Professional Documents
Culture Documents
I Kuselman Humanerrorsandqualityofchemicalanalyticalresults-Chem Int 2015
Uploaded by
P SRINIVAS RAOOriginal Title
Copyright
Available Formats
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
Available Formats
I Kuselman Humanerrorsandqualityofchemicalanalyticalresults-Chem Int 2015
Uploaded by
P SRINIVAS RAOCopyright:
Available Formats
See discussions, stats, and author profiles for this publication at: https://www.researchgate.
net/publication/276418061
Human Errors and Quality of Chemical Analytical Results
Article in Chemistry International · May 2015
DOI: 10.1515/ci-2015-0324
CITATIONS READS
3 589
1 author:
Ilya Kuselman
190 PUBLICATIONS 1,739 CITATIONS
SEE PROFILE
All content following this page was uploaded by Ilya Kuselman on 18 May 2015.
The user has requested enhancement of the downloaded file.
Reports from recent conferences and symposia
See also www.iupac.org/home/conferences.html
Section head
Conference NEW
Call
Human Errors and Quality of cycle: principles of quality of analytical results and de-
cisions which can be made on their base”. The lecture
Chemical Analytical Results described a logically structured approach consisting of
a closer examination of the customers´ needs by map-
by Ilya Kuselman ping them onto the performance characteristics of the
anticipated analytical procedure. This approach may be
An International Workshop on Human Errors and Quality regarded as a “measurement cycle” if it is followed up
of Chemical Analytical Results was held 13 January 2015, to the actual usage of measurement results in the deci-
in Kfar Maccabiah, Israel, as a milestone of IUPAC project sion process of the customer. More reliable decisions are
2014-027-1-500. An earlier workshop on human error reached by reducing the size of the guard band around a
was held in Israel in 2013 to discuss experiences classify- specification limit where no decisions can realistically be
ing and modeling human error accumulated in aviation, made, i.e. reducing the measurement uncertainty. An op-
medicine and other fields, which could be helpful in ana- timization of the analytical procedure may be necessary,
lytical chemistry, as well [1]. The idea of the current work- even resorting to an alternative measurement principle
shop arose from the quality management system ap- based on the quality-by-design methodology.
proach. Applied for routine chemical analytical (testing) Another keynote lecture “Metrology and quality
laboratories, this approach provides continual improve- of test results: documents of the Joint Committee for
ment of a laboratory quality system. One of the improve- Guides in Metrology (JCGM)” was delivered by Dr. Wal-
ment targets is the prevention of human error. However, ter Bich, Istituto Nazionale di Ricerca Metrologica (IN-
human activity is never free from error: they are the root RIM), Italy. Dr. Bich discussed the publications of JCGM,
cause of the majority of incidents and accidents. In ana- in particular the guides for measurement uncertainty
lytical chemistry, human error may lead to atypical test evaluation. The scope of the new revision of the guide
results of questionable reliability [2]. There are, for ex- to the expression of uncertainty in measurement (JCGM
ample, test results that fall outside the established speci- 100) and a separate collection of examples (JCGM 110)
fications in the pharmaceutical industry, or that do not attracted the attention of the workshop participants. It
comply with regulatory, legislative or specification limits was important to hear that methods for the evaluation
in other industries and fields, such as environmental and of measurement uncertainty will be agreed upon and ad-
food analysis. When an atypical test result is identified, it opted world-wide. This implies that the methods will be
is important to determine the root cause of the event and universal, in the sense that they can be useful in any ap-
to avoid recurrence of such results. Where no limits have plication, including analytical chemistry.
yet been established (e.g., for an environmental object Dr. Raphael Bar, BR Consulting, Israel, provoked a
or a new material), human error may lead to an incor- discussion by his lecture “Should the pharmaceutical
rect evaluation of the tested property. Thus, a study of laboratory report test results with uncertainty?” Dr. Bar
human error is necessary in any field of analytical chem- said that the current regulatory requirement in a phar-
istry. A laboratory demonstrating competence in analyti- maceutical laboratory is only the validation of analytical
cal chemistry and conformity assessment should also be methods. Yet, a typical certificate of analysis of a drug
able to develop relevant, human error-related corrective substance or product shows no uncertainty of the test
and preventive actions [3]. results. The new USP-suggested approach for validating
The workshop was organized by the Israel Analytical analytical methods will discuss measurement uncertainty
Chemistry Society (IACS) with the participation of the evaluation in the validation process.
