Professional Documents
Culture Documents
Ethylene Glycol Mind Maps
Uploaded by
Himanshu BhardwajCopyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
Ethylene Glycol Mind Maps
Uploaded by
Himanshu BhardwajCopyright:
Available Formats
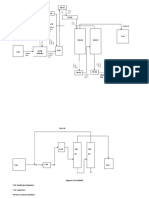
Na/
CH2ONa Na CH2ONa
01
323K
01
alk. KMnO 4
CH2 = CH2 + H2O + [O] 1 CH2OH 433 K CH2ONa
Ethene – H2 1
2 – H2
2
O
alk. KMnO 4
CH2 = CH2 + H2O + [O] Ag H2O
1 CH2ONO2
HNO3
Ethene
CH2–CH2
02
CH2 = CH2 + 2 O2
200 – 400°C H+ + 2H2O
O CH2ONO2
02
alk. KMnO 4
CH2 = CH2 + H2O + [O] Ag
1 H2O Ethylene dinitrate
CHEthene
2 = CH2 +
CH2–CH2
2 O2 200 – 400°C
OsO H+
4
CH2 = CH2 alk. KMnO 4 O
CH2 = CH2 + H2O + [O] H2O2
HIO4 or
Ethene Ag H2O
1
03
CH2 = CH2 + O2 OsO4 CH2–CH2
CH2 = CH2 2 200 – 400°C H+
H2O2
alk. KMnO 4 O
Cl2 + H2 O Lead tetra acetate
2HCHO + H2O
CH2 = CH2 + H2O + [O] Ag
CH H2ONa2CO3, H2O
CH2 = CH12 2–CH2
Ethene
CH2 = CH2 + 2 O2 CH2–CH2 +3[O]
03
200 OsO
– 400°C
4
| | H+
CH2 = CH2 Cl2H+2O
H22 O Cl Cl2CO3, H2O
Na
CH
alk. KMnO
2
= CH2 CHO2–CH2
CH2 = CH2 + H2O + [O] 4
alk. KMnO|4 |
CH2 = CH2 + 1H2O + [O] Ag H2O
Cl –CH
Cl KMnO4/H+
04
Ethene CH CH
2 = CH2 +
2 O2 200 –OsO
2HCOOH + H2O
Ethene 400°C
4
2 2
H +
CH2 = CH2 Na 2CO3/H 2O
ClH2 + HO
2O2 2
Na2CO3, H2O
CHBrCH
2
= CH42– CH2Br
Ethylene dibromide Na CO /H O |
CH2–CH2
|
+ 5[O]
ETHYLENE
1 Ag BrCH4 – CH2Br H2O2 3 2 Cl Cl
CH2 = CH2 + 2 O2 1 Ag
OsO H2O
Et2hylene
200 – 400°C
CH = CHdibromide
2 + O2 H+ 4
CH2 = CH2 2
O
04
200
ClH–2 2+O
400°C Na2H
775 K
+
H O CO3, H2O
GLYCOL
05
2 2
CH2 = CH2 CH2–CH2
COOC2H5 Na2CO 3
/H 2O
Na/C |
2H5OH
|
BrCH
| 4 – CH2Br
∆
OH Cl
OsO4 COOC
Ethylene2Hdibromide
| COOC2H5
5 Na/C2H5OH
OsO4
+ 8[H]
CH2 – CH2 + H2O
CH2OH
CH2 = CH2 Cl + H O Na2CO3, H2O
H2O2 CH
COOCDiethyl
2 = CH2H
oxalate 2 + 8[H]
2
CH2 = CH2 25
H2O2 CH2–CH2
Diethyl oxalate | |
Na2CO3/H 2O Cl Cl
COOC
BrCH H5 2Br
4 –2 CH Na/C2H5OH
ZnCl2 /∆
06
| CHO
Ethylene
COOC2H5
Cl2 + H2 O CHO
dibromide
Na/C2H5OH
+ 8[H] CH2
CH2OH
CH2 = CH2 |
Diethyl oxalate Na/C
Cl 2H5OH
2 + H2 O
CH3CHO
+ 4[H]
05
CH| 2 CHO
= CH
CHO 2
Glyoxal
+ 4[H]
Na CO
2
/H O
3 2 CHOH
BrCH 2 – CH2Br
Glyoxal
EtCOOC H5
hylene 2dibromide Na/C2H5OH
| CHO Na/C2H5OH
|
COOC 2H5
+ 8[H]
CHO oxalate + 4[H]
Diethyl
Na2CO3/H 2O
Glyoxal Na2CO3/H 2O
COOC2H5 Na/C2H5OH
|
COOC
CHO 2H5 + 8[H]
CH2 – CH2
H2 SO4 07
Na/C2H5OH
Diethyl
| oxalate
+ 4[H] O
O
CHO
Na/C2HCOOC H5
Glyoxal
5OH
∆
06
CH2 – CH2
2
Na/C2H5OH
PHYSICS
+ 8[H]
CHO Na/C 2H5OH
+ 8[H]
|
COOC2H5
WALLAH
CHO + 4[H]
Diethyl oxalate
Glyoxal
CHO Na/C2H5OH
CH |
CHO O – CH2
08
Gl
Glyoxal
CH3CHO/
07
CHO Na/C2H5OH CH3 – CH
|
CHO
Glyoxal
+ 4[H]
dry HCl O – CH2
You might also like
