Professional Documents
Culture Documents
Molecular Orbitals
Molecular Orbitals
Uploaded by
DeviOriginal Description:
Original Title
Copyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
Molecular Orbitals
Molecular Orbitals
Uploaded by
DeviCopyright:
Available Formats
Molecular Orbitals
An approach to bonding in which orbitals
encompass the entire molecule, rather than
being localized between atoms.
Muhammad Asif Pakistan
Molecular Orbitals
Molecular orbitals result from the
combination of atomic orbitals.
Since orbitals are wave functions, they can
combine either constructively (forming a
bonding molecular orbital), or destructively
(forming an antibonding molecular orbital).
Muhammad Asif Pakistan
Molecular Orbitals
Molecular orbitals form when atomic orbitals
with similar energies and proper symmetry can
overlap.
Atomic orbitals with differing energies or the
wrong spatial orientation (orthogonal) do not
combine, and are called non-bonding orbitals.
Muhammad Asif Pakistan
Need for MO Theory
Valence bond theory fails to explain the
bonding in many simple molecules.
The oxygen molecule has a bond length and
strength consistent with a double bond, and it
contains two unpaired electrons.
Muhammad Asif Pakistan
Need for MO Theory
Valence bond theory predicts the double
bond, but not the paramagnetism of oxygen.
O=O
: :
: :
Muhammad Asif Pakistan
Need for MO Theory
Resonance is another example of the
limitations of valence bond theory. Bond
lengths and strengths are intermediate between
single, double or triple bonds.
Molecular orbital theory is often a better
approach to use with molecules that have
extended π systems.
Muhammad Asif Pakistan
Molecular Orbital Theory
In order to simplify things, we’ll consider the
interaction of the orbitals containing valence
electrons to create molecular orbitals.
The wave functions of hydrogen atom A and
hydrogen atom B can interact either
constructively or destructively.
Muhammad Asif Pakistan
Molecular Orbital Theory
Constructively:
Ψ(σ) or Ψ+ = (1/√2 ) [φ(1sa) + φ(1sb) ]
Destructively:
Ψ(σ*) or Ψ- = (1/√2 ) [φ(1sa) - φ(1sb) ]
Muhammad Asif Pakistan
Molecular Orbital Theory
The bonding orbital
results in increased
electron density between
the two nuclei, and is of
lower energy than the
two separate atomic
orbitals.
Muhammad Asif Pakistan
Molecular Orbital Theory
The antibonding
orbital results in a node
between the two nuclei,
and is of greater energy
than the two separate
atomic orbitals.
Muhammad Asif Pakistan
Molecular Orbital Theory
The result is an
energy level diagram with
the bonding orbital
occupied by a pair of
electrons. The filling of
the lower molecular
orbital indicates that the
molecule is stable
compared to the two
individual atoms.
Muhammad Asif Pakistan
Molecular Orbital Theory
The bonding orbital is
+ - sometimes given the
notation σg, where the g
stands for gerade, or
+ symmetric with respect
to a center of inversion.
The signs on the molecular orbitals indicate the sign of
the wave function, not ionic charge.
Muhammad Asif Pakistan
Molecular Orbital Theory
The anti-bonding orbital
+ - is sometimes given the
notation σu, where the u
stands for ungerade, or
+ asymmetric with respect
to a center of inversion.
The signs on the molecular orbitals indicate the sign of
the wave function, not ionic charge.
Muhammad Asif Pakistan
Rules for Combining Atomic
Orbitals
1. The number of molecular orbitals = the
number of atomic orbitals combined.
2. The strength of the bond depends upon the
degree of orbital overlap.
Muhammad Asif Pakistan
Experimental Evidence
Photoelectron spectroscopy (PES) is a
technique in which a beam of ultraviolet light
with an energy of 21 eV is used to irradiate
molecules.
The energy is high enough to eject electrons.
The kinetic energy of the emitted electrons is
measured, and used to determine the energy
level of the electron.
Muhammad Asif Pakistan
Experimental Evidence
The technique allows
for the measurement
of specific ionization
energies (I). Each
ionization energy
represents the
removal of an
electron from a
specific molecular
orbital.
