Professional Documents
Culture Documents
Contaminated Blood
Uploaded by
mans sul0 ratings0% found this document useful (0 votes)
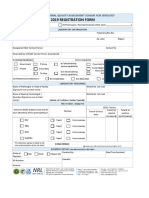
2 views1 pageThis document provides a template for a performance indicator to measure the percentage of contaminated blood culture bottles due to skin contamination. It defines the contaminated blood culture bottles as the numerator and total blood culture orders as the denominator. The target is to have less than 3% contaminated bottles, with thresholds of green less than 5%, amber between 5-8%, and red more than 8%. Data will be collected monthly from the MIS or log book by the Laboratories and Blood Banks department and reported electronically on a monthly basis.
Original Description:
Copyright
© © All Rights Reserved
Available Formats
DOCX, PDF, TXT or read online from Scribd
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentThis document provides a template for a performance indicator to measure the percentage of contaminated blood culture bottles due to skin contamination. It defines the contaminated blood culture bottles as the numerator and total blood culture orders as the denominator. The target is to have less than 3% contaminated bottles, with thresholds of green less than 5%, amber between 5-8%, and red more than 8%. Data will be collected monthly from the MIS or log book by the Laboratories and Blood Banks department and reported electronically on a monthly basis.
Copyright:
© All Rights Reserved
Available Formats
Download as DOCX, PDF, TXT or read online from Scribd
0 ratings0% found this document useful (0 votes)
2 views1 pageContaminated Blood
Uploaded by
mans sulThis document provides a template for a performance indicator to measure the percentage of contaminated blood culture bottles due to skin contamination. It defines the contaminated blood culture bottles as the numerator and total blood culture orders as the denominator. The target is to have less than 3% contaminated bottles, with thresholds of green less than 5%, amber between 5-8%, and red more than 8%. Data will be collected monthly from the MIS or log book by the Laboratories and Blood Banks department and reported electronically on a monthly basis.
Copyright:
© All Rights Reserved
Available Formats
Download as DOCX, PDF, TXT or read online from Scribd
You are on page 1of 1
General Directorate of Health Affaires Al Qassim Region
Quality & Patient Safety Administration
Performance Indicator Description Template
Deputyship/General Directorate: Department/Unit/Program:
Laboratories and Blood Banks Lab
KPI Name: CONTAMINATED BLOOD CULTURES
Rationale and Description: To assess the effectiveness of sample collection procedures and
the competence of clinical staff in proper collection site preparation by monitoring the percentage falls
positive blood culture bottles due to skin contamination.
Inclusion Exclusion Source of Data
Number of contaminated MIS or Log book
Numerator: blood culture bottles
Total number of blood culture MIS or Log book
orders
Denominator:
Formula: Output Value:
Numerator /Denominator x 100 %
Grouping: Sampling Rule:
Dept Full Volume Sampling
Indicator Level: Indicator Category:
Lab
Benchmark: Target: Less than 3
Threshold: Green: > Less than 5 Amber: 5-8 Red: < More than 8
Black:
Data Collection Responsibility: الوكالة المساعدة لشئون المختبرات و بنوك
Email: الدم
Mobile No.:
Data Collection Frequency: Monthly Reporting Frequency: Monthly
Data Collection Method: Reporting Method:
Manual System Electronic
Approved by: Approval Date:
You might also like
- Wwiy4300 PDFDocument3 pagesWwiy4300 PDFRani reddy45% (44)
- TyphoidDocument3 pagesTyphoidShivam Tomar83% (6)
- Xdue6378 2Document2 pagesXdue6378 2S Abedi50% (2)
- Wwiy4300 PDFDocument6 pagesWwiy4300 PDFvenkat ramanaNo ratings yet
