Professional Documents
Culture Documents
COVID-19, ACE2, and The Cardiovascular Consequences
Uploaded by
clara walukowOriginal Description:
Original Title
Copyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
COVID-19, ACE2, and The Cardiovascular Consequences
Uploaded by
clara walukowCopyright:
Available Formats
Invited Perspective “COVID-19, ACE2 …” Am J Physiol Heart Circ
1
2 COVID-19, ACE2, and the Cardiovascular Consequences
3
4
5 Andrew M. South, MD, MS, Debra I. Diz, PhD, FAHA,
6 and Mark C. Chappell, PhD, FAHA
7
8
9
10 Cardiovascular Sciences Center,
11 Wake Forest School of Medicine,
12 Winston-Salem, NC USA.
13
14 Short Title: Coronaviruses and ACE2
15 Total Number of Words: 5294
16 Total Number of Figures: 2
17 Total Number of Tables: 0
18
19
20 Address for Correspondence:
21 Mark C. Chappell, Ph.D.
22 Professor
23 Hypertension & Vascular Research Center
24 Wake Forest University School of Medicine
25 Biotech Place
26 575 N. Patterson Ave.
27 Winston-Salem, NC 27101
28 Telephone: 336-716-9236
29 Fax: 336-716-2456
30 E-mail: mchappel@wakehealth.edu
31 Orcid:
32
1|Page
Downloaded from journals.physiology.org/journal/ajpheart at Univ of Victoria (142.104.240.194) on April 4, 2020.
Invited Perspective “COVID-19, ACE2 …” Am J Physiol Heart Circ
33 Abstract
34 The novel SARS coronavirus SARS-CoV-2 pandemic may be particularly deleterious to
35 patients with underlying cardiovascular disease (CVD). The mechanism for SARS-CoV-
36 2 infection is the requisite binding of the virus to the membrane-bound form of
37 angiotensin-converting enzyme 2 (ACE2) and internalization of the complex by the host
38 cell. Recognition that ACE2 is the co-receptor for the coronavirus has prompted new
39 therapeutic approaches to block the enzyme or reduce its expression to prevent the
40 cellular entry and SARS-CoV-2 infection in tissues that express ACE2 including lung,
41 heart, kidney, brain, and gut. ACE2, however, is a key enzymatic component of the
42 renin-angiotensin-aldosterone system (RAAS); ACE2 degrades Ang II, a peptide with
43 multiple actions that promote CVD, and generates Ang-(1-7) which antagonizes the
44 effects of Ang II. Moreover, experimental evidence suggests that RAAS blockade by
45 ACE inhibitors, AT1 receptor antagonists and mineralocorticoid antagonists, as well as
46 statins enhance ACE2 that, in part, contribute to the benefit of these regimens. In lieu of
47 the fact that many older patients with hypertension or other CVDs are routinely treated
48 with RAAS blockers and statins, new clinical concerns have developed regarding
49 whether these patients are at greater risk for SARS-CoV-2 infection, whether RAAS and
50 statin therapy should be discontinued, and the potential consequences of RAAS
51 blockade to COVID-19-related pathologies such as acute and chronic respiratory
52 disease. The current perspective critically examines the evidence for ACE2 regulation
53 by RAAS blockade and statins, the cardiovascular benefits of ACE2, and whether ACE2
54 blockade is a viable approach to attenuate COVID-19.
55
2|Page
Downloaded from journals.physiology.org/journal/ajpheart at Univ of Victoria (142.104.240.194) on April 4, 2020.
Invited Perspective “COVID-19, ACE2 …” Am J Physiol Heart Circ
56 The rapid and progressive spread of the novel SARS coronavirus SARS-CoV-2
57 pandemic that causes coronavirus-induced disease (COVID-19) has profoundly affected
58 the health of thousands of individuals, strained national health care systems and
59 significantly impacted global economic stability. The characteristics of SARS-CoV-2
60 that particularly distinguish this disease from influenza are a higher transmission rate
61 combined with a greater risk of mortality from COVID-19 primarily due to acute
62 respiratory distress syndrome (ARDS) (16). While the major cause of mortality from
63 COVID-19, particularly in older adults and those with compromised immune systems, is
64 respiratory failure, a number of patients exhibit cardiovascular-related pathologies
65 including congestive heart failure (CHF) and brain medullary cardiorespiratory
66 dysfunction (6,16,32,33,55,60). The cardiovascular complications and the focus on
67 ACE2 as the co-receptor for SARS-CoV-2, as well as the apparent confusion in the
68 literature between the ACE and ACE2 components of the renin-angiotensin-aldosterone
69 system (RAAS) has prompted the current perspective.
70 Viral infections are dependent on cellular entry of the virus that utilize the cellular
71 machinery of the host to replicate multiple viral copies that are subsequently shed by
72 the host cell. Coronaviruses such as SARS-CoV-2 and SARS-CoV-1 are now known to
73 utilize the host protein angiotensin-converting enzyme-2 (ACE2, EC 3.4.17.23) as a co-
74 receptor to gain intracellular entry in the lungs and brain (17,30,52,53,62). ACE2 is a
75 membrane-bound peptidase with the majority of the protein that comprises the N-
76 terminal peptide domain including the catalytic site oriented extracellularly (3.4). ACE2
77 is expressed in essentially all tissues - with greatest activity in the ileum and kidney
78 followed by adipose tissue, heart, brainstem, lung, vasculature, stomach, liver, and
3|Page
Downloaded from journals.physiology.org/journal/ajpheart at Univ of Victoria (142.104.240.194) on April 4, 2020.
Invited Perspective “COVID-19, ACE2 …” Am J Physiol Heart Circ
79 nasal and oral mucosa based on activity data in the mouse that generally parallel ACE2
80 mRNA levels in humans (13,53,62), although discrepancies between mRNA levels and
81 ACE2 activity or expression are evident (10,11,47). ACE2 has access to peptides in
82 the circulation (both maternal and fetal), renal tubular fluid, cerebrospinal fluid, interstial
83 fluid and bronchial fluid. Consensus of evidence from various studies favors a primary
84 role of ACE2 to efficiently degrade Ang II to Ang-(1-7). ACE2 is not an aminopeptidase
85 as recently described by Zheng et al. (60) as its catalytic action that removes the C-
86 terminal phenylalanine residue of Ang II characterizes ACE2 as a carboxypeptidase.
87 This single catalytic event reduces Ang II - the major effector of the RAAS that promotes
88 hypertension (HTN) in part by attenuating baroreceptor sensitivity (BRS) for control of
89 heart rate, and promoting vasoconstriction, sodium retention, oxidative stress,
90 inflammation, and fibrosis, as well as increases the bioactive peptide Ang-(1-7) that
91 opposes the Ang II-AT1 receptor axis through its anti-inflammatory and anti-fibrotic
92 actions, as well enhancing BRS (Figure 1). Thus, the ACE2 peptidase pathway
93 constitutes a key inflexion point in the processing pathway of the RAAS. Consequently,
94 the loss of ACE2 may shift the system to an overall higher Ang II and lower Ang-(1-7)
95 tone (4,5,39). In contrast, ACE forms Ang II and degrades Ang-(1-7) which produces
96 the opposite processing of ACE2 and promotes an increase in blood pressure,
97 inflammation and fibrosis (Figure 1). ACE2 hydrolyzes other peptides including apelin
98 and des-arginine bradykinin (des-Arg1-BK); apelin exhibits cardioprotective actions (48)
99 while des-Arg1-BK promotes inflammation via stimulation of the B1 receptor (44). The
100 extent that these peptides functionally contribute to the effects of altered ACE2 activity
4|Page
Downloaded from journals.physiology.org/journal/ajpheart at Univ of Victoria (142.104.240.194) on April 4, 2020.
Invited Perspective “COVID-19, ACE2 …” Am J Physiol Heart Circ
101 is not well established, although increased levels of des-Arg1-BK enhancing pulmonary
102 inflammation would be deleterious (44).
