Professional Documents
Culture Documents
BIOTECHNOLOGY - Applications & Ethical Issues
Uploaded by
Korou LaikhaOriginal Title
Copyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
BIOTECHNOLOGY - Applications & Ethical Issues
Uploaded by
Korou LaikhaCopyright:
Available Formats
Study Notes – Class XII BIOTECHNOLOGY – Applications & Ethical Issues
APPLICATIONS OF BIOTECHNOLOGY
The applications of biotechnology include the following fields:
Therapeutics
Diagnostics
Genetically modified crops
Food processing
Bioremediation
Waste treatment
Energy production, etc.
Three critical research areas of biotechnology:
1. Developing best catalyst
a. Catalyst for biotechnology work include improved strains of organisms such as
microbes
b. Enzymes such as REE, ligase, etc.
2. Creating optimal conditions for the catalyst to act
a. Such as developing better growth medium for growing microorganisms and other
hosts
3. Developing better downstream processing technologies
a. The best quality product development depends on downstream processing
employed to purify the proteins or the products.
Applications of Biotechnology
Agriculture Medicine Transgenic animal
Recombinant Proteins Therapeutics Molecular Diagnosis
Applications in Agriculture
Genetically Modified Organisms (GMOs): organisms which are genetically engineered for
human benefit.
Genetic modifications of plants have following advantages:
1. Abiotic stress tolerant crops
a. GM plants can tolerate extreme cold, draught, heat, high salt conc. in soil, etc.
2. Reduced reliance on chemical pesticides
a. Reduces the side effects of pollution caused by chemical pesticides
b. Insect resistant and pest resistant plants were developed
3. Reduced post harvest losses
a. GM plants have higher shelf life
b. They are resistant to microbial infections
4. Increased efficiency of mineral usage by plants
a. So that the soil fertility is not exhausted early
5. Enhanced nutritional value
a. So that food security can be ensured
b. Plants rich in vitamins, calcium, etc are created
Notes prepared by MRINAL CHOUDHURY Page 1 of 11
Study Notes – Class XII BIOTECHNOLOGY – Applications & Ethical Issues
Bt Cotton – Insect Resistant Plant
Cotton plants are often infected by various insects such as
o Lepidopterans, e.g. tobacco budworm, armyworm
o Coleopterans, e.g. beetles
o Dipterans, e.g. flies, mosquitoes
Bacteria Bacillus thuringiensis contain cry genes – cryIAb, cryIAc and cryIIAb
Cry genes produce insecticidal proteins effective against the cotton infecting insects.
However, they are produced in inactive form inside bacteria and hence do not effect them.
Genetic engineering of cotton plants: the process is as follows
Cry genes form Bacillus thuringiensis are isolated
↓
Genes are introduced into cotton plant cells using Ti plasmid
(Agrobacterium mediated transfer)
↓
Genetically modified plant cells ate cultured to produce genetically modified plants
↓
GM cotton plants produce inactive Cry toxin protein in its cells
↓
When insects feed on the cotton tissues, inactive toxin enters the insect gut
↓
In the alkaline pH of insect gut, inactive toxin becomes active toxic protein
↓
Toxin binds the midgut surface and produce pores in the gut epithelium
↓
The cells swell up and burst, killing the insect.
Pest Resistant plant
Roots of tobacco plants are often infected by Meloidogyne incognita
They reduce the plants roots’ absorbing capacity and thus reduce the yield.
A novel strategy known as RNA interference or RNA Silencing is employed to make pest
resistant plants.
Notes prepared by MRINAL CHOUDHURY Page 2 of 11
Study Notes – Class XII BIOTECHNOLOGY – Applications & Ethical Issues
The process of RNA interference is as follows:
Usually source of RNA is Retrovirus infection or Transposons.
For pest resistance, an artificial pest specific DNA is constructed
↓
Pest specific DNA is introduced into the plant cells through Agrobacterium mediated transfer
↓
Transcription of the pest specific DNA from both the sides
produce a dsRNA
↓
Dicer protein acts on the dsRNA and cleaves it to form multiple
siRNAs (small interfering RNA)
↓
RISC (RNA induced silencing complex) binds to it the siRNAs to
form activated RISC
↓
Activated RISC* becomes single stranded which is
complementary to an important nematode mRNA
↓
When a nematode infects the plant, the RISC* encounters the
nematode mRNA and binds to it
↓
RISC* cleaves the nematode mRNA
↓
Thus nematode mRNA is silenced, i.e. cannot undergo translation
and leads to death of nematode
Recombinant Protein Production – Human Insulin (Humulin)
30 therapeutic recombinant proteins are approved for human use, 12 are marketed in India.
