Professional Documents
Culture Documents
Anatomy Histo Physio DR - Adil
Uploaded by
Hybat Elsheikh0 ratings0% found this document useful (0 votes)
16 views73 pagesThis document provides information on the anatomy, histology, and physiology of the skin, mucous membranes, and keratinization. It discusses the structure and layers of the epidermis, including the stratum basale, stratum spinosum, and stratum granulosum. It also describes keratinocyte differentiation and keratin gene expression during epidermal development and renewal.
Original Description:
Original Title
Anatomy Histo Physio Dr.adil Copy
Copyright
© © All Rights Reserved
Available Formats
PPTX, PDF, TXT or read online from Scribd
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentThis document provides information on the anatomy, histology, and physiology of the skin, mucous membranes, and keratinization. It discusses the structure and layers of the epidermis, including the stratum basale, stratum spinosum, and stratum granulosum. It also describes keratinocyte differentiation and keratin gene expression during epidermal development and renewal.
Copyright:
© All Rights Reserved
Available Formats
Download as PPTX, PDF, TXT or read online from Scribd
0 ratings0% found this document useful (0 votes)
16 views73 pagesAnatomy Histo Physio DR - Adil
Uploaded by
Hybat ElsheikhThis document provides information on the anatomy, histology, and physiology of the skin, mucous membranes, and keratinization. It discusses the structure and layers of the epidermis, including the stratum basale, stratum spinosum, and stratum granulosum. It also describes keratinocyte differentiation and keratin gene expression during epidermal development and renewal.
Copyright:
© All Rights Reserved
Available Formats
Download as PPTX, PDF, TXT or read online from Scribd
You are on page 1of 73
Anatomy, Histology and
Physiology of the skin,
mucous membranes
& Keratinization.
Dr. ADIL HAMID HASSAN
BASHIR, MD
DERMATOLOGIST.
orcid-org/0000-0003-4232-3601
researchgate.net
• Skin is the largest organ in human body.
• It has a surface area 1.5-2 m square.
• Weight about 15% of total body weight.
• It measures a body mass 3.6 Kg.
• Skin receives a blood flow 462ml/min.
• Arterio-venous O2 difference 25ml/L.
• O2 consumption 12ml/min.
• It has a resistance 11.7 R units (Absolute).
• The integument is composed of the skin,
which covers the entire body, in addition to
accessory organs derived from skin. The
accessory organs include the nails, hair,
and glands of various kinds.
• The skin is considered the largest organ of
the body and has many different functions.
• Skin serves many important functions:
(1) It is an impervious barrier that excludes harmful
substances and prevents desiccation;
(2) it plays an important role in the regulation of body
temperature;
(3) it readily repairs itself;
(4) it receives sensory stimuli (touch, pressure,
temperature, and pain);
(5) sweat glands excrete waste products;
(6) lacrimal glands produce an isotonic saline bathing
solution for the eyes;
(7) sebaceous and ceruminous glands secrete sebum
and cerumen ("wax"), respectively; and
(8) mammary glands secrete milk.
• The skin is divided into two main regions, the
epidermis, and the dermis, each providing a
distinct role in the overall function of the skin.
The dermis is attached to an underlying
hypodermis, also called subcutaneous
connective tissue, which stores adipose tissue
and is recognized as the superficial fascia of
gross anatomy.
• The epidermis is the most superficial layer of
the skin and provides the first barrier of
protection from the invasion of foreign
substances into the body. The principal cell of
the epidermis is called a keratinocyte.
Cell kinetics
• Are complicated in the epidermis by the
balance between growth with differentiation and
cell death.
• A major concept is that of turnover time, which
is the amount of time for the whole cell
population to replace itself (regeneration time or
replacement time).
• The cell cycle or intermitotic time (Tc)
represents the interval between two successive
mitoses (M).
Epidermal differentiation and keratin gene
expression
• The epidermis of the skin is a stratified
squamous epithelium, which plays an important
protective role. It manifests this role by building
an extensive cytoskeletal architecture, the
unique feature of which is the presence of
keratin filaments. There are two major pairs
of keratins in the epidermis: one pair is
expressed in dividing cells and the other
expressed in terminally differentiating cells.
As such, keratins provide useful biochemical
markers to explore the molecular mechanisms
underlying the balance between growth and
differentiation in the epidermis
Epidermal differentiation and keratin gene
expression.
• Epidermis, a continuously self-renewing and
differentiating organ, produces a protective
stratum corneum that shields us from external
chemical, physical and microbial threats.
Epidermal differentiation is a multi-step process
regulated by influences, some unknown, others
insufficiently explored. Detachment of
keratinocytes from the basement membrane is
one such pro-differentiation stimulus.
The regulation of epidermal differentiation
• In skin development, several signaling
pathways such as Hedgehog, Wnt and
Transforming Growth Factor- β (TGF-β) have
been implicated.
• Three main classes of transcription factors,
AP1, AP2 and Sp1,appear to be relevant.
