Professional Documents
Culture Documents
Simultaneous Determination of Candesartan and Hydrochlorothiazide in Human Plasma by LC-MS/MS
Uploaded by
Galileu E TamyOriginal Title
Copyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
Simultaneous Determination of Candesartan and Hydrochlorothiazide in Human Plasma by LC-MS/MS
Uploaded by
Galileu E TamyCopyright:
Available Formats
Brazilian Journal of
Article
Pharmaceutical Sciences
http://dx.doi.org/10.1590/s2175-97902018000117381
Simultaneous determination of candesartan and
hydrochlorothiazide in human plasma by LC-MS/MS
Mohan Gandhi Bonthu1*, Lakshmana Rao Atmakuri2,Venkateswara Rao Jangala3
1
Department of Pharmaceutical Analysis, Sri Vasavi Institute of Pharmaceutical Sciences, Tadepalligudem, Andhra Pradesh,
India, 2Department of Pharmaceutical Analysis, V. V. Institute of Pharmaceutical Sciences, Gudlavalleru, Andhra Pradesh,
India, 3Department of Pharmaceutical Analysis, St. Paul’s College of Pharmacy, Turkayamjal(V), Telangana, India
A simple, sensitive, rapid and highly efficient LC-MS/MS method was developed for the determination
of Candesartan and Hydrochlorothiazide simultaneously in human plasma. The method employed Zorbax
eclipse C18 (150 X 4.6 mm, 5µ) column using acetate buffer: acetonitrile (25:75%, v/v) as the mobile
phase. The mobile phase flow rate is 1 mL/min which was delivered into the mass spectrometer electron
spray ionization chamber. The Liquid/liquid extraction procedure was used in the method for the extraction
of analytes. The chromatograph was attached to a negative ion mode tandem mass spectrometer and the
method was validated for all the parameters as per the guidelines of US-FDA. The ions were detected
in multiple reaction monitoring mode and the transitions are m/z 439.00→309.10 and 295.80→268.80
for candesartan and hydrochlorothiazide respectively. Isotopic standards were used as internal standards
for effective recovery of the analytes. The drugs were analyzed over a calibration range of 1.027-302.047
ng/mL for candesartan and 1.044-306.945 ng/mL for hydrochlorothiazide respectively with regression
coefficient greater than 0.99. The mean extraction recoveries are 96.95±5.61 and 100.55±4.82 for
candesartan and hydrochlorothiazide respectively. The precision and accuracy values for all the studies
were within the range of ≤15% and 85-115%. The performed stability studies indicate that the developed
method is stable in plasma for 15 h at room temperature (bench top); 52 h (in injector); for 112 days at
-70 ºC for long term stability; five successive freeze and thaw cycles. The developed method could be
successfully employed for the determination of selected drugs in biological samples.
Keywords: Candesartan. Hydrochlorothiazide. LC-MS/MS. Method validation. Human plasma.
INTRODUCTION thiadiazine-7-sulfonamide. It is a prototypical member of
the thiazide diuretic. It helps in reduction of reabsorption
Candesartan (CAN), is chemically 2-ethoxy-1- of various electrolytes through renal tubules resulting
({4-[2-(2H-1, 2, 3, 4-tetrazol-5-yl) phenyl] phenyl} in excretion of water along with different electrolytes
methyl)-)-1H-1,3-benzodiazole-7-carboxylic acid. It is like sodium, potassium, chloride, magnesium etc. It is
an angiotensin receptor blocking agent which can be used widely used in the treatment of edema, hypertension,
alone or in combination with other drugs for the treatment hyperparathyroidism, and diabetes insipidus (The Merck
of hypertension. It competes with angiotensin-II for its index, 2006).
receptors there by lowering blood pressure. It is also used Thorough survey of literature disclosed good
as an effective alternative for the treatment of heart failure, number of analytical methods which include UV (Erk,
myocardial infarction, coronary diseases and systolic 2003a; Naseemet al., 2009), HPTLC (Bipin, Sachinet al.,
dysfunction (The Merck Index, 2006). 2008), HPLC (Qutab et al., 2007; Be et al., 1990; Richter,
Hydrochlorothiazide (HCT), is chemically Oertel, Kir, 1996; Erk, 2003b; Zendelovska, Stafilovm
6-chloro-1, 1-dioxo-3, 4-dihydro-2H-1,2,4-benzo Molisevski, 2004; Balamuralikrishna, Syamasundar,
2010; Annapurna, Narendra, Ravi, 2012; Veeranjaneyulu,
Aneesha, Nandakishore, 2012; Narendra, Satyanarayana,
*Correspondence: M. G. Bonthu. Assistant Professor, Sri Vasavi Institute of
Ganga, 2012), LC-MS (Brushinina et al., 2014; Surbhi et
Pharmaceutical Sciences, Tadepalligudem-534201, Andhra Pradesh, India.
E-mail:bmgandhipharma@gmail.com al., 2010; Bharathi et al., 2012) and UPLC-MS (Singh
Braz. J. Pharm. Sci. 2018;54(1):e17381 Page 1 / 10
M. G. Bonthu, L. R. Atmakuri,V. R. Jangala
et al., 2014) methods are available for the evaluation of pump (LC-20AD) in company with auto-sampler (SIL-
CAN and HCT in bulk, pharmaceutical dosage forms and HTC) was used. Zorbax Eclipse C18 (150 × 4.6 mm, 5
in biological samples individually or in combination with µm) column was employed for the separation of analytes,
other drugs. To our knowledge, no LC-MS/MS method is which was preserved at 40°C±2°C in column oven (CTO-
available for the simultaneous evaluation of these drugs 10AS). A mixture of 5 mM ammonium acetate: acetonitrile
in human plasma using isotopic internal standards. Hence, (25:75%, v/v) was filtered as mobile phase, which was
we made an attempt to develop a simple method for the delivered at a flow rate of 1.0 mL/min into the mass
appraisal of these drugs. The developed method has been spectrometer electrospray ionization chamber.