Muhammad Asif Pakistan
Experimental Evidence
Electrons in
lower energy levels
require more energy
to be removed, and
are ejected with less
kinetic energy.
hνo = I + Ekinetic
Muhammad Asif Pakistan
Period 2 Diatomic Molecules
For the second period, assume that, due to a
better energy match, s orbitals combine with s
orbitals, and p orbitals combine with p orbitals.
The symmetry of p orbitals permits end-on-
end overlap along the bond axis, or side-by-side
overlap around, but not along, the internuclear
axis.
Muhammad Asif Pakistan
MOs using p orbitals
+ - + -
- + -
With the x axis as the bond axis, the px
orbitals may combine constructively or
destructively. The result is a σ bonding orbital
and a σ anti-bonding orbital.
Muhammad Asif Pakistan
MOs using p orbitals
+ - + -
- + -
The designation σ indicates symmetric
electron density around the internuclear (x) axis.
The + and – signs indicate the sign of the wave
function, and not electrical charges.
Muhammad Asif Pakistan
MOs using p orbitals
+ - + -
- + -
Some texts will use the symmetry
designations of g (gerade) or u (ungerade) instead
of indicating bonding or anti-bonding.
Muhammad Asif Pakistan
MOs using p orbitals
+ - + -
- + - σg
For these orbitals, the bonding orbital is
gerade, or symmetric around the bond axis.
Muhammad Asif Pakistan
MOs using p orbitals
+ - + - σu
- + - σg
For these orbitals, the anti-bonding orbital is
asymmetric about the bond axis, and is
designated as σu. Note that the designations of u
or g do not correlate with bonding or anti-
bonding.
Muhammad Asif Pakistan
π Molecular Orbitals
+ -
- +
+
side-by-side -
overlap
The orbital overlap side-by-side is less than
that of overlap along the bond axis (end-on-
end). As a result, the bonding orbital will be
higher in energy than the previous example.
Muhammad Asif Pakistan
π Molecular Orbitals
+ -
- +
+
side-by-side -
overlap
π orbitals are asymmetric with respect to the
bond axis. There is electron density surrounding
the bond axis, with a node along the internuclear
axis.
Muhammad Asif Pakistan
π Molecular Orbitals
+ -
- +
+
πu
side-by-side -
overlap
Some texts use the subscripts g and u instead
of bonding and anti-bonding. In this example,
the bonding orbital is ungerade, or asymmetric
about a center of symmetry.
Muhammad Asif Pakistan
π Molecular Orbitals
+ - πg
- +
+
πu
side-by-side -
overlap
The anti-bonding orbital is gerade, or
symmetric about a center of symmetry.
Muhammad Asif Pakistan
Molecular Orbital Diagram
This is a molecular
orbital energy level σu
diagram for the p πg
orbitals. Note that the 2p 2p
σ bonding orbital is πu
lowest in energy due to σg
the greater overlap
end-on-end.
Muhammad Asif Pakistan
Molecular Orbital Diagram
The alternate σu
notation is provided πg
on the right side of the 2p 2p
energy level diagram. πu
σg
Muhammad Asif Pakistan
Molecular Orbital Diagrams
1. Electrons preferentially occupy molecular
orbitals that are lower in energy.
2. Molecular orbitals may be empty, or contain
one or two electrons.
3. If two electrons occupy the same molecular
orbital, they must be spin paired.
4. When occupying degenerate molecular
orbitals, electrons occupy separate orbitals
with parallel spins before pairing.
Muhammad Asif Pakistan
Molecular Orbital Diagrams
Although molecular orbitals form from inner
(core) electrons as well as valence electrons,
many molecular orbital diagrams include only
the valence level.
Muhammad Asif Pakistan
Molecular Orbital Diagrams
For O2, there
will be a total of
12 valence
electrons that
must be placed in
the diagram.
Muhammad Asif Pakistan
Molecular Orbital Diagrams
For O2, there
will be a total of
12 valence
electrons that
must be placed in
the diagram.
Muhammad Asif Pakistan
Molecular Orbital Diagrams
For O2, there
will be a total of
2p 2p 12 valence
electrons that
must be placed in
the diagram.
2s 2s
Muhammad Asif Pakistan
MO Diagram for O2
σ*u
The molecular
π*g
orbital diagram for
2p 2p oxygen shows two
πu
σg
unpaired electrons,
σ*u
consistent with
2s 2s experimental data.
