Professional Documents
Culture Documents
Mini Test 2
Mini Test 2
Uploaded by
niamhtheanimegorl280 ratings0% found this document useful (0 votes)
6 views1 pageThe document is a chemistry test with matching questions asking students to match keywords related to chemical calculations and measurements to their definitions. The keywords include the law of conservation of mass, relative atomic mass, relative formula mass, limiting reactant, range, mean, and concentration, while the definitions explain their meanings in chemistry.
Original Description:
Original Title
mini test 2 (5)
Copyright
© © All Rights Reserved
Available Formats
PPT, PDF, TXT or read online from Scribd
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentThe document is a chemistry test with matching questions asking students to match keywords related to chemical calculations and measurements to their definitions. The keywords include the law of conservation of mass, relative atomic mass, relative formula mass, limiting reactant, range, mean, and concentration, while the definitions explain their meanings in chemistry.
Copyright:
© All Rights Reserved
Available Formats
Download as PPT, PDF, TXT or read online from Scribd
0 ratings0% found this document useful (0 votes)
6 views1 pageMini Test 2
Mini Test 2
Uploaded by
niamhtheanimegorl28The document is a chemistry test with matching questions asking students to match keywords related to chemical calculations and measurements to their definitions. The keywords include the law of conservation of mass, relative atomic mass, relative formula mass, limiting reactant, range, mean, and concentration, while the definitions explain their meanings in chemistry.
Copyright:
© All Rights Reserved
Available Formats
Download as PPT, PDF, TXT or read online from Scribd
You are on page 1of 1
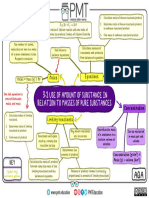
Name:______________________ Score : /7
Topic 3 Chemical calculations Test 2
Match up the keywords to the definitions:
Law of the difference between the
conservation of highest and lowest result in
mass an investigation
Relative atomic the average mass of atom
mass (Ar) compared to Carbon-12
having a mass of 12
Relative formula the reactant that is
mass (Mr) completely used up in a
reaction
Limiting reactant mass cannot be created or
destroyed (only
rearranged)
Range the average result –
calculated by adding all of
the results together and
dividing by number of
results
Mean the amount of a substance
in a set volume
Concentration the sum of the relative
atomic masses of the
atoms in the formula of a
compound or molecule
You might also like
- 59 Titration CalculationsDocument3 pages59 Titration CalculationsChimiste Chimiste100% (2)
- 10.1 Science Notebook (Answer Key)Document5 pages10.1 Science Notebook (Answer Key)Black arab GaladimaNo ratings yet
- Mole ConceptDocument24 pagesMole ConceptRaju SinghNo ratings yet
- Tugas KimiaDocument2 pagesTugas KimiaAries RahmawatiNo ratings yet
- Chapter 4 Calculations ANSWERSDocument2 pagesChapter 4 Calculations ANSWERSmollicksoham2907No ratings yet
- Syllabus BIDocument25 pagesSyllabus BICristhian GavilanezNo ratings yet
- CM011 - StoichiometryDocument4 pagesCM011 - StoichiometryDanise NicoleNo ratings yet
- 3.2. Use of Amount of Substance in Relation To Masses of Pure SubstancesDocument1 page3.2. Use of Amount of Substance in Relation To Masses of Pure Substancesameliarazaq12No ratings yet
- ACFrOgAsZJDlN9z fZja3YuD8P628as3XS-D8W2CT6nF02noP kYFimqGNFSpFwsZ0ECWDWxzBUO0JZs1f9RtWfcbdKro90aVpvzRB76QkBIect860ffb-HOv8djE6Xs0DH497mKXGfDU3ssMDOoDocument8 pagesACFrOgAsZJDlN9z fZja3YuD8P628as3XS-D8W2CT6nF02noP kYFimqGNFSpFwsZ0ECWDWxzBUO0JZs1f9RtWfcbdKro90aVpvzRB76QkBIect860ffb-HOv8djE6Xs0DH497mKXGfDU3ssMDOoAinalnaem GhidariNo ratings yet
- Reactions and Stoichiometry Cheat Sheet: by ViaDocument3 pagesReactions and Stoichiometry Cheat Sheet: by ViaNeia De JesusNo ratings yet
- Particulate Preview Wrap-Up: Avogadro Constant MoleDocument24 pagesParticulate Preview Wrap-Up: Avogadro Constant MoleBunny Smith100% (1)
- Stoichiometry Lesson StarterDocument2 pagesStoichiometry Lesson StarteroscarbecNo ratings yet
- CHEM NotesDocument4 pagesCHEM NotesShayne BonayonNo ratings yet
- SS End of Year Assessment Revision Guide 1Document28 pagesSS End of Year Assessment Revision Guide 1fvsousa2009No ratings yet
- Topic 3.1Document1 pageTopic 3.1duneraoreedNo ratings yet
- CHM1 Q1 0903 YieldDocument21 pagesCHM1 Q1 0903 YieldDumalay, Xenia L.No ratings yet
- IB Chemistry SL and HLDocument108 pagesIB Chemistry SL and HLVed JoshiNo ratings yet
- Chem Exam 2aDocument1 pageChem Exam 2aAnabelle McKenlyNo ratings yet
- Percent Yield Limiting Reactants: Study Guide For Content Mastery Study Guide For Content MasteryDocument1 pagePercent Yield Limiting Reactants: Study Guide For Content Mastery Study Guide For Content MasteryDaniel YosefNo ratings yet