validated as per the guidelines given by US-FDA for Analytes and IS were ascertained by MS/MS
biological samples (ICH, 1996; ICH, 1997). detection under negative ion mode using an MDS Sciex
(Foster City, CA, USA) API-4000 mass spectrometer. It is
EXPERIMENTAL furnished with a Turbo ion spray. The routine specifications
like curtain gas, nebulizer gas (GS1), auxiliary gas (GS2)
Chemicals and materials and collision gas (CAD) were fixed at 20, 21, 31 and
6 psi, respectively. The compounds specifications like
CAN and HCT were procured from Dr. Reddy’s declustering potential (DP), collision energy (CE), collision
Laboratories Limited, Hyderabad, India. Candesartan exit potential (CEP) and entrance potential (EP) for CAN,
d4 (CAN- IS), Hydrochlorothiazide 13C6 (HCT-IS) were HCT, CAN-IS and HCT-IS were -40, -30, -15, -10 V; -99,
obtained from Clear-synth Labs Limited, Mumbai, -29, -15, -10 V ; -40, -32, -15, -10 V and -99, -29, -15, -10
India. HPLC grade methanol, HPLC grade acetonitrile, V respectively. Diagnosis of the ions was accomplished in
analytical grade formic acid and HPLC grade diethyl ether the multiple reaction monitoring (MRM) mode, observing
were purchased from Merck Specialties, Mumbai, India. the transition of the m/z 439.00 parent ion to the m/z 309.10
K2EDTA human plasma was obtained from a registered daughter ion for CAN, m/z 295.80 parent ion to the m/z
blood bank, Secunderabad, India and was stored at –20°C 268.80 daughter ion for HCT, m/z 443.00 parent ion to the
prior to use. m/z 312 00 daughter ion for CAN-IS and m/z 301.80 parent
ion to the m/z 274.90 daughter ion for HCT-IS. The dwell
Liquid chromatographic and mass spectrometry time was 200 msec. The analytical data were exercised by
Analyst software (version 1.4.2). Summary of optimized
A Shimadzu HT (Shimadzu, Japan) LC system method parameters were tabulated in Table I and optimized
which was furnished with degasser (DGU-20A5), binary system parameters were portrayed inTable II.
TABLE I - Summary of optimized conditions
Parameter Condition
Column Zorbax Eclipse XDB-C18 4.6 x 150 mm, 5 µm (Make: Agilent Technologies)
Mobile phase HPLC grade acetonitrile:5 mM ammonium acetate in water(75:25, v/v)
Rinsing solution HPLC grade acetonitrile:milli-Q water or HPLC grade water (60:40, v/v)
Flow rate 1.0 mL/minute (with splitter)
Split ratio 50:50
Sample cooler temperature 10°C
Injection volume 15 µL
Needle rinsing volume 500 µL
Column oven temperature N/AP
Rinsing mode Before and after aspiration
Retention time Candesartan 1.10±0.1 minutes
Candesartan d4 1.10±0.1 minutes
Hydrochlorothiazide 1.40±0.1 minutes
Hydrochlorothiazide 13C6 1.40±0.1 minutes
Run time 3.00 minutes
Page 2 / 10 Braz. J. Pharm. Sci. 2018;54(1):e17381
Simultaneous determination of candesartan and hydrochlorothiazide in human plasma by LC-MS/MS
TABLE II - LC-MS/MS optimized system parameters
Parameter CAN CAN D4 HCT HCT 13C6
Ionization mode Negative Negative Negative Negative
Detection m/z 439.00 (parent) and 443.00 (parent) and 295.80 (parent) and 301.80 (parent) and
309.10 (product) 312.00 (product) 268.80 (product) 274.90 (product)
Ion Spray Voltage (IS) -4500.00 V -4500.00 V -4500.00 V -4500.00 V
Temperature (TEM0C) 500.00 500.00 500.00 500.00
Curtain Gas (CUR) 20.00V 20.00V 20.00V 20.00V
Collision Gas (CAD) 6.00psi 6.00psi 6.00psi 6.00psi
GS1 21.00 psi 21.00 psi 21.00 psi 21.00 psi
GS2 31.00psi 31.00psi 31.00psi 31.00psi
Declustering Potential (DP) -40.00V -40.00V -99.00V -99.00V
Collision Energy (CE) -30.00 V -32.00 V -29.00 V -29.00 V
Collision Cell Exit Potential (CEP) -15.00 V -15.00 V -15.00 V -15.00 V
Entrance Potential (EP) -10.00 V -10.00 V -10.00 V -10.00 V
Dwell time (m.sec) 200 200 200 200
Software Analyst software 1.4.2
Preparation of reagents grade water was added and mixed it well, then sonicated
it in an ultrasonic bath for 5 min, finally made up to
5 mM ammonium acetate buffer the mark.
About 385.4 mg of ammonium acetate was weighed
accurately and transferred into a clean and dry 1000 mL Preparation of internal standards (IS) solution
standrad flask. About 100 mL of HPLC/Milli Q grade Accurately weighed and transferred 2 mg of
water was added to mix the contents and then subjected Candesartan d4 and Hydrochlorothiazide 13C6 working
to sonication on an ultrasonicator for 5 min. Finally, the standards separately into 2 mL standard flasks. Dissolved
solution was made up to the mark with HPLC/Milli Q in HPLC grade methanol and made up the volume with
grade water. the same to produce a solution of 1.0000 mg/mL of
candesartan d4 and hydrochlorothiazide 13C6.
Mobile phase (25:75%, v/v) Combined stock dilution of candesartan d 4 and
500 mL of 5mM ammonium acetate was transferred hydrochlorothiazide 13C6 was prepared at a concentration
into a two liters volumetric flask. About 500 mL of HPLC of 5000.000 ng/mL and 5000.000 ng/mL, respectively
grade acetonitrile was added to it and mixed it well, using diluent.
sonicated in an ultrasonic bath for 5 min and made up to
the mark with acetonitrile. Preparation of candesartan and hydrochlorothiazide
stock solution
Diluent (v/v) Accurately weighed and transferred 2 mg of
A mixture of HPLC grade acetonitrile and Milli Q/ candesartan and hydrochlorothiazide working standards
HPLC grade water in the volume ratio of 60:40%, v/v separately into 2 mL volumetric flasks. Dissolved in HPLC
respectively was prepared and used as diluent. grade methanol and made up the volume with the same to
produce a solution of 1.0000 mg/mL of candesartan and
Rinsing Solution (v/v) hydrochlorothiazide.
The Diluent was employed as a rinsing solution.