One of the most common diseases among all human is Diabetes.
Since the time it was discovered that deficiency of hormone Insulin is one important reason
for its onset, different insulin molecules from various sources such as cow and pig have been
tried.
Although, they were functional to some extent and induces immunogenic response in the
body.
After the birth of genetic engineering, recombinant human insulin is being made in a
bacterial host.
The first such recombinant human insulin is branded as Humulin
It was manufactured by Eli Lilly and Co., and American company in 1983.
Structure of normal in vivo Human Insulin
o INS gene at chromosome no 11 codes for human insulin.
Notes prepared by MRINAL CHOUDHURY Page 3 of 11
Study Notes – Class XII BIOTECHNOLOGY – Applications & Ethical Issues
o Human insulin is translated as a fusion protein, Preproinsulin of Signal peptide, B
chain, C chain and A chain having a total of 110 amino acids.
Signal peptide – 24 amino acids
B chain – 30 amino acids
C chain – 35 amino acids
A chain – 21 amino acids
o Signal peptide directs the preproinsulin to golgi bodies for modifications
o C chain induces a folding of the preproinsulin
o Signal peptide is cleaved inside golgi and two disulfide bonds are formed between A
and B chains.
o C chain is cleaved and degraded
o Mature insulin has two chains A and B, having 51 amino acids and has a molecular
weight of 5808 Da.
Production of recombinant protein Insulin
A and B chains are artificially
synthesized separately
↓
They were ligated into two separate
plasmids along with β-gal gene
↓
Recombinant plasmids were
transformed into E.coli
↓
Expression of insulin gene led to
formation of β-gal/insulin fusion
proteins inside the cells
↓
They were extracted after mass
production
↓
Treatment with cyanogens bromide
cleaved β-gal from A and B chain
polypeptides
↓
Mixing the purified A and B chains produced active Insulin molecule by formation of two
disulfide bonds
Assembly of mature insulin by forming disulfide bonds between A and B chains is a
challenge, which often reduces the efficiency of the process.
This challenge was overcome by another approach by Genetech
o A proinsulin cDNA was used as gene of interest and proinsulin was produced by the
same process as followed by Eli lilly
o But the presence of C chain in the final protein, induced the natural folding and
bonding of A and B chains
o C chain was cleaved by treating with carboxypeptidase and thus mature insulin was
formed.
Notes prepared by MRINAL CHOUDHURY Page 4 of 11
Study Notes – Class XII BIOTECHNOLOGY – Applications & Ethical Issues
Gene Therapy
Gene therapy literally means replacing the defective gene by a functional copy of the same.
First gene therapy was successfully carried out for ADA-SCID in a 4 year old Sri Lankan girl
Ashanti de-Silva by Dr. William French Anderson at National Institute of Health, USA in
1990
ADA-SCID: Adenosine Deaminase Severe Combined Immunodeficiency
o Adenosine is an enzyme coded by ada gene in chromosome 20
o It is expressed in the lymphocytes necessary for proper immune system development
o Its deficiency causes severyly compromised immune system
o Conventional treatment includes –
Enzyme Replacement Therapy – not a permanent treatment procedure as
frequent injections of enzyme is necessary
Bone Marrow Transplantation – not feasible always due to non availability of
matching donor
Gene Therapy for ADA SCID
Patient lymphocytes are isolated and cultured
↓
The abnormal lymphocytes are treated with disarmed retrovirus carrying recombinant RNA of
normal ADA gene.
↓
Inside the cells recombinant RNA produce recombinant DNA by the action of enzyme reverse
transcriptase.