• Regulation of the Nuclear transcription Factor
NF-κB also appears to be significant in the
process of terminal differentiation in epidermis.
• Terminal differentiation is also influenced by Retinoids
that act at the mRNA level to inhibit aspects of
differentiation and to promote proliferation.
Growth stimulatory signals
family
• Epidermal Growth Factor (EGF) Stimulates
cell proliferation and differentiation in a wide
range of tissues.
• TGF-α, Synthesized by epidermal keratinocytes
and stimulates keratinocyte growth.
• interleukin-1 (IL-1), IL-6 and Granulocyte–
Macrophage Colony-Stimulating Factor (GM-
CSF) can also stimulate growth.
• Paracrine factors stimulate keratinocyte
growth.
Growth inhibitors for keratinocytes
• Epidermal growth is under the influence of a
negative-feedback mechanism.
• TGF-β present in normal cells and malignant
cell lines, stimulates fibroblast growth and
increases fibrosis, but inhibits growth of
keratinocytes.
• Interferon-α (IFN-α) and IFN-γ have cytostatic
effects on keratinocytes.
• Apoptosis programmed cell death.
•
Differentiation
Terminal differentiation of the epidermis in vivo
• As the epidermal keratinocytes move through
the epidermis after losing their attachment to
the basal lamina, they undergo the complex
process of terminal differentiation to produce
the stratum corneum. stratum spinosum have
lost this ability to divide, and are inevitably
committed to differentiation, stratum corneum
cells (squames or corneocytes) have lost their
nuclei and other recognizable organelles, and
comprise 65% insoluble cysteine-rich,
disulphide cross-linked, fibrous proteins or
keratins.
• keratin filaments aggregate into bundles
through the action of the histidine-rich basic
protein filaggrin is the process of
keratinization, epidermal differentiation
involves the synthesis of a highly insoluble
cornified envelope whose cross-linking is
catalysed by a keratinocyte specific
transglutaminase from a precursor protein
called involucrin, intercellular adhesion
proteins (integrins), cell-matrix adhesion
proteins and blood-group antigens.
Terminal differentiation is also influenced by several other
factors including expression of desmosome proteins and
signaling pathways such as Notch or phosphatidylinositol 3
kinase, extracellular matrix protein 1 (ECM1) , vitamin D
receptors, FGF10, cyclo-oxygenase enzymes, calcium-sensing
receptors, sodium channels and small heat shock protein.
Keratin biochemistry, synthesis and changes in epidermis.
• Simple epithelia are characterized by the keratin pair
K8/K18, and the stratified squamous epithelia by
K5/K14.
• In skin, suprabasal keratins K1/K10 are characteristic
of epidermal differentiation. In the stratum
granulosum, release of filaggrin from the
keratohyalin granules forms macrofibres.
• K19 is expressed in basal keratinocytes of the
hair-follicle bulge region at the site of
pluripotential stem cells. K9 and K2e
expression are site restricted in skin: K9 to
palmoplantar epidermis and K2e to superficial
interfollicular epidermis.
• epidermal hyperproliferation, as in wound
healing and psoriasis, expression of suprabasal
keratins K6/ K16/K17 is rapidly induced
suprabasally, probably due to previous
expression of their mRNAs with
posttranscriptional regulation.
Hair and nail differentiation
• The pilosebaceous units develop from epidermal down
growths under the influence of specific mesenchymal cell
condensations between the 10th and 14th week estimated
gestational age. They have complex groups of specialized cell
layers with distinctive pathways of differentiation.
• There are four classes of pilosebaceous unit: terminal on the
scalp and beard; apopilosebaceous in axilla and groin; vellus
on the majority of skin; and sebaceous on the chest, back and
face. The dermal papilla is located at the base of the hair follicle
with a rich ECM. Around the papilla are germinative (matrix)
cells that have a very high rate of division, and give rise to
spindle-shaped central cortex cells of the hair fiber, and the
single outer layer of flattened overlapping cuticle cells.
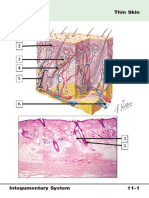
• The epidermis is 0.8 to 1.4 mm in thickness,
devoid of blood vessels but is nourished by
diffusion from capillaries in the underlying dermis
and subdivided into five layers or strata,
the stratum germinativum (SG)-basale,
the stratum spinosum(SS)-Prickle cell layer, the
stratum granulosum (SGR)-granular layer,
the stratum lucidum and the stratum corneum
(SC) in which a keratinocyte gradually migrates to
the surface and is sloughed off in a process called
desquamation.
• The strata basale and spinosum are also referred
to as the Malpighian layer.
• Three additional cell types are found in the
epidermis: (1) melanocytes, (2) Langerhans cells,
and (3) Merkel's cells.
Stratum germinativum, basale,
Malpighian.