Sample preparation
5% formic acid buffer(v/v) One set of calibration curve standards; one or more
5.00 mL of formic acid was carefully withdrawn sets of quality control samples and plasma samples were
using calibrated pipette and transferred into a 100 mL withdrawn from the deep freezer and allowed it to thaw
standard flask and to this few mL of Milli Q/HPLC at room temperature. The thawed samples were placed on
Braz. J. Pharm. Sci. 2018;54(1):e17381 Page 3 / 10
M. G. Bonthu, L. R. Atmakuri,V. R. Jangala
a vortex to ensure complete mixing of the contents. 100 extracted plasma QC standard samples (n=6) with the
µL of the sample was pipetted out into prelabelled 15 mL response of neat samples at equivalent concentrations.
stopper glass test tubes. 10 µL of internal standard dilution
(combined dilution of 5000.000 ng/mL Candesartan d4 Calibration curve
and Hydrochlorothiazide 13C6) was added, except in blank
wherein added 10 µL of diluent and vortexed. Then, added A nine-point calibration curve (1.027, 2.567, 5.135,
500 µL of 5% formic acid buffer and vortexed. 5 mL of 10.27, 25.674, 51.348, 102.696, 205.392 and 302.047 ng/
tertiary butyl methyl ether (TBME) was added and placed mL for CAN and 1.044, 2.609, 5.219, 10.438, 26.095,
the samples for 20 min on the reciprocating shaker at 200 52.189, 104.378, 208.757 and 306.995 ng/mL for HCT)
rpm. Then samples were centrifuged at 4000 rpm for 10 was plotted by taking the peak area ratio of each analyte:
minutes at 4°C. Then the superficial organic layer (4 mL) internal standard against the nominal concentration of
was shifted to prelabelled glass test tubes and evaporated calibration curve standards. Using the assessment of
to dryness under liquid nitrogen at 45°C. The samples were different weighing factors, the results were made to fit
reconstituted with 250 µL of mobile phase and 15 µL of by linear regression analysis with the help of 1/X2 (X:
the sample was injected into the system. concentration) weighting factor. The limit for calibration
curve is to possess a correlation coefficient (r) of 0.99 or
VALIDATION PROCEDURES more.
The developed method was validated for various Precision and accuracy
specifications to reach the acceptance criteria as per the
guidelines of US-FDA (ICH, 1996; ICH, 1997). For this study, six replicates containing CAN and
HCT at five different QC levels were selected [for CAN:
Matrix effect 1.08 (LLOQ), 3.087 (LQC), 35.079 (MQC1), 152.516
(MQC2) and 258.502 (HQC) ng/mL; for HCT: 1.092
The interference consequences of human plasma (LLOQ), 3.121 (LQC), 35.466 (MQC1), 154.199 (MQC2)
elements over the ionization of CAN, HCT, CAN-IS and and 261.354 (HQC) ng/mL]. The inter-assay precision was
HCT-IS were assessed in this test. The responses of the assessed by monitoring the mentioned QC level samples
post extracted plasma QC samples (n=6) were equated on four different runs. The benchmark for the prominence
with the response of analytes from neat (aqueous) standard of the data included accuracy within ±15% deviation (SD)
samples at equivalent concentrations. It was assessed and a precision of within ±15% relative standard deviation
at low and high concentrations viz., CAN; 3.10 and (RSD) except for LLOQ, where it should not exceed ±20%
256.20 ng/mL, HCT; 3.12 and 262.07 ng/mL, whereas of SD.
for the internal standards it was determined at a single
concentration of 250 ng/mL. Stability experiments
Specificity The stability of analytes and internal standards was
The specificity of the analyte was assessed by assessed randomly by injecting replicate freshly prepared
concealing six different batches of human blank plasma samples in autosampler after the initial injection. The
spiked with its contrary analyte. peak areas of the analytes and internal standard obtained
at initial run were marked as the reference to assess the
Sensitivity relative stability of the analytes at further stages. Stability
The sensitivity is determined by injecting six LLOQ of analytes for benchtop study is performed at two different
samples from six different lots of human plasma and by concentrations in six replicates. Freezer stability of the
spiking the concentration of HCT-ULOQ. The specificity analytes for the biosamples was assessed by analyzing
of the HCT was determined by screening six different the QC samples stored at –70 °C±15°C. The stability
batches of human blank plasma spiked with CAN-ULOQ. of analytes in biomatrix following repeated freeze-thaw
cycles (stored at –70 °C±15 °C between cycles) was
Recovery assessed using QC samples spiked with analytes. Samples
were considered to be stable if assay values were within
The recovery of CAN, HCT, CAN-IS and HCT-IS the acceptable limits of accuracy (i.e., ±15% SD) and
was assessed by correlating the responses of the post precision (i.e., 15% RSD).
Page 4 / 10 Braz. J. Pharm. Sci. 2018;54(1):e17381
Simultaneous determination of candesartan and hydrochlorothiazide in human plasma by LC-MS/MS
RESULTS m/z 439.00 → 309.10 was used for quantification of CAN
and m/z 295.80 → 268.80 was used for quantification of
Liquid chromatography and mass spectroscopy HCT. Similarly, for CAN-IS m/z 443.00 → 312.00 and
m/z 301.80 → m/z 274.90 was used for quantification of
Method development using various solvents such HCT-IS was used for assessment purpose. Mass tuning
as acetonitrile and methanol, using different buffers such peaks are depicted in Figures 1 and 2.
as ammonium formate, ammonium acetate and formic The ideal combination of Zorbax Eclipse column
acid; changed flow-rates (0.1-1.5 mL/min) were tried usage and negative ion mode for quantification of CAN and
for effective separation of analytes with good resolution. HCT helped in achieving greater resolution of the peaks
Finally, the desired resolution was accomplished with and also for concurrent quantification of both analytes with
5mM ammonium acetate: acetonitrile (25:75, v/v) at a flow low LLOQ and shorter run time compared with the earlier
rate of 1.0 mL/min, on Zorbax Eclipse XBD C18 (150 × reported independent methods. Moreover, the employment
4.6 mm, 5 µm) column for the evaluation of electrospray of isotopic internal standards helped in achieving recovery
response for CAN, HCT, CAN-IS and HCT-IS. In order to values on the higher end compared to previous methods.
finalize ESI conditions for CAN, HCT, CAN-IS and HCT-
IS, the assessment was carried out in negative ionization Matrix effect, specificity and sensitivity
mode with MRM scan. The mass spectra for CAN, HCT,
CAN-IS and HCT-IS observed peaks at m/z 439.00, Interferences were not observed at the retention
295.80, 443.00, and 301.80 respectively. The transition of times of the analytes when the blank plasma samples
FIGURE 1 - Mass tuning peaks of candesartan & candesartan D4.