Israel Laboratory Accreditation Authority (ISRAC), in co- Dr. Kuselman’s lecture reported on the progress of
operation with the International Union of Pure and Ap- IUPAC projects 2012-021-1-500 and 2014-027-1-500, as
plied Chemistry (IUPAC), Cooperation on International well as quantification of human error in an analytical lab-
Traceability in Analytical Chemistry (CITAC), and A Fo- oratory based on expert judgments. Examples were pro-
cus for Analytical Chemistry in Europe (Eurachem). The vided using earlier published sets of expert judgments
event was sponsored by Sigma-Aldrich Corporation and on human error in pH measurement of groundwater [3],
arranged by Bioforum Ltd. multi-residue analysis of pesticides in fruits and vegeta-
Opening remarks were given by Dr. Ilya Kuselman, bles [4], and elemental analysis of geological samples
Modiin, Israel, Chair of the Workshop Organizing Com- by inductively coupled plasma mass spectrometry [5].
mittee, and Prof. Wolfhard Wegscheider, the University Evaluation of the residual risk of human error remaining
of Leoben, Austria, Chair of Eurachem. Prof. Wegscheider after the error reduction by the laboratory quality sys-
then delivered the keynote lecture “The measurement tem and the risk influence on the quality of the analytical
30 CHEMISTRY International May-June 2015
Unauthenticated
Download Date | 5/18/15 9:17 AM
Conference Call
results and associated measurement uncertainty were problem is that Type A evaluation is defined as the meth-
discussed [6]. od of evaluation by statistical analysis. However, Type B
Monte Carlo simulation of expert behaviour in quan- evaluation is also based on rectangular, triangular or an-
tification of human error was the subject of the lecture other distribution of observations, in spite of the defini-
by Dr. Francesca Pennecchi, INRIM, Italy. Any expert is a tion in JCGM 100 and JCGM 200 as evaluation by means
human being whose judgments are influenced by intra- other than statistical analysis. Dr. Bich explained the his-
personal conflicts and other factors. Therefore, an evalu- torical reasons for that problem, and noted that such a
ation of the robustness of the error quantification scores classification of methods for uncertainty evaluation will
to the doubts of an expert is required. To that aim, a Mon- not be used in the new JCGM 100 issue under devel-
te Carlo simulation of expert judgments on human error opment. Dr. Kuselman briefly presented the position of
was used for determining the distributions of the error the task group of the IUPAC projects on the possibility
quantification scores. An expert judgment, represented of taking human error into account as a component of
by a choice on the scale (0, 1, 3, 9), is a discrete quantity measurement uncertainty. While gross errors are easily
characterized by a probability mass function (pmf). Ap- identifiable and corresponding results can be separated
propriate pmfs were considered in order to model confi- from the data set, small human errors are in principle not
dent, reasonably doubting, or irresolute expert behavior, distinguishable from other components of measurement
respectively. An R code was developed for the random uncertainty. Therefore, an uncertainty budget is not com-
generation of expert judgments as discrete values and plete when consequences of possible human error are
the propagation of the considered pmfs according to the not evaluated as a contribution to the budget. Quantify-
Monte Carlo approach [5]. ing residual risks of human error allows such evaluations
After these lectures Dr. Michela Sega, INRIM, Italy, to be made.
moderated the round-table discussion “Can human error The second half of the workshop day began with the
be taken into account as a component of measurement lecture by Dr. Kuselman “Knowledge is Power (Francis
uncertainty?” First, she proposed that the participants Bacon 1597): comparability concept and global metrol-
ask questions to the lecturers. The question posed to Dr. ogy system”. The lecture was dedicated to knowledge-
Bich was “how can one explain the difference between based mistakes, which are more frequent than other
Type A and Type B evaluations of measurement uncer- kinds of human error and have significant severity in dif-
tainty, as well as the benefit of this classification?” The ferent scenarios. On the other hand, these mistakes are
A group of the lecturers. From left to right: Prof. Wolfhard Wegscheider, Dr. Walter Bich,
Dr. Ilya Kuselman, Dr. Michela Sega, and Dr. Francesca Pennecchi.