σg
Muhammad Asif Pakistan
Bond Order
Bond order is an indicator of the bond
strength and length. A bond order of 1 is
equivalent to a single bond. Fractional bond
orders are possible.
The bond order of the molecule =
(# e- in bonding orbtls) - (# e- in anti-bonding orbtls)
2 2
Muhammad Asif Pakistan
MO Diagram for O2
The bond order of
σ*u
O2 is:
π*g
2p 2p 8-4 = 2
πu 2
σg
σ*u This is consistent
2s 2s with a double
σg bond.
Muhammad Asif Pakistan
MO Diagram for O2
This energy level
σ*u
diagram works well
π*g
for atoms in which
2p 2p the 2s and 2p levels
πu
are fairly far apart.
σg These are the
σ*u elements at the right
2s 2s of the table: O, F and
σg Ne.
Muhammad Asif Pakistan
Experimental Evidence
Oxygen is paramagnetic, consistent with having
two unpaired electrons. In addition, photoelectron
spectroscopy (PES) can be used for determining orbital
energies in molecules. The molecule is bombarded with
UV or X-rays to remove an electron from the molecule.
The kinetic energy of the emitted electron is measured
and subtracted from the incident radiation to determine
the binding energy of the electron.
Muhammad Asif Pakistan
Photoelectron Spectroscopy
The result is a spectrum of absorptions which are
correlated to the molecular orbitals of the molecule. In
addition, electrons ejected from bonding orbitals show
more vibrational energy levels than electrons emitted
from anti-bonding or non-bonding orbitals.
Muhammad Asif Pakistan
Muhammad Asif Pakistan
MO diagram for Li through N
The elements on the left side of period 2
have a fairly small energy gap between the 2s
and 2p orbitals. As a result, interaction between
s and p orbitals is possible. This can be viewed
in different ways.
Muhammad Asif Pakistan
MO diagram for Li through N
In some approaches, the s orbital on one atom
interacts with the p orbital on another. The interaction
can be constructive or destructive.
Muhammad Asif Pakistan
MO diagram for Li through N
In another approach, the s and p orbitals on
the same atom interact in what is called orbital
mixing.
Either approach yields the same result. The
σ bonding and anti-bonding orbitals are raised in
energy due to the interaction with a p orbital.
Muhammad Asif Pakistan
MO diagram for Li through N
σ*u
π*g
σg
πu
σ*u
σg
Muhammad Asif Pakistan
MO diagram for N2
σ*u N2 has 10
π*g valence
electrons.
σg
πu
σ*u
σg
Muhammad Asif Pakistan
Experimental Evidence
The photoelectronic
spectrum of nitrogen is
consistent with a
molecular orbital
approach.
Electrons emitted
σg from bonding orbitals
πu show vibrational
excitations.
σ*u
Muhammad Asif Pakistan
Experimental Evidence
σ*u
π*g
σg
σg πu
πu
σ*u
σ*u
σg
Muhammad Asif Pakistan
Heteronuclear Diatomic
Molecules
The more electronegative atom will have
orbitals of lower energy, and therefore
contribute more to the bonding orbitals.
The less electronegative atom has orbitals of
higher energy, and contributes more to the anti-
bonding orbitals.
Muhammad Asif Pakistan
Rules for Combining Atomic
Orbitals
For heteronuclear molecules:
1. The bonding orbital(s) will reside
predominantly on the atom of lower orbital
energy (the more electronegative atom).
2. The anti-bonding orbital(s) will reside
predominantly on the atom with greater orbital
energy (the less electronegative atom).
Muhammad Asif Pakistan
HF
The 2s and 2px orbitals
on fluorine interact with
the 1s orbital on hydrogen.
The py and pz orbitals
on fluorine lack proper
symmetry to interact with
hydrogen, and remain as
non-bonding orbitals.
Muhammad Asif Pakistan
HF
The anti-bonding
orbital resides primarily on
the less electronegative
atom (H).
Note that the
subscripts g and u are not
used, as the molecule no
longer has a center of
symmetry.
Muhammad Asif Pakistan
Carbon monoxide
In carbon
monoxide, the bonding
orbitals reside more on
the oxygen atom, and
the anti-bonding
orbitals reside more on
the carbon atom.
Muhammad Asif Pakistan
Carbon monoxide
CO is a highly
reactive molecule with
transition metals.