FIGURE 2 - Mass tuning peaks of hydrochlorothiazide & hydrochlorothiazide 13C6
Braz. J. Pharm. Sci. 2018;54(1):e17381 Page 5 / 10
M. G. Bonthu, L. R. Atmakuri,V. R. Jangala
from different lots were injected into the system. Under at the retention times of analytes and IS. The retention time
sensitivity studies, the %CV value was found to be 6.53 of CAN and CAN-IS was 1.10 min and HCT and HCT-IS
which is acceptable as per the guidelines. The average was 1.40 min. respectively. The total chromatographic run
matrix factor values calculated are 1.060 for CAN and time was 3.0 min.
1.033 for HCTZ at LLOQ QC and HQC concentrations.
Significant peaks were absent at the retention times of Recovery
analytes and no change in peak areas of the analytes was
observed which indicates very less effect of plasma extract Recovery was evaluated for each analyte at HQC
on the ionization of analytes and internal standards. concentration and the response in terms of peak area was
A representative chromatogram for the control employed for the calculations. Recovery was found to be
human plasma (free of the analyte and IS) and human to be 98.86%±1.17% and 99.72%±1.40% for CAN and
plasma spiked with CAN, HCT, CAN-IS, HCT-IS at HCT respectively. The recovery of CAN-IS and HCT-IS
LLOQ are shown in Figure 3 to Figure 8 respectively. No was 96.95%±3.69% and 100.55% ±2.10%. The results
interfering peaks from interfering compounds are observed obtained in recovery studies were tabulated in Table III.
FIGURE 3 - Representative chromatogram of blank plasma sample-CAN.
FIGURE 4 - Representative chromatogram of blank plasma sample-HCT.
FIGURE 5 - Representative chromatogram of blank plasma sample with CAN- IS.
Page 6 / 10 Braz. J. Pharm. Sci. 2018;54(1):e17381
Simultaneous determination of candesartan and hydrochlorothiazide in human plasma by LC-MS/MS
FIGURE 6 - Representative chromatogram of blank plasma sample with HCT-IS.
FIGURE 7 - Representative chromatogram of standard CAN (LLOQ sample).
FIGURE 8 - Representative chromatogram of standard HCT (LLOQ sample).
TABLE III - Recovery and matrix effect of CAN and HCT QC Calibration curve
samples
The calibration curve plotted shows outstanding
QC sample
IS normalized
recovery in the entire calibration range. The plot was
Analyte ID (ng/mL) % Recovery
matrix factor
drawn by taking peak-area ratios (peak area analyte/peak
n=6 area IS) on x-axis and concentration on y-axis considering
CAN LQC 3.087 99.78±7.06 1.060±6.12 their best fit using weighing factor (1/x 2). The mean
MQC 152.516 99.26±2.01 ------- regression (n=4) was found to be >0.995. The lowest
HQC 256.523 95.54±3.41 0.985±1.26
concentration with the RSD<20% was taken as LLOQ
and was found to be 1.080 and 1.092 ng/mL for CAN
CAN-IS ---- 250 96.95±3.69 -------
and HCT respectively. The %accuracy calculated for the
HCT LQC 3.121 98.31±3.92 1.033±5.98 mean of back-calculated concentrations for five calibration
MQC 154.199 101.11±2.24 ------- curves for CAN and HCT was within 97.54-99.78 and
HQC 261.354 99.73±3.21 1.015±0.86 97.39-100.67, respectively; while the precision (%CV)
values diverse from 7.06-7.02 and 1.49-4.39 for CAN and
HCT-IS ---- 250 100.55±2.19 -------
HCT, respectively.
Braz. J. Pharm. Sci. 2018;54(1):e17381 Page 7 / 10
M. G. Bonthu, L. R. Atmakuri,V. R. Jangala
Accuracy and precision It was indeed an essence in optimizing
chromatographic conditions, mass spectrometric
Accuracy and precision data for intra-day and inter- parameters and extraction techniques to develop
day plasma samples for CAN and HCT are presented in a selective, sensitive and rapid assay method for
Table IV. The assay values on both the occasions (intra and simultaneous determination of CAN and HCT in human
inter-day) were found to be within the accepted variable plasma. Under mentioned conditions, CAN, HCT, CAN-
limits. IS and HCT-IS exhibit a high sensitivity in negative
ion detection mode rather than positive ion detection
Stability mode. The final composition of mobile phase was 5mM
ammonium acetate: acetonitrile (25:75, v/v), both analytes
The selected concentrations for CAN and HCT and their internal standards got good responses with the
at LQC and HQC deviated within ±15% of the average possible best peaks and eluted within 3 minutes. The
concentrations in a series of stability tests viz., 53.00 h method got all the validation parameters in limits and has
(in-injector), 15.00 h (bench-top), and for 112 days freezer got greater recovery values compared to earlier reported
stability at –70 °C±15 °C (Table V). methods.
DISCUSSION CONCLUSION
Several methods have been reported for the A highly sensitive, specific, reproducible and
estimation of CAN and HCT using LC-MS/MS. Till date, high-throughput LC- MS/MS bioanalytical method was
no LC-MS/MS method was reported for the simultaneous developed and validated to quantify CAN and HCT
determination of these drugs using isotopic internal simultaneously in human plasma. This method could
standards. It is always recommended to select an internal be used to characterize the concentrations of CAN and
standard with close chemical properties to have effective HCT in pharmacokinetic studies. We believe that the
extraction and recovery values. Bearing this fact in mind, LC-MS/MS method presented by us has got good merits
we have selected isotopic internal standards for both the over the earlier reported methods. Advantages of the
drugs. In addition, the earlier reported methods have got current method is less sample volume, utilizes simple
fewer recovery values due to the employment of regular extraction method and enables simultaneous estimation
extraction procedures. of combination drugs; hence we believe that the future
TABLE IV - Intra and inter-day precision of CAN and HCT QC samples
Measured concentration (ng/mL)
Intra-day variation (Six replicates at each concentration)
Theoretical CAN HCT
Concentration (nm/mL) Mean SD % CV Accuracy (%) Mean SD % CV Accuracy (%)
LLOQ 1.02 0.05 4.72 94.17 1.06 0.06 6.30 97.12
LQC 3.05 0.29 9.62 98.90 3.19 0.17 5.46 102.50
MQC1 35.17 1.76 5.01 100.26 36.92 1.72 4.67 104.10
MQC2 154.47 5.51 3.56 101.28 159.00 4.79 3.02 103.12
HQC 256.20 17.51 6.84 99.11 262.07 7.64 2.92 100.28
Inter-day variation (Thirty replicates at each concentration)
LLOQ 1.05 0.07 6.94 97.17 1.08 0.09 8.00 98.79
LQC 3.10 0.24 7.75 100.45 3.14 0.15 4.79 100.76
MQC1 34.80 1.51 4.34 99.20 36.00 1.44 4.02 101.50
MQC2 154.89 5.20 3.36 101.56 156.97 4.25 2.71 101.80
HQC 257.56 12.11 4.71 99.64 262.40 5.61 2.14 100.40
RSD: Relative standard deviation (SD X 100/Mean)
Page 8 / 10 Braz. J. Pharm. Sci. 2018;54(1):e17381
Simultaneous determination of candesartan and hydrochlorothiazide in human plasma by LC-MS/MS
TABLE V - Stability data of CAN and HCT quality controls in human plasma
Mean ± SDa
Nominal conc. Precision(%)
Drug Stability (n=6) Accuracy (%)b
(ng/mL) % C.V.