• The stratum germinatum (SG) provides the
germinal (proliferative) cells necessary for the
regeneration of the layers of the epidermis,
constitute a single layer of columnar or cuboidal
cells in contact with the basement membrane
and connective tissue of the dermis. After a
mitotic division a newly formed cell will undergo
a progressive maturation called keratinization
(differentiation) as it migrates to the surface.
Stratum Spinosum, prickle cell
layer.
• The cells that divide in the stratum
germinativum soon begin to differentiate
into polyhedral cells, accumulate many
desmosomes on their outer surface which
provide the characteristic “prickles” of the
stratum spinosum (SS), which is often
called the prickle-cell layer.
Stratum granulosum
• The stratum granulosum is composed of a layer
of three to five cells in thick skin and 2-3 in thin
skin that contains keratohyalin granules of
irregular shape that stain with basic dyes,
contain lipids, which along with the desmosomal
connections help to form a waterproof barrier
(lipid barrier) that functions to prevent fluid loss
from the body (Trans-epidermal water loss).
• The progressive maturation of a keratinocyte is
characterized by the accumulation of keratin,
called keratinization.
Keratohyalin granules
• highly distinctive cellular inclusions in
differentiating epidermis forming the
characteristic granules of the stratum
granulosum. found in other cornifying epithelia,
such as palate and gingiva, but not in non-
cornified squamous epithelia. Filaggrin is
derived from a high-molecular-weight precursor
profilaggrin, which is insoluble, cannot
aggregate filaments and is highly
phosphorylated. The degradation of filaggrin
produces amino acids that help to retain
stratum corneum water.
Stratum Lucidum
• The stratum lucidum consists of a tightly packed
layer of cells without nuclei containing a
refractile substance called eleidin. This layer is
strongly eosinophilic.
• Epidermis varies in thickness throughout the
body depending mainly on frictional forces and
is thickest on the palms of the hands and soles
of the feet. The stratum lucidum is normally only
well seen in thick epidermis and represents a
transition from the stratum granulosum to the
stratum corneum, and act as an immunological
layer.
Stratum corneum
• The most superficial layer, the stratum corneum, is
composed of many dead cells without nuclei, which are
filled with keratin. As a cell accumulates keratinohyalin
granules, it is thought that rupture of lysosomal
membranes release lysosomal enzymes that eventually
cause cell death. The dead and dying cells filled with
mature keratin form the stratum corneum (SC)-fully
differentiated cell (corneocyte). The deeper cells of
the stratum corneum retain their desmosomal junctions,
but as they are pushed to the surface by newly forming
cells of the stratum germinativum (SG), the dead cells
gradually break apart and are lost, a process called
desquamation.
The cornified envelope
• highly insoluble by the formation of glutamyl-
lysysl isodipeptide bonds between envelope
proteins, catalysed by transglutaminases.
• There are three distinct transglutaminases:
• TG1 (keratinocyte
transglutaminase),Involucrin
The dermo epidermal junction
• One of the largest epithelial-mesenchymal junctions in
the body. It forms extensive interface between the
dermis and epidermis.
• Have a key role in a wide range of epithelial
mesenchymal interactions including:
*Epidermal cell anchorage, adhesion, migration and
differentiation. Signaling between the extracellular matrix
and basal cells, and serves as a barrier and filter.
Basement membranes allow migrating cells to
pass under physiological conditions, but act as
barriers against tumor cell invasion and regulate
the passage of molecules based on their
electronic charge and molecular size.
It encompasses:
• The basal plasma membrane of keratinocytes.
Melanocytes, Merkel’s cells and
Hemidesmosomes.
Immediately beneath:
• Basement Membrane Zone (BMZ), 0.5-1.0mm-
thick, which consists of three layers:
• Lamina Lucida, Lamina Densa, and Sub Lamina
Densa (fibroreticularis).
• Lamina densa is observable as an
electron-dense layer.
• lamina lucida is proposed to be an artifact
resulting from rapid dehydration during tissue
processing.
• The skin BMZ can be divided into four
ultrastructurally distinct areas: the
hemidesmosome/upper lamina lucida, the lower
lamina lucida, the lamina densa, and the sub-
lamina densa. Well-characterized BMZ
components include the hemidesmosome/upper
lamina lucida-located bullous pemphigoid
antigens (BP230 and BP180), a6b4 integrin, and
plectin; the lower lamina lucida-located laminin-1,
laminin-5 (previously named kalinin, epiligrin,
nicein, BM600), laminin-6 (previously named k-
laminin), p105, and entactin/nidogen; the lamina
densa-located type IV collagen and perlecan; and
the sub-lamina densa-located type VII collagen
(epidermolysis bullosa acquisita antigen).
Mucosal BMZ also contain identical components
as skin BMZ.
• basement membranes, consists of a number of
collagenous and non-collagenous
macromolecules.
• Type IV collagen is the main structural
constituent of the basement membrane-
immunolocalized mainly to the lamina densa.
• The type IV collagen molecule is able to
interact with laminin, nidogen, heparan sulphate
proteoglycan (PG), BM-40/SPARC and β1
integrin. Immunolabelling of normal human skin
with an antitype IV collagen antibody.