Reactivity typically
arises from the highest
occupied molecular
orbital (HOMO), when
donating electrons.
Muhammad Asif Pakistan
Carbon monoxide
When acting as an
electron pair acceptor,
the lowest
unoccupied molecular
orbital (LUMO), is
significant.
Muhammad Asif Pakistan
Carbon monoxide
When acting as an
electron pair donor,
the highest occupied
molecular orbital
(HOMO), is
significant.
Muhammad Asif Pakistan
The highest
occupied molecular
orbital of CO is a
molecular orbital
which puts
significant electron
density on the
carbon atom.
Muhammad Asif Pakistan
The lowest
unoccupied
molecular orbital of
CO is the π* orbitals.
The lobes of the
LUMO are larger on
the carbon atom than
on the oxygen atom.
Muhammad Asif Pakistan
CO as a Ligand
Carbon monoxide is known as a σ donor and
a π acceptor ligand. It donates electrons from its
HOMO to form a sigma bond with the metal.
Muhammad Asif Pakistan
CO as a Ligand
Carbon monoxide accepts electrons from
filled d orbitals on the metal into its antibonding
(LUMO) orbital.
Muhammad Asif Pakistan
CO as a Ligand
This phenomenon is called back bonding. The
increased electron density in the antibonding orbitals of
CO causes an increase in the C-O bond length and a
decrease in its stretching frequency.
Muhammad Asif Pakistan
MOs for Larger Molecules
Group theory is usually used to develop
molecular orbital diagrams and drawings of
more complicated molecules. When a central
atom is bonded to several atoms of the same
element (H2O, BF3, or PtCl42-], group theory can
be used to analyze the symmetry of the orbitals
of the non-central atoms, and then combine
them with the appropriate orbitals of the central
atom.
Muhammad Asif Pakistan
MOs for Larger Molecules
The orbitals of the non-central atoms are
called group orbitals. In considering a simple
example, H2O, we obtain group orbitals using
the two 1s orbitals on the hydrogen atoms.
Muhammad Asif Pakistan
The characters for the
group orbitals is obtained by
considering each hydrogen
as a spherical 1s orbital.
They remain in position for
identity, are exchanged
during rotation, remain in
place for σxz (the molecular
plane), and are exchanged
for σyz.
Muhammad Asif Pakistan
Group Orbitals of Water
Γred and its irreducible representations are:
Muhammad Asif Pakistan
Group Orbitals of Water
The A1 representation has both 1s orbitals with
positive wave functions: Ha+Hb.
The B1 representations is Ha+Hb.
Muhammad Asif Pakistan
Group Orbitals of Water
These group orbitals are combined with orbitals
on oxygen that have the same symmetry.
Muhammad Asif Pakistan
Group Orbitals of Water
The 2s and 2pz orbital on oxygen have
A1 symmetry, the 2px orbital has B1 symmetry,
and the 2py has B2 symmetry.
Muhammad Asif Pakistan
Molecular Orbitals of Water
Since the 2py orbital on oxygen doesn’t
match the symmetry of the group orbitals of
hydrogen, it will remain non-bonding. The
other orbitals on oxygen will combine with the
appropriate group orbitals to form bonding and
antibonding molecular orbitals.
Muhammad Asif Pakistan
Muhammad Asif Pakistan
MOs for Larger Molecules
Group theory is usually used to develop
molecular orbital diagrams and drawings of
more complicated molecules. A simplified
example will be shown for the π bonding of
benzene.
Muhammad Asif Pakistan
π Bonding of Benzene
Benzene belongs to point group D6h. In
determining the orbital combinations for π
bonding, we need to obtain Гπ by looking only at
the pz orbitals on each carbon atom.
We need only consider
those orbitals on carbon
atoms that remain in place
for a given symmetry
operation.