103 As to the mechanism for the intracellular entry by SARS-CoV-2 and SARS-CoV,
104 the viral coat expresses a protein termed SPIKE (S protein) that contains a receptor
105 binding region that binds to the extracellular domain of ACE2 with high affinity of 15 nM
106 (50). Cleavage of the S protein along dibasic arginine sites by the host protease
107 TMPRSS2 to generate the S1 and S2 subunits is a critical step for S2-induced
108 membrane fusion and viral internalization by endocytosis with ACE2 in the pulmonary
109 epithelium (17,21). The S protein is a not a substrate for ACE2 nor does SARS-CoV-2
110 bind to ACE. Wrapp and colleagues (50) suggest the greater virulence of SARS-CoV-2
111 may reflect that the S1 protein exhibits markedly higher affinity for ACE2 as compared
112 to that of SARS-CoV. ACE2 internalization by SARS-CoV-2 would potentially result in
113 the loss of ACE2 at the cell surface and voids a key pathway for the cell to degrade Ang
114 II and generate the CVD-protective Ang-(1-7). Indeed, an increase in the overall ratio of
115 Ang II:Ang-(1-7) following ACE2 internalization may exacerbate the pulmonary tissue
116 damage initially provoked by SARS-CoV-2. In turn, the reduction in ACE2 may
117 contribute to chronic loss of pulmonary function and increased tissue fibrosis due to
118 COVID-19.
119 The extent to which SARS-CoV-2 infects the heart or other cardiovascular
120 tissues once it enters the circulation and potentially contributes to the myocarditis
121 associated with COVID-19 is unknown (16,18,55). In fact, the impact of SARS-CoV-2
122 on the cardiovascular system apart from the lung is not established. Cardiovascular
123 tissues or cells that express ACE2 are potentially at risk for SARS-CoV-2 infection;
5|Page
Downloaded from journals.physiology.org/journal/ajpheart at Univ of Victoria (142.104.240.194) on April 4, 2020.
Invited Perspective “COVID-19, ACE2 …” Am J Physiol Heart Circ
124 however, other factors including expression of the host proteases that prime the virus
125 are required for infection as well (17,21). In patients with underlying CVD, the loss of
126 ACE2 by SARS-CoV-2 induced internalization would be predicted to exacerbate CVD
127 acutely and perhaps long-term (56). ACE2 is the primary route of Ang II metabolism
128 and Ang-(1-7) generation in the heart and the loss of this carboxypeptidase may
129 compromise cardiac function apart from or in addition to viral infection
130 (3,12,24,38,40,43,56). ACE2 is highly expressed in the tubular epithelium of the kidney
131 and the loss of the enzyme may contribute to altered sodium transport leading to an
132 increase in blood volume and pressure, as well as both acute and chronic effects on
133 kidney injury (4,5,10,22,24,43,50). On the basis of studies on SARS-CoV and recent
134 reports of the presence of viral load in the brainstem with SARS-CoV-2, a similar
135 transfer of virus to the brain by ACE2 may occur via internalization and transport by
136 various cranial nerves (32,35). Certainly neuronal cell death as a result of viral infection
137 would disrupt these vital functions (32). In addition, loss of ACE2 in brain cardiovascular
138 centers short of neuronal death may impair proper autonomic nervous system
139 regulation of blood pressure and potentially respiration (52). The loss of ACE2 in the
140 brainstem may facilitate an increase in sympathetic drive, alterations in the baroreflex
141 and exacerbation of hypertension (1,8,54). Reduced expression of ACE2 in the
142 vasculature may also promote endothelial dysfunction, inflammation and exacerbate
143 existing atherosclerosis and diabetes (9,34,42,45,56,59). A loss of pulmonary ACE2
144 may exacerbate hypertension, respiratory distress and fibrosis post-viral infection
145 (20,30,44). Cell surface diminution of ACE2 may contribute to widespread inflammation
146 observed with COVID-19. We note that the ACE2 protein collectrin that facilitates
6|Page
Downloaded from journals.physiology.org/journal/ajpheart at Univ of Victoria (142.104.240.194) on April 4, 2020.
Invited Perspective “COVID-19, ACE2 …” Am J Physiol Heart Circ
147 amino acid transport and is expressed in multiple tissues (kidney, brain, vasculature,
148 and pancreas) lacks the extracellular peptide domain of ACE2 and is not expected to
149 directly bind and enable internalization of SARS-CoV-2 (7).
150 In contrast to ACE, endogenous circulating levels of soluble ACE2 are generally
151 quite low to non-detectable and would not adequately sequester SARS-CoV-2 in the
152 circulation to prevent viral dissemination (3). While a clinical trial on infusion of
153 recombinant ACE2 was recently proposed and subsequently withdrawn
154 (NCT04287686), the extent that soluble ACE2 would compete for SARS-CoV-2 binding
155 to reduce viremia infection and alleviate tissue injury is unknown. This approach would
156 likely have little impact on viral infection via the respiratory system or the gut, although
157 an increase in circulating ACE2 to augment the Ang-(1-7):Ang II ratio may improve
158 SARS-CoV-2-induced organ injury such as ARDS and attenuate subsequent viral
159 infection of other tissues. However, ACE2 infusion may decrease circulating Ang II and
160 increase Ang-(1-7) levels to the extent that blood pressure dysregulation leading to
161 relative hypotension could occur in patients with COVID-19 patients in later stages of
162 disease including septic or cardiogenic shock (28).
163 Experimental studies generally support the notion that RAAS blockade stimulates
164 ACE2 expression and/or activity, although there appear to be differential responses to
165 AT1 receptor antagonists (ARBs) versus ACE inhibitors (ACEIs), as well as tissue-
166 dependent responses In normotensive Lewis and hypertensive mRen2.Lewis male rats,
167 we found that the ARB losartan increased ACE2 activity in the heart by 2 to 3-fold as
168 shown in Figure 2 (11,22); a similar increase in cardiac ACE2 activity (~2-fold) was
169 reported for the ARB eprosartan in rats with CHF (27). The ACEI lisinopril, however,
7|Page
Downloaded from journals.physiology.org/journal/ajpheart at Univ of Victoria (142.104.240.194) on April 4, 2020.
Invited Perspective “COVID-19, ACE2 …” Am J Physiol Heart Circ
170 either failed to increase cardiac ACE2 activity (Lewis) or stimulated to a lesser extent
171 than losartan (mRen2) (Figure 2), despite similar reductions in blood pressure (11,22)
172 Plasma and cardiac tissue contents of Ang-(1-7) paralleled the increase in ACE2 activity
173 following losartan treatment in the Lewis rats (11). In the kidney of both strains, losartan
174 and lisinopril increased ACE2 activity (10,22), although to a lesser degree compared to
175 the heart (Figure 2). Burchilll et al. (2) found that the ACEI ramipril reduced cardiac
176 ACE2 activity to the level of the control group in a rat model of acute kidney injury (AKI).
177 Wang et al. (49) recently showed that various ARBs (olmesartan, losartan, valsartan,
178 candesartan, telmisartan and irbesartan) all increased ACE2 protein to a similar extent
179 [~2-fold] in the hearts of aorta-constricted mice. In patients with chronic kidney disease
180 (CKD), urinary ACE2 levels (an index of renal tubular expression) those treated with
181 ACEIs or ARBs were similar to the untreated group (34). Furthermore, Lely et al. (31)
182 found no effect of ACEI treatment on ACE2 protein expression in renal biopsy samples
183 from patients with various renal pathologies, as well as in kidney transplant recipients.
184 In contrast, only ACEI-treated patients exhibited an increase in intestinal ACE2 mRNA
185 levels as compared to those on ARBs; however, ACE2 protein or activity were not
186 assessed to validate the mRNA results (46).
187 In the brainstem of older rats, losartan treatment increased ACE2 mRNA levels
188 2-fold; ACE2 was the primary peptidase to generate Ang-(1-7) in this brain region (8,14).
189 Chronic exercise may be another important stimulus of ACE2 in the brain and the
190 periphery (37). In the rostral ventrolateral medulla (RVLM), an exercise regimen
191 markedly increased ACE2 protein as compared to both the control and CHF
192 experimental groups (26). However, this raises the potential issue that while exercise is
8|Page
Downloaded from journals.physiology.org/journal/ajpheart at Univ of Victoria (142.104.240.194) on April 4, 2020.
Invited Perspective “COVID-19, ACE2 …” Am J Physiol Heart Circ
193 clearly associated with improved cardiovascular outcomes in chronic situations,
194 exercise may contribute to a greater risk of SARS-CoV-2 infection. Keidar et al. (24)
195 reported that the mineralocorticoid antagonist spironolactone increased ACE2 activity 4-
196 fold in monocyte-derived macrophages from CHF patients; however, spironolactone
197 failed to increase cardiac ACE2 significantly in experimental CHF (27). Apart from
198 RAAS blockade, experimental studies reveal that statins also augment the ACE2
199 expression. Tikoo et al. (45) reported an increase in ACE2 protein in both heart and
200 kidney [~2-fold] of atorvastatin-treated atherosclerotic rabbits that was associated with
201 epigenetic modifications of the ACE2 gene. Fluvastatin treatment significantly
202 enhanced the effects of insulin to augment cardiac ACE2 protein expression in diabetic
203 rats (41). To our knowledge, the influence of ARB or ACEI treatments combined with
204 statins on ACE2 expression has not been established. Finally, the peroxisome
205 proliferator-activated receptor-γ (PPAR-ɣ) may influence ACE-2 expression as well.
