Professional Documents
Culture Documents
Bab Iv Iso
Bab Iv Iso
Uploaded by
Akidah TahirOriginal Title
Copyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
Bab Iv Iso
Bab Iv Iso
Uploaded by
Akidah TahirCopyright:
Available Formats
BAB IV
HASIL DAN PEMBAHASAN
A. Hasil Pengamatan
1. Tabel Pengamatan
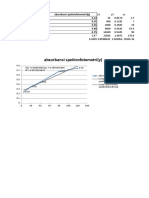
Tabel IV.1 Hasil Pengamatan Larutan Standar
x y
No. Larutan x.y x2 y2
(mg/L) (Adsorbansi)
1 Standar 1 0,5 0,0466 0,0233 0,25 0,00217156
2 Standar 2 1 0,0728 0,0728 1 0,00529984
3 Standar 3 2 0,1483 0,2966 4 0,02199289
4 Standar 4 4 0,3342 1,3368 16 0,11168964
5 Standar 5 8 0,6865 5,4920 64 0,47128225
n=5 Σ = 15,5 Σ =1,2884 Σ =7,2215 Σ = 85,25 Σ =0,61243618
Tabel IV.2 Hasil Pengamatan Larutan Contoh
No. Larutan x (mg/L) y (Adsorbansi) Panjang
Gelombang (nm)
1 Sampel 1 30 0,1325 0,0442
2 Sampel 2 45 0,0779 0,0959
3 Sampel 3 60 0,0833 4,0240
4 Sampel 4 75 0,1238 4,9520
5 Sampel 5 90 0,0739 8,7440
Tabel IV.3 Hasil Perhitungan Efektivitas Adsorpsi Larutan Sampel
Konsentrasi Kapasitas Efektivitas
No. Larutan Adsorbansi
Sisa Adsorpsi Adsorpsi
1 Sampel 30 ppm 0,1325 1,657163837 0,005668567 94,47 %
2 Sampel 45 ppm 0,0779 1,027839229 0,000879443 97,71 %
3 Sampel 60 ppm 0,0833 1,090080124 0,011781983 98,18 %
4 Sampel 75 ppm 0,1238 1,556886839 0,014688622 97,92 %
5 Sampel 90 ppm 0,0739 0,981734862 0,017803653 98,90 %
Tabel IV.4 Persamaan Freundlich
No. Larutan Ce Qe x (log Ce) y (log Qe)
1 Sampel 30 ppm 1,657163837 0,0056685672 0,2193654476 -2,246526698
2 Sampel 45 ppm 1,027839229 0,0008794432 0,0119251894 -3,055792197
3 Sampel 60 ppm 1,090080124 0,0117819839 0,0374584213 -1,928781572
4 Sampel 75 ppm 1,556886839 0,0146886226 0,1922570476 -1,833018926
5 Sampel 90 ppm 0,981734862 0,0178036530 -0,008005786 -1,749490878
Tabel IV.5 Persamaan Langmuir
No. Larutan Ce Qe x (1/Ce) y (1/Qe)
1 Sampel 30 ppm 1,657163837 0,0056685672 0,6034406361 176,4114219
2 Sampel 45 ppm 1,027839229 0,0008794432 0.9729148020 1137,083099
3 Sampel 60 ppm 1,090080124 0,0117819839 0.9173637588 84,87534938
4 Sampel 75 ppm 1,556886839 0,0146886226 0.6423074400 68,07990287
5 Sampel 90 ppm 0,981734862 0,0178036530 1,0186049601 56,16824816
2. Grafik
a. Larutan Standar
Larutan Standar
0.8
0.8000
0.7
0.7000
f(x) =f(x)
0.0864548387096774 x − 0.00924999999999998
= 0.0864548387096774 x − 0.00924999999999998
0.6
0.6000 R² = 0.998496213958368
R² = 0.998496213958368
Y (Absorbansi)
0.5
Adsorbansi (Y)
0.5000
0.4
0.4000 Absorbansi
Adsorbansi
0.3 LinearLinear
(Adsorbansi)
0.3000 (Ab-
sorbansi)
0.2
0.2000
0.1000
0.1
0.0000
0
0 0
1 12 2 3 3 44 55 6 6 7 78 9
8 9
Konsentrasi
Konsentrasi (mg/L) X (ppm)
Grafik IV.1 Larutan Standar
b. Larutan Contoh
Larutan
Konsentrasi ContohContoh
Larutan
0.14
10.0000
0.129.0000
8.0000 f(x) = 0.0499799756097561 x − 1.67587743902439
f(x) = 0.000732285714285714
0.17.0000 x + 0.0452857142857143
Y (Absorbansi)