Muhammad Asif Pakistan
π Bonding of Benzene
C″2 C′2
z axis
{
D6h E 2C6 2C3 C2 3C′2 3C″2 i 2S3 2S6 σh 3 σd 3 σv
Гπ
Muhammad Asif Pakistan
π Bonding of Benzene
C″2 C′2
z axis
{
D6h E 2C6 2C3 C2 3C′2 3C″2 i 2S3 2S6 σh 3 σd 3 σv
Гπ 6 0 0 0 -2 0 0 0 0 -6 0 2
Muhammad Asif Pakistan
π Bonding of Benzene
C″2 C′2
z axis
{
D6h E 2C6 2C3 C2 3C′2 3C″2 i 2S3 2S6 σh 3 σd 3 σv
Гπ 6 0 0 0 -2 0 0 0 0 -6 0 2
This reduces to: B2g + E1g + A2u + E2u
Muhammad Asif Pakistan
π Bonding of Benzene
Гπ: B2g + E1g + A2u + E2u
Group theory can be used to draw each of
the π molecular orbitals. Molecular orbitals with
fewer nodes are lower in energy (more bonding),
and those with more nodes are higher in energy
(more antibonding).
Muhammad Asif Pakistan
π Bonding of Benzene
Гπ: B2g + E1g + A2u + E2u
A2u fully bonding and
lowest in energy
E1g degenerate
bonding orbitals
with one node
Muhammad Asif Pakistan
π Bonding of Benzene
Гπ: B2g + E1g + A2u + E2u
E2u degenerate
largely anti-
bonding orbitals
with two nodes
B2g fully anti-
bonding orbital
with three nodes
Muhammad Asif Pakistan
π Bonding of Benzene
B2g
E2u
E1g
A2u
Muhammad Asif Pakistan
Molecular Orbitals of Complexes
Group theory is also used to construct
molecular orbital diagrams for the complexes of
metal atoms or ions. The symmetry
combinations of the atomic orbitals on the
ligands are determined, and then “matched”
with appropriate atomic orbitals on the central
metal. Both σ and π bonding between the metal
and ligands can be considered.
Muhammad Asif Pakistan
You might also like
- General Chemistry: More Than Two Electrons (With Opposite Spin) ."Document5 pagesGeneral Chemistry: More Than Two Electrons (With Opposite Spin) ."Marc Vincent CastilloNo ratings yet
- Point GroupsDocument34 pagesPoint GroupsDeviNo ratings yet
- Advertising ReportDocument64 pagesAdvertising Reportswatigupta88No ratings yet
- Management of Eyelid Tumors in Daily Practice: Department of Ophthalmology Faculty of Medicine Sultan Agung UniversityDocument41 pagesManagement of Eyelid Tumors in Daily Practice: Department of Ophthalmology Faculty of Medicine Sultan Agung UniversityWahyudi Bambang SukocoNo ratings yet
- Molecular OrbitalsDocument80 pagesMolecular Orbitals1balamanian100% (2)
- Flange ALignmentDocument5 pagesFlange ALignmentAnonymous O0lyGOShYG100% (1)
- Mo TheoryDocument80 pagesMo TheoryMridul Bhaskar0% (1)
- UV-Visible Spectroscopy: Dr. V. KrishnakumarDocument32 pagesUV-Visible Spectroscopy: Dr. V. KrishnakumarRafael HenriqueNo ratings yet
- Molecular Orbital TheoryDocument22 pagesMolecular Orbital TheoryRizaldi Al FauzanNo ratings yet
- UV-Visible Spectroscopy: Dr. V. KrishnakumarDocument32 pagesUV-Visible Spectroscopy: Dr. V. KrishnakumarDarpan KumarNo ratings yet
- Chemistry Questions OnlyDocument66 pagesChemistry Questions OnlyBHUVAN I (RA2111003011433)No ratings yet
- Molecular Orbital TheoryDocument11 pagesMolecular Orbital Theoryannym2665No ratings yet
- Chem Unit 1Document25 pagesChem Unit 1dgoutham4926No ratings yet
- Molecular Orbital TheoryDocument4 pagesMolecular Orbital TheoryYuexun ZhangNo ratings yet
- Electric Field in MatterDocument17 pagesElectric Field in MatterLasmaenita SiahaanNo ratings yet
- Unit - I: Molecular Structure and Theories of BondingDocument7 pagesUnit - I: Molecular Structure and Theories of BondingParadox 1883No ratings yet