206 The PPAR-ɣ agonist rosiglitazone increased ACE2 protein levels 2-fold in the aorta of
207 hypertensive rats following aortic coarctation (39). Oudit and colleagues (61) found
208 that telmisartan, a partial PPAR-ɣ agonist, also increased ACE2 protein expression in
209 aorta that was associated with greater PPAR-ɣ content in the spontaneously
210 hypertensive rat. The extent that ARBs with PPAR-ɣ agonistic actions such as
211 telmisartan and irbesartan exhibit a greater effect on ACE2 expression in different
212 tissues is unknown, although Wang et al. (49) found no difference in the increase in
213 cardiac ACE2 among six different ARBs that included both telmisartan and irebesartan.
214 The influence of RAAS blockade on ACE2 has not been evaluated thoroughly,
215 but ACEI and ARB treatment may improve outcomes in patients with ARDS (27). In
9|Page
Downloaded from journals.physiology.org/journal/ajpheart at Univ of Victoria (142.104.240.194) on April 4, 2020.
Invited Perspective “COVID-19, ACE2 …” Am J Physiol Heart Circ
216 experimental studies, Yuan et al. (57) reported reduced ACE2 protein in the lungs of
217 rats subjected to chronic smoking, and that losartan treatment was beneficial, but failed
218 to increase ACE2 in either the control or the smoking-exposed groups. However, there
219 are inconsistencies in the stated conclusions of this study that are not supported by the
220 data, as well as the extremely high Ang II content reported in the lung tissue (>10
221 µg/mg or 10 nmol/mg protein) whereby Ang II would comprise 1% of the total protein
222 content in lung (57). In a model of LPS-induced ARDS, losartan improved pulmonary
223 function and inflammation (51). Losartan treatment was associated with higher ACE2
224 activity in bronchoalveolar lavage fluid (BALF) compared to that of the ARB-treated
225 controls; losartan reduced ACE2 activity by 50% in the ventilated control group (49).
226 Changes in Ang II and Ang-(1-7) BALF content evaluated by HPLC-mass spectroscopy
227 paralleled alterations in ACE2 activity (51). We are not aware of studies in animals or
228 humans that have examined the effects of ACEI on pulmonary ACE2, and the
229 discrepancy between ARBs and ACEIs to augment ACE2 activity clearly requires
230 further evaluation. The effect of ARBs or ACEIs on the expression of the SPIKE
231 proteases on the host cell that facilitate binding and entry of SARS-CoV-2 is also
232 unknown. ARBs substantially increase the circulating levels of Ang II arising from the
233 disinhibition of kidney renin release and whether the higher Ang II levels compete for
234 SARS-CoV-2 binding to ACE2 is unclear. In collaborative studies with the McCray lab,
235 we reported that the SPIKE protein from SARS-CoV did not attenuate hydrolysis of Ang
236 II to Ang-(1-7) by soluble ACE2, thus it may be unlikely that Ang II or other peptide
237 substrates would directly interfere with SAR-CoV-2 binding and internalization (23).
10 | P a g e
Downloaded from journals.physiology.org/journal/ajpheart at Univ of Victoria (142.104.240.194) on April 4, 2020.
Invited Perspective “COVID-19, ACE2 …” Am J Physiol Heart Circ
238 Finally, our knowledge of the cardiovascular consequences of SARS-CoV-2
239 infection in patients at this early point is quite limited. Liu et al. (33) recently reported
240 that the circulating levels of Ang II were significantly higher in COVID-19 patients than
241 those of healthy controls that would be consistent with lower ACE2 activity. Moreover,
242 plasma Ang II content significantly correlated with both the viral load in BALF and
243 pulmonary function in the SARS-CoV-2 cohort (33). Thus, as suggested by Liu and
244 colleagues, whether a direct Ang II ACE2 interaction occurs or changes in pulmonary or
245 cardiac function in these patients alters RAAS expression cannot be ascertained.
246 However, this clinical study comprised only 12 patients and circulating ACE2 or ACE
247 levels were not determined. Moreover, an increase in circulating Ang II may reflect
248 changes in a number of RAAS components as opposed to solely a reduction in ACE2
249 activity (Figure 1). Plasma Ang II levels in the Liu et al. (33) study ranged from 100 pM
250 in healthy controls to 500 pM in the SARS-CoV-2 cohort [100-500 pg/ml] using an
251 ELISA-based method in which the patient plasma was directly assayed; these values
252 are 5-10 fold higher than expected Ang II levels in plasma, but are in an acceptable
253 range particularly if the patients were ventilated (3). In contrast, their previous study
254 using a different ELISA reported plasma Ang II values in control subjects of 5 nM [5,000
255 pg/ml] that increased up to 20 nM [20,000 pg/ml] in patients with the influenza A
256 H7N9N1 virus that far exceeds accepted plasma Ang II concentrations (19). This latter
257 study underscores that appropriate methods to accurately quantify Ang II, ACE2 and
258 other RAAS component are vital to establish the role of the RAAS in COVID-19 patients.
259 Biochemical approaches to assess the peptide and protein components of the RAAS in
260 plasma and tissues, as well as the expected endogenous peptide values were recently
11 | P a g e
Downloaded from journals.physiology.org/journal/ajpheart at Univ of Victoria (142.104.240.194) on April 4, 2020.
Invited Perspective “COVID-19, ACE2 …” Am J Physiol Heart Circ
261 reviewed by Chappell (3). The Liu et al. (33) study also failed to include blood pressure
262 data for each patient which could substantiate the higher circulating levels of Ang II.
263 Patients with more severe COVID-19 are reported to have hypokalemia and higher
264 blood pressure as compared to those with milder COVID-19 that would support a role
265 for a stimulated Ang II-AT1 receptor axis (6). Current clinical data on the ACE2-Ang-(1-
266 7) pathway, however, are quite limited relative to the experimental data, especially in
267 regards to the effects of ACEI and ARB. Furthermore, the existing evidence, though
268 novel and insightful, often comes from smaller cross-sectional observational studies
269 with incomplete RAAS measurements that cannot fully account for potential sources of
270 bias and confounding.
271 There are essentially no clinical data on how ACEIs or ARBs may impact the
272 ACE2-Ang-(1-7) pathway in lung, heart or brain. Thus, additional data are urgently
273 needed on the effects of ACEI and ARB on human pulmonary disease and RAAS
274 expression, particularly the response of the ACE2-Ang-(1-7)-Mas receptor axis. Ang-(1-
275 7) itself may potentially serve as a novel therapeutic to treat COVID-19. In both LPS-
276 and acid-induced ARDS with high-stretch ventilation, Ang-(1-7) infusion improved
277 oxygenation, reduced the acute inflammatory response, and reduced subsequent tissue
278 fibrosis (51,58). Clinical trials are in development to test the effects of the ARB losartan
279 in patients with COVID-19 (NCT04311177 and NCT04312009). Upregulation of the
280 ACE2-Ang-(1-7) pathway of the RAAS is well-known to counter-regulate the pro-
281 inflammatory and pro-fibrotic effects of the ACE-Ang II-AT1 receptor axis in experimental
282 models of HTN and CVD (4,5,24,40). Indeed, experimental studies demonstrate that the
283 ACE2-Ang-(1-7) pathway mediates some of the beneficial effects of ACEI and ARB in
12 | P a g e
Downloaded from journals.physiology.org/journal/ajpheart at Univ of Victoria (142.104.240.194) on April 4, 2020.
Invited Perspective “COVID-19, ACE2 …” Am J Physiol Heart Circ
284 these diseases including improved regulation of autonomic control of blood pressure
285 (3,5,40,56), though data in humans remains limited. Experimental evidence to date
286 strongly suggests that Ang II may promote acute lung injury and ARDS induced by
287 coronaviruses, including SARS-CoV, SARS-CoV-2/COVID-19, and possibly in MERS-
288 CoV (20,30,51).
289 We emphasize that further investigation into these potential mechanisms is
290 urgently required, now given the complex interplay of the RAAS and novel
291 coronaviruses such as SARS-CoV-2. Particularly relevant information includes the
292 status of the RAAS at baseline, during and after the infection and during progression of
293 COVID-19 and following recovery. Comprehensive assessments of the full complement
294 of RAAS components across this timeline particularly in concert with medical
295 management of the patient during different phases of the disease are required to
296 establish whether the ACE-Ang II-AT1 receptor versus ACE2-Ang-(1-7)-Mas receptor
297 pathways are beneficial or detrimental at a given point in time. This situation is not
298 surprising since the RAAS has both an immediate role in blood pressure and fluid
299 balance regulation and a longer-term impact on chronic oxidative stress, inflammation
300 and fibrosis. Clinical trials are required for a greater understanding of the impact of
301 treatment with ACEIs and ARBs (as well as statins and aldosterone blockers)
302 medications, known to be highly effective for mitigating the impact of hypertension and
303 diabetes on the heart, kidney and vasculature, on infection rates and severity of disease,
304 in order to directly target our understanding of proposed mechanisms of injury.
