Professional Documents
Culture Documents
Notes C12 121
Uploaded by
DonnavilleOriginal Title
Copyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
Notes C12 121
Uploaded by
DonnavilleCopyright:
Available Formats
Chapter 12.
Saturated Hydrocarbons
12.1 Organic and Inorganic Compounds
Organic vs. Inorganic
• Organic chemistry: Study of hydrocarbons (only carbon and hydrogen atoms) and their
various derivatives.
– Examples: natural gas, petroleum, plastics, rubbers, paper, carbohydrates (sugar,
starch), proteins, enzymes, fatty acids, food stuff, drugs, textiles, etc.
• Inorganic chemistry: Study of all substances other than hydrocarbons and their
derivatives.
– Examples of inorganic compounds: sulfuric acid, nitric acid, ores and minerals,
air, baking powder, caustic soda, table salt, metal alloys (brass, bronze), etc.
12.2 Bonding Characteristics of the Carbon Atom
• C-atom always makes total 4 Bonds
• The sharing of four valance electrons requires the formation of four covalent bonds
which are represented by four lines as shown here.
12.3 Hydrocarbons and Hydrocarbon Derivatives
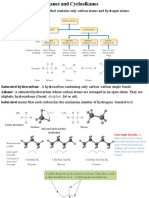
• Hydrocarbon: Compound that contains only carbon and hydrogen atoms.
• Hydrocarbon derivative: Compound that contains carbon and hydrogen and one or
more additional elements.
• Saturated hydrocarbon: Hydrocarbon with all carbon–carbon bonds are single bonds.
• Unsaturated hydrocarbon: Hydrocarbon with one or more carbon–carbon multiple
bonds (double bonds, triple bonds, or both).
12.4 Alkanes: Acyclic Saturated Hydrocarbons
• Alkanes: Open chain (straight or branched) binary compounds of carbon and hydrogen.
• All bonds are single bonds.
• General formula: CnH2n+2 .
12.5 Structural Formulas
Expanded Structural Formula: A two-dimensional structural representation that depicts
the bonding of all atoms in a molecule.
Condensed Structural Formula: Structural arrangements of different groupings (a
central atom connected with other atoms) in a molecule.
Skeletal Structural Formula: Structural arrangement of all bonded carbon atoms
without showing the attached hydrogen atoms.
•
12.6 Alkane Isomerism
Structural (Constitutional) Isomerism
Compounds having same molecular formula, but different structural arrangements
(different connectivity of atoms) are called Isomers, and the phenomenon is known as
isomerism.
Space-filing models for the three isomeric C5H12 alkanes:
Number of Isomers Possible for Alkanes of Various Carbon Chain Lengths
12.7 Conformations of Alkanes
Conformation: At a given instant, the specific three dimensional spatial arrangement of
atoms due to the rotation of C-C single bond in the same molecule.
Different conformational structures result from C-C single bond rotation, but no breaking of
bonds takes place.
Different structural isomers result from different connectivity of atoms, which requires
breaking and making of bonds in the same molecule.
12.8 IUPAC Nomenclature of Alkanes
Rules for Naming Stright-Chain Alkanes
Systematic approach for naming alkanes was introduced by the International Union of Pure
and Applied Chemistry (IUPAC), and the rules are being updated periodically.
The rules are applied to both types of alkanes:
– Straight chain (continuous) alkanes
– Branched chain (divided / substituted) alkanes
– Substituent: an atom or group of atoms attached to a chain (or ring) of carbon
atoms.
– Alkyl group: the group of atoms that would be obtained by removing a hydrogen
atom from an alkane.
Rules for Naming Branched-Chain Alkanes
Rule 1: Identify the longest continuous carbon chain and name that chain as the parent
Rule 2: Number the carbon atoms in the parent chain in such a way so that to give the
lowest possible number for a substituent (alkyl group).
Rule 3: If only one substituent present, name and locate it by number; then, prefix the
number and name to that of the parent carbon chain.
Rule 4: If two or more of the same kind of alkyl group are present in a molecule, indicate
the number with a Greek numerical prefix
(di-, tri-, tetra-, penta-, and so forth). In addition, a number specifying the location of each
identical group must be included. These position numbers, separated by commas, precede
the numerical prefix. Numbers are separated from words by hyphens.
