Professional Documents
Culture Documents
AA2-G8 - TrendsofPeriodic Table
Uploaded by
Mokshitha Reddy YanamalOriginal Description:
Original Title
Copyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
AA2-G8 - TrendsofPeriodic Table
Uploaded by
Mokshitha Reddy YanamalCopyright:
Available Formats
NPS International School
Periodic Table - Grade 8 Chemistry Investigation
Title of the investigation: An Investigation on periodic trends of elements.
What is the investigation about?
Arrangement of elements in the modern periodic table is based on the Periodic Law. The
Periodic Law states that the physical and chemical properties of the elements recur in a
systematic and predictable way when the elements are arranged in order of increasing atomic
number. The research aspect of this investigation is to explore trend in physical property of
elements in a group with change in atomic radius.
Research
Select any two physical properties of the elements in a group and investigate how these
properties are affected by atomic/ionic radius. In your research, your consideration should
cover but not limited to the following:-
Research Question:
How does physical properties of elements (melting point, boiling point, density etc.)
in a group affected by change in atomic radii/ionic radii?
Hypothesis:
Variables:
What is your Controlled variable?
What is your Dependent variable?
What is your Independent variable?
Data collection:
Use interactive periodic table or other similar secondary sources for data collection.
For eg: Periodic Table – Royal Society of Chemistry (rsc.org)
Tabulate your data collected here:
Data analysis:
Using appropriate axes, plot a graph of the data collected:-
Observations:
List your observations and suggest scientific reason for each of your observations.
Conclusion:
Refer to your hypothesis and answer the research question.
Extension:
Repeat the investigation for elements belonging to a period and compare the results.
NPSI/ Version1.0/22-23/Chem./8/AA2 Page No:1
NPS International School
Rubrics
Proficient Basic Novice
Hypothesis clear and Hypothesis is not clear but Hypothesis is neither
focussed on the topic have some relevance to the written clear nor relevant to
topic the topic.
Hypothesis
(2 marks) Hypothesis does not make
links to two variables.
(2 mark) (1 mark) (0 mark)
All variables listed Some variables listed are No variables listed are
are linked to the linked to the hypothesis. linked to the hypothesis.
Variables
hypothesis.
(3 marks)
(3 marks) (2-1 marks) (0 mark)
.
Data collected allows Some data collected allows Data collected is not
the hypothesis to be the hypothesis to be relevant to hypothesis and
confirmed or confirmed or rejected. not tabulated.
rejected.
No data is collected.
Data collected is Some data collected is
Data Collection
tabulated and linked tabulated.
(5 marks)
to the variables.
.
References are made References are made (1-0 mark)
for atomic radii
(3-2 marks)
(5-4 marks)
A graph is correctly A graph is incorrectly plotted A graph is incorrectly
plotted with correct with axes chosen that links to plotted with wrong axes or
Data Analysis axes chosen that the variables. no graph is plotted
(5 marks) links to the variables.
(3-2 marks)
(5-4 marks) (1-0 marks)
Observation, Experimental Some experimental Experimental observations
Conclusion, & observations are observations are correctly are not correctly inferred.
Extension correctly inferred inferred which allows the
(5 marks) which allows the hypothesis to be confirmed Conclusion made is not
hypothesis to be or rejected. scientifically correct.
confirmed or
rejected. Conclusion made is No extension of learning is
scientifically correct. demonstrated.
Conclusion made is
scientifically correct. Extension of learning is (1-0 marks)
demonstrated.
Extension of learning
which allows the (3-2 marks)
same hypothesis to
be tested.