CHEMISTRY International May-June 2015 31
Unauthenticated
Download Date | 5/18/15 9:17 AM
Conference Call
the simplest to prevent. The lecturer directed the atten- moderated by Prof. Emil Bashkansky, ORT Braude Col-
tion of the participants to the global metrology system lege, Israel, again allowed participants to receive an-
and publications in this system containing knowledge swers to their questions to the lecturers. Dr. Kuselman
important for chemical analytical laboratories. There are presented the scores for evaluation of the quality system
guidelines, recommendations and documents of the In- effectiveness developed by the task group of the IUPAC
ternational Bureau of Weights and Measures (BIPM), the projects. Prof. Bashkansky reported about his matrix
International Organization of Legal Metrology (OIML), approach to human error problems in engineering. The
the International Organization for Standardization (ISO), opinions on the effectiveness of a laboratory quality sys-
the International Laboratory Accreditation Cooperation tem against human error were diverse, but the majority
(ILAC), the National Conference of Standard Laborato- of the participants agreed with the optimistic position of
ries – International (NCSLI), IUPAC, CITAC, Eurachem and the IUPAC project task group.
others. It was a very fruitful, interesting, and pleasurable
Ms. Erica Pinco, ISRAC, Israel, reported on the im- meeting. Some participants expressed their wish to con-
provement of error management in accredited medical tinue the traditional biannual workshop, and supported
laboratories in the country. The identification and con- further development of the topic of human error and
trol of nonconformities of test results are a part of the quality in an analytical chemical laboratory.
quality management system, based on ISO 15189. Those The next two days, 14-15 January, the workshop lec-
results that are not in line with specifications may indi- turers and participants took part in the Isranalytica 2015
cate a medical condition, when in fact they result from Conference and Exhibition. A summary of these events is
human error. When the management identifies that the available at http://bioforumconf.com/isranalytica15 .
irregularities of the results are due to human error, a set
of activities including corrective actions should be taken References
in order to reduce the error probability. 1. Kuselman I, Fajgelj A (2013) Human errors and out-
Dr. Shula Levin, Waters (TC) Ltd, Israel, impressed of specification test results. Chem Int 35(3):30-31
the workshop participants with the lecture “Distinguish- 2. Kuselman I, Pennecchi F, Fajgelj A, Karpov Yu
ing between human error and instrumental malfunction (2013) Human errors and reliability of test results
in HPLC”. HPLC instrumentation and software have ad- in analytical chemistry. Accred Qual Assur 18:3-9
vanced significantly in recent years and have become 3. Kuselman I, Kardash E, Bashkansky E, Pennecchi
technologically sophisticated. Therefore, the task of F, Ellison SLR, Ginsbury K, Epstein M, Fajgelj A,
distinguishing between human error and system mal- Karpov Yu (2013) House of security approach
function has become more and more challenging, and to measurement in analytical chemistry: quanti-
requires collaboration between service engineers and fication of human error using expert judgments.
analytical chemists. Dr. Levin presented examples of Accred Qual Assur 18:459-467
investigations during which there were real instrument 4. Kuselman I, Goldshlag P, Pennecchi F (2014)
malfunction vs. human error; recommendations for mini- Scenarios of human errors and their quantification
mal details of the HPLC method which must be speci- in multi-residue analysis of pesticides in fruits and
fied in a standard operation procedure; and some useful vegetables. Accred Qual Assur 19:361-369
Chromatographic Data Systems’ modules and reference 5. Kuselman I, Pennecchi F, Epstein M, Fajgelj A,
chemical kits, which can reduce human error. Ellison SLR (2014) Monte Carlo simulation of
“Can human error be reduced in a pharmaceutical expert judgments on human errors in chemi-
laboratory?” asked Dr. Orna Dreazen of Nextar Chemp- cal analysis – a case study of ICP-MS. Talanta
harma Solutions Ltd, Israel, in her lecture. Human error, 130:462-469
said Dr. Dreazen, is the main source of unnecessary ac- 6. Kuselman I, Pennecchi F (2015) Human errors and
tivities in a laboratory, wasting energy and resources. In measurement uncertainty. Metrologia 52:238-243
the worst case, human error may cause an adverse effect
and the recall of pharmaceutical products. GMP, GLP and Dr. Ilya Kuselman <ilya.kuselman@gmail.com> worked at the National
ISO 13485 contain requirements designed to reduce hu- Physical Laboratory of Israel and is a member of the IUPAC Interdivisional
man error. The lecturer analyzed examples from a phar- Working Party on Harmonization of Quality Assurance.