- Gambar Blok Diagram Pembuatan Metanol Dari Gas Alam Proses Hidrogenasi Karbon Monoksida Tekanan RendahDocument2 pagesGambar Blok Diagram Pembuatan Metanol Dari Gas Alam Proses Hidrogenasi Karbon Monoksida Tekanan RendahAulia HasanahNo ratings yet
- O N-NH: Wolf-Kishner ReductionDocument4 pagesO N-NH: Wolf-Kishner ReductiondfghNo ratings yet
- Pra Rancangan Pabrik Pembuatan Urea Formaldehid Dari Urea Dan MetanolDocument1 pagePra Rancangan Pabrik Pembuatan Urea Formaldehid Dari Urea Dan MetanolAlfiyah An-NajmNo ratings yet
- Organic TuteDocument1 pageOrganic TuteDimuthu SandaruwanNo ratings yet
- C11 Alcohol, Phenol & EthersDocument1 pageC11 Alcohol, Phenol & EthersRonak kadamNo ratings yet
- Functional Group Interconversion Scheme PDFDocument1 pageFunctional Group Interconversion Scheme PDFBilal AhmadNo ratings yet
- Maestri Hot TopicDocument56 pagesMaestri Hot TopicRaamses DíazNo ratings yet
- ALCOHOL : AlcoholsDocument11 pagesALCOHOL : AlcoholsSamirNo ratings yet
- OXIDATIONS FinalDocument9 pagesOXIDATIONS Finalgamer boomerNo ratings yet
- Problem Set 8Document2 pagesProblem Set 8CARLOS ALBERTO OSORIO MARTINEZNo ratings yet
- Name Reaction TestDocument2 pagesName Reaction TestvikiasNo ratings yet
- Phenols and Ethers NotesDocument9 pagesPhenols and Ethers NotesDhanaranjani BNo ratings yet
- Roadmap Problem - 3Document1 pageRoadmap Problem - 3abhyudaipathwayNo ratings yet
- B PharmDocument23 pagesB PharmpurnimaNo ratings yet
- ZuzanaDocument58 pagesZuzanaAlexanderNo ratings yet
- Combustion. Adiabatic Flame Temperature - Jeff MunicDocument56 pagesCombustion. Adiabatic Flame Temperature - Jeff MunicChristopher LloydNo ratings yet
- TutorialDocument27 pagesTutorialSiti NuraqidahNo ratings yet
- X Uv H /PT HX Mno4 H or Oh NH (Alc) Heat NH (Alc) : PCL PCL Socl ZN/HCLDocument1 pageX Uv H /PT HX Mno4 H or Oh NH (Alc) Heat NH (Alc) : PCL PCL Socl ZN/HCLEmily McCullochNo ratings yet
- Alcohol, Phenol - EthersDocument1 pageAlcohol, Phenol - Etherssarthakyedlawar04No ratings yet
- Alcohols, Phenols and Ethers - Short Notes - Prayas JEE 2024Document6 pagesAlcohols, Phenols and Ethers - Short Notes - Prayas JEE 2024yash vardhanNo ratings yet
- OxygenDocument6 pagesOxygentruongvuthanhmai_324No ratings yet
- MINDMAP Alkene, Benzene, HaloalkaneDocument3 pagesMINDMAP Alkene, Benzene, HaloalkaneLeow JiashengNo ratings yet
- (Eduwaves360) OC Hydrocarbon EDocument100 pages(Eduwaves360) OC Hydrocarbon Ekrutika goharkar100% (1)
- Solucionario de Wade-306-323 PDFDocument18 pagesSolucionario de Wade-306-323 PDFCésar PastranoNo ratings yet
- OQR (Oragnic Quick Revision) (ALIPHATIC) : Mcpba Conc. H SO DDocument2 pagesOQR (Oragnic Quick Revision) (ALIPHATIC) : Mcpba Conc. H SO Dmanya9b32100% (1)