(ng/mL)
3.087 0 h (for all) 2.85 91.78 4.51
3.087 15 h (bench-top) 3.07 99.67 3.49
CAN 3.087 53.00 h (in injector) 2.86 92.84 2.24
LQC 3.087 112 days at -70±5 ˚C 3.03 98.42 6.12
3.087 Freeze-thaw stability 3.00 97.21 3.32
(5 cycles at-70±5 ˚C)
3.121 0 h (for all) 3.03 98.12 3.12
3.121 15 h (bench-top) 2.92 93.65 4.10
HCT 3.121 53.00 h (in injector) 3.27 104.92 8.23
LQC 3.121 112 days at -70±5 ˚C 3.13 100.48 7.75
3.121 Freeze-thaw stability 3.08 98.81 4.12
(5 cycles at-70±15 ˚C)
258.502 0 h (for all) 264.20 101.57 2.91
258.502 15 h (bench-top) 272.53 105.43 1.78
CAN 258.502 53.00 h (in injector) 254.67 98.52 1.76
HQC 258.502 112 days at -70±15 ˚C 259.08 100.22 2.39
258.502 Freeze-thaw stability 258.57 100.03 3.84
(5 cycles at-70±15 ˚C)
261.354 0 h (for all) 256.04 98.88 1.52
261.354 15 h (bench-top) 255.75 97.85 1.72
HCT 261.354 53.00 h (in injector) 270.92 103.66 6.36
HQC 261.354 112 days at -70±15 ˚C 265.69 101.66 4.77
261.354 Freeze-thaw stability 268.33 102.67 4.92
(5 cycles at-70±15 ˚C)
a
Back-calculated plasma concentration; b(Mean assayed conc. At 0 h) X 100
bioanalytical methods would utilize this method for Be SK, Arun M, David RO,Kin KH. Column-switching high
estimation of CAN and HCT in various biological matrices performance liquid chromatographic (HPLC) determination of
with little or no modification. hydrochlorothiazide in rat, dog and human plasma.Pharm Res.
1990;7(12):1257-1261.
REFERENCES
Bharathi DV, Hotha KK, Chatki PK, Satyanarayana
Annapurna MM, Narendra A, Ravi KK. Liquidchromatographic V,Venkateswarlu V. LC-MS/MS method for simultaneous
method for the simultaneous quantitative determination of estimation of candesartan and hydrochlorothiazide in human
candesartan cilexetil and hydrochlorthiazide in pharmaceutical plasma and its use in clinical pharmacokinetics. Bioanal.
dosage forms. J Drug Del Ther. 2012;2(2):48-54. 2012;4(10):1195-1204.
Balamuralikrishna K, Syamasundar B. Development and Bipin HM, Sachin BM. HPTLC densitometric analysis of
validation of high performance liquid chromatographic method candesartan cilexetil and hydrochlorothiazide in tablets. J
for the simultaneous estimation of candesartan cilexetil and PlanChrom. 2008;21(3):173-176.
hydrochlorothiazide in combined tablet dosage form. Der Pharm
Chem. 2010;2(6):231-237.
Braz. J. Pharm. Sci. 2018;54(1):e17381 Page 9 / 10
M. G. Bonthu, L. R. Atmakuri,V. R. Jangala
Brushinina OS, Gurto RV, Timofeev MS, Frelikh GA, Slepichev Qutab SS, Razzaq SN, Ashfaq M, Shuja ZA, Khan IU. Simple
VA, Yanovskaya EA, et al. Determination of candesartan and sensitive LC–UV method for simultaneous analysis of
andhydrochlorothiazide in human plasma by HPLC coupled hydrochlorothiazide and candesartan cilexetil in pharmaceutical
with mass spectrometry. Int J Anal Mass Spec Chrom. formulations.Acta Chrom. 2007;19:119-129.
2014;2(2):25-32.
Richter K, Oertel R, Kir W. New sensitive method for the
Erk N. Application of First Derivative UV-spectrophotometry determination of hydrochlorothiazide in human serum by
and ratio derivative spectrophotometry for the simultaneous high-performance liquid chromatography with electrochemical
determination of candesartan cilexetil and hydrochlorothiazide. detection.J Chrom A. 1996;729(1-2):293-296.
Pharmazie. 2003a;58(11):796-800.
Singh B, Lokhandae RS, Dwivedi A, Sharma S, Dubey
Erk N. Simultaneous analysis of candesartan cilexetil and N.Improved simultaneous quantitation of candesartan and
hydrochlorothiazide in human plasma and dosage forms using hydrochlorthiazide in human plasma by UPLC-MS/MS
hplc with a photodiode array detector. J LiqChromRel Tech. and its application in bioequivalence studies. J Pharm Anal.
2003b;26(15):2581-2591. 2014;4(2):144-152.
International Conference on Harmonization.ICH.Q2B, Surbhi M, Ravi PS, Rajkamal P, Saranjit S. LC and LC-MS/TOF
validation of analytical procedures methodology.1997, 62, US studies on stress degradation behaviour of candesartan cilexetil.J
FDA Federal Register. Geneva, Switzerland: ICH; 1997. Pharm Biomed Anal. 2010;52(3):345-354.