305 Moreover, precise, rigorous and appropriate methods are required to correctly
306 characterize the RAAS phenotype, whether in COVID-19 patients or experimental
13 | P a g e
Downloaded from journals.physiology.org/journal/ajpheart at Univ of Victoria (142.104.240.194) on April 4, 2020.
Invited Perspective “COVID-19, ACE2 …” Am J Physiol Heart Circ
307 studies in animals, particularly with respect to ACE2 activity or protein expression that
308 may provide insight into the role of ACE2 in the progression of SARs-CoV-2 infection
309 and, importantly, whether blockade of the peptidase is an appropriate step, at least
310 acutely and targeted to the pulmonary system rather than systemically (56). Thus, a
311 shift to studies of the multi-faceted roles of the RAAS in the setting of infectious disease
312 is warranted, which should not occur in isolation from the other well-known roles of the
313 system in cardiovascular homeostasis, particularly given the recent focus on the RAAS
314 and the balance of Ang II and Ang-(1-7) in critical care medicine such as in septic shock
315 (15,28).
316
317
14 | P a g e
Downloaded from journals.physiology.org/journal/ajpheart at Univ of Victoria (142.104.240.194) on April 4, 2020.
Invited Perspective “COVID-19, ACE2 …” Am J Physiol Heart Circ
318 ACKNOWLEDGMENTS
319
320 This work was supported by grants from NIH HL146818, HD-084227, HL56973;
321 American Heart Association AHA-151521, AHA-18TPA34170522; Cardiovascular
322 Sciences Center CVSC-830114; the Hypertension and Vascular Research Center, and
323 the Farley Hudson Foundation.
324
325
326 DISCLOSURES
327 No conflicts of interest are declared by the authors.
328
15 | P a g e
Downloaded from journals.physiology.org/journal/ajpheart at Univ of Victoria (142.104.240.194) on April 4, 2020.
Invited Perspective “COVID-19, ACE2 …” Am J Physiol Heart Circ
329 LITERATURE CITED
330
331 1. Alenina N, Bader M. ACE2 in brain physiology and pathophysiology: Evidence from
332 transgenic animal models. Neurochemical Research 2019; 44:1323-1329.
333 2. Burchill L, Velkoska E, Dean RG, Lew RA, Smith AI, Levidiotis V, Burrell LM.
334 Acute kidney injury in the rat causes cardiac remodeling and increases
335 angiotensin‐converting enzyme 2 expression. Exp Physiology 2008; 93:622-630.
336 3. Chappell MC. Biochemical evaluation of the renin-angiotensin system. Am. J.
337 Physiol Heart. Circ Physiol 2016; 310:H137-H152.
338 4. Chappell MC, Marshall AC, Alzayadneh EM, Shaltout HA, Diz DI. Update on the
339 ACE2-Angiotensin-(1-7)-MAS receptor axis. Front Endocrinol (Lausanne), 2014;
340 4:201-215.
341 5. Chappell MC. Emerging evidence for a functional ACE2-angiotensin-(1-7)-Mas
342 receptor axis: more than regulation of blood pressure. Hypertension, 2007; 50:
343 596-603.
344 6. Chen D, Li X, Song Q, Hu C, Su F, Dai J. Hypokalemia and clinical implications in
345 patients with Coronavirus Disease 2019 (COVID-19). MedRxiv preprint 2020.
346 7. Danilczyk U, Sarao R, Remy C, Benabbas C, Stange G, Richter A, Arya S,
347 Pospisilik JA, Singer D, Camargo SM, Makrides V, Ramadan T, Verrey F, Wagner
348 CA, Penninger JM. Essential role for collectrin in renal amino acid transport.
349 Nature 2006; 444:1088-1091.
350 8. Diz DI, Garcia-Espinosa MA, Gegick S, Tommasi EN, Ferrario CM, Ann Tallant E,
351 Chappell MC, Gallagher PE. Injections of angiotensin-converting enzyme 2 inhibitor
352 MLN4760 into nucleus tractus solitarii reduce baroreceptor reflex sensitivity for
353 heart rate control in rats. Exp Physiol. 2008; 93:694-700.
354 9. Dong B, Zhang C, Feng JB, Zhao XY, Li SY, Yang YP Overexpression of ACE2
355 enhances plaque stability in a rabbit model of atherosclerosis Arterioscler Thromb
356 Vasc Biol, 2008; 28:1270-1276.
357 10. Ferrario CM, Jessup J, Gallagher PE, Averill DB, Brosnihan KB, Smith R, Chappell
358 MC. Effect of renin-angiotensin system blockade on renal Angiotensin-(1-7)-
359 forming enzymes, ACE-2 and receptors. Kidney Int 2005; 68: 2189-2196.
360 11. Ferrario CM, Jessup J, Chappell MC, Averill DB, Brosnihan KB, Tallant EA, Diz DI,
361 Gallagher PE. Effect of angiotensin converting enzyme inhibition and angiotensin II
362 receptor blockers on cardiac angiotensin converting enzyme 2. Circulation 2005;
363 111:2605-2610.
364 12. Garabelli PJ, Modrall JG, Penninger JM, Ferrario CM, Chappell MC. Distinct roles
365 for angiotensin converting enzyme 2 and carboxypeptidase A in the processing of
366 angiotensins in the heart. Exp Physiology, 2008; 93:613-621.
367 13. Gembardt F, Sterner-Kock A, Imboden H, Spalteholz M, Reibitz F, Schultheiss HP,
368 Siems WE, Walther T. Organ-specific distribution of ACE2 mRNA and correlating
369 peptidase activity in rodents. Peptides 2005, 26:270-1277.
370 14. Gilliam-Davis S, Gallagher PE, Payne VS, Kasper SO, Tommasi EN, Westwood BM,
371 Robbins ME, Chappell MC, Diz DI. Long-term systemic Angiotensin II type 1
372 receptor blockade regulates mRNA expression of dorsomedial medulla renin-
373 angiotensin system components. Physiol Genomics, 2011; 43:829-835.
16 | P a g e
Downloaded from journals.physiology.org/journal/ajpheart at Univ of Victoria (142.104.240.194) on April 4, 2020.
Invited Perspective “COVID-19, ACE2 …” Am J Physiol Heart Circ
374 15. Gleeson PJ, Crippa IA, Mongkolpum W, Cavicchi FZ, Meerhaeghe TF, Brimioulle S,
375 Taccone FS, Vincent JL, Creteur J. Renin as a marker of tissue perfusion and
376 prognosis in critically ill patients. Crit Care Med 2019; 47:152-158.
377 16. Guan W. J., Ni Z. Y., Hu Y., Liang W. H., Ou C. Q., He J. X., et al.. Clinical
378 characteristics of 183 coronavirus disease 2019 in China. N Engl J Med. 2020; doi:
379 10.1056/NEJMoa2002032
380 17. Hoffmann M, Kleine-Wever H, Kruger N, Muller M, Drotsten C, Pholhlmann S. The
381 novel coronavirus 2019 (2019-nCoV) uses the SARS coronavirus receptor ACE2
382 and the cellular protease TMPRSS2 for entry in target cells. Cell 2020; 181: 1-10.
383 18. Huang C, Wang Y, Li X, Ren L, Zhao J, Hu Y, Zhang L, Fan G, Xu J, Gu X, Cheng
384 Z, Hu T, Xia J, Wei Y, Wu W, Xie X, Yin W, Li W, Liu M, Xiao Y, Gao Y , Guo L, Xie
385 J, Wang G, Jiang R, Gao Z, Jin Q, Wang J, Cao B. Clinical features of patients
386 infected with 2019 novel coronavirus in Wuhan, China. Lancet 2020; 395:497-506.
387 19. Huang F, Guo J, Zou Z. Angiotensin II plasma levels are linked to disease severity
388 and predict fatal outcomes in H7N9-infected patients Nature Commun 2014; 5:
389 3595-3602.
