20-year-
long civil war has made it difficult to implement an
effective eradication program. A program was first estab-
lished in Sudan in 1995 when former president Jimmy
Carter negotiated a successful 4-month “guinea worm
The Carter Center / Emily Howard
cease fire.” All fighting ceased during this period, allowing
health care workers to educate villagers about guinea worm
disease and to disseminate cloth filters in some of the re-
motest areas of the country.21,26,27 This did not have a lasting
effect, however, as the civil war currently prevents access to
affected villages, particularly in the southern part of the
Fig. 5: A Togolese woman strains her family’s drinking water country. It is estimated that at least 5 years will be required
through a cloth filter to prevent them from contracting guinea to eradicate guinea worm disease in Sudan once an effective
worm disease. program is established.21 Continued success of the Global
Dracunculiasis Eradication Campaign will depend on ade-
Ethiopia, 24; and Uganda, 6 [number not reported for quate funding, the ongoing cooperation of affected vil-
Central African Republic]). lagers, continued political support and political stability in
The ultimate success of an eradication program depends affected countries.
not only on favourable biological factors but also on soci-
etal acceptance of the intervention and the political will to Conclusion
carry it out. Guinea worm disease is a good candidate for
eradication for several biological reasons. First, the disease The Global Dracunculiasis Eradication Campaign has
has a limited geographic distribution and the marked sea- been enormously successful over the past 20 years, reflected
sonal occurrence allows for periods of more intensive inter- in a decrease of the global burden of the disease by 98%.
vention. Second, transmission is only from those with clini- This has been achieved through high-level political advo-
cal disease and there is no known animal reservoir. Third, cacy, sustained financial support and community-based
because symptoms develop within 1 year after infection, health interventions implemented by villagers. The chal-
with predictable timing, the disease is easy to recognize and lenge to complete the eradication of guinea worm disease
diagnose, which facilitates identification and containment worldwide remains in the resolution of the long-standing
of active cases. Finally, effective preventive measures such civil war in Sudan followed by the institution of effective
as health education and water filtration are available to pre- prevention programs in that country.
vent transmission.2,20,25
The main strategies of the Global Dracunculiasis Eradi-
cation Campaign have been to identify all communities
with endemic disease transmission, contain all active cases
(thereby preventing contamination of drinking water
sources) and educate the population about methods to avoid
ingestion of contaminated drinking water. These objectives
have been achieved through training volunteer village-based
health workers to educate villagers about the origin of the
disease, its mode of transmission and methods of preven-
tion. Villagers are taught to avoid ingestion of contaminated
water by filtering water through cloth filters (Fig. 5) and are
The Carter Center / Emily Howard
educated about the care and management of people with ac-
tive infections so that they do not contaminate sources of
drinking water (Fig. 6). Vector control through the addition
of Abate (a copecide) monthly to unsafe sources of drinking
water to control the copepod population (the intermediate
host) has also been used. Another important aspect of the
control program has been the provision of monthly reports, Fig. 6: A young man with guinea worm disease receives edu-
based on active surveillance of cases, to the community by cation on the life cycle of the guinea worm and how to pre-
the volunteer village-based health workers.2,21,23,25 vent contamination of drinking-water sources.
CMAJ • FEB. 17, 2004; 170 (4) 499
Greenaway
20. Aylward B, Hennessey KA, Zagaria N, Olive JM, Cochi S. When is a disease
This article has been peer reviewed.
eradicable? 100 years of lessons learned. Am J Public Health 2000;90(10):1515-20.
21. Progress toward global dracunculiasis eradication, June 2002. MMWR Morb
Chris Greenaway is an Assistant Professor in the Department of Medicine, McGill
Mortal Wkly Rep 2002;51(36):810-1.
University, and a staff physician in the Departments of Microbiology and Internal
22. Brieger WR, Otusanya S, Adeniyi JD, Tijani J, Banjoko M. Eradicating
Medicine, Sir Mortimer B. Davis Jewish General Hospital, Montréal, Que. She is a
guinea worm without wells: unrealized hopes of the Water Decade. Health
consultant at the McGill Centre for Tropical Diseases and spent 1 year at the
Policy Plan 1997;12(4):354-62.