↓
Normal recombinant DNA (ADA gene) is integrated into the lymphocytes’ DNA forming
genetically modified lymphocytes
↓
GM lymphocytes are selected for successful transformation and cultured to increase their number
↓
GM lymphocytes are injected into patient blood stream
Since the cell contain a copy of the normal gene, normal ADA is produced in the body
Since lymphocytes are of patient origin, there is no problem of matching donor
Limitation of the process:
Notes prepared by MRINAL CHOUDHURY Page 5 of 11
Study Notes – Class XII BIOTECHNOLOGY – Applications & Ethical Issues
o Since lymphocytes have a fixed life span, GM lymphocytes need to regularly injected
into patient’s blood.
o This limitation may be removed by carrying out gene therapy at an early embryonic
stage so that all the cells of body receive the normal copy of the gene.
Molecular Diagnosis
Treatment of a disease depends on its accurate and early detection/diagnosis.
Conventional diagnosis is done by testing samples such as serum, urine, stool, sputum, etc.
However, such diagnostic tools are not efficient enough for accurate and early diagnosis.
Accurate and early diagnosis can be done by using molecules such as DNA and proteins.
Use of molecules for diagnosis is known as Molecular Diagnosis
Various techniques such as PCR, ELISA and Radioactive probe are used for molecular
diagnosis.
Molecular Diagnosis – PCR
Many acute diseases such as AIDS and Cancer require early detection to be treated
efficiently.
However, their diagnosis depends upon the quantity of infectious agents or amount of
mutations present in the body.
Thus PCR is employed to detect the presence of bacteria or virus or mutations in DNA
Process of detection
o Primers are used to amplify the pathogen DNA or
the mutated DNA
o If the pathogen is present even in very minute
quantity, its DNA is amplified
o During the electrophoresis, a band for pathogen
DNA is observed along with the host DNA.
Types of PCR for diagnosis –
o Qualitative PCR – only presence or absence of a
pathogen is detected
o Quantitative PCR – quantity of pathogen or
mutation is detected.
Molecular Diagnosis – ELISA
ELISA (enzyme-linked immunosorbent assay) is a plate-based assay technique designed for
detecting and quantifying substances such as peptides, proteins, antibodies and hormones.
Whenever, there is an infection, an antigenic determinant is present in the serum, which may
be detected by ELISA
Else, when an antigen is present, immune system produces antibodies against it, such
antibodies can be detected by ELISA, which shows the presence of antigen.
Notes prepared by MRINAL CHOUDHURY Page 6 of 11
Study Notes – Class XII BIOTECHNOLOGY – Applications & Ethical Issues
The basic process of ELISA is as follows:
A microtitre ELISA plate is taken having multiple small wells
(as in the pic.)
↓
The buffered serum where antigen is to be tested is taken in the
wells of microtitre plate
↓
Some time is allowed for the antigens to adhere to the wells
↓
A solution of non reacting protein such as Bovine Serum
Albumin is added to coat any surface on the plate which is
uncoated with antigen ↓
↓
An antibody attached (linked) to an enzyme is added which
specifically binds to the antigen
↓
↓
A substrate is added for the enzyme which changes colour
when acted upon by the enzyme thus showing the presence of
antigen in the serum
↓
Qualitative Quantitative
estimate estimate
Appearance of color More is the quantity of antigen
indicates the presence of in the serum, more intense the
antigen color will be
↓ ↓
A positive test Can be estimated using
colorimeter
Molecular Diagnosis – Radio labelled Probe
Probe: an artificially synthesized single stranded DNA or RNA sequence of variable length
(usually 100-1000 bases long).
A radiolabelled probe has a radioactive substance coupled to it.
The sequence of the probe is complementary to the target DNA sequence to be identified.
This technique can be used for identifying the presence of mutations in a sample of tissues.
A brief overview of the principle:
A radiolabelled probe is synthesized having sequence complementary to normal gene
sequence
↓
The DNA from tissue sample is exposed to Radiolabelled probe for hybridisation
↓
Hybridised sample is exposed to X ray film for detecting radioactive signals
↓
Notes prepared by MRINAL CHOUDHURY Page 7 of 11
Study Notes – Class XII BIOTECHNOLOGY – Applications & Ethical Issues
Interpretation of observations:
If the gene is not mutated, probe binds to it and gives a positive signal for radioactivity
If the gene is mutated, its sequence changes, as a result, probe does not bind to it, and gives a
negative result
TRANSGENIC ANIMALS
Transgene: a foreign gene that is introduced into a host cell
Transgenic organisms: An organism produced by introducing a foreign gene into it
to study normal
physiology and
development
to study to study a
chemical human
toxicity disease and
new drug
TRANSGENIC trial
ORGANISMS
to produce
To study the useful
vaccine safety biological
products
Model organism: an organism that has a physiology similar to human beings, which is used
to study human physiology or pathophysiology.