- 4L OxidationsDocument16 pages4L OxidationsCarlos Javier Orellana OrtizNo ratings yet
- Diagram KualitatifDocument3 pagesDiagram KualitatifUci UtamiNo ratings yet
- Allen Organic QUICK RevisionDocument2 pagesAllen Organic QUICK RevisionChetna Ahlawat100% (2)
- Synthesis of Acetoaldehyde, Acetic Acid, and Others by The Dehydrogenation and Oxidation of EthanolDocument9 pagesSynthesis of Acetoaldehyde, Acetic Acid, and Others by The Dehydrogenation and Oxidation of EthanolJuan DavidNo ratings yet
- Qu 2014Document18 pagesQu 2014Melsi SihombingNo ratings yet
- Solution Felder 9.16Document3 pagesSolution Felder 9.16Alya Sholikhatul100% (1)
- AIEEE Chemistry Quick ReviewDocument1 pageAIEEE Chemistry Quick ReviewYashwanth KalyanNo ratings yet
- Tarea 7 Sistemas Dipróticos y TripróticosDocument2 pagesTarea 7 Sistemas Dipróticos y TripróticosSantiago GarciaNo ratings yet
- Road Maps Organic Chemistry Set 3 Eklavya @JEEAdvanced - 2024Document6 pagesRoad Maps Organic Chemistry Set 3 Eklavya @JEEAdvanced - 2024puneethrgcNo ratings yet
- Hints and SolutionsDocument9 pagesHints and SolutionsAnil YaduvansiNo ratings yet
- Roadmap Problem - 6Document1 pageRoadmap Problem - 6abhyudaipathwayNo ratings yet
- Aep 8Document6 pagesAep 8devkaushik0613No ratings yet
- Nucleus OC Hydrocarbon EDocument94 pagesNucleus OC Hydrocarbon Epoonam sharmaNo ratings yet
- Amines: (I) Basic Character of AminesDocument1 pageAmines: (I) Basic Character of AmineseliyachrisNo ratings yet
- 3CH CH CH CH + B H 3 (CH CH CH CH) B B (OH) +: (Basic Character)Document1 page3CH CH CH CH + B H 3 (CH CH CH CH) B B (OH) +: (Basic Character)SHADOW SNo ratings yet
- Block Flow Diagram Prarancangan Pabrik Dimethyl Ether Dari Limbah Biomassa Perkebunan Kelapa Sawit DenganDocument2 pagesBlock Flow Diagram Prarancangan Pabrik Dimethyl Ether Dari Limbah Biomassa Perkebunan Kelapa Sawit DengandesniaNo ratings yet
- The Sandmeyer Reaction: Replacement of The Diazonium Group by CL, BR, or CNDocument2 pagesThe Sandmeyer Reaction: Replacement of The Diazonium Group by CL, BR, or CNUsman GhaniNo ratings yet
- Xercise: Socl Pyridine, O CH CL MG (CH) O O CH MGCLDocument5 pagesXercise: Socl Pyridine, O CH CL MG (CH) O O CH MGCLPriyanshu RajNo ratings yet
- Super Important Reaction (Repaired)Document5 pagesSuper Important Reaction (Repaired)Rudra PratapNo ratings yet
- Super Important Reaction (Repaired)Document5 pagesSuper Important Reaction (Repaired)Madhav BhatiNo ratings yet
- Super Important Reaction (Repaired)Document5 pagesSuper Important Reaction (Repaired)AlphaNo ratings yet
- Alkyl Halides & Aryl Halides-02 - Solved ProblemsDocument13 pagesAlkyl Halides & Aryl Halides-02 - Solved ProblemsRaju SinghNo ratings yet
