Professional Documents
Culture Documents
(@bohring - Bot) Special Batch Assignment-6
Uploaded by
lash73752Original Description:
Original Title
Copyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
(@bohring - Bot) Special Batch Assignment-6
Uploaded by
lash73752Copyright:
Available Formats
Vidyamandir Classes
Special Batch Assignment-6 | Chemistry
[Introduction to Organic Chemistry & Hydrocarbons]
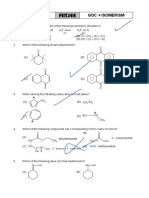
SECTION-1 SINGLE CORRECT ANSWER TYPE
1. Which has maximum dipole moment?
(A) (B)
(C) (D)
2. Which one among the following carbocations has the longest half-life?
(A) (B)
+
(C) (D) CH3 − C − CH3
|
CH3
3. The stable form of trans-1,4-dimethylcyclohexane is represented as:
(A) (B)
(C) (D)
4. The relationship among the following pairs of isomer is:
I. A : Constitutional II. B : Configurational
III. C : Conformational IV. D : Optical
(A) I-A, II-B, III-B, IV-D (B) I-A, II-A, III-B, IV-D
(C) I-B, II-A, III-B, IV-D (D) I-B, II-B, III-A, IV-B
VMC | Chemistry 1 Special Assignment-6 | JEE-2023
Vidyamandir Classes
5. Which of the following does not represent major product of that reaction?
(A)
(B)
(C)
(D)
6. ; Product (C) of the reaction is:
(A) (B) (C) (D)
7. Which of the following reactions results in the formation of a pair of diastereomers ?
(A) (B)
(C) (D)
8. (mCPBA is m-chloroperoxobenzoic acid)
(A) (B)
(C) (D) None of these
VMC | Chemistry 2 Special Assignment-6 | JEE-2023
Vidyamandir Classes
9. The major product of the following reaction sequence is:
(A) (B) (C) (D)
10.
(Styrene)
(A) Ph − C CNa (B) Ph − CH2 − C CH
(C) Ph − C C − CH3 (D) Ph − CH = C = CH2
11.
Identify the product (C).
O O CH2OH
|| || |
(A) CH3 − C − C − O − CH3 (B) CHOH
|
CH2OH
CHO CHO
| |
(C) CHOH (D) CH − OH
| |
CH 2 OH CHO
12. Select the reaction(s) that would result in the formation of 2-bromopropane.
I. CH3CH = CH2 + HBr ⎯⎯⎯⎯
peroxide
→ II. CH3CH = CH2 + HBr ⎯⎯⎯
CCl4
→
III. CH3CH2CH3 + Br 2 ⎯⎯
hv
→ IV. CH3CH = CH2 + Br2 ⎯⎯⎯
CCl4
→
(A) I and III (B) II and III (C) I, II and III (D) I and II
13.
Compound (C) in above sequence of reaction is:
(A) (B)
(C) (D)
VMC | Chemistry 3 Special Assignment-6 | JEE-2023
Vidyamandir Classes
14. Which is incorrect statement about heats of combustion?
(A) (B)
(C) Iso-butene > trans-2-butene > 1-butene (D) n-Hexane < n-Heptane < n-Octane
15.
(A) (B) (C) (D)
16.
17.
18.
19.
20.
VMC | Chemistry 4 Special Assignment-6 | JEE-2023
Vidyamandir Classes
21.
22.
23.
24.
25.
26.
VMC | Chemistry 5 Special Assignment-6 | JEE-2023
Vidyamandir Classes
27.
28.
29.
30.
31.
VMC | Chemistry 6 Special Assignment-6 | JEE-2023
Vidyamandir Classes
32.
33.
34.
35.
36.
37.
38.
39.
40.
VMC | Chemistry 7 Special Assignment-6 | JEE-2023
Vidyamandir Classes
SECTION-2 MULTIPLE CORRECT ANSWER TYPE
1. The correct statement regarding a pair of enantiomers is(are):
(A) They have opposite absolute configuration at stereocentre
(B) They have opposite relative configuration at stereocentre
(C) They react similarly with an optically active isomer of other substance
(D) They can be separated by chromatographic method of separation
2. What is(are) true regarding Fischer projection of an optically active isomer?
(A) Interchange of two groups position at chiral centre changes the relative configuration
(B) Interchange of groups position at chiral centre changes the absolute configuration
(C) Rotation of Fischer projection any way but by 180 does not change the configuration at chiral
centre
(D) Reaction involving breaking of bonds to chiral centre always changes the sign of optical
rotation
F
|
3. Consider the following molecule. CH3 − CH − CH = CH − CH − CH3
|
F
The correct statement concerning above molecule is(are):
(A) It has a total of six stereoisomers
(B) It's meso form upon ozonolysis followed by Zn-hydrolysis gives racemic mixture
(C) It's optically active isomers, each upon ozonolysis followed by Zn-hydrolysis gives a single
enantiomer
(D) It has only two optically active isomers
4. Consider the following free radical chlorination reaction.
The correct statement about product(s) is(are):
(A) Total four isomers are formed (B) Two optically active isomers are formed
(C) One pair of diastereomers is formed (D) Two pair of diastereomers are formed
5. The correct statement(s) regarding isomers of a compound with general name trichlorocyclobutane
is(are):
(A) It has total seven isomers (B) It has two meso-isomers
(C) It has two optically active isomers (D) One isomer is devoid of any chiral carbon
6. Among the butane conformers, when viewed about C2 − C3 bond, which occur at energy minima on a
graph of potential energy versus dihedral angle?