(5-4 marks)
NPSI/ Version1.0/22-23/Chem./8/AA2 Page No:2
NPS International School
NPSI/ Version1.0/22-23/Chem./8/AA2 Page No:3
You might also like
- Quantitative Method-Breviary - SPSS: A problem-oriented reference for market researchersFrom EverandQuantitative Method-Breviary - SPSS: A problem-oriented reference for market researchersNo ratings yet
- Nam Il KIM - EDITED Blank BC Template (Aug 2022)Document12 pagesNam Il KIM - EDITED Blank BC Template (Aug 2022)Nam Kim “Namil plaz” KimNo ratings yet
- Biology Chapter 1 Surface Tension LabDocument4 pagesBiology Chapter 1 Surface Tension LabjnfurstNo ratings yet
- Terminal Output. StatsdocxDocument2 pagesTerminal Output. StatsdocxGabs CastilloNo ratings yet
- Assn2-Hmef 5093quantitative Research MethodologyDocument8 pagesAssn2-Hmef 5093quantitative Research MethodologyivanNo ratings yet
- CNUR 860 - FALL - 2020 Stats 2 - InstructionDocument7 pagesCNUR 860 - FALL - 2020 Stats 2 - Instructionzobia100% (1)
- Correlational ResearchDocument34 pagesCorrelational ResearchTừ Quỳnh NhưNo ratings yet
- 1.2 MYP Lab Report Template NEW OCT 2018Document6 pages1.2 MYP Lab Report Template NEW OCT 2018stparth.taNo ratings yet
- Penelitian TrisaktiDocument51 pagesPenelitian TrisaktiWenny HuNo ratings yet
- Researchmethodslecture 2015Document25 pagesResearchmethodslecture 2015api-300762638No ratings yet
- Physics Labs 7-9Document2 pagesPhysics Labs 7-9ANGEL GUZMAN HERNANDEZNo ratings yet
- Lab Report RubricDocument2 pagesLab Report RubricjacnauNo ratings yet
- The Scientific Laboratory Experiment Rubric: CategoryDocument1 pageThe Scientific Laboratory Experiment Rubric: CategoryLeonard Joseph RuizNo ratings yet
- Science Lab Format and Rubric 2019Document2 pagesScience Lab Format and Rubric 2019api-396909076No ratings yet
- RUBRIC Internal FacultyDocument3 pagesRUBRIC Internal FacultyArpita SenNo ratings yet
- PSYC - 1001 - Chapter 2Document5 pagesPSYC - 1001 - Chapter 2Alaina PalmaNo ratings yet
- Penelitian Hypothetico Deductive MethodDocument56 pagesPenelitian Hypothetico Deductive Methodlorensa kristinaNo ratings yet
- Goodson, Buhi & Dunsmore 2002 - 2003Document4 pagesGoodson, Buhi & Dunsmore 2002 - 2003why daNo ratings yet
- SPSS Def + Job DescriptionDocument54 pagesSPSS Def + Job DescriptionmamtaNo ratings yet
- Department of Education: Republic of The PhilippinesDocument9 pagesDepartment of Education: Republic of The PhilippinesPrincess Loraine DuyagNo ratings yet
- Grade 8 Math Silly PuttyDocument4 pagesGrade 8 Math Silly Puttyrurbanik100% (2)
- Formal Lab Report RubricDocument2 pagesFormal Lab Report RubricBrian Andrew SmithNo ratings yet
- IA Introduction 2 - Snell's LawDocument11 pagesIA Introduction 2 - Snell's LawPhương Anh Cao HoàngNo ratings yet
- chi-SquaredTest - Vishal (21DM217) - Vatsal (21DM216) - Preeti (21DM242) - Absent On4th&11th July.Document17 pageschi-SquaredTest - Vishal (21DM217) - Vatsal (21DM216) - Preeti (21DM242) - Absent On4th&11th July.HITESH SHARMA-DM 21DM078No ratings yet
- J Gray Test Blueprint Assignment RevDocument3 pagesJ Gray Test Blueprint Assignment Revapi-528983071No ratings yet
- Scientific Method Fall 2021Document4 pagesScientific Method Fall 2021Angel D'AndriaNo ratings yet