- Wa0001.Document1 pageWa0001.Akanksha VermaNo ratings yet
- Aldehyde Ketone and AcidDocument15 pagesAldehyde Ketone and AcidSsNo ratings yet
- Allen Organic Quic RivisionDocument2 pagesAllen Organic Quic Rivisionsaisupreeth0913No ratings yet
- Roadmap Problem - 1Document1 pageRoadmap Problem - 1Siddharth SharmaNo ratings yet
- Roadmap Problem - 9Document1 pageRoadmap Problem - 9abhyudaipathwayNo ratings yet
- 100S120 CS19L01Document38 pages100S120 CS19L01b101112154No ratings yet
- Road Maps Organic Chemistry Set 2 Eklavya @JEEAdvanced - 2024 (2) (4 Files Merged)Document11 pagesRoad Maps Organic Chemistry Set 2 Eklavya @JEEAdvanced - 2024 (2) (4 Files Merged)puneethrgcNo ratings yet
- Phenolic Resin - BrosurDocument53 pagesPhenolic Resin - BrosurpaimannNo ratings yet
- 10 PrintDocument2 pages10 Printsubin v pNo ratings yet
- PhenolDocument6 pagesPhenolPASCAL EMMANUELNo ratings yet
- Water Disinfection Ferrate VI As The Gre PDFDocument10 pagesWater Disinfection Ferrate VI As The Gre PDFalejandra mNo ratings yet
- Exp 7 Preparation of AlkenesDocument14 pagesExp 7 Preparation of AlkenesGeorge PiliposyanNo ratings yet
- Heat Capacities of Inorganic and Organic Ideal Gas StateDocument7 pagesHeat Capacities of Inorganic and Organic Ideal Gas StateGabriel SugayaNo ratings yet
- Indifference CurvesDocument18 pagesIndifference CurvesjiaNo ratings yet
- Smart Money Concept Cryptocurrency Day Trading For A LivingDocument27 pagesSmart Money Concept Cryptocurrency Day Trading For A LivingZack Ming67% (3)
- Short Notes From Analysis of Derivatives by Don M. ChanceDocument5 pagesShort Notes From Analysis of Derivatives by Don M. ChancemmajidNo ratings yet
- Relative Strength Index, or RSI, Is A Popular Indicator Developed by A Technical AnalystDocument2 pagesRelative Strength Index, or RSI, Is A Popular Indicator Developed by A Technical AnalystvvpvarunNo ratings yet
- The Frankfurt School and The Culture IndustryDocument4 pagesThe Frankfurt School and The Culture IndustryJoon Bok NamleeNo ratings yet
- Economics Project: Topic-Inflation in IndiaDocument4 pagesEconomics Project: Topic-Inflation in IndiaTaiyabaNo ratings yet
- International Trade 3rd Edition Feenstra Solutions ManualDocument13 pagesInternational Trade 3rd Edition Feenstra Solutions ManualJenniferThompsongoacm100% (12)
- How To Avoid False BreakoutDocument14 pagesHow To Avoid False BreakoutLokesh YadavNo ratings yet
- The Stock Market For Dummies PDFDocument2 pagesThe Stock Market For Dummies PDFJoe D100% (1)
- Momentum Indicators: An Empirical Analysis of The Concept of DivergencesDocument10 pagesMomentum Indicators: An Empirical Analysis of The Concept of Divergencespderby1No ratings yet