Rule 5: When two kinds of alkyl groups are present on the same carbon chain, number each
group separately, and list the names of the alkyl groups in alphabetical order.
Rule 6: Follow IUPAC punctuation rules:
1. Separate numbers from each other by commas.
2. Separate numbers from letters by hyphens.
3. Do not add a hyphen or a space between the last-named substituent and the
name of the parent alkane.
Example
Answer:
a. 3,6-Dimethyloctane
b. 3,4,4,5-Tetramethyloctane
12.9 Line-Angle Structural Formulas for Alkanes
Line-angle (Bond-Angle) structural formula:
– A structural representation in which a line represents a carbon–carbon bond.
– A carbon atom is understood to be present at every point where two lines meet (
at the intersection).
– At the ends of each lines.
Example:
Structures of branched-chain alkanes designated using line-angle structural formulas.
Example: The five constitutional isomers of C6H14.
12.10 Classification of Carbon Atoms
Based on the skeletal structure of the C-atom in a hydrocarbon chain, each C-atom can
be classified as follows:
– Primary: If bonded with only one C- atom
– Secondary: If bonded with other 2 C- atoms
– Tertiary: If bonded with other 3 C-atoms
– Quaternary: If bonded with other 4 C- atoms
12.11 Branched-Chain Alkyl Groups
Continuous chain alkyl groups: The simplest type of alkyl group with continuous chain of
carbon atoms.
Branched-chain alkyl groups: Alkyl groups are added to the continuous parent chain of
carbon atoms (Alkenes with branched-chain alkyl groups).
12.12 Cycloalkanes
Cycloalkanes are alkanes that contain a ring of three or more carbons.
General Formula of Cycloalkane: CnH2n
12.13 IUPAC Nomenclature for Cycloalkanes
• Count the number of carbons in the ring, and name the parent cyclic chain (just like an
alkane).
• Add the prefix cyclo to the IUPAC name of the unbranched alkane, e.g., Cyclopentane
• Name any alkyl groups on the ring in the usual way
• Numbering conventions used in locating substituents on the ring:
1. If there is just one ring substituent, it is not necessary to locate it by number.
2. When two ring substituents are present, the carbon atoms in the ring are
numbered beginning with the substituent of higher alphabetical priority and
proceeding in the direction (clockwise or counterclockwise) that gives the other
substituent the lower number.
When three or more ring substituents are present, ring numbering begins at the
substituent that leads to the lowest set of location numbers. When two or more
equivalent numbering sets exist, alphabetical priority among substituents determines
the set used
.
3-Ethyl-1,1-dimethylcyclohexane
Example
Answers:
a. Methylcyclopropane
b. 1-Ethyl-4-methylcyclohexane
c. 4-Ethyl-1,2 dimethylcyclopentane
12.14 Isomerism in Cycloalkanes
Cycloalkanes also exhibit stereoisomerism.
– Stereoisomers have the same molecular and structural formulas but different
orientations of atoms in space.
– Several forms of stereoisomerism exist.
Di-substituted Cycloalkanes exhibit Cis-Trans isomerism.
– Cis–trans isomers have the same molecular and structural formulas but different
spatial orientations of atoms due to the restricted rotation about bonds.
Cis- is a prefix that means “on the same side.” In cis-1,2-dimethylcyclopentane, the two
methyl groups are on the same side of the ring.
Trans- is a prefix that means “across from.” In trans-1,2-dimethylcyclopentane, the two
methyl groups are on opposite sides of the ring.
12.15 Sources of Alkanes and Cycloalkanes
Natural Gas and Petroleum
Natural Gas:
– 50 - 90% Methane
– 1 - 10% Ethane
– Up to 8% Higher Alkanes (mostly Propane and Butanes)
Petroleum (crude oil):
– Complex mixture of acyclic and cyclic hydrocarbons
–
Petroleum Refining
Refining: Isolation of soil, sand, sulfur, etc., from the crude oil and physical separation of the
hydrocarbons mixture into several fractions.
Fractional Distillation: Process of separating different components from a mixture based
on their boiling points.
