Professional Documents
Culture Documents
Summary
Uploaded by
amina tahir amina tahir0 ratings0% found this document useful (0 votes)
5 views2 pagesThe document discusses the quantum numbers that describe electrons in atoms. It explains that each electron is defined by four quantum numbers - the principal quantum number (n) describing the electron shell or energy level, the azimuthal quantum number (l) describing the subshell and orbital angular momentum, the magnetic quantum number (m) describing orbital orientation, and the spin quantum number (s) describing the spin of the electron. These quantum numbers completely specify the characteristics of each electron in an atom.
Original Description:
Summary of an article
Original Title
summary
Copyright
© © All Rights Reserved
Available Formats
DOCX, PDF, TXT or read online from Scribd
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentThe document discusses the quantum numbers that describe electrons in atoms. It explains that each electron is defined by four quantum numbers - the principal quantum number (n) describing the electron shell or energy level, the azimuthal quantum number (l) describing the subshell and orbital angular momentum, the magnetic quantum number (m) describing orbital orientation, and the spin quantum number (s) describing the spin of the electron. These quantum numbers completely specify the characteristics of each electron in an atom.
Copyright:
© All Rights Reserved
Available Formats
Download as DOCX, PDF, TXT or read online from Scribd
0 ratings0% found this document useful (0 votes)
5 views2 pagesSummary
Uploaded by
amina tahir amina tahirThe document discusses the quantum numbers that describe electrons in atoms. It explains that each electron is defined by four quantum numbers - the principal quantum number (n) describing the electron shell or energy level, the azimuthal quantum number (l) describing the subshell and orbital angular momentum, the magnetic quantum number (m) describing orbital orientation, and the spin quantum number (s) describing the spin of the electron. These quantum numbers completely specify the characteristics of each electron in an atom.
Copyright:
© All Rights Reserved
Available Formats
Download as DOCX, PDF, TXT or read online from Scribd
You are on page 1of 2
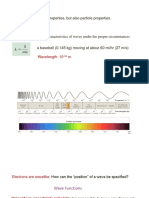
The research article paper about Quantum Chemistry basically
describes about the common terminology like electrons used
for Quantum numbersi.e; Principle or Azimuthal Quantum
Numbers then here comes some background concept about
their angular momentum.
Each orbital in an atom is specified by set of three quantum
no(n,l,m,)and each electron is desingnated by a set of 4
quantum no(n,l,m and s)they discribe electron in atom
completly. Principal quantum describes the electron shell,or
energy level of an electron the value range start from 1 shell to
shell contaning the outer most electron of an atom.Azimuthal
Quantum describe about the subshell and give the megnitude
of orbital angular momentum value range 0 TO n-
1&l=0,1,2,3.......n-1And shape of orbital also given by Azimuthal
quantum no .Megnatic quantum gives information about the
orientation of orbital in space also called orbital orientation
quantum no.it has value from depend upon value of "L".Spin
Quantum discribe the spin of electron in an atom. it is denoted
by s" its electron within each orbital give projection of spin
quantum .Electron also revolve about its axis called self
rotation .S quantum no of electron may be +1/2,-1/2.
Elementry partical contain many quantums but usually typical
quantum related to spin or typical internal symmetrics are
lepton baryon and electric charge.