- Internship Proof - TemplateDocument3 pagesInternship Proof - TemplateNaveen Venkatess V HNo ratings yet
- 5 Powerful Bearish Candlestick Patterns: Technical AnalysisDocument18 pages5 Powerful Bearish Candlestick Patterns: Technical Analysiswolf1No ratings yet
- Financial Derivative Ajitav AcharyaDocument45 pagesFinancial Derivative Ajitav AcharyaBhabani Sankar BrahmaNo ratings yet
- VWAP BreakOut Failure StrategyDocument29 pagesVWAP BreakOut Failure StrategymanenderNo ratings yet
- International Parity Conditions: Dr. Ch. Venkata Krishna Reddy Associate ProfessorDocument34 pagesInternational Parity Conditions: Dr. Ch. Venkata Krishna Reddy Associate Professorkrishna reddyNo ratings yet
- Binary Options Trading Strategies StrateDocument19 pagesBinary Options Trading Strategies StrateJoseph peelwadNo ratings yet
- Technical Analysis by Leavitt BrothersDocument441 pagesTechnical Analysis by Leavitt Brothersvinodtp100% (2)
- A Stock Market Model - NotesDocument12 pagesA Stock Market Model - Notesshadow slayerNo ratings yet
- Stock Exchange and Its Losses BST Project Class 12Document13 pagesStock Exchange and Its Losses BST Project Class 12Varoon RajeshNo ratings yet
- International Economics 12th Edition Salvatore Test BankDocument10 pagesInternational Economics 12th Edition Salvatore Test Bankbewet.vesico1l16100% (23)
- Introduction To Macro Economics Class 12 NotesDocument13 pagesIntroduction To Macro Economics Class 12 NotesAdnan Khan XI Com.Ext.No ratings yet
- Welcome To The Book of Finacial Freedom. Read and Read With Understandind. Learn Super Massive StrategyDocument21 pagesWelcome To The Book of Finacial Freedom. Read and Read With Understandind. Learn Super Massive StrategySthembiso ThusiNo ratings yet
- Mintec Overview Presentation Industrial 2023Document10 pagesMintec Overview Presentation Industrial 2023fcrpolykoteNo ratings yet
- Elliot Wave and Fibonacci Retrenchment of 10 StocksDocument10 pagesElliot Wave and Fibonacci Retrenchment of 10 StocksPRADIP.KUMAR SHUKLANo ratings yet
- Index NumbersDocument17 pagesIndex NumbersGudduNo ratings yet
- ABSTRACT LABOR AND COMMODITY PRODUCTION John WeeksDocument20 pagesABSTRACT LABOR AND COMMODITY PRODUCTION John WeeksTom Waspe TeachNo ratings yet
- An Impressive Start To The Year For Nickel As It Hits 10-Year HighDocument7 pagesAn Impressive Start To The Year For Nickel As It Hits 10-Year HighOwm Close CorporationNo ratings yet
- Bitcoin Price AnalysisDocument4 pagesBitcoin Price AnalysisNicholas GeeNo ratings yet
- Different Types of Exchange System: ReciprocityDocument2 pagesDifferent Types of Exchange System: Reciprocitymehazabin anamikaNo ratings yet