Professional Documents
Culture Documents
The Solubility Product of PBCL From Electrochemical Measurements
Uploaded by
Aitor PastorOriginal Description:
Original Title
Copyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
The Solubility Product of PBCL From Electrochemical Measurements
Uploaded by
Aitor PastorCopyright:
Available Formats
In the Laboratory
The Solubility Product of PbCl2 from Electrochemical W
Measurements
Jimmy S. Hwang* and Ghassan A. Oweimreen
Department of Chemistry, King Fahd University of Petroleum and Minerals, Dhahran 31261, Saudi Arabia;
*jimmy@kfupm.edu.sa
Electrochemical cell measurements have been used to 1 M KNO3 salt bridge
thermometer
determine the thermodynamic properties of chemical reac- AgAgCl electrode
AgAgCl sat KCl
tions (1, 2). The experiment presented in this article is not electrode
rubber stopper
found in physical chemistry laboratory textbooks; however, thermostated
water out
it reinforces material often discussed in lectures. The experi- internal
saturated PbCl2 beaker
ment illustrates how thermodynamic properties such as the solution
change in the standard Gibbs energy, ∆G°, and the equilib- PbCl2 crystals 1 M AgNO3
See https://pubs.acs.org/sharingguidelines for options on how to legitimately share published articles.
rium constant of a reaction are related to the emf, at different magnetic stirrer thermostated
temperatures, of electrochemical cells in which the reaction water in
takes place. The experiment is an application of a metal–
Downloaded via UNIV DE ALICANTE on October 16, 2022 at 13:40:47 (UTC).
insoluble salt electrode to determine the solubility of a spar- Figure 1. Diagram of the cell 1. Cell 2 is very similar to cell 1; the
ingly soluble salt, PbCl2. only difference is that the saturated PbCl2 is replaced by 1 m KCl.
PbCl2 Pb2+ + 2Cl−
Ksp = [Pb2+][Cl−]2
It is suited for a second- or third-year physical chemistry labo- electrode dipped in 1 m KCl. The left side electrode of cell 1
ratory. The students learn to use equipment for temperature is a Ag|AgCl(s) electrode dipped in a saturated PbCl2 solu-
control and to freshly prepare a Ag|AgCl electrode. tion. The diagram of cell 1 is shown in Figure 1.
The difference between the emf values of these cells mea-
Experiment sured at the same temperature T permits the calculation of
Ksp at T:
The following items are needed for the experiment.
∆E = E 2 − E 1 = (RT兾F )ln aCl᎑
• Two standard (sat. KCl)|AgCl(s)|Ag(s) electrodes
(Corning Glass) Ksp = a3Cl᎑兾2
• A high impedance voltmeter (Hewlett-Packard 34420
The metal–insoluble salt electrode is a second-order
Nano Volt/Micro Ohm Meter)
indicator electrode since it is used to measure the Cl −
• Magnetic stirrers activity, which is not directly involved in the electron-
• Two potassium nitrate salt bridges transfer process (3).
• An RCS Lauda Temperature Controller/Circulating
System Hazards
• Specially designed beakers with jackets for circulation
Powdered PbCl2 is harmful if swallowed; avoid breath-
of thermostated water to control the temperatures of
ing the PbCl2 dust. It is toxic if absorbed through the skin.
the solutions in the half-cells (Figure 1)
Concentrated nitric acid is toxic and may be fatal if swal-
• Silver electrode lowed or inhaled. It is extremely corrosive. Contact with skin
• Platinum electrode or eyes may cause severe burns and permanent damage.
• 1.5-V battery Hydrochloric acid (1M) is corrosive. Inhalation of vapor is
harmful and ingestion may be fatal. Liquid can cause severe
Two cells were used in the emf measurements at identi- damage to skin and eyes. Potassium nitrate is harmful if swal-
cal temperatures and with identical 1 M KNO3 salt bridges. lowed and may cause reproductive disorders.
These cells, numbered 1 and 2 respectively are:
Results
Ag(s) AgCl(s) Cl − (a = ?) (sat. KCl) AgCl(s) Ag(s)
Typical values obtained in an experimental run in the
physical chemistry laboratory are shown in Table 1. The
Ag(s) AgCl(s) Cl − (1m KCl) (sat. KCl) AgCl(s) Ag(s) variation of ln Ksp with the inverse of the temperature is pre-
sented linearly in Figure 2 and quadratically in Figure 3. The
The right side electrode is common to both cells. The left thermodynamic values calculated from the slope and inter-
side electrode in cell 2 is a standard reference Ag|AgCl(s) cept of the graphs are listed in Table 2.