maceutical laboratory and discussed corrective and pre-
ventive actions for error reduction. www.iupac.org/project/2014-027-1-500
Finally, a round-table discussion “Is a laboratory qual-
ity assurance system effective against human errors?”
32 CHEMISTRY International May-June 2015
Unauthenticated
Download Date | 5/18/15 9:17 AM
View publication stats
You might also like
- To Err Is Human A Case Study of Error Prevention in Process IsolationsDocument6 pagesTo Err Is Human A Case Study of Error Prevention in Process Isolationsairbuk doeingNo ratings yet
- Obsolescence Risk Assessment Process Best Practice-REDDocument11 pagesObsolescence Risk Assessment Process Best Practice-REDLORENZO RAMIREZ MATEOSNo ratings yet
- CDVR CommissioningDocument47 pagesCDVR CommissioningZamani MahdiNo ratings yet
- Introduction To The ASTM E3106 Standard Guide To Science-Based and Risk-Based Cleaning Process Developmentand ValidationDocument14 pagesIntroduction To The ASTM E3106 Standard Guide To Science-Based and Risk-Based Cleaning Process Developmentand Validationnsk79in100% (1)
- Peer Reviewed: Case StudyDocument11 pagesPeer Reviewed: Case StudyPiruzi MaghlakelidzeNo ratings yet
- 9G Visa Process MapDocument1 page9G Visa Process MapJohn Lawrence de lunaNo ratings yet
- Application of Design of Experiment (DOE) Techniques To Process Validation in Medical Device ManufactureDocument10 pagesApplication of Design of Experiment (DOE) Techniques To Process Validation in Medical Device ManufactureCamila MatheusNo ratings yet
- Handbook of Investigation and Effective CAPA SystemsFrom EverandHandbook of Investigation and Effective CAPA SystemsNo ratings yet
- What Causes Most Errors in Chemical AnalysisDocument2 pagesWhat Causes Most Errors in Chemical AnalysisImmerNo ratings yet
- Analysis of Laboratory Critical Value Reporting at A Large Academic Medical CenterDocument7 pagesAnalysis of Laboratory Critical Value Reporting at A Large Academic Medical CenterLevi GasparNo ratings yet
- Designing Aseptic Process SimulationsDocument13 pagesDesigning Aseptic Process SimulationsAmi AfiyatiNo ratings yet
- Technical Report Measurement Uncertainty 2007Document62 pagesTechnical Report Measurement Uncertainty 2007yousaf_zai_khan81995No ratings yet
- RiskAnalysisPreview PDFDocument28 pagesRiskAnalysisPreview PDFFrancesco De BellisNo ratings yet
- 10.1515 - CCLM 2015 0303Document4 pages10.1515 - CCLM 2015 0303adrianaNo ratings yet
- A Journey Through Quality ControlDocument20 pagesA Journey Through Quality Controlivan sekiddeNo ratings yet
- Lab Audit1Document5 pagesLab Audit1Yagna SubrmanianNo ratings yet
- Cleaning Process Development and Validation: Science, Risk and Statistics-BasedDocument12 pagesCleaning Process Development and Validation: Science, Risk and Statistics-BasedHami KeserciNo ratings yet
- The Shirokizawa Matrix Determining The Level of Effort Formality Documentation in Cleaning ValidationDocument7 pagesThe Shirokizawa Matrix Determining The Level of Effort Formality Documentation in Cleaning ValidationGESLIN JOSE BRUNO CHAVEZNo ratings yet
- Soriano Et Al. 2019 - Reliability - Unidad IDocument10 pagesSoriano Et Al. 2019 - Reliability - Unidad IaaNo ratings yet