o Transgenic Model Organisms: they are produced by introducing a foreign gene
into a model organism which can produce a typical phenotype similar to human.
o Knock out model organism: a transgenic animal in which a specific gene is
inactivated by introducing another similar gene (having enough changes in sequences
to produce an abnormal copy of the gene product)
The introduced gene due to its homology with the host gene gets incorporated in the
place of host gene by replacing the same.
Transgenic Model Organisms to study normal physiology and development of Human:
o Knock out mice models are made to inactivate specific genes similar to human and
study
the function of the gene product
its role in the normal physiology and development
how the gene is regulated
o e.g. – effect of Insulin like Growth Factor, PDGF, etc.
Transgenic Model Organisms to study Diseases and new Drug Effect:
o Knock out mice models are also made to deactivate certain genes responsible for
various single gene disorders and other genetic diseases.
o Such diseases can be studied to understand their pathophysiology and disease
progression
o Any new drug or other medical interventions developed for such diseases can be
tested in transgenic model organisms.
Notes prepared by MRINAL CHOUDHURY Page 8 of 11
Study Notes – Class XII BIOTECHNOLOGY – Applications & Ethical Issues
o E.g. - model organisms are available for cancer, cystic fibrosis, rheumatoid arthritis,
Alzheimer’s disease, etc.
Production of Biological Products:
o Various biological products have uses in medicine.
o But they are either difficult to extract from animal tissues or difficult or too expensive
to synthesize.
o Such products may be produced by genetic engineering as follows:
The specific human gene is isolated from human DNA
Unfertilised egg is obtained from a suitable donor
↓
Human gene is introduced into the egg by
microinjection
↓
The transgenic egg is introduced into the host female
organism (usually a surrogate goat or cow)
↓
The new offspring (transgenic) produced will carry
the human gene
↓
The transgenic adult will secrete the human protein in its
milk
o E.g. –
1. 1997, 1st transgenic cow, Rosie was born, produced 2.4 gm/ltr of α-lactalbumin
2. Human protein α-1-antitrypsin for treatment of emphysema
3. Tissue Plasminogen Activator from transgenic goat
4. Blood Clotting Factor VIII and IX from transgenic sheep
Testing Vaccine Safety using Transgenic mice models:
o Since vaccine contain antigens although in weakened state, it cannot tested in humans
directly, so they are tested in animal models first
o Transgenic mice are replacing monkeys for testing the safety of new vaccine.
o E.g. – Polio Vaccine
Testing safety of chemicals in hypersensitive transgenic models:
o Transgenic model organisms are made to be hypersensitive for specific toxic
chemicals so that even minute amount of toxic chemicals in foods or drugs or
cosmetics can be determined.
Rapid Growth promotion in commercial animals:
o Transgenic animals are made by introducing growth promoting genes to induce faster
and better growth or increase in size.
o Better meat can be obtained in a shorter duration in sheep and fish
Higher Wool production in Transgenic Sheep:
o Transgenic sheep is made by introducing bacterial genes for synthesis of amino acid
cysteine.
o Such sheep produces higher yield of wool
Transgenic Pigs to be used for xenotransplantation of human organs:
o Transgenic pigs are produced having human gene which make them more genetically
closer to human beings.
o The organs from such transgenic sheep can be used for transplantation in human with
reduced chances of immunoreactions.
Notes prepared by MRINAL CHOUDHURY Page 9 of 11
Study Notes – Class XII BIOTECHNOLOGY – Applications & Ethical Issues
ETHICAL ISSUES
Ethics: ethics is a set of moral principles and standards by which a community regulates its
behaviour and decides as to the legitimacy and illegitimacy of an activity.
Bioethics: a set of moral principles and standards which regulate our perceptions as to
exploitation of biological world
GEAC (Genetic Engineering Approval Committee): Government organisation that takes
decision regarding validity of Genetic Engineering / Genetic Modification research and their
safety of use in the environment.