(A) Gauche only (B) Gauche (C) Anti (D) Eclipsed
7. The correct statement regarding cyclopropane is(are):
(A) It has the highest angle strain among cycloalkanes
(B) It has smaller heat of hydrogenation per mole of − CH2 − than cyclohexane
(C) It has large torsional strain due to all eclipsed C − H bonds on adjacent carbons
(D) Conformations in cyclopropane is due to flipping of bonds
VMC | Chemistry 8 Special Assignment-6 | JEE-2023
Vidyamandir Classes
8. Select the correct statement(s) concerning conformers of ethane and hexachloroethane?
(A) Both have the same number of important conformers
(B) Potential energy versus dihedral angle curves have similar nature in both cases
(C) Least stable conformer in hexachloroethane is most polar
(D) In potential energy versus dihedral angle diagram, there is greater difference between maxima
and minima in ethane than in hexachloroethane
9. The correct statement concerning various conformers of 2-fluoroethanol is(are):
(A) Anti-conformer is the most stable conformer
(B) There is intramolecular H-bonding in its most stable conformer
(C) The most stable conformer has greater steric strain than 2nd most stable conformer
(D) Dissolving in water lower the percentage of most stable conformer
10. Consider the various conformers possible for meso-form and enantiomeric 1, 2-dibromo-1,
2-dichloroethane and select the correct statement(s):
(A) Both have exactly same energy barrier to rotation
(B) The most stable conformer of meso form has lower potential energy than of enantiomeric form
(C) In most stable conformer of an enantiomer, the dihedral angle between bromine atoms is 180°
(D) In the most of the conformation, meso form is non-polar
11. Select the correct statement(s) regarding conformers of 1, 2-dimethyl cyclohexane.
(A) In the most stable conformation, cis-isomer has methyl groups at both axial and equatorial
position
(B) In the most stable conformation, cis-isomer has methyl groups at equatorial positions only
(C) In the most stable conformation, trans-isomer has both methyl groups at axial positions only
(D) In the most stable conformation, trans-isomer has both methyl groups at equatorial positions
only
12. The correct statement regarding conformation in butane is(are):
(A) The two gauche forms, when viewed about C2 − C3 bond, are enantiomer
(B) There is greater barrier to rotation about C1 − C2 bond than to C2 − C3 bond
(C) Its gauche form has higher energy than an eclipsed form when CH3 − and H are eclipsing
(D) In the most stable conformer, dihedral angle between hydrogen atoms is only 60°
13. The correct statement(s) concerning the following Newman’s projections is(are):
(A) I and II correspond to conformers of enantiomers of 2, 3-dichlorobutane
(B) I and III are two conformers of the same enantiomer of 2, 3-dichlorobutane
(C) III is the most stable conformer of meso-2, 3-dichlorobutane
(D) I and III are conformers of a pair of diastereomers of 2, 3-dichlorobutane
14. Tautomerism is not exhibited by:
(A) (B) (C) (D)
VMC | Chemistry 9 Special Assignment-6 | JEE-2023
Vidyamandir Classes
15. Which of the following will show optical isomerism as well as geometrical isomerism?
(A) (B) (C) (D)
16. The correct statement regarding 1, 3-dichloropropadiene is(are):
(A) It can exist as a pair of geometrical isomers
(B) It shows enantiomerism
(C) The stereoisomers are polar
(D) The molecule is planar
17. The correct statements regarding following conformers is(are):
(A) The process is endothermic
(B) G for this process is greater than zero
(C) Increasing temperature will increase the percentage of I
(D) Conformer I has a plane of symmetry while II does not
18. Which is(are) true regarding the product of the following reaction?
() -2-butanamine + (+)-2-methyl butanoic acid ⎯⎯ →
(A) A single enantiomer of product will be formed
(B) A pair of enantiomers will be formed
(C) A pair of diastereomers will be formed
(D) Products mixture can be separated by fractional crystallization
19. What is(are) true about the following two conformers of 2-fluoroethanol?
(A) Both have equal percentage at equilibrium
(B) I has greater percentage at equilibrium due to less steric strain
(C) II has greater percentage at equilibrium due to intramolecular H-bonding
(D) Transformation of I to II will be an exothermic process
20. The correct statement(s) concerning the following structures is(are):
(A) P and Q are enantiomers
(B) P and Q are diastereomers
(C) Both are optically active
(D) P is optically inactive while Q is optically active
VMC | Chemistry 10 Special Assignment-6 | JEE-2023
Vidyamandir Classes
21. A solution containing stereoisomers A and B, is optically active. Which of the following statement(s)
concerning A and B can be true?
(A) A and B are enantiomers (B) A and B are diastereomers
(C) A and B are geometrical isomers (D) Neither A nor B contain any chiral carbon
22. The correct statement(s) concerning aldehydes of general formula C5H10O is(are):
(A) It has total five aldehyde isomers
(B) Two of the aldehydes are chiral
(C) Two of the aldehydes isomers have no enolic form
(D) All enols of aldehyde isomers are diastereomeric
23. In which of the following, enol form dominates over keto form?
(A) (B) (C) (D)
24. Which of the following is(are) true regarding a free radical?
(A) It is formed by homolytic cleavage of bond
(B) A chemical that is free radical initiator, i.e., peroxide, etc. is must for the formation
(C) It is a Lewis acid
(D) It undergoes disproportionation to some extent during the reaction
25. Which of the following compounds forms salts with KOH in aqueous solution?
(A) (B) O2 N − CH2 − NH2
(C) (D)
26. Which of the following is(are) regarding a carbene true?
(A) Carbon is sp2 hybridized
(B) It acts only as electrophile
(C) It can be both paramagnetic as well as diamagnetic
(D) All atoms lie in the same plane
27. The correct statement(s) regarding acidity of the following compounds is(are):
I. II.
(A) I is more acidic due to the greater –I effect
(B) II is more acidic due to the resonance effect of –NO2
(C) I is less acidic due to poor resonance delocalization of conjugate base
(D) Both have comparable acidity
28. The desiccants used for absorbing water during Liebig's method for estimation of carbon and hydrogen
are:
(A) anhydrous CaCl2 (B) anhydrous Na2SO4
(C) Mg(ClO4)2 (D) MgSO4 7H2O
VMC | Chemistry 11 Special Assignment-6 | JEE-2023
Vidyamandir Classes
29. In Lassaigne's test, the organic compound is first fused with sodium metal. The sodium metal is used
because:
(A) the melting point of sodium metal is low
(B) sodium reacts with elements of organic compound to form inorganic salts
(C) all sodium salts are water soluble
(D) sodium metal does not react with solvent water
30. Which of the following cannot be obtained as single major product in the Wurtz's coupling reaction of
halides with sodium metal?