12.16 Physical Properties of Alkanes and Cycloalkanes
Solubility and Density
Solubility: Follows the rule ‘like dissolves like’
– Soluble in non-polar solvents e.g., in hexanes
– Insoluble in polar solvents e.g., water, alcohol
– Application: protective wax coating on fruits to prevent loss of water as well as
from fungi or bacterial strain etc.
Density: Lighter than water
– Densities of alkanes and cycloalkanes range from
– 0.6 – 0.8 g/mL (water density at 4 degree C = 1.0 g/mL)
– Practical illustration: quick spreading of oil spills in water.
– Fire caused by floating oil can not be extinguished by water.
Boiling Points
The boiling points increase with an increase in the number of carbon atom in chain length
or ring size.
Branching in carbon chain lowers the boiling point of an alkane.
Cycloalkanes have higher boiling points than their noncyclic counterparts with the same
number of c-atoms
12.17 Chemical Properties of Alkanes and Cycloalkanes
Vigorous reaction with oxygen (usually from air)
Accompanied by the evolution of heat and light
Always an exothermic process
The final products of any complete combustion of hydrocarbons are:
– Carbon dioxide and Water (vapor)
– CH4 + 2O2 CO2 + 2H2O + Energy
– 2C6H14 + 19O2 12CO2 + 14H2O + Energy
– Incomplete / partial combustion results in a mixture of carbon dioxide and
carbon monoxide with water (g)
Halogenation
Direct reaction with halogens (chlorine, bromine, iodine)
Substitution of one or more H-atoms of hydrocarbons by halogen atoms (substitution
reaction of alkanes)
12.18 Nomenclature and Properties of Halogenated Alkanes
Halogenated Alkanes: Derivatives alkanes in which one or more hydrogen atoms are
substituted by halogen atoms.
Halogenated cycloalkanes: Cycloalkane derivatives in which one or more hydrogen
atoms are substituted by halogen atoms
These derivatives are named as:
– Haloalkanes or Halocycloalkanes (IUPAC naming)
– Alkyl Halides or Cycloalkyl halides (common naming)
IUPAC Rules
Follow the same rules for naming alkanes / cycloalkanes
Lowest locant number is given to the top priority group.
Alkyl and halogen substituents have the same ranking.
If 2 or more different substituents are present, the alphabetical order determines their
priority.
Physical Properties of halogenated alkanes
Density: Many halogen derivatives are heavier than water.
Polarity: They are mostly polar compounds (if unsymmetrical).
Boiling Points: Generally higher than parent hydrocarbons.
You might also like
- Organic Chemistry Study Guide: Key Concepts, Problems, and SolutionsFrom EverandOrganic Chemistry Study Guide: Key Concepts, Problems, and SolutionsRating: 3.5 out of 5 stars3.5/5 (10)
- Introduction To Organic ChemistryDocument142 pagesIntroduction To Organic ChemistryHafiz Hamidi100% (1)
- Schaum's Easy Outline of Organic Chemistry, Second EditionFrom EverandSchaum's Easy Outline of Organic Chemistry, Second EditionRating: 3.5 out of 5 stars3.5/5 (2)
- Organic 2 TwoDocument28 pagesOrganic 2 TwoNamwangala Rashid NatinduNo ratings yet
- Alkanes and CycloalkanesDocument49 pagesAlkanes and Cycloalkanesaron supanNo ratings yet
- McMurry9e PPT CH03Document48 pagesMcMurry9e PPT CH03김가영No ratings yet
- Organic-Chemistry-and-Alkanes(1)Document49 pagesOrganic-Chemistry-and-Alkanes(1)joyanne180No ratings yet
- 4 Allkanes StructureDocument22 pages4 Allkanes StructureJames ClementsNo ratings yet
- CHEM7-Organic ChemistryDocument30 pagesCHEM7-Organic ChemistryEmily SalibNo ratings yet