JChemEd.chem.wisc.edu • Vol. 80 No. 9 September 2003 • Journal of Chemical Education 1051
In the Laboratory
Table 1. A Typical Set of Student Data for Cells 1 and 2 Discussion
T/°C Ecell 1/mV Ecell 2 /mV To our knowledge this is the first variable-temperature
20.6 134.13 64.60 experiment in which simultaneous emf measurements were
25.3 134.58 65.31 made on two cells at identical temperatures and using two
identical salt bridges. This approach eliminates the need to
29.8 136.23 66.71
correct for the temperature dependence of the two standard
34.8 137.56 68.47 reference electrodes and minimizes errors arising from junc-
39.7 139.65 70.11 tion potentials across the salt bridge.
46.0 142.26 72.73 In the last 20 years, a number of solubility product ex-
50.3 144.03 74.45 periments have been published (4–16). None deal with the
57.0 147.28 77.39
determination of the solubility products through electro-
chemical measurements. Some of the articles deal with the
60.1 148.38 78.31
effect of ionic strength on solubility, a few deal with the com-
mon ion effect, and one deals with the determination of equi-
Table 2. Thermodynamic Quantities at 25 °C librium constant of some complex silver ions.
Obtained from ln Ksp versus T ᎑1 Least-Squares Fits The experiment was easily carried out within a three-
∆H°/ ∆S°/ ∆G°/ hour lab period and the students enjoyed both the practical
Least-Squares Fits
(kJ mol᎑1) (J mol᎑1 K᎑1) (kJ mol᎑1) aspect of the experiment and its relevance to their theory class.
Linear 18.8 ᎑10.2 21.8 It is recommended to freshly prepare the salt bridge a few
days prior to the experiment.
Quadratic 21.7 ᎑0.5 21.8
W
Supplemental Material
-7.8
The background, relevant theory, and details of the ex-
perimental procedure are available in this issue of JCE On-
-8.0
line.
8.2
Acknowledgments
-8.4
ln Ksp
The authors acknowledge support for this work from the
-8.6
chemistry department at King Fahd University of Petroleum
and Minerals. The technical help of Nasrullah Baig in the
-8.8 physical chemistry laboratory is also acknowledged.
-9.0 Literature Cited
-9.2 1. Laidler, K. L.; Meiser, J. H. Physical Chemistry, 3rd ed.;
2.9 3.0 3.1 3.2 3.3 3.4 3.5
Houghton Mifflin Co.: Boston, 1999; pp 344–346.
T ⴚ1/ (10ⴚ3 Kⴚ1) 2. Alberty, R. A.; Silbey, R. J. Physical Chemistry, 1st ed.; J. Wiley
& Sons, Inc.: New York, 1992; pp 255–260.
Figure 2. A linear least-squares fit of ln Ksp versus T ᎑1. 3. Skoog, R. A.; West, D. M. Fundamentals of Analytical Chem-
istry, 3rd ed.; Holt, Rinehart and Winston: New York, 1976;
-7.8 pp 382–383.
4. Lehman, T.; Everett, W. W. J. Chem. Educ. 1982, 59, 132.
-8.0 5. Sawyer, A. K. J. Chem. Educ. 1983, 60, 416.
6. Baca, G.; Lewis, D. A. J. Chem. Educ. 1982, 60, 762–763.
-8.2 7. Edmiston, M. D.; Suter, R. W. J. Chem. Educ. 1988, 65, 278–
280.
-8.4 8. Wruck, D. B.; Rechstein J. J. Chem. Educ. 1989, 66, 515–
ln Ksp
516.
-8.6 9. Rice, G. W.; Hall, C. D. J. Chem. Educ. 1990, 67, 430–431.
10. Scaife, C. W.; Hall, C. D. J. Chem. Educ. 1990, 67, 605–606.
-8.8 11. Gotlib, L. J. J. Chem. Educ. 1990, 67, 937–938.