- Rejected SamplesDocument1 pageRejected Samplesmans sulNo ratings yet
- Performance Indicator Description TemplateDocument1 pagePerformance Indicator Description Templatemans sulNo ratings yet
- TAT TherapeuticDocument1 pageTAT Therapeuticmans sulNo ratings yet
- Availability of Blood and Blood ProductsDocument2 pagesAvailability of Blood and Blood ProductsLovelydePerioNo ratings yet
- Wwiy4300 PDFDocument6 pagesWwiy4300 PDFShobhit MishraNo ratings yet
- Body Fluid Cell Counts by Automated MethodsDocument11 pagesBody Fluid Cell Counts by Automated MethodsntnquynhproNo ratings yet
- 1-1-5 (2) Blood Safety (Mar 08)Document4 pages1-1-5 (2) Blood Safety (Mar 08)umahsuthanNo ratings yet
- 11 Feb Denise PHD Final With Corrections For PrintingDocument272 pages11 Feb Denise PHD Final With Corrections For PrintingAbeer AbdullahNo ratings yet
- MANI Quality Control in Hematology AnalysersDocument65 pagesMANI Quality Control in Hematology Analyserscandiddreams100% (1)
- EQAS 2019 Form - New PDFDocument2 pagesEQAS 2019 Form - New PDFSharon ChuaNo ratings yet
- Eqas 2019 Form - NewDocument2 pagesEqas 2019 Form - NewAlfreah Jean Tambasen100% (3)
- Obeid Specialized Hospital - Riyadh: Quality Indicator Form 2018 2019Document2 pagesObeid Specialized Hospital - Riyadh: Quality Indicator Form 2018 2019LovelydePerioNo ratings yet
- Availability of Emergency MedicationDocument2 pagesAvailability of Emergency MedicationLovelydePerioNo ratings yet
- CoA For Source PlasmaDocument2 pagesCoA For Source PlasmaDavidNo ratings yet
- 型号未知 2019年在韩销售血球厂家对比Document8 pages型号未知 2019年在韩销售血球厂家对比郑伟健No ratings yet
- CBC Autoverification Paper Draft V 102Document22 pagesCBC Autoverification Paper Draft V 102Anj RodriguezNo ratings yet
- Bias in Error Estimation When Using Cross-Validation For Model Selection." BMC Bioinformatics, 7 (1), 91Document9 pagesBias in Error Estimation When Using Cross-Validation For Model Selection." BMC Bioinformatics, 7 (1), 91sterepavelNo ratings yet
- NACO - NARI GLP Training WorkshopDocument21 pagesNACO - NARI GLP Training WorkshopDinesh SreedharanNo ratings yet
- Semen Analysis Test Report: Patient Information Facility Information Contact InformationDocument6 pagesSemen Analysis Test Report: Patient Information Facility Information Contact Informationsoyuzmedimaging and diagnostics ltdNo ratings yet
- FrmPatientViewAllReport - 2023-04-06T211159.013 PDFDocument9 pagesFrmPatientViewAllReport - 2023-04-06T211159.013 PDFamir.khann4411No ratings yet
- Quality Control 2023 +CLIA 2024Document125 pagesQuality Control 2023 +CLIA 2024bashira khalidyNo ratings yet
- Electronic CrossmatchDocument4 pagesElectronic CrossmatchMano cempakaNo ratings yet
- N Tran Interferences in POC Devices 100918 PDFDocument118 pagesN Tran Interferences in POC Devices 100918 PDFsiscaNo ratings yet
- 179-Article Text-723-2-10-20200606Document7 pages179-Article Text-723-2-10-20200606Laboratorium RS BELLANo ratings yet
- Drug Discovery: Echo Liquid Handlers Enable 3456-Well AssaysDocument12 pagesDrug Discovery: Echo Liquid Handlers Enable 3456-Well Assaysschumi252No ratings yet
- BD FACSCount BrochureDocument12 pagesBD FACSCount BrochureneysaonlineNo ratings yet
- AT - Chapter 10-Notes - Part 2Document3 pagesAT - Chapter 10-Notes - Part 2hoxhiiNo ratings yet
- Lab Dept: Hematology Test Name: Reticulocyte Count: General InformationDocument2 pagesLab Dept: Hematology Test Name: Reticulocyte Count: General InformationTanveerNo ratings yet
- Lmy 030Document7 pagesLmy 030Tuan NguyenNo ratings yet
- Includes The Analysis Of: Water and Waste Water AnalysisDocument214 pagesIncludes The Analysis Of: Water and Waste Water AnalysisHasun MalingaNo ratings yet
- Whitepaper QAQCEnvironmentalLabDocument5 pagesWhitepaper QAQCEnvironmentalLabbudi_alamsyahNo ratings yet