R² = 0.959203989732364
R² = 0.253501624285846
Adsorbansi (Y)
0.086.0000 Absorbansi
5.0000 Adsorbansi
Linear
0.064.0000 (Ab-
Linear (Adsorbansi)
sorbansi)
0.043.0000
2.0000
0.021.0000
0
0.0000
0 100 20 5030 100 15060 200
40 50 70 250 90 100
80
Konsentrasi (mg/L) X (ppm)
Konsentrasi
Grafik IV.2 Larutan Contoh
c. Grafik Isoterm Adsorpsi Freundlich
Isoterm Adsorpsi
Persamaan Freundlich
Freundlich
0.25 0.0000
-1.0000 0.0000 1.0000 2.0000 3.0000
0.2 -0.5000
0.15 -1.0000
Persamaan Freundlich
Log Ce
Log Ce
log Qe
0.1 -1.5000 Linear (Persamaan
Linear (Log Ce)
Freundlich)
-2.0000
f(x) = 9.14285714285715E-05 x + 0.0674285714285714
0.05 R² = 0.00087491455912525
f(x) = 0.207928803173391 x − 2.46900705578348
-2.5000 R² = 0.748566526578363
0
0 10 20
-3.0000 30 40 50 60 70 80 90 100
-0.05
logKonsentrasi
Ce X (ppm)
Grafik IV.3 Isoterm Adsorpsi Freundlich
d. Grafik Isoterm Adsorpsi Langmuir
Isoterm Adsorpsi
Persamaan Langmuir
Langmuir
0
450.0000
0 10 20 30 40 50 60 70 80 90 100
400.0000
-0.5
350.0000 f(x) = 166.519500856621 x + 120.739080767104
R² = 0.870853610830886
-1
300.0000
f(x) = − 0.0141904761904762 x − 1.08714285714286
250.0000
R² = 0.210808480576108 Persamaan Langmuir
-1.5
Log Qe
1/Qe
200.0000 Linear (Persamaan
Log Qe
Langmuir)
150.0000
-2 Linear (Log Qe)
100.0000
-2.5
50.0000
-3 0.0000
0.00000.50001.00001.50002.0000
-3.5
1/Ce
Konsentrasi X (ppm)
Grafik IV.4 Isoterm Adsorpsi Langmuir
You might also like
- Lab Report (Atomic Absorption Spectroscopy)Document8 pagesLab Report (Atomic Absorption Spectroscopy)Shirley Cheong67% (6)
- (0000) (Collection) 木村拓哉ドラマと映画 Kimura Takuya - JDrama, Movies.torrent 108.4 GiBDocument4 pages(0000) (Collection) 木村拓哉ドラマと映画 Kimura Takuya - JDrama, Movies.torrent 108.4 GiBHoonchul KimNo ratings yet
- Lab Report Batch Reactor GGDocument25 pagesLab Report Batch Reactor GGFrost Orchid100% (1)
- UVDocument11 pagesUVShaun Loo100% (3)
- Chm580 Experiment 2Document8 pagesChm580 Experiment 2ohhi100% (1)
- Metric Conversion Chart: Metric Length Measurement: Word ProblemsDocument1 pageMetric Conversion Chart: Metric Length Measurement: Word ProblemsDemo NOWNo ratings yet
- Kurva Kalibrasi STD PCT: Analisis Data Pengujian Paracetamol Dengan HPLCDocument4 pagesKurva Kalibrasi STD PCT: Analisis Data Pengujian Paracetamol Dengan HPLCaprilia kurnia putriNo ratings yet
- IVDocument2 pagesIV12TĐ15 Bạch Long VĩNo ratings yet
- Kurva Kalibrasi Standarpct: Konsentrasi (%)Document3 pagesKurva Kalibrasi Standarpct: Konsentrasi (%)aprilia kurnia putriNo ratings yet
- Seminar 1 PDFDocument29 pagesSeminar 1 PDFDiana Andrade LarrañagaNo ratings yet
- Data EksperimenDocument1 pageData EksperimenAn Nur Zajar BmtNo ratings yet
- Fe CalibrationDocument4 pagesFe CalibrationShu YeeNo ratings yet
- X/MVSC Logx/Mvslogc: C Naoh 1.725 N MR Ch3Co 60 Mg/Mmol Karbon Aktif SerbukDocument4 pagesX/MVSC Logx/Mvslogc: C Naoh 1.725 N MR Ch3Co 60 Mg/Mmol Karbon Aktif SerbukadevirgieNo ratings yet
