Professional Documents
Culture Documents
Structural Characteristics de Ning High Barrier
Uploaded by
Christhy Vanessa Ruiz MadroñeroOriginal Title
Copyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
Structural Characteristics de Ning High Barrier
Uploaded by
Christhy Vanessa Ruiz MadroñeroCopyright:
Available Formats
Overview
Structural characteristics de ning high barrier
properties in polymeric materials
J. M. Lagaron, R. Catala and R. Gavara
The packaging industry has dramatically increased the number of packaging systems and designs made of plastics
over recent decades. Plastics, in contrast with more traditional packaging materials such as glass and metals, are
permeable systems that permit the exchange of low molecular weight compounds, e.g. gases and vapours, between
the inner and the outer atmosphere. Despite this drawback, the availability of shapes and forms in which they can be
manufactured, their ease of processing and handling, low price, excellent chemical resistance, etc., have made them
very attractive in many packaging elds. Consequently, much industrial and academic research has been devoted to
understanding the mechanisms of mass transport in polymers, to enable design of materials with improved barrier
properties. The present paper reviews some of these developments, and highlights the structural factors that cause
polymers to behave as high barrier materials, taking as benchmark the properties of one of the most widely used
family of high barrier materials, the ethylene – vinyl alcohol copolymers. MST/5992
Keywords: Polymers, High barrier properties
The authors are at the Packaging Laboratory, Institute of Agrochemistry and Food Technology, CSIC, Apdo. Correos 73, 46100
Burjassot, Spain (lagaron@iata.csic.es). Manuscript received 25 September 2003; accepted 8 October 2003.
# 2004 IoM Communications Ltd. Published by Maney for the Institute of Materials, Minerals and Mining.
use of plastic materials.1 – 3 In this respect, it has become a
Nomenclature topic of industrial and social awareness because of links
with current issues such as food commercialisation, food
aPA amorphous polyamide shelf life extension, quality and safety. Furthermore, it has
EPDM ethylene propylene diene monomer also become very relevant to a number of other applications
EVOH ethylene – vinyl alcohol copolymers including gas separation membranes, packaging of health
HDPE high density polyethylene care and pharmaceutical goods and chemicals and housing
LCP liquid crystal polymer of fuels (including oxygenated fuels) in tanks and lines in the
LDPE low density polyethylene automotive eld.
LLDPE linear low density polyethylene The reasons for the more recent interest in the develop-
PA polyamide ment of high barrier polymers and polymer based structures
PAN polyacrylonitrile are associated with a widespread trend to implement poly-
PA6 polyamide 6 (nylon) meric materials in an ever increasing number of applica-
PC polycarbonate tions. It is common knowledge that the attractiveness of
PE polyethylene plastics lies in their versatility and ability to offer a broad
PET polyethylene terephthalate variety of properties, and yet be cheap and readily processed
PHA polyhydroxyalkanoates and conformed into a myriad of shapes and sizes. However,
PK aliphatic polyketone copolymers polymers have a number of limitations for certain applica-
PLA polylactic acid tions when compared with more traditional materials such
PMMA polymethyl methacrylate as metals and alloys or ceramics. Among some of these
PP polypropylene limitations relevant to the subject of the present paper are
PS polystyrene their permeability and comparatively low thermal resis-
PVC polyvinyl chloride tance, and the strong interdependence between these two
PVDC polyvinylidene chloride properties. The permeability of plastics to the exchange of
PVOH polyvinyl alcohol gases and vapours imposes a number of challenges in those
applications where a high barrier, ideally impermeability,
is required. These applications were traditionally assumed
by, for instance, tinplate and glass in the food packaging
eld. However, polymer scientists, engineers and technol-
Introduction ogists in industry and academia have devoted a great deal of
effort and resources to pushing the limits of plastics
HIGH BARRIER CONCEPT performance towards impermeability, chie y because of
the overwhelming pressure exerted by the numerous other
A ‘high barrier’ is without doubt a very desirable property
advantages associated with the use of plastics in high barrier
to be exhibited by polymeric materials intended for use in
applications.
many packaging applications. The term high barrier is
usually applied in cases of low to very low permeability of a
material to low molecular weight chemical species, such as
gases and organic compounds. However, this property has PHENOMENOLOGY OF MASS TRANSPORT
perhaps never attracted so much industrial attention as over IN POLYMERS
recent decades, when it began to be associated with some According to the above, barrier properties in polymers are
modern food and beverage packaging technologies making necessarily associated with their inherent ability to permit
DOI 10.1179/026708304225010442 Materials Science and Technology January 2004 Vol. 20 1
2 Lagaron et al. High barrier properties in polymeric materials
the exchange, to a higher or lower extent, of low molecular equilibrium state and, consequently, there appears to exist
weight substances through mass transport processes such as a unique mode of penetrant transport for these polymers.
permeation. The permeation of low molecular weight Moreover, rubbery polymers are more amenable to showing
chemical species through a polymeric matrix is generally upwardly in ecting permeability responses with increasing
envisaged as a combination of two processes, i.e. solution penetrant partial pressure owing to plasticisation. This is
and diffusion.4 A permeate gas is rst dissolved into the typically the case with d-limonene, a common avour
upstream face of the polymer lm, and then undergoes component in fruit juices, contained in polymers such as
molecular diffusion to the downstream face of the lm polyethylene and polypropylene. By comparison, glassy
where it evaporates into the external phase again. A polymers are below their Tg , and hence require, on average,
solution – diffusion mechanism is thus applied, which can be long timescales to relax fully. Gas transport then typically
formally expressed in terms of permeability P, solubility S occurs in glassy polymers under non-thermodynamic equili-
and diffusion D coef cients by brium conditions. In this case, penetrant molecules can
P~DS : : : : : : : : : : : : : : : (1) allocate to holes on irregular cavities with very different
diffusional mobilities, and consequently more than one mode
The permeability coef cient P derives from application of of transport may be accessible. A ‘dual mode sorption’ model
Henry’s law of solubility to Fick’s rst law of diffusion, describes satisfactorilythe dependence of transport properties
according to on penetrant partial pressure in glassy polymers. This model
q ç c SDp Dp postulates the existence of two different molecular popula-
J~ ~{D ~D ~DS tions dissolved in a glass: one dissolved by an ordinary
At ç x l l
ql dissolution process which can typically follow Henry’s law
)P~DS~ : : : : : : : : : : (2) (c~Sp), and the other dissolved in a limited amount of xed
AtDp
microcavities which can be described by a Langmuir type
where J is the permanent ux, q is the heat quantity, A is the isotherm
cross-sectional area, t is the time, c is the concentration, CH bp
x is the direction of mass transport, p is the pressure and l is c~ : : : : : : : : : : : : : : (3)
the thickness. 1zbp
The solubility coef cient S is thermodynamic in nature, where CH is the hole saturation constant and b is the hole
and is de ned as the ratio of the equilibrium concentration af nity constant. More complex sorption behaviours have
of the dissolved penetrant in the polymer to its partial also been postulated for other glassy materials. For
pressure p in the gas phase (Henry’s law). In polymers, instance, a modi ed dual mode model requiring Langmuir
Henry’s law is usually obeyed at low penetrant concentra- and Flory – Huggings equations was suggested to explain
tions, i.e. when S is independent of concentration (or of the the sorption of water in an amorphous polyamide.9
partial pressure). The diffusion coef cient D characterises The present paper rst gives an overview of some relevant
the average ability of the sorbed permeate to move through structural factors de ning and/or altering high gas barrier
the polymer chain segments, and is determined from Fick’s properties in polymers, and then comments on recent
rst law of diffusion, i.e. the ux of the permeant J is pro- developments in the eld.