- Obeid Specialized Hospital - Riyadh: Quality Indicator Form 2018 2019Document2 pagesObeid Specialized Hospital - Riyadh: Quality Indicator Form 2018 2019LovelydePerioNo ratings yet
- Introduction To Franchisee Model: Step Towards A Prosperous PartnershipDocument27 pagesIntroduction To Franchisee Model: Step Towards A Prosperous PartnershipNaomi SargeantNo ratings yet
- Three Years' Experience of Quality Monitoring Program On Pre-Analytical Errors in ChinaDocument7 pagesThree Years' Experience of Quality Monitoring Program On Pre-Analytical Errors in ChinaCenyiqanita NurqanitaNo ratings yet
- AEB14 SM CH17 v2Document31 pagesAEB14 SM CH17 v2RonLiu350% (1)
- Lab Report NewDocument12 pagesLab Report Newoneoceannetwork3No ratings yet
- Interpretation: No. Investigation Observed Value Unit Biological Reference IntervalDocument2 pagesInterpretation: No. Investigation Observed Value Unit Biological Reference IntervalAbeer AliNo ratings yet
- Molecular Laboratory Report and CertificateDocument1 pageMolecular Laboratory Report and Certificateجوهوي ريسNo ratings yet
- Interpretation: No. Investigation Observed Value Unit Biological Reference IntervalDocument2 pagesInterpretation: No. Investigation Observed Value Unit Biological Reference IntervalAbeer AliNo ratings yet
- Solution Manual Auditing and Assurance Services 13e by Arens Chapter 17Document32 pagesSolution Manual Auditing and Assurance Services 13e by Arens Chapter 17vindhy 201512006No ratings yet
- LQMS-VL Specimen: ManagementDocument24 pagesLQMS-VL Specimen: ManagementagumasNo ratings yet
- Desalegn Et Al. - 2018 - Misdiagnosis of Pulmonary Tuberculosis and Associated Factors in Peripheral Laboratories A Retrospective StudyDocument7 pagesDesalegn Et Al. - 2018 - Misdiagnosis of Pulmonary Tuberculosis and Associated Factors in Peripheral Laboratories A Retrospective StudyRania MaalejNo ratings yet
- 2021 Errors Human Factor CHARLESRIVER - LGDocument2 pages2021 Errors Human Factor CHARLESRIVER - LGAdriana VillaverdeNo ratings yet
- BD FACSCount BrochureDocument12 pagesBD FACSCount BrochureAhmed Ghassany100% (1)
- Diacetyl and Acetyl Propionyl Testing On Suicide Bunny (Sucker Punch and Mother's Milk)Document9 pagesDiacetyl and Acetyl Propionyl Testing On Suicide Bunny (Sucker Punch and Mother's Milk)Russ Wishtart50% (2)
- Blood Bank ProceduresDocument102 pagesBlood Bank Proceduresahalimyousef70% (10)
- 5 Ways To Reduce Legionnaires' Disease RiskDocument3 pages5 Ways To Reduce Legionnaires' Disease Riskn_ashok689039No ratings yet
- PMLS Last Lesson and Current LessDocument8 pagesPMLS Last Lesson and Current LessGNo ratings yet
- Dilip Shah 70895932024 02 21 10 31 30 165 6 0 1 114 133529798901344240Document3 pagesDilip Shah 70895932024 02 21 10 31 30 165 6 0 1 114 133529798901344240kurmabhavinNo ratings yet
- Dini 5Document10 pagesDini 5Cenyiqanita NurqanitaNo ratings yet
- Intra Operative Aspiration PneumoniaDocument2 pagesIntra Operative Aspiration PneumoniaLovelydePerioNo ratings yet
- TRRCDocument9 pagesTRRCmans sulNo ratings yet
- Rejected Samples Sheet Critical Value Sheet: Hospital, City Laboratory and Blood Bank Department FormsDocument1 pageRejected Samples Sheet Critical Value Sheet: Hospital, City Laboratory and Blood Bank Department Formsmans sulNo ratings yet
- UntitledDocument1 pageUntitledmans sulNo ratings yet
- Equipment Name: Manufacturer: Model: S/N: SupplierDocument1 pageEquipment Name: Manufacturer: Model: S/N: Suppliermans sulNo ratings yet
- UntitledDocument1 pageUntitledmans sulNo ratings yet
- 138 - Accepting Suboptimal SpecimensDocument1 page138 - Accepting Suboptimal Specimensmans sulNo ratings yet
- 30 - Cleaning SheetDocument1 page30 - Cleaning Sheetmans sulNo ratings yet
- Unit . Computer No: Month: ..Document1 pageUnit . Computer No: Month: ..mans sulNo ratings yet
- 32 - Tracing MaterialDocument1 page32 - Tracing Materialmans sulNo ratings yet