12. Thomsen, M. W. J. Chem. Educ. 1992, 69, 328–329.
-9.0
13. Silbermann, R. G. J. Chem. Educ. 1996, 73, 426–427.
14. Green, D. B.; Rechsteiner, G.; Honodel, A. J. Chem. Educ.
-9.2
2.9 3.0 3.1 3.2 3.3 3.4 3.5
1996, 73, 789–792.
15. Marzzacco, C. J. J. Chem. Educ. 1998, 75, 1628–1629.
T ⴚ1/ (10ⴚ3 Kⴚ1)
16. Thompson, M. L.; Kateley, L. J. J. Chem. Educ. 1999, 76,
Figure 3. A quadratic least-squares fit of ln Ksp versus T ᎑1. 95–96.
1052 Journal of Chemical Education • Vol. 80 No. 9 September 2003 • JChemEd.chem.wisc.edu
You might also like
- 1 s2.0 S0013468613011997 MainDocument7 pages1 s2.0 S0013468613011997 MainDang MinhNo ratings yet
- Chem Math MinorDocument5 pagesChem Math MinorPeter OgollaNo ratings yet
- 2009-Solmaz-NiFe para Eletrolise Alcalina Da AguaDocument9 pages2009-Solmaz-NiFe para Eletrolise Alcalina Da Aguaadalbajunior8285No ratings yet
- Ghosh 2015Document6 pagesGhosh 2015Arijit GhoshalNo ratings yet
- Carbon ClothDocument6 pagesCarbon ClothRomano AlbertNo ratings yet
- Scanlon Tafel Silver Anode Oxidrile 1968Document6 pagesScanlon Tafel Silver Anode Oxidrile 1968Jose garciaNo ratings yet
- CHEM REV UonDocument5 pagesCHEM REV UonPeter OgollaNo ratings yet
- Electrochemical and DFT Studies of - Amino-Alcohols As Corrosion Inhibitors For BrassDocument6 pagesElectrochemical and DFT Studies of - Amino-Alcohols As Corrosion Inhibitors For BrassVaishnavi SinghNo ratings yet
- ElectrochemistryDocument5 pagesElectrochemistryAnil SuryawanshiNo ratings yet
- Modified Carbon Fiber Electrodes For Carbon Dioxide ReductionDocument6 pagesModified Carbon Fiber Electrodes For Carbon Dioxide ReductionAli hassanNo ratings yet
- Air-Sensitive: Argon Techniques Manipulation ofDocument1 pageAir-Sensitive: Argon Techniques Manipulation ofAitor PastorNo ratings yet
- Analysis of The Impact of Individual Glass Constituents On Electrocatalysis On PT Electrodes in Alkaline SolutionDocument4 pagesAnalysis of The Impact of Individual Glass Constituents On Electrocatalysis On PT Electrodes in Alkaline SolutionshojihallNo ratings yet
- Molecule-Independent Electrical Switching in PT Organic Monolayer Ti DevicesDocument4 pagesMolecule-Independent Electrical Switching in PT Organic Monolayer Ti Devicesraymond wellNo ratings yet
- TMP 47 A7Document13 pagesTMP 47 A7FrontiersNo ratings yet
- Electrochemical Oxidation of Benzoic Acid at Boron-Doped Diamond ElectrodesDocument5 pagesElectrochemical Oxidation of Benzoic Acid at Boron-Doped Diamond ElectrodesSebas ValenciaNo ratings yet
- Conformal Coating of Ni (OH) 2 Nanoflakes On Carbon Fibers by Chemical Bath Deposition For Efficient Supercapacitor ElectrodesDocument7 pagesConformal Coating of Ni (OH) 2 Nanoflakes On Carbon Fibers by Chemical Bath Deposition For Efficient Supercapacitor ElectrodeshijerNo ratings yet
- Electrochemical Study and Recovery of PB Using 1 2 Choline ChlorideDocument11 pagesElectrochemical Study and Recovery of PB Using 1 2 Choline ChlorideaytajNo ratings yet
- Tang 2014Document11 pagesTang 2014Jeff DiffNo ratings yet
- Correlations Between Structural and Electrical Properties of Baceo, Studied by Coupled In-Situ Raman Scattering and Impedance SpectrosDocument10 pagesCorrelations Between Structural and Electrical Properties of Baceo, Studied by Coupled In-Situ Raman Scattering and Impedance SpectrosWendell LopesNo ratings yet