Medical Research Council Laboratories in the Gambia, West Africa, doing a re-
23. Hopkins DR, Ruiz-Tiben E, Ruebush TK, Diallo N, Agle A, Withers PC. Dra-
search fellowship.
cunculiasis eradication: delayed, not denied. Am J Trop Med Hyg 2000;62(2):163-8.
Competing interests: None declared. 24. Dracunculiasis. Global surveillance summary, 2000. Wkly Epidemiol Rec 2001;
76(18):133-9.
Acknowledgements: The photographs and map as well as the latest data about the 25. Hopkins DR, Ruiz-Tiben E. Strategies for dracunculiasis eradication. Bull
Global Dracunculiasis Eradication Campaign were provided by The Carter Center World Health Organ 1991;69(5):533-40.
and Emily Howard. Insights and details about the eradication campaign were ob- 26. Hopkins DR, Ruiz-Tiben E, Diallo N, Withers PC, Macquire JH. Dracuncu-
tained through discussions with Dr. Ernesto Ruiz-Tiben, Technical Director, Dra- liasis eradication: and now, Sudan. Am J Trop Med Hyg 2002;67(2):415-22.
cunculiasis Eradication, The Carter Center, drawn from his more than 20-year in- 27. Implementation of health initiatives during a cease-fire — Sudan, 1995 [pub-
volvement with the campaign. Dr. Ruiz-Tiben also reviewed the manuscript. lished erratum in MMWR Morb Mortal Wkly Rep 1995;44(25):479]. MMWR
Morb Mortal Wkly Rep 1995;44(23):433-6.
References
Correspondence to: Dr. Chris Greenaway, Department of
1. Smith GS, Blum D, Huttley SRA, Okeke N, Kirkwood BR, Feachem RG.
Disability from dracunculiasis: effect on mobility. Ann Trop Med Parasitol Microbiology, Sir Mortimer B. Davis Jewish General Hospital,
1989;83(2):151-8. Rm. G-143, 3755 Cote Ste. Catherine Rd., Montréal QC
2. Cairncross S, Muller R, Zagaria N. Dracunculiasis (guinea worm disease) and H3T 1E2; fax 514 340-7578; ca.greenaway@mcgill.ca
the eradication initiative. Clin Microbiol Rev 2002;15(2):223-46.
3. Belcher DW, Wurapa FK, Ward WB, Lourie IM. Guinea worm in southern
Ghana: its epidemiology and impact on agricultural productivity. Am J Trop
Med Hyg 1975;24(2):243-9.
4. Watts SJ, Brieger WR, Yacoob M. Guinea worm: an in-depth study of what
happens to mothers, families and communities. Soc Sci Med 1989;29(9):1043-9.
5. Chippaux JP, Banzou A, Agbede K. Impact social et économique de la dra-
cunculose : une étude longitudinale effectuée dans deux villages du Bénin.
Bull World Health Organ 1992;70(1):73-8.
6. Ilegbodu VA, Kale OO, Wise RA, Christensen BL, Steele JH, Chambers LA.
Impact of guinea worm disease on children in Nigeria. Am J Trop Med Hyg
1986;35(5):962-4.
7. Nwosu ABC, Ifezulike EO, Anaya AO. Endemic dracontiasis in Anambra State
of Nigeria: geographical distribution, clinical features, epidemiology and so-
cio-economic impact of the disease. Ann Trop Med Parasitol 1980;76:187-200.
8. Department of Health and Human Services. Detect every case, contain every worm!
8th meeting of national coordinators held in Kampala [Guinea worm wrap-up no
132]. Atlanta: US Centers for Disease Control and Prevention; 2003 Apr 25.
Available: www.cdc.gov/ncidod/dpd/parasites/guineaworm/wrapup/word132.pdf
(accessed 2004 Jan 12).
9. McMahon JE, Simonsen PE. Filariases. In: Cook G, editor. Manson’s tropical
diseases. London: W.B. Saunders Co; 1996. p. 1358-62.
10. Kale OO. The clinico-epidemiological profile of Guinea worm in the Ibadan
District of Nigeria. Am J Trop Med Hyg 1977;26(2):208-14.