- X/MVSC Logx/Mvslogc: C Naoh 1.725 N MR Ch3Co 60 Mg/Mmol Karbon Aktif SerbukDocument4 pagesX/MVSC Logx/Mvslogc: C Naoh 1.725 N MR Ch3Co 60 Mg/Mmol Karbon Aktif SerbukAidah NaufiaNo ratings yet
- Analisi Data KromatografiDocument3 pagesAnalisi Data KromatografiSAHRUL ADI SAPUTRANo ratings yet
- Penentuan Uji Kelarutan KTI ParacetamolDocument6 pagesPenentuan Uji Kelarutan KTI ParacetamolMinul1412No ratings yet
- Supplementary MaterialDocument7 pagesSupplementary MaterialInigo JohnsonNo ratings yet
- Analisis Data Uv-VisDocument8 pagesAnalisis Data Uv-VisStarvilla HNo ratings yet
- Kurva Kalibrasi Parasetamol λ maks 243 nmDocument2 pagesKurva Kalibrasi Parasetamol λ maks 243 nmGelisaNo ratings yet
- Lab Report Beer Lambert'S Law Experiment: Determing The Concentration of Unknown Asa SolutionsDocument5 pagesLab Report Beer Lambert'S Law Experiment: Determing The Concentration of Unknown Asa Solutionsumair saleemNo ratings yet
- No Label Sampel Konsentrasi AbsorbansiDocument2 pagesNo Label Sampel Konsentrasi AbsorbansiJoanna AdelineNo ratings yet
- Enzyme Kinetics ReportDocument9 pagesEnzyme Kinetics ReportRudshut CudleNo ratings yet
- Orde 1 Orde 2: T (S) T (S)Document3 pagesOrde 1 Orde 2: T (S) T (S)Lukman El-lathifNo ratings yet
- 2) Result For Coffea RobustaDocument6 pages2) Result For Coffea RobustaVinda PatriciaNo ratings yet
- Estimasi ThiaminDocument9 pagesEstimasi ThiaminDwi VaniaNo ratings yet
- Pengolahan Data H10 - Kelompok 07Document6 pagesPengolahan Data H10 - Kelompok 07Iqbal RoroaNo ratings yet
- %kadar (B/V) (X) Absorbansi Spektrofotometri (Y) 5 0.24 20 0.35 40 0.45 80 0.68 120 0.75Document4 pages%kadar (B/V) (X) Absorbansi Spektrofotometri (Y) 5 0.24 20 0.35 40 0.45 80 0.68 120 0.75Nida AdillahNo ratings yet
- Experiment 2Document5 pagesExperiment 2Alya HaifaNo ratings yet
- Analisa Air LautDocument8 pagesAnalisa Air LautUfafa AnggariniNo ratings yet
- Lou 1 - Practica N2Document9 pagesLou 1 - Practica N2Anthony Quispe CarbajalNo ratings yet
- Contoh Val-Ver Ortofosfat-OkDocument15 pagesContoh Val-Ver Ortofosfat-OkRANY RAMADHANINo ratings yet
- Weir CalculationDocument8 pagesWeir CalculationWade LiNo ratings yet
- In Calibrating A 10Document4 pagesIn Calibrating A 10GrenlyKerehNo ratings yet
- Antioksidan FRAP FIXDocument4 pagesAntioksidan FRAP FIXizzaaaaawNo ratings yet
- Experiment 1: The Visible Spectra of Soft Drinks: A. Pre-Laboratory QuestionsDocument5 pagesExperiment 1: The Visible Spectra of Soft Drinks: A. Pre-Laboratory QuestionsMuhd Mirza HizamiNo ratings yet
- Kurva Kalibrasi Larutan Standar C-Organik: 1. Penentuan LinieritasDocument4 pagesKurva Kalibrasi Larutan Standar C-Organik: 1. Penentuan Linieritasaprilia kurnia putriNo ratings yet