- Best Practices For Zinc Metal BatteriesDocument2 pagesBest Practices For Zinc Metal Batteriespeizx123No ratings yet
- Cambridge IGCSE Chemistry Workbook 2nd Edition PDFDocument97 pagesCambridge IGCSE Chemistry Workbook 2nd Edition PDFfran85% (13)
- بحث رقم 62Document16 pagesبحث رقم 62ahmed eldesokyNo ratings yet
- Materials Chemistry and Physics: 2 Chin-Shen Lim, K.H. Teoh, Chiam-Wen Liew, S. RameshDocument7 pagesMaterials Chemistry and Physics: 2 Chin-Shen Lim, K.H. Teoh, Chiam-Wen Liew, S. Rameshkhellouf940101No ratings yet
- Electrochemical Sensor For Amoxicillin Using Cu/Poly (Modified Carbon Paste ElectrodeDocument9 pagesElectrochemical Sensor For Amoxicillin Using Cu/Poly (Modified Carbon Paste ElectrodeQuímica InvestChimNo ratings yet
- A Computational and Experimental Study of The CyclicDocument24 pagesA Computational and Experimental Study of The Cyclicscheau-anaNo ratings yet
- Rivas Romero 2017Document5 pagesRivas Romero 2017Astuti SuwarnoNo ratings yet
- 2013 Li Pyroelectric and Electrocaloric MaterialsDocument15 pages2013 Li Pyroelectric and Electrocaloric MaterialsNacho Delgado FerreiroNo ratings yet
- J Jpcs 2004 01 010Document5 pagesJ Jpcs 2004 01 010student1No ratings yet
- The Solubility Product of PbCl2 From Electrochemical MeasurementsDocument2 pagesThe Solubility Product of PbCl2 From Electrochemical MeasurementsJosé Urpez0% (1)
- J Jelechem 2006 11 008Document7 pagesJ Jelechem 2006 11 008Mateo bolañosNo ratings yet
- Sodium Sulphate ConductivityDocument13 pagesSodium Sulphate Conductivitysandypu08No ratings yet
- Potential of A Galvanic CellDocument6 pagesPotential of A Galvanic CellRalph Andrew EsperonNo ratings yet
- Electrochemical Properties of Composite Materials Based On Platinum Modified With Molybdenum CompoundsDocument6 pagesElectrochemical Properties of Composite Materials Based On Platinum Modified With Molybdenum CompoundsFabricio CarrilloNo ratings yet
- Douiri 2016Document4 pagesDouiri 2016Vidyadhara VNo ratings yet
- Journal of Power Sources: P. Mazúr, J. Mrlík, J. Bene Š, J. Pocedi Č, J. Vrána, J. Dundálek, J. KosekDocument10 pagesJournal of Power Sources: P. Mazúr, J. Mrlík, J. Bene Š, J. Pocedi Č, J. Vrána, J. Dundálek, J. KosekSơn PhạmNo ratings yet
- 01 Coulometric Titration ExperimentDocument3 pages01 Coulometric Titration ExperimentAndres VaronNo ratings yet
- Electrochemical Activation of Graphite Felt ElectrodeDocument7 pagesElectrochemical Activation of Graphite Felt ElectrodeThuu TrangNo ratings yet
- Powder Metallurgy & Mining: Electrochemical Chelation of Heavy Metals by 2-BenzimidazoleDocument5 pagesPowder Metallurgy & Mining: Electrochemical Chelation of Heavy Metals by 2-Benzimidazolecharaf lNo ratings yet
- Buchwald 2007Document9 pagesBuchwald 2007jitha sathishNo ratings yet
- EG For EDLC PerformanceDocument9 pagesEG For EDLC PerformanceNisreen MohamedNo ratings yet
- Review On Different Components of Solid Oxide Fuel Cells 2168 9806 1000181Document4 pagesReview On Different Components of Solid Oxide Fuel Cells 2168 9806 1000181Asim IdreesNo ratings yet
- Acs Inorgchem 9b00112Document5 pagesAcs Inorgchem 9b00112pandiaraj1988No ratings yet