11. Ilegbodu VA, Ilegbodu AE, Wise RA, Christensen BL, Kale OO. Clinical
manifestations, disability and use of folk medicine in dracunculus infection in
Nigeria. J Trop Med Hyg 1991;94(1):35-41.
12. Edungbola LD. Babana Parasitic Diseases Project. II. Prevalence and impact
of dracontiasis in Babana District, Kwara State, Nigeria. Trans R Soc Trop Med
Hyg 1983;77(3):310-5.
13. Imported dracunculiasis — United States, 1995 and 1997. MMWR Morb
Mortal Wkly Rep 1998;47(11):209-11.
14. Ruiz-Tiben E, Gutierrez Y. Dracunculiasis. In: Guerrant RL WD, Weller
PF, editors. Tropical infectious diseases. Principles, pathogens and practice. Vol 2.
Philadelphia: Churchill Livingstone; 1999. p. 903-6.
15. Hours M, Cairncross C. Long-term disability due to guinea worm disease.
Trans R Soc Trop Med Hyg 1994;88(5):559-60.
16. Magnussen P, Yakubu A, Bloch P. The effect of antibiotic and hydrocorti-
sone-containing ointments in preventing secondary infection in guinea worm
disease. Am J Trop Med Hyg 1994;51(6):797-9.
17. Issaka-Tinorgah A, Magnussen P, Bloch P, Yakubu A. Lack of effect of iver-
mectin on prepatent guinea-worm: a single-blind, placebo-controlled trial.
Trans R Soc Trop Med Hyg 1994;88(3):346-8.
18. Chippaux JP. Mebendazole treatment of dracunculiasis. Trans R Soc Trop Med
Hyg 1991;85(2):280.
19. Hopkins DR. The guinea worm eradication effort: lessons for the future.
Emerg Infect Dis 1998;4(3):414-5.
500 JAMC • 17 FÉVR. 2004; 170 (4)
You might also like
- Dracunculus MedinensisDocument59 pagesDracunculus Medinensistriambulo justinNo ratings yet
- 2 - Reducing Guinea Worm ArticleDocument8 pages2 - Reducing Guinea Worm Articleapi-375285021No ratings yet
- Term Paper DraftDocument5 pagesTerm Paper DraftBello SakanauNo ratings yet
- Dracunculiasis: Group 7Document15 pagesDracunculiasis: Group 7FatemaNo ratings yet
- Soil Transmitted Helminths: Dr. Madhusudan Swarnkar Professor, Community Medicine, JMC, JhalawarDocument34 pagesSoil Transmitted Helminths: Dr. Madhusudan Swarnkar Professor, Community Medicine, JMC, Jhalawarmadhusudan swarnkarNo ratings yet
- 2018 SabourinDocument13 pages2018 SabourinEDGAR DANIEL GONZALEZ BAUTISTANo ratings yet
- SCHISTOSOMIASIS Handouts FinalDocument2 pagesSCHISTOSOMIASIS Handouts Finalmaglangitjoannamarie1920No ratings yet
- GUINEA WORM Dracunculus Medinesisi PDFDocument22 pagesGUINEA WORM Dracunculus Medinesisi PDFLesbeth Ann GamotNo ratings yet
- Progress Toward The Eradication of Dracunculiasis (Guinea Worm Disease) : 1994Document3 pagesProgress Toward The Eradication of Dracunculiasis (Guinea Worm Disease) : 1994Birhanu RetaNo ratings yet
- Hookworm Infection: Submitted By: Desarey C. Dela Cruz (BSN-1C) Submitted To: Emerson G. Parcon, RMTDocument3 pagesHookworm Infection: Submitted By: Desarey C. Dela Cruz (BSN-1C) Submitted To: Emerson G. Parcon, RMTcooky maknaeNo ratings yet
- Schistosoma Japonicum: Schistosoma Japonicum or The Oriental Blood Fluke Causes Schistosomiasis Japonica. It IsDocument7 pagesSchistosoma Japonicum: Schistosoma Japonicum or The Oriental Blood Fluke Causes Schistosomiasis Japonica. It IsPau SorianoNo ratings yet
- MLS 223 Clinical Bacteriology Module 6 Unit 2Document28 pagesMLS 223 Clinical Bacteriology Module 6 Unit 2p7pxrdjhwvNo ratings yet