- Evaluation Human Errorin Control RoomDocument6 pagesEvaluation Human Errorin Control RoomJesús SarriaNo ratings yet
- FMEA - Improving Quality Risk Manag During Qualific, Validation, and Change ControlDocument15 pagesFMEA - Improving Quality Risk Manag During Qualific, Validation, and Change ControlLuis Gustavo PachecoNo ratings yet
- A Failure Mode and Effect Analysis FMEA-based ApprDocument12 pagesA Failure Mode and Effect Analysis FMEA-based ApprTrần Thanh TiếnNo ratings yet
- Analytical Performance Specifications in Clinical ChemistryDocument12 pagesAnalytical Performance Specifications in Clinical ChemistrymohammedNo ratings yet
- 10.1515 - CCLM 2016 0929Document10 pages10.1515 - CCLM 2016 0929adrianaNo ratings yet
- Harmonized Guidelines For Quality ControlDocument18 pagesHarmonized Guidelines For Quality ControlJosé Esqueda LeyvaNo ratings yet
- Mistakes in A Stat Laboratory: Types and Frequency: Clinical Chemistry September 1997Document5 pagesMistakes in A Stat Laboratory: Types and Frequency: Clinical Chemistry September 1997Othman MaaiNo ratings yet
- Best Practices For Conducting An RCADocument10 pagesBest Practices For Conducting An RCAzabuawadNo ratings yet
- A Matrix Model For Phlebotomy Quality AssuranceDocument15 pagesA Matrix Model For Phlebotomy Quality AssuranceandhiNo ratings yet
- Standards 02 00014 v2Document8 pagesStandards 02 00014 v2ANOBA LABNo ratings yet
- PRPOSALDocument7 pagesPRPOSALAhmedAhmedNo ratings yet
- Some Practical Examples of Method Validation in The Analytical LaboratoryDocument10 pagesSome Practical Examples of Method Validation in The Analytical LaboratoryImad AghilaNo ratings yet
- Simulated Clinical TrialsDocument32 pagesSimulated Clinical TrialsAnand SrinivasanNo ratings yet
- Safety and Health at Work: Belky P. Castaño, Vladimir Ramírez, Julio A. CanceladoDocument7 pagesSafety and Health at Work: Belky P. Castaño, Vladimir Ramírez, Julio A. CanceladodiegoNo ratings yet
- Obsolescence Risk Assessment Process Best PracticeDocument12 pagesObsolescence Risk Assessment Process Best Practiceعلي الاسمريNo ratings yet
- Quality Assurance and Quality Control in Neutron Activation Analysis: A Guide to Practical ApproachesFrom EverandQuality Assurance and Quality Control in Neutron Activation Analysis: A Guide to Practical ApproachesNo ratings yet
- TG40Document39 pagesTG40jiar001No ratings yet
- Risk Assessment and Quality Control - QCNetDocument3 pagesRisk Assessment and Quality Control - QCNetSuryana MamanNo ratings yet
- Sci-Com SSC Out252 enDocument81 pagesSci-Com SSC Out252 enKangan KhannaNo ratings yet
- Análisis de Errores Humanos en Un Sistema de Permiso de Trabajo Un Estudio de Caso en Una Planta QuímicaDocument6 pagesAnálisis de Errores Humanos en Un Sistema de Permiso de Trabajo Un Estudio de Caso en Una Planta QuímicaNESTOR DURANNo ratings yet
- Analysing The Shape of An OC Curve For An AcceptanDocument10 pagesAnalysing The Shape of An OC Curve For An AcceptanAnh Tran Thi VanNo ratings yet
- Metrological Traceability of Test Results: An Essential Practice That Must Be Adhered ToDocument3 pagesMetrological Traceability of Test Results: An Essential Practice That Must Be Adhered ToWang YehanNo ratings yet
- Process Analytical Technology Concepts and Principles PDFDocument8 pagesProcess Analytical Technology Concepts and Principles PDFhdmnauNo ratings yet