You might also like
- The Mysterious World of Fundamental Particles: Cosmic BeginningsFrom EverandThe Mysterious World of Fundamental Particles: Cosmic BeginningsRating: 4 out of 5 stars4/5 (2)
- Elementary Particles: The Commonwealth and International LibraryFrom EverandElementary Particles: The Commonwealth and International LibraryNo ratings yet
- Exercises No. 2Document6 pagesExercises No. 2John Miguel BarbaNo ratings yet
- Quantum NumbersDocument31 pagesQuantum NumbersDianne de la TorreNo ratings yet
- Screenshot - 2023-01-07-23-13-07-99 - (7 Files Merged)Document7 pagesScreenshot - 2023-01-07-23-13-07-99 - (7 Files Merged)Prashant ChaureNo ratings yet
- Atomic Orbitals: Quantum NumbersDocument16 pagesAtomic Orbitals: Quantum NumberslostgirlNo ratings yet
- GenChem Reviewer Q2Document3 pagesGenChem Reviewer Q2Khent Calvin SanchezNo ratings yet
- Quantum Numbers: Learning Objective Key PointsDocument3 pagesQuantum Numbers: Learning Objective Key PointsCandyAnonymousNo ratings yet
- Quantum Numbers and ECDocument3 pagesQuantum Numbers and ECShraddha Deshmukh-KelkarNo ratings yet
- Hydrogen Spectrum and Atomic Terms NotesDocument32 pagesHydrogen Spectrum and Atomic Terms Notesoyamo markNo ratings yet
- q2 General Chemistry Rev (Complete)Document14 pagesq2 General Chemistry Rev (Complete)rosedancel52No ratings yet
- Electronic Structure of The Atom: Charlito R. AligadoDocument32 pagesElectronic Structure of The Atom: Charlito R. AligadoElaine Mata100% (1)
- General Chemistry: Use Quantum Numbers To Describe An Electron in An AtomDocument16 pagesGeneral Chemistry: Use Quantum Numbers To Describe An Electron in An AtomDenver John Caloza LamarcaNo ratings yet
- Quantum Numbers NotesDocument8 pagesQuantum Numbers NotesMamidanna SashankNo ratings yet
- Quantum NumbersDocument3 pagesQuantum NumbersMia Pearl Tabios ValenzuelaNo ratings yet
- Applied Chemistry Lecture 2Document62 pagesApplied Chemistry Lecture 2Muhammad RafayNo ratings yet
- Quantum Numbers: Introduction To ChemistryDocument24 pagesQuantum Numbers: Introduction To Chemistryhimanshu yadavNo ratings yet
- Atomic StructureDocument7 pagesAtomic Structuregovindkrishnatv2020No ratings yet
- CHEM 2101 Lecture 2 (Qunatum Number)Document5 pagesCHEM 2101 Lecture 2 (Qunatum Number)Asif UddinNo ratings yet
- Max Plank:: Learning Objectives For This ChapterDocument4 pagesMax Plank:: Learning Objectives For This Chapterdomer2011No ratings yet
- Atomic Structure RewindDocument86 pagesAtomic Structure RewindRudransh JNo ratings yet
- Quantum Number and Electronic ConfigurationDocument18 pagesQuantum Number and Electronic ConfigurationNoor Mohammad NofaerNo ratings yet
- Quantum Numbers and Their Physical InterpetationDocument15 pagesQuantum Numbers and Their Physical InterpetationMomina LaraibNo ratings yet
- Principal Quantum NumberDocument11 pagesPrincipal Quantum NumberPraveen KumarNo ratings yet
- Energy StateDocument30 pagesEnergy StateTahirullah KhanNo ratings yet
- Quantum Theory and Atomic StructureDocument42 pagesQuantum Theory and Atomic StructureMBalbuena, Daryll A.No ratings yet
- 1A The Shapes and Structures of Molecules Student Handout Part Two 2022.23Document70 pages1A The Shapes and Structures of Molecules Student Handout Part Two 2022.23Music MaestroNo ratings yet
- Quantum Numbers and Periodic Trends: by Sarah C. Petitto Using Materials From Jack F. MckennaDocument14 pagesQuantum Numbers and Periodic Trends: by Sarah C. Petitto Using Materials From Jack F. MckennaMuhammad HarisNo ratings yet
