Professional Documents
Culture Documents
Volumetric Analysis Determines HCl Concentration
Uploaded by
Amelia LlewellynOriginal Description:
Original Title
Copyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
Volumetric Analysis Determines HCl Concentration
Uploaded by
Amelia LlewellynCopyright:
Available Formats
Volumetric Analysis
Name: Amelia Llewellyn
Grade: 11/55
Date: November 9, 2021
Aim: To determine the concentration Hydrochloric acid by titration with sodium hydroxide
Theory: in this experiment you will determine the concentration of hydrochloric acid. You will
obtain a standard solution of 0.05M sodium hydroxide and then titrate it against Hydrochloric
acid using screened ethyl orange indicator. The results will then be used to determine the molar
concentration of the acid.
Apparatus: 100cm3 beakers, conical flasks, balance, 25cm3 pipette, white tile, retort stand, 50
cm3 burette, filter funnel, pipette fillers
Materials: distilled water, phenolphthalein indicator, 0.05moldm-3 NaOH, hydrochloric acid
Procedure:
1. 25cm3 of the Sodium hydroxide was added into a conical flask using a pipette.
2. A few drops of screened methyl orange indicator was added.
3. The burette was filled with HCl and the initial reading of the burette was recorded.
4 HCl was slowly added to the conical flask with the NaOH until the colour changed.
5. The final reading of the burette was recorded in a table.
6. The above steps were repeated until 2 values which are within 0.1cm3 of each other were
obtained.
7. The average volume of HCl used in the reaction was calculated.
Results
Run Rough 1 2
Final volume / cm3 37.10 38.75 37.20
Initial volume / cm3 10.65 10.35 8.80
Volume Used / cm3 26.45 28.40 28.40
Calculations:
Balanced Equation
NaOH(aq) + HCl(aq) NaCl(aq) + H2O(l)
# of Mols of NaOH in the Reaction
1 of NaOH : 1 of HCl
Mol = conc. x volume (dm3 )
= 0.05mol/dm3 x (25 / 1000)
= 0.05mol/dm3 x 0.025 dm3
= 0.00125 mol NaOH
# of Mols of HCl Reacted with the NaOH
1:1
0.00125 mol : x
X = 0.00125 mol x 1 / 1
X = 0.00125 mol
Molar conc. Of HCl
Average vol. = (28.40 + 28.40) / 2
= 28.40 dm3
Conc. = mol / vol
= 0.00125 mol / 28.40 dm3
= 4.01 mol / dm3
Sources of Error
Misjudging when the endpoint of neutralization take place. This is where you stop
before or go past the point at which a colour change happens.
Misreading the volume on the burette.
Precautions
Ensure to read the burette at eye level to get the best reading.
Ensure that after placing the evaporating dish onto the balance that you rest the
reading because the reading from the dish will affect the reading of the Sodium
Hydroxide.
Conclusion
In conclusion, the concentration of Hydrochloric acid found by titration using the Sodium
hydroxide was 4.01 mol / dm3.
