Professional Documents
Culture Documents
Sulfuric Acid Pressure Leaching of Nickeliferous Limonites: Metallurgical and Materials Transactions B January 1977
Uploaded by
andyOriginal Title
Copyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
Sulfuric Acid Pressure Leaching of Nickeliferous Limonites: Metallurgical and Materials Transactions B January 1977
Uploaded by
andyCopyright:
Available Formats
See discussions, stats, and author profiles for this publication at: https://www.researchgate.
net/publication/226382174
Sulfuric acid pressure leaching of nickeliferous limonites
Article in Metallurgical and Materials Transactions B · January 1977
DOI: 10.1007/BF02669329
CITATIONS READS
41 217
3 authors, including:
Paul B. Queneau
The Bear Group; Colorado School of Mines
46 PUBLICATIONS 319 CITATIONS
SEE PROFILE
All content following this page was uploaded by Paul B. Queneau on 16 July 2015.
The user has requested enhancement of the downloaded file.
Sulfuric Acid Pressure Leaching of
Nickeliferous Limonites
E. C. CHOU, P. B. Q U E N E A U , AND R. S. RICKARD
T h i s l a b o r a t o r y study e x a m i n e s the s u l f u r i c a c i d p r e s s u r e l e a c h i n g c h a r a c t e r i s t i c s of
n i c k e l i f e r o u s l i m o n i t e s a m p l e s f r o m South New C a l e d o n i a . T h e e f f e c t of t e m p e r a t u r e
(225~ to 300~ a c i d - t o - o r e r a t i o (0.21 to 0.30), p ct s o l i d s (10 to 45), p a r t i c l e s i z e (--20
to - 2 7 0 m e s h ) , and a g i t a t i o n (50 to 600 r p m ) on n i c k e l e x t r a c t i o n and s e l e c t i v i t y a r e d i s -
c u s s e d . F o r o v e r a l l i m p r o v e m e n t of the l e a c h r e s p o n s e , l e a c h i n g i s b e s t c o n d u c t e d at
t e m p e r a t u r e s h i g h e r than 250~ and at s o l i d s l e s s than 33 pct. V a r i a t i o n of g o e t h i t e ,
h e m a t i t e , and b a s i c a l u m i n u m s u l f a t e c on t en t of the l e a c h r e s i d u e was d e t e r m i n e d s e m i -
q u a n t i t a t i v e l y a s a function of l e a c h i n g t i m e . S u l f a t e co n t en t of the r e s i d u e r i s e s s h a r p l y
d u r i n g the f i r s t s e v e r a l m i n u t e s of l e a c h i n g , then f a l l s , and f i n a l l y e q u i l i b r a t e s at f i v e
to s i x p e r c e n t . T h i s c h a n g e in s u l f a t e a n a l y s i s can be e x p l a i n e d by a s s u m i n g a s e r i e s of
c o n s e c u t i v e i r o n and a l u m i n u m h y d r o l y s i s r e a c t i o n s .
T HE w o r l d ' s l a r g e s t known land b a s e d r e s e r v e s of t i c u l a r a t t e n t i o n is g i v e n to the f i r s t p o r t i o n of the
n i c k e l o c c u r as n i c k e l i f e r o u s l i m o n i t e s . l L i m o n i t e s leaching period.
a r e r i c h in i r o n (40 to 50 pct), c h i e f l y as g o e t h i t e ,
(FeOOH), but low in m a g n e s i a (<5 pct) and s i l i c a (<10
CHARACTERIZATION OF ORE
pct). N i c k e l content s e l d o m e x c e e d s 1.5 pct and
usual l y l i e s b e t w e e n 0.9 and 1.4 pct; the cobalt a s s a y The weight distribution of the size fractions and as-
is t y p i c a l l y 0.1 pct. 2 L a r g e s c a l e n i c k e l e x t r a c t i o n of sociated chemical analyses of the South New Caledonia
l i m o n i t e s u t i l i z e d e i t h e r r e d u c t i v e - r o a s t i n g plus a m - oxide ore used in this study are tabulated in Table I
moniacal atmospheric leaching, or sulfuric acid below.
p r e s s u r e l e a c h i n g . 3 T h i s p a p e r e x a m i n e s the l a t t e r , The - 3/+20 mesh fraction was removed by screen-
which has the a d v a n t a g e of high n i c k e l and cobalt ex - ing because of its low nickel and cobalt content. The
t r a c t i o n at the e x p e n s e of high p r e s s u r e o p e r a t i o n . remaining minus 20 mesh ore was designated Sample
R e s u l t s p r e s e n t e d w e r e obtained at A M A X ' s E x t r a c - "A", and was the feed material used in this study.
t i v e M e t a l l u r g y L a b o r a t o r y a s p a r t of t h e i r d e v e l o p - The fine fractions (-270 mesh of Sample A) are rich
m e n t p r o g r a m f o r r e c o v e r i n g n i c k e l and cobalt f r o m in nickel and iron, whereas the coarse fractions are
oxide o r e s . rich in cobalt, magnesium, manganese, chromium
P u b l i s h e d i n f o r m a t i o n on s u l f u r i c a c i d p r e s s u r e (mostly as chromite) and aluminum. X-ray diffraction
l e a c h i n g of l i m o n i t e is s c a r c e . W o r k p e r f o r m e d in analysis showed the major mineral constituent of Sam-
s u p p o r t of a l a r g e s c a l e o p e r a t i o n at Moa Bay in Cuba ple A was goethite. Also present were lesser quanti-
was f i r s t d e t a i l e d by C a r l s o n and S i m o n s , 4 then e x - ties of quartz, kaolinite, talc, hematite, serpentine,
tended by Sobol s-7 and C h a v e s 8 At Moa Bay, o r e was montmorillonite, chromite and lithiophorite. The in-
wet s c r e e n e d at 20 m e s h , t h i c k e n e d to 45 pct s o l i d s , frared spectra both of Sample A and of a typical limoni-
and p r e s s u r e l e a c h e d at 240 to 250~ with s u l f u r i c tic ore sample from Moa Bay were also obtained. Figure
a c i d. To a t t a i n 95 pct n i c k e l e x t r a c t i o n in 60 to 90 1 shows that both samples have similar characteristic
min, 0.22 pounds of c o n c e n t r a t e d s u l f u r i c a c i d p e r absorption bands except for Sample A's typical sili-
pound of o r e was pumped into the f i r s t a u t o c l a v e of a cate and quartz bands at wavenumber 1020 and II00
four-autoclave train. The pregnant liquor generated cm -I, respectively. The presence of this band indicates
a s s a y e d 6 gpl Ni, 0.8 gpl F e , 2.3 gpl A1, and 28 gpl
f r e e acid. C a r l s o n and S i m o n s r e p o r t e d on the k i n e t i c s
and m e c h a n i s m of this }oressure l e a c h , d e m o n s t r a t i n g
the i m p o r t a n c e of a c i d / o r e r a t i o and t e m p e r a t u r e ; Table I. Size Fractions and Analyses of New Cale4~nian Laterite Ores
k i n e t i c s of n i c k e l e x t r a c t i o n w e r e e x p l a i n e d by t h r e e
Sample
n i c k e l - b e a r i n g s p e c i e s , but no p r o o f was given. T h e Size Fraction, -3 -20 100 -270 "A"
r e m a r k a b l e f e a t u r e of t h e i r w o r k was the s h a p e of the Mesh +20 +100 +270 +400 400 -20
n i c k e l e x t r a c t i o n c u r v e s . Most of the l e a c h i n g was
c o m p l e t e in the f i r s t few m i n u t e s , with l i t t l e e x t r a c - Wt Pct 12 8 7 4 69 88
Assays,Pct
tion o c c u r r i n g d u r i n g the r e m a i n i n g h o u r . T h e p r e s e n t Ni 0.67 1.08 1.77 1.84 1.80 1.73
i n v e s t i g a t i o n e x t e n d s that of C a r l s o n and S i m o n s . P a r - Co 0.11 0.37 0.42 0.25 0.11 0.16
Fe 45.9 30.9 29.2 35.0 44.5 41.6
Mg 1.4 3.0 2.4 2.2 1.3 1.5
Mn 0.69 1.6 1.6 1.0 0.50 0.71
E. C. CHOU and P. B. QUENEAU are Section Chiefs in AMAX's Cr 2.9 7.0 4.9 3.3 1.3 2.2
Extractive Metallurgy Laboratory, Golden, CO 80401. R. S. RICKARD A1 3.5 4.1 3.0 2_5 2.4 2.6
is Vice-President, Engineering, Earth Sciences, Inc., Golden, CO. SiO2 8.0 16.3 26.1 21.6 10.8 13.0
Manuscript submitted December 31, 1976.