- Atoms NotesDocument6 pagesAtoms NotesLonely SoulNo ratings yet
- AU Chemistry Unit-1Document26 pagesAU Chemistry Unit-1Aarush PitlaNo ratings yet
- Electronic Strucure of AtomsDocument26 pagesElectronic Strucure of AtomsSAKE TvNo ratings yet
- Quantum Number: How Many Quantum Numbers?Document6 pagesQuantum Number: How Many Quantum Numbers?kakulitan93No ratings yet
- General Chemistry 1: Quarter 2 - Week 1Document13 pagesGeneral Chemistry 1: Quarter 2 - Week 1Janzelle BorbonNo ratings yet
- De Broglie Relation: EinsteinDocument19 pagesDe Broglie Relation: EinsteinMaisha MaimunaNo ratings yet
- 1 Quantum Numbers-1Document34 pages1 Quantum Numbers-1Carlo James SablanNo ratings yet
- Electronic Structure of MatterDocument3 pagesElectronic Structure of Matterilikegay2dmensNo ratings yet
- Chapter 3 - Electronic Structure of Elements - RozainaDocument75 pagesChapter 3 - Electronic Structure of Elements - RozainaEzzarenNo ratings yet
- Quantum Numbers: Name Anuj Class 11Document5 pagesQuantum Numbers: Name Anuj Class 11Anuj MorNo ratings yet
- Atomic Structure... (3) PPTDocument32 pagesAtomic Structure... (3) PPTVaibhav KargetiNo ratings yet
- Quantum Theory and The Electronic Structure of AtomsDocument17 pagesQuantum Theory and The Electronic Structure of AtomsSalama NaumanNo ratings yet
- Structure of AtomDocument112 pagesStructure of AtomAnanya SinghNo ratings yet
- BiophysicsDocument80 pagesBiophysicsBibo BobiNo ratings yet
- Gen-Chem-1 Handout Q2 Week1 PDFDocument4 pagesGen-Chem-1 Handout Q2 Week1 PDFFrancisco de GuzmanNo ratings yet
- Che 126 Complete SlideDocument658 pagesChe 126 Complete Slideabuabdmuqseet2001No ratings yet
- tilde : (N,, M) Define An Orbital (N,, M, M) Define An Electron in An Determined OrbitalDocument1 pagetilde : (N,, M) Define An Orbital (N,, M, M) Define An Electron in An Determined OrbitalRichardA.TorresNo ratings yet
- Quantum Mechanical Description of The AtomDocument3 pagesQuantum Mechanical Description of The AtomShiro ChanNo ratings yet
- The Quantum NumbersDocument2 pagesThe Quantum NumbersArun Kumar SrivastavaNo ratings yet
- The Quantum Mechanical Model of The AtomDocument13 pagesThe Quantum Mechanical Model of The AtomnehaNo ratings yet
- 1.1.1 Quantum Numbers, Atomic Orbitals and Electron ConfiguartionDocument6 pages1.1.1 Quantum Numbers, Atomic Orbitals and Electron Configuartionmy3gr8kidoosNo ratings yet
- Atomic Structure - 3Document27 pagesAtomic Structure - 3suhiermai3No ratings yet
- Quantum Mechanical ModelDocument26 pagesQuantum Mechanical ModelMaye AporadorNo ratings yet
- Chapter 6 NewDocument59 pagesChapter 6 NewhoiminhNo ratings yet
- Gen Chem 1 - Q2Document55 pagesGen Chem 1 - Q2pingcj9No ratings yet
- Chemistry Notes Part 1Document59 pagesChemistry Notes Part 1SharleneNo ratings yet
- DME I Year CHEDocument91 pagesDME I Year CHERadha KrishnaNo ratings yet
- Quantum Numbers GENCHEM1 Q2Document2 pagesQuantum Numbers GENCHEM1 Q2Glean EsmileNo ratings yet
- Lecture 2 EGM 241 - @2022 EditedDocument41 pagesLecture 2 EGM 241 - @2022 EditedIwell PhiriNo ratings yet
- Atomic OrbitalDocument25 pagesAtomic OrbitalCarolle PerezNo ratings yet
- Fundamentals of Energy Dispersive X-Ray Analysis: Butterworths Monographs in MaterialsFrom EverandFundamentals of Energy Dispersive X-Ray Analysis: Butterworths Monographs in MaterialsRating: 5 out of 5 stars5/5 (1)