390 20. Imai Y, Kuba K, Rao S, Huan Y, Guo F, Guan B, Yang P, Sarao R, Wada T, Leong-
391 Poi H, Crackower MA, Fukamizu A, Hui CC, Hein L, Uhlig S, Slutsky AS, Jiang C,
392 Penninger JM. Angiotensin-converting enzyme 2 protects from severe acute lung
393 failure. Nature 2005; 436:112–116.
394 21. Iwata-Yoshikawa N, Okamura T, Shimizu Y, Hasegawa H, Takeda M, Nagata N.
395 TMPRSS2 contributes to virus spread and immunopathology in the airways of
396 murine models after coronavirus infection. J Virol. 2019; 93:e01815-18.
397 22. Jessup J, Gallagher PE, Averill DB, Brosnihan KB, Ann TE, Chappell MC, Ferrario
398 CM. Effect of Angiotensin II blockade on a new congenic model of hypertension
399 derived from the transgenic Ren-2 rat. Am J Physiol Heart Circ, 2006; 291:H2166-
400 7H2172.
401 23. Jia HP, Look DC, Tan P, Shi L, Hickey M, Gakhar L, Chappell MC, Wohlford-
402 Lenane C, McCray Jr PB. Ectodomain shedding of ACE2 in human airway
403 epithelial. Am J Physiol Lung Cell Mol Physiol. 2009: 297:L84-L96.
404 24. Jiang F, Yang J, Zhang Y, Dong M, Wang S, Zhang Q, Liu FF, Zhang K, Zhang C.
405 Angiotensin-converting enzyme 2 and Angiotensin-(1–7): novel therapeutic targets.
406 Nature Reviews Cardiology, 2014; 45:413-420.
407 25. Keidar S, Gamliel-Lazarovich A, Kaplan M, Pavlotzky E, HamoudS, Hayek T, Karry
408 R, Abassi Z. Mineralocorticoid receptor blocker increases angiotensin-converting
409 enzyme 2 activity in congestive heart failure patients. Circ Res 2005; 97:946-53.
410 26. Kar S, Gao L, Zucker IH. Exercise training normalizes ACE and ACE2 in the brain
411 of rabbits with pacing-induced heart failure J Appl Physiol 2010; 108:923-932.
412 27. Karram T, Abbasi A, Keidar S, Golomb E, Hochberg I, Winarver J, Hoffman A,
413 Abassi. Effects of spironolactone and eprotosartan on cardiac remodeling and ACE
414 isoforms in rats with experimental heart failure. Am J Physiol – Heart Circ 2005;
415 289:H135-H1358.
416
417
17 | P a g e
Downloaded from journals.physiology.org/journal/ajpheart at Univ of Victoria (142.104.240.194) on April 4, 2020.
Invited Perspective “COVID-19, ACE2 …” Am J Physiol Heart Circ
418 28. Khanna A, English SW, Wang XS, Ham K, Tumlin J, Szerlip H, Busse LW, Altaweel
419 L, Albertson TE, Mackey C, McCurdy MT, Boldt DW, Chock S, Young PJ, Krell K,
420 Wunderink RG, Ostermann M, Murugan R, Gong MN, Panwar R, Hästbacka J,
421 Favory R, Venkatesh B, Thompson BT, Bellomo R, Jensen J, Kroll S, Chawla LS,
422 Tidmarsh GF, Deane AM; ATHOS-3 Investigators. Angiotensin II for the Treatment
423 of Vasodilatory Shock. N Engl J Med. 2017; 377:419-430.
424 29. Kim J, Choi SM, Lee J, Park YS, Lee CH, Yim JJ, Yoo CH, Kim YW, Han SK, Lee
425 SM. Effect of Renin-Angiotensin System blockage in patients with Acute
426 Respiratory Distress Syndrome: A retrospective case control study. Korean J Crit
427 Care Med. 2017; 32154–163.
428 30. Kuba K, Imai Y, Rao S, Gao H, Guo F, Guan B, Huan Y, Yang P, Zhang Y, Deng
429 W, Bao L, Zhang B, Liu G, Wang Z, Chappell MC, Liu Y, Zheng D, Leibbrandt A,
430 Wada T, Slutsky AS, Liu D, Qin C, Jiang C, Penninger JM. A crucial role of
431 angiotensin converting enzyme 2 (ACE2) in SARS coronavirus–induced lung injury.
432 Nature Med 2005; 11:875–879.
433 31. Lely AT, Hamming I, van Goor H, Navis GJ. Renal ACE2 expression in human
434 kidney disease. J Pathol 2004; 204:587-593.
435 32. Li YC, Bai WZ, Hashikawa T. The neuroinvasive potential of SARS-CoV2 may play
436 a role in the respiratory failure of COVCID-19 patients. J Med Virol, 2020; 1:1-4.
437 33. Liu Y, Yang Y, Zhang C, Huang F, Wang F, Yuan J, Wang Z, Li J, Li J, Feng C,
438 Zhang Z, Wang L, Peng L, Chen L, Qin Y, Zhao D, Tan S, Yin L, Xu J, Zhou C,
439 Jiang C, Liu L. Clinical and biochemical index form COVID-19 infected patients
440 linked to viral loads and lung injury. Sci China Life Sci 2002; 63:364-374.
441 34. Lovren F, Pan Y, Quan A, Teoh H, Wang G, Shukla PC, Levitt KS, Oudit GY, Al-
442 Omran M, Stewart DJ, Slutsky AS, Peterson MD, Backx PH, Penninger JM, Verma
443 S. Angiotensin converting enzyme-2 confers endothelial protection and attenuates
444 atherosclerosis. Am J Physiol Heart Circ Physiol. 2008; 295:H1377-H1384.
445 35. Mao L, Wang M, Chen S, He Q, Chang J, Hong C, Zhou Y, Wang D, Li Y, Jin H, Bo
446 H. Neurological Manifestations of Hospitalized Patients with COVID-19 in Wuhan,
447 China: a retrospective case series study MedRExiv, 2020,
448 https://doi.org/10.1101/2020.02.22.20026500
449 36. Mizuiri S, Aoki T, Hemmi H, Arita M, Sakai K, Aikawa A. Urinary angiotensin-
450 converting enzyme 2 in patients with CKD. Nephrology 2011; 16:567-572.
451 37. Nunes-Silva A, Rocha GC, Magalhaes DM, Vaz LN, Salviano de Faria MH, Simoes
452 ESAC. Physical exercise and ACE2-angiotensin-(1-7)-mas receptor Axis of the
453 renin angiotensin system. Protein Pept. Lett., 2017; 24:809-81
454 38. Patel VB, Zhong JC, Grant MB, Oudit GY. Role of the ACE2/Angiotensin 1-7 Axis of
455 the Renin-Angiotensin System in Heart Failure. Circ Res. 2016; 118:1313-1326.
456 39. Sánchez-Aguilar M, Ibarra-Lara L, Del Valle-Mondragón L, Rubio-Ruiz ME,
457 Aguilar-Navarro AG, Zamorano-Carrillo A, Ramírez-Ortega MC, Pastelín-
458 Hernández G, Sánchez-Mendoza A. Rosiglitazone, a ligand to PPAR, improves
459 blood pressure and vascular function through Renin-Angiotensin System regulation.
460 PPAR Res 2019; 1371758:1-12.
18 | P a g e
Downloaded from journals.physiology.org/journal/ajpheart at Univ of Victoria (142.104.240.194) on April 4, 2020.
Invited Perspective “COVID-19, ACE2 …” Am J Physiol Heart Circ
461 40. Santos RAAS, Oudit GY, Verano-Braga T, Canta G, Steckelings UM, Bader M. The
462 renin-angiotensin system: going beyond the classical paradigms. Am J Physiol
463 Heart Circ Physiol. 2019; 316:H958-H970.
464 41. Shin YH, Min JJ, Lee JH, Kim EH, Kim GE, Kim MH, Lee JJ, Ahn HJ. The effect of
465 fluvastatin on cardiac fibrosis and angiotensin-converting enzyme-2 expression in
466 glucose-controlled diabetic rat hearts. Heart and Vessels 2017; 32:618–627.
467 42. Sahara M, Ikutomi M, Morita T, Minami Y, Nakajima T, Hirata Y, Nagai R, Sata M.
468 Deletion of angiotensin-converting enzyme 2 promotes the development of
469 atherosclerosis and arterial neointima formation. Cardiovasc Res. 2014; 101:236-
470 246.
471 43. Simões E Silva AC, Teixeira MM. ACE Inhibition, ACE2 and angiotensin-(1-7) axis
472 in kidney and cardiac inflammation and fibrosis. Pharmacol Res 2016, 107: 154-
473 162.