International Conference on Harmonization.ICH.International The Merck Index.14th edition.Whitehouse Station, NJ:Merck
Conference on Harmonizationof technical requirements for Research Laboratories Division of Merck and Co. Inc.; 2006;
registration of pharmaceuticals for human use (ICH) guideline p. 1742, 4785
on validation of analytical procedure-methodology: Geneva,
Switzerland; 1996. Veeranjaneyulu D, Aneesha A,Nandakishore A. Stability
indicating RP-HPLC method for the simultaneous determination
Narendra D, Satyanarayana T, Ganga RB. Simultaneous of candesartan cilexetil and hydrochlorothiazide in bulk and
determination of candesartan and hydrochlorothiazide in dosage forms. Indian J Rese Pharm Bio. 2012;1(5):720-724.
combined pharmaceutical dosage form by new RP-HPLC
method. Res J PharmBioChem Sci. 2012;3(1):270-278. Zendelovska D, Stafilov T,Milosevski P. Development of solid-
phase extraction method and its application for determination of
Naseem AC, Mohammad B, Enas A,Khalid IH. Determination hydrochlorothiazide in human plasma using HPLC. Bio Chrom.
of candesartan cilexetil in tablet dosage forms and dissolution 2004;18(2):71-76.
testing samples by first derivative uv spectrophotometric
method. Anal Lett. 2009;42(14):2232-2243. Received for publication on 27th June 2017
Accepted for publication on 20th September 2017
Page 10 / 10 Braz. J. Pharm. Sci. 2018;54(1):e17381
You might also like
- Instrumental Methods of Drug AnalysisFrom EverandInstrumental Methods of Drug AnalysisRating: 3 out of 5 stars3/5 (3)
- Development and Validation of A LC/MS/MS Method For The Determination of Duloxetine in Human Plasma and Its Application To Pharmacokinetic StudyDocument14 pagesDevelopment and Validation of A LC/MS/MS Method For The Determination of Duloxetine in Human Plasma and Its Application To Pharmacokinetic StudyMohamed Medhat AliNo ratings yet
- Development and Validation of New Analytical Methods For Simu - 2017 - Arabian JDocument7 pagesDevelopment and Validation of New Analytical Methods For Simu - 2017 - Arabian Jlucian_lovNo ratings yet
- Abhinanda Et Al.,2020Document11 pagesAbhinanda Et Al.,2020Aakanksha SinhaNo ratings yet
- Research PratikshaDocument8 pagesResearch PratikshaNutan Desai RaoNo ratings yet
- Development and Validation of Reversed-Phase HPLC Method For Simultaneous Estimation of Rosuvastatin and Fenofibrate in Tablet Dosage FormDocument6 pagesDevelopment and Validation of Reversed-Phase HPLC Method For Simultaneous Estimation of Rosuvastatin and Fenofibrate in Tablet Dosage FormshraddhaJPNo ratings yet
- Simultaneous RP-HPLC determination of valsartan and hydrochlorothiazideDocument3 pagesSimultaneous RP-HPLC determination of valsartan and hydrochlorothiazideEllie satrianiNo ratings yet
- 2011 - Rapid Simultaneous Determination of Telmisartan, Amlodipine Besylate and Hydrochlorothiazide in A Combined Poly Pill Dosage Form by Stability-Indicating Ultra Performance Liquid ChromatographyDocument16 pages2011 - Rapid Simultaneous Determination of Telmisartan, Amlodipine Besylate and Hydrochlorothiazide in A Combined Poly Pill Dosage Form by Stability-Indicating Ultra Performance Liquid ChromatographyStefana SzántóNo ratings yet
- OutDocument11 pagesOutAngel EivinoNo ratings yet
- 7.meto Hydro TLCDocument6 pages7.meto Hydro TLCBaru Chandrasekhar RaoNo ratings yet
- 2015 - Analytical Techniques For Determination of Hydrochlorothiazide and Its CombinationsDocument13 pages2015 - Analytical Techniques For Determination of Hydrochlorothiazide and Its CombinationsStefana SzántóNo ratings yet
- Tablet Formulation and Development of A Validated Stability Indicating HPLC Method For Quantification of Valsartan and Hydrochlorthiazide Combination 1Document7 pagesTablet Formulation and Development of A Validated Stability Indicating HPLC Method For Quantification of Valsartan and Hydrochlorthiazide Combination 1Wildan SuhadiNo ratings yet
- 116-Article Text-421-2-10-20120317Document7 pages116-Article Text-421-2-10-20120317Fajar PutraNo ratings yet
- 1 Reverse HPLC Method BudesonideDocument11 pages1 Reverse HPLC Method Budesonidenonick18No ratings yet
- 1 s2.0 S2095177912000482 MainDocument8 pages1 s2.0 S2095177912000482 MaindewimachfudNo ratings yet
- 21 Vol. 9 Issue 7 July 2018 IJPSR RA 9344Document7 pages21 Vol. 9 Issue 7 July 2018 IJPSR RA 9344ChilaNo ratings yet
- Jurnal InternasionalDocument12 pagesJurnal InternasionalIsra Tri HardiantiNo ratings yet
- LC-MS Method for Quantifying Diabetes DrugDocument13 pagesLC-MS Method for Quantifying Diabetes DrugManishaNo ratings yet
- A Very Sensitive Bioanalytical Method For The Estimation of Escitalopram in Rat Plasma Using Liquid Chromatography WithDocument10 pagesA Very Sensitive Bioanalytical Method For The Estimation of Escitalopram in Rat Plasma Using Liquid Chromatography WithvinayNo ratings yet
- AbstractDocument11 pagesAbstractEllie satrianiNo ratings yet
- Method Development and Validation of Canagliflozin in Human Plasma by Liquid Chromatography Tandem Mass Spectrometry (LC-MS/MS)Document9 pagesMethod Development and Validation of Canagliflozin in Human Plasma by Liquid Chromatography Tandem Mass Spectrometry (LC-MS/MS)Mahesh with UshaNo ratings yet
- 1 s2.0 S165836551630019X MainDocument10 pages1 s2.0 S165836551630019X MainFauzia HusnaNo ratings yet