- Saturated HydrocarbonDocument60 pagesSaturated HydrocarbonMarvin K. Candor100% (1)
- Plugin-495014180310kuliah Ke 2 Alkana SikloalkanaDocument59 pagesPlugin-495014180310kuliah Ke 2 Alkana SikloalkanaYunitaNo ratings yet
- Alkanes and Cycloalkanes - PPTX 2Document57 pagesAlkanes and Cycloalkanes - PPTX 2eysabelagwenNo ratings yet
- P4 AlkanesDocument71 pagesP4 AlkanesHazell SangalangNo ratings yet
- Hydrocarbons: ObjectivesDocument29 pagesHydrocarbons: Objectivesjohn ryan piolNo ratings yet
- UntitledDocument46 pagesUntitled양우경No ratings yet
- Organic Compounds: Alkanes and Their Stereochemistry: Based On Mcmurry'S Organic Chemistry, 7 EditionDocument29 pagesOrganic Compounds: Alkanes and Their Stereochemistry: Based On Mcmurry'S Organic Chemistry, 7 EditionLois EuniceNo ratings yet
- CHE 112 - Lecture 2Document103 pagesCHE 112 - Lecture 2Martias WambiNo ratings yet
- Chapter 2 AlkanesDocument61 pagesChapter 2 AlkanesKonoli NuingNo ratings yet
- Stereo ChemistryDocument27 pagesStereo ChemistryAtul KambleNo ratings yet
- 2019 c3.5 Organic ChemistryDocument197 pages2019 c3.5 Organic Chemistryhydesh100% (1)
- Organic Chemistry HydrocarbonsDocument12 pagesOrganic Chemistry HydrocarbonsAlshaimaa SolimanNo ratings yet
- Alkane and AlkylDocument12 pagesAlkane and AlkylHanna GalatiNo ratings yet
- Naming Organic CompoundsDocument22 pagesNaming Organic CompoundsNovira ChandisaNo ratings yet
- Chapter 2 Alkanes and CycloalkanesDocument62 pagesChapter 2 Alkanes and CycloalkanesJitherDoromalDelaCruzNo ratings yet
- Lecture Outline: - Nomenclature - ConformationDocument65 pagesLecture Outline: - Nomenclature - Conformation박승민No ratings yet
- Leaning Packet 6 Engg Chem 1Document42 pagesLeaning Packet 6 Engg Chem 1Ritchel Conde BoholNo ratings yet
- 10 Alkanes NomenclatureDocument29 pages10 Alkanes Nomenclaturesomayya waliNo ratings yet
- Orgchem Lec3 FinalDocument42 pagesOrgchem Lec3 FinalSummer Eisheriel JamoraNo ratings yet
- Chapter 2Document48 pagesChapter 2민규강No ratings yet
- Organic Chemistry Compound NamingDocument35 pagesOrganic Chemistry Compound NamingRENE N. RAMILONo ratings yet
- Lecture 3 Theory 2Document12 pagesLecture 3 Theory 2Noor FarhanNo ratings yet
- 2,5,8-trimethyl-octaneDocument42 pages2,5,8-trimethyl-octaneMa Faustina GeronaNo ratings yet
- CYCLOALKANESDocument5 pagesCYCLOALKANESRosalynda Talosig MabbunNo ratings yet
- Chapter04 Smith3e PPTDocument71 pagesChapter04 Smith3e PPTLuisa Palomo100% (1)
- Chem 3 - Alkanes PDFDocument79 pagesChem 3 - Alkanes PDFFeaid Aina OrnedoNo ratings yet
- Alkene: University of Zakho College of Basic Education General Science Department 2 Stage, Class ADocument14 pagesAlkene: University of Zakho College of Basic Education General Science Department 2 Stage, Class AasaNo ratings yet
- Families of Organic CompoundsDocument8 pagesFamilies of Organic CompoundsJessa Mae LangcuyanNo ratings yet
- Functional Group NamesDocument21 pagesFunctional Group NamesAdine RaissaNo ratings yet
- Chem1280 Topic 03Document37 pagesChem1280 Topic 03setrcicuscienceNo ratings yet
- AlkenesAlkynes-nomenclature-and-properties-for - College-Bio StudentsDocument23 pagesAlkenesAlkynes-nomenclature-and-properties-for - College-Bio Studentshixevab276No ratings yet
- Organic Chemistry BKF1323: 2.1 Alkanes and CycloalkanesDocument47 pagesOrganic Chemistry BKF1323: 2.1 Alkanes and Cycloalkanes0JTINGNo ratings yet