- Dar Et Al-2015-P Columella Resistencia A Calicophoron DaubneyiDocument6 pagesDar Et Al-2015-P Columella Resistencia A Calicophoron Daubneyicba_ssNo ratings yet
- 408 2022 Article 528Document8 pages408 2022 Article 528nathanaellee92No ratings yet
- Intoduction To Guinea WormDocument10 pagesIntoduction To Guinea WormChika JonesNo ratings yet
- Yash Bhargava, Et AlDocument6 pagesYash Bhargava, Et Alamit vishenNo ratings yet
- Paramphistomiasis PDFDocument13 pagesParamphistomiasis PDFSumit Sharma PoudelNo ratings yet
- Cattle Meat Inspection Chapter 2Document35 pagesCattle Meat Inspection Chapter 2hadiminshawi2029No ratings yet
- Chapter 1Document9 pagesChapter 1Leonard allan PerezNo ratings yet
- First Page PDFDocument1 pageFirst Page PDFferhanmu419No ratings yet
- Causal Agents:: Uncinaria Stenocephala) - Occasionally A. Caninum Larvae May Migrate To The Human Intestine, CausingDocument9 pagesCausal Agents:: Uncinaria Stenocephala) - Occasionally A. Caninum Larvae May Migrate To The Human Intestine, CausingJulia WLNo ratings yet
- Siwila 2020Document14 pagesSiwila 2020GTA5ON360No ratings yet
- Health Issues Caused by Water Pollution To Predict Using Logistic Regression ModelDocument3 pagesHealth Issues Caused by Water Pollution To Predict Using Logistic Regression ModelInternational Journal of Innovative Science and Research TechnologyNo ratings yet
- COMMON WORM INFESTATION DrASBDocument54 pagesCOMMON WORM INFESTATION DrASBDr AMAL S BABUNo ratings yet
- ParamphistomiasisDocument13 pagesParamphistomiasisI Gede Putu Alit Anggara PutraNo ratings yet
- Review On Dracunculus Medinensis in DI Khan Region of PakistanDocument9 pagesReview On Dracunculus Medinensis in DI Khan Region of PakistanProf. Dr. Akram Shah, Department of Zoology, UoPNo ratings yet
- BioDocument10 pagesBioapi-401402382No ratings yet
- ASCARISLUMBRICOIDESDocument5 pagesASCARISLUMBRICOIDESIonela VișinescuNo ratings yet
- Dracunculus Medinensis Onchocerca Loa LoaDocument45 pagesDracunculus Medinensis Onchocerca Loa LoaMeenachi Chidambaram100% (1)
- Hookworm RephraseDocument6 pagesHookworm Rephrasecooky maknaeNo ratings yet
- GY339 Environmental Classification of Excreta Related Disease 2017Document58 pagesGY339 Environmental Classification of Excreta Related Disease 2017Lorraine100% (1)
- EJVS - Volume 52 - Issue 3 - Pages 351-360Document10 pagesEJVS - Volume 52 - Issue 3 - Pages 351-360Ervin Juliet, ANo ratings yet
- TP40 1 IntroductionDocument10 pagesTP40 1 Introductionnermeen ahmedNo ratings yet
- Notes - Sporozoa PDFDocument15 pagesNotes - Sporozoa PDFJezzah Mae CañeteNo ratings yet
- Ascaris Lumbricoides: Infective DiagnosticDocument17 pagesAscaris Lumbricoides: Infective DiagnosticRaymundo III DoceNo ratings yet
- ASCARISLUMBRICOIDESDocument5 pagesASCARISLUMBRICOIDESWira NababanNo ratings yet
- Hymenolepis DiminutDocument10 pagesHymenolepis DiminutLois RazonNo ratings yet
- PD FilariasisDocument8 pagesPD FilariasisNarisha SmaradhantiaNo ratings yet
- MedicalentomologyDocument89 pagesMedicalentomologyRajeev NepalNo ratings yet
- Javma-Javma 22 06 0258Document8 pagesJavma-Javma 22 06 0258Insyirah DaudNo ratings yet