- Incertidumbre Rev MEx Pat Clin 19Document7 pagesIncertidumbre Rev MEx Pat Clin 19Lorena Ek MaciasNo ratings yet
- Assessment of Visual Function in Glaucoma - A Report by The American Academy of OphthalmologyDocument17 pagesAssessment of Visual Function in Glaucoma - A Report by The American Academy of OphthalmologyJose Antonio Fuentes VegaNo ratings yet
- Three Years' Experience of Quality Monitoring Program On Pre-Analytical Errors in ChinaDocument7 pagesThree Years' Experience of Quality Monitoring Program On Pre-Analytical Errors in ChinaCenyiqanita NurqanitaNo ratings yet
- ISO-GUM and Supplements Are Utilized For QA of BCA Data: Yasuo IwakiDocument31 pagesISO-GUM and Supplements Are Utilized For QA of BCA Data: Yasuo IwakiKemo TufoNo ratings yet
- Chapter 2Document14 pagesChapter 2AdityaDhanrajSinghNo ratings yet
- A Process-Based Approach in Failure Analysis: EditorialDocument3 pagesA Process-Based Approach in Failure Analysis: EditorialNoven PutraNo ratings yet
- Review: Harmonization of Pre-Analytical Quality IndicatorsDocument9 pagesReview: Harmonization of Pre-Analytical Quality IndicatorsadrianaNo ratings yet
- Practical Aspects of Cosmetic Testing: How to Set up a Scientific Study in Skin PhysiologyFrom EverandPractical Aspects of Cosmetic Testing: How to Set up a Scientific Study in Skin PhysiologyJoachim W. FluhrNo ratings yet
- ISO 9001 2015 Standard Implementation in Clinical Trial CentersDocument4 pagesISO 9001 2015 Standard Implementation in Clinical Trial CentersBRYAN GEORGENo ratings yet
- Detecting GMP FailuresDocument7 pagesDetecting GMP Failuresbsetiawan0% (1)
- Computer Aided Process EngineeringDocument7 pagesComputer Aided Process EngineeringIndah Fitriany PurwaningtyasNo ratings yet
- Sampling Waters Flow and LoadDocument316 pagesSampling Waters Flow and LoadPauline Anne WilsonNo ratings yet
- Integration of HOFs Into ATEX Risk AssesDocument6 pagesIntegration of HOFs Into ATEX Risk AssesNurettin TERZİOĞLUNo ratings yet
- Validation of AnalyticalDocument307 pagesValidation of AnalyticalJagdish ChanderNo ratings yet
- Project Quality Case StudyDocument8 pagesProject Quality Case StudySlawkterDeEspritoSlawkterNo ratings yet
- JOURNAL Errors in Clinical Laboratories or Errors in LaboratoryDocument11 pagesJOURNAL Errors in Clinical Laboratories or Errors in LaboratoryVincent ReyesNo ratings yet
- Risk Considerations For Installation of A New Autoclave in A Pharmaceutical Manufacturing FacilityDocument29 pagesRisk Considerations For Installation of A New Autoclave in A Pharmaceutical Manufacturing Facilitygadhang dewanggaNo ratings yet
- Plebani Etal 2014Document8 pagesPlebani Etal 2014vivi maykasariNo ratings yet
- Comparison Usp 1790 Nf41 6 and Nf41 1Document27 pagesComparison Usp 1790 Nf41 6 and Nf41 1Apar DholakiaNo ratings yet
- Proposed New USP General Chapter: The Analytical Procedure LifecycleDocument9 pagesProposed New USP General Chapter: The Analytical Procedure LifecycleLeonardo GiraldoNo ratings yet
- CFA Level 2 - 2020 Curriculum Changes (300hours) PDFDocument1 pageCFA Level 2 - 2020 Curriculum Changes (300hours) PDFJOAONo ratings yet
- BS BuzzDocument8 pagesBS BuzzBS Central, Inc. "The Buzz"No ratings yet