METALLURGICAL TRANSACTIONS B VOLUME 8B, DECEMBER 1977 547
Screening Analysis
The weight d i s t r i b u t i o n of the s i z e f r a c t i o n s was d e -
t e r m i n e d by w e t - s c r e e n technique u s i n g T y l e r s c r e e n s .
RELATIVE
Surface A r e a D e t e r m i n a t i o n
ABSORBANCE The s u r f a c e a r e a of Sample A was m e a s u r e d with a
Q u a n t a c h r o m e s u r f a c e m e a s u r e m e n t unit m a n u f a c t u r e d
by Q u a n t a s o r b Company.
CHEMISTRY OF LEACHING
,ooo 3~oo ~oo ,5'oo ,o~o ~o Sulfuric acid leaching of n i c k e l i f e r o u s l i m o n i t e is
p e r f o r m e d at t e m p e r a t u r e s high enough to s o l u b i l i z e
WAVENUMSER, CM -I
n i c k e l and cobalt as aqueous s u l f a t e s while c o n v e r t -
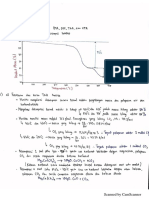
Fig. 1--The infrared spectra of Moa Bay Limonite and Sam- ing most of the o r e ' s i r o n and a l u m i n u m content to in-
ple A and the residues obtained by leaching Sample A at
250~ 33 pct solids and 0.24 acid/ore ratio. solubIe h e m a t i t e and a l u m i n u m s u l f a t e s : 4
2FeOOH + 6H* ~ 2Fe §247247
+ 4H20 [1]
2Fe §247+ SO4= + 3H20 ~-~ Fe203 'SO3 'H20 + 4H § [2]
that Sample A is not a pure l i m o n i t e , but contains some
FeeO3 "SO3 "HeO ~-~ FezO3 + SO4= + 2H § [3]
s i l i c a t e m i n e r a l s , which a r e m a j o r c o n s t i t u e n t s in
s e r p e n t i n i c r o c k s . Such c o n t a m i n a t i o n is difficult to 6A1~§ + 4SO4= + 14H20 ~- 3A1203 94SO3 99H20
avoid when m i n i n g l i m o n i t e deposits in which the
+ 10H § [4]
b o u n d a r y between the l i m o n i t e and u n d e r l y i n g s e r -
p e n t i n e o r e is not well defined. F i g u r e 1 is d i s c u s s e d 2AI~§ + 2SO4: + 2H20 ~ A1203 " 2SO3 "HeO + 2H*. [5]
f u r t h e r in the E x p e r i m e n t a l R e s u l t s Section.
R e a c t i o n [2] takes place d u r i n g the f i r s t few m i n u t e s
of leaching when acidity is high, 9 then the b a s i c i r o n
E X P E R I M E N T A L PROCEDURE sulfate p r o d u c t of R e a c t i o n [2] is h y d r o l y z e d to h e m a -
tite by R e a c t i o n [3]. A l u m i n u m h y d r o l y z e s m o r e
Leaching
slowly, g e n e r a t i n g mostly 3 94 99 hydrate below 250~
A i r - d r i e d o r e was held at 40~ overnight u n d e r 15 (Reaction [4]) and the 1 . 2 91 hydrate above 280~
m m m e r c u r y p r e s s u r e (to a s s u r e c o n s i s t e n t m o i s t u r e (Reaction [5]).s The quantity of 1 . 2 91 hydrate also in-
content), weighed, s a m p l e d and mixed with w a t e r for c r e a s e s with acidity, even at t e m p e r a t u r e s below
15 m i n in a r o i l e d bottle. The r e s u l t i n g s l u r r y was 250~176
t r a n s f e r r e d into a g l a s s - l i n e d t w o - l i t e r P a r r t i t a n i u m
autoclave and brought to t e m p e r a t u r e over a 40 rain
E X P E R I M E N T A L RESULTS
p e r i o d . Two axial t u r b i n e s (5.7 cm diam) mounted on a
c e n t r a l shaft r o t a t i n g at 600 r p m provided a m p l e m i x - I n f r a - r e d s p e c t r a of Sample A and of its leach r e s i -
ing. To begin a test, s u l f u r i c acid (96 pct) was in- due (250~ 0.24 a c i d / o r e , 33 pct solids) a p p e a r in Fig.
jected u n d e r n i t r o g e n o v e r p r e s s u r e through a tube 1. The five b a n d s at 440, 600, 800, 900 and 3180 cm -1
e x t e n d i n g about 5 c m downward f r o m the autoclave a r e a t t r i b u t e d to goethite. The i n t e n s i t y of t h e s e bands
head. S a m p l e s were taken through s a m p l i n g b o m b s at d e c r e a s e s d r a m a t i c a l l y d u r i n g the f i r s t 5 m i n of l e a c h -
p r o g r a m m e d t i m e s , filtered, and the f i l t e r cakes ing and b e c o m e s i n s i g n i f i c a n t a f t e r 15 min. The bands
washed with pH 2 s u l f u r i c acid solution. Cakes were at 1020 cm -1 and 1100 cm -1 a r e a t t r i b u t e d to s i l i c a t e
d r i e d o v e r n i g h t at l l 0 ~ m i n e r a l s and q u a r t z , r e s p e c t i v e l y . The 1020 cm -1 band
d e c r e a s e s and the 1100 cm -~ band r e m a i n s unchanged
with i n c r e a s i n g leaching t i m e , i n d i c a t i n g that the s i l i -
I n f r a - R e d S p e c t r o s c o p y (IR)
cate m i n e r a l s d i s s o l v e d u r i n g l e a c h i n g w h e r e a s q u a r t z
The i n f r a - r e d s p e c t r a of Sample A and its leach does not. (Si a n a l y s i s of p r e g n a n t liquor is typically
r e s i d u e were obtained u s i n g the KBr p e l l e t technique 1 gpl.) T h r e e bands at 340, 475, and 550 cm -~ a r e at-
with a B e c k m a n Model 4250 I n f r a - R e d S p e c t r o p h o t o m e - t r i b u t e d to h e m a t i t e . The i n t e n s i t y of these bands
ter. show the i n c r e a s i n g f o r m a t i o n of h e m a t i t e with in-
c r e a s i n g leach t i m e . T h e r e is an unidentified band at
X - R a y D i f f r a c t i o n (XRD) about 1200 cm -~ which may be a t t r i b u t e d to the a l u n i t e .
Two bands at 1640 cm -1 and 3440 cm -~ probably o r i g i -
The XRD study of Sample A and its leach r e s i d u e nate f r o m c o m b i n e d water (i.e., hydroxyl group) in
was conducted u s i n g a c o p p e r - t a r g e t X - r a y tube with v a r i o u s m i n e r a l s . T h u s , the IR data do give an indi-
a Norelco 2500 XRD unit and an AMR m o n o c h r o m a t o r . cation of the b e h a v i o r of m i n e r a l s d u r i n g leaching.
In addition, goethite and h e m a t i t e identified by XRD
D i f f e r e n t i a l T h e r m a l A n a l y s i s (DTA) and a l u n i t e identified by DTA w e r e found in the s a m -
pie and its leach r e s i d u e . The r e l a t i v e q u a n t i t i e s as
The DTA study of Sample A and its leach r e s i d u e c o m p a r e d to the c o r r e s p o n d i n g pure m i n e r a l s a r e il-
was conducted with a D e l t a t h e r m III Unit m a n u f a c t u r e d l u s t r a t e d in T a b l e II as a function of l e a c h i n g t i m e .