(A) (B)
(C) (D)
31. Which of the following decarboxylation, reaction and their product is(are) incorrectly matched?
(A) (B)
(C) (D)
32. Which of the following reduction reaction and their product is(are) correctly matched?
(A) (B)
(C) (D)
33. What product(s) is(are) expected in the electrolysis of aqueous solution of potassium propanoate?
(A) Butane (B) Ethyl ethanoate
O O
|| ||
(C) Ethyl propanoate (D) CH3 − C − O − O − C − CH3
34. In the following free radical bromination reaction, the important product(s) is(are):
(A) (B) (C) (D)
35. What is(are) true regarding free radical iodination of an alkane?
(A) It occurs very rapidly due to very small value of bond enthalpy of I2
(B) Direct iodination of alkane with I2 in the presence of light is impractical
(C) Iodination of an alkane can be achieved successfully using an oxidizing agent catalyst
(D) Presence of some HCl activate the free radical iodination of alkane
VMC | Chemistry 12 Special Assignment-6 | JEE-2023
Vidyamandir Classes
36. Consider the following bromination reaction.
If a pure enantiomer of reactant is taken in the above reaction, the correct statement concerning product
dibromide is(are):
(A) A racemic mixture is formed
(B) Two optically active isomers are formed
(C) A pair of diastereomers in equal amount is formed
(D) A pair of enantiomers but in unequal amounts is formed
37. Which of the following reactions can bring about chlorination of cyclohexane?
Cyclohexane + Cl2 ⎯⎯⎯ → Cyclohexane + Cl2 ⎯⎯⎯
3→
Heat AlCl
(A) (B)
(C) Cyclohexane + SO2Cl2 ⎯⎯⎯
Heat
→ (D) Cyclohexane + HCl ⎯⎯
→
38. Which of the following reactions results in formation of an alkene?
(A) CH3 − CH2 − CHBr2 ⎯⎯⎯⎯
Zn
→ (B) CH3 − CH2 − CHBr2 ⎯⎯⎯
Ether
→
CH OH 3 Na/
Br
|
(C) CH3 − CH − CH2 Br ⎯⎯⎯⎯
Zn
CH OH
→ (D) CH2 − CH2 − CH2 ⎯⎯⎯⎯
Zn
CH3OH
→
3 | |
Br Br
39. Which of the following reaction(s) produces propene as one of the important organic product?
O
||
(A) CH3 − C − CH3 + Zn(Hg) ⎯⎯⎯ HCl
→
+
(B) CH3 − CH2 − CH2 − N(CH3 )3 Br − ⎯⎯⎯→
AgOH
(C) CH3 − CH − CH2 − COOK(aq) ⎯⎯⎯⎯⎯
Electrolysis
→
|
COOH
(D) CH3 − CHO + (C6H5 )3 P = CH2 ⎯⎯ →
40. Consider the following reaction,
The correct statement concerning product alkene(s) is(are):
(A) A single pure enantiomer of product is formed
(B) Racemic mixture of product is formed
(C) Product alkene and the starting compound have same configuration
(D) Starting compound and product have same sign of optical rotation
41. Consider the following dehydration of alcohol.
The important dehydration product(s) is(are):
(A) (B) (C) (D)
VMC | Chemistry 13 Special Assignment-6 | JEE-2023
Vidyamandir Classes
42. In which of the following reaction(s), major product is formed by the rearrangement of reaction
intermediate?
(A)
(B)
(C)
(D)
43. Consider the following reaction,
The correct statement(s) is(are):
CH3
|
(A) X is a tertiary bromide (B) Y is Ph − C C − CH2 − CH − CH3
(C) Z is
(D) Z is formed through heterogeneous catalysis reaction
44. In which of the following reactions, reactants and products are correctly matched?
(A)
(B)
(C)
(D) H − C C − H ⎯⎯⎯⎯NaNH2
Liq. NH3
→ ⎯⎯→
D2
Pt
CH2 − CH2
| |
D D
45. Which is the important reactive intermediate in the following reaction?
(A) (B) (C) (D)
46. In which of the following halogen addition reactions, racemic mixture of products is formed?
(A) (B)
(C) (D) CH3 − CH = CH2 + Br2 − CCl4 ⎯⎯
→
VMC | Chemistry 14 Special Assignment-6 | JEE-2023
Vidyamandir Classes
47. Consider the following addition reaction on a pure enantiomer of the shown bromoalkene.
What is(are) true regarding products of the above reaction?
(A) Four stereoisomers of products are formed
(B) A pair of enantiomers and a meso-dibromide are formed
(C) Only a pair of diastereomers are formed
(D) One of the products is meso-dibromide
48. In which of the following reaction(s), an alkyne product is formed?
(A) (B) CaC2 + H2O ⎯⎯
→
(C) Mg2C3 + H2O ⎯⎯
→ (D) Al4C3 + H2O ⎯⎯
→
49. Which of the following reagent gives an effervescence when reacted with 1-butyne?
(A) Na / (B) NaH (C) NaNH2 (D) CH3MgBr
50. Which of the following reagent(s) can be used to distinguish between 1-hexyne and 2-hexyne?
(A) Ammoniacal AgNO3 solution (B) Ammoniacal solution of Cu(II) tartarate
(C) Cold, dilute and alkaline KMnO4 (D) H2SO4/HgSO4
51. Terminal alkyne being thermodynamically less stable than an internal alkyne, instead when 2,
2-dibromo butane is treated with NaNH2, 1-butyne is formed as major product, not 2-butyne because:
(A) NaNH2 is a very strong base
(B) NaNH2 abstract H from less hindered carbon
(C) terminal bromo alkene is formed in first step of elimination reaction
(D) terminal alkyne forms salt with NaNH2, phased out of equilibrium
52. Predict product(s) in the following reaction.
(A) (B)
(C) (D)
53. The correct statement(s) regarding following reaction is(are):
CH2 = CH − CH = CH2 + HBr ⎯⎯ → CH2 = CH − CH − CH3 + CH2 − CH = CH − CH3
| |
Br Br
[I] [II]
(A) At –80°C I is the major product
(B) At room temperature, II is the major product
(C) Both of the products I and II are always formed in comparable amounts
(D) Product I isomerizes into II on heating above 40C
VMC | Chemistry 15 Special Assignment-6 | JEE-2023
Vidyamandir Classes
54. Predict product(s) of the following reactions.
(A) (B)
(C) (D)
55. The correct statement(s) regarding 1, 2-butadiene(I) and 1, 3-butadiene(II) is(are):
(A) I has perpendicular pi planes while II has parallel pi planes
(B) Both gives the same product on adding 1.0 equivalent of HBr at −80C
(C) I gives 2, 2-dibromobutane while II gives 2, 3-dibromobutane, when reacted with excess of
HBr at very low temperature
(D) Both have same length of C = C double bonds
56. Which of the following species is(are) aromatic?
(A) (B) (C) (D)
57. What makes the following compound aromatic?
−
(A) Add one electron to -bond to give C9H10
2−
(B) Add two electrons to -bond to give C9H10
(C) Remove an ion, H+, from sp3 carbon
(D) Remove an ion, H+, from sp2 carbon
58. Which is true about the compound calicene?
(A) It is highly soluble in water
(B) It exists mainly as dipolar ion with +ve charge in the three membered ring while –ve charge in
five membered ring
(C) It exists mainly as dipolar ion with –ve charge in the three membered ring and +ve charge in
five membered ring
(D) In solution it shows very high electrical conductivity
59. Which of the following species is(are) anti-aromatic?
(A) (B)
(C) (D)
VMC | Chemistry 16 Special Assignment-6 | JEE-2023
Vidyamandir Classes
60. What is(are) true regarding following reaction?
(A) Bromination occur above at a rate faster than at benzene
(B) Bromination occur above at rate slower than at benzene
(C) Bromination occur at ortho and para position giving para isomer as major product
(D) Bromination occur predominantly at meta position
61. What is(are) true about chlorination of toluene?
(A) In presence of light, benzyl chloride is formed
(B) In presence of AlCl3, ortho and para chlorotoluene’s are formed
(C) Once a chlorine atom is introduced into benzene ring, further chlorination at benzene ring
becomes difficult compared to first chlorination
(D) Chlorination of toluene in presence of AlCl3 is more difficult than benzene
62. Consider the following reaction,
The correct statement(s) is(are):
(A) Major bromination occur at C-2
(B) Major bromination occur at C-3
(C) Bromination at C-3 and C-4 are equally probable
(D) Bromination occur at a rate slower than that at benzene
63. The compound(s) that direct incoming electrophile predominantly at meta position is(are):
(A) (B) (C) (D)
64. The compound(s) that undergo slower electrophilic substitution than benzene is(are):
(A) (B) (C) (D)
65. Which of the following compound(s) fail in Friedel-Crafts alkylation reaction?
(A) (B) (C) (D)
66. What is(are) true about the following reaction?
(A) The major product is
(B) The reaction involves a cyclic oxonium ion intermediate
(C) The product formed is chiral
(D) If the product is further reacted with Br2 − AlBr3 , para bromination occur predominantly
VMC | Chemistry 17 Special Assignment-6 | JEE-2023
Vidyamandir Classes
67. Consider the following reaction,
What is the expected product(s)?
(A) (B)
(C) (D)
68. Consider the following reaction, C6H4Cl2 (X) + CH3Cl ⎯⎯⎯
AlCl3
→ C6H3Cl2 (CH3 ) (Y) "Single isomer".
The correct statement(s) concerning X and Y is(are):
(A) X is a non-polar dichloride isomer
(B) X is more reactive than benzene in the same reaction
(C) Y gives three isomers of dimethylation product
(D) X and Y have different positions of chlorine atoms
69. What is(are) true regarding Friedel-Crafts methylation reaction of phenol using CH3Cl/AlCl3?
(A) Methylation succeeds only if large excess of AlCl3 is taken
(B) AlCl3 reacts with phenol to give C6H5OAlCl2
(C) Methylation occurs mainly at meta position
(D) Ortho/para methylation occur with para isomer as major product
70. What is(are) the principal products of the following reaction?
(A) (B)
(C) (D)
71. Which of the following alkene cannot be prepared by controlled hydrogenation of an alkyne?
(A) (B)
(C) (D)
72. Consider the following reaction,
Which of the following is(are) true statement?
(A) Reaction initiated by protonation of − OH followed by formation of carbocation
(B) Reaction is initiated at C = C forming a tertiary carbocation
(C) Here intramolecular reaction is favoured by entropy of reaction
(D) The same reaction can also be accomplished using NaOH as catalyst
VMC | Chemistry 18 Special Assignment-6 | JEE-2023
Vidyamandir Classes
73. Which of the following reactants, reagents and products are correctly matched?
(A)
(B) H3C − C CH ⎯⎯⎯⎯⎯⎯ ⎯
Lindlar 's catalyst
→ ⎯⎯⎯
D2 /Pt
→ CH3CHDCH2D
(C) H − C C − H ⎯⎯⎯⎯Br2
1.0 equiv.
→ ⎯⎯⎯
Cl2
→ Racemic mixture
(D) C6H5 − C C − H ⎯⎯⎯
HBr
ROOR
⎯
→ C6H5 − CH = CHBr
74. Which of the following reactions, reactants and products are correctly matched?
(A) (B)
(C) (D)
75. Which of the following will evolve hydrogen gas on heating with potassium metal?
(A) (B) (C) (D)
76. What product(s) are expected when excess of benzene is treated with 3-propenol in presence of HF?
(A) 3-phenyl propene (B) 1, 3-diphenyl propane
(C) 1, 2-diphenyl propane (D) 1, 1-diphenyl propane
SECTION-3 LINK-COMPREHENSION TYPE
PARAGRAPH FOR QUESTIONS 1 - 2
Qualitative analysis of organic compound is performed by Lassaigne’s test by fusion with metallic sodium; by
which the covalent compounds are converted into ionic compounds. Extra elements like N, S, P, and halogens
are detected by their usual tests.