- Chapter 2 Alkanes - 2Document49 pagesChapter 2 Alkanes - 2farah amaniNo ratings yet
- Hydrocarbons Class 11 Notes Chemistry Chapter 13 - Learn CBSEDocument16 pagesHydrocarbons Class 11 Notes Chemistry Chapter 13 - Learn CBSERishabh Singh RajputNo ratings yet
- 1.2 Nomenclature of HydrocarbonsDocument40 pages1.2 Nomenclature of HydrocarbonsElina NaberezhnovaNo ratings yet
- AlkanesDocument42 pagesAlkanesShahina ShayneNo ratings yet
- Functional Groups and Alkanes FinalDocument9 pagesFunctional Groups and Alkanes FinalJoecel May Gabino HalipanNo ratings yet
- Hydrocarbon and PetroleumDocument38 pagesHydrocarbon and PetroleumVincentius Matthew YonathanNo ratings yet
- Nomenclature of Alkane,Alkene & AlkyneDocument19 pagesNomenclature of Alkane,Alkene & Alkynea6446674No ratings yet
- Alkanes and Cycloalkanes: Structures and NomenclatureDocument37 pagesAlkanes and Cycloalkanes: Structures and Nomenclaturemohammad 3yaNo ratings yet
- CA Lesson 2 AlkanesDocument31 pagesCA Lesson 2 AlkanesAlbaraaAliNo ratings yet
- Alkanes and Cycloalkanes: Hybridized and Tetrahedral, With All Bond Angles of 109.5°Document5 pagesAlkanes and Cycloalkanes: Hybridized and Tetrahedral, With All Bond Angles of 109.5°Alecs Elinor AmoguisNo ratings yet
- Hydrocarbons NotesDocument15 pagesHydrocarbons Notesarjunrkumar2024No ratings yet
- CAPE CHEMISTRY CRASH COURSEDocument75 pagesCAPE CHEMISTRY CRASH COURSEKewi Love100% (3)
- Organic Chemistry - Basic Principles and TechniquesDocument34 pagesOrganic Chemistry - Basic Principles and Techniquesjkc collegeNo ratings yet
- Alkana-1Document61 pagesAlkana-1ayundhaNo ratings yet
- Cape Chemistry Unit 2 ReviewDocument7 pagesCape Chemistry Unit 2 Reviewkayanna100% (1)
- ALKANES2Document41 pagesALKANES2Shiki Asagami BrunestedNo ratings yet
- Families of Organic CompoundsDocument13 pagesFamilies of Organic CompoundsFabe Maria Feb VeranoNo ratings yet
- Organic Compounds EditedDocument52 pagesOrganic Compounds EditedbrendonNo ratings yet
- ND Veterinary Laboratory TechnologyDocument158 pagesND Veterinary Laboratory TechnologyTeklewNo ratings yet
- Ch8 SQDocument16 pagesCh8 SQhihiNo ratings yet
- Ionic Bonding Lab ActivityDocument3 pagesIonic Bonding Lab ActivityMBOTAKE LawsonNo ratings yet
- ChemistryDocument4 pagesChemistryMalik Ameer Hamza BalochNo ratings yet
- Year 12 A Level Chemistry Mock Exam June 2022: Please Write Clearly in Block CapitalsDocument28 pagesYear 12 A Level Chemistry Mock Exam June 2022: Please Write Clearly in Block CapitalsJaweria bibiNo ratings yet
- CY 1001 - Organic Chemistry: Anbarasan CY1001 Anbarasan CY1001Document21 pagesCY 1001 - Organic Chemistry: Anbarasan CY1001 Anbarasan CY1001Rashmi AgrawalNo ratings yet
- Biology 100 Exam 1 Study GuideDocument5 pagesBiology 100 Exam 1 Study GuideAllison ANo ratings yet
- Chemistry MCQs For Class 11 Test 2Document5 pagesChemistry MCQs For Class 11 Test 2Mubashar Rehmat AnsariNo ratings yet
- Chemistry of Solid State (AutoRecovered)Document53 pagesChemistry of Solid State (AutoRecovered)amehnrNo ratings yet
- Resonance in FormamideDocument7 pagesResonance in FormamideRodrigo YepsenNo ratings yet
- Basic Chemistry 5th Edition Timberlake Test BankDocument28 pagesBasic Chemistry 5th Edition Timberlake Test Banksamuelfintanult1jj100% (30)
- King of Zion School Science Exam Covers Atomic ModelsDocument5 pagesKing of Zion School Science Exam Covers Atomic ModelsRamil Jayme Damiles0% (1)