Major Bioethical issues are:
o Unfamiliar proteins are introduced into the organisms which react to form toxins and
allergens.
o Gene for high yield introduced into crops can pass into weeds through pollen transfer,
which may increase their population indiscriminately.
o Animals used in biotechnology experiments are made to suffer
o Animals being used to produce biological products are reduced to the status of
factories.
o As a foreign gene is introduced into the organism, it loses the integrity as a species
o The their race to gain supremacy over others, companies and individuals are rushing
for biopatents even of those products which are produced through traditional
knowledge of tribals, communities and societies.
Two major bioethical issues are: Biopatent and Biopiracy
Biopatent
Patent: an official right from a government granting an inventor or establishment the sole
monopoly to make and sell a particular article for a certain period.
Biopatent: an official right from a government granting an inventor or establishment the sole
monopoly to use a particular biological material for commercial exploitation of
i. Strains of microorganisms
ii. Cell lines
iii. Genetically modified plants and animals
iv. DNA sequences
v. Protein encoded by DNA sequences
vi. A particular biotechnological procedure
vii. A production process
viii. Product
ix. Product application
Ethical issues related to biopatenting: Cases study
o Certain developed countries are issuing broad patents for whole group of organisms
which prevent others from using any related organism or for carrying out any
research.
o Case of Basmati rice:
27 documented varieties of Basmati rice are grown in India.
An American company developed a new variety of basmati by crossing an Indian
farmer’s variety and semi dwarf variety.
The company got patent for the new variety from US patent and Trademark
office, which allowed the company to sell the new variety in the US and abroad.
The US patent extends to functional equivalents which mean it prevents anyone
else from selling Basmati rice.
Notes prepared by MRINAL CHOUDHURY Page 10 of 11
Study Notes – Class XII BIOTECHNOLOGY – Applications & Ethical Issues
Biopiracy
Use of bio-resources by multinational companies and other organisations without proper
authorization from the countries and people concerned without compensatory payments.
Mostly industrialised nations are involved which are rich financially.
Underdeveloped nations having rich biodiversity are usually victimized of biopiracy.
Action taken
Indian Parliament has amended the Indian Patent Bill.
It now considers emergency granting of patent for research and development initiative.
Notes prepared by MRINAL CHOUDHURY Page 11 of 11
You might also like
- Class XII English Vistas Chapter 6. On The Face of It' by Susan HillDocument15 pagesClass XII English Vistas Chapter 6. On The Face of It' by Susan HillChetna Singh100% (1)
- Sophie's Fantasies About Meeting Soccer Star Danny CaseyDocument18 pagesSophie's Fantasies About Meeting Soccer Star Danny Caseydragon draksterNo ratings yet
- Indigo NOTESDocument4 pagesIndigo NOTESDZ GamesNo ratings yet
- Memories of Childhood - AnswersDocument6 pagesMemories of Childhood - AnswersVarun KumarNo ratings yet
- On The Face of It PDFDocument10 pagesOn The Face of It PDFlalitkumarNo ratings yet
- Vistas Prose - 1 The Third LevelDocument6 pagesVistas Prose - 1 The Third LevelMohammad AyaanNo ratings yet
- Indigo, Class 12thDocument3 pagesIndigo, Class 12thdheeraj sharmaNo ratings yet
- Rattrap story highlights human natureDocument5 pagesRattrap story highlights human natureEducation academy / Arin tech and vlogNo ratings yet