by T e c h n i c a l E q u i p m e n t C o r p o r a t i o n . A p o r t i o n of the c h r o m i u m content of the ore also
548-VOLUME 8B, DECEMBER 1977 METALLURGICALTRANSACTIONS B
III p e r f o r m e d in g r a d u a t e d c y l i n d e r s on the d i s c h a r g e
Table II. Phases Found in Acid Leach Residues of "Sample A" Ore s l u r r y with no r a k e s o r f l o c c u l a n t s i n d i c a t e d that
(250~ 0.24 acid/ore, 33 pct solids)
u l t i m a t e c o m p a c t i o n d e c r e a s e d and t i m e to r e a c h the
Leaching Goethite, Hematite, Alunite, Free Acid, c o m p r e s s i o n p o i n t i n c r e a s e d with i n c r e a s i n g a g i t a t i o n
Time, min FeOOH Fe~O3 3AI~O3-4SO4"9H20 gpl d u r i n g the l e a c h . In g e n e r a l , the r a t e of s e t t l i n g in-
c r e a s e s with i n c r e a s i n g of p a r t i c l e s i z e . F i n e p a r t i -
0 40 Trace Absent 130
1 30 30 Absent 38 c l e s p r o d u c e d u n d e r high s h e a r c o n d i t i o n s p r o b a b l y in-
3 20 45 Absent 34 hibit the s e t t l i n g ; u n f o r t u n a t e l y , the p a r t i c l e s i z e d i s -
5 10 60 Trace 34 t r i b u t i o n was not d e t e r m i n e d d u r i n g the p e r i o d of t e s t -
15 5 65 Minor 37 ing, which m a y e x p l a i n why the b e s t s e t t l i n g c o n d i t i o n s
30 Nil 70 Minor 33 a r e in the 2 7 5 ~ pct s o l i d s - l o w s h e a r c a s e .
60 Nil 75 Minor 38
Low s h e a r r e f e r s to m i x i n g with a p e r f o r a t e d pad-
dle at 50 r p m , and high s h e a r r e f e r s to m i x i n g with the
twin t u r b i n e s at 600 r p m . In a l l of the 'n o a g i t a t i o n "
t e s t s , the a c i d added to b e g i n the l e a c h was m i x e d with
Table III. Effect of Agitation on Thickening Response When the o r e - w a t e r s l u r r y f o r 30 s at 600 r p m .
Pressure Leaching Sample "'A"
(Acid/Ore = 0.24)
E f f e c t of T e m p e r a t u r e
Leach
F i g u r e 3 i l l u s t r a t e s the e f f e c t of t e m p e r a t u r e on the
Type of Temperature, 24 h Compression r a t e and e x t e n t of n i c k e l e x t r a c t i o n . T e m p e r a t u r e s a p-
Agitation ~ Solids, Pct Compaction,Pet Point, h
p r o a c h i n g 250~ a r e r e q u i r e d f o r r a p i d l e a c h i n g .
None 250 33 44 4.9 C o m p a r a b l e e x t r a c t i o n , but at a h i g h e r i n i t i a l r a t e , is
Low shear 250 33 44 6.3 a t t a i n e d at 275~ l e a c h i n g at 300~ l o w e r s the n i c k e l
Highshear 250 33 42 7.3 y i e l d . In a n a l y z i n g data p r e s e n t e d in F i g , 3, the f i r s t
None 250 45 53 2.5
Low shear 250 45 48 None o r d e r a s s u m p t i o n f i t s the d a t a f a i r l y w e l l in the r a n g e
Highshear 250 45 46 None of 75 p ct to 90 pct n i c k e l l e a c h e d , but the r a t e is
None 275 45 52 4.0 m u c h h i g h e r e a r l y in the r e a c t i o n . B a s e d on A r r h e n i u s
Low shear 275 45 60 2.3 equation, a plot of log k v s 1/T (K) in F i g . 4 show that
High shear 275 45 51 None
the e n e r g y of a c t i v a t i o n c a l c u l a t e d b e t w e e n 225 ~ and
275~ is a p p r o x i m a t e l y 30 K c a l / g m o l e .
C u r v e s s i m i l a r to t h o s e shown in F i g . 3 could have
b e e n p l o t t ed f o r c o b a l t e x t r a c t i o n , and would have
d i s s o l v e s , but is to a l a r g e extent h y d r o l y z e d d u r i n g shown that u l t i m a t e cobalt e x t r a c t i o n l a g s that of
the l a t e r s t a g e of l e a c h i n g (probably as Cr3(SO4)2 • n i c k e l by 3 to 4 pct. H o w e v e r , the i n i t i a l r a t e of c o -
(OH)sHeO (Ref. 12) and 3A12Oa 9 2CRO3 9 6HeO. 13 About b a l t e x t r a c t i o n was a l w a y s h i g h e r than that of n i c k e l .
half of S a m p l e A ' s m a g n e s i u m and m a n g a n e s e content
a r e s o l u b l e , as is m o s t of the t r a c e c o p p e r and z i n c
content of the o r e .
~.O!
E f f e c t of P a r t i c l e S i z e 100%-270 Mesh
To d e t e r m i n e the effect of p r i o r g r i n d i n g on l e a c h -
ing, t h r e e s a m p l e s of S a m p l e A w e r e ground to 100
2.6
p c t - 20 m e s h (78 p c t - 4 0 0 m e s h ) , 100 p c t - 100 m e s h
(82 p c t - 4 0 0 m es h ) and 100 p c t - 2 7 0 m e s h (91 pct
- 4 0 0 m e s h ) , r e s p e c t i v e l y . T h e s u r f a c e a r e a s of t h e s e
ground s a m p l e s w e r e 60.5, 65.3 and 66.8 m 2 / g r e s p e c -
t i v e l y . E a c h of t h e s e f r a c t i o n s was then p r e s s u r e 2.2
l e a c h e d at 250~ 0.24 a c i d / o r e r a t i o (based on 96 pct Ni/(Fe +AI)
H2SO~) and 33 pct s o l i d s . N e i t h e r the r a t e nor the ex -
tent of n i c k e l e x t r a c t i o n was a f f e c t e d by the o r e grind;
h o w e v e r , the n i c k e l - t o - i m p u r i t y r a t i o s in the p r e g n a n t 1.8
l i q u o r i n c r e a s e d s u b s t a n t i a l l y when l e a c h i n g the
gound m a t e r i a l . See F i g . 2. A p p a r e n t l y the i n c r e a s e d e
s u r f a c e g e n e r a t e d by g r i n d i n g p r o v i d e s a d d i t i o n a l nu-
c l e a t i n g s i t e s f o r h y d r o l y s i s of the i r o n and a l u m i -
num, s o l u b i l i z e d d u r i n g the i n i t i a l s t a g e s of l e a c h i n g . 1.4
E f f e c t of A g i t a t i o n
1,5 3'0 4'5 6'0
A f t e r the acid had b e e n t h o r o u g h l y m i x e d with the
s l u r r y fo r 30 s, no a d d i t i o n a l a g i t a t i o n was r e q u i r e d . LEACHING TIME minutes
T h u s , t u r n i n g the a g i t a t o r off t h i r t y s e c o n d s a f t e r a c i d Fig. 2--Nickel-to-impurity ratios in Sample A leach liquor
i n j e c t i o n h i n d e r e d n e i t h e r the r a t e n o r the e x t e n t of as a function of leaching time and grind (250~ 0.24 acid/ore
n i c k e l e x t r a c t i o n . T h i c k e n i n g t e s t s a s shown in T a b l e and 33 pct solids).
METALLURGICAL TRANSACTIONS B VOLUME SB, DECEMBER 1977-549
I00 9 I i i I i i i
050 ~,7~oc ~0o/~c
90
2 ~ 225"C o.z'r INi/Fe: , 4 . 0 / J
Ni ACID/ ORE
80
EXTN, RATIO
%
70 / 55 % Solids
0.24 Acid/Ore
/ /// 2~o.c~
600 RPM
6C
.... I I I I I I
I 3 5 15 30 60
LEACHING TIME-minutes(l~ scale)
15 50 45 60 Fig. 5--Leaching time required to attain a given nickel-to-
LEACHING T I M E - minutes impurity ratio as a function of temperature and acid/ore ratio.
Fig. 3--Nickel extraction as a functien ofleaching time and
temperature.
T h e f r e e a c i d in the l e a c h l i q u o r d e c r e a s e d f r o m the
i n i t i a l c o n c e n t r a t i o n of o v e r 100 gpl to about 25 to 50
gpl a f t e r the f i r s t s e v e r a l m i n u t e s of l e a c h i n g , and
r e m a i n e d at this e q u i l i b r i u m c o n c e n t r a t i o n throughout
the r e s t of the l e a c h i n g p e r i o d . The e q u i l i b r i u m con-
60
c e n t r a t i o n was independent of t e m p e r a t u r e v a r i a t i o n
50
f r o m 250 to 275~ (Fig. 7). One might have e x p e c t e d
40
LOg k= 2.30~'RT the f r e e a c i d l e v e l of the h o t t e r l i q u o r s to be h i g h e r ,
30 * Constant b e c a u s e in t h e s e s o l u t i o n s m e t a l h y d r o l y s i s was m o s t
c o m p l e t e . H o w e v e r , at the h i g h e r t e m p e r a t u r e , m o r e
c a I/g- molq
20 s u l f a t e was tied up in the a l u m i n u m p r e c i p i t a t e , so
h y d r o l y s i s at 275~ d o e s not g e n e r a t e the quantity of
kxlOZ,min- t f r e e a c i d that would have b e e n f o r m e d at 250~
I0
8 Effect of A c i d / O r e R a t i o
6 \ One v a r i a b l e r e q u i r i n g c a r e f u l c o n t r o l is the weight
498 2.73 of a c i d a d d e d to a given weight of o r e (i.e., a c i d / o r e
4 5?_.3 17.0 r a t i o ) . A l r e a d y shown was the i n c r e a s e in r e s i d u a l
548 51.4 s u l f u r content with i n c r e a s e d a c i d addition. M o r e i m -
3
p o r t a n t is the effect of a c i d / o r e r a t i o on the t i m e r e -
q u i r e d to a t t a i n a given l e v e l of n i c k e l e x t r a c t i o n .