1. Which of the following will give blood-red colour in Lassaigne’s test for nitrogen.
(A) PhNH2 (B) PhNO2
(C) (D) PhSO3H
2. An organic compound containing N, S, and O as extra elements is fused with metallic sodium and then
extracted with distilled water. Which species is not present in the Lassaigne’s extract?
(A) NO3− (B) CN − (C) CNS− (D) S2−
PARAGRAPH FOR QUESTIONS 3 - 5
Compound (A) (C13H24O) is a sex-attractant pheromone of codling moth. On hydrogenation, compound (A)
absorbs two molar equivalents of H2 and is converted to 3-ethyl-7-methyl-decan-1-ol. On reductive ozonolysis,
it gives the following compounds.
VMC | Chemistry 19 Special Assignment-6 | JEE-2023
Vidyamandir Classes
3. (A) has two structural isomers, one isomer is:
(A1)
The other isomer (A2) is:
(A)
(B)
(C)
(D)
4. The number of stereoisomers for (A) is:
(A) 2 (B) 4 (C) 6 (D) 8
5. The product of (A) on ozonolysis followed by reduction with LAH are:
(A)
(B)
(C)
(D)
VMC | Chemistry 20 Special Assignment-6 | JEE-2023
Vidyamandir Classes
SECTION-4 COLUMN MATCHING TYPE
1. Match the reactions from column I with the properties of their products from column II.
Column I Column II
(A) (p) Forms a pair of enantiomers
(B) (q) Forms a meso compound
Proceed through a cyclic
(C) (r)
intermediate
Proceeds through a free
(D) (s)
radical intermediate
2. Match the structures in column A with the properties in Column B.
Column I Column II
Has plane of symmetry
(A) (p)
Has axis of symmetry
(B) (q)
(C) (r) Has centre of symmetry
(D) (s) Rotate plane polarized light
3. Match the following Columns:
Column I Column II
(A) CH3 − C C − CH3 (p) cis-product with H2/Pd-BaSO4
(B) CH3 − CH2 − C CH (q) Trans-product with Na/liq. NH3
(C) CH3 − C CH (r) White ppt. with amm. AgNO3
(D) CH3 − C C − Et (s) H2 gas with Na
VMC | Chemistry 21 Special Assignment-6 | JEE-2023
Vidyamandir Classes
4.
5.
6.
VMC | Chemistry 22 Special Assignment-6 | JEE-2023
Vidyamandir Classes
7.
SECTION-5 INTEGER | SUBJECTIVE TYPE
1. ( A) ⎯⎯⎯
2H2
Pt
→
Unit of unsaturation in compound (A)?
2. How many distinct monochlorinated products, (including stereoisomers) may be obtained when the
alkane shown below is heated in the presence of Cl2 ?
3. 1.216 gm of an organic compound was reacted under Kjeldahl’s method and the ammonia evolved was
absorbed in 100 ml N H2SO4. The remaining acid solution was made up to 500 ml by the addition of
water. Twenty millilitres of this dilute solution required 32 ml of N/10 caustic soda solution for
complete neutralization. Calculate the percentage of nitrogen in the compound. (Nearest Integer)
4.
Sum of molecular mass of gas (A + C) is ________.
5. P = Number of anti-aromatic compound, so the value of x is:
Q = total number of resonating structures of carbonate ion [CO32− ]
R = Number of − hydrogen in given carbocation.
S = Number of geometrical isomers of CH3 − CH = CH − CH = CH2
T = Number of compounds more acidic than CH3CH2OH
VMC | Chemistry 23 Special Assignment-6 | JEE-2023
Vidyamandir Classes
Sum of (P + Q + R + S + T) – 15 is ________.
6. Identify relationship between following pairs:
(A) (B)
(C) (D)
If they are enantiomer answer will be 1, if they are diastereomers answer will be 2, if they are
constitutional isomers answer will be 3 and if they are identical use 4 as the answer. Sum of answer of
each part A + B + C + D is ________.
7. total number of di-chloro product S-2-chloro hexane.
8. 1. 2.
3. 4.
Sum of molecular mass of A, B, C, D (i.e., A + B + C + D) is equal to ________.
9. How many carbon-hydrogen bond orbitals are available for overlap with the vacant p-orbital in ethyl
carbocation?
10. How many reagents from the following list will produce visible change when treated with propyne?
(I) H2 / Pt, Heat (II) K/ heat (III ) CH3MgBr (IV) Ag ( NH3 )+2
( V) Cu ( NH3 )+4 ( VI) Br2 − H2O ( VII) KMnO4 / HO− ( VIII) NaNH2
( IX) HCl ( X) O3 / H2O
11. How many different alkynes, on treatment with HgSO4 / H2SO4 can give 2-pentanone as one of the
product?
12. How many different cyclic isomers exist for the hydrocarbon C5H10 ?
VMC | Chemistry 24 Special Assignment-6 | JEE-2023
Vidyamandir Classes
13. If an aqueous solution containing both potassium ethanoate and potassium propanoate is subjected to
Kolbe’s electrolysis, how many products from the list below would be formed?
CH3CH3(I) CH3CH2CH3(II) CH3CH2CH2CH3(III) CH3COOC2H5(IV)
CH3CH3COOC2H5(V) CH3COOCH3(VI) CH3 CH2COOCH3(VII) CH4(VIII)
CH2 = CH2(IX) CH3CH=CHCH3(X)
14.
How many products will be formed in above reaction?
15. How many different six carbon hydrocarbons on reductive ozonolysis give the same product shown
below.
16.
17.
18.
VMC | Chemistry 25 Special Assignment-6 | JEE-2023
Vidyamandir Classes
19.