474 44. Sodhi CP, Wohlford-Lenane C, Yamaguchi Y, Prindle T, Fulton WB, Wang S,
475 McCray PB Jr, Chappell MC, Hackam DJ, Jia H. Attenuation of pulmonary ACE2
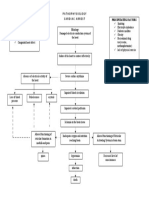
476 activity impairs inactivation of des-Arg9 bradykinin/BKB1R axis and facilitates LPS-
477 induced neutrophil infiltration. Am J Physiol Lung Cell Mol Physiol. 2018; 314:L17-
478 L31.
479 45. Tikoo K, Patel G, Pinakin K, Karpe A, Sanghavi M, Malek V, Srinivasan K. Tissue
480 specific up regulation of ACE2 in rabbit model of atherosclerosis by atorvastatin:
481 Role of epigenetic histone modifications. Biochemical Pharmacology 2015; 93:343-
482 351.
483 46. Vuille-dit-Bille RN, Camargo SM, Emmenegger L, Sasse T, Kummer E, Jando J,
484 Hamie QM, Meier CF, Hunziker S, ForRAAS-Kaufmann Z, Kuyumcu S, Fox M,
485 Schwizer W, Fried M, Lindenmeyer M, Götze O, Verrey F. Human intestine luminal
486 ACE2 and amino acid transporter expression increased by ACE-inhibitors. Amino
487 Acids. 2015; 47:693-705.
488 47. Wang G, Lai FM, Lai KB, Chow KM, Kwan CH, Li KT, Szeto CC. Discrepancy
489 between intrarenal messenger RNA and protein expression of ACE and ACE2 in
490 human diabetic nephropathy. Am J Nephrol. 2009; 29:524-31.
491 48. Wang W, McKinnie SM, Farhan M, Paul M, McDonald T, McLean B, Llorens-Cortes
492 C, Hazra S, Murray AG, Vederas JC, Oudit GY. Angiotensin-Converting Enzyme 2
493 Metabolizes and Partially Inactivates Pyr-Apelin-13 and Apelin-17: Physiological
494 Effects in the Cardiovascular System. Hypertension. 2016; 68:365-377.
495 49. Wang X, Ye Y, Gong H, Wu J, Yuan J, Wang S, Yin P, Ding Z, Kang L, Jiang Q,
496 Zhang W, Li Y, Ge J, Zou Y. The effects of different Angiotensin II Type 1 Receptor
497 blockers on the regulation of the ACE-AngII-AT1 and ACE2-Ang-(1-7)-Mas axes in
498 pressure overload-induced cardiac remodeling in male mice. Mol Cell Cardiol 2016;
499 97:180-190.
500 50. Williams VR, Scholey JW. Angiotensin-converting enzyme 2 and renal disease.
501 Curr Opin Nephrol Hypertens. 2018; 27:35-41.
502
503
19 | P a g e
Downloaded from journals.physiology.org/journal/ajpheart at Univ of Victoria (142.104.240.194) on April 4, 2020.
Invited Perspective “COVID-19, ACE2 …” Am J Physiol Heart Circ
504 51. Woesten-van Asperen RM, Lutter R., Specht P. A., Moll G. N., van Woensel J. B.,
505 van der Loos M, van Goor H, Kamilic J, Florquin S, Bos AP. Acute respiratory
506 distress syndrome leads to reduced ratio of ACE/ACE2 activities and is prevented
507 by angiotensin-(1-7) or an angiotensin II receptor antagonist. J Pathol. 2011;
508 225:618-627.
509 52. Wrapp D, Wang N, Corbett KS, Goldsmith JA, Hsieh CL, Abiona O, Graham BS,
510 McLellan JS. Cryo-EM structure of the 2019-nCoV spike in the prefusion
511 conformation. Science. 2020; 367:1260-1263.
512 53. Xu H, Zhong L, Deng J, Peng J, Dan H, Zeng X, Li T, Chen Q. High expression of
513 ACE2 receptor of 2019-nCoV on the epithelial cells of oral mucosa. Int J Oral
514 Science 2020; 12:8-15.
515 54. Xu P, Sriramula S, Lazartigues E. ACE2/ANG-(1-7)/Mas pathway in the brain: the
516 axis of good. Am J Physiol Regul Integr Comp Physiol. 2011; 300:R804-R817.
517 55. Yang J. Zheng Y, Gou X, Pu K, Chen Z, Guo Q, Ji R, Wang H, Wang Y, Zhou Y.
518 Prevalence of comorbidities in the novel Wuhan coronavirus (COVID-19) infection:
519 a systematic review and meta-analysis, International Journal of Infectious Diseases
520 2020, 9712:30136-3.
521 56. Yousif MHM, Dhaunsi GS, Makki BM, Qabazard BA, Akhtar S, Benter IF.
522 Characterization of Angiotensin-(1-7) effects on the cardiovascular system in an
523 experimental model of type-1 diabetes. Pharmacol Res 2012: 66:269-275.
524 57. Yuan YM, Luo L, Guo Z, Yang M, Ye RS, Luo C. Activation of the renin-angiotensin
525 aldosterone system in the lung smoking induced pulmonary arterial hypertesnion in
526 rats. J Renin-Angiotensin-Aldosterone Syst 2015; 16:249-253.
527 58. Zambelli V, Bellani G, Borsa R, Pozzi F, Grassi A, Scanziani M, Castiglioni V,
528 Masson S, Decio A, Laffey JG, Latini R, Pesentit A. Angiotensin-(1-7) improves
529 oxygenation, while reducing cellular infiltrate and fibrosis in experimental Acute
530 Respiratory Distress Syndrome. Intensive Care Med Exp 2015; 3:8-17.
531 59. Zhang, C, Zhao, YX, Zhang, YH. Angiotensin-converting enzyme 2 attenuates
532 atherosclerotic lesions by targeting vascular cells. Proc Natl Acad Sci U S A 2010;
533 107: 5886–15891
534 60. Zheng, Y., Ma, Y., Zhang, J. Xie X. COVID-19 and the cardiovascular system. Nat
535 Rev Cardiol 2020. https://doi.org/10.1038/s41569-020-0360-5.
536 61. Zhong JC, Ye JY, Jin HY, Yu X, Yu HM, Zhu DL, Gao PJ, Huang DY, Shuster M,
537 Loibner H, Guo JM, Yu JY, Xiao BX, Gong ZH, Penninger JM, Oudit GY
538 Telmisartan attenuates aortic hypertrophy in hypertensive rats by the modulation of
539 ACE2 and profilin-1 expression. Regul Pept, 2011; 166: 90-97.
540 62. Zou X, Chen K, Zou J, Han P, Hao J. Single-cell RNA-seq data analysis on the
541 receptor ACE2 expression reveals the potential risk of different human organs
542 vulnerable to 2019-nCoV infection. Front Med DOI: 10.1007/s11684-020-0754-0,
543 2020.
544
20 | P a g e
Downloaded from journals.physiology.org/journal/ajpheart at Univ of Victoria (142.104.240.194) on April 4, 2020.
Invited Perspective “COVID-19, ACE2 …” Am J Physiol Heart Circ
545 FIGURE LEGENDS
546
547 Figure 1. Processing and functional scheme of the renin-angiotensin system.
548 The protease renin converts the precursor angiotensinogen to Angiotensin I (Ang I)
549 which is subsequently converted to Ang II by dipeptidyl carboxypeptidase angiotensin
550 converting enzyme (ACE). Ang II binds to the AT1 receptor (AT1R) to stimulate
551 inflammation, fibrosis, oxidative stress and an increase in blood pressure. Ang II is
552 metabolized to Ang III and Ang IV through various aminopeptidases (APs). Ang I and
553 Ang II are converted to Ang-(1-7) via endopeptidases (NEP) and the
554 monocarboxypeptidase ACE2, respectively. Ang-(1-7) binds to the Mas-R to exert anti-
555 inflammatory and anti-fibrotic actions, stimulate the release of nitric oxide and reduce
556 blood pressure. Ang-(1-7) is metabolized to Ang-(1-5) by ACE. Major forming and
557 degrading pathways depicted by solid and dashed lines, respectively. SARS-CoV-2
558 binds to ACE2 to stimulate internalization of both the virus and peptidase that may
559 remove ACE2 from this pathway.