- Review Article On ValsartanDocument8 pagesReview Article On ValsartanEditor IJTSRDNo ratings yet
- Stability-indicating RP-HPLC method for racecadotril quantificationDocument8 pagesStability-indicating RP-HPLC method for racecadotril quantificationAnjay MalikNo ratings yet
- Development and Validation of Stability Indicating RPHPLC Method For The Determination of Axitinib in Bulk and Its PharmDocument10 pagesDevelopment and Validation of Stability Indicating RPHPLC Method For The Determination of Axitinib in Bulk and Its PharmAllNo ratings yet
- International Research Journal of PharmacyDocument8 pagesInternational Research Journal of PharmacyVivek SagarNo ratings yet
- Sun 2003Document9 pagesSun 2003adolfo olmosNo ratings yet
- Development and Validation of Dissolution Test for Candesartan TabletsDocument7 pagesDevelopment and Validation of Dissolution Test for Candesartan TabletsElvita SariNo ratings yet
- UV method for metformin quantification in tabletsDocument4 pagesUV method for metformin quantification in tabletsWilliam SmithNo ratings yet
- PCM PhenergenDocument4 pagesPCM PhenergenYoobNorismawandiNo ratings yet
- D Jothiewari Et. Al.. (UV)Document5 pagesD Jothiewari Et. Al.. (UV)EVELYN SOLANHS ACERO RODRIGUEZNo ratings yet
- Journal of Chemical and Pharmaceutical Research: J. Chem. Pharm. Res., 2011, 3 (4) :404-409Document6 pagesJournal of Chemical and Pharmaceutical Research: J. Chem. Pharm. Res., 2011, 3 (4) :404-409J.k. KiranNo ratings yet
- Hidroklortiazid Dan TelmisartanDocument3 pagesHidroklortiazid Dan TelmisartanRetno Rela MahananiNo ratings yet
- AJPS Volume 65 Issue 1 Pages 85-106Document22 pagesAJPS Volume 65 Issue 1 Pages 85-106yusriyah zulfaNo ratings yet
- RP-HPLC Method For The Quantitative Determination of Fexofenidine HCL in Coated Tablets and HumanDocument13 pagesRP-HPLC Method For The Quantitative Determination of Fexofenidine HCL in Coated Tablets and HumanchiralicNo ratings yet
- Sian Ournal of HemistryDocument5 pagesSian Ournal of Hemistryjkc collegeNo ratings yet
- Arnhard 2012Document14 pagesArnhard 2012malwanafhc.adNo ratings yet
- V3I303Document12 pagesV3I303Harrizul RivaiNo ratings yet
- Development and Validation of Stability Indicating Assay Method For Simultaneous Estimation of Amoxicillin Trihydrate and Cloxacillin Sodium in Pharmaceutical Dosage Form by Using RP-HPLCDocument13 pagesDevelopment and Validation of Stability Indicating Assay Method For Simultaneous Estimation of Amoxicillin Trihydrate and Cloxacillin Sodium in Pharmaceutical Dosage Form by Using RP-HPLCJermy ErmiNo ratings yet
- 20 Manuscript PDFDocument11 pages20 Manuscript PDFBaru Chandrasekhar RaoNo ratings yet
- Simultaneous Determination of Cefotaxime Sodium and Paracetamol by LC-MSDocument7 pagesSimultaneous Determination of Cefotaxime Sodium and Paracetamol by LC-MSIOSR Journal of PharmacyNo ratings yet
- Microchemical Journal: SciencedirectDocument8 pagesMicrochemical Journal: SciencedirectErika Alejandra Pardo AcostaNo ratings yet
- Quantification of Methyldopa in Human Plasma by HPLC-MS/MSDocument8 pagesQuantification of Methyldopa in Human Plasma by HPLC-MS/MSNgọc MaiNo ratings yet
- Development and Validation of An RP-HPLC Method For Simultaneous Estimation of Amlodipine Besylate and Indapamide in Bulk and Tablet Dosage FormDocument10 pagesDevelopment and Validation of An RP-HPLC Method For Simultaneous Estimation of Amlodipine Besylate and Indapamide in Bulk and Tablet Dosage FormBaru Chandrasekhar RaoNo ratings yet
- Development and Validation of A Reversed Phase HPLC Method For Simultaneous Determination of Curcumin and Piperine in Human Plasma For Application inDocument15 pagesDevelopment and Validation of A Reversed Phase HPLC Method For Simultaneous Determination of Curcumin and Piperine in Human Plasma For Application inMaryem SafdarNo ratings yet
- High Performance Liquid Chromatography Method ForDocument8 pagesHigh Performance Liquid Chromatography Method ForWalther YepesNo ratings yet
- Study On Stress Degradation Behaviour of Abiraterone Acetate in Film Coated Tablets and Identification of Major Stress DDocument11 pagesStudy On Stress Degradation Behaviour of Abiraterone Acetate in Film Coated Tablets and Identification of Major Stress DvinayNo ratings yet
- Stability Indicating RP-LC Assay Method For Carisoprodol: Original ArticleDocument7 pagesStability Indicating RP-LC Assay Method For Carisoprodol: Original ArticleNguyen binhNo ratings yet
- New RP-HPLC Method For The Determination of Olmesartan Medoxomil in Tablet Dosage FormDocument7 pagesNew RP-HPLC Method For The Determination of Olmesartan Medoxomil in Tablet Dosage FormsanjeevbhatNo ratings yet
- European Journal of Pharmaceutical Sciences: Mahmoud Kamel Mohamed, John Takyi-Williams, Bertrand Baudot, Anne GroblerDocument8 pagesEuropean Journal of Pharmaceutical Sciences: Mahmoud Kamel Mohamed, John Takyi-Williams, Bertrand Baudot, Anne GroblerLeidys Pérez SerpaNo ratings yet
- BisopDocument11 pagesBisopAlinaDianaNo ratings yet
- 263-Article Text-921-2-10-20190727Document9 pages263-Article Text-921-2-10-20190727ankit AcharyaNo ratings yet
- Determination Pyrazinamide in Human PlasmaDocument7 pagesDetermination Pyrazinamide in Human Plasmaervano1969No ratings yet