• Type VII collagen was recognized that crystallites of
SLS collagen were similar to anchoring fibrils.
• The laminins an α chain, β chain, and γ chain.
laminins 1, 2, 5, 6 and 10 are known to occur in
epidermal basement membrane.
• Nidogen key stabilizer of basement membrane,
binding to type IV collagen, perlecan and
fibulins.
• Perlecan is the main PG of basement
membranes, binds to Nidogen, dystroglycan
and ECM1. Its functions include stabilization of
basement membrane and regulating basement
membrane synthesis and turnover.
The hemidesmosome - anchoring filament-
anchoring fibril adhesion complex
• Hemidesmosomes are thought to form a
continuous link between the intracellular keratin
filament network and the extracellular
basement membrane.
• hemidesmosomes are important not only in
adhesion but also as a pathway for signal
transduction between the extracellular and
intracellular compartments.
• Major intracellular (plaque) components of the
hemidesmosome include the 230 kDa bullous
pemphigoid antigen (BP230 or BPAG1) and
plectin.
• basement membrane synthesis and turnover through
binding cytokines and growth factors. The major
component involved in adhesion between basal cells
and ECM.
• The α6β4 integrin and the 180-kDa bullous
pemphigoid antigen (BP180, BPAG2 or collagen type
XVII) are both transmembrane components of
hemidesmosomes.
• The anchoring filament associated proteins include
laminin 5, laminin 6, the extracellular segment of type
XVII collagen, the linear IgA antigen, LAD-1 (which
constitutes part of the extracellular domain of type
XVII collagen and the as yet unknown antigen
recognized by 19-DEJ-1 monoclonal antibody .
• Laminin 5 (previously known as nicein, kalinin
or epiligrin)
Dermis (corium)
• The dermis (D), 0.3 to 4.0 mm in thickness, assumes the
important functions of thermoregulation and supports the
vascular network to supply the avascular epidermis with
nutrients. The dermis is typically subdivided into two
zones, a papillary dermis includes the ridges and
papillae and a reticular layer. The dermis contains
mostly fibroblasts which are responsible for secreting
collagen, elastin and ground substance that give the
support and elasticity of the skin. Also present are
immune cells that are involved in defense against foreign
invaders passing through the epidermis.
Papillary dermis
• The papillary layer includes the ridges and papillae,
which protrude between the epidermal pegs. The
papillae contain tactile corpuscles of Meissner's and
small blood vessels. The papillary dermis (PD) contains
vascular networks that have two important functions. The
first being to support the avascular epidermis with vital
nutrients and secondly to provide a network for
thermoregulation. The vasculature is organized so that
by increasing or decreasing blood flow, heat can either
be conserved or dissipated. The vasculature
interdigitates in areas called dermal papillae (DP). The
papillary dermis also contains the free sensory nerve
endings and structures called Meissner’s corpuscles in
highly sensitive areas.
Reticular dermis
• The reticular layer of the dermis (RD) consists of
dense irregular connective tissue, which differs
from the papillary layer (PD), which is made up,
of mainly loose connective tissue (note the
difference in the number of cells). The reticular
layer of the dermis is important in giving the skin
it overall strength and elasticity, as well as
housing other important epithelial derived
structures such as glands and hair follicles.
Subcutaneous Layer (Panniculus
Adiposus)
• This layer, not part of the skin, is also called
superficial fascia and is a loose network of
connective tissue bundles and septa, which
blend indistinctly with the dermis. This
arrangement of the connective tissue of the
superficial fascia allows the movement of skin
except on the palm of the hand and sole of the
foot, where the skin is firmly anchored to deeper
structures. In most places, particularly the
abdominal wall, lobules of fat may be abundant;
the layer may then also be called the panniculus
adiposus.
Skin Appendages
• The skin contains a variety of
appendages, mainly hair follicles (HF),
sweat glands , and sebaceous glands
(SG), which are all embrylogically
epidermal in origin.
• In most parts of the body, such as
abdominal skin, the keratin layer is thin. Thick
keratin is most prominent on the soles and
palms. In this abdominal skin, the basal layer of
epidermal cells has faint pigmentation, and the
basement membrane can be seen beneath the
basal layer. The difference between the thin,
outer papillary dermis and the broader, deeper
reticular dermis is subtle. A granular layer with
prominent keratohyaline granules is often not
distinct in normal skin, except where keratin is
present in large amounts.
• Melanocytes appear
within the basal layer as cells with clear cytoplas
m.
They exist in a ratio of 1-to-10 with the basal
keratinocytes, i.e., one melanocyte for every ten
basal cells (every 6). In the above diagrams, the
melanocytes are shown in light blue (for clarity);
under normal circumstances they are not
pigmented. Although melanocytes do produce
the brown pigment, melanin, they transfer it to
neighboring epidermal cells and do not normally
store it in visible amounts.
Histopathological keys
• Histopathological examination of a skin
biopsy taken from an appropriate lesion is
a great help in assisting the clinician to
come to a correct diagnosis, and therefore
come to a decision regarding management
of the patient.