20.
21.
VMC | Chemistry 26 Special Assignment-6 | JEE-2023
Vidyamandir Classes
Answers to Special Batch Assignment-6
[Introduction to Organic Chemistry & Hydrocarbons]
SECTION-1 SINGLE CORRECT ANSWER TYPE
1 2 3 4 5 6 7 8 9 10
C A C B D C B B B C
11 12 13 14 15 16 17 18 19 20
B B B C C B C B C D
21 22 23 24 25 26 27 28 29 30
C A A C C D B D A B
31 32 33 34 35 36 37 38 39 40
C D C B A D C B C C
SECTION-2 MULTIPLE CORRECT ANSWER TYPE
1 2 3 4 5 6 7 8 9 10
ABD AB ABC AB ABD BC AC AB BCD BD
11 12 13 14 15 16 17 18 19 20
CD AD ACD BD ACD BC ABD CD CD BD
21 22 23 24 25 26 27 28 29 30
ABCD ABD AB AD ABC ACD BC AC ABC AD
31 32 33 34 35 36 37 38 39 40
AB AD AC BC BC BC AC ABC BCD AC
41 42 43 44 45 46 47 48 49 50
BCD AD BCD AB BD ACD CD BC ABCD AB
51 52 53 54 55 56 57 58 59 60
D B ABD D AC ACD C AB AD BC
61 62 63 64 65 66 67 68 69 70
ABC BD AC BCD BC ABCD ABC AC ABD BC
71 72 73 74 75 76
AC BC ABD ABC AC ACD
VMC | Chemistry 27 Special Assignment-6 | JEE-2023
Vidyamandir Classes
SECTION-3 LINK-COMPREHENSION TYPE
1 2 3 4 5
C A B B B
SECTION-4 COLUMN MATCHING TYPE
1 2 3
[A-p, r] [B-q] [C-q, r] [D-p, s] [A-p, r] [B-q, s] [C-p, q, r] [D-p] [A-p, q] [B-r, s] [C-r, s] [D-p, q]
4 5 6 7
[A-P] [B-R] [C-S] [D-Q] [A-S] [B-R] [C-P] [D-R, S] [A-Q] [B-R] [C-S] [D-P] [A-Q] [B-P] [C-S] [D)-R]
SECTION-5 INTEGER | SUBJECTIVE TYPE
1 2 3 4 5 6 7 8 9 10
9 8 23 90 4 13 9 312 3 8
11 12 13 14 15
2 7 9 4 2
VMC | Chemistry 28 Special Assignment-6 | JEE-2023
Vidyamandir Classes
[Introduction to Organic Chemistry & Hydrocarbons]
Name : ___________________________________ Roll No.
SECTION-1 SINGLE CORRECT ANSWER TYPE
1 2 3 4 5 6 7 8 9 10
11 12 13 14 15 16 17 18 19 20
21 22 23 24 25 26 27 28 29 30
31 32 33 34 35 36 37 38 39 40
SECTION-2 MULTIPLE CORRECT ANSWER TYPE
1 2 3 4 5 6 7 8 9 10
11 12 13 14 15 16 17 18 19 20
21 22 23 24 25 26 27 28 29 30
31 32 33 34 35 36 37 38 39 40
41 42 43 44 45 46 47 48 49 50
51 52 53 54 55 56 57 58 59 60
61 62 63 64 65 66 67 68 69 70
71 72 73 74 75 76
VMC | Chemistry 29 Special Assignment-6 | JEE-2023
Vidyamandir Classes
SECTION-3 LINK-COMPREHENSION TYPE
1 2 3 4 5
SECTION-4 COLUMN MATCHING TYPE
SECTION-5 Numerical Value Type (Nearest Integer)
1 2 3 4 5 6 7 8 9 10
11 12 13 14 15
VMC | Chemistry 30 Special Assignment-6 | JEE-2023
You might also like
- ACA-2B Full Organic Chemistry Class (11+12) (63 Questions+Answers)Document15 pagesACA-2B Full Organic Chemistry Class (11+12) (63 Questions+Answers)Biswajit GhoshNo ratings yet
- Carbonyl CompoundsDocument34 pagesCarbonyl CompoundsprinceNo ratings yet
- Edited - Class Test-1Document6 pagesEdited - Class Test-1Dhruv Kumar BansalNo ratings yet
- ProjectDocument17 pagesProjectrudrashah4777No ratings yet
- Home Assignment-4Document66 pagesHome Assignment-4ansh guptaNo ratings yet
- Practice Paper - Phase 3Document6 pagesPractice Paper - Phase 3Dhairya VinayakNo ratings yet
- Alkene DPPDocument20 pagesAlkene DPPKalyan ReddtNo ratings yet
- OrganicDocument9 pagesOrganicjitesh100kushwahaNo ratings yet
- Carbon and Its Compounds WSDocument7 pagesCarbon and Its Compounds WSShankar Balajee SambasivamNo ratings yet
- Homework-9 SolutionsDocument9 pagesHomework-9 SolutionsbishoppairisdeadNo ratings yet
- Guided Plan-6 (E)Document7 pagesGuided Plan-6 (E)abhiraw30062005No ratings yet
- Alkyl Halide PDFDocument6 pagesAlkyl Halide PDFTvisha ViraniNo ratings yet
- Organic Chemistry: Daily Practice ProblemsDocument10 pagesOrganic Chemistry: Daily Practice ProblemsRaju SinghNo ratings yet
- CHM207Document7 pagesCHM207Aqilah NajwaNo ratings yet
- Alkene Exercise Eng Module-4Document21 pagesAlkene Exercise Eng Module-4Raju SinghNo ratings yet
- JEE 2024-Fortnightly Test-9 - PaperDocument17 pagesJEE 2024-Fortnightly Test-9 - PaperAayush NagpalNo ratings yet
- Class Test-1-Aldehydes & Ketones - PreparationDocument5 pagesClass Test-1-Aldehydes & Ketones - PreparationSarthak VermaNo ratings yet
- Basic Concept in Org. Chem - Exercise-Eng Module-4Document25 pagesBasic Concept in Org. Chem - Exercise-Eng Module-4Raju SinghNo ratings yet
- Diwali Assignment 13thDocument8 pagesDiwali Assignment 13thRaju SinghNo ratings yet
- Date Planned: - / - / - Daily Tutorial Sheet-10 Expected Duration: 90 Min Actual Date of Attempt: - / - / - Level-2 Exact DurationDocument2 pagesDate Planned: - / - / - Daily Tutorial Sheet-10 Expected Duration: 90 Min Actual Date of Attempt: - / - / - Level-2 Exact DurationVIDYA SRI GANESHNo ratings yet