- Atomic Absorption Spectroscopy: CHEMY 313 Analytical ChemistryDocument7 pagesAtomic Absorption Spectroscopy: CHEMY 313 Analytical ChemistryJassim123 SabtNo ratings yet
- Book1 (Autosaved) CourbDocument4 pagesBook1 (Autosaved) CourbayoubalkheirNo ratings yet
- Quality ControlDocument6 pagesQuality ControlAinun SyarifNo ratings yet
- IuyhtdgrsfghjklDocument10 pagesIuyhtdgrsfghjklJose LuisNo ratings yet
- RESULT and DISCUSSIONDocument5 pagesRESULT and DISCUSSIONnisasoberiNo ratings yet
- La Courbe D'étalonnage: C (PPM) ABS 10 0.1 20 0.3 30 0.45 40 0.6 50 0.75 60 0.9Document19 pagesLa Courbe D'étalonnage: C (PPM) ABS 10 0.1 20 0.3 30 0.45 40 0.6 50 0.75 60 0.9ouarasismahanNo ratings yet
- Reactorpfr 1 Practica 4 ReactoresDocument6 pagesReactorpfr 1 Practica 4 ReactoresLeticia AlvaradoNo ratings yet
- Homework No. 3..Document7 pagesHomework No. 3..Katherine PaterninaNo ratings yet
- Tugas QC Bu AnaDocument4 pagesTugas QC Bu AnaJuw EllahNo ratings yet
- ValmetDocument9 pagesValmetAhmad PrayogaNo ratings yet
- EXP 2Document12 pagesEXP 2tanaygupta1011No ratings yet
- Concentration Vs Conductivity: 1.0 Results, Discussion and Analysis 1.1 Calibration CurveDocument9 pagesConcentration Vs Conductivity: 1.0 Results, Discussion and Analysis 1.1 Calibration CurveAhZaiSkyNo ratings yet
- Lab Report AASDocument8 pagesLab Report AASMohamad Saiful Mohd RaffiahNo ratings yet
- Data Prak Kimling 4Document4 pagesData Prak Kimling 4Putri Nur AngelinaNo ratings yet
- Data Mentah PenelitianDocument6 pagesData Mentah PenelitianIrham KusumaNo ratings yet
- Kurva Kalibrasi Fe Absorban Vs KonsentrasiDocument1 pageKurva Kalibrasi Fe Absorban Vs KonsentrasiKinanthi SekarNo ratings yet
- Chart Title: X (Fraccion Molar, Liquido)Document3 pagesChart Title: X (Fraccion Molar, Liquido)alberto quispe huamanNo ratings yet
- Kurva Kalibrasi SO4Document2 pagesKurva Kalibrasi SO4medya leaciliaNo ratings yet
- Chemy 310 Expriment 4Document6 pagesChemy 310 Expriment 4Faisal MumtazNo ratings yet
- Lab AnalysisDocument4 pagesLab AnalysisErnestasBlaževičNo ratings yet
- Reporte SedimentscionDocument13 pagesReporte SedimentscionANDREA CAMARGONo ratings yet
- Bab Iv Hasil Dan PembahasanDocument4 pagesBab Iv Hasil Dan PembahasanIndah Suci RamadhaniNo ratings yet
- CPP Lab 1 MuahDocument16 pagesCPP Lab 1 Muahelynnmasrof100% (1)
- Pengolah Data PFR (Abang)Document6 pagesPengolah Data PFR (Abang)Anonymous vjTWskvNo ratings yet
- Hasil Adsorben KontrolDocument5 pagesHasil Adsorben KontrolDwi NoviantiNo ratings yet
- Inductively Coupled Plasma-Mass Spectrometry: Practices and TechniquesFrom EverandInductively Coupled Plasma-Mass Spectrometry: Practices and TechniquesNo ratings yet
- ConversionsDocument3 pagesConversionsBoedisantosoNo ratings yet
- Torque Conversions Technical Information - MSC Industrial Supply CoDocument2 pagesTorque Conversions Technical Information - MSC Industrial Supply CoMarcelo OliveiraNo ratings yet