- Revise Chemistry PDFDocument11 pagesRevise Chemistry PDFAli AshrafNo ratings yet
- Stoichiometry: Faculty of Engineering and Technology at Sampoerna UniversityDocument32 pagesStoichiometry: Faculty of Engineering and Technology at Sampoerna UniversityYuni AntariNo ratings yet
- AQA GCSE Chemistry Separate Science U3 Quantitative Chemistry Knowledge OrganiserDocument6 pagesAQA GCSE Chemistry Separate Science U3 Quantitative Chemistry Knowledge OrganiserBikramNo ratings yet
- Urethane Terms NEW PDFDocument9 pagesUrethane Terms NEW PDFgirish balodiNo ratings yet
- Combustion Eng'g PITDocument26 pagesCombustion Eng'g PITDaveRaphaelDumanatNo ratings yet
- Stoichiometry ChecklistDocument2 pagesStoichiometry Checklistpicket1019No ratings yet
- 02 Chap 2 ChemF4 Bil 2018 (CSY3p) PDFDocument27 pages02 Chap 2 ChemF4 Bil 2018 (CSY3p) PDFalanislnNo ratings yet
- Gas Mixtures: Thermodynamics: An Engineering Approach, 6 EditionDocument13 pagesGas Mixtures: Thermodynamics: An Engineering Approach, 6 Editionjulito paquitNo ratings yet
- Chapter 9 - MoleDocument2 pagesChapter 9 - MoleKayla WNo ratings yet
- Documentation SolubilityParameterCalculation V2Document6 pagesDocumentation SolubilityParameterCalculation V2Camilo PardoNo ratings yet
- Basic Principles of Pathway ChemistryDocument15 pagesBasic Principles of Pathway ChemistrySanket AherNo ratings yet
- © Cambridge University Press IGCSE Chemistry: Summary: Chapter 6Document1 page© Cambridge University Press IGCSE Chemistry: Summary: Chapter 6Princess KimNo ratings yet
- GCSE Chemistry - 1.5 Rate of Chemical Change - Lesson Plan 4Document4 pagesGCSE Chemistry - 1.5 Rate of Chemical Change - Lesson Plan 4Eiji OkumuraNo ratings yet
- Unit 1 Introduction-Equilibrium Constants For Chemical ReactionsDocument30 pagesUnit 1 Introduction-Equilibrium Constants For Chemical ReactionsKhánh Vy NguyênNo ratings yet
- Lecture Combustion1Document22 pagesLecture Combustion1Shariq SalahudeenNo ratings yet
- Condensation Reactions ADocument28 pagesCondensation Reactions ANino FelicesNo ratings yet
- 4.3 Quantitative Chemisty Checklist LatymerDocument3 pages4.3 Quantitative Chemisty Checklist LatymerEashwar RajakumarNo ratings yet
- Grim Me 2012Document11 pagesGrim Me 2012Adel KridNo ratings yet
- Synthetic Organic ChemistryDocument157 pagesSynthetic Organic ChemistryPhương MaiNo ratings yet
- 1.2 Amount of SubstanceDocument18 pages1.2 Amount of SubstanceBhPO2023No ratings yet
- 1.3 StoichiometryDocument18 pages1.3 Stoichiometrymishant1980No ratings yet
- Appendix A3 Derivation of Blend Rule For Solubility Parameters 2014 Cleaning With SolventsDocument3 pagesAppendix A3 Derivation of Blend Rule For Solubility Parameters 2014 Cleaning With SolventsMenee Love U TooNo ratings yet
- Bit Atd1Document7 pagesBit Atd1saikrishnaps31No ratings yet
- 1.2. Amount of SubstanceDocument4 pages1.2. Amount of Substancelilysingh2006No ratings yet
- 8 - Reaction Kinetics IIDocument2 pages8 - Reaction Kinetics IINelsonNo ratings yet
- Estimation of Pure Compound Properties Using Group-Interaction ContributionsDocument7 pagesEstimation of Pure Compound Properties Using Group-Interaction ContributionsSofia Velez VazquezNo ratings yet
- IB Chemistry Learning OutcomesDocument25 pagesIB Chemistry Learning OutcomesjszNo ratings yet
- Calculating Hansen Solubility Parameters of Polymers With Genetic AlgorithmsDocument12 pagesCalculating Hansen Solubility Parameters of Polymers With Genetic AlgorithmsVởSạchChữĐẹpNo ratings yet
- Learning Tracker ExperimentDocument2 pagesLearning Tracker Experimentyear2025jeeNo ratings yet
- Lecture Note 5Document24 pagesLecture Note 5DebryNo ratings yet
- Week 12 NotesDocument5 pagesWeek 12 NotesBethel Joy S. TapallaNo ratings yet
- Chapter 12 Study GuideDocument8 pagesChapter 12 Study GuideAegislash 777No ratings yet
- Equilibrium and Equilibrium Concentration CalculationsDocument4 pagesEquilibrium and Equilibrium Concentration CalculationsEvanNo ratings yet
- Estimation Solvent Activities in Polymer Solutions Using A Group-Contribution MethodDocument7 pagesEstimation Solvent Activities in Polymer Solutions Using A Group-Contribution MethodlauraNo ratings yet
- Department of Chemical Engineering: GMR Institute of Technology Rajam, Andhra PradeshDocument6 pagesDepartment of Chemical Engineering: GMR Institute of Technology Rajam, Andhra PradeshsagarNo ratings yet
- Falsified Kinetics: Criterion For in The PelletDocument16 pagesFalsified Kinetics: Criterion For in The Pelletrodrigo pereiraNo ratings yet
- Volumetric SolutionsDocument10 pagesVolumetric SolutionsjycortesNo ratings yet
- Module 8 The Mole ConceptDocument32 pagesModule 8 The Mole ConceptJA BerzabalNo ratings yet
- Revision List - StoichiometryDocument2 pagesRevision List - StoichiometryjuliafigueiredomendesbarreiraNo ratings yet