- Psychological Assessment Module 2Document30 pagesPsychological Assessment Module 2Judyangaangan03No ratings yet
- Animal+Behavior+Pre LabDocument2 pagesAnimal+Behavior+Pre LabodomeishomeNo ratings yet
- Rubrics For Lab Exercise 2023 StickersDocument1 pageRubrics For Lab Exercise 2023 StickersJared Clive Palo AlvarezNo ratings yet
- FINAL (SG) - PR 2 11 - 12 - UNIT 7 - LESSON 2 - Testing The Difference of Two MeansDocument27 pagesFINAL (SG) - PR 2 11 - 12 - UNIT 7 - LESSON 2 - Testing The Difference of Two MeansCamille Darcy AguilarNo ratings yet
- 1204 23S Assignment#2-HypTestingDocument2 pages1204 23S Assignment#2-HypTestingmea.uk151No ratings yet
- RubricDocument1 pageRubricapi-450605415No ratings yet
- Exploratory Factor Analysis (Efa)Document56 pagesExploratory Factor Analysis (Efa)Madhavii PandyaNo ratings yet
- Assessment Criteria: Cambridge Lower SecondaryDocument3 pagesAssessment Criteria: Cambridge Lower Secondarygugel cromNo ratings yet
- Identifying The Context: Your Sources in MLA Format in Your Bibliography at The End of The DocumentDocument11 pagesIdentifying The Context: Your Sources in MLA Format in Your Bibliography at The End of The DocumentFiza MahtaniNo ratings yet
- Research Iii (Introduction-Week 1) : Prepared By: Mia M. BidesDocument30 pagesResearch Iii (Introduction-Week 1) : Prepared By: Mia M. BidesMiKouShi -chanNo ratings yet
- 1906 EM202322SHA527HUM 18907EDN Assessment 2 Planning Document RubricDocument3 pages1906 EM202322SHA527HUM 18907EDN Assessment 2 Planning Document RubricAryjeet K.No ratings yet
- Science Inquiry RubricDocument1 pageScience Inquiry RubricPasheena RamathasNo ratings yet
- PDF DocumentDocument2 pagesPDF DocumentMiguel BarbosaNo ratings yet
- Practical StatisticsDocument26 pagesPractical Statisticssunilverma2010No ratings yet
- Research IssuesDocument41 pagesResearch IssuesRyma UzairNo ratings yet
- Independent VariableDocument6 pagesIndependent VariableChinara SafarliNo ratings yet
- Lab Experiment Name: - : RubricDocument1 pageLab Experiment Name: - : RubricChristiCamelNo ratings yet
- Master (Without Thesis) Process Safety and Loss Prevention - Rubrics For Research ProposalDocument3 pagesMaster (Without Thesis) Process Safety and Loss Prevention - Rubrics For Research ProposalM HNo ratings yet
- Introduction To Meta Analysis SDGDocument72 pagesIntroduction To Meta Analysis SDGBes GaoNo ratings yet
- Reliability and ValidityDocument29 pagesReliability and ValidityR-wah LarounetteNo ratings yet
- CHEM2A - Midterm Examination (Test Draft)Document3 pagesCHEM2A - Midterm Examination (Test Draft)Christian DeanNo ratings yet
- 01 Scientific Method-StoesselDocument26 pages01 Scientific Method-StoesselMDolo GSNo ratings yet
- Factor AnalysisDocument56 pagesFactor Analysissanzit0% (1)
- Chapter 5 Data AnalysisRevisedDocument33 pagesChapter 5 Data AnalysisRevisedBantamkak FikaduNo ratings yet
- Rubrics For A Scientific Investigation ProjectDocument4 pagesRubrics For A Scientific Investigation ProjectAlyssa Mae DapadapNo ratings yet
- Rubric Crit C Enthalpy of Combustion of FuelsDocument2 pagesRubric Crit C Enthalpy of Combustion of FuelsShreyaNo ratings yet
- TA T3 PCS Social Problem Within School Society 23-24Document3 pagesTA T3 PCS Social Problem Within School Society 23-24davidx17011No ratings yet