- PCST - MODULE - 0.2 Process Control Systems Technician ProgramDocument15 pagesPCST - MODULE - 0.2 Process Control Systems Technician Programkali bangonNo ratings yet
- OpenSSH - 7.4 Multiple Vulnerabilities - Nessus - InfosecMatterDocument1 pageOpenSSH - 7.4 Multiple Vulnerabilities - Nessus - InfosecMatterdrgyyNo ratings yet
- Lesson 8-BridgesDocument2 pagesLesson 8-BridgesMohamed MraouiNo ratings yet
- Cylinder: Data Sheet No. 2.29.001E-1Document10 pagesCylinder: Data Sheet No. 2.29.001E-1sancsa_74No ratings yet
- Pineapple Extract For Skin Care: FacialsDocument2 pagesPineapple Extract For Skin Care: Facialsrobustauger1567No ratings yet
- Saeed Zaman CVDocument2 pagesSaeed Zaman CVSheeraz Ali khakiNo ratings yet
- Hazard Register: Type Make Model Location Sale Number Lot Number Serial NumberDocument3 pagesHazard Register: Type Make Model Location Sale Number Lot Number Serial NumberShawn WairisalNo ratings yet
- Material Safety Data Sheet: 1 Chemical Product and Company Identification FENOL LFR (6015-6020-6025-6035)Document5 pagesMaterial Safety Data Sheet: 1 Chemical Product and Company Identification FENOL LFR (6015-6020-6025-6035)Zirve PolimerNo ratings yet
- SIG-DE-01 Especificaciones Cargaor LONKING CDM858Document2 pagesSIG-DE-01 Especificaciones Cargaor LONKING CDM858Jorge PalacioNo ratings yet
- Honeywell 5800CO-Installation-InstructionsDocument4 pagesHoneywell 5800CO-Installation-InstructionsIzah FriendsNo ratings yet
- VIP 32 Hybrid VentDocument8 pagesVIP 32 Hybrid VentsagarNo ratings yet
- Welding ParametersDocument34 pagesWelding ParametersmilindNo ratings yet
- Performance RubricsDocument2 pagesPerformance RubricsMaria Manoa GantalaNo ratings yet
- Aircom - UMTS Advanced Cell Planning and Optimisation PS-TR-005-O036 - v5.0Document292 pagesAircom - UMTS Advanced Cell Planning and Optimisation PS-TR-005-O036 - v5.0Igor MalianovNo ratings yet
- Catalog Tranzistori NPN de PutereDocument4 pagesCatalog Tranzistori NPN de PutereSmigun CorneliuNo ratings yet
- Chest Tubes Nursing CareDocument2 pagesChest Tubes Nursing CareYousef KhalifaNo ratings yet
- Mangayar Malar Recipes - 01-09-2013 - Tamil MagazinesDocument4 pagesMangayar Malar Recipes - 01-09-2013 - Tamil MagazinesiyappangokulNo ratings yet
- Name GroupDocument4 pagesName GroupAmit KashikarNo ratings yet
- Solution Manual For Mis Cases Decision Making 4th Edition by MillerDocument8 pagesSolution Manual For Mis Cases Decision Making 4th Edition by MillerElizabethSteelefocgk100% (81)
- VariationDocument6 pagesVariationPSYCHO / FLOWNo ratings yet
- Pattern Making SyllabusDocument5 pagesPattern Making Syllabusapi-4617069890% (1)
- Managing Risk: Jean B. Ganub, RPHDocument32 pagesManaging Risk: Jean B. Ganub, RPHJean GanubNo ratings yet
- PACER US Vs Adam Hageman Complaint - Nov 12 2020Document5 pagesPACER US Vs Adam Hageman Complaint - Nov 12 2020jpeppardNo ratings yet
- Toyo - Soal Dasar Akuntansi UTS - Gasal 2122Document2 pagesToyo - Soal Dasar Akuntansi UTS - Gasal 2122Dita nNo ratings yet
- Datacard150i ManualDocument2 pagesDatacard150i Manualb00sey0% (1)
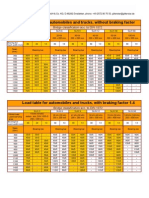
- 9 Gitterstar LoadTable Autom TrucksDocument1 page9 Gitterstar LoadTable Autom TrucksSnaz_nedainNo ratings yet