portional to the local gradient of concentration c through
the thickness of the polymer lm l. During sorption kinetic
experiments, if Fickian transport (case I) is assumed, a
linear behaviour in the penetrant uptake versus t1 /2 curve at Structural factors determining high
small times is usually observed.5 Case II diffusion is de ned barrier properties
when linear behaviour is observed in the uptake versus t
curve. This behaviour is observed in a number of systems, The main structural factor determining inherent high barrier
and is associated with large uptakes and plasticisation of the properties in polymers is fundamentally the chemistry, but
structure by the penetrant. When complex sorption there are also other relevant factors having a signi cant
behaviours, e.g. sigmoidal shapes, are observed, it is usually impact on barrier properties for a given chemistry, including
assumed that an ‘anomalous’ or non-Fickian transport polymer morphology (crystallinity, thermal history, mole-
occurs. Nevertheless, from recent studies, better rationalisa- cular orientation, etc.), polymer molecular architecture
tion of these ‘anomalous’ behaviours has been achieved, (branches, molecular weight and tacticity), polymer plasti-
whereby contributions from the effect of macroscopic cisation and others.
elastic constraints arising during the swelling process
(geometric effects) in adsorption experiments have been
pointed out.6 ,7
Concerning mechanisms of the mass transport process CHEMISTRY IS KEY
through polymeric materials, two general approaches can Nowadays, there is a large number of chemical combinations
be found, namely (a) molecular models studying the speci c and high throughput and selective catalyst technologies,
penetrant and chain motions in conjunction with the accessible via cutting edge polymer chemistry, to generate
corresponding intermolecular forces, and (b) ‘free volume’ polymeric materials with tailormade structures and proper-
models which pay attention to the relationships between the ties. As would be reasonable to expect, then, the chemistry is
transport coef cients and the free volume present in the the basic and main de ning factor determining barrier
polymeric matrix, without consideration of molecular scale properties in polymeric materials. Thus, by varying the
mechanisms (see Ref. 8 for a review of these). chemistry of the macromolecule,often by simply adjusting the
It is also relevant to emphasise here that the mass trans- pendant group along the polymer chain, a signi cantly large
port mechanisms as well as their dependence on permeant variation in barrier properties can be achieved. Figure 1
partial pressure and testing temperature are thought to be shows that the barrier properties to oxygen increase by more
generally different depending on whether the polymer is than ve orders of magnitude as a result of a change in the
in a rubbery or glassy state. Rubbery polymers are above functional group present in a repetitive basic ethylenic unit.
their glass transition temperature Tg , and therefore Some commonly employed acronyms applied to well known
exhibit very short relaxation times and respond quickly to commercial plastics are also indicated in Fig. 1; generic
physical changes. Thus, the absorption of small molecules names for the acronyms used throughout the paper are
or penetrants causes immediate adjustments to a new listed in the ‘Nomenclature’ above.
Materials Science and Technology January 2004 Vol. 20
Lagaron et al. High barrier properties in polymeric materials 3
2 Oxygen permeability P versus ratio of fractional free
volume (FFV)/cohesive energy density (CED) for some
widely used commercial polymers: unpublished data
after Duc et al.
1 Given functional groups pendant from a basic ethylenic
polymer chain and their relative oxygen permeability:
note that values along x axis are equally spaced for illus-
transient, to diffuse through the polymer matrix. The
trative purposes and therefore are not properly scaled transport properties of a permeant are therefore dependent
on the number and size of these microcavities. This concept
Associated with the signi cant changes in barrier proper- is usually expressed through the so called fractional free
ties resulting from chemistry variations are, for instance, the volume parameter Vf , and is indeed strongly related to
introduction of apolar voluminous groups at the low barrier chemistry (cohesive energy density), but is also related to a
side of the permeability spectrum, or the incorporation of number of other relevant factors having an impact on
small and strongly self-interacting chemical groups at the barrier properties, for example thermal history, polymer Tg ,
high barrier side of the permeability spectrum. As is well crystallinity and/or conformational order, etc. The frac-
known, most polymeric materials comprise exceedingly tional free volume can be readily determined by the
long, high molecular weight molecules (called polymer following simple expression
chains) which, for the case of the most widely used plastics, V {V0
the thermoplastics family, do not possess in the amorphous Vf ~ : : : : : : : : : : : : : (4)
V
state intermolecular links other than secondary forces of,
where V is the speci c volume of a particular polymer
for example, the van der Waals type. Consequently, the
sample determined from the density, and V0 is the speci c
presence of these various pendant groups can either disrupt
or enhance the high intermolecular cohesion necessary to volume at zero solubility (volume exclusively occupied by
maintain high barrier ef ciency against the transport of low polymer chains). The latter parameter can be experimen-
molecular weight substances. tally determined by, for instance, extrapolation of experi-
Moreover, the chemistry also de nes the af nity between mental data,1 1 or can be estimated from additive group
a potential permeant and the polymer matrix. As the pro- contribution models.1 0
cess of permeation is a bimodal process comprising solution A very useful concept for free volume is that proposed by
and diffusion, low solubility based on the chemical disparity Cohen and Turnbull12 ,13 and Fujita,1 4 through a general
of a permeant and the polymer matrix will also result in low expression
permeability, irrespective of whether the kinetics of diffu- D!exp({Bd =Vf ) : : : : : : : : : : : (5)
sion are going to be favourable to permeant transport. The
present paper concentrates, owing to its relevance and ease In this expression, D is the diffusion coef cient and Bd is a
of generalisation, on the barrier properties of non-interacting constant that depends only on the size of the penetrant
chemicals, as is usually the case for permanent gases. molecule. This model has been shown adequately to des-
A physical quantity termed the cohesive energy density cribe the transport kinetics of organic vapours and small gas
can be a useful parameter to help explain, quantify or even molecules in a number of polymers.
predict the behaviour in terms of barrier properties of More recent efforts, however, have led to the develop-
polymeric materials. The cohesive energy of a substance in a ment of an experimental methodology based on a technique
condensed state is de ned as the increase in internal energy called positronium annihilation spectroscopy. This metho-
per mole of substance if all the intermolecular forces are dology provides an experimental approach to determine
eliminated. For low molecular weight substances, this free volume, as it enables measurement of hole size on a
energy can be experimentally calculated from the heat of nanoscale and free volume fraction.1 5 Nevertheless, the
evaporation. However, for polymers, the cohesive energy absolute value of the fractional free volume cannot be
density (de ned as the cohesive energy per unit volume) can directly obtained from only positron lifetime measurements.
be estimated using additive group contribution models such Despite this, a recent study making use of positronium
as those devised by, for instance, Van Krevelen for cohesive annihilation spectroscopy showed that there exists an
energy and Traube for molar volume.1 0 These models excellent correlation between oxygen permeability and a
propose contribution values for each of the chemical entities relative fractional free volume parameter as determined by
building up the polymer chain. Consequently, this para- this technique in a number of EVOH copolymers.1 6 From
meter informs about the strength of the interaction between the experiments, it was clear that the fractional free volume
molecules, and how this interaction changes when different in these materials concerns mainly the free volume size, as
chemical groups are added to the polymer chain. The only the free volume size and not the orthopositronium o-Ps
cohesive energy density is often given as the square of the lifetime intensity, i.e. the number of holes, varied across
solubility parameter. composition in these polymers.