1. Acanthosis:
Increase in number of cells in the Malpighian or prickle cell
layer of the epidermis.
Features may be seen:
-Naevoid conditions.
-Virally induced papillomas.
-Chronic inflammatory conditions, for example Chronic
eczema, psoriasis.
- Pemphigus, Darier's disease.
-Malignant melanoma, Paget’s disease, Mycosis Fungoids
and Histiocytosis X.
*Pseudoacanthosis; increase thickness of the epidermis
due to enlarged keratinocytes.
*Pseudoepitheliomatous hyperplasia; reactive epidermal
proliferation
2. Acantholysis is the term used to describe loss of
cohesion between keratinocytes, due to breakdown of
intercellular bridges.
• Primary acantholysis:
• Pemphigus and its variants.
• Darier's disease.
• Transient and persistent acantholytic dermatosis.
• Warty dyskeratoma.
• Secondary acantholysis:
• Benign familial Pemphigus (Hailey-Hailey disease).
• Bullous impetigo.
• Viral disorders.
• Solar keratoses.
• Squamous cell carcinoma (some forms).
3. Dyskeratosis
• This term relates to some abnormality in
the process of epidermal cell
keratinization.
The process occurs in two main contexts:
• Firstly, in malignant and premalignant
epithelial lesions, such as
• Squamous cell carcinoma,
• Bowen's disease and
• Solar keratosis.
• Secondly, in various forms of acantholytic disorder such as
• Darier's disease.
In this condition, specific types of dyskeratotic cell include corps ronds
and grains.
• Benign familial Pemphigus (Hailey-Hailey disease).
• Transient and persistent acantholytic dermatosis.
• Warty dyskeratoma.
• Solitary dyskeratoma.
• Acantholytic dyskeratotic epidermal naevus
• (Grover’s disease)- focal Acantholytic dyskeratosis.
• Porokeratosis of Mantoux.
• Condylamata accuminata of uterine cervix.
• Chronic radiodermatitis.
• Graft Versus Host disease.
• Necrolytic migratory erythema with carcinoma of pancreas.
• Familial dyskeratotic comedones.
• Hereditary benign intraepidermal dyskeratosis
• (Witkop- Von Sallman syndrome)- focal Acantholytic dyskeratosis.
4. Grenz zone:
A zone of normal dermis between the epidermis and
pathological changes deeper in the dermis. This
comes from the German word for border.
• Lepromatous leprosy.
• Leukocytoclastic vasculitis.
• Early stage 1ry CTCL.
• Histiocytosis; Fat- storing hamartoma of dermal
dendrocytes.
• Melanocytic Naevi and Malignant Melanoma.
5. Hypergranulosis:
Increase in thickness of the granular layer of
the epidermis.
-Chronic lichenification, and lichen planus
(commonly).
6. Hyperkeratosis:
Increased thickness of the stratum corneum.
-Keratodermas.
-Icthyotic disorders.
-Chronic Discoid Lupus Erythematosus.
- Kyrle’s disease.
• 7. Necrolysis:
The separation of tissue constituents as a
consequence on cell death.
Epidermal changes of Necrolysis are seen in
various inflammatory reactions such as:
• Erythema multiforme, EM,
• Toxic epidermal Necrolysis, TEN and
• Necrolytic migratory erythema seen in
association with the glucagonoma
syndrome.
8. Papillomatosis:
Elongation upwards of the dermal papillae,
giving an accentuated and sometimes
irregular, undulating configuration to the
dermo-epidermal junction.
The feature is commonly seen in:
*Viral warts (Verrucae).
*Pemphigus vegetans.
9. Parakeratosis:
The retention of keratinocyte nuclei within the horny
cell layer,
it represents a:
• Disturbance of keratinization, and
• Is normally associated with an absence or
reduction in thickness of the granular cell layer.
• Closely associated either with:
* Increased epidermal cell turnover or
* with inflammatory changes in the epidermis itself.
It is commonly seen in:
-An earlier disturbance in the underlying
epidermis such as:
• Psoriasis,
• Subacute eczematous reactions, and
• Pityriasis lichenoides.
• -Dysplastic epithelial changes, such as:
*Actinic keratoses and
* Bowen's disease.
• 10. Spongiosis ( Intercellular edema):
-Widening of the intercellular spaces between
keratinocytes due to fluid accumulation.
*Acute and Subacute eczema (characteristically).