- ASCARSISDocument39 pagesASCARSISkajal thakurNo ratings yet
- Health 3Document4 pagesHealth 3daniellexdlNo ratings yet
- Dracunculus MedinensisDocument1 pageDracunculus MedinensisEm KayNo ratings yet
- ROLL NO 30 (Assignment On Zoonotic Importance of Nematodes)Document3 pagesROLL NO 30 (Assignment On Zoonotic Importance of Nematodes)barsha subediNo ratings yet
- Jurnal Int YyyyDocument3 pagesJurnal Int YyyyAnonymous kXH84rVeNo ratings yet
- Dracunculiasis (Guinea Worm Disease) : Eradication Without A Drug or A VaccineDocument29 pagesDracunculiasis (Guinea Worm Disease) : Eradication Without A Drug or A VaccineDina sayunaNo ratings yet
- Https Jmpas - Com Abstract 1132Document9 pagesHttps Jmpas - Com Abstract 1132KabirNo ratings yet
- Filariasis Control ProgramDocument17 pagesFilariasis Control ProgramLharra Cagulada-PostranoNo ratings yet
- A Rare Case of Vulvar Myiasis: Gupta Shivani MS, Mirza Nooren MS, Simlot Anita MS and Meena Kiran MSDocument3 pagesA Rare Case of Vulvar Myiasis: Gupta Shivani MS, Mirza Nooren MS, Simlot Anita MS and Meena Kiran MSshivani guptaNo ratings yet
- Waterborne Diseases: Jump To Navigation Jump To SearchDocument16 pagesWaterborne Diseases: Jump To Navigation Jump To SearchDennis NjorogeNo ratings yet
- "Helminths and SanitationDocument12 pages"Helminths and SanitationSyed Ali Akbar BokhariNo ratings yet
- General Methods: Monitoring Mosquito PopulationsDocument5 pagesGeneral Methods: Monitoring Mosquito PopulationsRamesh KolekarNo ratings yet
- Cestodes (Tapeworms)Document5 pagesCestodes (Tapeworms)hudamNo ratings yet
- FilariasisDocument40 pagesFilariasisAarzoo SainiNo ratings yet
- Epidemiological and Clinical Characteristics of Hookwormrelated Cutaneous Larva MigransDocument9 pagesEpidemiological and Clinical Characteristics of Hookwormrelated Cutaneous Larva MigransShelyAzradNo ratings yet
- Cryptosporidium ParvumDocument7 pagesCryptosporidium ParvumKalebNo ratings yet
- Epidemiology Presentation With Dr. TendongforDocument60 pagesEpidemiology Presentation With Dr. TendongforNjeodoNo ratings yet
- Project in Microbiolog Y: Submitted To: Yolanda Ilagan Ph. D Submitted By: Irish Jane B. Cubillo BSN 2-2Document28 pagesProject in Microbiolog Y: Submitted To: Yolanda Ilagan Ph. D Submitted By: Irish Jane B. Cubillo BSN 2-2Irish Jane Bayle CubilloNo ratings yet
- Research ArticleDocument8 pagesResearch ArticleJeromeNo ratings yet
- Febs 13591Document12 pagesFebs 13591JeromeNo ratings yet
- 1vanrensburg1971 PDFDocument62 pages1vanrensburg1971 PDFJeromeNo ratings yet
- BJD 17896Document8 pagesBJD 17896JeromeNo ratings yet
- DR Axe 5 Essential Oils Proven Bymedical Science PDFDocument7 pagesDR Axe 5 Essential Oils Proven Bymedical Science PDFDubravka Duda100% (1)
- Protocol SummaryDocument4 pagesProtocol Summaryapi-635954562No ratings yet
- Complementary and Alternative Health Care Client Bill of RightsDocument2 pagesComplementary and Alternative Health Care Client Bill of Rightsapi-31121402No ratings yet
- Myringoplasty and TympanoplastyDocument20 pagesMyringoplasty and TympanoplastyAsiya Zaidi100% (1)
- Abnormal PuerperiumDocument23 pagesAbnormal PuerperiumLynee OlvianaNo ratings yet