Another important factor strongly associated with It is relevant, therefore, to realise that high barrier
barrier properties is the free volume. The free volume com- polymers are the result of a permeable structure (amor-
prises the microcavities present in a polymeric material. phous phase) with a high cohesive energy density and very
Permeants make use of these cavities, whether permanent or low fractional free volume. Figure 2 plots the oxygen
Materials Science and Technology January 2004 Vol. 20
4 Lagaron et al. High barrier properties in polymeric materials
permeability of a number of plastics versus the ratio of
the fractional free volume/cohesive energy density. From
Fig. 2, it can be seen that the high barrier EVOH copolymer
(with 32 mol-% ethylene) is one of the most ef cient oxygen
barrier materials owing to its high intermolecular cohesion
and low fractional free volume. Consequently, this material
is being increasingly introduced in packaging applications
where high barrier properties to gases are required. In
contrast, polymers such as HDPE have much lower gas
barrier properties owing to low intermolecular cohesion and
large fractional free volume. However, high intermolecular
cohesion can be distorted by, for instance, chemical alter-
ations in the material (polymer degradation) as a result of
thermal treatments.1 7
Polymer chain rigidity or polymer Tg also plays a relevant
role in barrier properties since, as explained above,
penetrant transport mechanisms are greatly altered depend-
ing on whether the permeation process occurs above
(rubbery state) or below (glassy state) the polymer glass
transition temperature. There is a very general trend which
indicates that the higher is the polymer Tg , the lower is the
gas permeability and the better is the permselectivity.
However, this does not apply to common polymers such as
PS or PC, which are very rigid glassy materials with Tg
values above 100°C and very high permeability. This is of
course a consequence of the voluminous side groups, which 3 Schematic diagram of gas permeation process through
indeed reduce chain segment mobility owing to steric amorphous phase only
hindrance but in turn generate large fractional free volumes.
On the other hand, polymers such as EVOH copolymers, permeant has to travel across a lm thickness divided by its
PK copolymers or PVDC have lower Tg values than, for actual thickness. Furthermore, and as noted above, the
instance, PS, PC or other materials including PET, and yet presence of these crystalline blocks also affects the
have outstanding barrier properties. This is again a result of surrounding conformationally disordered amorphous
very high cohesive energy density and low fractional free phase. The constraining effects imposed by crystals to the
volume exhibited by the former materials. chain segments in the amorphous phase depend typically on
factors such as crystal surface area and penetrant size. This
phenomenon is substantiated from extensive mechanical
POLYMER MORPHOLOGY and transport data, which indicate clearly that the
An important issue implicit in all the above considerations segmental mobility of the non-crystalline fraction is much
is the well known characteristic that polymers are not less than that in the fully amorphous polymer.1 8 ,1 9 This
able to crystallise fully, some of them being in fact totally effect is taken into account in calculations of the transport
amorphous. Many polymers used in packaging applications coef cients (see equation (6)) by the so called chain
have therefore a semicrystalline nature, and hence are, from immobilisation factor t
a structural viewpoint, heterogeneous materials. These Damorphous
polymers contain, under the most simplistic two phase Dsemicrystalline ~ (1{x) : : : : : : : (6)
model visualisation, both a fraction of chain segments com- bt
prising highly packed and conformationally ordered three- As a result of this, being aware of the implications of
dimensional structures, the polymer crystalline fraction x, crystallinity and its morphology for the barrier properties is
and another fraction in an amorphous state without a relevant issue, because by adequate processing (thermal
conformational regularity and lateral order. As a large history) of polymers these parameters can be optimised to
body of experimental evidence suggests that polymer obtain specimens, based on the same chemistry, with
crystals are impermeable to the transport of most low enhanced permeability.
molecular weight substances, it is broadly accepted that the Polymer molecular orientation as a result of drawing or
amorphous phase is the only phase available for permeation processing leads generally to an increase in barrier proper-
of these substances (shown schematically in Fig. 3). ties. This is usually attributed to (a) orientation induced
It is therefore this particular structural feature, i.e. crystallisation, (b) fractionation and alignment (perpendi-
polymer crystallinity, together with a low intermolecular cular to the permeant transport) of the crystals in the
cohesion between polymer chains in the amorphous phase, straining direction (increase in the tortuosity factor) and (c)
that best de nes many of the most characteristic polymer to densi cation (reduction in free volume) of the amor-
properties including permeability. However, polymer crys- phous phase owing to an increase in conformational order
tals not only ll the molecular structure of semicrystalline in the non-crystalline chain segments. A recent paper1 1
materials with microscopic impermeable blocks, but also reported that oxygen permeability, diffusivity and solubility
affect the surrounding amorphous phase. To begin with, the parameters decreased with the amount of uniaxial orienta-
presence of crystallinity, its morphology (for instance tion in PET owing to conformational transformations of
crystal width/thickness ratio) and orientation present glycol linkages from gauche to trans. Orientation is then
additional considerations in terms of permeability, as the seen as the process of decreasing excess free volume,
penetrant molecules have to circumvent the crystallites and bringing the non-equilibrium glassy polymer closer to the
thereby travel through a more tortuous diffusive path equilibrium condition.
than in a fully amorphous material. This effect is usually A special case in barrier properties is that of liquid crystal
accounted for in calculations of the transport coef cients polymers. These materials can have gas barrier properties as
(see equation (6) below for diffusion) by the so called good as those of high barrier EVOH copolymers. Liquid
tortuosity or geometric impedance factor b. Thus, the crystal polymers are often termed ‘mesomorphic’ because
tortuosity factor is in essence the path length that a they have structures between those of amorphous polymers
Materials Science and Technology January 2004 Vol. 20
Lagaron et al. High barrier properties in polymeric materials 5
against the transport of oxygen. This is also the case, albeit
to a lesser extent, for other polar polymers such as the
polyamide family. Thus, it is often the situation that high
barrier polymers to gases have very low barrier performance
to polar solvents such as water. This behaviour is associated
with the disruption by moisture of the existing polymer
intermolecular self-association promoted by, for instance,
hydrogen bonding in EVOH, PVOH and PA.2 3 – 2 5 As
opposed to this behaviour, polymers such as polyole ns, PE
and PP have low barrier properties to gases owing to weak
self-association, but are extremely good barrier materials to
water as a result of their ole nic hydrophobic character.