,,~ ,.~ 2'.o ( R e f e r to F i g . 8). 90 p c t n i c k e l e x t r a c t i o n is a t t a i n e d
I/T• OK-I in 5 rain at 250~ 25 pct s o l i d s and 0.30 a c i d / o r e ;
t h r e e t i m e s l o n g e r is r e q u i r e d if only 0.27 units of
Fig. 4--Rate of nickel extraction as a function of temperature.
a c i d a r e a d d e d . U n f o r t u n a t e l y , s h o r t l e a c h t i m e s at
high a c i d / o r e a l s o l e a v e s u b s t a n t i a l q u a n t i t i e s of i r o n
and a l u m i n u m in the p r e g n a n t liquor. The s a m e
It is not s u f f i c i e n t to c o n s i d e r only the n i c k e l and change f r o m 0.27 up to 0.30 a c i d ~ o r e r a t i o i n c r e a s e d
c o b a l t r e c o v e r y ; s e l e c t i v i t y i s a l s o an i m p o r t a n t f a c t o r the quantity of i r o n and a l u m i n u m in the p r e g n a n t
for s u b s e q u e n t m e t a l r e c o v e r y . F i g u r e 5 shows the l i q u o r by 100 pct and 33 pct, r e s p e c t i v e l y , at about 30
l e a c h i n g t i m e r e q u i r e d to a t t a i n N i / F e and Ni/A1 r a - min of l e a c h i n g . (See F i g . 9). The N i / F e r a t i o in-
tios of four in the p r e g n a n t l i q u o r at s e v e r a l a c i d / o r e c r e a s e s f a s t e r with t i m e than does the Ni/A1 r a t i o ,
r a t i o s . It i s c l e a r that i n c r e a s i n g the t e m p e r a t u r e r e f l e c t i n g the r e l a t i v e r a t e of h y d r o l y s i s of t h e s e i m -
f r o m 250 ~ up to 275~ s u b s t a n t i a l l y i m p r o v e s p r e g n a n t p u r i t i e s . T h e i m p u r i t y c u r v e s g e n e r a l l y have a p o s i -
l i q u o r q u a l i t y o v e r the r a n g e of l e a c h i n g t i m e and tive slope, a t r e n d which is m a i n t a i n e d a s t e m p e r a t u r e
acid/ore ratios investigated. i n c r e a s e s . T h i s s l o p e r e f l e c t s the i n c r e a s e d s o l u b i l i t y
L e a c h i n g t e m p e r a t u r e a l s o has a s h a r p effect on the of the i r o n and a l u m i n u m h y d r a t e s with the i n c r e a s e d
a m o u n t of s u l f a t e r e p o r t i n g in the r e s i d u e , (Sulfur a c i d i t y g e n e r a t e d by the h i g h e r a c i d to o r e r a t i o s .
is a m a j o r o p e r a t i n g e x p e n s e , and that p o r t i o n lost to
the r e s i d u e is the m o s t difficult to u t i l i z e o r r e -
Effect of P e r c e n t Solids
cycle) .14 See F i g . 6. The sulfate a s s a y of the r e s i d u e
r i s e s s h a r p l y d u r i n g the f i r s t s e v e r a l m i n u t e s of T h i s v a r i a b l e has been s a v e d until l a s t , b e c a u s e the
leaching, then f a l l s , and f i n a l l y e q u i l i b r a t e s at five to l e v e l of s o l i d s which gives the b e s t n i c k e l e x t r a c t i o n
s i x pct. Sulfur l o s s e s a r e g r e a t e s t at high t e m p e r a - d e c r e a s e s with i n c r e a s i n g t e m p e r a t u r e at a given
t u r e and high a c i d / o r e , and l e a s t at low l e v e l s of t e m - a c i d a d d i t i o n and l e a c h t i m e ( T a b l e s IV and V). L i m o -
p e r a t u r e and a c i d / o r e . A s a t i s f a c t o r y s u l f a t e b a l a n c e nite a c i d l e a c h i n g l i t e r a t u r e e m p h a s i z e s the d e s i r a -
was obtained between s o l i d and s o l u t i o n in t h e s e t e s t s . b i l i t y of t h i c k e n i n g the o r e feed to at l e a s t 33 pct
550-VOLUME 8B, DECEMBER 1977 METALLURGICAL TRANSACTIONS B
-- I I I I 1 I I i E I I
7.0 7
i
o,e q
AC
R
ID
oC
A
Io~
;2
E
.~
1
o
,]2
'.557~
5E
%
,N
~X
~iTN
,9l.0
,%,~.,
275~ 0.21 A / O i
5.0
SULFATE / j o~oA,o~-
IN
RESIDUE,
%
3.0
25% Solids I 3 5 15 30 60
LEACHING TIME- minutes(log scale)
Fig. 8--Nickel extraction as a function of acid/ore ratio and
I I 1
15 30 45 60 leaching time (250~ 25 pet solids).
LEACHING TIME - minutes
I ! I ! i i i i i I
9.0
1.0
o.~ /
ACID/ORE o.27 / / O'O~ff
RATIO
IN
SULFATE 7 . 0 ~
RESIDUE, : ~' 0.24
%
5.0
0.21
! ~ 250 ~ , 0.21 A / O
A~___ I _ i t __ /
3 5 15 3O 6o
LEACHING TIME- minutes (log scale)
3'~ t 33% Solids
F T- - f ..... ~ - - 9
I I L I 0.3C / ~ ~
o 15 30 45 60
1.2
LEACHING TIME - minutes
Fig. 6--Sulfate content of Sample A ' s leach residue as a
function of leaching time, temperature, acid/ore ratio RATIO .5 Ni/AI =2.O
and pct solids.
0.2~
50
I % I ~ I I 0.21
I 3 5 115
,9
30 60
Q 33 275 LEACHING TIME- minutes(log .~cale)
Fig, 9--Nickel-to-impurity ratio as a function of acid/ore
ratio and leaching time (250~ 25 pet solids).
4O
AVERAGE
FREE
ACID, s o l i d s and p r e f e r a b l y to 45 pct s o l i d s . 2 Such t h i c k e n -
g/I ing a p p e a r s j u s t i f i e d when o p e r a t i n g in the 240 ~ to
260~ t e m p e r a t u r e r a n g e u t i l i z e d by p a s t p r a c t i c e .
30 The d a t a shown in T a b l e IV and F i g . 10 i l l u s t r a t e this
point.
Although l e a c h i n g at high s o l i d s is p r e f e r r e d p r a c t i c e
at 250~ the s a m e is not t r u e in the 260 to 275~ t e m -
p e r a t u r e r a n g e . At t h e s e h i g h e r t e m p e r a t u r e s , l e a c h -
20 ing should be c a r r i e d out at low s o l i d s (20 to 30 pct)
I I I ]
0.21 0,24 0.27 0.30 f o r b e t t e r n i c k e l e x t r a c t i o n and s o l u t i o n q u a l i t y . T h e
ACID/ORE RATIO r e s u l t is m o r e n i c k e l , but l e s s i r o n and a l u m i n u m b e -
Fig. 7--Average free acid in leach liquor as a function of i n g c a r r i e d into the p r e g n a n t l i q u o r ( T a b l e V).
acid/ore ratio, temperature and pet solids. T h u s , the c o m b i n a t i o n of low s o l i d s and high t e r n -
METALLURGICAL TRANSACTIONS B VOLUME 8B, DECEMBER 1977-551
r i a l . F o r the nucleation of products of h y d r o l y s i s of
Table IV. Effect of Percent Solids on Metal Extraction When iron and aluminum, it a p p e a r s that the additional s u r -
Leaching Samples "A" at 250~ for 60 Min face provided by grinding the c o a r s e fraction is more
active than the s u r f a c e of the r e a d i l y d i s s o l v e d min-
Pregnant Liquor Pct Ni
erals.