560
561 Figure 2. Influence of AT1 receptor or ACE blockade on ACE2 activity in the heart
562 and kidney of normotensive Lewis and hypertensive mRen2.Lewis rats. Chronic
563 blockade with losartan (LOS) increased cardiac ACE2 activity by 3-fold in normotensive
564 Lewis and 2-fold hypertensive mRen2.Lewis (mRen2) male rats. Lisinopril (LIS)
565 treatment had little or no effect on cardiac ACE2 activity in these strains. Chronic LOS
566 or LIS treatment increased renal ACE2 activity in the Lewis [1.3- and 1.7-fold,
567 respectively] and mRen2 [1.3- and 1.2-fold, respectively]. ACE2 activity is the amount
568 of Ang-(1-7) converted from Ang II in the plasma membrane fraction [fmol of Ang-(1-7)/
569 mg protein/minute – fmol/mg/min] sensitive to the ACE2 inhibitor MLN4760 (3). Data
570 are the means ± SEM, *P<0.05 from n of 6-8 (10,11,22).
571
21 | P a g e
Downloaded from journals.physiology.org/journal/ajpheart at Univ of Victoria (142.104.240.194) on April 4, 2020.
Angiotensinogen
Renin
Ang I
ACE NEP
Ang II Ang-(1-7)
AP ACE2 ACE
Ang III Ang-(1-5)
SARS-CoV2
Ang IV
AT1R MAS-R
Inflammation Anti-inflammatory
Fibrosis Anti-fibrotic
Oxidative Stress Nitric Oxide
Vasoconstriction Vasorelaxation
Downloaded from journals.physiology.org/journal/ajpheart at Univ of Victoria (142.104.240.194) on April 4, 2020.
ACE2 Activity
100 Heart – Lewis 100 Heart – mRen2
80 80
fmol/mg/min
60 * * 60
40 40
* *
20 20
0 0
CON LOS LIS CON LOS LIS
Kidney – Lewis Kidney – mRen2
100 100
*
* *
80 80 *
fmol/mg/min
60 60
40 40
20 20
0 Downloaded from journals.physiology.org/journal/ajpheart at Univ of0Victoria (142.104.240.194) on April 4, 2020.
CON LOS LIS CON LOS LIS
You might also like
- Anatomy Dec 2019 - One LinersDocument19 pagesAnatomy Dec 2019 - One LinersJijoNo ratings yet
- OSCE SkillsDocument9 pagesOSCE SkillsgoodbyethereNo ratings yet
- Body Awareness FactsheetDocument2 pagesBody Awareness FactsheetDr.GladsonNo ratings yet
- Alpha Male State of Mind PDFDocument12 pagesAlpha Male State of Mind PDFRideIT0% (1)
- Basic Pharmacokinetic and Pharmacodynamic - Sara - E. - RosenbaumDocument658 pagesBasic Pharmacokinetic and Pharmacodynamic - Sara - E. - Rosenbaumazizah nurul50% (2)
- Hormonal ImbalanceDocument4 pagesHormonal ImbalanceSeeraphNo ratings yet
- Surya KriyaDocument9 pagesSurya KriyaAnonymous VXIcsYNo ratings yet
- Dobutamine Drug StudyDocument1 pageDobutamine Drug Studyzyr2189100% (2)
- Pathophysiology Cardiac ArrestDocument1 pagePathophysiology Cardiac ArrestPATHOSHOPPE100% (2)
- Journal Pre-Proof: European Journal of Internal MedicineDocument30 pagesJournal Pre-Proof: European Journal of Internal MedicineNabil Sangga BuanaNo ratings yet
- Ijms 23 01049Document14 pagesIjms 23 01049Manjeev GuragainNo ratings yet
- EliabethDocument6 pagesEliabethmadelineNo ratings yet
- 1 s2.0 S0014299921000522 MainDocument9 pages1 s2.0 S0014299921000522 MainMuhammad Reza FirdausNo ratings yet
- Bosso Et Al 2020 Role of ACE2 Polymorphisms in HT and COVID19Document7 pagesBosso Et Al 2020 Role of ACE2 Polymorphisms in HT and COVID19Golden Era BookwormNo ratings yet
- Outcomes in Patients With COVID-19 Infection Taking ACEI/ARBDocument4 pagesOutcomes in Patients With COVID-19 Infection Taking ACEI/ARBfk unswagatiNo ratings yet
- 11ACE2 - The Key Molecule For Understanding The Pathophysiology of Severe and Critical Conditions of COVID-19 - Demon or Angel?Document11 pages11ACE2 - The Key Molecule For Understanding The Pathophysiology of Severe and Critical Conditions of COVID-19 - Demon or Angel?chimansqmNo ratings yet
- Artigo 5Document5 pagesArtigo 5Paloma PaixãoNo ratings yet
- Complicaciones Neurológicas de La COVID-19 Desde La Fisiopatología Hasta La Rehabilitación. Una Visión GeneralDocument17 pagesComplicaciones Neurológicas de La COVID-19 Desde La Fisiopatología Hasta La Rehabilitación. Una Visión GeneralB10. Oscar Alonso Díaz PintoNo ratings yet
- Apb 12 641Document4 pagesApb 12 641Irin Susan VargheseNo ratings yet
- COVID 19 in The Heart and The Lungs: Could We "Notch" The Inflammatory Storm?Document8 pagesCOVID 19 in The Heart and The Lungs: Could We "Notch" The Inflammatory Storm?laax_siuyenNo ratings yet
- Brojakowska 2020Document11 pagesBrojakowska 2020Anett Pappné LeppNo ratings yet
- Covid Paper 2Document3 pagesCovid Paper 2John SharkeyNo ratings yet
- Angiotensin-Converting Enzyme 2 (ACE2) Expression Increases With Age in Patients Requiring Mechanical VentilationDocument17 pagesAngiotensin-Converting Enzyme 2 (ACE2) Expression Increases With Age in Patients Requiring Mechanical VentilationJuan Vera SanchezNo ratings yet
- CardiologíaDocument3 pagesCardiologíajakpowerNo ratings yet
- Investigación - CovidDocument12 pagesInvestigación - CovidMauricio FemeniaNo ratings yet
- 2-A Comprehensive Investigation of The MRNA and Protein Level of ACE2, The Putative Receptor of SARS-CoV-2, in Human Tissues and Blood CellsDocument10 pages2-A Comprehensive Investigation of The MRNA and Protein Level of ACE2, The Putative Receptor of SARS-CoV-2, in Human Tissues and Blood CellschimansqmNo ratings yet
- Role of Angiotensin-Converting Enzyme 2 (ACE2) in COVID-19: Review Open AccessDocument10 pagesRole of Angiotensin-Converting Enzyme 2 (ACE2) in COVID-19: Review Open AccessjakpowerNo ratings yet
- Pharmacology of Renin-Angiotensin-System Blockers and COVID-19: Confusion Around ACE2Document3 pagesPharmacology of Renin-Angiotensin-System Blockers and COVID-19: Confusion Around ACE2Sharan SahotaNo ratings yet
- Role of AngiotensinDocument14 pagesRole of Angiotensindebby nirmasariNo ratings yet
- Ace2 & HipertensiDocument21 pagesAce2 & HipertensiPulmoFKUI 2011No ratings yet
- ACE2 OriginalDocument18 pagesACE2 OriginalErika BennaiaNo ratings yet
- How Does Sars-Cov-2 Cause Covid-19?: The Viral Receptor On Human Cells Plays A Critical Role in Disease ProgressionDocument3 pagesHow Does Sars-Cov-2 Cause Covid-19?: The Viral Receptor On Human Cells Plays A Critical Role in Disease ProgressionMiguel Chimal MuñozNo ratings yet
- 2021 Article 2395Document15 pages2021 Article 2395danyescalerapumas24No ratings yet
- Angiotensin InhibitionDocument3 pagesAngiotensin Inhibitionvivitri.dewiNo ratings yet
- 2021 - Association Between Ace2 and Tmprss2 Expression and Covid19Document9 pages2021 - Association Between Ace2 and Tmprss2 Expression and Covid19Gustavo ResendeNo ratings yet
- Exercise Training and Cardiac Rehabilitation in COVID-19 Patients With Cardiovascular Complications: State of ArtDocument16 pagesExercise Training and Cardiac Rehabilitation in COVID-19 Patients With Cardiovascular Complications: State of Artyayu latifahNo ratings yet
- 5400-Article Text-31589-2-10-20210820Document12 pages5400-Article Text-31589-2-10-20210820Voltaire Méndez RodríguezNo ratings yet
- Coronavirus 2019 (Covid-19) Eassociated Encephalopathies and Cerebrovascular Disease: The New Orleans ExperienceDocument10 pagesCoronavirus 2019 (Covid-19) Eassociated Encephalopathies and Cerebrovascular Disease: The New Orleans ExperienceAmandaNo ratings yet