- Solomons Testbank1 Struktur Bindn + SvarDocument7 pagesSolomons Testbank1 Struktur Bindn + SvarTahirat NasiruNo ratings yet
- Molecular Orbital Theory: Inorganic ChemistryDocument15 pagesMolecular Orbital Theory: Inorganic ChemistryGokul GanesanNo ratings yet
- Cell Physics 1Document24 pagesCell Physics 1Ahmad Al-ThunebatNo ratings yet
- Behavior of The EBehavior of The Electronic Dielectric Constant in Covalent and Ionic Materialslectronic Dielectric Constant in Covalent and Ionic MaterialsDocument14 pagesBehavior of The EBehavior of The Electronic Dielectric Constant in Covalent and Ionic Materialslectronic Dielectric Constant in Covalent and Ionic MaterialsHoang Van DungNo ratings yet
- 4.5 Metallic BondDocument12 pages4.5 Metallic BondALIS SUHAIRIN BT ABD GHANI BMNo ratings yet
- PMC Practice Test 01 2022Document11 pagesPMC Practice Test 01 2022Farhan GhanghroNo ratings yet
- A-Level-Chemistry Edexcel FACER Sample-Chapter PDFDocument36 pagesA-Level-Chemistry Edexcel FACER Sample-Chapter PDFahamedNo ratings yet
- 5 Ionic, Covalent, Metallic Bonding 10th GradeDocument9 pages5 Ionic, Covalent, Metallic Bonding 10th GradeMBOTAKE LawsonNo ratings yet
- Biomedical - Engineering 2011 12 PDFDocument86 pagesBiomedical - Engineering 2011 12 PDFNashitah AlwazNo ratings yet
- Physical Science 4Document5 pagesPhysical Science 4Morelei FernandezNo ratings yet
- The Reality of ConsciousnessDocument3 pagesThe Reality of ConsciousnesstimtoyNo ratings yet
- Lasgen Chemistry 2 Q3 W2Document3 pagesLasgen Chemistry 2 Q3 W2Franzhean Balais CuachonNo ratings yet
- Lecture NotesDocument3 pagesLecture NotesIdk UlitNo ratings yet
- Answers To E3 PDFDocument5 pagesAnswers To E3 PDFJowel MercadoNo ratings yet
- Chemistry ? ? ?? ?? ? Support MaterialDocument400 pagesChemistry ? ? ?? ?? ? Support MaterialAdityaNo ratings yet
- Brain Test System: For Solution Purchase Brain Question Bank 042-37113344Document4 pagesBrain Test System: For Solution Purchase Brain Question Bank 042-37113344Mohammad AshfaqNo ratings yet
- Scientiæ Baccalaureus: A Physics Research TextDocument258 pagesScientiæ Baccalaureus: A Physics Research TextPerry Lee GordonNo ratings yet
- Mid-Semester Examination: (March 2012 Session)Document12 pagesMid-Semester Examination: (March 2012 Session)diraazlanNo ratings yet
- Meltdown: Nuclear disaster and the human cost of going criticalFrom EverandMeltdown: Nuclear disaster and the human cost of going criticalRating: 5 out of 5 stars5/5 (5)
- Is That a Fact?: Frauds, Quacks, and the Real Science of Everyday LifeFrom EverandIs That a Fact?: Frauds, Quacks, and the Real Science of Everyday LifeRating: 4.5 out of 5 stars4.5/5 (3)
- Periodic Tales: A Cultural History of the Elements, from Arsenic to ZincFrom EverandPeriodic Tales: A Cultural History of the Elements, from Arsenic to ZincRating: 3.5 out of 5 stars3.5/5 (137)
- Organic Chemistry for Schools: Advanced Level and Senior High SchoolFrom EverandOrganic Chemistry for Schools: Advanced Level and Senior High SchoolNo ratings yet
- Essential Oil Chemistry Formulating Essential Oil Blends that Heal - Aldehyde - Ketone - Lactone: Healing with Essential OilFrom EverandEssential Oil Chemistry Formulating Essential Oil Blends that Heal - Aldehyde - Ketone - Lactone: Healing with Essential OilRating: 5 out of 5 stars5/5 (1)