Pct Solids Acid/Ore Ni/Fe Ni/AI Extraction
Nickel e x t r a c t i o n was insensitive to agitation a f t e r
25 0.21 16 6 91 the f i r s t 30 s of leaching, showing that during most
33 0,21 14 8 93 of the r e a c t i o n p e r i o d the diffusion of a r e a c t i n g
25 0.24 13 4 96
s p e c i e s through a liquid boundary l a y e r was not l i m i t -
33 0.24 14 6 95
ing. This study of agitation during acid p r e s s u r e leach-
ing of l a t e r t t e did v e r i f y one of S. I. Sobol's concepts:
to attain high leaching r a t e s and complete conversion,
r a p i d and p e r f e c t mixing of the o r e and acid is r e -
Table V. Effect of Percent Solids on Metal Ext~ction When quired. 5 However, Sobol a l s o s t a t e d that " i n t e n s i v e
Leaching Samples "'A" at 275~ for 60 Min agitation is r e q u i r e d during the time the sludge r e -
mains in the r e a c t o r s to attain complete c o n v e r s i o n " . 6
Pregnant Liquor Pct Ni This s t a t e m e n t is quite c o n t r a r y to the r e s u l t s ob-
Pct Solids Acid/Ore Ni/Fe Ni/AI Extraction tained in the p r e s e n t study. Our work shows that
25 0.21 23 12 94 nickel e x t r a c t i o n is independent of agitation after a
33 0,21 16 12 93 b r i e f initial mixing p e r i o d .
25 0.24 18 8 97 When sulfuric acid is mixed with limonite pulp, the
33 0.24 9 5 95 aluminum and iron in the feed (except that portion tied
with chromite), a r e r e a d i l y converted to soluble sul-
fates. As this conversion p r o g r e s s e s , the acidity of
I00
the liquor d e c r e a s e s from a few hundred to about 25
I I I I
to 50 gram p e r l i t e r . This drop in acidity at the p r e -
vailing high r e a c t i o n t e m p e r a t u r e r e s u l t s in the hy-
d r o l y s i s of i m p u r i t i e s as d i s c u s s e d e a r l i e r in the
C h e m i s t r y of Leaching Section.
Studies of the FezO3-SO3-H~O s y s t e m p e r f o r m e d by
/
90
Posnjak and Haigh 15'16 have shown that the only stable
iron phases above 200~ a r e Fe20~, Fe203" 2SO3" H20
and Fe203 93SO3. S i m i l a r studies p e r f o r m e d by Davey
on the A1203-SO3-H20 s y s t e m d e m o n s t r a t e d that the
h y d r o l y s i s of aluminum sulfate solutions above 220~
p r o d u c e s hydronium alunite (3A1203 94SO3 ' 9H20) over
EXTN., a wide range of o p e r a t i n g conditions.9 The SO4=/A1
%
weight r a t i o in alunite is 2.4 to 1. The SO4:/A1 weight
r a t i o found in the final leach r e s i d u e was 2.3 to 1.
70
T/J E] - ~0
Thus, the predominant forms of iron and aluminum in
/~ O - 20
the final leach r e s i d u e were probably hematite and
alunite, r e s p e c t i v e l y (i.e., a s s i g n all of the sulfur to
/ & - 25
the aluminum and none to the iron). The IR s p e c t r u m
O 33 shown in Fig. 1 and the XRD data s u m m a r i z e d in
60 9 45 Table H support this conclusion.
The t e m p e r a t u r e dependence of the nickel e x t r a c t i o n
I" t ~ 70 I r a t e between 250 and 275~ is pronounced during the
0 30 60 120
f i r s t ten min of leaching, but becomes a l m o s t t e m p e r a -
LEACHING TIME - minutes ture independent after 15 min (Fig. 3). Also, the 225 ~
Fig. 1 0 - - E f f e c t of pulp d e n s i t y on nickel e x t r a c t i o n b a s e d on and 300~ r e a c t i o n t e m p e r a t u r e s r e s u l t in significantly
r e s i d u e a n a l y s e s as a function of l e a c h i n g t i m e . S a m p l e A lower nickel e x t r a c t i o n than does the 250 to 275~
l a t e r i t e leached at 250~ and 0.24 a c i d / o r e .
i n t e r m e d i a t e range. Low t e m p e r a t u r e apparently does
not provide the d r i v i n g force needed for rapid nickel
extraction, and v e r y high t e m p e r a t u r e apparently r e -
p e r a t u r e r e q u i r e s 10 pct l e s s acid to attain a com- sults in c o p r e c i p i t a t i o n of nickel with the solid hy-
p a r a b l e level of nickel extraction than does the low d r o l y s i s products. 7
temperature-high solids combination. The sulfur content or the leach r e s i d u e i n c r e a s e s
with i n c r e a s i n g t e m p e r a t u r e , probably because the
DISCUSSION stable f o r m of the aluminum h y d r o l y s i s product
changesfl At 250~ and 20 to 50 gpl H2SO4 c o n c e n t r a -
The r a t e of nickel e x t r a c t i o n is not affected by p r i o r tion, alunite (3A1203 "4SO3" 9H20) is the most stable
grinding of the ore. This i n s e n s i t i v i t y is probably due aluminum h y d r o l y s i s product. At 285~ the compound
to the insignificant amount of soluble nickel in the A1203 92SO3 "H20 becomes the stable phase.l~ The
c o a r s e fraction ( r e f e r back to Table I). However, the higher t e m p e r a t u r e aluminum product contains 50 pct
n i c k e l - t o - i m p u r i t y r a t i o in the pregnant liquor in- m o r e sulfur, and thus accounts for a portion of the in-
c r e a s e d substantially when leaching the ground m a t e - c r e a s e d sulfur content of the leach r e s i d u e s generated
552-VOLUME 8B, DECEMBER 1977 METALLURGICAL TRANSACTIONS B
at the higher leach t e m p e r a t u r e s . T h e s u l f u r content
of the l e a c h r e s i d u e also i n c r e a s e s with i n c r e a s i n g 95
a c i d / o r e r a t i o due to the i n c r e a s e d s t a b i l i t y of Fe2Oa 4.0
9SOn 9H20 and A1203 92SOs 9H20 in high acid media.iX 85
T h e a c t i v a t i o n e n e r g y , c a l c u l a t e d b e t w e e n 225 ~ and
275~ was a p p r o x i m a t e l y 30 K c a l / g mole 9 Such a high 3.6
a c t i v a t i o n e n e r g y is a s s o c i a t e d e i t h e r with diffusion 75 NICKEL
of a r e a c t a n t through a solid (such as h y d r o l y s i s p r o - Ni/(Fe*AI) a.2 EXTRACTION
ducts coated on a l i m o n i t e p a r t i c l e ) o r with a c h e m i -
PERCENT
cal r e a c t i o n . Agitation data i n d i c a t e that the l e a c h i n g
r e a c t i o n is not a diffusion c o n t r o l l e d r e a c t i o n of a 28
liquid b o u n d a r y layer 9
T h e c h a r a c t e r i s t i c shape of the c u r v e s d r a w n in
2.4
Fig. 6 can be c o n s t r u c t e d by a s s u m i n g that f i r s t a hy-
d r o l y s i s p r o d u c t high i n s u l f u r , such a s b a s i c a l u m i -
n u m sulfate o r b a s i c i r o n s u l f a t e o r a m i x t u r e of both, ,b 2b 3b 4b 50
is f o r m e d . T h e s e compounds a r e s e n s i t i v e to i n i t i a l
a c i d / o r e r a t i o and t e m p e r a t u r e . They b e c o m e u n s t a - PERCENT SOLIDS
b l e as l e a c h i n g conditions a p p r o a c h e q u i l i b r i u m ( i . e . , Fig. 12--Nickel-to-impurities ratio in laterlte leach liquor
a f t e r the i n i t i a l charge is c o n s u m e d ) and t r a n s f o r m and nickel extraction as a function of pet solids. Sample A ,
leached for 1 h at 250~ and 0.24 acid/ore ratio.
to p r o d u c t s c o n t a i n i n g l e s s s u l f u r s u c h a s a l u n i t e and
h e m a t i t e . F i g u r e 11 i l l u s t r a t e s how two h y d r o l y s i s
c u r v e s additively can r e p r o d u c e the r a t h e r s t r a n g e at v e r y low s o l i d s r e s u l t s in low n i c k e l s o l u b i l i z a t i o n
looking c u r v e s of Fig. 6. X - r a y d i f f r a c t i o n a n a l y s i s due to the d i s p r o p o r t i o n a t e a m o u n t of acid lost to the
of leach r e s i d u e s (Table II) c o n f i r m s that this i s what end l i q u o r . (See F i g s . 6 and 10).
is t a k i n g p l a c e within the a u t o c l a v e a s l e a c h i n g t i m e N i c k e l - t o - i m p u r i t y r a t i o s in l a t e r i t e l e a c h l i q u o r
i n c r e a s e s . Goethite is not fully c o n v e r t e d to h e m a t i t e a s a f u n c t i o n of p e r c e n t s o l i d s a r e p l o t t e d i n F i g . 12,
u n t i l about 10 rain have e l a p s e d (250~ 33 pet s o l i d s , which show that the h i g h e s t n i c k e l e x t r a c t i o n o c -
0.24 a c i d / o r e ) and h e m a t i t e and a l u n i t e a r e the m a j o r c u r r e d at about 25 pct s o l i d s in one h o u r , l e a c h e d at
c r y s t a l l i n e products in the f i n a l leach r e s i d u e s .