- Section (A) : Molecular Orbital Theory (MOT) : Chemical Bonding-IVDocument8 pagesSection (A) : Molecular Orbital Theory (MOT) : Chemical Bonding-IVwanderedNo ratings yet
- Unit - I: Molecular Structure and Theories of BondingDocument13 pagesUnit - I: Molecular Structure and Theories of BondingAthirath VeldandaNo ratings yet
- Chemical Bonding Ncert-Part-2Document10 pagesChemical Bonding Ncert-Part-2gameing samrajayNo ratings yet
- Molecular Orbital TheoryDocument15 pagesMolecular Orbital TheoryDr. Md. Ehtesham Ul HoqueNo ratings yet
- Molecular Orbital Theory: Chemistry PresentationDocument15 pagesMolecular Orbital Theory: Chemistry PresentationMuntaha DaudpotaNo ratings yet
- Molecular Orbital Theory: Inorganic ChemistryDocument15 pagesMolecular Orbital Theory: Inorganic ChemistryGokul GanesanNo ratings yet
- 10 Lecture Notes-All UnitsDocument109 pages10 Lecture Notes-All UnitsSathish Kumar KurapatiNo ratings yet
- Final Exam Study GuideDocument43 pagesFinal Exam Study GuideAndrés Ignacio Irribarra RamírezNo ratings yet
- Struktur Atom Dan Ikatan - Kimor 1Document30 pagesStruktur Atom Dan Ikatan - Kimor 1akbar_rozaaqNo ratings yet
- Chem 7Document10 pagesChem 70No ratings yet
- Lecture9 SMatrixCompoundNucleusHypothesisOpticalModel2019Document17 pagesLecture9 SMatrixCompoundNucleusHypothesisOpticalModel2019Sagar RawalNo ratings yet
- (12942) Sheet Chemical Bonding 4 Theory eDocument8 pages(12942) Sheet Chemical Bonding 4 Theory eAnurag SinghNo ratings yet
- Superconducting Spintronics: Review ArticlesDocument9 pagesSuperconducting Spintronics: Review ArticlesSriNo ratings yet
- Review Basic Chemical ConceptsDocument4 pagesReview Basic Chemical ConceptsMary♡No ratings yet
- 1.2,1.3, 1.4Document55 pages1.2,1.3, 1.4Firdaus HadiNo ratings yet
- Valence Bond Theory VBTDocument13 pagesValence Bond Theory VBTWassachol SumarasinghaNo ratings yet
- Theories of Covalent Bond and Shapes of MoleculeDocument12 pagesTheories of Covalent Bond and Shapes of Moleculemuhwaqar80No ratings yet
- CH 9 (Cont'd)Document8 pagesCH 9 (Cont'd)PineraserNo ratings yet
- 1.2! Describing Chemical Bonds - Molecular Orbital TheoryDocument2 pages1.2! Describing Chemical Bonds - Molecular Orbital TheorySadeeq ArtxzNo ratings yet
- Lecture 07 - OM - EM PDFDocument33 pagesLecture 07 - OM - EM PDFHemant KumarNo ratings yet
- Applied ChemistryDocument11 pagesApplied ChemistryMaqsood Ahmad KhanNo ratings yet
- PK Sinha (LCAO PPT) @-1Document8 pagesPK Sinha (LCAO PPT) @-1MIN 121 Ravi RanjanNo ratings yet
- Lec #7 - Electric Field in Matter-II - 18PHY303 ElectrodynamicsDocument23 pagesLec #7 - Electric Field in Matter-II - 18PHY303 Electrodynamicskrishnanunnimanoj2004No ratings yet
- Chemistry Unit-1Document32 pagesChemistry Unit-1RajeshNo ratings yet
- XI Chem Unit-4Document4 pagesXI Chem Unit-4Prateek ChaudharyNo ratings yet
- Semiconductor Theory and Devices 11.1 - 11.2Document35 pagesSemiconductor Theory and Devices 11.1 - 11.2jamalur.lNo ratings yet
- Tutorial 12 Chemical Bond - Molecular Orbital Theory 09 May 2023Document18 pagesTutorial 12 Chemical Bond - Molecular Orbital Theory 09 May 2023SNEHANSHU BANERJEENo ratings yet
- Chapter 5 IC 2021Document87 pagesChapter 5 IC 2021김막자No ratings yet
- Mot& CFTDocument81 pagesMot& CFTBHUVAN I (RA2111003011433)No ratings yet
- PericyclicsDocument28 pagesPericyclicsEdward PittsNo ratings yet