- Sample Paper Chem3333333333333333333Document1 pageSample Paper Chem3333333333333333333maria b chackoNo ratings yet
- Answer Assignment-04 On Hydrocarbon (Alkane, Alkene & Alkynes)Document2 pagesAnswer Assignment-04 On Hydrocarbon (Alkane, Alkene & Alkynes)mew090506No ratings yet
- Home Assignment-3Document32 pagesHome Assignment-3ansh guptaNo ratings yet
- Guided Plan-5 (E)Document4 pagesGuided Plan-5 (E)abhiraw30062005No ratings yet
- HydrocarbonsDocument116 pagesHydrocarbonsabhisheksingh27zxNo ratings yet
- Homework 9Document8 pagesHomework 9bishoppairisdeadNo ratings yet
- GOC Rev Ex 6Document3 pagesGOC Rev Ex 6mitsuhaNo ratings yet
- STEP Chemistry 2021 Practice Book - MDCAT by Sidhu-1Document238 pagesSTEP Chemistry 2021 Practice Book - MDCAT by Sidhu-1lailasafdar01No ratings yet
- Goc + IsomerismDocument5 pagesGoc + IsomerismRohail HussainNo ratings yet
- DPP GoccccDocument10 pagesDPP GoccccMayur Khichi0% (1)
- Date Planned: - / - / - Daily Tutorial Sheet-9 Expected Duration: 90 Min Actual Date of Attempt: - / - / - Level-2 Exact DurationDocument1 pageDate Planned: - / - / - Daily Tutorial Sheet-9 Expected Duration: 90 Min Actual Date of Attempt: - / - / - Level-2 Exact DurationVIDYA SRI GANESHNo ratings yet
- GRP #05Document4 pagesGRP #05rajkaran3765No ratings yet
- Nsec National Standard Examination in Chemistry: Class: Xi DATE: 22.11.2020Document13 pagesNsec National Standard Examination in Chemistry: Class: Xi DATE: 22.11.2020KritikaNo ratings yet
- Oc Taas Quiz 1 StudentDocument2 pagesOc Taas Quiz 1 Studentattackerasp1234No ratings yet
- Organic+Dpps A1-A14 PDFDocument43 pagesOrganic+Dpps A1-A14 PDFAditya ChakraniNo ratings yet
- DPP (22 To 25) 12th OcDocument25 pagesDPP (22 To 25) 12th OcRaju SinghNo ratings yet
- Isomerism Level Wise Practice Sheet by Mr. Dhirendra Kumar For Class 11th ChemistryDocument7 pagesIsomerism Level Wise Practice Sheet by Mr. Dhirendra Kumar For Class 11th ChemistryManoj SisodiaNo ratings yet
- Halogen Derivatives (13th)Document31 pagesHalogen Derivatives (13th)Raju SinghNo ratings yet
- Xii Chemistry Practice Test - 16-10-2021Document9 pagesXii Chemistry Practice Test - 16-10-2021Manoj RacerNo ratings yet
- Chem WS HydrocarbonDocument7 pagesChem WS HydrocarbonshrutiNo ratings yet
- Hydrocarbon (SCH Sir)Document5 pagesHydrocarbon (SCH Sir)ArthGadaNo ratings yet
- GOC (13th)Document34 pagesGOC (13th)Raju SinghNo ratings yet
- Vibrant Academy: (India) Private LimitedDocument6 pagesVibrant Academy: (India) Private LimitedRk ChaudharyNo ratings yet
- 40 Questions Organic JEE Mains 2022 14 JuneDocument51 pages40 Questions Organic JEE Mains 2022 14 JuneAyush RanjaNNo ratings yet
- JEE (Adv) 2012 - Paper-1 - Q +solnDocument8 pagesJEE (Adv) 2012 - Paper-1 - Q +solnJanmedra Kumar TripathyNo ratings yet
- Carbanion and Other Intermediate DPPDocument9 pagesCarbanion and Other Intermediate DPPJeetNo ratings yet
- Chemistry Advanced Level Problem Solving (ALPS-6) - PaperDocument14 pagesChemistry Advanced Level Problem Solving (ALPS-6) - PaperAnanmay ChauhanNo ratings yet
- Neral Organic Chemistry (62-80)Document19 pagesNeral Organic Chemistry (62-80)udaysrinivasNo ratings yet
- Previous Years Iit - Jee Questions: General Organic ChemistryDocument8 pagesPrevious Years Iit - Jee Questions: General Organic ChemistryudaysrinivasNo ratings yet
- Chemistry - Mains2 (Entire 11th)Document7 pagesChemistry - Mains2 (Entire 11th)Ravi Kiran KoduriNo ratings yet
- Class 12 Chemistry Sample Paper 01Document15 pagesClass 12 Chemistry Sample Paper 01milanraj9148No ratings yet
- ChTest (Set1) - D06 Dec 2023Document12 pagesChTest (Set1) - D06 Dec 2023Rishi ParmarNo ratings yet
- ChTest (Set1) - D06 Dec 2023Document12 pagesChTest (Set1) - D06 Dec 2023Rishi ParmarNo ratings yet
- Alkyl Halide: Organic ChemistryDocument56 pagesAlkyl Halide: Organic ChemistryAaryan KeshanNo ratings yet
- Marking Scheme: Single Correct (+3,-1) M M: 140 Time: 1 HR 30 MinDocument8 pagesMarking Scheme: Single Correct (+3,-1) M M: 140 Time: 1 HR 30 Minarryan keshanNo ratings yet
- Unit Test 4 NeetDocument5 pagesUnit Test 4 Neetmanan10jas1529No ratings yet
- GOC & EAS CPP-II - PMDDocument14 pagesGOC & EAS CPP-II - PMDVansh sareenNo ratings yet
- Goc Till Isomerism DPPDocument16 pagesGoc Till Isomerism DPPpranav.padhle6No ratings yet