(1) Langerhans cells in the epidermis
(2) Lymphocytes in the epidermis
(3) Spongiosis
(4) Hyperkeratosis & parakeratosis
(5) Elongated rete ridges
(6) Lymphocytes surrounding dermal vessels
You might also like
- Integumentary System: Stratum BasaleDocument21 pagesIntegumentary System: Stratum BasalerinaNo ratings yet
- Meth Elab Topic N1Document23 pagesMeth Elab Topic N1Group M1869No ratings yet
- Histology by - Dr.abdirahman Gagajir 1Document40 pagesHistology by - Dr.abdirahman Gagajir 1HusseinNo ratings yet
- 1st Lecture On The Histology of Skin by Dr. RoomiDocument14 pages1st Lecture On The Histology of Skin by Dr. RoomiMudassar RoomiNo ratings yet
- The 3 Layers of Skin: Epidermis, Dermis and Subcutaneous FatDocument11 pagesThe 3 Layers of Skin: Epidermis, Dermis and Subcutaneous FatPanggih Sekar Palupi IINo ratings yet
- Keratinocytes Undergo A Differentiation Process, Starting From A Single Layer of Stem Cells at The Basis ofDocument14 pagesKeratinocytes Undergo A Differentiation Process, Starting From A Single Layer of Stem Cells at The Basis ofHarish KakraniNo ratings yet
- Dermatology Questions and AnsDocument147 pagesDermatology Questions and AnsEl FaroukNo ratings yet
- Lecture 27 - Histology of The Integumentary System 2Document89 pagesLecture 27 - Histology of The Integumentary System 2spitzmark2030No ratings yet
- Integumentary System - Pptx-For Fundamentals of Zoology 2020Document25 pagesIntegumentary System - Pptx-For Fundamentals of Zoology 2020Islam SamirNo ratings yet
- Keratinisation and CornificationDocument38 pagesKeratinisation and Cornificationlizamjen100% (3)
- Vertebrates'Integumentary System 2021 UpdatedDocument62 pagesVertebrates'Integumentary System 2021 UpdatedLumanlan ArcadioNo ratings yet
- Structure of Skin SeminarDocument65 pagesStructure of Skin SeminarVikas PurushottamNo ratings yet
- Skin Organ Structure and FunctionsDocument20 pagesSkin Organ Structure and FunctionsDr. Mary MjeedNo ratings yet
- Semisolid Dosage FormsDocument200 pagesSemisolid Dosage Formsved.g007100% (8)
- Lecture Presented by:-ALI Waheed /yasmin Falah /zainab Mazin Supervised By:-Dr. Sattar Jabbar Jasim /cell and Tissue BiomedicalDocument24 pagesLecture Presented by:-ALI Waheed /yasmin Falah /zainab Mazin Supervised By:-Dr. Sattar Jabbar Jasim /cell and Tissue BiomedicalAli Waheed jolan100% (1)
- Core Curriculum WOCNSDocument1,212 pagesCore Curriculum WOCNSJerry MaguireNo ratings yet
- 1,2-Introduction To DermatologyDocument20 pages1,2-Introduction To Dermatologyhesham attiaNo ratings yet
- Intug SystemDocument42 pagesIntug SystemdrkumaranNo ratings yet
- Anatomy, Skin (Integument), Epidermis - StatPearls - NCBI BookshelfDocument8 pagesAnatomy, Skin (Integument), Epidermis - StatPearls - NCBI Bookshelfpka25No ratings yet
- Skin LayersDocument3 pagesSkin LayersCrackmcat CrackmcatNo ratings yet
- Skin AnatomyDocument71 pagesSkin AnatomyHardik DaveNo ratings yet
- Integumentary SystemDocument24 pagesIntegumentary SystemWadabiNo ratings yet
- Anatomy and PhysiologyDocument25 pagesAnatomy and PhysiologyNaidin Catherine De Guzman-AlcalaNo ratings yet
- "Vision Without Action Is A Daydream. Action Without Vision Is A Nightmare." Japanese ProverbDocument22 pages"Vision Without Action Is A Daydream. Action Without Vision Is A Nightmare." Japanese Proverb123456100% (1)
- Integumentary System: Dr. Abdul Ahad ShaikhDocument55 pagesIntegumentary System: Dr. Abdul Ahad ShaikhPharmacist DinaNo ratings yet
- Characteristics of Living Organisms and The Integumentary SystemDocument11 pagesCharacteristics of Living Organisms and The Integumentary SystemSHYRE COLEEN CATAYAONo ratings yet
- Library DissertationDocument201 pagesLibrary Dissertationhaneefmdf100% (2)
- M13 v2 Dermatology & STI - LO3Document8 pagesM13 v2 Dermatology & STI - LO3medipro clinicNo ratings yet
- The Integumentary System: Protection, Temperature Regulation & SensationDocument97 pagesThe Integumentary System: Protection, Temperature Regulation & SensationPrynz Tumaliuan MalsiNo ratings yet
- Chapter-1: 1.1 Anatomy of SkinDocument59 pagesChapter-1: 1.1 Anatomy of SkinLydia Elezabeth AlappatNo ratings yet