250 C and 0.24 a c i d / o r e r a t i o . F u r t h e r m o r e , data
The effect of s o l i d s content on l i m o n i t e l e a c h i n g
g e n e r a t e d at 275~ show that the l e v e l of s o l i d s which
can be c o n s i d e r e d an e x t e n s i o n of the a c i d c o n c e n t r a -
gives the b e s t n i c k e l e x t r a c t i o n d e c r e a s e s with i n c r e a s -
tion effect. High pet s o l i d s g e n e r a t e high a c i d c o n c e n -
ing t e m p e r a t u r e 9 G e n e r a l l y s p e a k i n g , i n c r e a s i n g
t r a t i o n s . It has a l r e a d y been shown that an i n c r e a s i n g
t e m p e r a t u r e i n c r e a s e s the i n i t i a l r a t e of n i c k e l ex-
s o l u t i o n acidity i n c r e a s e s the q u a n t i t y of a l u m i n u m
traction; i n c r e a s i n g a c i d / o r e ratio i n c r e a s e s total
and i r o n in the p r e g n a n t liquor (Fig. 9). T h u s , the
n i c k e l e x t r a c t i o n and n i c k e l e x t r a c t i o n r a t e , and de-
lower the pct solids, the l o w e r the i m p u r i t y l e v e l in
c r e a s i n g the p e r c e n t s o l i d s i n c r e a s e s l e a c h i n g s e l e c -
the p r o d u c t s o l u t i o n . What r e a l l y c o u n t s , h o w e v e r , is t i v i t y and a c i d u t i l i z a t i o n ( i . e . , l e s s s u l f u r i s l o s t to
the n i c k e L / i m p u r i t y c o n c e n t r a t i o n r a t i o in this s o l u - the l e a c h r e s i d u e . )
tion. F o r a given a c i d / o r e r a t i o , t h e r e is a pct s o l i d s C o n c e r n i n g o v e r a l l i m p r o v e m e n t of the l e a c h r e -
that m a x i m i z e s the n i c k e l / i m p u r i t y r a t i o , O p e r a t i n g
s p o n s e , l a t e r i t e l e a c h i n g is b e s t conducted at t e m -
p e r a t u r e s h i g h e r than 250~ and at s o l i d s l e s s than 30
pct. The a c i d / o r e l e v e l should be that r e q u i r e d to a t -
I -- I I F ~ t a i n r e a s o n a b l e n i c k e l e x t r a c t i o n at a n a c c e p t a b l e
zo rate.
r 2 5 0 * C . 0 . 3 0 Acid l o r e , 5 3 % Solid~
ACKNOWLEDGMENTS
5.0 I The a u t h o r s thank C. S. S i m o n s (AMAX N i c k e l D i v i -
f sion) for h i s c o n t i n u e d s u p p o r t and e n c o u r a g e m e n t , and
SULFATE
IN II ,~--Stable
/ Hydrolysis p a r t i c u l a r l y for u n s e l f i s h l y s h a r i n g the knowledge and
RESIDUE, ~ PfOauctts) e x p e r i e n c e he gained d u r i n g d e v e l o p m e n t of the Moa
% 3.0 /~ Bay o p e r a t i o n 9 S p e c i a l thanks a r e extended to P r o f .
H. J. R o o r d a of Delft U n i v e r s i t y f o r his d i r e c t i o n d u r -
ing p r e p a r a t i o n of this m a n u s c r i p t 9
I .
I \ <- Tronsient Sydroly~t~
V Product(s)
\ REFERENCES
/ \,
15 30 45 60 1. H. J. Roorda and P. E. Queneau:Institution of Mining and Metallurgy, Sec.C,
pp. 79-87,June 1973.
LEACHING TIME - minutes 2. C. S. Simons:J. of Metals, 1971,pp. 48-58.
Fig. ll--Hypothetical construction of observed residual sul- 3. J. H. Canterford:Rev. Pure AppL Chem., 1972,vol. 22, pp. 17-51.
fate curve by addition of a transient hydrolysis curve to a 4. E. T. Carlsonand C. S. Simons:Extractive Metallurgy of Copper, Nickel and
jarosite-alunite formation curve. Cobalt, IntersciencePublishers,New York, N.Y., !96 I-
METALLURGICALTRANSACTIONSB VOLUME 8B, DECEMBER 1977-553
5. S. I. Sobol: Rev. Technol., 1966, vol. 4, no. 5, pp. 3-19. 11. C. Nikolic: Thesis, Department of Metallurgy, The University of British
6. S. 1. Sobol: Rev. Technol., 1968, vol. 6, nos. 5-6, pp. 3-24. Columbia, February 1971.
7. S. I. Sobol: Rev. Technol., 1969, vol. 7, no. 1, pp. 3-23. 12. P. Bayliss: Can. J. Chem., 1971, vol. 49, pp. 2433-41.
8. R. A. Chaves, V. V. Karelin, and B. P. Sobolex: Tsvet. Metal., 1968, vol. 9, no. 13. S. H. C. Briggs: J. Chem. Soc. Proc., 1929, pp. 242-46.
4, pp. 66-70. 14. J. G. Morrow: U.S. Patent 2,872,306, February 3, 1959.
9. P. T. Davey and T. R. Scott: Aust. J. Appl. Chem., 1962, vol. 13, pp. 229-41. 15. E. Posnjak and H. E. Merwin: 3". Amer. Chem. Soc., 1922, vol. 44, pp.
10. V. I. Goryachkin and S. I. Sobol: Sb. Nauch. Tr. Gos. Nauch.-lssled. Inst. 1965-94.
Tsvet. Metal, 1965, vol. 23, pp. 252-62. 16. C. J. Haigh: Proc. Australas. Inst. Mining Met., 1967, vol. 9, pp. 49-56.