- 1471524general ScienceDocument623 pages1471524general ScienceAnurag ShrivastavNo ratings yet
- Units & AbbreviationsDocument3 pagesUnits & AbbreviationsSiti_Balqis_Is_6819No ratings yet
- Test Bank For College Physics 7th Edition Jerry D WilsonDocument24 pagesTest Bank For College Physics 7th Edition Jerry D WilsonJames Cunningham100% (35)
- Q 6Document358 pagesQ 6orsbsoulNo ratings yet
- List of Length UnitsDocument7 pagesList of Length UnitsDiscover LearnNo ratings yet
- Units of Measurement - Print - QuizizzDocument4 pagesUnits of Measurement - Print - QuizizzAliyah LuharNo ratings yet
- Construction Cost Management Systems: Lecture 1: Course Introduction, Basics of Cost Management, Intro To IS 1200Document29 pagesConstruction Cost Management Systems: Lecture 1: Course Introduction, Basics of Cost Management, Intro To IS 1200Er Inamul HassanNo ratings yet
- Tuercas y SegurosDocument15 pagesTuercas y SegurosMonserrat Hernández SánchezNo ratings yet
- SIGuide ConversionsDocument12 pagesSIGuide Conversionscalvin.bloodaxe4478No ratings yet
- Chemical Process Calculations: Sweta C Balchandani Faculty, SOT-Chemical, PdpuDocument32 pagesChemical Process Calculations: Sweta C Balchandani Faculty, SOT-Chemical, PdpuParikh DhruvNo ratings yet
- Lesson Plan Measuring of MassDocument3 pagesLesson Plan Measuring of MassJason XavierNo ratings yet
- MV Chilean Bulker Proposed Stowplan 2Document1 pageMV Chilean Bulker Proposed Stowplan 2Gaurav MantriNo ratings yet
- Lab 4: Itecalculator: Storageconverter StorageconverterDocument5 pagesLab 4: Itecalculator: Storageconverter StorageconverterChhay LayNo ratings yet
- U Cân Laumax W200 BaseDocument36 pagesU Cân Laumax W200 BaseJose Luna0% (1)
- Complete List of Energy Units For ConversionDocument2 pagesComplete List of Energy Units For Conversionreksian bungasarrangNo ratings yet
- Basic & Derived Quantity (SI Units)Document2 pagesBasic & Derived Quantity (SI Units)Muhammad Noman Mughal0% (1)
- DRG Size - Scale SelectionDocument10 pagesDRG Size - Scale SelectionkrishNo ratings yet
- Summary Notes - Topic 1 Edexcel Physics GCSEDocument3 pagesSummary Notes - Topic 1 Edexcel Physics GCSEAli AbdullahNo ratings yet
- Chapter 1 - Feb 2017Document70 pagesChapter 1 - Feb 2017Firdaus RoslimNo ratings yet
- Initial Inputs Current Body Stats: Goal Weight Loss Per WeekDocument8 pagesInitial Inputs Current Body Stats: Goal Weight Loss Per WeekrapinoNo ratings yet
- Lesson 2 Customary Units of Liquid MeasureDocument4 pagesLesson 2 Customary Units of Liquid Measureapi-248726622No ratings yet
- Mechanics of Materials (2015)Document258 pagesMechanics of Materials (2015)theocrite100% (12)
- Units of VilocityDocument2 pagesUnits of VilocityvinodbankheleNo ratings yet
- Converting Metric Units To English UnitsDocument14 pagesConverting Metric Units To English Unitsliezel nosnosanNo ratings yet