Table 1 gives typical oxygen permeability and water permea-
bility values for a number of commercial polymers and
multilayer structures used in food packaging applications.2 6
An exceptional case is that of amorphous polyamide
4 Oxygen permeability P versus water activity for given (aPA) and some polyimides, for which oxygen permeability
ethylene – vinyl alcohol copolymers (EVOH) with vary- decreases with increasing relative humidity.8 For aPA, even
ing contents of ethylene though the presence of moisture decreases to a high extent
the polymer Tg , the oxygen permeability does not increase
with no regular order and those with a three-dimensional but surprisingly decreases (see Table 1). Recent spectro-
crystal lattice. The unique packing arrangement of these scopic work suggests that moisture has a speci c interaction
polymeric systems has raised some fundamental questions with this particular polymer.2 7 The results indicate that
about the permeation mechanisms of low molecular weight moisture molecules do not disrupt the originally existing
molecules, i.e. whether they behave more like glasses or hydrogen bonding intermolecular interactions between
conventional crystals.8 amide groups but rather link to the few remaining free
amide groups, and most of the sorbed water molecules self-
associate, forming clusters, which all together act as a free
POLYMER MOLECULAR ARCHITECTURE volume blocking mechanism to the diffusion of oxygen
Some relevant routes to modify the molecular architecture molecules.
of polymers, and hence the barrier properties, include
copolymerisation, i.e. introduction of a few side groups or
branches along the main chain, or modi cation of the
molecular weight or the stereoisomerism. Linear polyethy- Recent developments in the eld
lene (HDPE) is more crystalline than both branched
polyethylenes (e.g. LLDPEs and LDPE) and ultrahigh Recent developments in the high barrier eld come mainly
molecular weight polyethylenes, and is therefore found to from three sources, namely (a) new polymers, (b) polymer
be more dense, less permeable and stiffer, albeit less tough. blends and (c) nanocomposites. Polymeric materials for
Moreover, the homogeneous or heterogeneous character of high barrier applications are challenged today by a broad
the incorporation of branches along the polymer chains has range of stringent property requirements, including ease of
a large impact on properties, including barrier proper- processing, higher barrier properties to permanent gases, to
ties.2 0 ,2 1 The new polyole ns obtained by single site catalyst moisture and to low molecular weight organic compounds,
technologies can lead to very low density materials with excellent chemical resistance, permselectivity, low relative
unprecedented very low barrier properties, which in thin humidity dependence for barrier performance and ease of
lm form can serve as excellent packaging materials for recycling and biodegradability. Among the new high barrier
products that have breathing necessities, e.g. fruit and
vegetables. Another signi cant factor is the stereoisomerism Table 1 Water permeability P (at 38 C and 90% relative
(tacticity). This results from the various stereochemical humidity) and oxygen permeability P (23 C) of a
arrangements that can be present along the polymer number of commercial plastics and multilayer
backbone and that cannot be changed by rotation along structures: data from Ref. 26
the C-C bond. A polymer in which the pendant groups
contain the same con guration is said to be isotactic. Oxygen P, 1021 m3 m
(m2 s Pa) 2 1
Polymers in which alternate carbon atoms have the same Water P, 1018 kg m
con guration are called syndiotactic, and in which the Material (m2 s Pa) 2 1 Dry Wet
con guration is random are called atactic. The atactic
con guration is in principle more permeable, as it usually PVOH 485 000 0.17 900
EVOH 17 000 0.77 91
yields amorphous polymers (e.g. PS or PMMA). PAN 2420 1.9 ¼
PAN (70%AN) 8250 10 .5 31
PVDC 30 .53 4.5 ¼
POLYMER PLASTICISATION PA6 20 600 52 225
In this context, it is relevant to add here that polymer aPA 2420 83 60
PET 2300 135 ¼
plasticisation (Tg depletion) owing to polymer – permeant PP 726 6750 ¼
interactions or polymer and surrounding media chemical PC 19 400 10 500 ¼
interactions has very detrimental effects, which usually lead LDPE 1200 21 500 ¼
to losses in intermolecular cohesion and a decrease in LCP 10 0.42 ¼
PET ± PVDC 170 17 .5 ¼
overall barrier performance. As an example, Fig. 4 shows PA ± PVDC 160 18 .2 ¼
the variation in oxygen permeability of a number of EVOH PP ± PVDC 43 25 ¼
copolymers as a function of the percentage of relative PET (metallised) 58 3.5 ¼
humidity present in the environment.2 2 PET ± AlO x ± PE 21 7 15
PET ± SiOx ± PE 16 4.9 ¼
From Fig. 4, it can be readily observed that relative PA ± SiOx ± PE 32 7.7 ¼
humidity has a tremendous detrimental impact on the PP ± SiOx ± PE 13 81 ¼
outstanding barrier capacity of these EVOH copolymers
Materials Science and Technology January 2004 Vol. 20
6 Lagaron et al. High barrier properties in polymeric materials
containers.3 3 – 3 5 Also, a large number of blends have been
developed in which a high gas barrier polymer such as
EVOH is added to improve the barrier properties of a low
gas barrier material and, conversely, in which a high water
barrier polymer is added to a high gas barrier material to
reduce relative humidity dependence of the barrier proper-
ties of the latter.2 4
More recently, there has been a signi cant increase in the
amount of research devoted to enhancing relevant polymer
properties, mainly mechanical and barrier properties, but
also surface hardness, thermal stability and re retardancy,
by dispersing low loadings (usually less than 5 wt-%) of
exfoliated clay platelets with thickness on a nanometre scale
and with high aspect ratios in a conventional polymer
matrix. Gas and water vapour permeabilities have been
5 Modelling of oxygen permeability for various disposi- found to decrease, in some cases, to a large extent in the
tions of EVOH – amorphous polyamide (aPA) blend nanocomposites owing to, among other parameters,
components facing transport of oxygen gas as func- increased tortuosity factors.3 6 For example, an EPDM –
tion of volume fraction of EVOH: experimental data clay nanocomposite with a 4 wt-% loading had decreased
(indicated by arrow) for 80/20 EVOH – aPA and EVOH – N2 permeability by 30%, compared with EPDM alone.3 7
ionomer melt mixed blends recently developed in pre- Oxygen permeability decreased by a factor of three in
sent authors’ laboratory are also provided35 polyester – clay nanocomposites at 2.5 wt-% loading.3 4 A
60% reduction in the water permeability was measured in a
polymers being developed are materials such as the PK 5 wt-% loaded poly(vinyl alcohol) – sodium montmorillo-
copolymers (aliphatic polyketones).2 8 ,2 9 These semicrystal- nite nanocomposite, and the material still retained its
line materials have an outstanding range of mechanical, optical clarity.3 8
thermal and high barrier properties (comparable to some There are also a large number of technological develop-
EVOH copolymers), chemical resistance and reduced ments making use of existing materials and associated with
relative humidity dependence for barrier properties, which modern packaging technologies, aimed to tailor designs to
give them signi cant commercial potential in a broad range speci c performances and packaged product needs. These
of engineering, barrier packaging, bre and blend applica- developments include (a) multilayer systems comprising
tions. Other new ranges of promising materials currently various polymeric materials made by lamination, coextru-
being investigated for packaging applications are those sion or coinjection, (b) aluminium metallised polymeric
derived from biomass, and, therefore, to a higher or lower lms obtained by vacuum deposition technologies and (c)
extent readily biodegradable/compostable.3 0 ,3 1 Among oxide (AlOx or SiOx ) coated polymeric lms. Multilayer
these materials, it is possible to nd: (a) polymers systems in the food packaging eld usually comprise high
synthesised from bioderived monomers such as polylac- gas barrier polymers such as EVOH or PA sandwiched
tates; (b) polymers produced directly by organisms such as between structural layers of other polymers, which usually
PHAs, bacterial cellulose, etc.; and (c) polymers derived provide the assembly with a high barrier to moisture as well
directly from biomass such as polysaccharides (cellulose, as having other properties, i.e. thermal, mechanical, optical
starch, chitosan). These polymers can have excellent barrier or processing properties, printability, thermosolderability,
properties to gases, for instance in the case of plasticised etc. Coating plastics with vacuum deposited aluminium
chitosan, albeit the barrier performance is dramatically seeks to increase barrier properties to gases, moisture and
reduced in the presence of moisture. However, other poly- organic vapours, and results in better exibility, consumer
mers such as the PHAs have very high water barrier appeal and lower enviromental impact owing to a reduction
properties. Hence, in principle, a biomass derived high in metal consumption and better recyclability than in the
barrier multilayer system could be developed whereby an case of conventional lamination with aluminium foil.3 9 On
inner layer of plasticised chitosan could be sandwiched the other hand, the metal coating of polymeric lms imposes
between high moisture barrier PHA layers. An interesting reductions in exibility, stretchability and thermoformabil-
property of some of these biobased polymers, e.g. PLA and ity compared with the performance of the polymer lms
starch, is that the permeability of carbon dioxide compared alone. Moreover, aluminium coatings render the package
to that of oxygen (permselectivity) is higher than that of opaque, and it cannot be microwaved. To circumvent these
most conventional mineral oil based plastics. This is of problems, oxide coated polymer lms can be obtained that
interest for some food packaging applications where a high exhibit transparency and high barrier properties, albeit they
barrier to oxygen is required, but CO2 generated by the can be susceptible to coating microcraking during proces-
product should be allowed to exit the package head space to sing or handling.