5 5 4 - V O L U M E 8B, DECEMBER 1977 METALLURGICAL TRANSACTIONS B
View publication stats
You might also like
- Palygorskite-Sepiolite: Occurrences, Genesis and UsesFrom EverandPalygorskite-Sepiolite: Occurrences, Genesis and UsesRating: 1 out of 5 stars1/5 (1)
- Activities and Phase Boundaries in The Cr-Ni System Using A Solid Electrolyte TechniqueDocument6 pagesActivities and Phase Boundaries in The Cr-Ni System Using A Solid Electrolyte Techniquehoushi_miroku_151610No ratings yet
- Esca and Ftir Studies of Bituminous CoalDocument6 pagesEsca and Ftir Studies of Bituminous Coalcraigorio616No ratings yet
- Wang 1974Document5 pagesWang 1974G.L.R StudiosNo ratings yet
- 23 - 4 - Miami Beach - 09-78 - 0072Document9 pages23 - 4 - Miami Beach - 09-78 - 0072adrian2009-2020No ratings yet
- Design of Pillars in Shrinkage StopingDocument13 pagesDesign of Pillars in Shrinkage StopingManuel QuirozNo ratings yet
- Vol24 2 PDFDocument378 pagesVol24 2 PDFAnonymous 3XvNuVNo ratings yet
- 8.5 Treatment of The Leach Liquor Prior To SX-EW: Eco-Tec - Co MDocument1 page8.5 Treatment of The Leach Liquor Prior To SX-EW: Eco-Tec - Co MLeo Santiago GallegosNo ratings yet
- A Study of The Kinetics and Mechanism of The Drying Process in Ceramic MaterialsDocument3 pagesA Study of The Kinetics and Mechanism of The Drying Process in Ceramic MaterialsRafaelNo ratings yet
- Basic Solar Pond ModelDocument7 pagesBasic Solar Pond ModelNeale NeelamegghamNo ratings yet
- Physical Properties of Uranium Process Solutions byDocument15 pagesPhysical Properties of Uranium Process Solutions byShrishma PaikNo ratings yet
- (TID, 4500._ UC (Series)., 25, Metals, Ceramics, and Materials._ AEC research and development report) Oak Ridge National Laboratory. Metallurgy Division._ Union Carbide Corporation - Metallurgy Divisi.pdfDocument502 pages(TID, 4500._ UC (Series)., 25, Metals, Ceramics, and Materials._ AEC research and development report) Oak Ridge National Laboratory. Metallurgy Division._ Union Carbide Corporation - Metallurgy Divisi.pdfCarlosNo ratings yet
- Johnv. Elackay, Gleiss.: Apo2ChDocument7 pagesJohnv. Elackay, Gleiss.: Apo2ChagyaaNo ratings yet
- DHBuckley and RLJohnson - Co Friction and Wear Under VaccumDocument20 pagesDHBuckley and RLJohnson - Co Friction and Wear Under VaccumJiahui QiNo ratings yet
- Design Considerations For Merrill-Crowe PlantsDocument5 pagesDesign Considerations For Merrill-Crowe PlantsLuis Miguel La Torre100% (1)
- The Densities and Structures of Melts in the CaO-FeO-SiO2 SystemDocument8 pagesThe Densities and Structures of Melts in the CaO-FeO-SiO2 Systemjagadish mahataNo ratings yet
- (Received April 1988 Revision 26 Sept. 1988) : Minerals Engineering, Vol. 2, No. 2, Pp. 217-227, 1989Document11 pages(Received April 1988 Revision 26 Sept. 1988) : Minerals Engineering, Vol. 2, No. 2, Pp. 217-227, 1989alinoriNo ratings yet
- Fatigue Crack Initiation and Microcrack Growth in 4140 SteelDocument5 pagesFatigue Crack Initiation and Microcrack Growth in 4140 SteelEdda Andrade RosalesNo ratings yet
- 10 - 3 - New York - 09-66 - 0058Document14 pages10 - 3 - New York - 09-66 - 0058Juan Bayron MuñozNo ratings yet
- Development and Verification of NASA Standard Initiator-2 (NSI-2)Document51 pagesDevelopment and Verification of NASA Standard Initiator-2 (NSI-2)구자춘No ratings yet
- Metal Notes PlumbingDocument8 pagesMetal Notes PlumbingPatrick MachariaNo ratings yet
- Desulfurization of Liquid Steel Containing Aluminum or Silicon With Lime PDFDocument6 pagesDesulfurization of Liquid Steel Containing Aluminum or Silicon With Lime PDFRasul BzNo ratings yet
- Determination of Carbohydrates by Anion Exchange Chromatography With Pulsed Amperometric DetectionDocument16 pagesDetermination of Carbohydrates by Anion Exchange Chromatography With Pulsed Amperometric DetectionXYZUSPNo ratings yet
- Structural Properties of the Pb02 Active Mass Determining-복사Document8 pagesStructural Properties of the Pb02 Active Mass Determining-복사에푸디ᅳᅳNo ratings yet
- Copper Electrowinning From Cyanide Solutions by Lu Jianming PDFDocument306 pagesCopper Electrowinning From Cyanide Solutions by Lu Jianming PDFFerudun AkyolNo ratings yet
- UT Rotary Tube TestingDocument4 pagesUT Rotary Tube Testingach soNo ratings yet
- Victoria Brown Coal PropertiesDocument401 pagesVictoria Brown Coal PropertiesShahabuddin SuzanNo ratings yet
- Prasad1992 PDFDocument3 pagesPrasad1992 PDFummu atiqah maisaraNo ratings yet
- Chemistry, The University, Leyden (NetDocument4 pagesChemistry, The University, Leyden (NetCristhian ParedesNo ratings yet
- 15 - 2 - Washington DC - 09-71 - 0150Document8 pages15 - 2 - Washington DC - 09-71 - 0150Matias MancillaNo ratings yet
- Nicolle and RistDocument10 pagesNicolle and RistMariaNo ratings yet
- Rockfill Deformation From Large-Scale Tests: Essais A Grande Echelle Pour Les Barrages RocheuxDocument5 pagesRockfill Deformation From Large-Scale Tests: Essais A Grande Echelle Pour Les Barrages RocheuxChandra PutraNo ratings yet
- Evaluation of Toughness in AISI 4340 Alloy Steel Austenitized at Low and High TemperaturesDocument2 pagesEvaluation of Toughness in AISI 4340 Alloy Steel Austenitized at Low and High TemperaturesSyahid AzhariNo ratings yet
- Recuperción de Níquel ECOTECDocument11 pagesRecuperción de Níquel ECOTECJesús RiberaNo ratings yet
- Mechanical Properties of Modified Low Cobalt Powder Metallurgy UDIMET 700 Type AlloysDocument30 pagesMechanical Properties of Modified Low Cobalt Powder Metallurgy UDIMET 700 Type AlloysEnary SalernoNo ratings yet
- International Journal of Mine Water - © International Mine Water Association 2006 - WWW - IMWA.infoDocument13 pagesInternational Journal of Mine Water - © International Mine Water Association 2006 - WWW - IMWA.infoGIANCARLONo ratings yet
- Monografia Captopril ConnorsDocument3 pagesMonografia Captopril ConnorsAngelica MedinaNo ratings yet
- Of An: Attention inDocument17 pagesOf An: Attention inRhecelNo ratings yet
- High Damping in Grey Cast IronDocument5 pagesHigh Damping in Grey Cast IronOutvaluing Vanmen GreensNo ratings yet
- Removing H2S from Coal Gases Using Zinc Oxide-Zeolite SorbentsDocument8 pagesRemoving H2S from Coal Gases Using Zinc Oxide-Zeolite Sorbentsngnm0No ratings yet
- Intermtionaz Tournazof Pcne:,Later, 3Document12 pagesIntermtionaz Tournazof Pcne:,Later, 3RiswanNo ratings yet
- 08TARTAR WINESTONE ANTIMONY - Unknown PDFDocument8 pages08TARTAR WINESTONE ANTIMONY - Unknown PDFJohnny jonesNo ratings yet
- 16 4 Boston 04-72 0070 PDFDocument9 pages16 4 Boston 04-72 0070 PDFGurusangmeshHiremathNo ratings yet
- Estimating Methane Content of Bituminous Coalbeds From AdsorptionDocument26 pagesEstimating Methane Content of Bituminous Coalbeds From AdsorptionAdhitya KuswantoroNo ratings yet
- Amorphous GaDocument4 pagesAmorphous GaGolompoNo ratings yet
- Ali 1983Document32 pagesAli 1983Andres Rengifo BotinaNo ratings yet
- Butadiene PproductionDocument5 pagesButadiene PproductionYoteshYadavNo ratings yet
- Blast Furnace Panel Discussion: Experience with Stave CoolersDocument14 pagesBlast Furnace Panel Discussion: Experience with Stave CoolersGabriel MáximoNo ratings yet
- The Effect of Carbide and Nitride Additions On The Heterogeneous Nucleation Behavior of Liquid IronDocument9 pagesThe Effect of Carbide and Nitride Additions On The Heterogeneous Nucleation Behavior of Liquid IronJoe Iurk PereiraNo ratings yet
- Effect of Shot-Peening On Surface Crack Propagation in Plane-Bending FatigueDocument8 pagesEffect of Shot-Peening On Surface Crack Propagation in Plane-Bending FatigueaapennsylvaniaNo ratings yet
- Jennite Molecuar ModelDocument11 pagesJennite Molecuar ModelProdip SarkarNo ratings yet