- Enhancement of Reversibility of CR (III) /CR (II) Redox Couple in Fe-Cr Flow Cell With Bi-Pb Electroplated Carbon FeltsDocument7 pagesEnhancement of Reversibility of CR (III) /CR (II) Redox Couple in Fe-Cr Flow Cell With Bi-Pb Electroplated Carbon FeltsInternational Organization of Scientific Research (IOSR)No ratings yet
- 1-S2.0-S1388248119301614-Main - Sergio LugoDocument5 pages1-S2.0-S1388248119301614-Main - Sergio LugoSergio LugoNo ratings yet
- Full Text 01Document9 pagesFull Text 01Tahir BeshirNo ratings yet
- Electrolysis of Mgcl2 With A Top Inserted Anode and An MG-PB CathodeDocument8 pagesElectrolysis of Mgcl2 With A Top Inserted Anode and An MG-PB CathodeFarid EluddinNo ratings yet
- Schalenbach 2016 J. Electrochem. Soc. 163 F3197Document13 pagesSchalenbach 2016 J. Electrochem. Soc. 163 F3197Avishek JaiswalNo ratings yet
- 10 1016@j Molliq 2017 11 114Document8 pages10 1016@j Molliq 2017 11 114s-hadnineNo ratings yet
- 20.1999.kinetic and Catalytic Aspects in The Hydrogen Peroxide Production Via AnthraquinoneDocument8 pages20.1999.kinetic and Catalytic Aspects in The Hydrogen Peroxide Production Via AnthraquinonesophixNo ratings yet
- Nayak 2008 JES 2008Document7 pagesNayak 2008 JES 2008Prasant NayakNo ratings yet
- Anode Polarization of Pb-Bi Alloys in The KCL-PBCL MeltDocument5 pagesAnode Polarization of Pb-Bi Alloys in The KCL-PBCL MeltIuliaHortNo ratings yet
- Electrochimica Acta: Yuanqing Zhou, Chao Chen, Jia Zhao, Junjie Fei, Yonglan Ding, Yuanli CaiDocument9 pagesElectrochimica Acta: Yuanqing Zhou, Chao Chen, Jia Zhao, Junjie Fei, Yonglan Ding, Yuanli CaiSaulo MunizNo ratings yet
- Dismantling, Recovery, and Reuse of Spent Nickel-Metal - 2006Document5 pagesDismantling, Recovery, and Reuse of Spent Nickel-Metal - 2006Daiana NavarreteNo ratings yet
- Experiment 1 FINALDocument12 pagesExperiment 1 FINALMaiSakurajimaNo ratings yet
- Delhi Public School Class 12 - Chemistry Chapter 3:electrochemistryDocument9 pagesDelhi Public School Class 12 - Chemistry Chapter 3:electrochemistryAvishi OjNo ratings yet
- Jurnal Reduksi ElektrolitDocument7 pagesJurnal Reduksi ElektrolitIqbal SaharaNo ratings yet
- Electrochemical and AFM Study of Cobalt Nucleation Mechanisms On Glassy Carbon From Ammonium Sulfate SolutionsDocument14 pagesElectrochemical and AFM Study of Cobalt Nucleation Mechanisms On Glassy Carbon From Ammonium Sulfate SolutionsReksi Bayu El HanifNo ratings yet
- Nasa Molten SaltDocument10 pagesNasa Molten SaltLuis Pedrero OjedaNo ratings yet
- Ed066p853 2Document1 pageEd066p853 2Aitor PastorNo ratings yet
- 6 CHM 5710 Using Character TablesDocument35 pages6 CHM 5710 Using Character TablesAitor PastorNo ratings yet
- Low-Noise Simplex Optimization Experiment: FutilityDocument2 pagesLow-Noise Simplex Optimization Experiment: FutilityAitor PastorNo ratings yet
- Of Chemistry: Calculation Factors UndergraduateDocument3 pagesOf Chemistry: Calculation Factors UndergraduateAitor PastorNo ratings yet
- Instrumental: Simplex OptimizationDocument4 pagesInstrumental: Simplex OptimizationAitor PastorNo ratings yet
- Numerical Evaluation of Energy Levels and Wave Functions For Hindered Internal RotationDocument8 pagesNumerical Evaluation of Energy Levels and Wave Functions For Hindered Internal RotationAitor PastorNo ratings yet