avoid package swelling. These materials still suffer, how-
ever, from high production costs, competing with the
conventional plastic materials now on the market.
Blending of polymers is another route for accessing the References
desired balance of properties, by controlling the polymer
phase interaction and/or morphology.3 2 Figure 5 shows an 1. g. bureau and j. l. multon (ed.): ‘Food packaging technol-
example of modelling of the barrier properties of EVOH – ogy’, Vols. 1 and 2; 1995, New York, VCH Publishers.
aPA blends as a function of blend composition and blend 2. a. l. brody and k. s. marsh, (ed.): ‘The Wiley encyclopedia of
constituent disposition facing oxygen transport. High packaging technology’, 2nd edn; 1997, New York, Wiley.
barrier blends of EVOH with an ionomer and an amor- 3. j. f. hanlon, r. j. kelsey and h. e. forcinio: ‘Handbook of
phous polyamide have recently been developed in the package engineering’, 3rd edn; 1998, Lancaster, PA, Technomic
Publishing Co.
present authors’ laboratory.2 4 ,2 5 These blends show excel- 4. j. crank and g. s. park (ed.): ‘Diffusion in polymers’, 1968,
lent barrier properties to gases compared with neat EVOH New York, Academic Press.
(see experimental results for EVOH – aPA 80/20 blends in 5. z. zhang, i. j. britt and m. a. tung: J. Polym. Sci. B, Polym.
Fig. 5), and yet much better thermoformabilitythan EVOH Phys., 1999, 37, 691.
alone to produce thermoformed multilayer rigid food 6. m. a. samus and g. rossi: Macromolecules, 1996, 29, 2275.
Materials Science and Technology January 2004 Vol. 20
Lagaron et al. High barrier properties in polymeric materials 7
7. g. rossi: Trends Polym. Sci., 1996, 4, 337. 25. j. m. lagaron, e. gimenez, j. j. saura and r. gavara: Polymer,
8. w. j. koros (ed.): ‘Barrier polymers and structures’, ACS 2001, 42, 7381.
Symposium Series 423, 23; 1990, Washington, DC, American 26. ‘Permeability and other lm properties of plastics’, PDL
Chemical Society. Handbook Series; 1995, New York, Plastics Design Library.
9. r. j. hernandez, j. r. giacin and e. a. grulke: J. Membrane 27. j. m. lagaron, e. gimenez, r. catala and r. gavara: Macrom.
Sci., 1992, 65, 187. Chem. Phys., 2003, 202, Issue 4.
10. d. w. van krevelen (ed.): ‘Properties of polymers’; 1990, New 28. j. g. bonner and a. k. powell: in ‘213th National American
York, Elsevier. Chemical Society meeting’; 1997, Washington, DC, ACS
11. r. y. f. liu, d. a. schiraldi, a. hiltner and e. baer: J. Polym. Materials Chemistry Publications.
Sci. B, Polym. Phys., 2002, 40, 862. 29. j. m. lagaron and a. k. powell: US Patent WO 0042089,
12. m. h. cohen and d. turnbull: J. Chem. Phys. S., 1959, 31, 2000.
1164. 30. k. petersen, p. v. nielsen and m. b. olsen: Starch, 2001, 53,
13. d. turnbull and m. h. cohen: J. Chem. Phys., 1970, 52, 3038. 356.
14. h. fujita: Fortschr. Hochpolym. Forsch., 1961, 3, 1. 31. c. j. weber, v. haugaard, r. festersen and g. bertelsen: Food
15. k. tanaka, t. kawai, h. kita, k. okamoto and y. ito: Addit. Contam., 2002, 19, 172.
Macromolecules, 2000, 33, 5513. 32. d. r. paul and c. b. bucknall (ed.): ‘Polymer blends’, Vol. 2,
16. k. ito, y. saito, t. yamamoto, y. ujihira and k. nomura: ‘Performance’; 2000, New York, Wiley.
Macromolecules, 2001, 34, 6153. 33. j. m. lagaron, e. gimenez and r. gavara: Proc. 7th Symp. on
17. j. m. lagaron, e. gimenez and j. j. saura: Polym. Int., 2001, 50, ‘Polymer blends’, Lyon, France, May 2002.
635. 34. r. gavara, r. catala, e. gimÉnez, j. m. lagarÓn and c. sanz:
18. r. h. boyd: Polym. Eng. Sci., 1979, 19, 1010. in ‘WorldPack2002, improving the quality of life through
19. m. hedenqvist, a. angelstok, l. edsberg, p. t. larsson and packaging innovation’, Vol. 1, 400; 2002, Boca Raton, FL,
u. w. gedde: Polymer, 1996, 37, 2887. CRC Press.
20. j. m. lagaron, s. lopez-quintana, j. c. rodriguez-cabello, 35. j. m. lagaron, e. gimenez, v. del-valle, b. altava and
j. c. merino and j. m. pastor: Polymer, 2000, 41, 2999. r. gavara: Macromol. Symp., 2003, 198, 473.