- 32 - 3 - New Orleans - 08-87 - 0255Document5 pages32 - 3 - New Orleans - 08-87 - 0255Abdul AzizNo ratings yet
- Kinetics of Phase Layer Growth During Aluminide Coating of NickelDocument10 pagesKinetics of Phase Layer Growth During Aluminide Coating of NickelAbhishek ThakurNo ratings yet
- Lin 1971Document20 pagesLin 1971Xshf AkNo ratings yet
- Methane Oxidative Coupling for Ethylene ProductionDocument15 pagesMethane Oxidative Coupling for Ethylene ProductionGabriel F RuedaNo ratings yet
- Nickel Leaching Using Pressured Sulphuric AcidDocument8 pagesNickel Leaching Using Pressured Sulphuric AcidAG RahmanNo ratings yet
- 1975 Precipitation Hardening of Aluminum AlloysDocument6 pages1975 Precipitation Hardening of Aluminum AlloysyhgrinmNo ratings yet
- Mele (1982) - Characterisation of Runoff Water From Coalwaste Disposal Sites in South Wales IllionisDocument14 pagesMele (1982) - Characterisation of Runoff Water From Coalwaste Disposal Sites in South Wales IllionisRiswanNo ratings yet
- Metal Extraction (Recovery Systems) PDFDocument13 pagesMetal Extraction (Recovery Systems) PDFMary JohnsonNo ratings yet
- Kuliah 3a Thermodinamika 2020Document67 pagesKuliah 3a Thermodinamika 2020andyNo ratings yet
- MORE THAN JUST ANOTHER NICKEL MINERDocument28 pagesMORE THAN JUST ANOTHER NICKEL MINERandyNo ratings yet
- 06-TROUBLE SHOOTING Wear PartsDocument50 pages06-TROUBLE SHOOTING Wear PartsandyNo ratings yet
- High-Temperature Oxidation of Fe Si Alloys in The Temperature Range 900-1250°CDocument7 pagesHigh-Temperature Oxidation of Fe Si Alloys in The Temperature Range 900-1250°CandyNo ratings yet
- The Application of Potential - PH Diagrams To Solvent Extraction SystemsDocument14 pagesThe Application of Potential - PH Diagrams To Solvent Extraction SystemsandyNo ratings yet
- TEPZZ 86 95 - A - T: European Patent ApplicationDocument19 pagesTEPZZ 86 95 - A - T: European Patent ApplicationandyNo ratings yet
- Mass Transfer: Lecture 08: Leaching and ExtractionDocument15 pagesMass Transfer: Lecture 08: Leaching and ExtractionThe New WorldNo ratings yet
- Full Text 01Document86 pagesFull Text 01andyNo ratings yet
- 10 30797-Madencilik 580147-739472Document14 pages10 30797-Madencilik 580147-739472andyNo ratings yet
- Rupa Sing He 2016Document2 pagesRupa Sing He 2016andyNo ratings yet
- Atmospheric Acid Leaching of Nickel Laterites Review Part I. Sulphuric Acid TechnologiesDocument21 pagesAtmospheric Acid Leaching of Nickel Laterites Review Part I. Sulphuric Acid TechnologiesandyNo ratings yet
- 0003 - Crusher 21 & 31 Process DiagramDocument1 page0003 - Crusher 21 & 31 Process DiagramandyNo ratings yet
- Autoclaves Cale PQDocument9 pagesAutoclaves Cale PQandyNo ratings yet
- PDFDocument28 pagesPDFGaneshbhoiteNo ratings yet
- Midrexshaftprocess 141215100600 Conversion Gate02 PDFDocument10 pagesMidrexshaftprocess 141215100600 Conversion Gate02 PDFandyNo ratings yet
- TesDocument67 pagesTesandyNo ratings yet
- Madencilikdergimakalesi2019 1Document15 pagesMadencilikdergimakalesi2019 1andyNo ratings yet
- Intro Fracture MechanicsDocument51 pagesIntro Fracture MechanicsTsabit AymanNo ratings yet
- 00400011a3 Papers 00400011A3 00400011A30172 PDFDocument13 pages00400011a3 Papers 00400011A3 00400011A30172 PDFandyNo ratings yet
- Komnum Minggu 3Document6 pagesKomnum Minggu 3andyNo ratings yet
- Andy Kurnia - 1606838956 - Tugas Prosman 3Document2 pagesAndy Kurnia - 1606838956 - Tugas Prosman 3andyNo ratings yet
- 1 s2.0 0950061895000178 MainDocument12 pages1 s2.0 0950061895000178 MainandyNo ratings yet
- Multiple Hearth FurnaceDocument6 pagesMultiple Hearth FurnaceandyNo ratings yet
- CamScanner Scans PDFs QuicklyDocument4 pagesCamScanner Scans PDFs QuicklyandyNo ratings yet
- Dokumen - Tips Inspeksi KorosiDocument16 pagesDokumen - Tips Inspeksi KorosiandyNo ratings yet
- Jenny Randles - Mind Monsters - Invaders From Inner Space (1990)Document219 pagesJenny Randles - Mind Monsters - Invaders From Inner Space (1990)DirkTheDaring11100% (15)
- Challan FormDocument2 pagesChallan FormSingh KaramvirNo ratings yet
- In2it: A System For Measurement of B-Haemoglobin A1c Manufactured by BIO-RADDocument63 pagesIn2it: A System For Measurement of B-Haemoglobin A1c Manufactured by BIO-RADiq_dianaNo ratings yet
- Hydro Distillation Method Extraction of Eucalyptus Oil Lemongrass OilDocument9 pagesHydro Distillation Method Extraction of Eucalyptus Oil Lemongrass OilSIVANESAN JOTHIVELNo ratings yet
- Fuel Equivalent Factor - Marine Services PDFDocument9 pagesFuel Equivalent Factor - Marine Services PDFMax Kolonko100% (1)
- Sophiajurgens Resume EdtDocument2 pagesSophiajurgens Resume Edtapi-506489381No ratings yet
- Module 4.Document16 pagesModule 4.Sania MaqsoodNo ratings yet
- Blood Smear PreparationDocument125 pagesBlood Smear PreparationKim RuizNo ratings yet
- Chapter-10 - Biomechanics in Sports Class 12Document29 pagesChapter-10 - Biomechanics in Sports Class 12Matrix craft4100% (1)
- Risk Assessment: Overall RatingDocument3 pagesRisk Assessment: Overall RatingandirizaNo ratings yet
- BOYSEN® Marmorino™ Italian Marble Finish: Liters UseDocument1 pageBOYSEN® Marmorino™ Italian Marble Finish: Liters UseJohn Ray Esmama CalasicasNo ratings yet
- Lun Mapping DisksDocument11 pagesLun Mapping DisksKarn GusainNo ratings yet
- Call Log ReportDocument44 pagesCall Log ReportHun JhayNo ratings yet
- PC200-8 SM - 013 Troubleshooting by Failure Code Part-3Document50 pagesPC200-8 SM - 013 Troubleshooting by Failure Code Part-3t5442071100% (2)
- Performance Evaluation and Life Testing of The SPT-100Document12 pagesPerformance Evaluation and Life Testing of The SPT-100ThaSupaflyNo ratings yet
- Filipinism 3Document3 pagesFilipinism 3Shahani Cel MananayNo ratings yet
- Very Basic GSADocument46 pagesVery Basic GSATim ChongNo ratings yet
- Phrase Structure AnalysisDocument2 pagesPhrase Structure Analysismerlino99No ratings yet
- Mobilization and Participation: Social-Psychological Expansisons of Resource Mobilization TheoryDocument19 pagesMobilization and Participation: Social-Psychological Expansisons of Resource Mobilization TheoryJoaquim OliveiraNo ratings yet
- Welder Training in SMAW, GTAW & GMAW Welding Engineering & NDT Consultancy Welding Engineering Related TrainingDocument4 pagesWelder Training in SMAW, GTAW & GMAW Welding Engineering & NDT Consultancy Welding Engineering Related TrainingKavin PrakashNo ratings yet
- Volkswagen 2.0L TDI Common Rail Engine Service TrainingDocument90 pagesVolkswagen 2.0L TDI Common Rail Engine Service TrainingАлла Харютина100% (1)
- TMF1014 System Analysis & Design Semester 1, 2020/2021 APPENDIX C: Group Assignment Assessment RubricDocument4 pagesTMF1014 System Analysis & Design Semester 1, 2020/2021 APPENDIX C: Group Assignment Assessment RubricWe XaNo ratings yet
- Advanced Long Range Proximity Reader PDFDocument1 pageAdvanced Long Range Proximity Reader PDFPhangkie RecolizadoNo ratings yet
- Introduction To Globalization: Module DescriptionDocument3 pagesIntroduction To Globalization: Module DescriptionyowNo ratings yet
- Cosmos Carl SaganDocument18 pagesCosmos Carl SaganRabia AbdullahNo ratings yet
- Function Apollo Amadeus: Sign In/OutDocument16 pagesFunction Apollo Amadeus: Sign In/OutMabs GaddNo ratings yet
- 3 6 17weekly Homework Sheet Week 23 - 5th Grade - CcssDocument3 pages3 6 17weekly Homework Sheet Week 23 - 5th Grade - Ccssapi-328344919No ratings yet
- UCT APM M2 U1 - TP Leadership QuestionnaireDocument4 pagesUCT APM M2 U1 - TP Leadership QuestionnaireLincolyn MoyoNo ratings yet
- IPPD Presentation Roll OutDocument34 pagesIPPD Presentation Roll OutGracel Alingod Galay100% (1)
- Lanco Solar EPC leaderDocument19 pagesLanco Solar EPC leaderShabir TrambooNo ratings yet