You might also like
- The Sympathizer: A Novel (Pulitzer Prize for Fiction)From EverandThe Sympathizer: A Novel (Pulitzer Prize for Fiction)Rating: 4.5 out of 5 stars4.5/5 (119)
- Devil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaFrom EverandDevil in the Grove: Thurgood Marshall, the Groveland Boys, and the Dawn of a New AmericaRating: 4.5 out of 5 stars4.5/5 (266)
- The Little Book of Hygge: Danish Secrets to Happy LivingFrom EverandThe Little Book of Hygge: Danish Secrets to Happy LivingRating: 3.5 out of 5 stars3.5/5 (399)
- A Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryFrom EverandA Heartbreaking Work Of Staggering Genius: A Memoir Based on a True StoryRating: 3.5 out of 5 stars3.5/5 (231)
- Grit: The Power of Passion and PerseveranceFrom EverandGrit: The Power of Passion and PerseveranceRating: 4 out of 5 stars4/5 (587)
- Never Split the Difference: Negotiating As If Your Life Depended On ItFrom EverandNever Split the Difference: Negotiating As If Your Life Depended On ItRating: 4.5 out of 5 stars4.5/5 (838)
- The Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeFrom EverandThe Subtle Art of Not Giving a F*ck: A Counterintuitive Approach to Living a Good LifeRating: 4 out of 5 stars4/5 (5794)
- Team of Rivals: The Political Genius of Abraham LincolnFrom EverandTeam of Rivals: The Political Genius of Abraham LincolnRating: 4.5 out of 5 stars4.5/5 (234)
- Shoe Dog: A Memoir by the Creator of NikeFrom EverandShoe Dog: A Memoir by the Creator of NikeRating: 4.5 out of 5 stars4.5/5 (537)
- The World Is Flat 3.0: A Brief History of the Twenty-first CenturyFrom EverandThe World Is Flat 3.0: A Brief History of the Twenty-first CenturyRating: 3.5 out of 5 stars3.5/5 (2219)
- The Emperor of All Maladies: A Biography of CancerFrom EverandThe Emperor of All Maladies: A Biography of CancerRating: 4.5 out of 5 stars4.5/5 (271)
- The Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreFrom EverandThe Gifts of Imperfection: Let Go of Who You Think You're Supposed to Be and Embrace Who You AreRating: 4 out of 5 stars4/5 (1090)
- The Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersFrom EverandThe Hard Thing About Hard Things: Building a Business When There Are No Easy AnswersRating: 4.5 out of 5 stars4.5/5 (344)
- Her Body and Other Parties: StoriesFrom EverandHer Body and Other Parties: StoriesRating: 4 out of 5 stars4/5 (821)
- Hidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceFrom EverandHidden Figures: The American Dream and the Untold Story of the Black Women Mathematicians Who Helped Win the Space RaceRating: 4 out of 5 stars4/5 (894)
- Elon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureFrom EverandElon Musk: Tesla, SpaceX, and the Quest for a Fantastic FutureRating: 4.5 out of 5 stars4.5/5 (474)
- The Unwinding: An Inner History of the New AmericaFrom EverandThe Unwinding: An Inner History of the New AmericaRating: 4 out of 5 stars4/5 (45)
- The Yellow House: A Memoir (2019 National Book Award Winner)From EverandThe Yellow House: A Memoir (2019 National Book Award Winner)Rating: 4 out of 5 stars4/5 (98)
- My CoursesDocument18 pagesMy CoursesOsama SyedNo ratings yet
- On Fire: The (Burning) Case for a Green New DealFrom EverandOn Fire: The (Burning) Case for a Green New DealRating: 4 out of 5 stars4/5 (73)
- Julie Bui - Building Dna GizmosDocument3 pagesJulie Bui - Building Dna Gizmosapi-5226505140% (2)
- Periodic Trends GizmoDocument9 pagesPeriodic Trends GizmoGabe Itch67% (61)
- Cell SignalingDocument38 pagesCell SignalingShyrene Mamanao GumbanNo ratings yet