- Principles Project Introduction To BiomechanicsDocument3 pagesPrinciples Project Introduction To BiomechanicsBhavesh HotwaniNo ratings yet
- Statistics in Neurosciences: Basics - Field Chapter 1-5Document33 pagesStatistics in Neurosciences: Basics - Field Chapter 1-5lisamartinova747No ratings yet
- Final Exam (Eda Vinluan, Jericho)Document6 pagesFinal Exam (Eda Vinluan, Jericho)jek vinNo ratings yet
- Raws q4 Homework FileDocument13 pagesRaws q4 Homework Filennczr2vm55No ratings yet
- ENG40001 Final Year Research Project 1 Semester 2, 2020: RubricsDocument8 pagesENG40001 Final Year Research Project 1 Semester 2, 2020: RubricsAS V KameshNo ratings yet
- 05 - Statistical Processing and Analysis of Medical DataDocument14 pages05 - Statistical Processing and Analysis of Medical DataHarshit SodhiNo ratings yet
- Examiners ' Comments and Key Points: FEEDBACK To Candidates June 2015 Part A ExaminationDocument31 pagesExaminers ' Comments and Key Points: FEEDBACK To Candidates June 2015 Part A ExaminationsubiovefrigidoNo ratings yet
- National Poetry MonthDocument4 pagesNational Poetry MonthMokshitha Reddy YanamalNo ratings yet
- Housing in SingaporeDocument4 pagesHousing in SingaporeMokshitha Reddy YanamalNo ratings yet
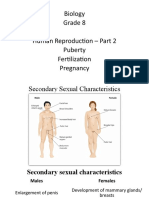
- Biology Grade 8 - Human Reproduction 2Document13 pagesBiology Grade 8 - Human Reproduction 2Mokshitha Reddy YanamalNo ratings yet
- Black History Mont1Document4 pagesBlack History Mont1Mokshitha Reddy YanamalNo ratings yet
- Biology Grade 8 - Variation & InheritanceDocument27 pagesBiology Grade 8 - Variation & InheritanceMokshitha Reddy YanamalNo ratings yet
- ReflectionDocument2 pagesReflectionMokshitha Reddy YanamalNo ratings yet
- Physics BibliographyDocument1 pagePhysics BibliographyMokshitha Reddy YanamalNo ratings yet
- Asynchronous Learning G8Document2 pagesAsynchronous Learning G8Mokshitha Reddy YanamalNo ratings yet
- Biology Grade 8 - ExcretionDocument16 pagesBiology Grade 8 - ExcretionMokshitha Reddy YanamalNo ratings yet
- Rodrigo 5Document12 pagesRodrigo 5Mokshitha Reddy YanamalNo ratings yet
- Agatha ChristieDocument3 pagesAgatha ChristieMokshitha Reddy YanamalNo ratings yet
- History BookletDocument2 pagesHistory BookletMokshitha Reddy YanamalNo ratings yet
- Investigation On Periodic Trends of ElementsDocument3 pagesInvestigation On Periodic Trends of ElementsMokshitha Reddy YanamalNo ratings yet
- Renewable and Non-Renewable ResourcesDocument40 pagesRenewable and Non-Renewable ResourcesMokshitha Reddy YanamalNo ratings yet
- Weather and Climate SummaryDocument20 pagesWeather and Climate SummaryMokshitha Reddy YanamalNo ratings yet
- PsheDocument3 pagesPsheMokshitha Reddy YanamalNo ratings yet
- Grade 7 Unseen Passage 3Document2 pagesGrade 7 Unseen Passage 3Mokshitha Reddy YanamalNo ratings yet
- BibliographyDocument1 pageBibliographyMokshitha Reddy YanamalNo ratings yet
- TechnologyDocument2 pagesTechnologyMokshitha Reddy YanamalNo ratings yet
- Hindi Term Paper English TranslationDocument3 pagesHindi Term Paper English TranslationMokshitha Reddy YanamalNo ratings yet
- Inquiries and Investigations ResearchDocument24 pagesInquiries and Investigations ResearchKenjay CelesteNo ratings yet