- Anatomy and Physiology of The SkinDocument30 pagesAnatomy and Physiology of The SkinNancy VargasNo ratings yet
- Skin Structure and Function: Applied Dermatotoxicology. DOI: © 2014 Elsevier Inc. All Rights ReservedDocument10 pagesSkin Structure and Function: Applied Dermatotoxicology. DOI: © 2014 Elsevier Inc. All Rights ReservedArsene AngelaNo ratings yet
- Null (1) (Autosaved)Document82 pagesNull (1) (Autosaved)SamNo ratings yet
- Oral Mucous MembraneDocument71 pagesOral Mucous MembraneHarleen GrewalNo ratings yet
- Online Practice Tests, Live Classes, Tutoring, Study Guides Q&A, Premium Content and MoreDocument43 pagesOnline Practice Tests, Live Classes, Tutoring, Study Guides Q&A, Premium Content and MoreYoAmoNYCNo ratings yet
- Insect IntegumentDocument31 pagesInsect IntegumentUbaid Khan50% (4)
- Human Anatomy 5Th Edition Saladin Solutions Manual Full Chapter PDFDocument28 pagesHuman Anatomy 5Th Edition Saladin Solutions Manual Full Chapter PDFdora.ivy892100% (16)
- Human Anatomy 5th Edition Saladin Solutions Manual 1Document36 pagesHuman Anatomy 5th Edition Saladin Solutions Manual 1donnawugnwsjrzcxt100% (22)
- Human Anatomy 5th Edition Saladin Solutions Manual 1Document7 pagesHuman Anatomy 5th Edition Saladin Solutions Manual 1katherine100% (40)
- Learning Outcomes:: Layers of The SkinDocument4 pagesLearning Outcomes:: Layers of The SkinRumaisa ChowdhuryNo ratings yet
- Histology of Integument: Structure and Function of Skin and Its AppendagesDocument50 pagesHistology of Integument: Structure and Function of Skin and Its AppendagesMsatriaNo ratings yet
- ReviewerDocument15 pagesReviewerYza Grace S. PayangdoNo ratings yet
- The anatomy and functions of human skinDocument9 pagesThe anatomy and functions of human skinmaNo ratings yet
- Skin 2020Document60 pagesSkin 2020Yousef KhallafNo ratings yet
- Anatomy and Physiology of The Skin.3Document11 pagesAnatomy and Physiology of The Skin.3alinaNo ratings yet
- 1 SS Skin Cancer - Chapter 1Document12 pages1 SS Skin Cancer - Chapter 1Muhammad IqbalNo ratings yet
- Physiology of Integumentary SystemDocument38 pagesPhysiology of Integumentary SystemAnisa AnisatusholihahNo ratings yet
- 2. Connective TissueDocument63 pages2. Connective TissueJeniNo ratings yet
- Histology SkinDocument5 pagesHistology SkinFahd Abdullah Al-refaiNo ratings yet
- Histology Structure of SkinDocument4 pagesHistology Structure of SkinLIEBERKHUNNo ratings yet
- Policy For B.ed HonsDocument67 pagesPolicy For B.ed HonsJameelaNo ratings yet
- The Following Notes Are References According To Figures and Tables in Gartner and Hiatt (2 Edition, 2001)Document5 pagesThe Following Notes Are References According To Figures and Tables in Gartner and Hiatt (2 Edition, 2001)gerginNo ratings yet
- Keratinization DisordersDocument156 pagesKeratinization DisordersAhmad YousryNo ratings yet
- 2.2.integumentary SystemDocument97 pages2.2.integumentary Systemmichot feleguNo ratings yet
- Chapter 2Document26 pagesChapter 2cabdinuux32No ratings yet
- The Histology ofDocument15 pagesThe Histology ofEveryday FundayNo ratings yet
- Bab 7. Struktur KulitDocument56 pagesBab 7. Struktur KulityumizoneNo ratings yet
- Skin cell, Functions, Diseases, A Simple Guide To The Condition, Diagnosis, Treatment And Related ConditionsFrom EverandSkin cell, Functions, Diseases, A Simple Guide To The Condition, Diagnosis, Treatment And Related ConditionsNo ratings yet
- GR2 MATHS WORKSHEET - -Document5 pagesGR2 MATHS WORKSHEET - -Hybat ElsheikhNo ratings yet
- final comprehension & Writing - Grade 3Document8 pagesfinal comprehension & Writing - Grade 3Hybat ElsheikhNo ratings yet
- 4 5791904539871281964 PDFDocument521 pages4 5791904539871281964 PDFElmira RissadiqahNo ratings yet
- 101-Local RX MCQsDocument31 pages101-Local RX MCQsHybat ElsheikhNo ratings yet
- MCQs on Bacterial DiseasesDocument14 pagesMCQs on Bacterial DiseasesHybat ElsheikhNo ratings yet
- 101-Normal Skin MCQsDocument25 pages101-Normal Skin MCQsHybat ElsheikhNo ratings yet