- Gandhi's Struggle for Peasants in ChamparanDocument4 pagesGandhi's Struggle for Peasants in ChamparanBala SuryaNo ratings yet
- Going Places Notes and QuestionsDocument3 pagesGoing Places Notes and QuestionsSeema S. Baghel100% (1)
- RattrapDocument48 pagesRattrapvandana khatriNo ratings yet
- Pb23che04 MSDocument5 pagesPb23che04 MSLalitranjan karNo ratings yet
- Self InductanceDocument15 pagesSelf Inductancesindhukannan2006No ratings yet
- Pearl Harbor Enemy Tells Story of Japanese Surgeon Saving Injured American POWDocument10 pagesPearl Harbor Enemy Tells Story of Japanese Surgeon Saving Injured American POWSouri DasguptaNo ratings yet
- Going Places Summary Class 12th EnglishDocument17 pagesGoing Places Summary Class 12th EnglishLohit DasNo ratings yet
- Indigo: Answer The Following Questions: (Long Answer Type)Document3 pagesIndigo: Answer The Following Questions: (Long Answer Type)RootNo ratings yet
- Article Writing Class 12th 2023Document14 pagesArticle Writing Class 12th 2023gokulNo ratings yet
- Investegatory Project Physics TESLA COILDocument24 pagesInvestegatory Project Physics TESLA COILMohd Ali17% (6)
- Useful To Human. Analogues For Products and Services: BIOTECHNOLOGY - Principles and ProcessesDocument10 pagesUseful To Human. Analogues For Products and Services: BIOTECHNOLOGY - Principles and ProcessesKorou LaikhaNo ratings yet
- Phyisc Internal Resistance Sonal PDFDocument14 pagesPhyisc Internal Resistance Sonal PDFAnkit Bharti100% (1)
- Extra Questions and Very Important Questions For Going PlacesDocument6 pagesExtra Questions and Very Important Questions For Going PlacesshradhaNo ratings yet
- Delta English school physics project on cell resistance factorsDocument16 pagesDelta English school physics project on cell resistance factorsaifafidu2005No ratings yet
- THE ENEMY NotesDocument9 pagesTHE ENEMY NotesDieHard TechBoyNo ratings yet
- EngProj12ScB 1Document25 pagesEngProj12ScB 1Kshitij SinghNo ratings yet
- THE THIRD LEVEL ChapterDocument4 pagesTHE THIRD LEVEL ChapterSunil rathi100% (1)
- Chemistry ProjectDocument33 pagesChemistry ProjectRakshit Sharma50% (2)
- The Lost Spring: The Theme of The Chapter Is The Grinding Poverty and The Traditions Which Condemn PoorDocument4 pagesThe Lost Spring: The Theme of The Chapter Is The Grinding Poverty and The Traditions Which Condemn PoorCrazy about Jungles100% (1)
- Gemini Studios JourneyDocument8 pagesGemini Studios JourneySharonNo ratings yet
- By: Asokamitran: "At The Touch of Pancakes, Everyone Becomes A Poet" - PalotDocument9 pagesBy: Asokamitran: "At The Touch of Pancakes, Everyone Becomes A Poet" - PalotHimanshu Jindal100% (2)
- On The Face of It Important Questions Class 12 EnglishDocument7 pagesOn The Face of It Important Questions Class 12 Englishaadi marwah100% (1)
- Lost Spring English Sample PaperDocument8 pagesLost Spring English Sample Paperpalak yadavNo ratings yet
- A Thing of Beauty Class 12thDocument8 pagesA Thing of Beauty Class 12thPrekshi SinghalNo ratings yet
- The Third LevelDocument31 pagesThe Third LevelKrishna SarafNo ratings yet
- Invitation and Replies English Core Class 12: DR Shahzad Ahmad SiddiquiDocument11 pagesInvitation and Replies English Core Class 12: DR Shahzad Ahmad SiddiquiRitzzzzNo ratings yet
- Class 12 Physics Project FileDocument26 pagesClass 12 Physics Project FileDeepan KumarNo ratings yet
- The Lost SpringDocument35 pagesThe Lost SpringAKSHITA GUPTANo ratings yet
- Genetic Disorders ProjectDocument15 pagesGenetic Disorders ProjectAshwin ShajithNo ratings yet