- An Introduction to the Periodic Table of Elements : Chemistry Textbook Grade 8 | Children's Chemistry BooksFrom EverandAn Introduction to the Periodic Table of Elements : Chemistry Textbook Grade 8 | Children's Chemistry BooksRating: 5 out of 5 stars5/5 (1)
- The Disappearing Spoon: And Other True Tales of Madness, Love, and the History of the World from the Periodic Table of the ElementsFrom EverandThe Disappearing Spoon: And Other True Tales of Madness, Love, and the History of the World from the Periodic Table of the ElementsRating: 4 out of 5 stars4/5 (146)
- Coating and Drying Defects: Troubleshooting Operating ProblemsFrom EverandCoating and Drying Defects: Troubleshooting Operating ProblemsRating: 5 out of 5 stars5/5 (1)
- Chemistry at Home - A Collection of Experiments and Formulas for the Chemistry EnthusiastFrom EverandChemistry at Home - A Collection of Experiments and Formulas for the Chemistry EnthusiastNo ratings yet
- The Periodic Table of Elements - Post-Transition Metals, Metalloids and Nonmetals | Children's Chemistry BookFrom EverandThe Periodic Table of Elements - Post-Transition Metals, Metalloids and Nonmetals | Children's Chemistry BookNo ratings yet
- Guidelines for Asset Integrity ManagementFrom EverandGuidelines for Asset Integrity ManagementRating: 5 out of 5 stars5/5 (1)
- Science Goes Viral: Captivating Accounts of Science in Everyday LifeFrom EverandScience Goes Viral: Captivating Accounts of Science in Everyday LifeRating: 5 out of 5 stars5/5 (1)
- Chemistry: a QuickStudy Laminated Reference GuideFrom EverandChemistry: a QuickStudy Laminated Reference GuideRating: 5 out of 5 stars5/5 (1)
- The Periodic Table: A Very Short IntroductionFrom EverandThe Periodic Table: A Very Short IntroductionRating: 4.5 out of 5 stars4.5/5 (3)
- The Regenerative Grower's Guide to Garden Amendments: Using Locally Sourced Materials to Make Mineral and Biological Extracts and FermentsFrom EverandThe Regenerative Grower's Guide to Garden Amendments: Using Locally Sourced Materials to Make Mineral and Biological Extracts and FermentsRating: 5 out of 5 stars5/5 (3)
- The Elements We Live By: How Iron Helps Us Breathe, Potassium Lets Us See, and Other Surprising Superpowers of the Periodic TableFrom EverandThe Elements We Live By: How Iron Helps Us Breathe, Potassium Lets Us See, and Other Surprising Superpowers of the Periodic TableRating: 3.5 out of 5 stars3.5/5 (22)
- Stuff Matters: Exploring the Marvelous Materials That Shape Our Man-Made WorldFrom EverandStuff Matters: Exploring the Marvelous Materials That Shape Our Man-Made WorldRating: 4 out of 5 stars4/5 (289)
- Chemistry for Breakfast: The Amazing Science of Everyday LifeFrom EverandChemistry for Breakfast: The Amazing Science of Everyday LifeRating: 4.5 out of 5 stars4.5/5 (14)
- Gas-Liquid And Liquid-Liquid SeparatorsFrom EverandGas-Liquid And Liquid-Liquid SeparatorsRating: 3.5 out of 5 stars3.5/5 (3)
- Monkeys, Myths, and Molecules: Separating Fact from Fiction in the Science of Everyday LifeFrom EverandMonkeys, Myths, and Molecules: Separating Fact from Fiction in the Science of Everyday LifeRating: 4 out of 5 stars4/5 (9)
- Monkeys, Myths, and Molecules: Separating Fact from Fiction, and the Science of Everyday LifeFrom EverandMonkeys, Myths, and Molecules: Separating Fact from Fiction, and the Science of Everyday LifeRating: 4 out of 5 stars4/5 (1)
- Chemical Elements Pocket Guide: Detailed Summary of the Periodic TableFrom EverandChemical Elements Pocket Guide: Detailed Summary of the Periodic TableNo ratings yet