- Lattice Prediction Kapustinskii Equations and Born-Haber: Energy and Chemical The TheDocument7 pagesLattice Prediction Kapustinskii Equations and Born-Haber: Energy and Chemical The TheAitor PastorNo ratings yet
- For Conceptualization The Franck-Condon PrincipleDocument1 pageFor Conceptualization The Franck-Condon PrincipleAitor PastorNo ratings yet
- Spectroscopy,: Laser-Induced Fluorescence Dynamics, DiagnosticsDocument10 pagesSpectroscopy,: Laser-Induced Fluorescence Dynamics, DiagnosticsAitor PastorNo ratings yet
- Coordination: Complexes CobaltDocument3 pagesCoordination: Complexes CobaltAitor PastorNo ratings yet
- Franck-Condon Factors and Their Use in Undergraduate Quantum MechanicsDocument7 pagesFranck-Condon Factors and Their Use in Undergraduate Quantum MechanicsAitor PastorNo ratings yet
- Kinetics: Electrode ProcessesDocument8 pagesKinetics: Electrode ProcessesAitor PastorNo ratings yet
- Ed5b00170 Si 001Document27 pagesEd5b00170 Si 001Aitor PastorNo ratings yet
- Active: of Optically ComplexDocument2 pagesActive: of Optically ComplexAitor PastorNo ratings yet
- Electronic Tetrahedral Complexes: Nickel (LL)Document2 pagesElectronic Tetrahedral Complexes: Nickel (LL)Aitor PastorNo ratings yet
- Air-Sensitive: Argon Techniques Manipulation ofDocument1 pageAir-Sensitive: Argon Techniques Manipulation ofAitor PastorNo ratings yet
- Error Titrations Mixtures: MinimumDocument4 pagesError Titrations Mixtures: MinimumAitor PastorNo ratings yet
- Acssuschemeng 2c00095Document10 pagesAcssuschemeng 2c00095Aitor PastorNo ratings yet
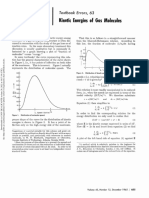
- Textbook Errors, 63 Kinetic Molecules: EnergiesDocument2 pagesTextbook Errors, 63 Kinetic Molecules: EnergiesAitor PastorNo ratings yet
- TheBaldwinRules RevisedandExtendedDocument29 pagesTheBaldwinRules RevisedandExtendedAitor PastorNo ratings yet
- Ab 036Document6 pagesAb 036Aitor PastorNo ratings yet
- Textbook Errors, 62 Difference Between and Liquids and SolidsDocument2 pagesTextbook Errors, 62 Difference Between and Liquids and SolidsAitor PastorNo ratings yet
- Acs Jchemed 5b00170Document5 pagesAcs Jchemed 5b00170Aitor PastorNo ratings yet
- Styer 2000Document7 pagesStyer 2000Aitor PastorNo ratings yet
- Claisen Rearrangement Over The Past Nine Decades: Ana M. Martı N CastroDocument64 pagesClaisen Rearrangement Over The Past Nine Decades: Ana M. Martı N CastroAitor PastorNo ratings yet
- The Multistep: Is Rate-Limiting ofDocument5 pagesThe Multistep: Is Rate-Limiting ofAitor PastorNo ratings yet
- Appendix A - Conversion From Molar To Molal: PL PLDocument57 pagesAppendix A - Conversion From Molar To Molal: PL PLAitor PastorNo ratings yet
- Acs Joc 8b00707Document14 pagesAcs Joc 8b00707Aitor PastorNo ratings yet
- Textbook: ForumDocument5 pagesTextbook: ForumAitor PastorNo ratings yet
- Microscale Preparation of Alcl3 Journal of ChemicaDocument2 pagesMicroscale Preparation of Alcl3 Journal of ChemicaAitor PastorNo ratings yet
- Lecture - Chapter7 2020 Part1Document45 pagesLecture - Chapter7 2020 Part1Qassem MohaidatNo ratings yet
- Engineering Standards Manual ISD 341-2 Chapter 13, Welding & Joining GWS 1-07 - Consumable MaterialsDocument2 pagesEngineering Standards Manual ISD 341-2 Chapter 13, Welding & Joining GWS 1-07 - Consumable MaterialsMUHAMMAD WARIS SQNo ratings yet