21. b. neway, m. s. hedenqvist, v. b. f. mathod and u. w. gedde: 36. r. k. bharadwaj, a. r. hehrabi, c. hamilton, c. trujillo,
Polymer, 2001, 42, 5307. m. murga, r. fan, a. chavira and a. k. thompson: Polymer,
22. s. aucejo, r. catala and r. gavara: Food Sci. Technol. Int., 2002, 43, 3699.
2000, 6, 159 – 164. 37. a. usuki, a. tukigase and m. kato: Polymer, 2002, 43, 2185.
23. j. m. lagaron, a. k. powell and j. g. bonner: Polym. Test., 38. k. e. strawhecker and e. manias: Chem. Mater., 2000, 12,
2001, 20, (5), 569. 2943.
24. j. m. lagaron, e. gimenez, r. gavara and j. j. saura: Polymer, 39. r. s. a. kelly: Proc. IzD Packaging Conf., Seville, Spain,
2001, 42, 9531. 1992.
Materials Science and Technology January 2004 Vol. 20
You might also like
- Catalog VALSIR Canalizare PDFDocument60 pagesCatalog VALSIR Canalizare PDFAlexandru Ilioiu100% (1)
- Final Year Project Edi IrawanDocument75 pagesFinal Year Project Edi IrawanEdi IrawanNo ratings yet
- DSP UPG CU EngDocument1 pageDSP UPG CU EngArtur KwiatkowskiNo ratings yet
- Karakteristik PPDocument12 pagesKarakteristik PPRestu KusumawardaniNo ratings yet
- INTRODUCTION TO FOOD PACKAGING Chapter 5Document21 pagesINTRODUCTION TO FOOD PACKAGING Chapter 5Suleyman MiftahNo ratings yet
- Biodegradable Polymer Blends Based On Thermoplastic StarchDocument17 pagesBiodegradable Polymer Blends Based On Thermoplastic Starchelias llamasNo ratings yet
- Polymers 14 01626 v3Document44 pagesPolymers 14 01626 v3Pablo RaimondaNo ratings yet
- Distilasi PDFDocument8 pagesDistilasi PDFLusiana Eka KurniawatiNo ratings yet
- Hindaw I Publishing Cor P or Ation in Ter N Ational Jour N Al of Po Ly Mer S Cience Vo Lume 2012, Ar Ticle I D 302029, 11 Pages Doi:10.1155/2012/302029Document12 pagesHindaw I Publishing Cor P or Ation in Ter N Ational Jour N Al of Po Ly Mer S Cience Vo Lume 2012, Ar Ticle I D 302029, 11 Pages Doi:10.1155/2012/302029Sheren Mohamed NagibNo ratings yet
- Making Plastics From Monomer To PolymerDocument6 pagesMaking Plastics From Monomer To PolymerMichell Camargo JimenezNo ratings yet
- Migration of Phthalate and Non PhthalateDocument14 pagesMigration of Phthalate and Non PhthalateFREDY CORREANo ratings yet
- Biological Degradation of Polymers in The Environment: John A. GlaserDocument22 pagesBiological Degradation of Polymers in The Environment: John A. GlaserRahaf AljboriNo ratings yet
- Hypercrosslinked Polymers A ReviewDocument42 pagesHypercrosslinked Polymers A ReviewnkazemiNo ratings yet
- High Barrier Biodegradable Food PackagingDocument5 pagesHigh Barrier Biodegradable Food PackagingSarfrazNo ratings yet
- Advanced Industrial and Engineering Polymer Research: Manas ChandaDocument18 pagesAdvanced Industrial and Engineering Polymer Research: Manas ChandaElena RomeroNo ratings yet
- Effect of Chlorine Dioxide GasDocument9 pagesEffect of Chlorine Dioxide GasCarinna Saldaña - PierardNo ratings yet
- Water Vapor Transport Properties of Polyurethane FDocument13 pagesWater Vapor Transport Properties of Polyurethane FRosmery Naupari AlvarezNo ratings yet
- Mechanical Color and Barrier Properties of Biodegradable Nanocomposites Polylactic Acidnanoclay 2155 6199 1000455Document5 pagesMechanical Color and Barrier Properties of Biodegradable Nanocomposites Polylactic Acidnanoclay 2155 6199 1000455yurinnelNo ratings yet
- Extrusion Blow MoldingDocument12 pagesExtrusion Blow MoldingmaheshgupteNo ratings yet
- Mechanical Properties Molecular Structures HDPE Polymers Accepted-ManuscriptDocument29 pagesMechanical Properties Molecular Structures HDPE Polymers Accepted-Manuscriptsiddiqui4303379No ratings yet
- Polymer Engineering Sci - 2022 - Ramezani Dana - Synthesis Properties and Applications of Polylactic Acid BasedDocument22 pagesPolymer Engineering Sci - 2022 - Ramezani Dana - Synthesis Properties and Applications of Polylactic Acid Basedsabitapurohit04No ratings yet
- Study of The Degradation Mechanisms of Polyethylene During ReprocessingDocument9 pagesStudy of The Degradation Mechanisms of Polyethylene During ReprocessingJoselyn GaliciaNo ratings yet
- A Review On The Thermomechanical Properties and Biodegradation BehaviourDocument31 pagesA Review On The Thermomechanical Properties and Biodegradation BehaviourvalentinaNo ratings yet
- Hydrolytic Degradation of Bio-Based Polyesters-Effect of PH and TimeDocument8 pagesHydrolytic Degradation of Bio-Based Polyesters-Effect of PH and TimeAnnie LauNo ratings yet
- Qasim 2019 - Soil Burial Degradation of Polypropylene Starch BlendDocument7 pagesQasim 2019 - Soil Burial Degradation of Polypropylene Starch BlendDibe DivolorNo ratings yet
- Adv Ind & Eng Polymer Research 5 (2022) 60-69 - Chapter 2 - High-Performance Fibers and Tapes Based On PP and PEDocument10 pagesAdv Ind & Eng Polymer Research 5 (2022) 60-69 - Chapter 2 - High-Performance Fibers and Tapes Based On PP and PEAlexandre Ferro RochaNo ratings yet
- Progress in Organic Coatings: Short CommunicationDocument5 pagesProgress in Organic Coatings: Short CommunicationJuan Manuel Montoya SalazarNo ratings yet
- 2018 JMEMS AdhesionDocument12 pages2018 JMEMS AdhesionDr. Alois FabianiNo ratings yet
- CCL 31 2 365Document4 pagesCCL 31 2 365Fahtin NabilahNo ratings yet
- European Polymer Journal: Marina P. Arrieta, Juan López, Alberto Hernández, Emilio RayónDocument16 pagesEuropean Polymer Journal: Marina P. Arrieta, Juan López, Alberto Hernández, Emilio RayónORGEX TMNo ratings yet
- 0424 Ecsmge 2019 - VerstDocument9 pages0424 Ecsmge 2019 - VerstVetriselvan ArumugamNo ratings yet
- Polymers 13 01309Document19 pagesPolymers 13 01309Huy Tuan QuachNo ratings yet
- IPTC 11465 Enhancing Natural Gas Purification With Advanced Polymer/Molecular Sieve Composite MembranesDocument6 pagesIPTC 11465 Enhancing Natural Gas Purification With Advanced Polymer/Molecular Sieve Composite MembranesKaroll GeraldineNo ratings yet