- Topic:: The Magnetic Field Lines Around Current Carrying SolenoidDocument2 pagesTopic:: The Magnetic Field Lines Around Current Carrying SolenoidAbhishek SinghNo ratings yet
- Biology Investigatory Project (Recovered)Document25 pagesBiology Investigatory Project (Recovered)shubham25% (4)
- The InterviewDocument4 pagesThe InterviewRamita Udayashankar100% (1)
- Important Questions For CBSE Class 11 English Hornbill Chapter 1 - The Portrait of A LadyDocument6 pagesImportant Questions For CBSE Class 11 English Hornbill Chapter 1 - The Portrait of A LadyPannaNo ratings yet
- 8 Human Health N Disease-NotesDocument5 pages8 Human Health N Disease-NotesAnanth DharanidharanNo ratings yet
- Birth FinalDocument60 pagesBirth FinalSoujany Agnihotri 9th E100% (2)
- Eng Project On RattrapDocument19 pagesEng Project On Rattrapsparshkumar2438No ratings yet
- Bio Updated ProjectDocument17 pagesBio Updated Projectbholakumar1739No ratings yet
- Physics Project File: MADE BY: - CLASS-12 SectionDocument12 pagesPhysics Project File: MADE BY: - CLASS-12 SectionAryan JindalNo ratings yet
- INDIGODocument11 pagesINDIGORohan ShahNo ratings yet
- Lost Spring NotesDocument2 pagesLost Spring NotesAnujNo ratings yet
- The RattrapDocument5 pagesThe RattrapGarvitNo ratings yet
- The RattrapDocument10 pagesThe RattrapArnab GayenNo ratings yet
- Self Inductance of A CoilDocument8 pagesSelf Inductance of A CoilUnknown RiderNo ratings yet
- A Thing of BeautyDocument12 pagesA Thing of BeautyqfeadvNo ratings yet
- Notes AdvertisementsDocument14 pagesNotes AdvertisementsSentlaNo ratings yet
- Lost SpringDocument39 pagesLost SpringNidhi rastogiNo ratings yet
- The Third LevelDocument2 pagesThe Third LevelHarsha HinklesNo ratings yet
- A Thing of BeautyDocument5 pagesA Thing of BeautyAnanya RayNo ratings yet
- Tiger KingDocument6 pagesTiger KingAman VermaNo ratings yet
- Molecular Basis of Inheritance PyqDocument12 pagesMolecular Basis of Inheritance PyqSoumya Ranjan NaikNo ratings yet
- 12 Biotechnology N Its Applications-NotesDocument3 pages12 Biotechnology N Its Applications-NotesAnanth DharanidharanNo ratings yet
- BIOTECHNOLOGY and Its Applications Class 12Document4 pagesBIOTECHNOLOGY and Its Applications Class 12Albina Zainab P.TNo ratings yet
- Class XII Mock Test PDFDocument1 pageClass XII Mock Test PDFKorou LaikhaNo ratings yet
- Guidelines For Viva and Practical BIOLOGY (Class - XII)Document1 pageGuidelines For Viva and Practical BIOLOGY (Class - XII)Korou LaikhaNo ratings yet
- Useful To Human. Analogues For Products and Services: BIOTECHNOLOGY - Principles and ProcessesDocument10 pagesUseful To Human. Analogues For Products and Services: BIOTECHNOLOGY - Principles and ProcessesKorou LaikhaNo ratings yet
- Guidelines For Viva and Practical BIOLOGY (Class - XII)Document1 pageGuidelines For Viva and Practical BIOLOGY (Class - XII)Korou LaikhaNo ratings yet
- Deleted Portions Class Xi: Biology (Code No. 044)Document5 pagesDeleted Portions Class Xi: Biology (Code No. 044)AarthiKalyanNo ratings yet
- Bullying: Definition, Types, Causes, Consequences and Intervention: BullyingDocument15 pagesBullying: Definition, Types, Causes, Consequences and Intervention: BullyingAlbert JeanezelleNo ratings yet
- Physical Education (Code No.048)Document10 pagesPhysical Education (Code No.048)RachitNo ratings yet
- (Elephas Maximus) During The First Wet Season at TheDocument44 pages(Elephas Maximus) During The First Wet Season at TheAvinash Krishnan100% (1)
- Soal Ulangan Harian Bab 7Document1 pageSoal Ulangan Harian Bab 7Rosma WatiNo ratings yet
- The Pollination and Fertilization Behavior of Fig PlantDocument4 pagesThe Pollination and Fertilization Behavior of Fig PlantIndutripathiNo ratings yet