- Durability Test PaintDocument9 pagesDurability Test PaintMohamed NouzerNo ratings yet
- Course Code EEE-2310 EX-5Document3 pagesCourse Code EEE-2310 EX-5Fardin ChowdhuryNo ratings yet
- Precast Driven Pile 450x450-350x350Document55 pagesPrecast Driven Pile 450x450-350x350civixxNo ratings yet
- Experiment 2 Melting Point Determination: Group 6Document11 pagesExperiment 2 Melting Point Determination: Group 6Iboniks Beponpiks DabondatskiNo ratings yet
- Structural SectionsDocument45 pagesStructural SectionsAlex SinclairNo ratings yet
- ACI 523.3 (1993) GuideCellularConcretesAbove50pcf&AggConcretesAbove50pcfwithCompStrLessThan2500psiDocument16 pagesACI 523.3 (1993) GuideCellularConcretesAbove50pcf&AggConcretesAbove50pcfwithCompStrLessThan2500psiphilipyap100% (2)
- Humidity Draft PDFDocument3 pagesHumidity Draft PDFneeraj aggarwalNo ratings yet
- Experiment 4: Chemical Equilibrium: Present Well: 1 Point Clear Picture: 1 Point Total 15 PointsDocument24 pagesExperiment 4: Chemical Equilibrium: Present Well: 1 Point Clear Picture: 1 Point Total 15 PointsjulyNo ratings yet
- KleentekDocument8 pagesKleenteksurawutwijarn100% (1)
- Fatigue Analysis of Combined - Bending-Torsion - LoadingDocument10 pagesFatigue Analysis of Combined - Bending-Torsion - LoadingjcbalthazarNo ratings yet
- Auxiliary Tables: o o o o o o o o oDocument10 pagesAuxiliary Tables: o o o o o o o o oselanne67No ratings yet
- Specification: KADPOL 980 / 941 / 940Document1 pageSpecification: KADPOL 980 / 941 / 940Михаил КузьминNo ratings yet
- Nitrogen Plant Genertor CDocument8 pagesNitrogen Plant Genertor CDEVAM PATELNo ratings yet
- Guidelines For Maintenance of Stainless Steel WagonsDocument20 pagesGuidelines For Maintenance of Stainless Steel WagonsPratap Singh GangwarNo ratings yet
- 5 Year Recommended Gearbox Maintenance Service BulletinDocument1 page5 Year Recommended Gearbox Maintenance Service BulletinFaisal Ali KhanNo ratings yet
- Worksheet On Oscillations 2022 (QP) - Final Version (With Cover Page)Document45 pagesWorksheet On Oscillations 2022 (QP) - Final Version (With Cover Page)Rafsun bin HossainNo ratings yet
- CONCLUSION-food AnalysisDocument2 pagesCONCLUSION-food AnalysisFadhlin Sakinah100% (1)
- Structures Lab ManualDocument40 pagesStructures Lab ManualAakif AmeenNo ratings yet
- EXP 9 Reynold's Number Shell & Tube Heat ExchangerDocument11 pagesEXP 9 Reynold's Number Shell & Tube Heat ExchangerHanish JierdhNo ratings yet
- Experiment 4 Test For Dimensions and Tolerance of Burnt Clay BricksDocument2 pagesExperiment 4 Test For Dimensions and Tolerance of Burnt Clay BricksJigme SingyeNo ratings yet
- Acoustically Induced Vibration Mitigations in Compressor Piping SystemsDocument10 pagesAcoustically Induced Vibration Mitigations in Compressor Piping SystemsaerosanthNo ratings yet
- MAE 589 Heat Transfer Syllabus (Spring 2015)Document8 pagesMAE 589 Heat Transfer Syllabus (Spring 2015)Pratik SankheNo ratings yet
- Dynamics of System of ParticlesDocument5 pagesDynamics of System of ParticlesTrilok AkhaniNo ratings yet
- 1 SMDocument12 pages1 SMPurnama Solusi IndoNo ratings yet
- Open Channel FlowDocument62 pagesOpen Channel Flowhammadi100% (1)
- Reinforced Brick MasonryDocument21 pagesReinforced Brick MasonryMihaela Batalan100% (1)
- HP RecoupDocument5 pagesHP Recouplaouadi abdelheqNo ratings yet