- Nac Papel Antioxidantes Cov-19 0221 FrattaDocument23 pagesNac Papel Antioxidantes Cov-19 0221 FrattaLuis C Ribon VNo ratings yet
- ACE2 CovidDocument15 pagesACE2 CovidLaura Kristel Sánchez HernandezNo ratings yet
- Potential Harmful Effects of Discontinuing Ace-Inhibitors and Arbs in Covid-19 PatientsDocument8 pagesPotential Harmful Effects of Discontinuing Ace-Inhibitors and Arbs in Covid-19 PatientsmadelineNo ratings yet
- Cardiovascular and Renal Risk Factors and ComplicationsDocument16 pagesCardiovascular and Renal Risk Factors and ComplicationskajelchaNo ratings yet
- Covid and Cardiovascular SystemDocument4 pagesCovid and Cardiovascular SystemChristine Jane Rodriguez100% (1)
- Two Things About COVID-19 Might Need AttentionDocument10 pagesTwo Things About COVID-19 Might Need AttentionRobertus RonnyNo ratings yet
- Imaging in COVID 19 Related Myocardial InjuryDocument12 pagesImaging in COVID 19 Related Myocardial Injuryorkut verificationNo ratings yet
- A Brand-New Cardiorenal Syndrome in The COVID-19 SettingDocument6 pagesA Brand-New Cardiorenal Syndrome in The COVID-19 SettingfannyNo ratings yet
- Covid y Sistema EndocrinoDocument22 pagesCovid y Sistema EndocrinocristinaNo ratings yet
- E134 FullDocument3 pagesE134 FullRahmanu ReztaputraNo ratings yet
- A Nicotinic Approach To CovidDocument11 pagesA Nicotinic Approach To Covidraphael.hunzingerNo ratings yet
- Molecular Basis of Cardiac and Vascular Injuries Associated With COVID-19Document15 pagesMolecular Basis of Cardiac and Vascular Injuries Associated With COVID-19Aji Prasetyo UtomoNo ratings yet
- Clinical Implications of SARS ESPDocument12 pagesClinical Implications of SARS ESPAnthony HopkinsNo ratings yet
- Endothelial Dysfunction in COVID-19 Potential Mechanisms and Possible Therapeutic OptionsDocument24 pagesEndothelial Dysfunction in COVID-19 Potential Mechanisms and Possible Therapeutic OptionssamuelNo ratings yet
- SARS-CoV-2 Viremia Is Associated With Distinct Proteomic Pathways and Predicts COVID-19 OutcomesDocument13 pagesSARS-CoV-2 Viremia Is Associated With Distinct Proteomic Pathways and Predicts COVID-19 OutcomesYoga Nugraha WNo ratings yet
- COVID-19 Update: Covid-19-Associated Coagulopathy: Richard C. BeckerDocument14 pagesCOVID-19 Update: Covid-19-Associated Coagulopathy: Richard C. Beckersumenep nursalamNo ratings yet
- Cholesterol Covid19Document9 pagesCholesterol Covid19Rheum RaponticumNo ratings yet
- 8 DBFDocument4 pages8 DBFFatimaNo ratings yet
- 2020 Article 2134Document14 pages2020 Article 2134Maysa HasanahNo ratings yet
- 13 ACE2 Receptor For Covid19Document11 pages13 ACE2 Receptor For Covid19mmaNo ratings yet
- Estudio - Secuelas COVID - TesticulosDocument8 pagesEstudio - Secuelas COVID - TesticulosmaryserranoNo ratings yet
- Covid-19 Science Report:: Pathogenesis and Host Immune Response To Sars-Cov-2Document12 pagesCovid-19 Science Report:: Pathogenesis and Host Immune Response To Sars-Cov-2ChavdarNo ratings yet
- Consequences of COVID-19 For The PancreasDocument14 pagesConsequences of COVID-19 For The PancreasNormanNo ratings yet
- Covid-19: The Potential Role of Copper and N-Acetylcysteine (Nac) in A Combination of Candidate Antiviral Treatments Against Sars-Cov-2Document22 pagesCovid-19: The Potential Role of Copper and N-Acetylcysteine (Nac) in A Combination of Candidate Antiviral Treatments Against Sars-Cov-2FREILEN BRAYAN HUANCA CUSINo ratings yet
- A Review On Sars Cov 2 Virology, Pathophysiology, Animal Models, and Anti Viral InterventionsDocument22 pagesA Review On Sars Cov 2 Virology, Pathophysiology, Animal Models, and Anti Viral InterventionsalodiarkNo ratings yet
- International Journal of Cardiology HypertensionDocument3 pagesInternational Journal of Cardiology HypertensionEndah StyaNo ratings yet
- Kelor DocingDocument12 pagesKelor DocingtridewantiwNo ratings yet
- Antihypertensive Treatment With ACEIARB of Patients With COVID-19 Complicated by HypertensionDocument3 pagesAntihypertensive Treatment With ACEIARB of Patients With COVID-19 Complicated by HypertensionDoctoRides 46No ratings yet
- Covid y CorazónDocument16 pagesCovid y CorazóndanielaNo ratings yet
- JMV 9999 NaDocument2 pagesJMV 9999 NamadelineNo ratings yet
- Class 10 Notes CH Life ProcessesDocument15 pagesClass 10 Notes CH Life ProcessesNITU AGARWALNo ratings yet
- Anatomy & Physiology: Seeley'sDocument48 pagesAnatomy & Physiology: Seeley'sNatron PickettNo ratings yet
- Chapter 7: Muscular System: Movement: PSYN 103: Human Anatomy and PhysiologyDocument67 pagesChapter 7: Muscular System: Movement: PSYN 103: Human Anatomy and PhysiologyWindy LavarientosNo ratings yet
- Physiological Demands of The Firefighter Candidate Physical Ability TestDocument11 pagesPhysiological Demands of The Firefighter Candidate Physical Ability TestNicholaus Leka MushiNo ratings yet
- Histology of The Endocrine System: Ma. Minda Luz M. Manuguid, M.DDocument27 pagesHistology of The Endocrine System: Ma. Minda Luz M. Manuguid, M.Dchocoholic potchiNo ratings yet
- Parkinsonism and Related Disorders: David A. Low, Ekawat Vichayanrat, Valeria Iodice, Christopher J. MathiasDocument5 pagesParkinsonism and Related Disorders: David A. Low, Ekawat Vichayanrat, Valeria Iodice, Christopher J. MathiasRafael Yokoyama FecchioNo ratings yet
- Award Ceremony SpeechDocument3 pagesAward Ceremony Speechapi-192784092No ratings yet
- Understanding Motor Awareness Through Normal and Pathological BehaviorDocument7 pagesUnderstanding Motor Awareness Through Normal and Pathological BehaviorDiretoria CientíficaNo ratings yet
- CiticolineDocument2 pagesCiticolineKerima Danica Lising GayoNo ratings yet
- Naw TrainingDocument34 pagesNaw Trainingganesh pNo ratings yet
- Racial, Ethnic, Religious and Linguistic Elements in Indian PopulationDocument50 pagesRacial, Ethnic, Religious and Linguistic Elements in Indian PopulationPD18100% (3)
- BacteriophageDocument4 pagesBacteriophageapi-294992872No ratings yet
- PhenacetinDocument16 pagesPhenacetinJericSalcedoNo ratings yet
- Terapi Cairan Resusitasi: DR Arief Munandar, Span, Kic Bagian Anestesi - Icu Rsud BrebesDocument34 pagesTerapi Cairan Resusitasi: DR Arief Munandar, Span, Kic Bagian Anestesi - Icu Rsud BrebesWistha MiyakiNo ratings yet
- Pancytopenia - A Clinicopathological Analysis of 132 CasesDocument11 pagesPancytopenia - A Clinicopathological Analysis of 132 CasesKaye Antonette AntioquiaNo ratings yet
- Artikel SkripsiDocument12 pagesArtikel SkripsiSari RofiqohNo ratings yet
- Diagnostic Approach of Short Stature / Stunted (Jan 2015) - Prof DR Jose BatubaraDocument49 pagesDiagnostic Approach of Short Stature / Stunted (Jan 2015) - Prof DR Jose BatubaraGerakan Kesehatan Ibu dan AnakNo ratings yet
- PharmD Course StructureDocument3 pagesPharmD Course StructureHaripratap SikarwarNo ratings yet
- PacemakerDocument24 pagesPacemakerرزاق ابو خالدNo ratings yet
- Urinalisis MikroskopisDocument26 pagesUrinalisis MikroskopisShandy Bethan100% (1)
- Hes 386 Poster Breanna CrockettDocument2 pagesHes 386 Poster Breanna CrockettManuel FerreiraNo ratings yet