- Solution Manual for The Elements of Polymer Science and EngineeringFrom EverandSolution Manual for The Elements of Polymer Science and EngineeringRating: 4 out of 5 stars4/5 (3)
- MAS 10 CirclesDocument14 pagesMAS 10 Circleslash73752No ratings yet
- 2.2 Indian WeaversDocument1 page2.2 Indian Weaverslash73752No ratings yet
- 19-01-21 SR - Iit Jee-Main SPCL Gtm-1 QPDocument20 pages19-01-21 SR - Iit Jee-Main SPCL Gtm-1 QPlash73752No ratings yet
- Test On Areas 19-09-2020Document5 pagesTest On Areas 19-09-2020lash73752No ratings yet
- Practical - 2: Preparation of The FixativeDocument14 pagesPractical - 2: Preparation of The FixativeIseth ISethNo ratings yet
- Marxism and The Oppression of Women Lise VogelDocument259 pagesMarxism and The Oppression of Women Lise VogelMoises SaavedraNo ratings yet
- Soccer Training DiaryDocument1 pageSoccer Training DiaryMark DeaconNo ratings yet
- FP Lecture Midterm Exam Sec - Sem.2020Document4 pagesFP Lecture Midterm Exam Sec - Sem.2020SAEEDA ALMUQAHWINo ratings yet
- Benefits of OTN in Transport SDNDocument9 pagesBenefits of OTN in Transport SDNGhallab AlsadehNo ratings yet
- Osce05ans 110918053819 Phpapp01Document20 pagesOsce05ans 110918053819 Phpapp01masood alamNo ratings yet
- Exploded View & Parts Listing Air Operated Double Diaphragm PumpDocument2 pagesExploded View & Parts Listing Air Operated Double Diaphragm PumpCarlos AvalosNo ratings yet
- DLP Physical Science Week1Document2 pagesDLP Physical Science Week1gizellen galvezNo ratings yet
- 3592 Operator GuideDocument103 pages3592 Operator GuideNaim GhattasNo ratings yet
- Design of CEB BuildingDocument20 pagesDesign of CEB BuildingVishalya Nipuni Lankeshi100% (1)
- Farmhouse Style Plans - Farm & CountryDocument6 pagesFarmhouse Style Plans - Farm & Countryhanif azriNo ratings yet
- Bitsat Paper 5Document19 pagesBitsat Paper 5pranka5240100% (1)
- Assignment 1 - Statistics ProbabilityDocument3 pagesAssignment 1 - Statistics ProbabilityAzel Fume100% (1)
- Sandvik Saf 31803 Tube and Pipe, Seamless: DatasheetDocument9 pagesSandvik Saf 31803 Tube and Pipe, Seamless: DatasheetPaul NeedhamNo ratings yet
- Medical CodingDocument5 pagesMedical CodingBernard Paul GuintoNo ratings yet
- ForewordDocument96 pagesForewordkkcmNo ratings yet
- Unit-1 Infancy: S.Dharaneeshwari. 1MSC - Home Science-Food &nutritionDocument16 pagesUnit-1 Infancy: S.Dharaneeshwari. 1MSC - Home Science-Food &nutritionDharaneeshwari Siva-F&NNo ratings yet
- Penetration Test & Softening PointDocument7 pagesPenetration Test & Softening PointAli M. Chehadeh100% (4)
- Notice - Appeal Process List of Appeal Panel (Final 12.1.24)Document13 pagesNotice - Appeal Process List of Appeal Panel (Final 12.1.24)FyBerri InkNo ratings yet
- Introduction To Kalman FilterDocument4 pagesIntroduction To Kalman FilterArghya MukherjeeNo ratings yet
- Conceptual FrameworkDocument3 pagesConceptual Frameworkprodiejigs36No ratings yet
- Pelton2014 Para-Equilibrium Phase DiagramsDocument7 pagesPelton2014 Para-Equilibrium Phase DiagramsAbraham Becerra AranedaNo ratings yet
- 02-09-18 SR - IZ Jee-Adv 2011-P1 PTA-03 Q PDFDocument31 pages02-09-18 SR - IZ Jee-Adv 2011-P1 PTA-03 Q PDFswarupNo ratings yet
- Qa/Qc Mechanical Monthly Progress Report For June 2015: Area/System Description Status RemarksDocument1 pageQa/Qc Mechanical Monthly Progress Report For June 2015: Area/System Description Status RemarksRen SalazarNo ratings yet
- Effect of Vino Gano Ginger and Herbal Liquor On The Heamatological Parameters of The Wistar RatsDocument5 pagesEffect of Vino Gano Ginger and Herbal Liquor On The Heamatological Parameters of The Wistar RatsInternational Journal of Innovative Science and Research TechnologyNo ratings yet
- Pertanyaan TK PDBDocument4 pagesPertanyaan TK PDBHardenNo ratings yet
- Lecture 1 1489485680Document52 pagesLecture 1 1489485680Dato TevzadzeNo ratings yet
- American Pile Driving Equipment Equipment CatalogDocument25 pagesAmerican Pile Driving Equipment Equipment CatalogW Morales100% (1)
- Section 08630 Metal-Framed SkylightDocument4 pagesSection 08630 Metal-Framed SkylightMØhãmmed ØwięsNo ratings yet
- EU - Guidance On GMP For Food Contact Plastic Materials and Articles (60p)Document60 pagesEU - Guidance On GMP For Food Contact Plastic Materials and Articles (60p)Kram NawkNo ratings yet