- Determination of 17 B-Estradiol in Pharmaceutical Preparation by UV Spectrophotometry and High Performance Liquid Chromatography MethodsDocument7 pagesDetermination of 17 B-Estradiol in Pharmaceutical Preparation by UV Spectrophotometry and High Performance Liquid Chromatography MethodsMariayolanita PajangNo ratings yet
- Article Wjpps 1425131684Document17 pagesArticle Wjpps 1425131684VOOGLS PUBLICATIONNo ratings yet
- Development and Validation of A Dried Blood Spot LC-MS/MS Assay To Quantify Furosemide in Human Whole Blood: An Approach To Improve The Design of PK Studies in Pediatric Population.Document7 pagesDevelopment and Validation of A Dried Blood Spot LC-MS/MS Assay To Quantify Furosemide in Human Whole Blood: An Approach To Improve The Design of PK Studies in Pediatric Population.IOSRjournalNo ratings yet
- Metoclopramida DegradacionDocument9 pagesMetoclopramida DegradacionYazminNo ratings yet
- Method Development and Validation of Paracetamol Drug by RP-HPLC 1Document7 pagesMethod Development and Validation of Paracetamol Drug by RP-HPLC 1Anonymous ncDgoMONo ratings yet
- Ashraf M. Mahmoud, Saad A. Alqahtani: American Journal of Analytical Chemistry, 2016, 7, 179-191Document13 pagesAshraf M. Mahmoud, Saad A. Alqahtani: American Journal of Analytical Chemistry, 2016, 7, 179-191DrGajanan VaishnavNo ratings yet
- 1947-489X 210755Document9 pages1947-489X 210755게임 뉴비에 게임 모험No ratings yet
- Group2 Magtarayo Mariano PecaocoDocument20 pagesGroup2 Magtarayo Mariano PecaocoMarco MalasanNo ratings yet
- Adsorption of Ammonia On Regenerable Carbon Sorbents: Advanced Fuel Research, Inc., East Hartford, CT, 06108 andDocument11 pagesAdsorption of Ammonia On Regenerable Carbon Sorbents: Advanced Fuel Research, Inc., East Hartford, CT, 06108 andKamalesh KumarNo ratings yet
- Primer 116970Document6 pagesPrimer 116970willis kristianNo ratings yet
- Final - HE 2.4 Performing Simple Calculations in CaregivingDocument36 pagesFinal - HE 2.4 Performing Simple Calculations in CaregivingJasmine EnriquezNo ratings yet
- What Defines An Industrial GasDocument2 pagesWhat Defines An Industrial GasYuri YamirnovNo ratings yet
- MSP Modular Divider Valves: Your Key To Maximum Design Flexibility With Series-Progressive ReliabilityDocument12 pagesMSP Modular Divider Valves: Your Key To Maximum Design Flexibility With Series-Progressive ReliabilityCedric ChatonNo ratings yet
- AITS-2021-OPEN TEST-JEEA-PAPER-1-SolDocument15 pagesAITS-2021-OPEN TEST-JEEA-PAPER-1-SolAyush SrivastavNo ratings yet
- A.B.paterakis - PHD - The Formation of Acetate Corrosion On Bronze AntiquitiesDocument362 pagesA.B.paterakis - PHD - The Formation of Acetate Corrosion On Bronze AntiquitiesDan Octavian PaulNo ratings yet
- Introduction To Organic ChemistryDocument16 pagesIntroduction To Organic ChemistryJan Mykel PestañoNo ratings yet
- Fire Watcher& Safety WatcherDocument6 pagesFire Watcher& Safety WatcherFarman ShaikhNo ratings yet
- BS 60079-10-2Document30 pagesBS 60079-10-2Ade KalejaiyeNo ratings yet
- Data Sheet: E 44/3 U0/V20 GreenDocument3 pagesData Sheet: E 44/3 U0/V20 GreenedNo ratings yet
- Chisti1998 PDFDocument80 pagesChisti1998 PDFAlpha ValerioNo ratings yet
- Iliade 453:2019 - Clen MethodDocument7 pagesIliade 453:2019 - Clen MethodMonirNo ratings yet
- Flange Dimensions Based On Tables D and E of BS 10: 1962Document6 pagesFlange Dimensions Based On Tables D and E of BS 10: 1962윤규섭No ratings yet
- Laporan PertaminaDocument51 pagesLaporan PertaminaDeni HermawanNo ratings yet
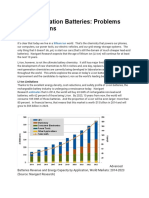
- Next-Generation Batteries: Problems and Solutions: Sam JaffeDocument4 pagesNext-Generation Batteries: Problems and Solutions: Sam JaffeNIBIN MARTINNo ratings yet
- SCH4U Chemistry 12 University Prep Solutions ManualDocument611 pagesSCH4U Chemistry 12 University Prep Solutions Manualkomal sheikh83% (12)
- Disperbyk-110 Disperbyk-111: Wetting and Dispersing Additives For Solvent-Borne SystemsDocument2 pagesDisperbyk-110 Disperbyk-111: Wetting and Dispersing Additives For Solvent-Borne SystemsLê Tiến100% (1)
- NG Et Al 2017Document13 pagesNG Et Al 2017rinifiahNo ratings yet
- Hydrazine in Water: Standard Test Method ForDocument4 pagesHydrazine in Water: Standard Test Method Forastewayb_964354182100% (1)
- International Journal of Innovative Pharmaceutical ResearchDocument5 pagesInternational Journal of Innovative Pharmaceutical Researchdini hanifaNo ratings yet
- CHEM-Types of SolidDocument4 pagesCHEM-Types of SolidMark Joseph PulintanNo ratings yet
- Salt Preparation and AnalysisDocument7 pagesSalt Preparation and AnalysisNyomie CoxNo ratings yet
- Kollidon-90-F Technical Information PDFDocument9 pagesKollidon-90-F Technical Information PDFPalak AgarwalNo ratings yet
- Standard Operating Procedures For Water Treatment Plants: Frequently Asked QuestionsDocument7 pagesStandard Operating Procedures For Water Treatment Plants: Frequently Asked QuestionsModi JayNo ratings yet
- Toxicology of Cleaning AgentsDocument38 pagesToxicology of Cleaning AgentsThe AbyssinicansNo ratings yet
- Phys Sci 2024 National Annual Teaching Plan Grade 12Document8 pagesPhys Sci 2024 National Annual Teaching Plan Grade 12kwandegquma62No ratings yet
- Na Benzoat Spektro UVDocument24 pagesNa Benzoat Spektro UVdevinaNo ratings yet
- Safdar Chemistry - Project - On - STUDY - OF - ADULTERANTDocument15 pagesSafdar Chemistry - Project - On - STUDY - OF - ADULTERANTSAFDAR HafizNo ratings yet