- Induction Program Performance and MoraleDocument107 pagesInduction Program Performance and MoralePaul John BerdanNo ratings yet
- Types of Speech PresentationDocument23 pagesTypes of Speech PresentationAndrea GeilNo ratings yet
- IRCEELT 2018 ProceedingsDocument520 pagesIRCEELT 2018 Proceedingsდავით იაკობიძეNo ratings yet
- IPCRF MOVs For MTs I-IVDocument5 pagesIPCRF MOVs For MTs I-IVSimonette CastrenceNo ratings yet
- The Five Minds of A ManagerDocument8 pagesThe Five Minds of A ManagerAashutosh PandeyNo ratings yet
- What Is The Behavioural Science Approach To ManagementDocument4 pagesWhat Is The Behavioural Science Approach To ManagementPahulpreet SinghNo ratings yet
- Hope11 Q3 Week1 PDFDocument12 pagesHope11 Q3 Week1 PDFIan Joshua Sanchez100% (1)
- Rocio Glez Romero TFMDocument64 pagesRocio Glez Romero TFMaridimeglioNo ratings yet
- TEACHER: Simona Pu Cău Ș: Main Aims: Assumptions: The Students MayDocument2 pagesTEACHER: Simona Pu Cău Ș: Main Aims: Assumptions: The Students MayOrastean Andreea SimonaNo ratings yet
- Appraisal Forms 1 3 Q32022 GLDDocument3 pagesAppraisal Forms 1 3 Q32022 GLDGiancarla Maria Lorenzo DingleNo ratings yet
- Questions 1Document7 pagesQuestions 1Abdirahman AbdullahiNo ratings yet
- Lesson Plan: Industrial University of Ho Chi Minh CityDocument8 pagesLesson Plan: Industrial University of Ho Chi Minh CityThư TrầnNo ratings yet
- ENTREPRENEURIAL MOTIVATION AssignmentDocument26 pagesENTREPRENEURIAL MOTIVATION AssignmentxakilNo ratings yet
- Lecture 7 CrosstabsDocument20 pagesLecture 7 CrosstabsibmrNo ratings yet
- M1 Research MethodologyDocument18 pagesM1 Research MethodologyLarissa Rodrigues GCHSNo ratings yet
- Detailed Lesson Plan Music 1Document6 pagesDetailed Lesson Plan Music 1Michelle Dang-iw100% (1)
- The Teaching ProfessionDocument3 pagesThe Teaching ProfessionMichael V. MagallanoNo ratings yet
- Problem Solving For ELTDocument6 pagesProblem Solving For ELTMoez LamtiNo ratings yet
- Management and Behavioural Training E-Brochure (Revised)Document7 pagesManagement and Behavioural Training E-Brochure (Revised)Muhammad Imran QaziNo ratings yet
- RT Research1 Mod2 Chapter1Document16 pagesRT Research1 Mod2 Chapter1Lance VillanuevaNo ratings yet
- Score Sheet For Interview and Demo Elem and JHS XLSX SIR DRIO ROGER (Version 1) .XLSBDocument4 pagesScore Sheet For Interview and Demo Elem and JHS XLSX SIR DRIO ROGER (Version 1) .XLSBRoger DrioNo ratings yet
- Question Bank of MCQs - IDT For IA3Document10 pagesQuestion Bank of MCQs - IDT For IA3Srujan SNo ratings yet
- Course Syllabus: Catering 101Document6 pagesCourse Syllabus: Catering 101Sandre WaldenNo ratings yet
- Summary of DecisionsDocument2 pagesSummary of DecisionsKaran TalwarNo ratings yet
- Hot English MagazineDocument50 pagesHot English MagazineJahs Praiser100% (1)
- PM2 Methodology - Leaflet.v.2.1.4 18052018Document8 pagesPM2 Methodology - Leaflet.v.2.1.4 18052018AlainspamNo ratings yet
- AdolescenceDocument29 pagesAdolescenceivypearlbelicanNo ratings yet
- g1 Introduction Chapters 1 2Document25 pagesg1 Introduction Chapters 1 2Yoru BuffNo ratings yet
- Differenciate Between Ralph TylerDocument3 pagesDifferenciate Between Ralph TylerSudheer AyazNo ratings yet