- Anatomy Histo Physio DR - AdilDocument73 pagesAnatomy Histo Physio DR - AdilHybat ElsheikhNo ratings yet
- Hair MCQsDocument35 pagesHair MCQsHybat Elsheikh100% (1)
- Polycythemia - 2Document27 pagesPolycythemia - 2Naveen SudevanNo ratings yet
- Esmé Weijun Wang's Insightful Look at Mental HealthDocument4 pagesEsmé Weijun Wang's Insightful Look at Mental Healthgghyo88No ratings yet
- Antibiotics in Periodontal DiseaseDocument57 pagesAntibiotics in Periodontal DiseaseReshmaaRajendranNo ratings yet
- Argumentative Essay FinalDocument10 pagesArgumentative Essay Finalapi-550035247No ratings yet
- Parkinson's Disease: A Review: Neurology India March 2018Document11 pagesParkinson's Disease: A Review: Neurology India March 2018Alina Vitali TabarceaNo ratings yet
- PH 1 8FLC Ipa GenapDocument5 pagesPH 1 8FLC Ipa GenapNurtri NunuNo ratings yet
- Case Analysis Essay The Children of Sitio MabolonDocument2 pagesCase Analysis Essay The Children of Sitio MabolonisabelNo ratings yet
- Appoach To Brittle Diabetes: Clinical Algorithm To Determine The EtiologyDocument2 pagesAppoach To Brittle Diabetes: Clinical Algorithm To Determine The EtiologyMudassar SattarNo ratings yet
- Antenatal Obstetric ComplicationsDocument40 pagesAntenatal Obstetric ComplicationsArhum SiddiqiNo ratings yet
- White Blue Modern Marine Pollution InfographicDocument2 pagesWhite Blue Modern Marine Pollution InfographicJanella Mae C. RiegoNo ratings yet
- Garispanduan COVID19 Edisi 5 - 2020 PDFDocument242 pagesGarispanduan COVID19 Edisi 5 - 2020 PDFNurul Amelia MustaffaNo ratings yet
- Arduino Guided Smart Rodent Trap (Trapuino)Document9 pagesArduino Guided Smart Rodent Trap (Trapuino)Eunice Nicole Masecampo LautingcoNo ratings yet
- Fluoroquinolones Mechanism Action Uses Side EffectsDocument2 pagesFluoroquinolones Mechanism Action Uses Side EffectsHlaSoe WinNo ratings yet
- Pelagio-Drug Study-NewbornDocument6 pagesPelagio-Drug Study-NewbornShiena Mae PelagioNo ratings yet
- Goldman Report-Corona Impact PDFDocument25 pagesGoldman Report-Corona Impact PDFsumitbajaj100% (1)
- Fillable How To Check Payslips Online As Fidelity Security Worker FormDocument20 pagesFillable How To Check Payslips Online As Fidelity Security Worker FormBang YehNo ratings yet
- 1.3 Respiratory DiseasesDocument14 pages1.3 Respiratory DiseasesgalvezchestertimothyNo ratings yet
- Endocrine Nursing Review: Thyroid and Adrenal GlandsDocument9 pagesEndocrine Nursing Review: Thyroid and Adrenal GlandsJohn denver FloresNo ratings yet
- Dinaxine® Flyer SDocument2 pagesDinaxine® Flyer SPhilip BlairNo ratings yet
- 28 - Rosita Febrianti Jurnal Review (Bahasa Inggris)Document20 pages28 - Rosita Febrianti Jurnal Review (Bahasa Inggris)Restu YasalamNo ratings yet
- 2-Shortness of Breath by Dr.hananDocument49 pages2-Shortness of Breath by Dr.hananSoon SheedNo ratings yet
- Soap Note Template 03Document5 pagesSoap Note Template 03Razan HaimounyNo ratings yet
- Neutrophil-To-Lymphocyte Ratio, Past, Present and Future PerspectivesDocument15 pagesNeutrophil-To-Lymphocyte Ratio, Past, Present and Future PerspectivesWilliamRayCassidyNo ratings yet
- COVID-19 Self-Assessment Declaration - ANISURDocument1 pageCOVID-19 Self-Assessment Declaration - ANISURNethiyaaRajendranNo ratings yet
- Renal Function Test (RFT) : Muhammad Asif Shaheen Lecturer Pathology Kemu, LahoreDocument14 pagesRenal Function Test (RFT) : Muhammad Asif Shaheen Lecturer Pathology Kemu, LahoreRimsha MustafaNo ratings yet
- Gene Environment InteractionDocument26 pagesGene Environment InteractionAwais GulNo ratings yet
- Med Surg Report Sheet Night Shift FINAL2Document2 pagesMed Surg Report Sheet Night Shift FINAL2Cindy Ann100% (1)
- HEALTH 8: Communicable DiseaseDocument31 pagesHEALTH 8: Communicable DiseaseElissah S PabilonaNo ratings yet
- Regional Trade Contacts Must Have Made Periodic Outbreaks Unavoidable in The PhilippinesDocument5 pagesRegional Trade Contacts Must Have Made Periodic Outbreaks Unavoidable in The PhilippinesGreen CrossNo ratings yet
- The Brown ADD Scales: January 1996Document6 pagesThe Brown ADD Scales: January 1996Francisco Ribeiro RachinasNo ratings yet