- Io PlasticsDocument7 pagesIo PlasticserpandianNo ratings yet
- Valsir C PP l02882Document60 pagesValsir C PP l02882Budihardjo Sarwo SastrosudiroNo ratings yet
- Surfacebulk PDFDocument11 pagesSurfacebulk PDFVishwarup GoswamiNo ratings yet
- TrendsMbDesignPervaporation Subm PDFDocument28 pagesTrendsMbDesignPervaporation Subm PDFLugon HuaccachiNo ratings yet
- Cheng2021 Article OxygenBarrierFilmsOfScCO2-assiDocument16 pagesCheng2021 Article OxygenBarrierFilmsOfScCO2-assiPablo RaimondaNo ratings yet
- Processing and CharacterizationDocument18 pagesProcessing and CharacterizationKawaiiBunnehSukiNo ratings yet
- Muthu Raj 2015Document13 pagesMuthu Raj 2015Shivani BehareNo ratings yet
- Characterization of Metallized Biaxially Oriented Polypropylene FilmDocument10 pagesCharacterization of Metallized Biaxially Oriented Polypropylene FilmLaboratory Plant 7No ratings yet
- NCL Report - Chapter 4Document6 pagesNCL Report - Chapter 4Aparna YaduNo ratings yet
- The Effect of Extensive Mechanical RecycDocument11 pagesThe Effect of Extensive Mechanical RecycAhmet AltunNo ratings yet
- HdpeDocument9 pagesHdpeXuân Giang NguyễnNo ratings yet
- Permeation - Oxygen Packaging - 18 - 2016Document11 pagesPermeation - Oxygen Packaging - 18 - 2016muriloinnocentiniNo ratings yet
- Activation Energy For The Pyrolysis of Polymer WastesDocument6 pagesActivation Energy For The Pyrolysis of Polymer WastesSwiftTGSolutionsNo ratings yet
- Hidrolisis, Pirosis....Document26 pagesHidrolisis, Pirosis....Elena RomeroNo ratings yet
- Advances in Polyamide Nanocomposites A ReviewDocument20 pagesAdvances in Polyamide Nanocomposites A ReviewSalman khanNo ratings yet
- Polymers 14 02317Document35 pagesPolymers 14 02317Ana ursNo ratings yet
- Evaluation and Identification of Degradative Processes in Post Consumer Recycled HDPEDocument7 pagesEvaluation and Identification of Degradative Processes in Post Consumer Recycled HDPEAroop Ratan SenNo ratings yet
- Resources, Conservation & Recycling: ReviewDocument25 pagesResources, Conservation & Recycling: ReviewElena RomeroNo ratings yet
- App 38945Document11 pagesApp 38945Annisya ZahraNo ratings yet
- Supercritical Gel Drying: A Powerful Tool For Tailoring Symmetric Porous PVDF-HFP MembranesDocument10 pagesSupercritical Gel Drying: A Powerful Tool For Tailoring Symmetric Porous PVDF-HFP MembranesArdila Hayu TiwikramaNo ratings yet
- Lecture FIVE: Classification of Polymer MaterialsDocument8 pagesLecture FIVE: Classification of Polymer MaterialsSadiq SalamNo ratings yet
- Mechanical, Color and Barrier, Properties of Biodegradable. 2018Document7 pagesMechanical, Color and Barrier, Properties of Biodegradable. 2018yurinnelNo ratings yet
- Polymers and Polymers BlendsDocument51 pagesPolymers and Polymers Blendsziradagreat539No ratings yet
- Study of Liquid CrystalDocument10 pagesStudy of Liquid CrystalXinshi ChenNo ratings yet
- 1 s2.0 S0379677997806583 Main PDFDocument8 pages1 s2.0 S0379677997806583 Main PDFMuhammad Faisal AminNo ratings yet
- Articulo 6 PDFDocument7 pagesArticulo 6 PDFDeiby LealNo ratings yet
- Plastic Polymers PDFDocument8 pagesPlastic Polymers PDFnoor emadNo ratings yet
- Atmospheric Pressure Plasma Treatment of Polymers: Relevance to AdhesionFrom EverandAtmospheric Pressure Plasma Treatment of Polymers: Relevance to AdhesionNo ratings yet
- Plastics as Corrosion-Resistant Materials: The Commonwealth and International Library: Plastics DivisionFrom EverandPlastics as Corrosion-Resistant Materials: The Commonwealth and International Library: Plastics DivisionNo ratings yet
- Current Research Trends On Plastic Pollution and Ecological Impacts OnDocument9 pagesCurrent Research Trends On Plastic Pollution and Ecological Impacts OnMaria-MirabelaGherasimNo ratings yet
- Beyond Mechanical Recycling Giving New Life To Plastic WasteDocument38 pagesBeyond Mechanical Recycling Giving New Life To Plastic WasteChristhy Vanessa Ruiz MadroñeroNo ratings yet
- Solutions and Integrated Strategies For The Control and Mitigation of Plastic and Microplastic PollutionDocument19 pagesSolutions and Integrated Strategies For The Control and Mitigation of Plastic and Microplastic Pollutionashadi asriNo ratings yet
- Solvent-Based Separation and Recycling of Waste Plastics A ReviewDocument14 pagesSolvent-Based Separation and Recycling of Waste Plastics A ReviewChristhy Vanessa Ruiz MadroñeroNo ratings yet
- Sub - and Supercritical Water For Chemical Recycling of Plastic WasteDocument22 pagesSub - and Supercritical Water For Chemical Recycling of Plastic WasteChristhy Vanessa Ruiz MadroñeroNo ratings yet
- Shah2008 PDFDocument20 pagesShah2008 PDFadri2115No ratings yet
- Increased Plastic Pollution Due To COVID-19 Pandemic Challenges andDocument9 pagesIncreased Plastic Pollution Due To COVID-19 Pandemic Challenges andChristhy Vanessa Ruiz MadroñeroNo ratings yet
- Biodegradation of Micro-Polyethylene Particles by BacterialDocument7 pagesBiodegradation of Micro-Polyethylene Particles by BacterialChristhy Vanessa Ruiz MadroñeroNo ratings yet
- Polymer-Plastics Technology and EngineeringDocument9 pagesPolymer-Plastics Technology and EngineeringChristhy Vanessa Ruiz MadroñeroNo ratings yet
- Plastics MaterialsDocument206 pagesPlastics MaterialsChristhy Vanessa Ruiz MadroñeroNo ratings yet
- Recently Emerging Trends in PolymerDocument57 pagesRecently Emerging Trends in PolymerChristhy Vanessa Ruiz MadroñeroNo ratings yet
- Plastic Waste As A Global Challengeare Biodegradable PlasticsDocument9 pagesPlastic Waste As A Global Challengeare Biodegradable PlasticsChristhy Vanessa Ruiz MadroñeroNo ratings yet
- Plastics Recycling Challenges and OpportunitiesDocument13 pagesPlastics Recycling Challenges and OpportunitiesChristhy Vanessa Ruiz MadroñeroNo ratings yet
