Professional Documents
Culture Documents
Liquid Nitrogen in Styrofoam
Uploaded by
John Joseph0 ratings0% found this document useful (0 votes)
9 views1 pageOriginal Title
Liquid Nitrogen in styrofoam
Copyright
© © All Rights Reserved
Available Formats
ODT, PDF, TXT or read online from Scribd
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
© All Rights Reserved
Available Formats
Download as ODT, PDF, TXT or read online from Scribd
0 ratings0% found this document useful (0 votes)
9 views1 pageLiquid Nitrogen in Styrofoam
Uploaded by
John JosephCopyright:
© All Rights Reserved
Available Formats
Download as ODT, PDF, TXT or read online from Scribd
You are on page 1of 1
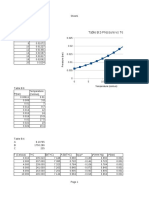
Liquid Nitrogen because of its very low boiling point ((−195.
79 °C (77 K; −320 °F) ) will boil once
it is placed in the styrofoam cup as the temperature of the room is much higher than the boiling
point of liquid Nitrogen.
The boiling of liquid Nitrogen means that liquid is being converted to gas and if this gas is confined
to a space (say a closed styrofoam container) it would explode as the gas expands. It s critical not to
have the top of the cup covered.
However if the volume of the cup is much larger than the volume of the Nitrogen, the top of the
container need not be left open.
Once all the liquid Nitrogen boils away, the Nitrogen in its gaseous state occupies a larger volume
than the Nitrogen in its liquid state.
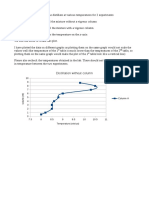
The temperature gradient : Now the Nitrogen exposed at the surface is being boiled away. Boiling
requires a heat source and the heat for the boiling is supplied from the surrounding air. As the heat is
taken away from the air, the water vapor in the air condenses and freezes around the styrofoam.
This lowers the temperature of the styrofoam at the top. This effect does not happen at the bottom
where the styrofoam is much warmer than at the top. Therefore, the temperature gradient between
the cup and the liquid is much greater at the bottom than at the top.
You might also like
- The Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeFrom EverandThe Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeRating: 4 out of 5 stars4/5 (5795)
- The Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreFrom EverandThe Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreRating: 4 out of 5 stars4/5 (1091)
- Never Split the Difference: Negotiating As If Your Life Depended On ItFrom EverandNever Split the Difference: Negotiating As If Your Life Depended On ItRating: 4.5 out of 5 stars4.5/5 (838)
- Hidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceFrom EverandHidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceRating: 4 out of 5 stars4/5 (895)
- Grit: The Power of Passion and PerseveranceFrom EverandGrit: The Power of Passion and PerseveranceRating: 4 out of 5 stars4/5 (588)
- Shoe Dog: A Memoir by the Creator of NikeFrom EverandShoe Dog: A Memoir by the Creator of NikeRating: 4.5 out of 5 stars4.5/5 (537)
- The Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersFrom EverandThe Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersRating: 4.5 out of 5 stars4.5/5 (345)
- Elon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureFrom EverandElon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureRating: 4.5 out of 5 stars4.5/5 (474)
- Her Body and Other Parties: StoriesFrom EverandHer Body and Other Parties: StoriesRating: 4 out of 5 stars4/5 (821)
- The Emperor of All Maladies: A Biography of CancerFrom EverandThe Emperor of All Maladies: A Biography of CancerRating: 4.5 out of 5 stars4.5/5 (271)
- The Sympathizer: A Novel (Pulitzer Prize for Fiction)From EverandThe Sympathizer: A Novel (Pulitzer Prize for Fiction)Rating: 4.5 out of 5 stars4.5/5 (121)
- The Little Book of Hygge: Danish Secrets to Happy LivingFrom EverandThe Little Book of Hygge: Danish Secrets to Happy LivingRating: 3.5 out of 5 stars3.5/5 (400)
- The World Is Flat 3.0: A Brief History of the Twenty-first CenturyFrom EverandThe World Is Flat 3.0: A Brief History of the Twenty-first CenturyRating: 3.5 out of 5 stars3.5/5 (2259)
- The Yellow House: A Memoir (2019 National Book Award Winner)From EverandThe Yellow House: A Memoir (2019 National Book Award Winner)Rating: 4 out of 5 stars4/5 (98)
- Devil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaFrom EverandDevil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaRating: 4.5 out of 5 stars4.5/5 (266)
- A Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryFrom EverandA Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryRating: 3.5 out of 5 stars3.5/5 (231)
- Team of Rivals: The Political Genius of Abraham LincolnFrom EverandTeam of Rivals: The Political Genius of Abraham LincolnRating: 4.5 out of 5 stars4.5/5 (234)
- On Fire: The (Burning) Case for a Green New DealFrom EverandOn Fire: The (Burning) Case for a Green New DealRating: 4 out of 5 stars4/5 (74)
- The Unwinding: An Inner History of the New AmericaFrom EverandThe Unwinding: An Inner History of the New AmericaRating: 4 out of 5 stars4/5 (45)
- ZrO2-CaO Phase DiagramDocument1 pageZrO2-CaO Phase DiagramJohn Joseph100% (2)
- Liquidus, Isothermal and Isopleth PlotsDocument1 pageLiquidus, Isothermal and Isopleth PlotsJohn JosephNo ratings yet
- Koshchmeider EquationDocument1 pageKoshchmeider EquationJohn JosephNo ratings yet
- Molar Heat Capacity of AluminumDocument1 pageMolar Heat Capacity of AluminumJohn JosephNo ratings yet
- Van't Hoff Factor Colligative PropertiesDocument3 pagesVan't Hoff Factor Colligative PropertiesJohn JosephNo ratings yet
- Synthesis of AspirinDocument2 pagesSynthesis of AspirinJohn JosephNo ratings yet
- Titration of CH3NH3+Document3 pagesTitration of CH3NH3+John JosephNo ratings yet
- Running Shoe LCADocument2 pagesRunning Shoe LCAJohn JosephNo ratings yet
- Distillation Without Column: Temperature (Celcius)Document2 pagesDistillation Without Column: Temperature (Celcius)John JosephNo ratings yet
- Ammonia Removal by Air StrippingDocument1 pageAmmonia Removal by Air StrippingJohn JosephNo ratings yet
- Antoine and Steam TablesDocument6 pagesAntoine and Steam TablesJohn JosephNo ratings yet
- Heat of Vaporization of Silica and GalliumDocument2 pagesHeat of Vaporization of Silica and GalliumJohn JosephNo ratings yet
- Drug Distribution VolumeDocument1 pageDrug Distribution VolumeJohn JosephNo ratings yet
- Specific Heat and Specific Gas ConstantDocument1 pageSpecific Heat and Specific Gas ConstantJohn JosephNo ratings yet
- Atomic Absorption SpectrometryDocument1 pageAtomic Absorption SpectrometryJohn JosephNo ratings yet
- ALEKS Assignment TimeDocument1 pageALEKS Assignment TimeJohn JosephNo ratings yet