- Chem3420 Topic3 5 New2Document28 pagesChem3420 Topic3 5 New2justinhadinata283No ratings yet
- Theory of Ultraviolet SpectrosDocument49 pagesTheory of Ultraviolet Spectrosanusha jrNo ratings yet
- Spin PolDocument24 pagesSpin PolMayank TiwariNo ratings yet
- 무기화학 1 ch2Document5 pages무기화학 1 ch2최마리아No ratings yet
- SL Chapter 2 ReviewDocument27 pagesSL Chapter 2 ReviewakikoNo ratings yet
- 8.4 Molecular Orbital TheoryDocument14 pages8.4 Molecular Orbital TheorySTRICTLY CONFIDENTIALNo ratings yet
- General Chemistry 1: Module 4, Lesson 1: Quantum Mechanical Model of An AtomDocument4 pagesGeneral Chemistry 1: Module 4, Lesson 1: Quantum Mechanical Model of An AtomKeano GelmoNo ratings yet
- The Atomic Construct A DiscussionDocument17 pagesThe Atomic Construct A DiscussionBranko R BabicNo ratings yet
- Introduction To Plasma Physics: Plasma Definition (S. Ichimaru, Statistical Plasma Physics, Vol I)Document26 pagesIntroduction To Plasma Physics: Plasma Definition (S. Ichimaru, Statistical Plasma Physics, Vol I)Noor RehmanNo ratings yet
- Electronic Structure of Solids: Chemical BondsDocument23 pagesElectronic Structure of Solids: Chemical BondsEli S. Alves Jr.No ratings yet
- Introductory Nuclear Physics - 1 The Liquid Drop ModelDocument45 pagesIntroductory Nuclear Physics - 1 The Liquid Drop ModelGourab BhattacharyaNo ratings yet
- Chemical Bonding.4Document4 pagesChemical Bonding.4VIVEK RASTOGINo ratings yet
- Electrical Transport in SolidsDocument130 pagesElectrical Transport in SolidsSaroshan DeshapriyaNo ratings yet
- Formation of Cooper Pairs in Quantum Oscillations of Electrons in PlasmaDocument8 pagesFormation of Cooper Pairs in Quantum Oscillations of Electrons in PlasmaGherghe BogdanNo ratings yet
- Nuclear Qudrupole Resonance Spectroscopy: Eq Xyzr DDocument2 pagesNuclear Qudrupole Resonance Spectroscopy: Eq Xyzr DcyrimathewNo ratings yet
- # Week 4 NotesDocument17 pages# Week 4 Notestimx123yNo ratings yet
- The Liquid Drop Model: 4.1 Some Nuclear NomenclatureDocument6 pagesThe Liquid Drop Model: 4.1 Some Nuclear NomenclatureNeeraj SinghNo ratings yet
- Struktur Atom Dan MolekulDocument35 pagesStruktur Atom Dan MolekulDeviNo ratings yet
- Simple BondingDocument41 pagesSimple BondingDeviNo ratings yet
- The Point Group of An Object or Molecule Can Be Determined by Following This Decision TreeDocument11 pagesThe Point Group of An Object or Molecule Can Be Determined by Following This Decision TreeDeviNo ratings yet
- A Matrix Is An Array of Numbers, A: Columns Column Matrix Row MatrixDocument17 pagesA Matrix Is An Array of Numbers, A: Columns Column Matrix Row MatrixDeviNo ratings yet
- Degrees of Freedom X yDocument14 pagesDegrees of Freedom X yDeviNo ratings yet
- Point Group D 2. 3. Make Reducible Reps For Outer Atoms: X y Z X y ZDocument22 pagesPoint Group D 2. 3. Make Reducible Reps For Outer Atoms: X y Z X y ZDeviNo ratings yet
- So Far We Can Say Staggered Ethane Has Three Operations: E, C, and CDocument16 pagesSo Far We Can Say Staggered Ethane Has Three Operations: E, C, and CDeviNo ratings yet
- Orbitals: 2h 2v H VDocument21 pagesOrbitals: 2h 2v H VDeviNo ratings yet
- Clean Water PollutionDocument8 pagesClean Water PollutionDeviNo ratings yet
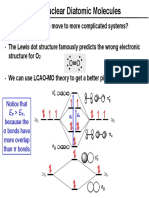