- Sub - and Supercritical Water For Chemical Recycling of Plastic WasteDocument22 pagesSub - and Supercritical Water For Chemical Recycling of Plastic WasteChristhy Vanessa Ruiz MadroñeroNo ratings yet
- Plastic Pollutantseffective Waste Management For PollutionDocument13 pagesPlastic Pollutantseffective Waste Management For PollutionChristhy Vanessa Ruiz MadroñeroNo ratings yet
- A Life Cycle Assessment ofDocument18 pagesA Life Cycle Assessment ofChristhy Vanessa Ruiz MadroñeroNo ratings yet
- Methods For Plastic RecyclingDocument19 pagesMethods For Plastic RecyclingAshik Shah100% (2)
- Recycling of PET: Firas Awaja, Dumitru PavelDocument25 pagesRecycling of PET: Firas Awaja, Dumitru PavelFernanda TenesacaNo ratings yet
- Plastics Recycling Challenges and OpportunitiesDocument13 pagesPlastics Recycling Challenges and OpportunitiesChristhy Vanessa Ruiz MadroñeroNo ratings yet
- Plastics Recycling Challenges and OpportunitiesDocument13 pagesPlastics Recycling Challenges and OpportunitiesChristhy Vanessa Ruiz MadroñeroNo ratings yet
- Polymer-Plastics Technology and EngineeringDocument9 pagesPolymer-Plastics Technology and EngineeringChristhy Vanessa Ruiz MadroñeroNo ratings yet
- Plastic Pollutantseffective Waste Management For PollutionDocument13 pagesPlastic Pollutantseffective Waste Management For PollutionChristhy Vanessa Ruiz MadroñeroNo ratings yet
- Recovery of PolyhydroxyalkanoatesDocument45 pagesRecovery of PolyhydroxyalkanoatesChristhy Vanessa Ruiz MadroñeroNo ratings yet
- Dehydration of Waterplasticized Poly (Vinylalcohol) Systemsparticular Behavior Ofisothermal Mass TransferDocument8 pagesDehydration of Waterplasticized Poly (Vinylalcohol) Systemsparticular Behavior Ofisothermal Mass TransferChristhy Vanessa Ruiz MadroñeroNo ratings yet
- IntercalationCarboxylicAcids LDHDocument12 pagesIntercalationCarboxylicAcids LDHChristhy Vanessa Ruiz MadroñeroNo ratings yet
- Characterization of Polyvinyl AlcoholDocument11 pagesCharacterization of Polyvinyl AlcoholChristhy Vanessa Ruiz MadroñeroNo ratings yet
- Polymer-Plastics Technology and EngineeringDocument9 pagesPolymer-Plastics Technology and EngineeringChristhy Vanessa Ruiz MadroñeroNo ratings yet
- Recently Emerging Trends in PolymerDocument57 pagesRecently Emerging Trends in PolymerChristhy Vanessa Ruiz MadroñeroNo ratings yet
- Crystal Structure of PVADocument2 pagesCrystal Structure of PVAChristhy Vanessa Ruiz MadroñeroNo ratings yet
- Research ProposalDocument2 pagesResearch Proposalsmh9662No ratings yet
- Generation Transmission and Distribution Notes.Document88 pagesGeneration Transmission and Distribution Notes.Abhishek NirmalkarNo ratings yet
- BLE Catalogue 2013Document21 pagesBLE Catalogue 2013Shahina Parvin ShaikNo ratings yet
- Physics 715 HW 1Document13 pagesPhysics 715 HW 1Antonildo PereiraNo ratings yet
- BarDocument1 pageBarJoannalyn Libo-onNo ratings yet
- كتالوج وصفي للعملات الرومانية النادرة وغير المحررة من الفترة الأولى للعملات المعدنية الرومانية إلى انقراض الإمبراطورية تحت حكDocument570 pagesكتالوج وصفي للعملات الرومانية النادرة وغير المحررة من الفترة الأولى للعملات المعدنية الرومانية إلى انقراض الإمبراطورية تحت حكFayza MaghrabiNo ratings yet
- 990XP Bandit ChipperDocument5 pages990XP Bandit ChipperFrancisco ConchaNo ratings yet
- Brand Loyalty & Competitive Analysis of Pankaj NamkeenDocument59 pagesBrand Loyalty & Competitive Analysis of Pankaj NamkeenBipin Bansal Agarwal100% (1)
- Cognitive Benefits of Language LearningDocument11 pagesCognitive Benefits of Language LearningIlhamdi HafizNo ratings yet
- CREDEDocument10 pagesCREDEDaffodilsNo ratings yet
- Teacher Thought For InterviewDocument37 pagesTeacher Thought For InterviewMahaprasad JenaNo ratings yet
- Anti LeproticDocument9 pagesAnti LeproticMeenakshi shARMANo ratings yet
- PlayStation MagazineDocument116 pagesPlayStation MagazineFrank Costello67% (3)
- Exercise 3 - Wireframe Geometry Creation and Editing - Rev ADocument33 pagesExercise 3 - Wireframe Geometry Creation and Editing - Rev AdevNo ratings yet
- John Deere CaseDocument2 pagesJohn Deere CaseAldo ReynaNo ratings yet
- Screen 2014 Uricchio 119 27Document9 pagesScreen 2014 Uricchio 119 27NazishTazeemNo ratings yet
- BS en 13369-2018 - TC - (2020-11-30 - 09-45-34 Am)Document164 pagesBS en 13369-2018 - TC - (2020-11-30 - 09-45-34 Am)Mustafa Uzyardoğan100% (1)
- Advantage Dis OqpskDocument5 pagesAdvantage Dis OqpskHarun AminurasyidNo ratings yet
- RTI SpicesDocument226 pagesRTI SpicesvivebajajNo ratings yet
- Q1 WK 2 To 3 Las Fabm2 Kate DionisioDocument8 pagesQ1 WK 2 To 3 Las Fabm2 Kate DionisioFunji BuhatNo ratings yet
- Language - Introduction To The Integrated Language Arts CompetenciesDocument7 pagesLanguage - Introduction To The Integrated Language Arts CompetenciesHari Ng Sablay100% (1)
- With Pneumatic and Electric Actuators: Datasheet 448001 EnglishDocument7 pagesWith Pneumatic and Electric Actuators: Datasheet 448001 EnglishPinak ProjectsNo ratings yet
- eLearnMarkets OptionsBuying HindiDocument17 pageseLearnMarkets OptionsBuying Hindisrinivas20% (1)
- FreeBSD HandbookDocument26 pagesFreeBSD Handbookhembeck119No ratings yet
- Cement Grouted Rock BoltsDocument28 pagesCement Grouted Rock BoltsBhaskar ReddyNo ratings yet
- Analysis and Design of Suspended Buildings: Creative and Innovative Report-1Document14 pagesAnalysis and Design of Suspended Buildings: Creative and Innovative Report-1Nirmal RaviNo ratings yet
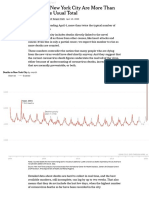
- Deaths in New York City Are More Than Double The Usual TotalDocument3 pagesDeaths in New York City Are More Than Double The Usual TotalRamón RuizNo ratings yet
- Draft Technical Notes SGLG 2023 - National OrientationDocument109 pagesDraft Technical Notes SGLG 2023 - National OrientationZane ZyneNo ratings yet