- Experiment 2 - Calibration of Volumetric GlasswareDocument22 pagesExperiment 2 - Calibration of Volumetric GlasswareJoemer Absalon Adorna73% (15)
- Iso 14344 2010Document9 pagesIso 14344 2010MOHAMEDNo ratings yet
- Live StreamingDocument7 pagesLive StreamingAmelia LlewellynNo ratings yet
- Factors That Impact The Mobile User ExperienceDocument4 pagesFactors That Impact The Mobile User ExperienceAmelia LlewellynNo ratings yet
- LAB #1 DiffusionDocument2 pagesLAB #1 DiffusionAmelia LlewellynNo ratings yet
- 5 Audio and Video Principles Graded Group AssignmentDocument2 pages5 Audio and Video Principles Graded Group AssignmentAmelia LlewellynNo ratings yet
- LAB #2 ChromatographyDocument3 pagesLAB #2 ChromatographyAmelia LlewellynNo ratings yet
- LAB #4 Properties of Ionic and Covalent CompoundsDocument2 pagesLAB #4 Properties of Ionic and Covalent CompoundsAmelia LlewellynNo ratings yet
- LAB #3 Trends in Group IIDocument3 pagesLAB #3 Trends in Group IIAmelia LlewellynNo ratings yet
- Biomedicine & Pharmacotherapy: Osasenaga Macdonald Ighodaro TDocument7 pagesBiomedicine & Pharmacotherapy: Osasenaga Macdonald Ighodaro TMohammed RickyNo ratings yet
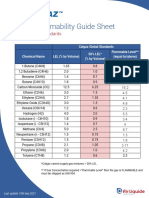
- Calgaz LEL ListDocument2 pagesCalgaz LEL ListPhan Cao An TruongNo ratings yet
- Renna2013 Carrot Jam MethodDocument8 pagesRenna2013 Carrot Jam MethodBalqish WahabNo ratings yet
- Chapter 2 Atoms Molecules and IonsDocument32 pagesChapter 2 Atoms Molecules and IonsCheska CapioNo ratings yet
- Measure Distances with Laser AccuracyDocument210 pagesMeasure Distances with Laser Accuracydan_1967No ratings yet
- Experiment No.1 TitrationDocument4 pagesExperiment No.1 Titrationoc dhogs TvNo ratings yet
- Biodegradation of Polyacrylic and Polyester PolyurethaneDocument12 pagesBiodegradation of Polyacrylic and Polyester PolyurethaneKarla ChavezNo ratings yet
- Guía de Resistencia de MaterialesDocument36 pagesGuía de Resistencia de MaterialesFelix AlorNo ratings yet
- Rewocare HV 100 TDSDocument2 pagesRewocare HV 100 TDShenriquefxs2926No ratings yet
- New Biochemical and Physicochemical Insights On A Muskmelon (Cucumis Melo (L.) ) ChitinaseDocument12 pagesNew Biochemical and Physicochemical Insights On A Muskmelon (Cucumis Melo (L.) ) ChitinaseEdnésioNo ratings yet
- 4020-GE-LST-001 - A ITY DED - Deliverable ListDocument51 pages4020-GE-LST-001 - A ITY DED - Deliverable Listthoriq ikhwanNo ratings yet
- Biotek 1Document23 pagesBiotek 1Ratna AnjarNo ratings yet
- Experiment 1 - RecoverDocument13 pagesExperiment 1 - RecoverChristina ApriliaNo ratings yet
- Chapt 4Document33 pagesChapt 4Ahmed AL-HarthiNo ratings yet
- 7697A AdvOperationDocument92 pages7697A AdvOperationEdgar Ordoñez OrjuelaNo ratings yet
- CHM 1102 Worksheet 2 2021Document2 pagesCHM 1102 Worksheet 2 2021kevin persaudNo ratings yet
- Slot 1 - Chapter 2-5 (Edit)Document51 pagesSlot 1 - Chapter 2-5 (Edit)Rabbi 08No ratings yet
- Haun SaDocument20 pagesHaun SaHerry SatriadiNo ratings yet
- LIPIDS QUIZ - Google FormsDocument25 pagesLIPIDS QUIZ - Google FormsJOHN MIKE RAMIREZNo ratings yet
- Genesis of Australian Iron Ore DepositsDocument11 pagesGenesis of Australian Iron Ore DepositszaNo ratings yet
- Gel ElectrophoresisDocument5 pagesGel ElectrophoresisROMELIE CHELSEA ESCOBIANo ratings yet
- Cdi 6 DotDocument5 pagesCdi 6 DotChella VhicNo ratings yet
- Atom Pusat - Merged PDFDocument95 pagesAtom Pusat - Merged PDFCHERLLIN EVANIANo ratings yet
- 4 6050711263309204057 PDFDocument89 pages4 6050711263309204057 PDFLakshya wardhan singh ShekhawatNo ratings yet