- What Happens When We Move To More Complicated Systems? Consider O - The Lewis Dot Structure Famously Predicts The Wrong Electronic Structure For O We Can Use LCAO-MO Theory To Get A Better PictureDocument11 pagesWhat Happens When We Move To More Complicated Systems? Consider O - The Lewis Dot Structure Famously Predicts The Wrong Electronic Structure For O We Can Use LCAO-MO Theory To Get A Better PictureDeviNo ratings yet
- Atkins' Physical Chemistry: Quantum Theory: Techniques and ApplicationsDocument24 pagesAtkins' Physical Chemistry: Quantum Theory: Techniques and ApplicationsDeviNo ratings yet
- Adempiere ManualDocument19 pagesAdempiere ManualMaria Yan Li100% (1)
- Electro-Thermal Analysis of Peltier Cooling Using Fem: D. Enescu, E.O. Vîrjoghe, M. Ionel, M.F. StanDocument6 pagesElectro-Thermal Analysis of Peltier Cooling Using Fem: D. Enescu, E.O. Vîrjoghe, M. Ionel, M.F. StanAziz AhmedNo ratings yet
- Harvest Moon Back To Nature Complete Guide BookDocument88 pagesHarvest Moon Back To Nature Complete Guide BookAgung Dwi PrasetiyoNo ratings yet
- Practice: ListeningDocument8 pagesPractice: ListeningChi Hoàng Minh TrươngNo ratings yet
- AndroidDocument6 pagesAndroidnikunjpatel90No ratings yet
- 9th Language Hindi 3Document168 pages9th Language Hindi 3Lokesh Pg0% (1)
- ReflectiveModelRolf 4Document2 pagesReflectiveModelRolf 4solar_powerNo ratings yet
- Harry Zaroogian, Alias, Etc. v. United States, 367 F.2d 959, 1st Cir. (1966)Document5 pagesHarry Zaroogian, Alias, Etc. v. United States, 367 F.2d 959, 1st Cir. (1966)Scribd Government DocsNo ratings yet
- Properties of Steel Fibre Reinforced ConcreteDocument8 pagesProperties of Steel Fibre Reinforced ConcreteCuriosoNo ratings yet
- Journal, Ledger Trial BalanceDocument15 pagesJournal, Ledger Trial BalanceOmar Galal100% (1)
- Mathai's "She"Document6 pagesMathai's "She"Sanjeev GoyalNo ratings yet
- Ppe3 Table Reflection and Signed OffDocument4 pagesPpe3 Table Reflection and Signed Offapi-318846856No ratings yet
- Saved QueriesDocument3 pagesSaved Queriesspace 4 meNo ratings yet
- MGT 420 Management Case Study - Motivation: Present To: Roseamilda BT MansorDocument4 pagesMGT 420 Management Case Study - Motivation: Present To: Roseamilda BT MansorkimimuraNo ratings yet
- Intelligence Studies On The ContinentDocument28 pagesIntelligence Studies On The ContinentSara Perley100% (1)
- MIS Midterm ReviewDocument38 pagesMIS Midterm ReviewAshley OjaNo ratings yet
- Fun WalkDocument14 pagesFun WalkGojo KaisenNo ratings yet
- The Forgotten Glory of Vijayanagara EmpireDocument12 pagesThe Forgotten Glory of Vijayanagara EmpireShravan Kumar CivilNo ratings yet
- Session 4 - English ViDocument11 pagesSession 4 - English ViJhefNo ratings yet
- LAC Session Program For ScienceDocument1 pageLAC Session Program For ScienceJessica Curay Salugsugan100% (1)
- CASP Case Control Drug ToxicityDocument6 pagesCASP Case Control Drug ToxicityNani Lestari Kendek AlloNo ratings yet
- Writing Headlines: (And Not Titles!)Document22 pagesWriting Headlines: (And Not Titles!)leemacaleNo ratings yet
- Tecnica de ChampyDocument7 pagesTecnica de Champyboye022694No ratings yet
- Night: by Elie WieselDocument9 pagesNight: by Elie WieselsarimkzamanNo ratings yet
- Spinster - Sylvia PlathDocument3 pagesSpinster - Sylvia PlathATHIRA CHANDRANNo ratings yet
- Order 59Document10 pagesOrder 59isharkilifatiniNo ratings yet
- Time Study EquipmentsDocument6 pagesTime Study EquipmentsGanesh ZopeNo ratings yet