- Production Guideline For 1 Hectare of GroundnutsDocument1 pageProduction Guideline For 1 Hectare of GroundnutsShadreck MabvoroNo ratings yet
- 1122 - B.sc. Botany Semester-I, IIDocument16 pages1122 - B.sc. Botany Semester-I, IIgungunjain132005No ratings yet
- Lesson 2Document8 pagesLesson 2Kezia Shayen RagualNo ratings yet
- Food Chains and Food Webs DFDocument2 pagesFood Chains and Food Webs DFSYEDA ARSHINo ratings yet
- Klironomos and Hart 2001Document2 pagesKlironomos and Hart 2001José Manuel MejíaNo ratings yet
- AVES - Crop Integration and Crop Production System InventoryDocument4 pagesAVES - Crop Integration and Crop Production System InventoryARLENE MARASIGANNo ratings yet
- Step-by-Step Guide to Sowing SeedsDocument1 pageStep-by-Step Guide to Sowing SeedsJohn Paul Dela PeñaNo ratings yet
- Medical Genetics - I Year, I Semester - List of QuestionsDocument3 pagesMedical Genetics - I Year, I Semester - List of QuestionsGiviko GvinjiliaNo ratings yet
- TranscriptionTranslation ActivityDocument3 pagesTranscriptionTranslation ActivityDylan DiazNo ratings yet
- Objective Plant Pathology BookDocument99 pagesObjective Plant Pathology BookaksriduNo ratings yet
- Types of Succulents ID Cards Alphabetical 3 2 1Document349 pagesTypes of Succulents ID Cards Alphabetical 3 2 1drshamen100% (3)
- Durian CultivationDocument4 pagesDurian CultivationCharlie UyNo ratings yet
- Orchids Possess Minute Seeds Devoid of Endosperm To Nourish The Embryo. This EndospermDocument4 pagesOrchids Possess Minute Seeds Devoid of Endosperm To Nourish The Embryo. This EndospermAdonis MiñanoNo ratings yet
- Cytoplasmic InheritanceDocument13 pagesCytoplasmic Inheritanceanay_379100% (1)
- Cytoplasmic InheritanceDocument7 pagesCytoplasmic Inheritanceanwesh choudhuryNo ratings yet
- DNA Structure and Analysis: Chapter 14 (Translation and Protein)Document2 pagesDNA Structure and Analysis: Chapter 14 (Translation and Protein)Cobe Christian LascunaNo ratings yet
- DnaDocument3 pagesDnaJohn Van Dave TaturoNo ratings yet
- Herbarium Collections of The Botany Department, Fergusson College, Pune, IndiaDocument13 pagesHerbarium Collections of The Botany Department, Fergusson College, Pune, IndiaJa sala DasNo ratings yet
- Gad Kar 2016Document6 pagesGad Kar 2016lacisagNo ratings yet
- Karakteristik Morfologi, Biokimia, Dan Molekuler Isolat Khamir Ik-2 Hasil Isolasi Dari Jus Buah Sirsak (Annona Muricata L.)Document8 pagesKarakteristik Morfologi, Biokimia, Dan Molekuler Isolat Khamir Ik-2 Hasil Isolasi Dari Jus Buah Sirsak (Annona Muricata L.)Rauzah SaniraNo ratings yet
- Adiciones A Chrysobalanaceae PDFDocument270 pagesAdiciones A Chrysobalanaceae PDFBrandon CruzNo ratings yet
- Cyanobacteria and Algae TypesDocument35 pagesCyanobacteria and Algae TypesDreyden Halo100% (1)
- 28 Population Distribution-S Manuel TzulDocument5 pages28 Population Distribution-S Manuel Tzulapi-323831182100% (17)
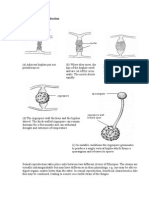
- Rhizopus - Sexual Reproduction: HyphaDocument1 pageRhizopus - Sexual Reproduction: HyphaNovalia Rachmah100% (1)
- rdr2 Spawn FunctionsDocument30 pagesrdr2 Spawn FunctionsFranco F. ZavalaNo ratings yet
- The effects of plant hormones on rooting Hibiscus cuttingsDocument9 pagesThe effects of plant hormones on rooting Hibiscus cuttingsLaurentiu BurlacuNo ratings yet
- Arthropods Associated With Dipterocarp Saplings at Eco-Park Conservation Area, Los Baños, Laguna, PhilippinesDocument7 pagesArthropods Associated With Dipterocarp Saplings at Eco-Park Conservation Area, Los Baños, Laguna, PhilippinesEzekiel InfantadoNo ratings yet