- Non-Invasive Methods of Penile Lengthening: Fact or Fiction?Document5 pagesNon-Invasive Methods of Penile Lengthening: Fact or Fiction?BJ VeuxNo ratings yet
- The Impact of Workplace Wellness Programs On Employee Health and ProductivityDocument43 pagesThe Impact of Workplace Wellness Programs On Employee Health and ProductivityVishal VishwakarmaNo ratings yet
- Metacarpal FractureDocument45 pagesMetacarpal FractureJinnasit TeeNo ratings yet
- Singapore Red Cross Application FormDocument2 pagesSingapore Red Cross Application FormOPSS NPCCNo ratings yet
- Naranjo AlgorithmDocument3 pagesNaranjo AlgorithmmilkymilkyNo ratings yet
- Agesta Sacred Codes ListDocument22 pagesAgesta Sacred Codes Listmimigogo92% (12)
- Case:: Biddy's Bakery (BB)Document3 pagesCase:: Biddy's Bakery (BB)tehreem chohanNo ratings yet
- Anaphylaxis: Learning ObjectivesDocument4 pagesAnaphylaxis: Learning Objectivesmohamed fahmyNo ratings yet
- Yoana Cardenas: InternshipDocument2 pagesYoana Cardenas: Internshipapi-395953402No ratings yet
- NCP Shoulder DystociaDocument4 pagesNCP Shoulder DystociaTrishia CaroNo ratings yet
- Dry Acide Dessolution Unit FreesenuisDocument59 pagesDry Acide Dessolution Unit FreesenuisokboNo ratings yet
- UCD School of Medicine Undergraduate Prospectus 2022-1Document36 pagesUCD School of Medicine Undergraduate Prospectus 2022-1ARab COMPutersNo ratings yet
- Screw Air Compressor L07-L22: User ManualDocument56 pagesScrew Air Compressor L07-L22: User ManualЯни Хандрулев100% (4)
- Pubmed - Australia - Clinical - brokenAffLastHistoDocument131 pagesPubmed - Australia - Clinical - brokenAffLastHistoHNNo ratings yet
- Cox, D.R. - Donnelly, C.A. - Principles of Applied StatisticsDocument209 pagesCox, D.R. - Donnelly, C.A. - Principles of Applied Statisticstgabi2012No ratings yet
- USAA Tax Exempt Intermediate-Term Fund 2Q '22Document2 pagesUSAA Tax Exempt Intermediate-Term Fund 2Q '22ag rNo ratings yet
- Psychopharma 1Document7 pagesPsychopharma 1Mitchee Zialcita100% (1)
- The Practical Design & Implementation of Small-Sided Conditioned Games (SSCGS)Document1 pageThe Practical Design & Implementation of Small-Sided Conditioned Games (SSCGS)Husan ThapaNo ratings yet
- ACLS (Notes) (Printable)Document2 pagesACLS (Notes) (Printable)mike_germain1172No ratings yet
- C. Stages of Growth and Development - The LearnerDocument13 pagesC. Stages of Growth and Development - The Learner18shar_07No ratings yet
- iHFG Part C SignageDocument5 pagesiHFG Part C SignageSCQA SHEIKHPURANo ratings yet
- VND Openxmlformats-Officedocument Wordprocessingml Document&rendition 1Document45 pagesVND Openxmlformats-Officedocument Wordprocessingml Document&rendition 1sadhana karapeNo ratings yet
- N222 Lecture Notes Sp13Document198 pagesN222 Lecture Notes Sp13Jacinth Florido FedelinNo ratings yet
- Short Bowel SyndromeDocument20 pagesShort Bowel SyndromeMohamed El-sayedNo ratings yet
- The Standing Meditation of Chinese Soaring Crane QigongDocument7 pagesThe Standing Meditation of Chinese Soaring Crane QigongTom HillNo ratings yet
























































![Sewage and sewerage of farm homes [1928]](https://imgv2-1-f.scribdassets.com/img/word_document/544942443/149x198/ae3275e6b6/1676295883?v=1)


































